Abstract
Although intraoperative rapid diagnosis is conventionally performed using hematoxylin–eosin (HE)-stained specimens, the use of additional special staining, together with immunostaining techniques, has been examined in recent years to improve diagnostic accuracy. In intraoperative rapid diagnosis, immunostaining should be completed within 7–10 min, because the pathologist is typically presented with an HE-stained specimen within the same time period. We hypothesized that ultrasound may enhance antigen–antibody reactions and reduce the number of immunostaining steps. To clarify the ability of ultrasound to support immunostaining, we first created an ultrasonic generator specifically for immunostaining. Next, we explored the optimal conditions for immunostaining of formalin-fixed specimens to examine the utility of the ultrasonic generator. Finally, we tried immunostaining with the ultrasonic generator using frozen specimens to simulate intraoperative rapid diagnosis. We report herein that ultrasound enables immunostaining of frozen specimens in ×10 min.
T
It has recently been reported that ultrasound can reduce the reaction time of processes such as fixation, decalcification, and defatting in the production of pathological specimens and can increase protein and nucleic acid stability (Chu et al. 2006; Kitayama and Yamada 2006; Reineke et al. 2006). The utility of microwaves in pathological specimen production and rapid immunostaining is believed to be the result of the greater agitation effect produced by high-frequency vibrations (Leong et al. 1985; Kumada et al. 2004; Hatta et al. 2006). On the other hand, ultrasound is also known to produce high churning/osmosis effects resulting from repeated compression and depression of the liquid level, and these are expected to have the same effects as microwaves (Chu et al. 2006; Kitayama and Yamada 2006; Reineke et al. 2006).
We hypothesized that immunostaining reaction time would be reduced using an ultrasonic generator, and thus for this purpose, we created an ultrasonic generator specifically for immunostaining. Next, we explored the optimal conditions for immunostaining using a formalin-fixed specimen to examine the utility of immunostaining with the ultrasonic generator. Finally, we attempted to immunostain frozen specimens using the ultrasonic generator, as a simulation of its application in intraoperative rapid diagnosis, to verify the amount of time required, its staining precision, and its potential for practical use.

Ultrasound generator device for immunostaining and its attachments. This device has six separate channels. In the image, six channels (channels 1–6) were used for immunostaining
Materials and Methods
Creation of an Ultrasound Generator for Immunostaining
In collaboration with the Kurokawa Corporation (Toyama, Japan) and Honda Electronics Corporation (Aichi, Japan), we experimentally produced an ultrasound generator exclusively for immunostaining; this generator has equal irradiation states in all vibration plates and enables the simultaneous investigation of two or more glass slides (Figure 1). The driving frequency of 1 MHz was utilized to minimize the chemical effects of ultrasound resulting from cavitation (Koda et al. 2003). This device has an independent vibration plate at 1 MHz (plate diameter: 3 cm) with six channels and allows the simultaneous processing of six glass slides under identical conditions. All the channels produce continuous waves at 1000 kHz. Between the glass slides and the vibration plate, a gel sheet was inserted to ensure that no gaps were present. This gel sheet protects the attenuation of ultrasound transmission by less than 1 dB.
Immunostaining Study
Examination of Ultrasonic Intensity and Time Required for Primary Antibody Incubation Using Formalin-fixed Paraffin-embedded Sections. The most effective combination of ultrasonic power [intensity = ultrasonic power (W)/area of plate(cm2)] and irradiation time in each stained specimen was examined using ultrasonic power (intensity) to 0.4 W (0.057 W/cm2), 1.9 W (0.269 W/cm2), 3.3 W (0.467 W/cm2), and 4.4 W (0.628 W/cm2), and ultrasound exposure times of 1, 2, and 3 min. Five different mouse monoclonal antibodies (cytokeratin AE1/AE3, cytokeratin CAM.5.2, CD3, CD20, and Ki-67) frequently used in routine laboratory tests were used as primary antibodies, and Envision peroxidase for mouse monoclonal antibodies (DAKO; Carpinteria, CA) was used as the secondary antibody. A much higher concentration of primary antibodies was used in this examination. Details of the antibodies are shown in Table 1. The irradiation time of the secondary antibody was set at 2 min, and color development was performed using the DAB substrate kit (DAKO). The color development time was set at 1 min. Formalin-fixed paraffin-embedded sections of lymph nodes including metastatic lesions of colorectal cancers were used as specimens. Each section was used for experimentation after performing the typical antigen retrieval process using a target retrieval solution (DAKO).
Five common antibodies
Validation Study of the Ultrasound-supported Method Using Formalin-fixed Paraffin-embedded Sections. To evaluate the practical utility of the ultrasonic method, we selected 10 formalin-fixed paraffin-embedded blocks of common malignant tumors and prepared 30 unstained specimens and a hematoxylin–eosin (HE)-stained specimen. Then, we performed immunostaining with 10 primary antibodies, including 5 common antibodies (cytokeratin AE1/AE3, cytokeratin CAM5.2, CD3, CD20, and Ki-67) and 5 other antibodies, using three different staining procedures (common method, microwave-supported method, and ultrasound-supported method). In brief, the common method is a standard immunostaining procedure with 30-min incubation of primary and secondary antibodies at room temperature, and the microwave-supported method is a procedure with 10-min incubation of primary and secondary antibodies under intermittent microwave irradiation (Kumada et al. 2004). For the ultrasound-supported method, we performed a 3-min incubation of primary and secondary antibodies under 4.4 W ultrasound using our aforementioned equipment. The 10 malignant tumors and selected antibodies are described in Table 2. Each section was used for experimentation after performing the typical antigen retrieval process using a target retrieval solution (DAKO).
Ten malignant tumors and selected antibodies
Immunostaining with five common primary antibodies (AE1/AE3, CAM5.2, CD3, CD20, Ki-67) was also performed in all organs.
Examination of Irradiation Intensity and Time Required for Incubation of the Primary Antibody in Cryosections. A protocol in which immunostaining of frozen specimens was completed within 10 min was developed, and staining quality was evaluated. We used frozen specimens that were presented for rapid diagnosis at Toyama University Hospital. These specimens also had sufficient slices to act as controls for actual diagnoses. After making 5-μm-thick sliced sections, the frozen specimens were fixed for 2 min with acetone (4C), and endogenous peroxidase was removed by a 30-sec incubation with 3% H2O2 (DAKO). The ultrasonic power (intensity) was fixed at 4.4 W (0.628 W/cm2) during the 90-sec incubation with the primary antibody, and the secondary antibody was incubated for 90 sec after washing the specimens for 15 sec with Tris-buffered saline. After DAB coloring for 1 min, nuclear counterstaining was performed for 15 sec using hematoxylin, and the specimens were then covered with coverslips (Table 3).
Immunostaining protocol for frozen specimens
Results
Staining Conditions Employing Formalin-fixed Paraffin-embedded Sections
The best-suited staining conditions were examined using 5 different primary antibodies (cytokeratin AE1/AE3, cytokeratin CAM5.2, CD3, CD20, and Ki-67) by adjusting the intensity and duration of ultrasound irradiation. With cytokeratin AE1/AE3, the staining intensity increased with irradiation intensity at all reaction times, and thus, the maximal power (intensity) of 4.4 W (0.628 W/cm2) was considered to be the optimal ultrasound irradiation intensity. Although staining intensity increased with a reaction time of 2 min compared with 1 min, attenuation of staining intensity was occasionally observed with a reaction time of 3 min (Figure 2). Long ultrasound exposure times were assumed to have harmful effects (destruction, denaturing, etc.) on the antibody (Figure 2). Similar tendencies were recognized in other antibodies examined. Taken together, the optimal conditions in formalin-fixed paraffin-embedded sections were 4.4 W of ultrasound irradiation intensity and 2–3 min irradiation time for both primary and secondary antibodies.
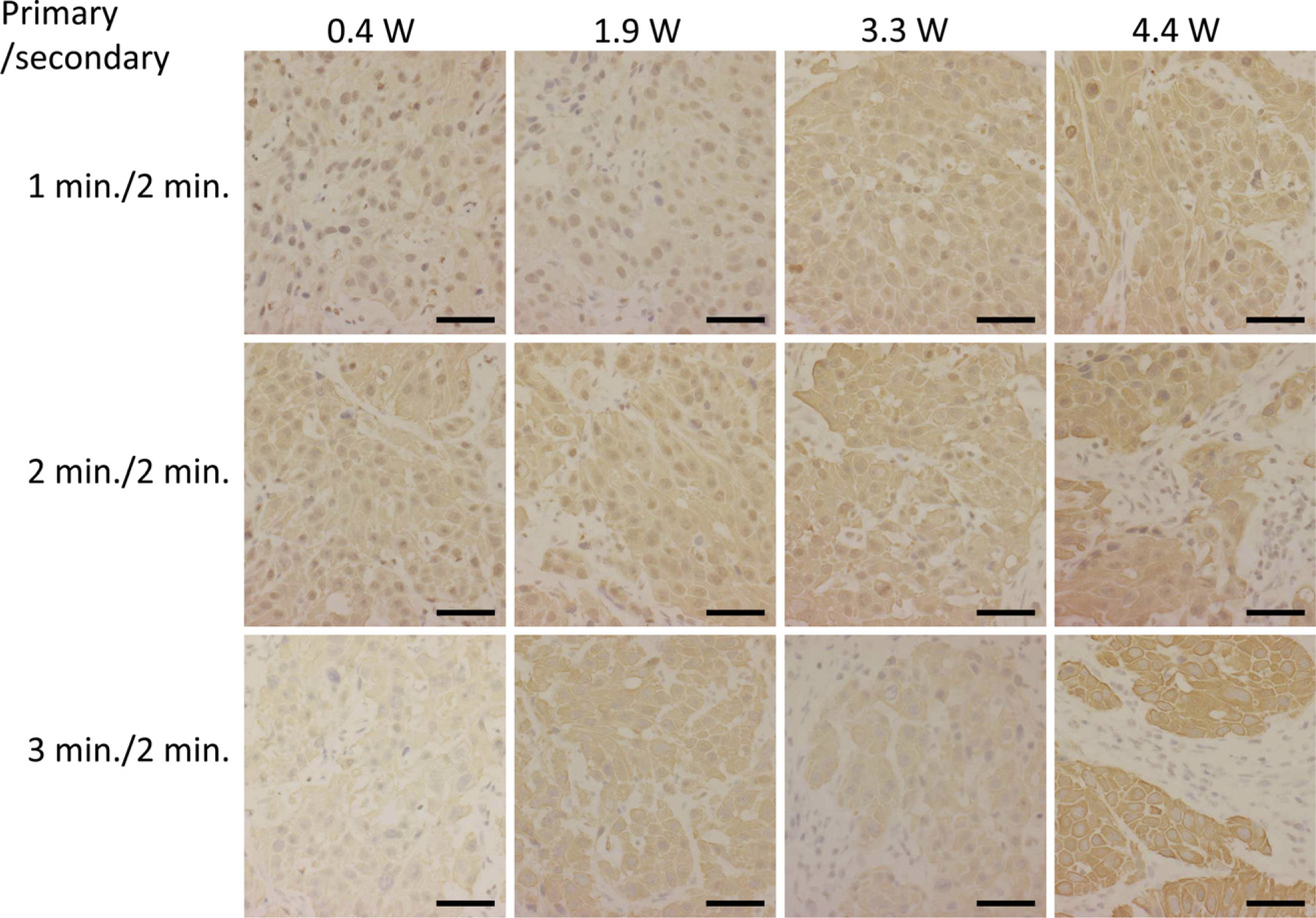
Examination of the irradiation intensity and staining time required for the primary antibody cytokeratin AE1/AE3, using formalin-fixed paraffin-embedded sections of lymph nodes including metastatic lesions of colorectal cancers. The optimal ultrasound irradiation intensity was considered to be 4.4 W. Although staining intensity was reinforced at the reaction time of 2 min compared with 1 min, attenuation of staining intensity was occasionally observed in irradiation for 3 min. Bar = 0.1 mm.
Validation of Ultrasound-supported Method
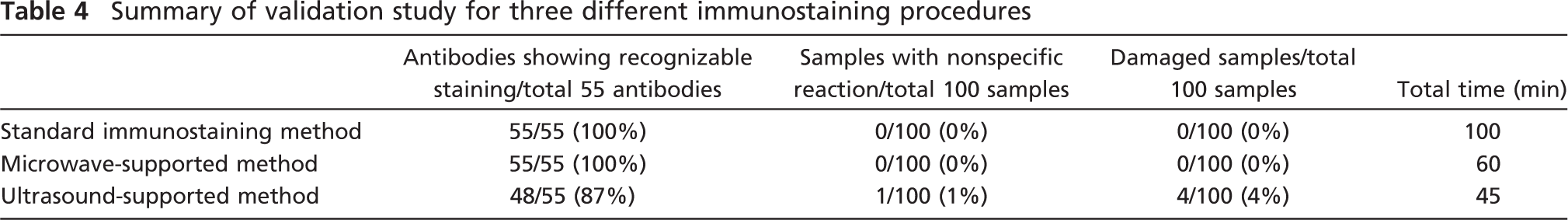
We fixed the staining condition of the ultrasound method as 3 min of incubation for primary and secondary antibodies with 4.4 W ultrasound irradiation and compared its ability to the common staining procedure and the microwave-supported procedure for a validation study. We then performed immunostaining of a total of 55 primary antibodies (5 common antibodies and 50 other antibodies) in 10 samples of malignant tumors using three different immunostaining procedures. Of the 10 primary antibodies, 5 were common primary antibodies (cytokeratin AE1/AE3, cytokeratin CAM5.2, CD3, CD20, and Ki-67) and 5 were other primary antibodies that were chosen based on the source of the tumor. In total, 55 different primary antibodies were examined in this validation study (Table 2). On staining with all 55 primary antibodies using the common and microwave-supported methods, all 100 specimens showed reasonable immunostaining with clear background. All the samples stained by the common and microwave-supported methods retained good morphology. The total staining time of the method using microwave (60 min) was substantially shorter than that of the common method (100 min). In the method using ultrasound, of the 55 primary antibodies, 39 produced strong immunostaining and 9 produced weak but reasonable immunostaining. Taken together, 48 of 55 primary antibodies (87%) were effective using the ultrasound-supported method. Figure 3 shows an example of HE staining and 10 examples of immunostaining of prostate adenocarcinoma. Although 7 primary antibodies (CD5, ER, PgR, β-catenin, Olig-2, Neu N, and CD61) did not produce recognizable immunostaining, 5 common primary antibodies (cytokeratin AE1/AE3, cytokeratin CAM5.2, CD3, CD20, and Ki-67) produced appropriate immunostaining in all cases examined. Almost all slides indicated a reasonable immunostaining pattern; however, cytokeratin CAM5.2 not only occasionally reacted with epithelial cells but also produced nonspecific staining on some foamy macrophages (Figure 4). Of 10 samples, 4 showed various degrees of morphological damage. Bone marrow samples tend to show morphological damage (Figure 4). The ultrasonic-supported method took the shortest time for staining (45 min). These results are described in Table 4.
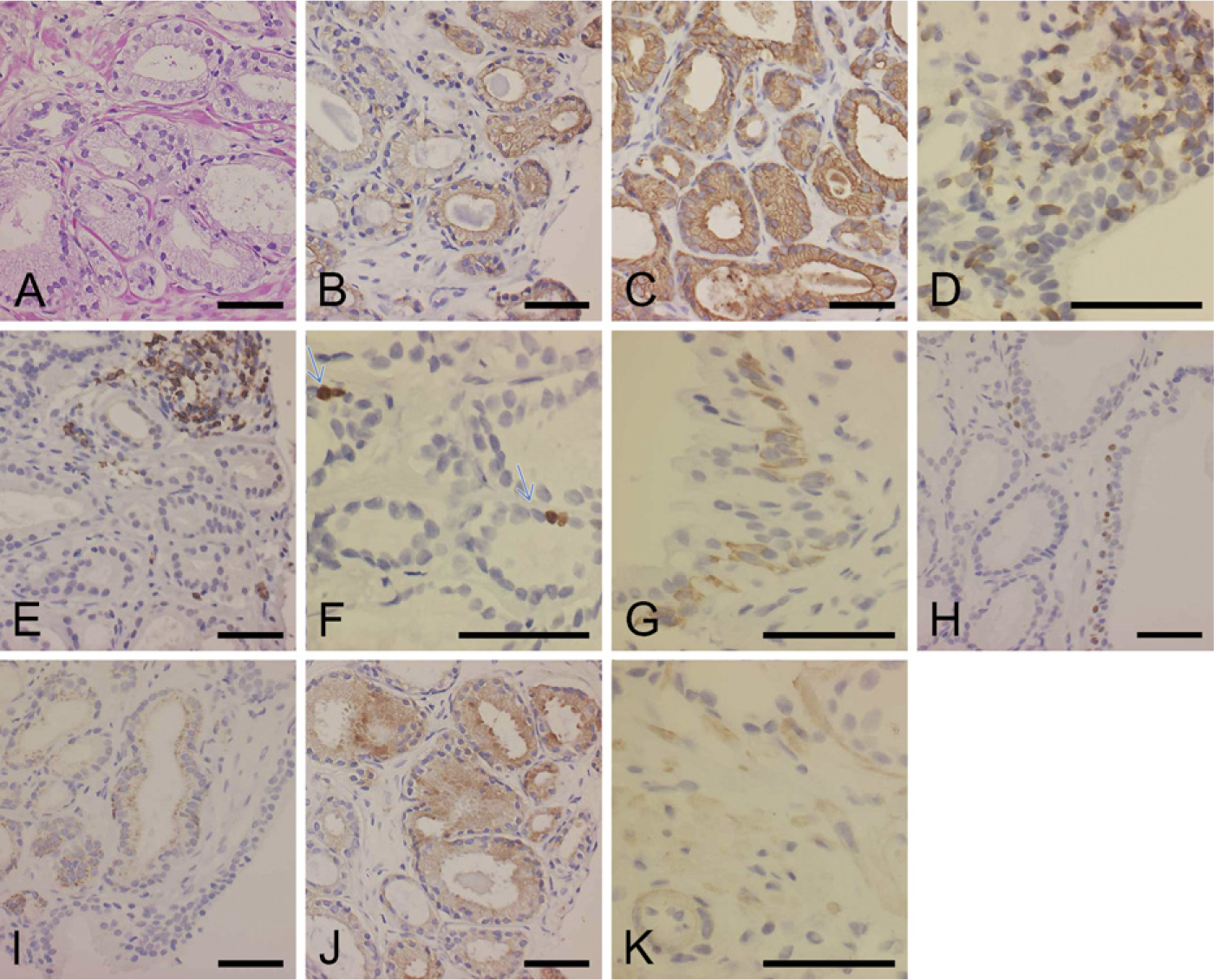
Results of validation study of formalin-fixed paraffin-embedded sections. In prostate adenocarcinoma, all 10 primary antibodies showed reasonable immunostaining by the ultrasound method.
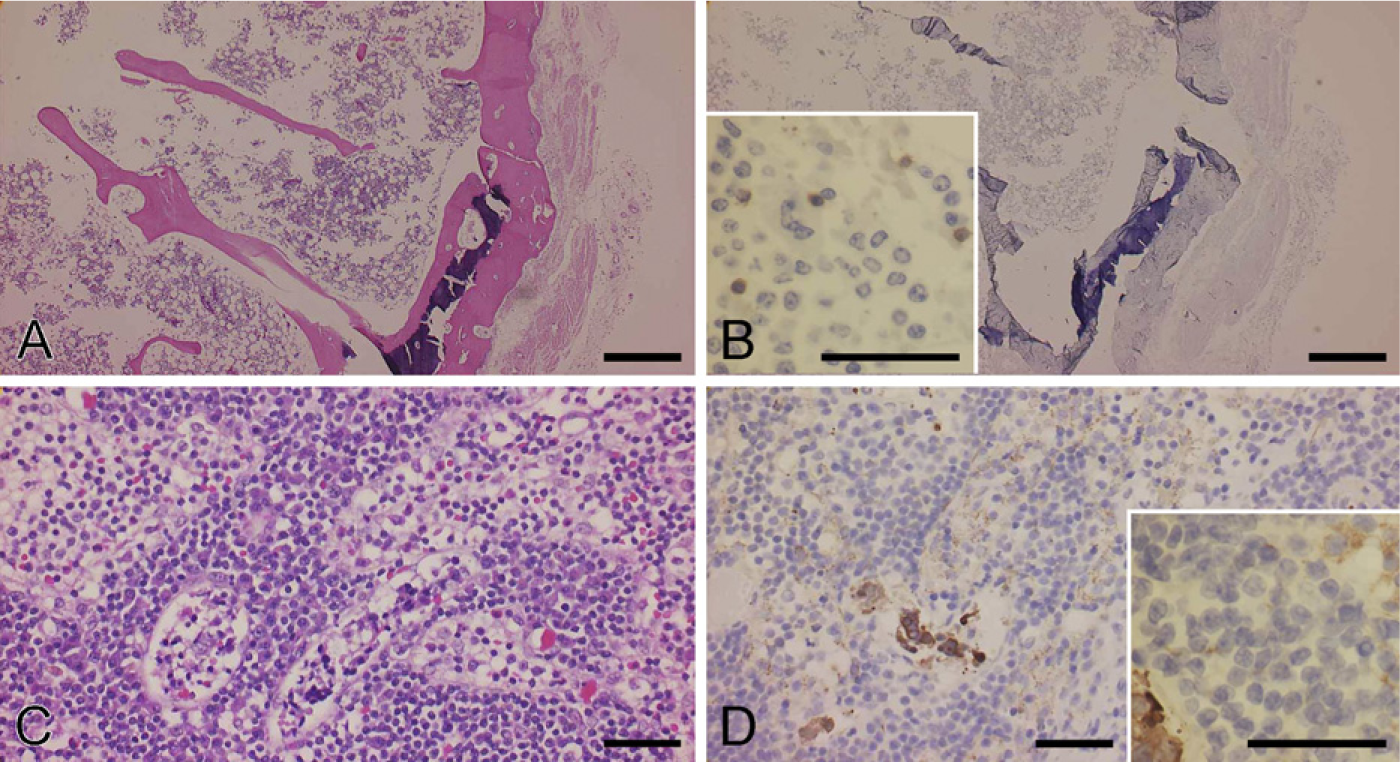
Examples of the disadvantage of the ultrasound method. Morphological damage was observed occasionally.
Summary of validation study for three different immunostaining procedures
Staining Conditions in Frozen Sections
We then examined 75 cases of frozen sections using 4 different primary antibodies (cytokeratin AE1/AE3, cytokeratin CAM5.2, CD3, and CD20) with 10-min sonication. Although 10 cases showed slight morphological damage at the edge of the specimens, they had sufficient area without morphological damage to be included in this experiment. In all the tested specimens, reasonable immunostaining results were obtained using 10-min sonication. Representative immunostaining images are shown in Figure 5. The staining intensity of each specimen was sufficient, the staining pattern was reasonable, and fewer nonspecific reactions were seen in most of the cases examined.
Discussion
Although intraoperative rapid diagnosis is conventionally performed using HE-stained specimens, the use of additional special staining, together with immunostaining techniques, has been examined in recent years to improve diagnostic accuracy (Tsutsumi et al. 1995; Viberti et al. 2001; Matsumoto et al. 2003; Salem et al. 2003; Hatta et al. 2006; Monig et al. 2006; Jylling et al. 2008). However, a certain amount of reaction time is required for an antigen–antibody reaction, and the time required for the common immunostaining procedure (dextran polymer method) can only be shortened to around 13–20 min (Ichihara et al. 2001). In intraoperative rapid diagnosis, immunostaining should be completed within 7–10 min, because the pathologist is typically presented with an HE-stained specimen within the same time period.
In this study, we experimentally produced an ultrasonic device specifically for immunostaining using 6 different antibodies simultaneously and conducted a preliminary examination of the optimal immunostaining conditions for this device (data not shown). As a result, when a particular intensity of sonication was exceeded, the reaction time for immunostaining was significantly shortened, but the quality of the immunostaining was reduced and the specimen was damaged by long-term ultrasonic irradiation. These facts indicate that regulation of output and reaction time is essential for immunostaining with ultrasonic irradiation equipment. Using 5 different commonly used antibodies, we observed that the optimal conditions for our equipment were 4.4 W of irradiation and 2–3 min of antigen–antibody reaction time. Then we performed a validation study using a total of 55 primary antibodies (5 common antibodies and 50 other antibodies), and compared the quality of staining using three different immunostaining procedures (common method, microwave-supported method, and ultrasound-supported method). Of the 55 primary antibodies examined, only 7 did not produce recognizable immunostaining using ultrasound. However, 5 commonly used primary antibodies (cytokeratin AE1/AE3, cytokeratin CAM 5.2, CD3, CD20, and Ki-67) produced recognizable immunostaining. Four of 100 cases showed morphological damage by ultrasound, but most of them were soft, easily damaged organs such as brain and bone marrow. In one case, nonspecific reaction of CAM5.2 to foamy macrophages was observed. Care should be taken while using this antibody for micrometastasis detection. The ultrasound-supported method is beneficial because it reduces the staining time to 45 min.
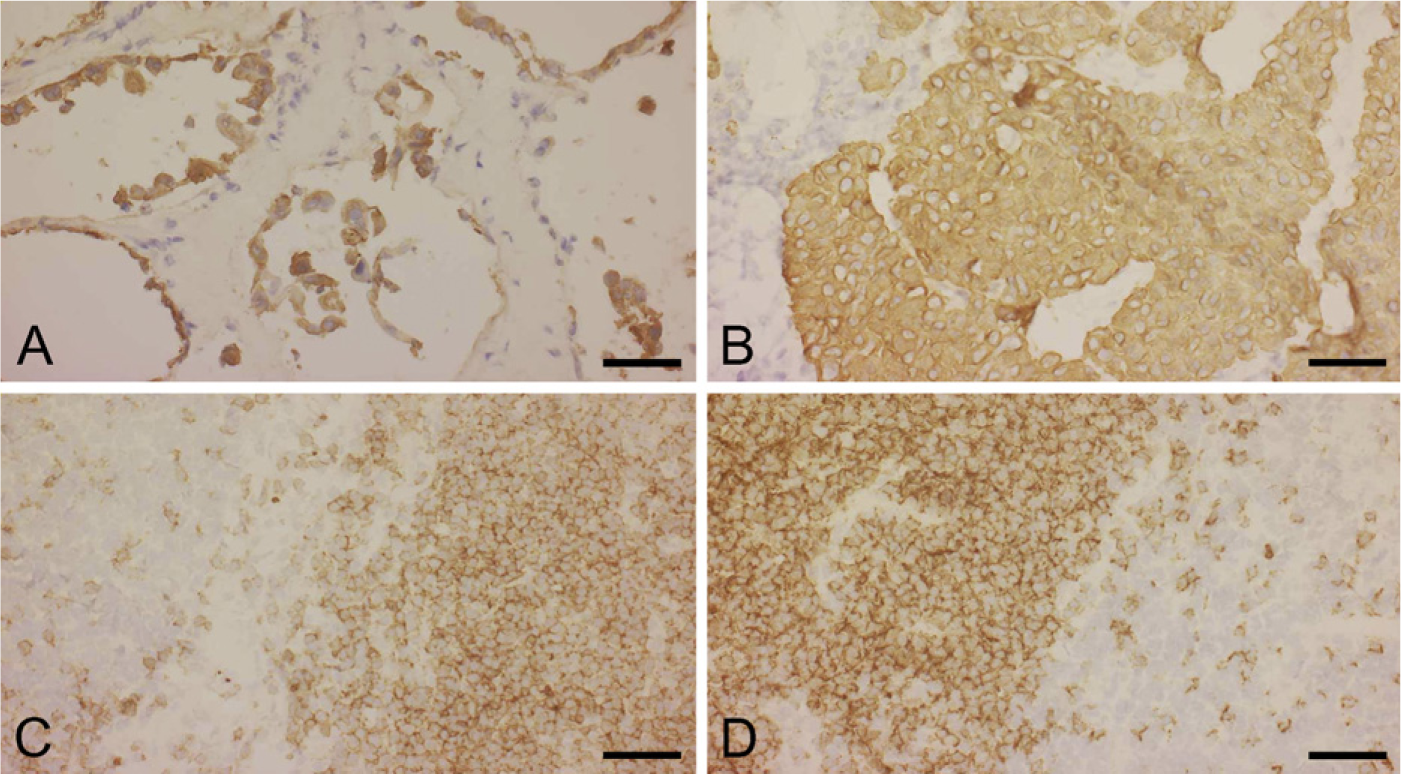
Immunostaining images of cryosections according to the protocol shown in Table 3.
Next, we attempted to establish a staining protocol in which the immunostaining of frozen specimens could be completed in 10 min. Although various steps are needed for immunostaining in addition to the antigen–antibody reaction, the antigen–antibody reaction time is the rate-determining step of the overall reaction time. Although frozen specimens tend to suffer mechanical damage, their antigenicity is more strongly retained compared with formalin-fixed paraffin-embedded specimens. Considering this fact, the antigen–antibody reaction time of the primary and secondary antibodies was reduced by 90 sec compared with the conditions for formalin-fixed paraffin-embedded specimens, thus producing a protocol with an overall reaction time of 10 min. When immunostaining was performed in 75 cases using this protocol, reasonable and recognizable immunostaining was observed in all cases examined, although slight morphological damage was apparent in some cases.
The mechanism by which antigen–antibody reaction time is shortened by ultrasound or microwave irradiation has not been fully clarified. We assume that when ultrasound waves emitted from a vibration plate repeatedly compress and depress the fluid level in a preparation, a high agitation effect is induced and the opportunity for antigens and antibodies to interact significantly improves (Leong et al. 1985, 2002; Kumada et al. 2004; Hatta et al. 2006). Because the same effect can also be induced by raising the dilution concentration of antibodies, an antibody concentration two to four times greater than normal is often used for rapid immunostaining. However, increased concentration of antibodies results in cost increase, introduction of nonspecific reactions, and reduction in immunostaining quality. Using our ultrasonic device resulted in a decrease in nonspecific reactions without producing morphological damage, suggesting that ultrasound can produce sufficient vibration and agitation to enhance specific reactions and inhibit nonspecific reactions.
Using the ultrasonic device developed in this study, immunostaining in frozen specimens can be performed in ×10 min. It is believed that routine immunostaining under the abovementioned conditions is now a viable method for rapid diagnosis.
Footnotes
Acknowledgements
This study was supported by Toyama New Industry Organization (2007).
We thank Tokimasa Kumada (University of Toyama), Shinichi Kurokawa (Kurokawa Co.), and Kazuto Kobayashi (Honda Electronics Co.) for their help and technical assistance.
