Abstract
In immune checkpoint inhibitor (ICI)-related colitis, tumor necrosis factor (TNF)-α blockade is recommended if symptoms are not alleviated with corticosteroids. Although TNF has been shown to be associated with steroid resistance, the early prediction of steroid resistance is challenging in clinical practice. Therefore, in the present study, we evaluated the potential of vascular E-selectin expression, which is induced by TNF, to serve as a predictor of steroid resistance in ICI-related colitis. We performed immunohistochemical analysis of 29 cases of ICI-related colitis using the U12-12 monoclonal antibody, which specifically recognizes E-selectin, and examined the association between U12-12 staining and clinical features. The result showed that the proportion of steroid-resistant cases was significantly higher in the U12-12-positive group (8 of 9 cases; 88.9%) than in the U12-12-negative group (1 of 20 cases; 5.0%) (p<0.001). Furthermore, the proportion of steroid-resistant cases was 100% (6 of 6 cases) in patients with U12-12-positive and Common Terminology Criteria for Adverse Events (CTCAE) grade 3 or higher and 0% (0 of 12 cases) in patients with U12-12-negativity and CTCAE grade 1 or 2. Thus, evaluation of vascular E-selectin expression using the U12-12 monoclonal antibody is useful for predicting steroid resistance in ICI-related colitis.
Introduction
Immune checkpoint inhibitors (ICIs), such as programmed cell death 1 (PD-1), programmed cell death ligand 1 (PD-L1), and cytotoxic T-lymphocyte associated protein 4 (CTLA-4) inhibitors, are widely used as effective anticancer drugs for various types of malignancies. However, ICIs may cause specific adverse events called immune-related adverse events (irAEs). 1 Although any organ can be affected, gastrointestinal tract-associated irAEs, such as colitis, are among the most common irAEs. 2
Several potential mechanisms underlying the occurrence of irAEs have been proposed, including increased T-cell activity and increased levels of proinflammatory cytokines, such as tumor necrosis factor (TNF). 1 However, because it is difficult to strictly distinguish between these mechanisms in clinical practice, irAEs are treated only in accordance with established protocols. Several guidelines recommend corticosteroids as the initial line of treatment for ICI-related colitis and infliximab, a TNF-α blockade, as the next treatments if the symptoms are not alleviated after approximately 3 days of corticosteroid administration.3,4 Colitis with Common Terminology Criteria for Adverse Events (CTCAE) grade 3 or higher is more likely to require infliximab treatment than colitis with CTCAE grade 1 or 2. 5 However, it is difficult to accurately predict steroid resistance and the need for infliximab based on the CTCAE grade alone. Furthermore, because unnecessary corticosteroid administration may diminish the therapeutic effects of ICI, 6 early prediction of steroid resistance and infliximab suitability is desirable. Although the usefulness of blood tests such as serum C-reactive protein levels has been investigated, it was also insufficient to predict steroid resistance in ICI-related colitis. 5 Therefore, predictive markers for steroid resistance in ICI-related colitis that can be used in clinical practice are required for the timely and effective management of this condition.
TNF-α secreted by macrophages upon activation by inflammatory stimuli induces E-selectin expression in endothelial cells.7,8 In other words, vascular E-selectin expression reflects the involvement of TNF-α. Based on this mechanism, we hypothesized that evaluating vascular E-selectin expression in colon biopsy samples from patients with ICI-related colitis may help identify subtypes that are strongly associated with TNF. Furthermore, as TNF has been shown to induce steroid resistance,9,10 evaluating vascular E-selectin expression in ICI-related colitis may help predict steroid resistance. We previously generated the U12-12 monoclonal antibody, which specifically recognizes E-selectin; this antibody can be used to assess E-selectin expression in formalin-fixed paraffin-embedded (FFPE) tissue sections. 11 There exist several anti-E-selectin monoclonal antibodies, including BB11,12,13 BBIG-E6, 14 and 1.2B6, 15 but these antibodies are reported to be usable only on frozen tissue sections and not on FFPE tissue sections. Moreover, the latter two antibodies were later found to cross-react with P-selectin. 16 In fact, there was an antibody designated 16G4 that could be usable on FFPE tissue sections, but this antibody was also later found to cross-react with P-selectin. 8 Thus, to our knowledge, U12-12 is the only E-selectin-specific monoclonal antibody that can be used on FFPE tissue sections. Accordingly, in the present study, using U12-12 as a marker for E-selectin, we aimed to investigate the potential of E-selectin to serve as a predictive biomarker for ICI-related colitis, with the goal of achieving the early identification of patients likely to require infliximab.
Materials and Methods
Study Design and Human Tissue Samples
This multicenter retrospective study was conducted at the University of Fukui, Municipal Tsuruga Hospital, Fukui Saiseikai Hospital, and Shinshu University School of Medicine. The study protocol was approved by the Ethics Committee of the University of Fukui (reference number: 20220190; approved on February 6, 2023).
Based on a previous study, 17 we made the diagnosis of ICI-related colitis when the following two criteria were met: (1) gastrointestinal symptoms, such as diarrhea, bloody stools, or abdominal pain, occurred during or after ICI administration and (2) other diseases that could cause these symptoms were clinically or pathologically excluded. FFPE tissue blocks prepared from patients with ICI-related colitis that met the above criteria were retrieved from pathology archives. The clinical data of the patients were collected from their medical records. The present study was conducted in accordance with the Declaration of Helsinki and patients were provided with the opportunity to opt out.
Immunohistochemical Assessment Using the U12-12 Monoclonal Antibody
Immunohistochemical staining for E-selectin was performed using the U12-12 antibody, as described in a previous study. 11 Briefly, after the FFPE tissue sections were deparaffinized and rehydrated, the endogenous peroxidase activity was blocked by immersing them in absolute methanol containing 0.3% H2O2 for 30 min. Next, antigen retrieval was performed by autoclaving the sections in 10 mM citrate buffer (pH 6.0) at 105C for 15 min. The sections were then blocked with 1% bovine serum albumin in Tris-buffered saline for 30 min, followed by overnight incubation with neat culture medium of U12-12 hybridoma cells (72 hr culture under standard condition, no dilution or concentration) at 4C and then with horseradish peroxidase-conjugated anti-rat IgG (Jackson ImmunoResearch, West Grove, PA) for 30 min. The color reaction was developed using the Betazoid DAB Chromogen Kit (Biocare Medical, Pacheco, CA). Finally, the sections were briefly counterstained with hematoxylin. U12-12 positivity was defined as the presence of at least one vessel stained with U12-12 in a specimen (Fig. 1, upper right panel). In contrast, specimens without U12-12-positive vessels were considered U12-12 negative (Fig. 1, lower right panel).
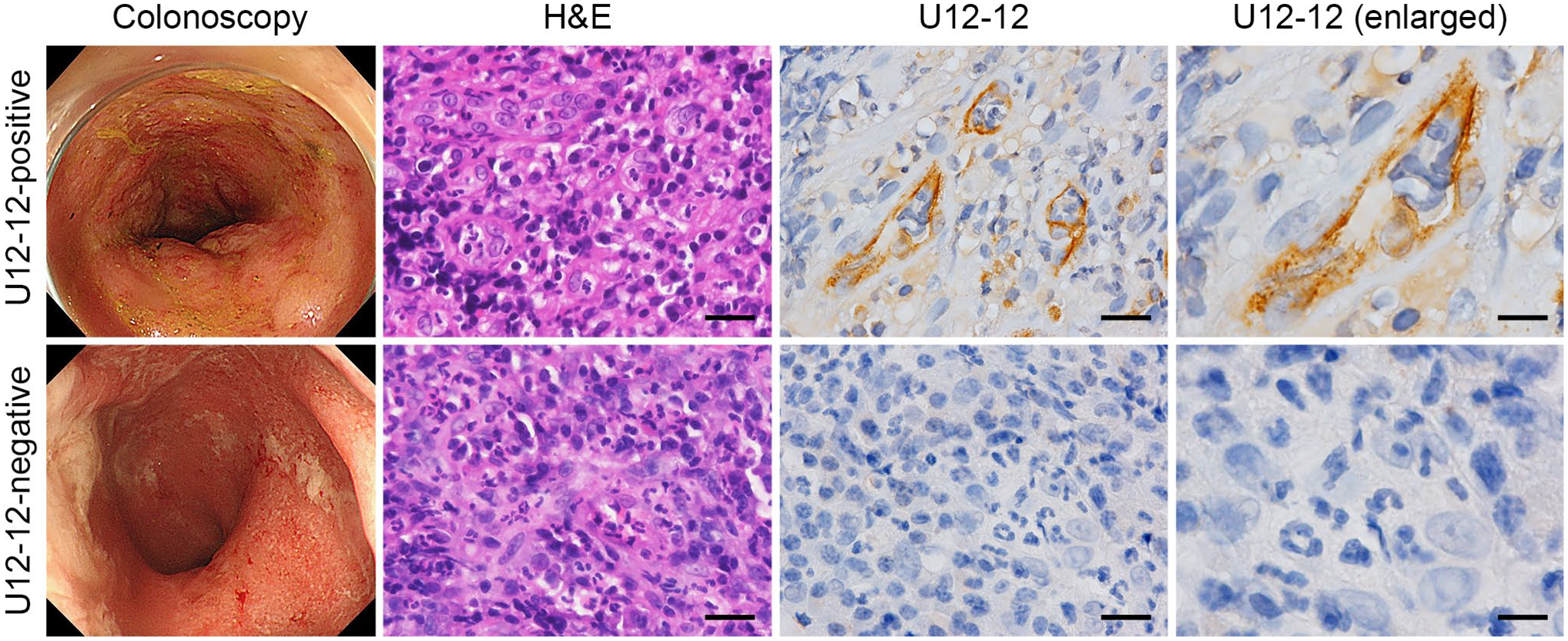
Two representative cases of immune checkpoint inhibitor (ICI)-related colitis. The upper panels show a U12-12-positive case. A man in his 60s with non-small-cell lung cancer (NSCLC) who received a PD-1 inhibitor for 3 months developed severe abdominal pain and diarrhea. The symptoms were not ameliorated despite corticosteroid administration. Therefore, infliximab was administered; this ameliorated the symptoms. The lower panels show a U12-12-negative case. A man in his 70s with NSCLC who received a PD-1 inhibitor for 11 months developed a loss of appetite and diarrhea. The symptoms were ameliorated with corticosteroid administration and no flare up was observed after the cessation of corticosteroid use. Colonoscopy photographs (left panels). Hematoxylin and eosin (H&E)-stained tissue sections showing acute inflammatory granulation tissue formed by erosions (second panels). Immunohistochemical staining with U12-12 shows the induction of E-selectin expression on the luminal surface of small vessels in a U12-12-positive case (upper right two panels), while the small vessels showed no E-selectin expression (in a U12-12-negative case; two panels in the lower right region). Scale bar = 25 µm for the panels in the second and third columns and 12.5 µm for the panels on the right.
Outcomes
The “steroid-sensitive” status was defined when symptoms associated with ICI-related colitis improved with corticosteroids alone. The “steroid-resistant” status was defined when: (1) symptoms associated with ICI-related colitis were not ameliorated with corticosteroid administration alone and/or (2) immunosuppressive drugs (including infliximab) were required in addition to corticosteroids. Patient characteristics, including U12-12 expression, were compared between the steroid-sensitive and steroid-resistant groups.
Statistical Analysis
Differences in clinical features between the groups were statistically analyzed by the Fisher’s exact test using EZR statistical software version 1.55 (Jichi Medical University Saitama Medical Center, Saitama, Japan). Differences with probability values (p values) <0.05 were considered statistically significant.
Results
Patient Characteristics and Clinical Courses
The patient characteristics are shown in Fig. 2. The median age of the patients was 72 years (range: 40–88 years); 24 patients were men. The types of malignancies included malignant melanoma (n=10), non-small-cell lung cancer (NSCLC; n=9), renal cell carcinoma (RCC; n=4), hepatocellular carcinoma (HCC; n=2), oropharyngeal cancer (n=1), gastric cancer (n=1), maxillary gingival cancer (n=1), and renal pelvis and ureter cancer (n=1). The types of ICIs administered were anti-PD-1 antibody monotherapy (n=17), anti-PD-L1 antibody monotherapy (n=4), anti-CTLA-4 antibody monotherapy (n=3), and combination therapy with anti-PD-1 and anti-CTLA-4 antibodies (n=5). Four patients had CTCAE grade 1 colitis, 11 patients had grade 2 colitis, 12 patients had grade 3 colitis, and two had grade 4 colitis. Nine patients met the aforementioned definition of steroid resistance.
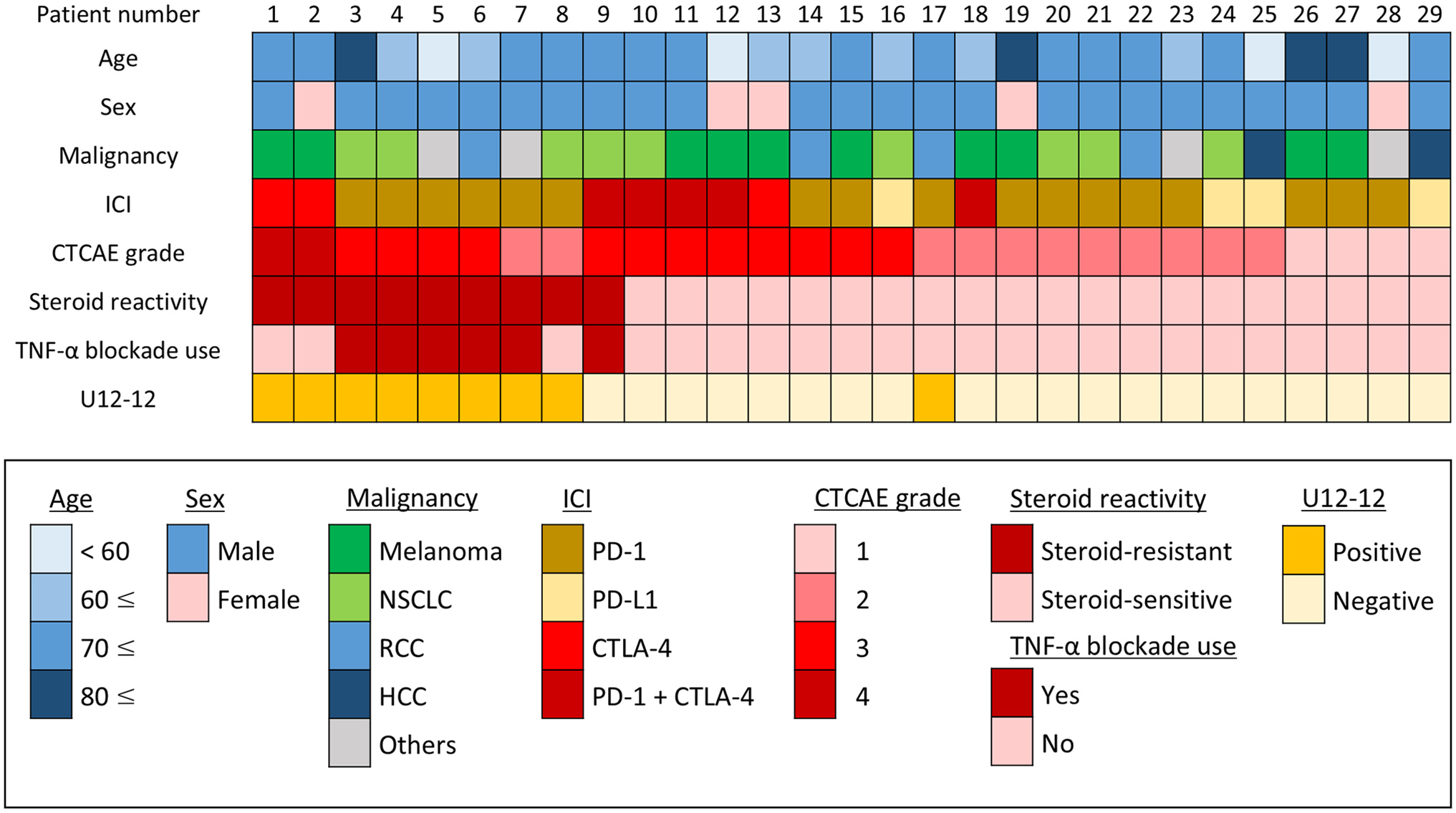
Patient characteristics and results of U12-12 staining are shown as an integrative list. ICI, immune checkpoint inhibitor; CTCAE, Common Terminology Criteria for Adverse Events; TNF, tumor necrosis factor; NSCLC, non-small-cell lung cancer; RCC, renal cell carcinoma; HCC, hepatocellular carcinoma; PD-(L)1, programmed cell death (ligand) 1; CTLA-4, cytotoxic T-lymphocyte associated protein 4.
The main reasons for steroid resistance were the need for infliximab in addition to corticosteroids in six cases (patients 3–7 and 9 in Fig. 2) and the failure of corticosteroids to ameliorate symptoms in three cases (patients 1, 2, and 8 in Fig. 2). In all six of the former cases (patients 3–7 and 9), the symptoms were ameliorated with infliximab administration. Among the latter three patients (patients 1, 2, and 8), two showed worsening symptoms despite corticosteroid administration and underwent surgery for intestinal perforation (patients 1 and 2 in Fig. 2), while the third patient continued taking antidiarrheal medication until death as the symptoms were not ameliorated with corticosteroid administration (patient 8 in Fig. 2).
U12-12 Staining
Of the 29 patients (27 of whom underwent biopsy only and the remaining two underwent surgical resection without biopsy), nine were U12-12-positive and the remaining 20 were U12-12-negative. Representative cases are shown in Fig. 1 (the upper panels correspond to patient 4 in Fig. 2 and the lower panels correspond to patient 20 in Fig. 2). A total of 23 biopsy specimens were obtained from the seven patients whose biopsies were U12-12-positive. The median number of biopsies per patient was 3 (range: 1–4) and the median number of U12-12-positive vessels per biopsy was 3 (range: 0–37).
Association of U12-12 Staining With Clinical Features
We examined the association between U12-12 staining and clinical features, such as steroid resistance and CTCAE grade, as well as the association between CTCAE grade and steroid resistance (Fig. 3A–C). As shown in Fig. 3A, the percentage of steroid-resistant cases was significantly higher in the U12-12-positive group [8 out of 9 cases; 88.9% (95% confidence interval, CI: 51.8–99.7)] than in the U12-12-negative group [1 out of 20 cases; 5.0% (95% CI: 0.1–24.9)] (p<0.001). The percentage of cases with CTCAE grade 3 or 4 was also higher in the U12-12-positive group [7 out of 9 cases; 77.8% (95% CI: 40.0–97.2)] than in the U12-12-negative group [7 out of 20 cases; 35.0% (95% CI: 15.4–59.2)]; however, this difference was not significant (p=0.050) (Fig. 3B). Furthermore, when patients were divided into two groups on the basis of the CTCAE grade, the percentage of steroid-resistant cases was higher in the CTCAE grade 3 or 4 group [7 out of 14 cases; 50.0% (95% CI: 23.0–77.0)] than in the CTCAE grade 1 or 2 group [2 out of 15 cases; 13.3% (95% CI: 1.7–40.5)]; however, this difference was not significant (p=0.050) (Fig. 3C).
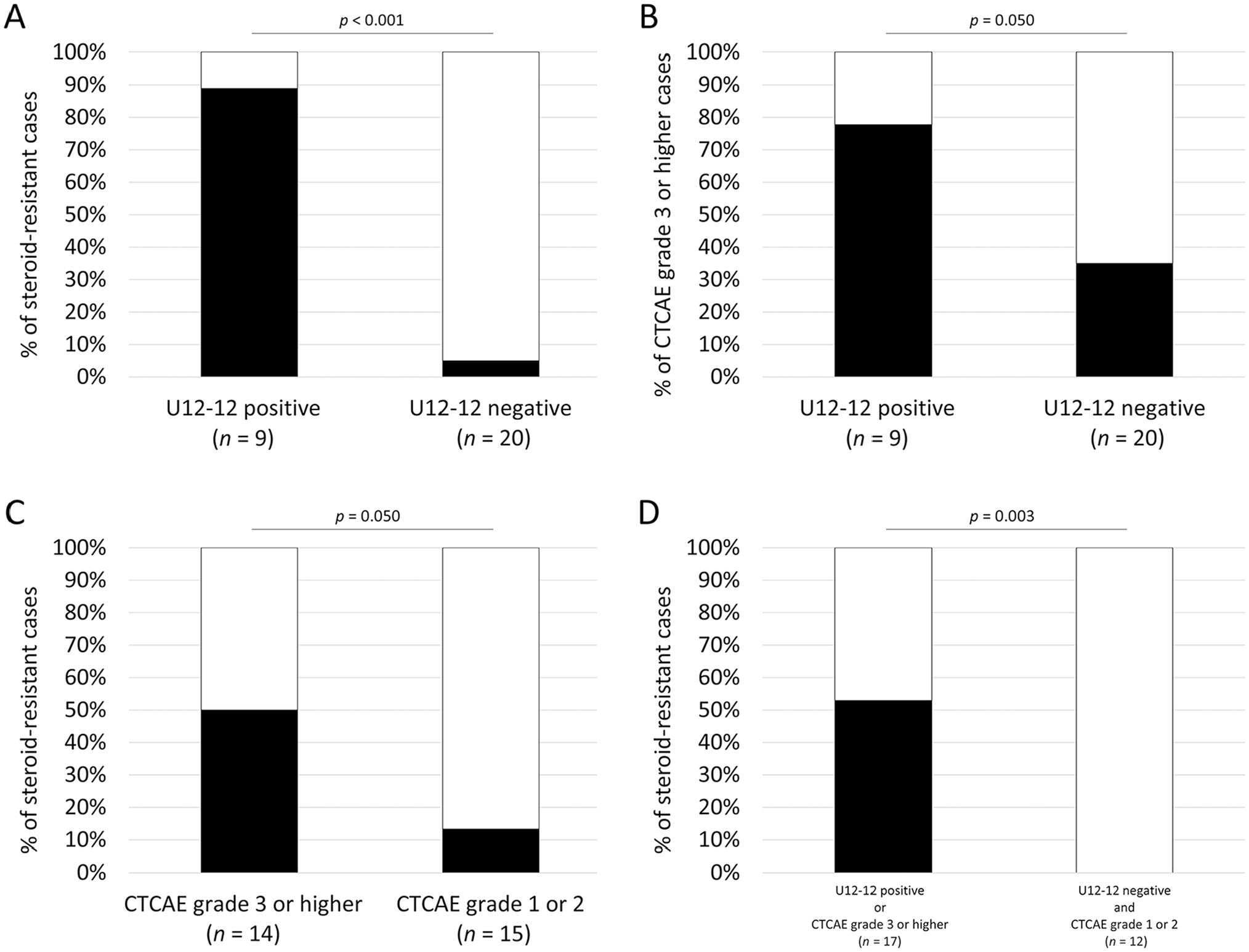
Association of U12-12 staining and clinical features. (A, B) Association between U12-12 staining and steroid resistance (A) and Common Terminology Criteria for Adverse Events (CTCAE) grade (B). (C) Association between steroid resistance and CTCAE grade. (D) Association between a composite parameter consisting of U12-12 staining and CTCAE grade and steroid resistance.
Association of A Composite Parameter Consisting of U12-12 Staining and CTCAE Grade With Steroid Resistance
As shown in Fig. 3D, to examine the association between a composite parameter consisting of U12-12 staining and symptom severity (CTCAE grade) and steroid resistance, we divided the patients into two groups: U12-12-positive or CTCAE grade 3 or higher (n=17) and U12-12-negative and CTCAE grade 1 or 2 (n=12). The percentage of steroid-resistant cases was significantly higher in the former [9 out of 17 cases; 52.9% (95% CI: 27.8–77.0)] than in the latter group [0 out of 12 cases; 0% (95% CI: 0–26.5)] (p=0.003). Moreover, when we divided the patients into the following three groups: (1) U12-12-positive and CTCAE grade 3 or higher (n=6), (2) U12-12-positive or CTCAE grade 3 or higher (n=11), and (3) U12-12-negative and CTCAE grade 1 or 2 (n=12), the percentage of steroid-resistant cases in the first group was 100% [6 out of 6 cases; 100% (95% CI: 54.1–100)], which was significantly higher than that in the second [3 out of 11 cases; 27.3% (95% CI: 6.0–61.0)] (p=0.009) and third groups (p<0.001) (Appendix Fig. 1).
Discussion
In this study, utilizing U12-12 as a specific E-selectin marker, we investigated the potential of E-selectin to serve as a predictive biomarker for the early identification of patients with ICI-related colitis who are likely to require infliximab treatment. We found that U12-12-positive ICI-related colitis was significantly more likely to be steroid resistant than U12-12-negative colitis. Furthermore, U12-12 staining, in combination with symptom severity assessment based on the CTCAE grade, was a more accurate predictor of steroid resistance (than the CTCAE grade alone). Based on these results, we propose that vascular E-selectin expression, recognized using the U12-12 antibody, can serve as a predictive marker of steroid resistance in ICI-related colitis.
TNF has been shown to induce steroid resistance9,10 and has been implicated in the resistance to steroid treatment in various diseases. The pathogenesis of ICI-related colitis has also been shown to be closely associated with TNF. The TNF gene expression levels were shown to be augmented in colon biopsy samples of ICI-related colitis compared with those in normal colon samples from humans 18 and mice. 19 The prophylactic administration of TNF-α blockade in mouse models has also been reported to reduce intestinal wall thickness and ameliorate symptoms such as weight loss due to ICI-related colitis.18,19 Thus, TNF is strongly implicated in the development and symptoms of ICI-related colitis. In our study, U12-12-positive cases tended to have higher CTCAE grades, suggesting an association between TNF and symptom severity of ICI-related colitis. However, the early identification of patients with ICI-related colitis with a strong suspected association with TNF has been challenging in clinical practice.
To address this issue, we focused on the expression of vascular E-selectin. As the expression of E-selectin reflects the TNF levels,7,8 we hypothesized that evaluating the vascular E-selectin expression using the U12-12 antibody may enable the early identification of cases of ICI-related colitis with a strong association with TNF. If this association is true, given the link between TNF and steroid resistance described above, our method may also enable the early prediction of steroid resistance. As expected, the proportion of steroid-resistant cases was significantly higher in the U12-12-positive group than in the U12-12-negative group; this supports our hypothesis. Moreover, the fact that the symptoms associated with ICI-related colitis were ameliorated in all five patients with U12-12-positive ICI-related colitis who received TNF-α blockade further supports our hypothesis. To evaluate TNF concentrations in biopsy specimens, Coutzac et al. 20 incubated colon biopsy specimens in a complete medium and measured TNF concentrations in the supernatants using ELISA; these authors found that high TNF-α concentrations in colon biopsies from patients with ICI-related colitis were associated with steroid resistance and the need for TNF-α blockade. The advantages of our method include the relative ease of the immunohistochemical staining of FFPE colonic biopsy specimens using the U12-12 antibody and the ability to predict steroid resistance before the initiation of treatment for ICI-related colitis.
In the present study, symptom severity, as assessed using the CTCAE grade of colitis, was also suggested to be associated with steroid resistance; however, the difference was not statistically significant. A similar previous study with a larger sample size showed that ICI-related colitis patients with CTCAE grade 3 or higher were significantly more likely to need TNF-α blockade; 5 interestingly, the authors of this study showed that 13% of patients with a CTCAE grade of 1 or 2 and 58% of patients with a CTCAE grade of 3 or higher required TNF-α blockade. The proportion of steroid-resistant patients in the present study was almost the same as that in the aforementioned study (13.3% in CTCAE of 1 or 2 and 50.0% in CTCAE grade of 3 or higher), suggesting an association between symptom severity, measured using CTCAE grade, and steroid resistance, which may be further validated in studies using larger sample sizes. In contrast, however, approximately half of the patients with a CTCAE grade of 3 or higher were steroid-sensitive, indicating the challenge associated with accurately predicting steroid resistance using the CTCAE grade alone. Therefore, a combination of U12-12 staining and CTCAE grade can more accurately predict steroid resistance. Based on this finding, we recommend that TNF-α blockade be administered early in patients with U12-12 positivity and CTCAE grade 3 or higher. However, symptom amelioration can be expected with steroid treatment alone in patients with U12-12 negativity and CTCAE grade 1 or 2.
The present study has a few limitations. First, as this was a retrospective cohort study with a relatively small number of cases, our findings must be validated in larger patient cohorts, including patients with CTCAE grade 1 or 2. The association between the type of ICI used and U12-12 staining should also be evaluated. Second, as E-selectin expression is induced not only by TNF but also by interleukin (IL)-1β,7,8 U12-12 staining does not exclusively reflect TNF activity; in other words, E-selectin expression is only an indirect indicator of TNF activity. Thus, it would be virtually impossible to completely distinguish between these subtypes (i.e., those induced by TNF and those induced by IL-1β). In fact, one U12-12-positive patient was classified as steroid-sensitive, which may represent the difference of these subtypes. Conversely, as U12-12 is an antibody with high specificity but relatively low sensitivity, it is also difficult to completely exclude cases with high TNF activity even if U12-12 is negative. Further researches are needed to assess the validity of our hypothesis. Nevertheless, the present study is valuable as it provides preliminary evidence regarding the association between E-selectin expression (recognized by U12-12) and irAEs.
In the present study, we demonstrated that the evaluation of vascular E-selectin expression, recognized using the U12-12 antibody, is useful for predicting steroid resistance in ICI-related colitis. Moreover, U12-12 staining in combination with symptom severity assessment based on the CTCAE grade can be a more accurate predictor of steroid resistance (than the CTCAE grade alone). Patients with CTCAE grade 3 or higher and U12-12 positivity may benefit from the early administration of TNF-α blockade. Our findings will help reduce the use of corticosteroids and aid the formulation of individualized, safe, and effective treatment regimens for ICI-related colitis patients.
Footnotes
Appendix
Acknowledgements
We thank Hisataka Kato and Maiko Yamanaka for technical assistance.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contributions
All authors have contributed to this article as follows: KN conceived of, designed, and performed the research, analyzed the data, and wrote the manuscript; TN collected the data; MI contributed to pathological evaluation; KH collected the data; TU contributed to pathological evaluation; YN organized the project team and collected the data; TI organized the project team and supervised the project; MK conceived of and designed the research, organized the project team, analyzed the data, and wrote the manuscript. All the authors have read and approved the final manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
