Abstract
Summary
Trefoil factor family (TFF) peptides have been examined primarily in the gastrointestinal tract, where they play an important role in the epithelial regeneration. The therapeutic effects of TFFs, particularly the TFF3 protein, have been well studied in humans and in animal models of gastrointestinal injury, whereas little is known about their occurrence and function in the urinary bladder. In this study, we investigated the presence, location, and function of Tff3 in the urinary bladders of wild-type mice (Tff3WT) and compared them with Tff3 knockout mice (Tff3KO) using molecular and microscopic methods at the light and electron microscopic level. Our results show that Tff3 is expressed in the superficial cells of the urothelium, where it colocalizes with the uroplakin UP1b as one of the fundamental structural components of the apical plasma membrane, which is an important component of the blood-urine permeability barrier. Analysis of the urothelium with experimentally induced injury revealed that injury is more severe in Tff3KO mice and urothelial regeneration is attenuated compared with Tff3WT mice, suggesting that Tff3 plays a fine-tuned role in homeostasis and protection of the urothelium. This study provides the first data on the precise location and function of Tff3 in the bladder epithelium.
Introduction
Trefoil factor protein 3 is a small peptide (80 amino acids, 8.6 kDa) belonging to the trefoil factor protein (TFF) family, which consists of three members: TFF1, TFF2, and TFF3. Each of them contains one (TFF1 and TFF3) or two (TFF2) typical, highly conserved structural motifs in the form of three loops known as trefoil domains, formed by intramolecular disulphide bridges between cysteine residues.1,2 All TFF peptides play a crucial role in protecting, maintaining, and restoring the body’s mucosa. They are well studied in the gastrointestinal tract, but are also found in the respiratory tract, salivary glands, eyes, and genitourinary system as exocrine products, while they are secreted as endocrine products in the immune system and central nervous system.3 –5 High concentrations of TFF peptides are particularly detected in the gastrointestinal tract, where they are integral components of the mucosal barrier, regulating mucus viscosity and interacting with surface proteins and receptors.6 –8 All TFF peptides function as lectins and can therefore be specifically associated with various cell surface and extracellular glycoproteins, including β-integrin, IgG Fc-binding protein (FCGBP), and mucins. Recently, the disaccharide GlcNAc-α-1, 4-Gal was confirmed to be an essential ligand for cross-linking TFF peptides with mucus glycoproteins in a lectin-like manner. 9
The TFF peptides share some functional properties, such as promoting epithelial repair by enhancing cell migration, but TFF1 and TFF3 also protect epithelia by inhibiting apoptosis.7,10,11 TFF3 is mainly found in the intestinal epithelium, where it plays an important role in increasing the impermeability of the intestinal barrier by altering the composition of tight junctions. 12 TFF3 has also been detected in the bloodstream and urine, and its presence in urine is a valuable biomarker in chronic kidney disease.13 –15
Although numerous functions have been described for TFF peptides, their role in the urinary tract and especially in the urinary bladder is still poorly understood. The epithelium of the urinary bladder, also called urothelium, covers the luminal surfaces of the urinary tract from the renal pelvis to the proximal urethra and belongs to a group of »tight« epithelia. 16 It forms the vital blood-urine permeability barrier, which is the tightest barrier in the human body. The superficial urothelial cells that are in contact with urine play a crucial role in urothelial permeability due to their specialized apical plasma membrane, the surface glycocalyx, and tight junctions between them.17 –19 There is some data in the literature on the presence of TFF3 in the urinary bladder,20,21 but the exact location and function of TFF3 in the bladder have hardly been investigated.
The aim of this study was to investigate the precise location of Tff3 in the mouse urinary bladder, which has never been described before. We also aimed to investigate the functional role of Tff3 by analyzing and comparing normal and experimentally injured urothelial tissue from wild-type and Tff3-knockout mice. Our results obtained in this manner provide the first evidence for the predominant expression of Tff3 in superficial urothelial cells and its protective role for the urothelium of the urinary bladder.
Materials and Methods
Chemicals
Chitosan hydrochloride (hereinafter chitosan) was purchased from Heppe Medical Chitosan GmbH (Halle, Germany). The 0.5% (w/v) dispersion of chitosan was prepared in phosphate buffer (pH 7.4). The pH value of dispersion was then adjusted to pH 4.5 and mixed for 4 hours to become clear liquid.
Animals
The experiments were performed with Tff3 knockout mice (Tff3-/-/C57BL/6NCrl or Tff3KO mice) and the corresponding wild-type C57BL/6NCrl strain (Tff3WT mice). A Tff3 knockout mouse strain on a genetic background of C57BL/6NCrl (Charles River) was developed from an existing mixed background strain (C57BL/6J/SV129) using a speed congenics approach as previously described. 22
Mice were housed in the laboratory animal facility of the Ruder Bošković Institute in open-bar cages with bedding and under controlled conditions of temperature (21°C), humidity (60%), and light (12/12-hour light/dark cycle) with unrestricted access to water and food (standard rodent diet Mucedola, 4RF21). Tff3 knockout mice (9 females and 3 males) and wild-type mice (9 females and 3 males) aged 5–8 months and weighing 20–30 g were included in this study. After sacrificing the animals, the urinary bladders were removed and prepared for further analyses. The animal experiments were conducted according to the guidelines of the Directive 2010/63/EU and approved by the Institutional Review board (Ethics Committee) of Institute of Ruđer Bošković and Croatian Ministry of Agriculture (Class: UP/I-322-01/16-01/81; URBROJ:525-10/0256-16-4; date of approval 15 December 2016).
mRNA Extraction and qPCR Analysis
The excised urinary bladders were cut in half and snap-frozen with liquid nitrogen for short-term storage at −80°C. After thawing, the tissue was lysed with QIAzol Lysis Reagent and TissueLyser LT (both from Qiagen, Hilden, Germany) using 5 mm stainless steel beads at 50 Hz (2 × 5 minutes). Total RNA was extracted using the RNeasy Plus Universal Mini Kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. The eluted content of RNA and its purity in each sample were measured using the NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA).
The RNA was then transcribed into cDNA using a 1st Strand cDNA Synthesis Kit for RT-PCR (Roche, Basel, Switzerland) according to the manufacturer’s instructions. A qPCR analysis of Tff3 was performed using the TaqMan Gene Expression Assay (Mm00495590) and the TaqMan Universal PCR Master Mix (both from Thermo Fisher Scientific, Carlsbad, SA, USA). The qPCR was performed using a LightCycler 480 system (Roche, Basel, Switzerland). Duplicates of each sample were assayed in a run consisting of polymerase activation (95°C for 10 minutes), followed by 60 cycles of denaturation (95°C for 15 seconds) and annealing/elongation (60°C for 1 minute). Data were analyzed using the comparative Ct method relative to the expression of the endogenous control (L32) and presented as 2-ΔΔCt between the average Ct of the Tff3 gene and the average Ct of the endogenous control. In addition, to monitor the expression of Tff1, Tff2, and Tff3, we developed Syber green-specific reaction conditions to ensure specific products (Appendix: Table A1 and Fig. A1). The level of Tffs gene expression was very low, that is, ct values in Tff3WT mice are ct = 28 ± 0.048 for Tff1, ct = 32.3 ± 0.043 for Tff2, and ct = 30 ± 0.048 for Tff3. These values are at the lowest detection limit of the Syber green-based qPCR method, indicating that the copy number of Tff molecules in the mouse bladder is really low, which is why we have presented the data as gel images (Fig. A1).
Protein Isolation and Western Blot Assay
The halves of the excised urinary bladders were snap-frozen in liquid nitrogen and homogenized using a tissue pulverizer. The homogenate was lysed in a RIPA buffer (Merck, Kenilworth, NJ, USA) with the addition of a cocktail of protease and phosphatase inhibitors (Thermo Fisher Scientific, Waltham, MA, USA). After centrifugation of the homogenates at 12,000 × g for 20 minutes at 4°C, the total protein concentration in the supernatant of each sample was measured using a Pierce BCA Protein Assay Kit (Thermo Fisher Scientific, Waltham, MA, USA). Equal amounts of protein for each sample (50 µg per lane) were separated on 4% to 20% Novex WedgeWell Tris-Glycine gels (Invitrogen, Carlsbad, CA, USA) and transferred to a nitrocellulose membrane (Sigma-Aldrich, St. Louis, MO, USA). The membranes were blocked in 5% nonfat dry milk dispersed in 0.1% Tris-buffered saline/Tween 20 (TBS-T) for 2 hours at room temperature (RT) and incubated in a suspension of primary antibodies against Tff3 (1:1000; prepared from rabbit sera in the laboratory of prof. dr. N. Blin, University of Tübingen, Germany) and against β-actin (1:2000, Sigma-Aldrich, St. Louis, MO, USA; #A2066) at 4°C overnight. The next day, the membranes were incubated in a suspension of horseradish peroxidase-conjugated secondary antibody (1:1000, Sigma-Aldrich, St. Louis, MO, USA; #A6154) for 2 hours at RT. Visualization of protein bands was performed using the SuperSignal West Femto Maximum Sensitivity Substrate on the iBright FL1500 Imaging System (both from Thermo Fisher Scientific, Waltham, MA, USA). Mouse small intestine tissue samples, prepared in the same way as the bladder tissue samples, served as positive controls.
Mouse Model of Urothelial Injury
Induction of urothelial injury was performed as previously described. 23 In brief, female mice were anesthetized with an intraperitoneal injection of ketamine HCl (100 mg/kg; Vetoquinol, Poland) and xylazine (10 mg/kg; Chanelle Pharmaceuticals Manufacturing Ltd., Ireland), placed in the dorsal position and immobilized on the surgical board. A polyethylene tube with an inner diameter of 0.28 mm (Intramedic, Becton Dickinson, USA) was inserted into the bladder through the urethra and sheathed with a 30 G needle connected to a 1-mL injection syringe. First, the bladder of each mouse was emptied by applying gentle manual pressure to the lower abdomen. Then, 80 µL of a 0.5% chitosan dispersion was instilled intravesically to induce cell desquamation in the bladder urothelium. After 30 minutes, the tube and syringe were carefully withdrawn from the bladder and the bladders were removed from the mice.
Control treatment was performed in female Tff3WT mice (n = 3) and female Tff3KO mice (n = 2) with intravesically injected sterile saline using the same procedure as for chitosan treatment (Appendix, Fig. A2).
Preparation of Paraffin Sections
Excised urinary bladders were cut in half and fixed in 10% neutral buffered formalin for 24 hours at 4°C, dehydrated in an increasing ethanol series, cleared in xylene and embedded in paraffin for 24 hours. The bladders were then cut into 5 µm thick tissue sections and stained with the routine histological dyes Hematoxylin and Eosin (H&E staining).
Preparation of Cryosections
Excised urinary bladders were cut in half and fixed in 3% paraformaldehyde in phosphate-buffered saline (PBS) for 2 hours at 4°C. After overnight incubation in 30% sucrose at 4°C, the bladder tissue was embedded in Tissue Freezing Medium (Leica, UK), frozen, and cut into 5-µm-thick cryosections in a cryostat chamber (CM3000, Leica, Germany).
Immunofluorescence Labeling
The cryosections of the bladder samples were first rinsed in PBS (Merck, Germany). Nonspecific labeling was blocked by incubating the tissue sections in 5% bovine serum albumin (BSA; Sigma-Aldrich, Germany) in PBS for 1 hour at RT. For the single immunolabeling of Tff3 or double immunolabeling of Tff3 and UP1b, immunopurified primary rabbit polyclonal antibodies against Tff3 (1:200; prepared from rabbit sera in the laboratory of prof. dr. N. Blin, University of Tübingen, Germany) were applied and incubated overnight at 4°C. After rinsing in PBS, secondary donkey anti-rabbit antibodies (1:400; Alexa Flour 488, Invitrogen, USA) were applied at 37°C for 90 minutes. For double immunolabeling of Tff3 and uroplakin UP1b, primary rabbit polyclonal antibodies against Tff3 (1:200) were applied simultaneously with primary goat polyclonal antibodies against uroplakin UP1b (1:100; Santa Cruz Biotechnology, Dallas, USA; sc-15174). Subsequently, secondary rabbit anti-goat antibodies (1:400, Alexa Flour 555, Invitrogen USA) were used. Positive controls for Tff3 immunolabeling were performed on sections of normal mouse small intestine, while negative controls for both selected antigens were performed by omitting the primary antibodies during the immunolabeling process. Sections were embedded in mounting medium containing DAPI (Vector Laboratories, Burlingame, USA) and examined with the AxioImager.Z1 fluorescence microscope equipped with ApoTome (Carl Zeiss MicroImaging GmbH; Munich, Germany).
Confocal Microscopy and Colocalization Quantification by Microscopy Image Analysis
Cryosections of bladders from Tff3WT mice were examined with the LSM900 confocal microscope (Zeiss, Germany) using an oil objective (63x/NA 1.40) and the Airyscan 2 detection system. The field of view for imaging was selected with the DAPI channel showing only the cell nuclei. This ensured that at least one nucleus of the superficial urothelial cells was detected and that the signals of the green (Tff3) and red (UP1b) channels did not influence the selection of the imaging area. All samples were imaged with the same laser settings. Colocalization analysis was performed using the colocalization mode of the ZEN blue 3.6 software. On each image, we selected a region of interest (ROI) limited to the superficial urothelial cells only. The crosshair thresholds in the scatter plot were determined using an automatic Costes threshold estimation method included in the ZEN blue software for each individual ROI. The analysis was performed on a total of 30 images of the bladder urothelium from three Tff3WT mice.
Immunogold Labeling for Transmission Electron Microscopy (TEM)
The bladder tissue samples were fixed in a mixture of 2% paraformaldehyde and 0.05% glutaraldehyde in PBS buffer for 1 hour at RT and cut into small pieces (1 mm3). Samples were dehydrated by progressive lowering the temperature and embedded in Lowicryl HM20 resin (Polysciences, Warrington, USA) in the Leica EM AFS instrument (Leica Microsystems, Wetzlar, Germany) according to the following protocol: 30% ethanol for 30 minutes at 0°C, 55% ethanol for 30 minutes at −15°C, 70% ethanol for 30 minutes at −30°C, 100% ethanol for 1 hour at −50°C, 75% ethanol/25% HM20 for 1 hour at −50°C, 50% ethanol/50% HM20 for 1 hour at −50°C, 25% ethanol/75% HM20 for 1 hour at −50°C, 100% HM20 for 1 hour at -50°C, 100% HM20 overnight at −50°C. HM20 was polymerized for 48 hours at −50°C and then for 24 hours at −20°C under UV light. Ultrathin sections (60 nm thick) were cut and collected on nickel grids. Sections were washed in washing buffer (0.1% Na-azide, 0.8% BSA and 0.1% IGSS gelatine in PBS), blocked in blocking buffer (5% fetal calf serum in washing buffer) for 30 minutes at room temperature and incubated with primary rabbit polyclonal antibodies against Tff3 (1:100) overnight at 4°C. After washing in washing buffer, secondary goat anti-rabbit antibodies conjugated with 18-nm colloidal gold (Au) (1:40 in blocking buffer) were applied for 90 minutes. The sections were counterstained with uranyl acetate and lead citrate. The grids were observed at 80 kV in a Philips CM100 transmission electron microscope (Philips, Eindhoven, Netherlands).
Tissue Preparation for Scanning Electron Microscopy (SEM)
Excised urinary bladders were cut in half lengthwise and fixed in a mixture of 2% paraformaldehyde and 2% glutaraldehyde in 0.1 M cacodylate buffer (pH 7.4) for 3–4 hours at 4°C. After an overnight rinse in 0.1 M cacodylate buffer and post-fixation in 1% osmium tetroxide (Roth, Germany) in the same buffer for 1 hour at 4°C, the bladder halves were dehydrated to 100% acetone via a graded acetone series and dried in a critical point dryer. The tissue pieces were then mounted on aluminum holders, sputter-coated with gold, and examined at 25 kV in a Tescan Vega3 scanning electron microscope (Brno, Czech Republic).
Results
Tff3 Is Expressed in the Urinary Bladder Urothelium of Tff3WT Mice, Predominantly in Superficial Cells
Our first aim was to verify the presence of Tff3 and thus analyze the expression of Tff3 mRNA and Tff3 protein in mouse urinary bladders. The qPCR analysis confirmed the expression of Tff3 mRNA in the urinary bladders of Tff3WT mice, whereas, as expected, no Tff3 mRNA was detected in the bladders of Tff3KO mice (Fig. 1A). The presence of Tff3 protein was confirmed in the bladders of Tff3WT mice using the Western blot assay, albeit at a low level. No Tff3 signal was detected in the bladders of Tff3KO mice (Fig. 1B).

The confirmation of Tff3 mRNA and Tff3 protein expression in the urinary bladders of mice. (A) The results of qPCR analysis show the expression of the Tff3 gene in the urinary bladders of Tff3WT mice, while the signal is completely absent in the urinary bladders of Tff3KO mice. The graph shows summary statistics of 2-ΔΔCt determined for eight Tff3WT mice and four Tff3KO mice. (B) Representative blots of Western blot analysis of Tff3 show a Tff3-specific signals (band at 10 kDa corresponds to Tff3 monomer and band at 13 kDa corresponds to glycosylated form of Tff3 monomer) in the urinary bladder tissue samples of Tff3WT mice and a strong Tff3-specific signal in the small intestine samples (positive control) of Tff3WT mice. No Tff3-specific bands were detected in the urinary bladder tissue samples from Tff3KO mice.
Immunofluorescence labeling revealed the expression of the Tff3 protein predominantly in the cytoplasm of the superficial urothelial cells of Tff3WT mice (Fig. 2A and B). TEM analysis of immunogold-labeled ultrathin sections of Tff3WT mouse bladders confirmed the immunoreaction in the superficial urothelial cells showing Tff3 mainly at the apical plasma membrane, at the membranes of the fusiform vesicles and in the cytoplasm, mainly at the cytoskeletal filaments (Fig. 2C and D). Immunofluorescence against Tff3 was negative in the bladders of Tff3KO mice (Fig. 2E and F).
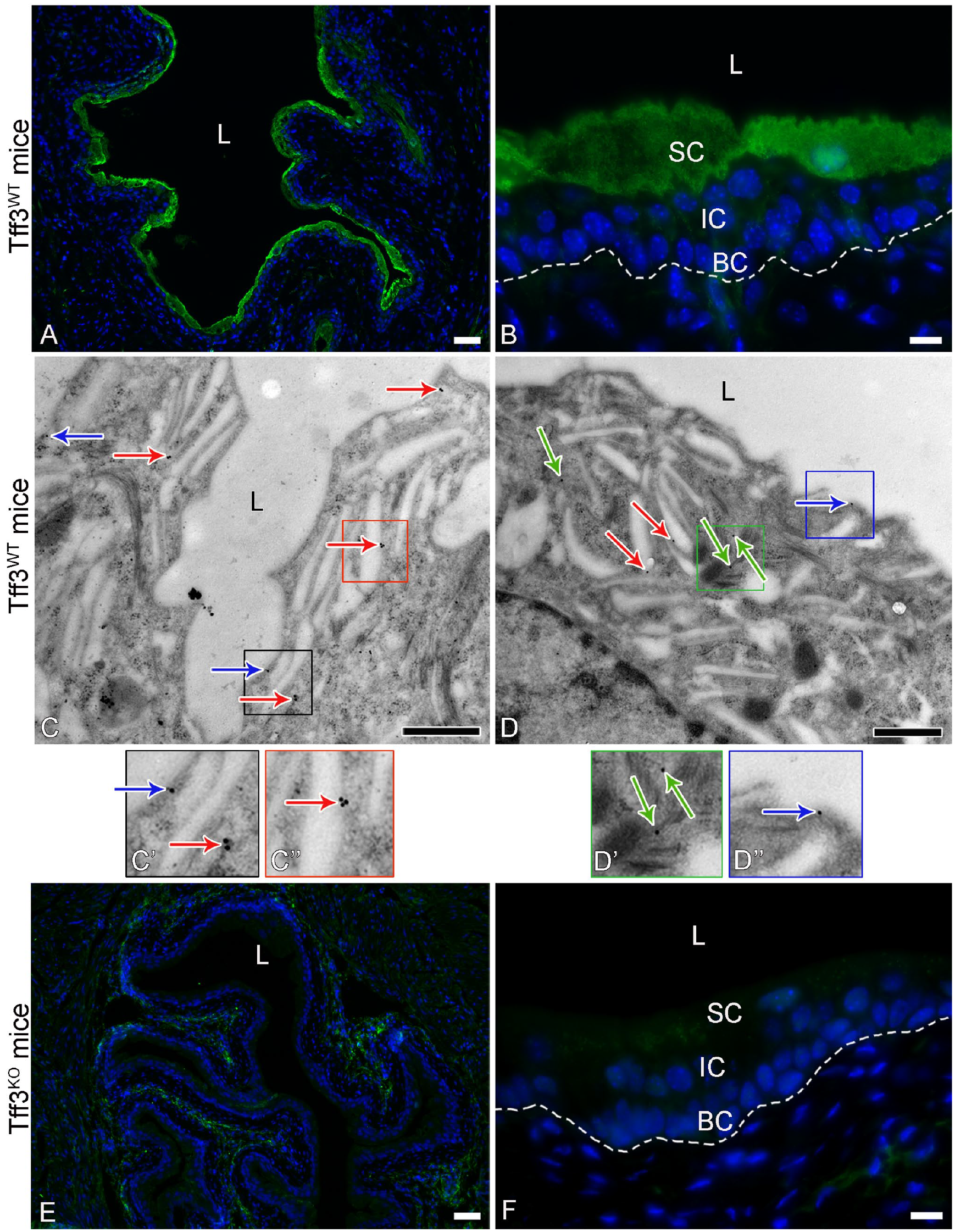
Representative micrographs of immunoreactions against Tff3 in the urinary bladders of Tff3WT and Tff3KO mice. (A, B) Strong and specific cytoplasmic immunofluorescence reaction (green fluorescence) in superficial cells of the urothelium of Tff3WT mice. The cell nuclei are labeled with the DNA-specific fluorescent dye DAPI (blue fluorescence). The dotted line represents the basal lamina in image B. (C, D) Immunogold labeling of Tff3 at the apical plasma membrane (blue arrows), at the membrane of the fusiform vesicles (red arrows), and at the cytoskeletal filaments (green arrows) of the superficial urothelial cells of Tff3WT mice. C’, C’’, D’, and D’’ are higher magnifications of the framed areas in C and D, in which the gold nanoparticles are clearly visible. (E, F) No immunofluorescence reaction against Tff3 in the urothelium of Tff3KO mice. The dashed line represents the basal lamina in image F. The cell nuclei are labeled with the DNA-specific fluorescent dye DAPI (blue fluorescence). SC-superficial cells, IC-intermediate cells, BC-basal cells, L-lumen of the urinary bladder. Images A, B, E, and F were taken with the wide-field fluorescence microscope. Scale bars: 50 µm (A, E), 10 µm (B, F), 600 nm (C, D).
Tff3 Colocalizes to Urothelium-Specific Transmembrane Protein Uroplakin UP1b
GlcNac-α-1, 4-Gal is the terminal disaccharide in the extracellular domain of uroplakin UP1b, one of the four uroplakin proteins that form urothelial plaques in the apical plasma membrane of superficial urothelial cells. 24 It is also the binding site for Tff3, 9 suggesting that these two proteins may interact in the urothelium. Using confocal microscopy, we were able to show that the signals for the proteins Tff3 and UP1b are indeed colocalized in the superficial urothelial cells (Fig. 3A to C).
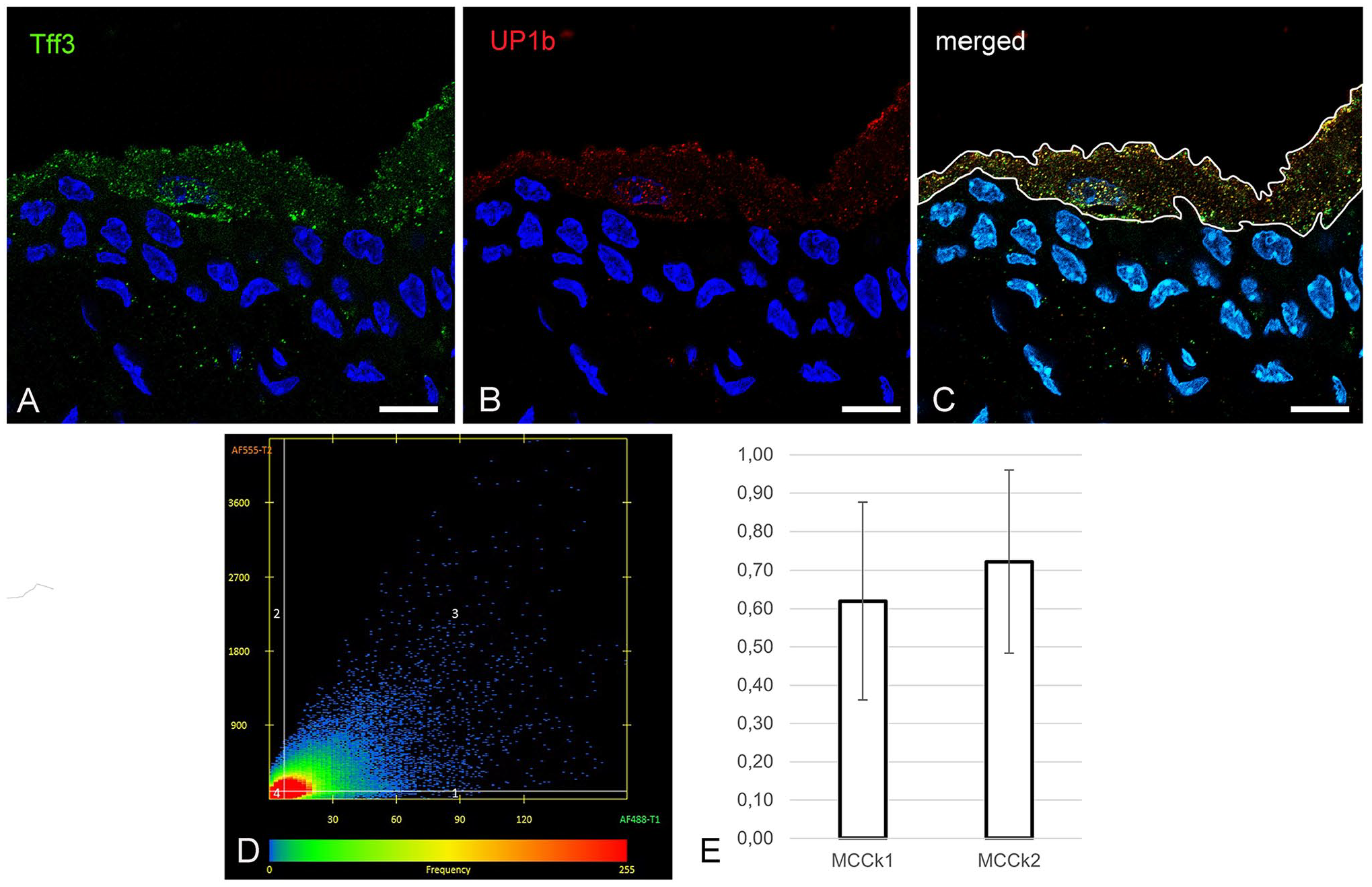
The colocalization analysis of Tff3 and UP1b in superficial urothelial cells of Tff3WT mice. (A-C) Representative micrographs of double immunofluorescence labeling of Tff3 (green fluorescence) and UP1b (red fluorescence) on cryosections of urinary bladders from Tff3WT mice. The cell nuclei are labeled with the DNA-specific fluorescent dye DAPI (blue fluorescence). The white line on image C shows the region of interest (ROI) selected for colocalization analysis, which was restricted to superficial urothelial cells with detected nuclei. (D) The points in the scatter plot are arranged based on the signal expressed in each pixel in the selected ROI. The points in quadrant 3 represent pixels with both signals (colocalized pixels). The frequency of pixels with a certain signal ratio is represented by a color scale (blue: low frequency; red: high frequency). (E) The graph shows the Manders’ coefficients MCCk1 and MCCk2 for the proteins Tff3 and UP1b in superficial urothelial cells. The results are presented as mean ± SD. The quantitative analysis was performed on 30 images of two technical and three biological replicates. Images A-C were taken with the confocal microscope. Scale bars: 10 µm (A, B, C).
For the quantitative analysis of colocalization we used Manders’ colocalization coefficients (MCCk1 and MCCk2). In the superficial urothelial cells, we determined the mean MCCk1 of 0.62 ± 0.26, meaning that 62% of pixels with a signal for Tff3 correlated with pixels with a signal for UP1b, and the mean MCCk2 of 0.72 ± 0.24, meaning that 72% of pixels with a signal for UP1b correlated with pixels with a signal for Tff3 (Fig. 3D and E).
Acute Urothelial Injury Is More Severe in Tff3KO Mice and Consequently Urothelial Regeneration Is Attenuated Compared With Tff3WT Mice
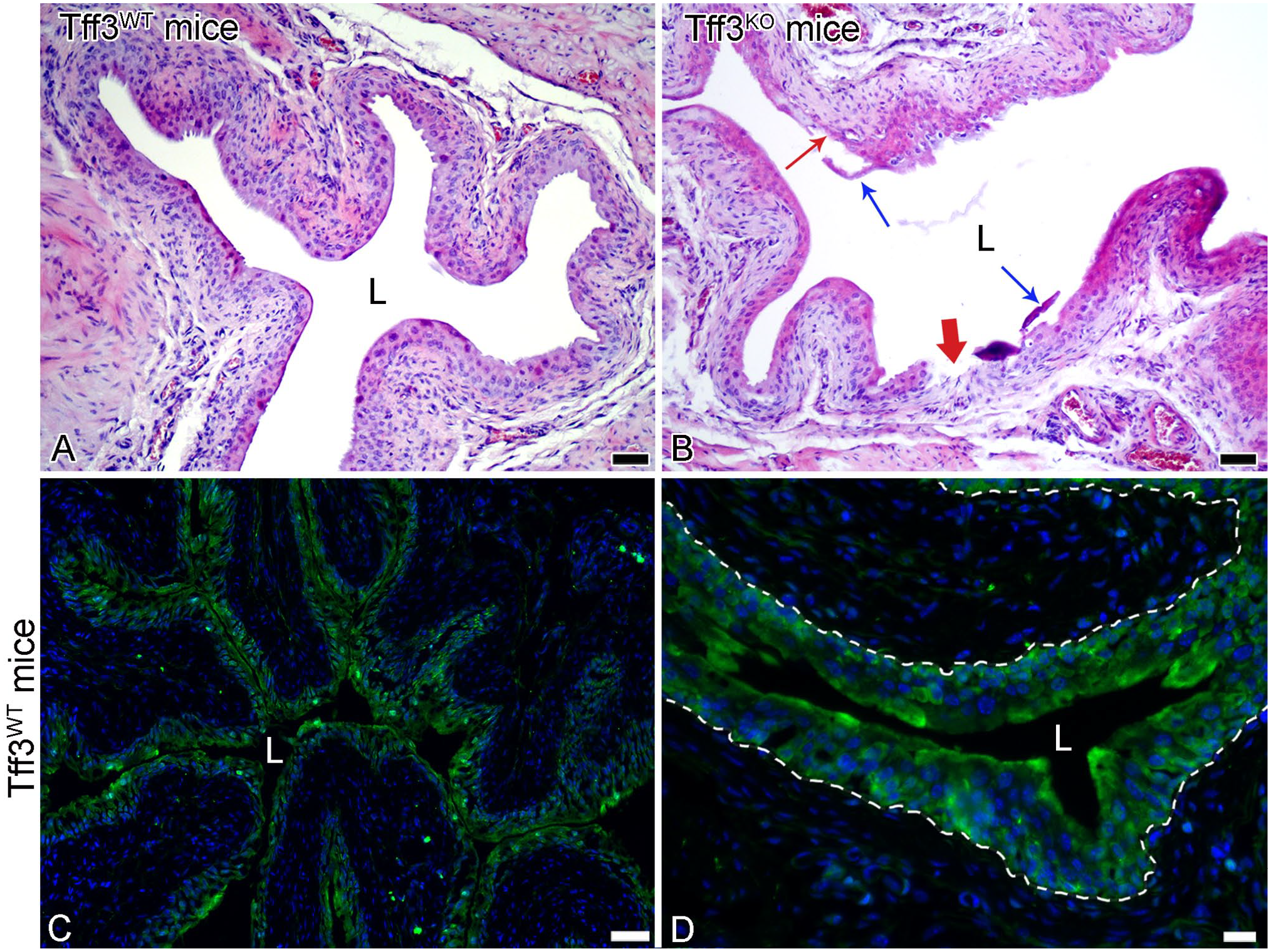
Based on literature data reporting the role of Tff3 in mucosal integrity and wound healing,25 –27 we intended to verify the potential protective role of Tff3 in the urothelium. Therefore, we induced urothelial injury in Tff3WT and Tff3KO mice with chitosan, a chemical agent that causes desquamation of urothelial cells, 23 and comparatively analyzed the extent of urothelial injury in histological bladder sections from both groups of mice. In the sections from Tff3WT mice, predominantly bilayered urothelium with desquamated superficial cells in the bladder lumen was observed, indicating that the desquamation was limited to the superficial cell layer only (Fig. 4A and B). In the tissue sections from Tff3KO mice, the urothelial injury was more severe. Most of the urothelium was bilayered, but there were also some single-layered areas or even completely denuded areas in the urothelium. Numerous desquamated superficial, intermediate and basal cells were therefore detected in the bladder lumen (Fig. 4C and D).
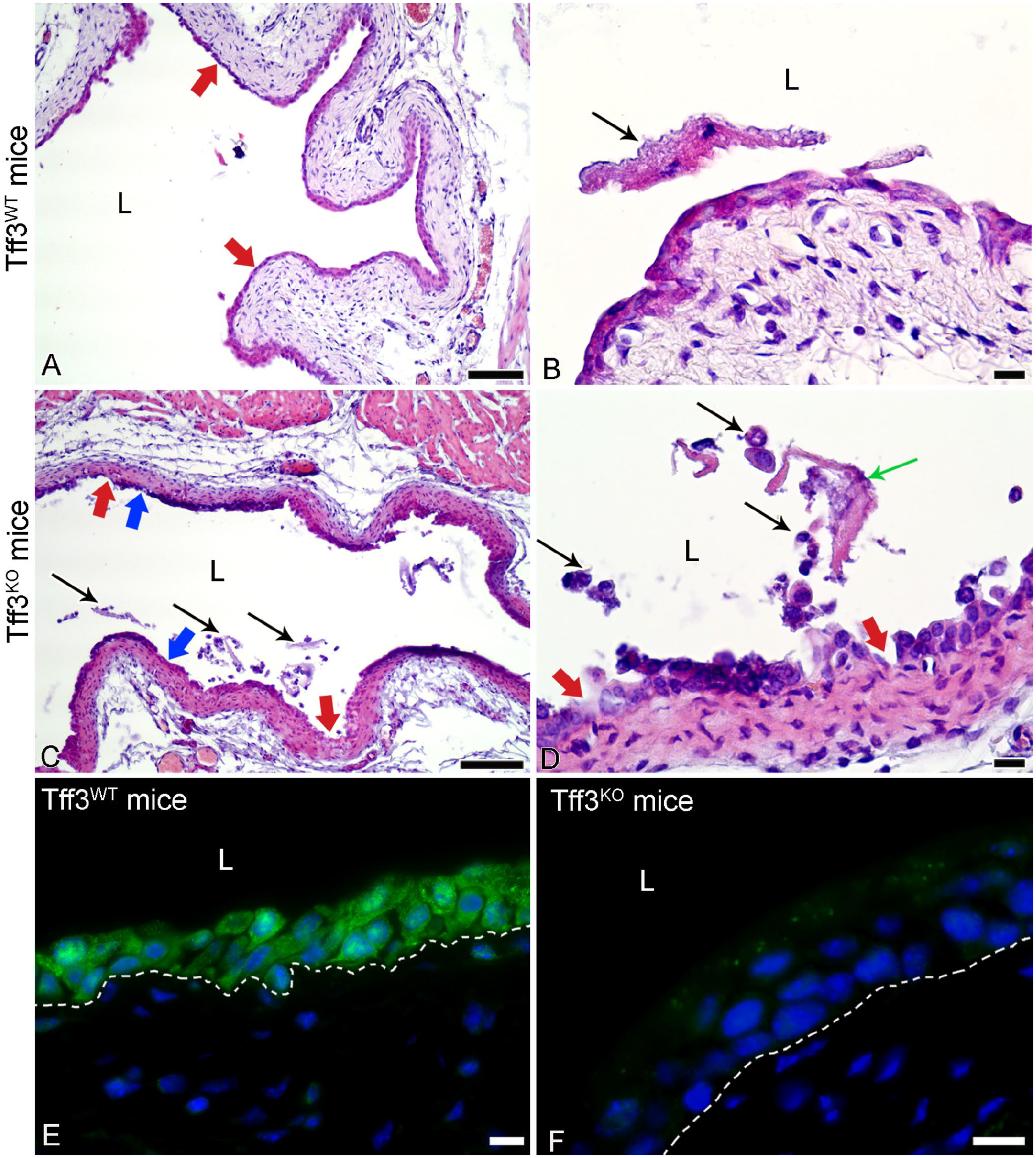
Representative micrographs of urinary bladders with induced urothelial injury in Tff3WT and Tff3KO mice. (A) Mainly bilayered urothelium (thick arrows) of Tff3WT mice due to induced desquamation of the superficial urothelial cells. (B) Desquamated cells (arrow) in the lumen (L) of the urinary bladder of Tff3WT mice. (C) In the bladders of Tff3KO mice, the urothelium is mainly bilayered, but with some deeper erosions of only single-layered urothelium (blue thick arrows) or completely denuded urothelium with exposed basal lamina (red thick arrows). There are numerous desquamated urothelial cells in the lumen (black thin arrows). (D) A higher magnification view of desquamated superficial cells (green thin arrow) and smaller intermediate or basal urothelial cells (black thin arrows) in the lumen of the urinary bladder of Tff3KO mice. Red thick arrows show deep erosions with exposed basal lamina. (E) Strong and specific immunofluorescence reaction against Tff3 (green fluorescence) in the cytoplasm of all cells of the bilayered urothelium in Tff3WT mice. The cell nuclei are labeled with the DNA-specific fluorescent dye DAPI (blue fluorescence). The dotted line represents the basal lamina. (F) Negative immunofluorescence reaction against Tff3 in the urothelium of Tff3KO mice. The cell nuclei are labeled with the DNA-specific fluorescent dye DAPI (blue fluorescence). The dashed line represents the basal lamina. L-lumen of the urinary bladder. Images A-D: H&E staining. Images E and F were taken with the wide-field fluorescence microscope. Scale bars: 100 µm (A, C), 10 µm (B, D, E, F).
Immunolabeling of Tff3 resulted in a positive immunoreaction in all urothelial cells of the bilayered injured urothelium of Tff3WT mice, while the immunoreaction in the injured urothelium of Tff3KO mice was completely negative as expected and served us only as an additional control for antibody specificity (Fig. 4E and F).
SEM analysis revealed that most of the superficial cell layer in the urothelium of Tff3WT mice was desquamated and the exposed urothelial surface, which consisted of former intermediate cells, was completely intact (Fig. 5A and B). In the urothelium of Tff3KO mice, the superficial cell layer was completely desquamated, so that exposed former intermediate cells composed the entire bladder luminal surface, which was also damaged. Namely, disrupted intercellular junctions, the desquamation of single or multiple newly exposed cells and the surface lesions with exposed basal cells were present on the bladder surface of Tff3KO mice (Fig. 5C and D).
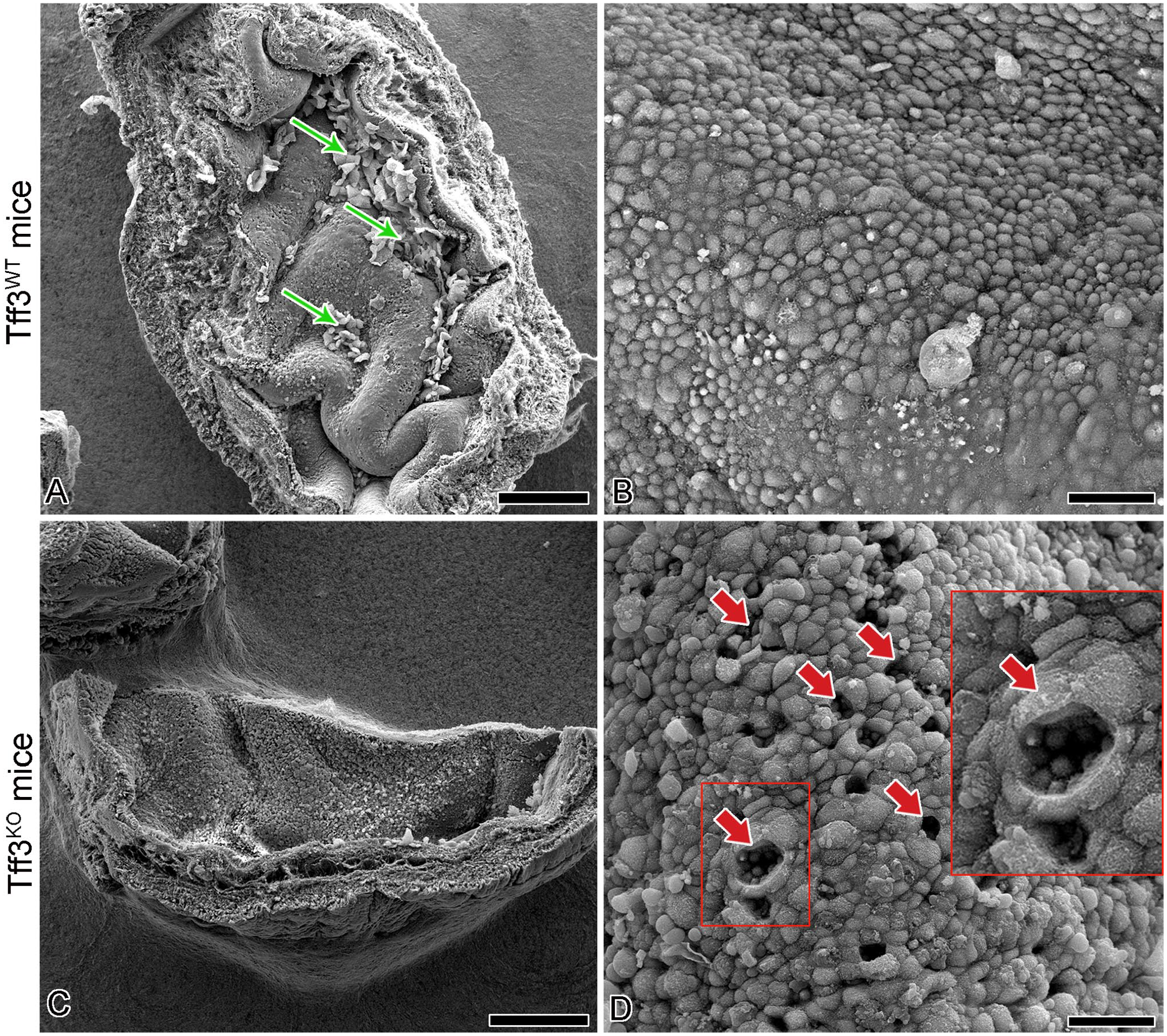
Representative SEM micrographs of the luminal surface ultrastructure of the injured urothelium in Tff3WT and Tff3KO mice. (A) Most of the luminal urothelial surface is desquamated but some areas with attached superficial cells are still present (arrows). (B) Due to desquamation of the superficial cell layer, smaller former intermediate cells are exposed on the urothelial surface as new superficial cells with intact cell junctions between them. (C) Luminal surface of the bladder with completely desquamated superficial cell layer. (D) The luminal surface consists mainly of former intermediate cells as new superficial cells, but with several punctate erosions due to desquamation of these cells. At those places, the basal urothelial cells are exposed on the urothelial surface (arrows). In the upper right corner is a higher magnification view of the punctate erosion in the boxed area. Note the small basal urothelial cells exposed on the luminal surface. Scale bars: 500 µm (A, C), 50 µm (B, D).
To evaluate the potential impact of Tff3 on urothelial regeneration after experimentally induced urothelial injury, the bladders of Tff3WT and Tff3KO mice were examined during the regeneration period, specifically 7 days after induction of urothelial injury. In the histological sections of the bladders from Tff3WT mice, the urothelium was again three-layered and completely recovered on regeneration day 7. At the same time-point of regeneration, the urothelium in Tff3KO mice was predominantly three-layered, but some injured areas were still preserved (Fig. 6A and B). Immunoreaction against Tff3 is present in all three cell layers of the restored urothelium in Tff3WT mice at regeneration day 7 (Fig. 6C and D).
Representative micrographs of the urothelium in Tff3WT and Tff3KO mice on regeneration day 7 after induced urothelial injury. The urothelium of Tff3WT mice is normoplastic and intact (A), while the urothelium of Tff3KO mice is still injured in some areas, where cell desquamation (blue thin arrows) and shallow (red thin arrow) or deep erosions (red thick arrow) are present (B). Strong and specific immunofluorescence reaction against Tff3 (green fluorescence) in all cells of the three-layered urothelium in Tff3WT mice (C), clearly visible at higher magnification (D). The cell nuclei are labeled with the DNA-specific fluorescent dye DAPI (blue fluorescence in images c and d). The dashed line represents the basal lamina. L-lumen of the urinary bladder. Images A and B: H&E staining. Images C and D were taken with the wide-field fluorescence microscope. Scale bars: 100 µm (A, B), 50 µm (C), 20 µm (D).
Images of Tff3 immunofluorescence labeling of urinary bladders from Tff3WT mice showing the progression of changes in Tff3 expression in normal urothelium, acutely injured, recovering and fully recovered urothelium can be found in the Appendix (Fig. A3).
Discussion
The TFF peptides are small peptides that are found in various mammalian tissues and have multiple functions. All TFF members are typical exocrine products of mucous epithelia. TFF3 is present in high concentrations in the intestine and its role in this tissue is well examined.3,28,29 In contrast, there is very little literature data on the presence and function of TFF3 in the mouse urinary bladder, which is not a mucous epithelium. In the present study, we were able to detect the expression of all three Tffs in the urinary bladder, but we were particularly interested in identifying the role of Tff3 in the urinary bladder using different methods at the molecular and cellular level. Using immunofluorescence labeling, we were able to demonstrate that Tff3 is expressed in the urothelium, predominantly in its superficial cells. Immunogold labeling and subsequent analysis by TEM, which enables high-resolution analysis, confirmed the localization of Tff3 in the superficial urothelial cells at their apical plasma membrane, the membranes of the fusiform vesicles, and the cytoskeletal filaments. Fusiform vesicles are specialized vesicles of the superficial urothelial cells that are responsible for the intracellular transport of uroplakin-containing urothelial plaques to the apical plasma membrane. 30 Uroplakin UP1b is one of the four uroplakins in urothelial plaques that form a membrane of fusiform vesicles and an asymmetrically thickened apical plasma membrane.31,32 This specifically structured apical plasma membrane of the superficial cells makes an important contribution to the barrier function of the urothelium.33,34 UP1b contains a recognizable disaccharide in its extracellular domain that is necessary for TFF3 binding. 9 Our quantitative analysis of colocalization of pixels with the signal for Tff3 and UP1b proteins in superficial urothelial cells from Tff3WT mice showed that the signals for both proteins are quite colocalized (MCCk1 = 0.62 ± 0.26, MCCk2 = 0.72 ± 0.24). These results suggest that Tff3 interacts with UP1b in superficial urothelial cells, which is consistent with the results of immunogold labeling of Tff3 confirming its presence at the membranes of fusiform vesicles and the apical plasma membrane. Since the uroplakin UP1b forms the apical plasma membrane of superficial urothelial cells and contributes to the barrier function of the urothelium, we speculate that Tff3 interacting with UP1b may play a role in the urothelial permeability barrier or even in the fine-tuned regulation of cell surface receptors through interaction with carbohydrate moieties.
Trefoil factor family peptides are integral components of the mucus in the mucosal barriers and as such play a crucial role in protecting the barrier and promoting the rapid repair of mucous epithelia after injury.10,12,35,36 TFF3 has been shown to act as a mitogen in the intestinal mucosa and is involved in the orderly dissociation of intercellular contacts to release viable cells for movement toward the injured area, in cytoskeletal reorganization, and signal production to promote cell migration. 8 Although the urothelium does not secrete mucus with TFF peptides entrapped, as is the case with mucous epithelia, we have demonstrated the role of Tff3 in protecting the urothelium. Indeed, we showed that chitosan-induced urothelial injury was more severe in the bladders of Tff3KO mice than in Tff3WT mice. The desquamation of the urothelium extended deeper and involved more urothelial cells in the bladders of Tff3KO mice than in those of Tff3WT mice. These results provide indirect but clear evidence that Tff3 plays a protective role in the urothelium. Interestingly, after induction of acute injury, Tff3 was expressed in all cells of the damaged urothelium of Tff3WT mice and not only in the superficial cells as observed in the normal urothelium of Tff3WT mice. Since the superficial urothelial cells are the main site of Tff3 expression according to the present data, we could speculate that after desquamation of the superficial cells, the remaining urothelial cells started to express Tff3 to maintain the protection of the damaged urothelium. These results could be a further indication of a protective role of Tff3. The exact mechanisms of action of Tff3 in urothelial restitution are still unknown and beyond the scope of our study. We can only speculate that the molecular mechanisms of Tff3 action on urothelial protection and renewal are unique because the urothelium is a special epithelium in many respects.
We also investigated whether the absence of Tff3 in Tff3KO mice affects the regenerative capacity of the injured urothelium. Histological examination of bladders obtained 7 days after chitosan-induced urothelial injury revealed that the urothelium of Tff3KO mice was mostly normoplastic, with only occasional erosions and almost complete recovery. This finding was surprising, as delayed recovery of the urothelial architecture had been expected due to the greater extent of injury in Tff3KO mice. The present results suggest that the urothelium lacking Tff3 retains the ability to regenerate but may be more susceptible to damaging factors. At the same time, it is worth noting that Tff3 was expressed in all urothelial cell layers of Tff3WT mice 7 days after injury, indicating that the urothelium has not yet fully recovered at the subcellular level. Overall, our results suggest that Tff3 contributes to the maintenance of urothelial homeostasis, as previously found in a study of induced epithelial injury in the mouse small intestine. 37 Our results are also consistent with the findings of Mashimo H and colleagues, 25 who reported that Tff3KO mice were more sensitive to dextran sulfate sodium (DSS) compared with wild-type mice in the DSS-induced colitis model. They also observed that self-renewal of colon epithelium was severely delayed in Tff3KO mice due to poor epithelial regeneration after injury. However, for a more comprehensive analysis of the contribution of Tff3 to the regenerative capacity of the urothelium, the urothelial tissue should be analyzed more consistently at many time points during regeneration using additional and different methodological approaches, but this is beyond the scope of the present study.
This study is the first to describe the exact location and role of Tff3 in the mouse urinary bladder. All TFF members are evolutionarily highly conserved, with 70% amino acid homology between the human and rodent Tff3 peptide 38 and similar genomic organization of all three Tff genes in humans and mice.39,40 Therefore, mice are established and suitable preclinical models for the study of Tff peptides in the pathogenesis of the urinary tract.
In summary, we hypothesize that Tff3 plays a fine-tuned role in urothelial homeostasis and protection. The present study is one of the first analyses of TFF3 in the urinary bladder. It contributes to new insights into the biology of the urothelium as an important body barrier and thus provides a basis for further studies on TFF3 in the prevention or therapy of various forms of urothelial injury caused by bladder stones or cystitis.
Footnotes
Appendix
Acknowledgements
We thank Nada Pavlica and Sabina Železnik for technical support.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AE designed the study, performed some methods and the data analyses and drafted the manuscript, UDJ performed confocal microscopy and image analysis and contributed to the preparation of the figures, DP performed molecular genetic methods, KŠ and IB contributed substantially to the animal experiments, and MBL helped to perform the study with constructive discussions and contributed to the animal experiments. All authors read the manuscript, gave constructive suggestions, and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This research was funded by the Slovenian Research and Innovation Agency (ARIS) (grant no. P3-0108 and J3-2521) and by the Croatian Science Foundation (grant IP-06-2016-2717). AE has received research support from the Slovenian Research and Innovation Agency and MBL has received research support from the Croatian Science Foundation.
