Abstract
Branched-chain amino acids (BCAAs) play vital roles in metabolic and physiological processes, with their catabolism initiated by two branched-chain aminotransferase isozymes: cytosolic (BCATc) and mitochondrial (BCATm). These enzymes have tissue and cell-specific compartmentalization and are believed to shuttle metabolites between cells and tissues. Although their expression and localization have been established in most tissues, ocular tissues remain unknown. In this study, we used immunohistochemical analyses to investigate the expression and localization of BCAT enzymes in the normal eye tissues. As expected, BCATc was highly expressed in the neuronal cells of the retina, particularly in the ganglion cell layers, inner nuclear layer, and plexiform layer, with little to no expression in Müller cells. BCATc was also present in the cornea, retinal pigment epithelium (RPE), choroid, ciliary body, and iris but not in the lens. In contrast, BCATm was expressed across all ocular tissues, with strong expression in the Muller cells of the retina, the endothelial and epithelial layers of the cornea, the choroid and iris, and the epithelial cells at the lens’s front. The extensive expression and distribution of BCAT isozymes in the ocular tissue, suggests that BCAA transamination is widespread in the eye, potentially aiding in metabolite transport between ocular tissues. The findings provide new insights into the physiological role of BCATs in the eye, particularly within the neuronal retina.
This is a visual representation of the abstract.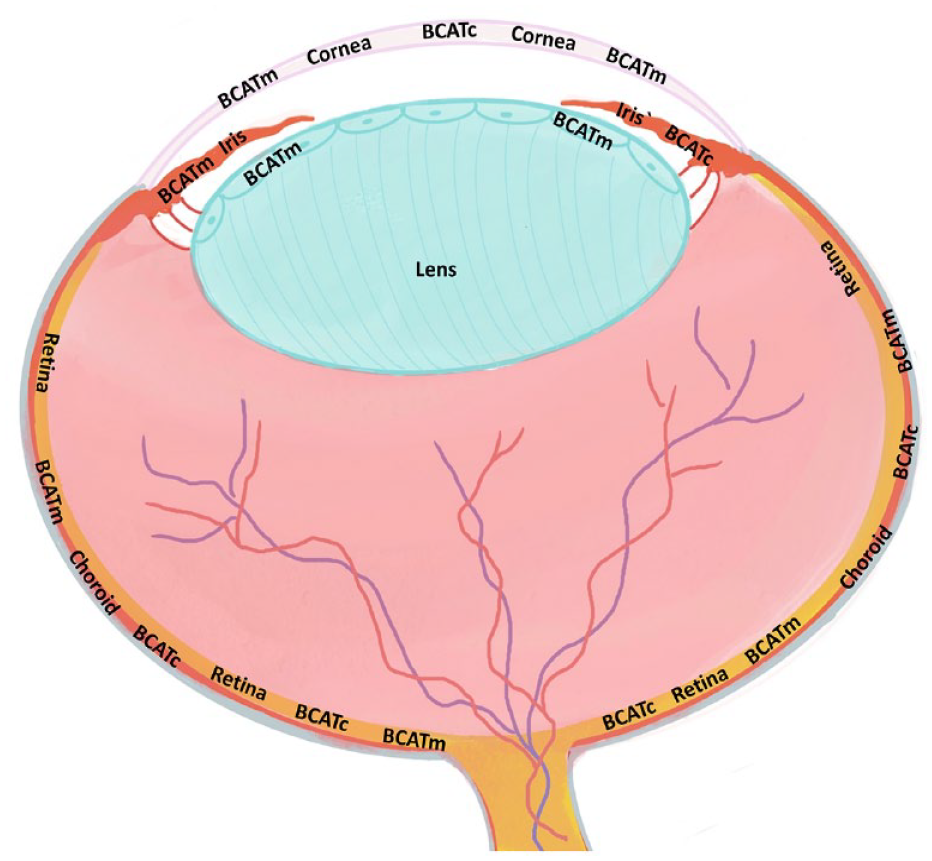
Introduction
Branched-chain amino acids (BCAAs), Leucine (Leu), Isoleucine (Ile), and valine (Val) are essential amino acids with significant metabolic and physiological functions.1,2 Branched-chain aminotransferase (BCAT) catalyzes the reversible transfer of the amino group from a BCAA to α-ketoglutarate (α-KG) to produce branched-chain keto acids (BCKA) and glutamate. BCAAs and their metabolites play essential physiological functions in nutrition signaling, glucose and lipid homeostasis, immunological response, neurotransmission, and energy homeostasis.3,4 The BCAT enzyme exists in two primary isoforms in mammals: cytosolic BCATc (also known as BCAT1) and mitochondrial BCATm (also known as BCAT2). These isozymes are 58% identical in amino acid sequence and have the same substrate specificity, but differ in cellular location, tissue distribution, catalytic efficiency, and regulation.3,5 The mitochondrial isozyme BCATm is expressed widely in most tissues, but high levels are mainly reported in the kidneys, stomach, and brain.3,6 The cytosolic isozyme BCATc is expressed only in the brain, peripheral nerves, ovaries, and placenta.7,8 Although the expression of BCAT isozymes has been established in most tissues, little is known regarding the expression and functional roles of BCAT enzymes in the ocular tissues, particularly in the retina.
The compartmentalization of BCAT isozymes expression in various tissues and cells is thought to play a functional role in transporting BCAA metabolites between tissues and cells.3,8,9 For instance, the muscle-specific BCATm metabolizes BCAAs to produce nitrogen to maintain glutamate, alanine, and glutamine pools. It facilitates the export of some of these nitrogen-rich amino acids from muscle to be taken up by the liver, kidneys, and intestines for energy production during gluconeogenesis. 3 BCATc has been detected in neurons from all brain regions.9,10 However, BCATm is expressed in rat brain astrocytes and human vascular endothelial cells.7,9,11 Glutamate is a major neurotransmitter that also serves as a precursor in synthesizing γ-aminobutyric acid (GABA), an inhibitory neurotransmitter. The presence of BCAT enzymes in brain cells supports glutamate synthesis, which plays an essential role in several metabolic functions of the brain. Moreover, BCAT controls the glutamate/glutamine cycle for neurotransmission by maintaining and regulating glutamate levels between neurons and glial cells/endothelial cells. 3 Although BCAAs and their metabolites are essential in normal tissue function, their levels are altered in several diseases, such as diabetic retinopathy, brain disorders, and cancer.12–14 Determining the local and cellular distribution of BCAT enzymes in the eye and neuronal retina provides a rationale since dysregulated BCAA substrates and their intermediates may disrupt the steady-state operation of neuronal function in the retina.
BCAA amino acids’ homeostasis is critical, which may be achieved by regulating BCAT enzymes. The differential expression of BCAT enzymes in ocular tissues has not been reported except of a few studies that assessed enzyme activity and demonstrated expression through Western blot analysis.10,11 In cell culture studies, BCATm is highly expressed within the Muller cells, which are the primary glial cells of the retina.10,11 Moreover, LaNoue and coworkers 10 demonstrated that inhibiting neuronal BCATc with gabapentin, an analog of leucine, reduced de novo synthesis of glutamate by 30%. The study also suggested that the distribution of the BCATc and BCATm isozymes in retinas could be similar to that observed in rat brains. In this study, we sought to study the cellular distribution of BCAT isozymes in the retina and other parts of eye tissues to understand better these enzymes’ role in BCAA metabolism and their function.
Materials and Methods
Experimental Animals
Male Sprague-Dawley rats (250–300 g, age 8–12 weeks) were purchased from the Experimental Surgery and Animal (ESAL) Laboratory, College of Medicine, King Saud University, Riyadh, Saudi Arabia. Animals were kept in controlled environmental conditions (25oC and a 12-h light/dark cycle) with free access to food and water. The rats were given anesthesia, sevoflurane (Tabuk pharmaceutical, KSA), and the eyeballs from five different rats were enucleated and processed for immunohistochemistry/immunofluorescence. All experimental procedures and protocols were performed in accordance with ARVO guidelines for the Care and Use of Experimental Animals and the Guidelines of the Experimental Surgery and Animal (ESAL) Laboratory, College of Medicine, King Saud University, Riyadh, Saudi Arabia. This study was approved by the Animal Ethics Committee at King Saud University (Reference No: KSU-SE-21-04).
Immunohistochemistry (IHC)
The whole eyes of rats were fixed in 4% paraformaldehyde buffer for 24 hr. Then, they were kept in 70% ethanol for 24 hr before being embedded in paraffin. For classic histological analysis, samples were embedded in paraffin blocks, serially cut into 5-µm-thick parts using a rotary microtome, and stained with hematoxylin-eosin. The automatic system (BenchMark XT; Ventana, Basel, Switzerland) was used for IHC analysis. Briefly, the eye-embedded blocks were cut into 3-µm-thick pieces and placed on coated slides, five to six sections per eye were used. After being deparaffinized, sections were subjected to epitope retrieval using Cell Conditioning Solution 1 (CC1) (Ventana) for 64 min before incubation with Anti-Rat BCAT1 (ABIN718856, 1:100; Antibodies-online, Aachen, Germany) or BCAT2 polyclonal antibodies (ABIN718511, 1:100; Antibodies-online, Aachen, Germany), for 40 min. Slides stained on the Ventana instruments were detected with an UltraView DAB kit (Ventana, Basel, Switzerland). Slides were stained using Hematoxylin II and incubated in a bluing reagent for four min. Finally, coverslips were applied. Positive controls are shown in Figure A4,6. Negative control sections were simultaneously stained but with omitted primary antibody incubation. The immunostained sections were viewed and photographed by Olympus light microscope and DP71 Olympus digital camera with magnification 10×, 20×, 40×, and 100×. We confirmed our results using manual IHC methods using an Abcam kit; details are in the Appendix. Each experiment has been repeated 3 times using serial sections (every fourth and eighth section).
Immunofluorescence
Eye sections on slides were dewaxed and rehydrated, then washed twice with distilled water and once with phosphate buffer saline (PBS). The deparaffinized sections were treated in 10 mM sodium citrate buffer containing 0.05% tween 20 for antigen retrieval. Eye sections were blocked with 10% normal goat serum for an hour to inhibit nonspecific sites and then incubated overnight with primary antibodies at 4oC: rabbit anti-BCAT1 (ABIN718856, 1:300 dilution; Antibodies-online), rabbit anti-BCAT2 (ABIN718511, 1:300 dilution; Antibodies-online) and mouse anti-glutamine synthetase (1.300 dilutions; Santa Cruz Biotechnology, Dallas, USA). After incubations, all the sections were rinsed with phosphate buffer saline (PBS) and incubated with corresponding secondary antibodies: goat anti-rabbit (Alexa Fluor 594, 1:500, Abcam, Cambridge, UK) and/or goat anti-mouse IgG H&L (Alexa Fluor 488, 1:500; Abcam, Cambridge, UK) for 45 min at room temperature in the dark. The slides were washed with PBS and incubated in DABI solution (Abcam) for 3 min away from light. Finally, coverslips were applied, and sections were examined and photographed using a ZEISS spinning disk confocal microscope (Zeiss, Germany).
Results
We performed IHC analysis on ocular tissues to determine the cellular expression and localization of BCAT isozymes in the retina, retinal pigment epithelium (RPE), choroid, ciliary body, iris, cornea, and lens. Histology with hematoxylin and eosin staining (H&E) images was included with all findings to identify the layers and tissues of the eyes.
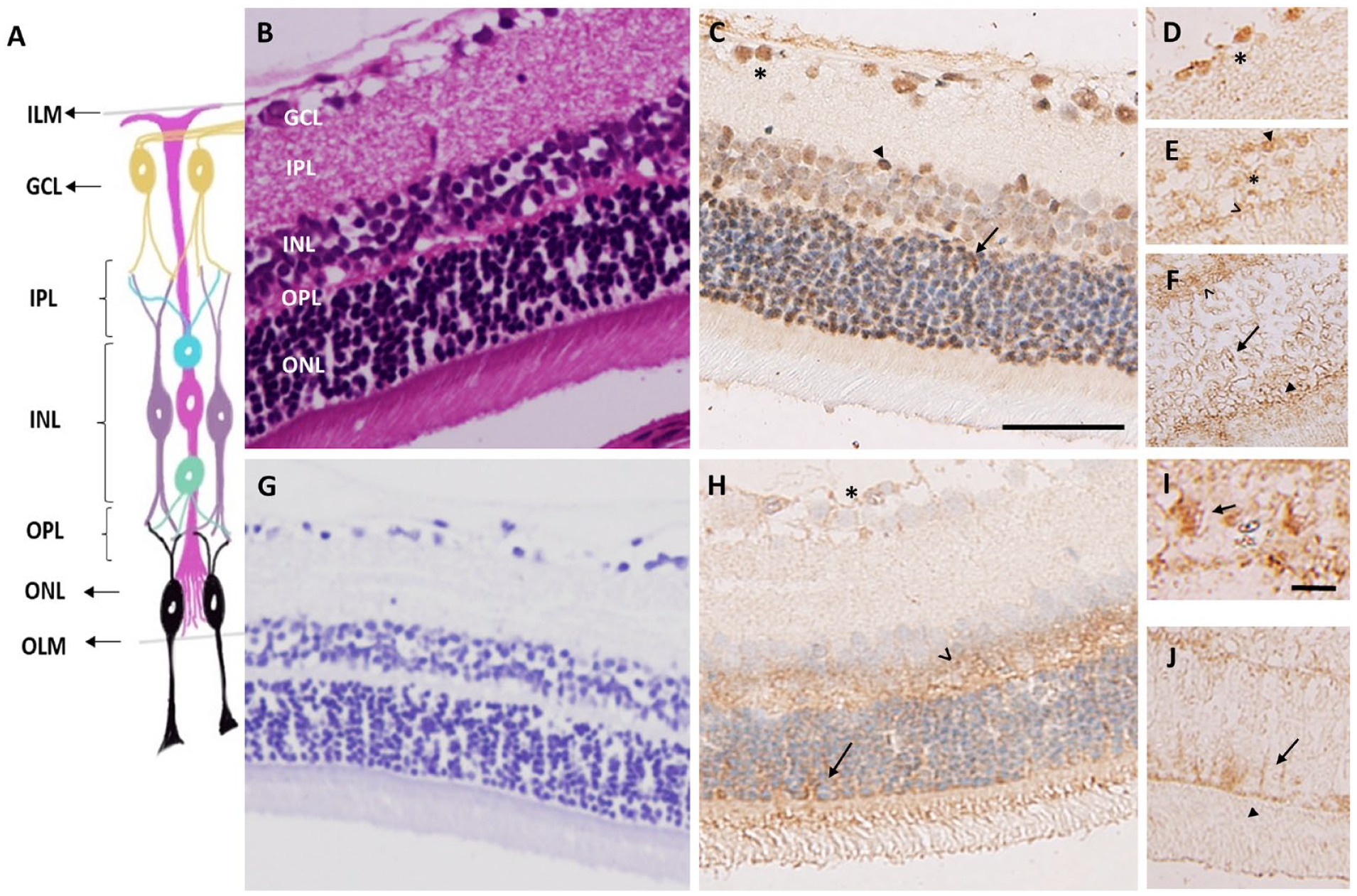
BCAT Expression in the Retina Layers
To localize the BCATc and BCATm enzyme expression, we used specific antibodies against those enzymes and performed immunohistochemical analyses in the intact sections of the rat retina (Fig. 1). Fig.1C shows immunolabeling of BCATc in the rat retina. Cell bodies within the ganglion cells layer (GCL) were strongly immunopositive for BCATc (Fig. 1D and A5). In addition, strong immunostaining was observed in the inner and outer nuclear layer (INL), which reflects the cell body of horizontal and amacrine cells (Fig. 1E). The outer nuclear layer, which contains the cell bodies of photoreceptors, also showed little immunostaining of BCATc. Almost all the neuronal layers of the retina appeared to be BCATc positive. Figure 1H, A7 shows immunostaining of BCATm in the retina. A low immunoreactivity of BCATm was detected throughout the retina. However, moderate immunolabeling was observed in the inner segment of the photoreceptor, the internal limiting membrane, and the outer limiting membrane. Muller cells extend from the inner membrane to the outer limiting membrane of the retina (Fig. 1I and J), indicating that BCATm is mainly expressed in Muller cells and some types of neuronal cells in the retina.
Expression and Immuno-Colocalization of the BCATc and BCATm With Glutamine Synthetase in the Rat Retina
We further investigated the expression of BCAT isozymes in the retina and their colocalization with glutamine synthetase (GS), a marker enzyme of retinal Muller cells (Fig. 2). As evident from panel A, BCATc was highly expressed in neuronal cells, but no or minimal expression was observed within Müller cells of the retina. The overlay panel indicates little or no overlap in the distribution of the two proteins. Furthermore, we determined the expression of BCATm in the retina by immuno-colocalization with GS (Fig. 2B and A8). Figure 2B shows an overlap between BCATm and GS, with the yellow color of BCATm and GS, suggesting BCATm co-localizes with the GS. Thus, BCATc appears to be expressed within neuronal cells of the intact rat retina. However, the BCATm isozyme is localized within Müller cells and a few neuronal cells in the INL (Fig. A8).
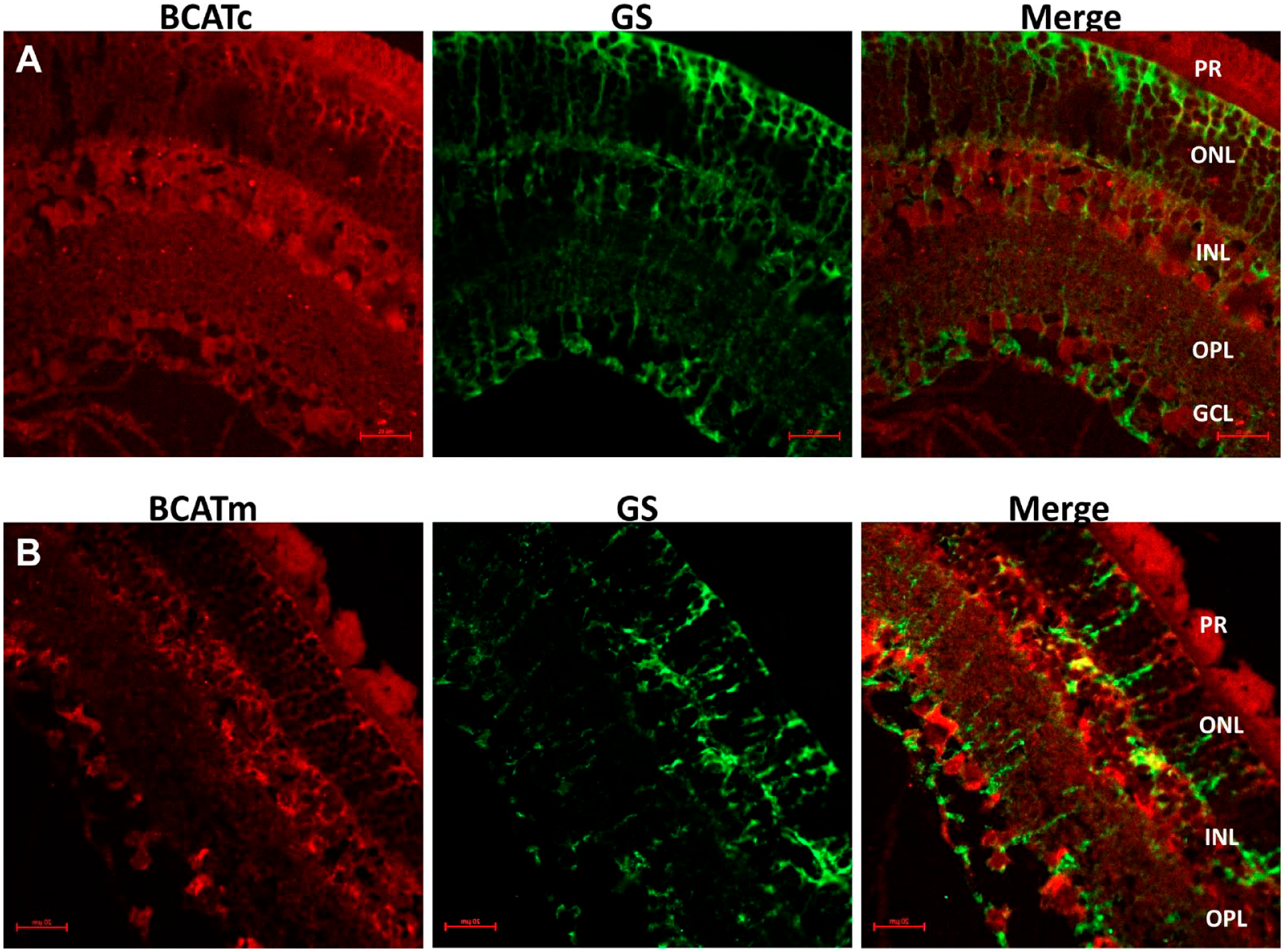
Expression and immuno-colocalization of branched-chain aminotransferase isozymes in the retina. (A) Red immunofluorescence stained for cytosolic branched-chain aminotransferase (BACTc), green immunofluorescence stained for glutamine synthetase (GS) (a marker of Muller cell), and merge image of BCATc and GS overlap. (B) Red immunofluorescence stained for mitochondrial branched-chain aminotransferase (BCATm, green immunofluorescence stained for GS, and merge image showing a BCATm and GS overlap. Scale bar; 20 µm.
BCATs Expression in the RPE and Choroid
The RPE and choroid section of the eye support and nourish metabolites, including amino acids in the retina. Using IHC methods, we analyzed the expression and distribution of the BCAT isozymes in the choroid and the outer part of the retina (RPE). Figure 3B shows BCATc immunopositive staining within the RPE and choroid especially in Haller’s layer and other layers of choroid (Fig. A9). The BCATm was also expressed in the choroid (Fig. 3C), with intense staining observed in the medium-sized vessels in the Sattler’s layer.
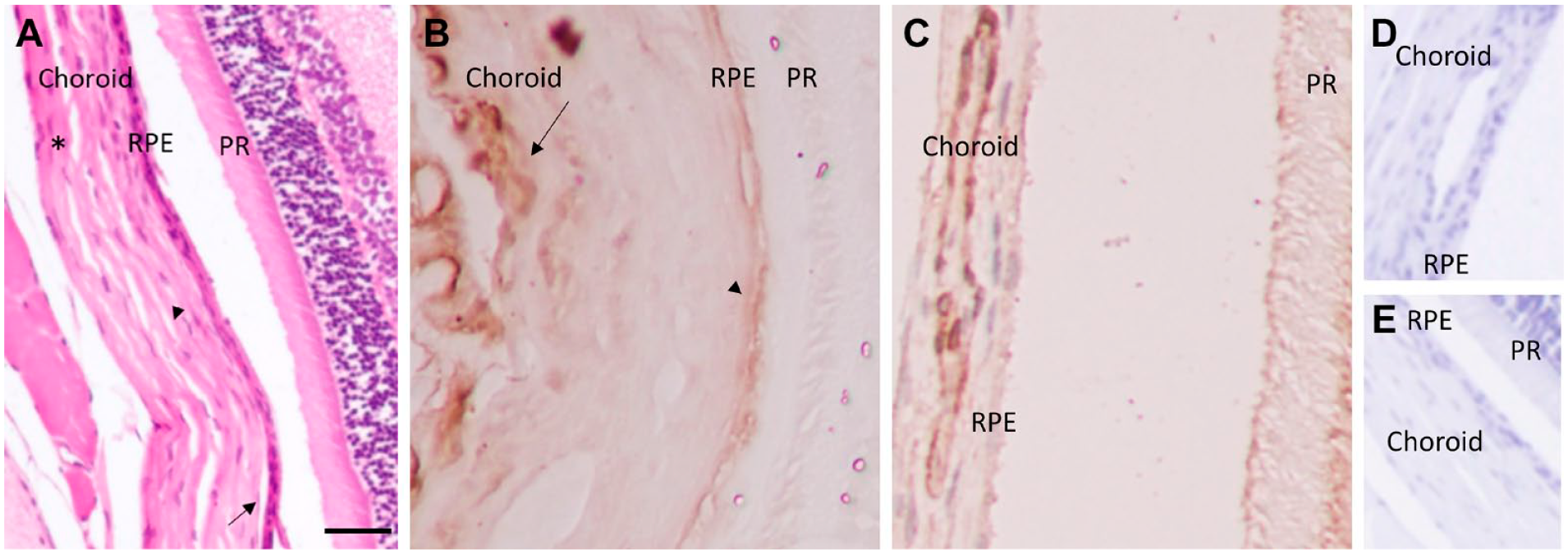
Expression of Branched-chain aminotransferase isoforms in the choroid and RPE. (A) H&E staining of the RPE and choroid layers; Choriocapillaris (large arrow), Sattler’s layer (small arrow), and Haller’s layer (*). (B) Staining of cytosolic branched-chain aminotransferase (BCATc) in the choroid (large arrow) and RPE (small arrow). (C) Staining of mitochondrial branched-chain aminotransferase (BCATm) within choroid. D&E) Negative control, without primary antibody. Scale bar 20 µm, 40× magnification.
Expression of the BCATc and BCATm in the Ciliary Body
We also determined the expression of BCAT enzymes in the ciliary body, which is the forward continuation of the retina and choroid. Figure 4B shows BCATc labeling within the ciliary body, especially in pigmented epithelial cells. BCATm was also expressed in the non-pigment epithelium (NPE), and pigment epithelium (PE) of the ciliary body (Fig. 4C), but intense staining was observed in the NPE. We further confirmed our results using immunofluorescence techniques, as shown in Fig. 5.
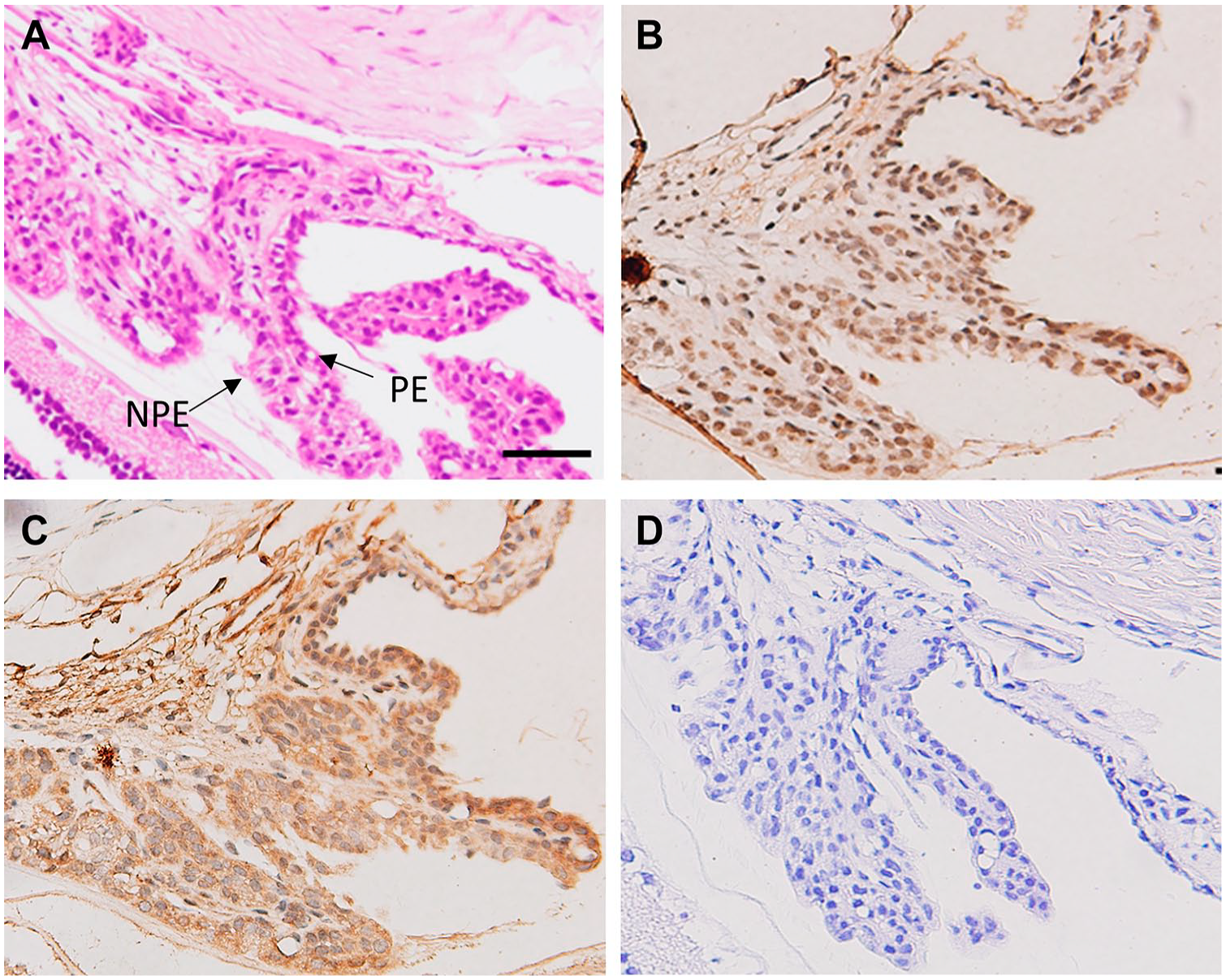
Expression of cytosolic branched-chain aminotransferase (BCATc) and mitochondrial branched-chain aminotransferase (BCATm) in the section of rat ciliary body. (A) H&E staining of the ciliary body. (B) BCATc staining in the pigment epithelium (PE) of the ciliary body, (C) BCATm staining in the PE & NPE ciliary body, (D) Negative control, without primary antibody. Scale bar 20 µm, 40× magnification.
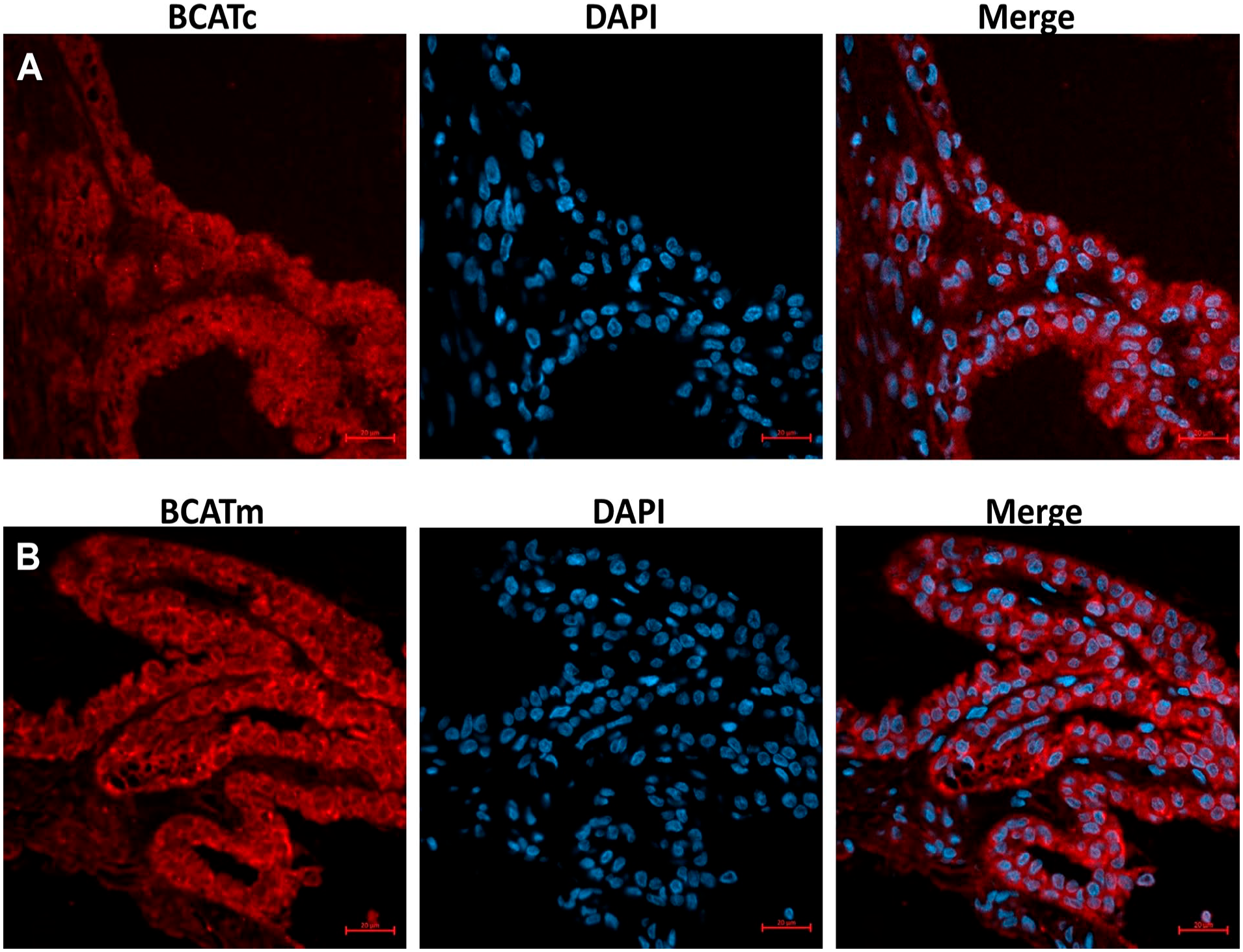
Immunofluorescence staining of branched-chain aminotransferase isozymes of the section of rat ciliary body. (A) Immunofluorescence stained for cytosolic branched-chain aminotransferase (BACTc). (B) Immunofluorescence stained for mitochondrial branched-chain aminotransferase (BACTm). DAPI was used as a nuclear stain. Scale bar 20 µm.
BCATs Expression in the Iris
We examined the expression of BCAT isozymes in the iris. Figure 6B shows a moderate immunoreactivity of BCATc throughout the iris. However, a strong immunoreactivity of BCATm in the iris was observed (Fig. 6C). The epithelium layers of the iris were strongly immunopositive with BCATm, and moderate staining was detected in the stroma and anterior limiting layer. We further confirmed our results using immunofluorescence methods to avoid the interference between nuclear staining and brown peroxidase reaction (Fig. 7). Similar to histochemistry results, a moderate expression of BCATc was observed in the iris. Still, a high expression of BCATm was found in the epithelial and stroma of the iris.
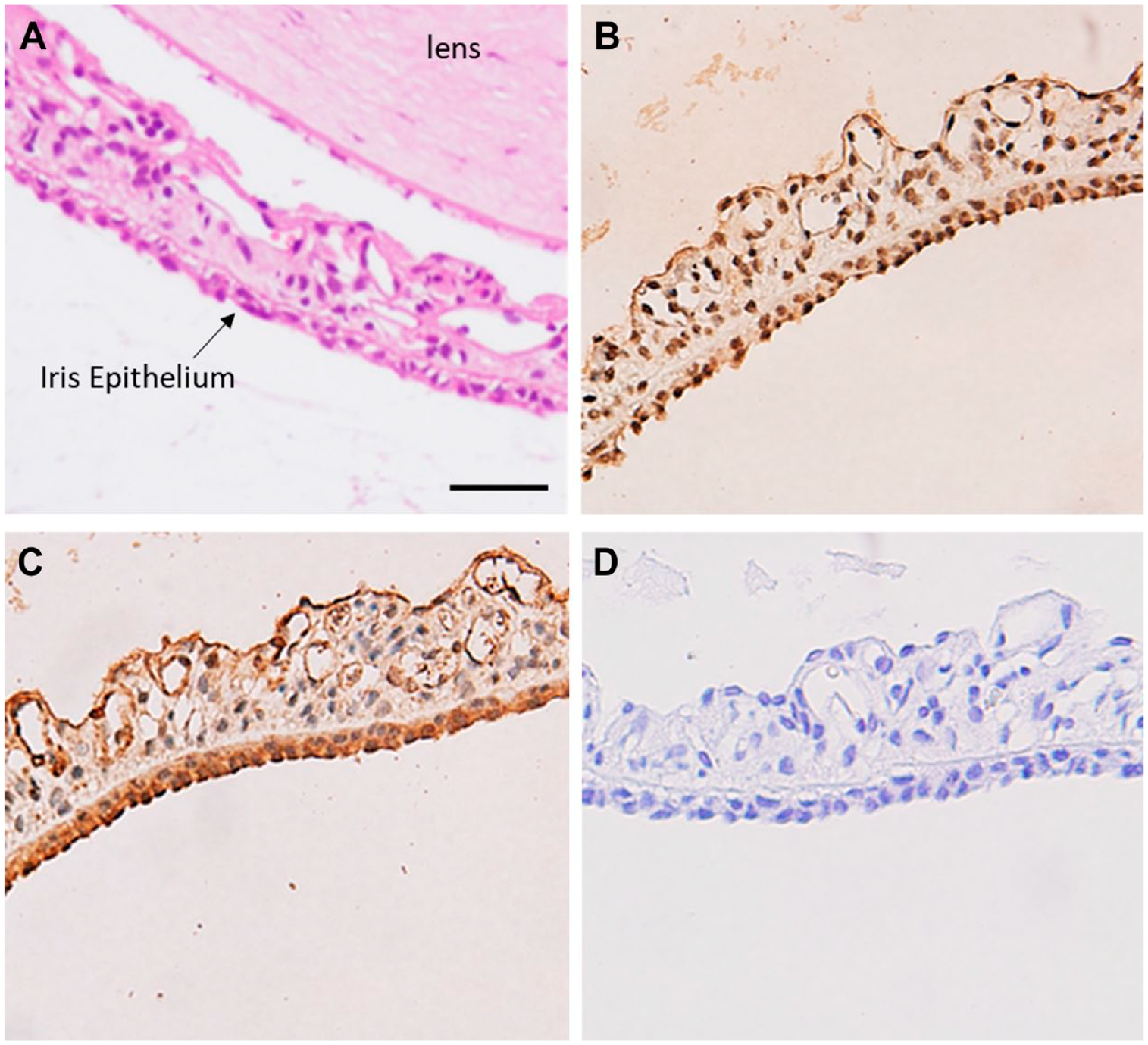
Expression and distribution of cytosolic branched-chain aminotransferase (BCATc) and mitochondrial branched-chain aminotransferase (BCATm) in the section of rat iris. (A) H&E staining of the iris. (B) BCATc staining of the iris layers. (C) BCATm staining of the iris layers. (D) Negative control, without primary antibody. Scale bar 20 µm, 40× magnification.
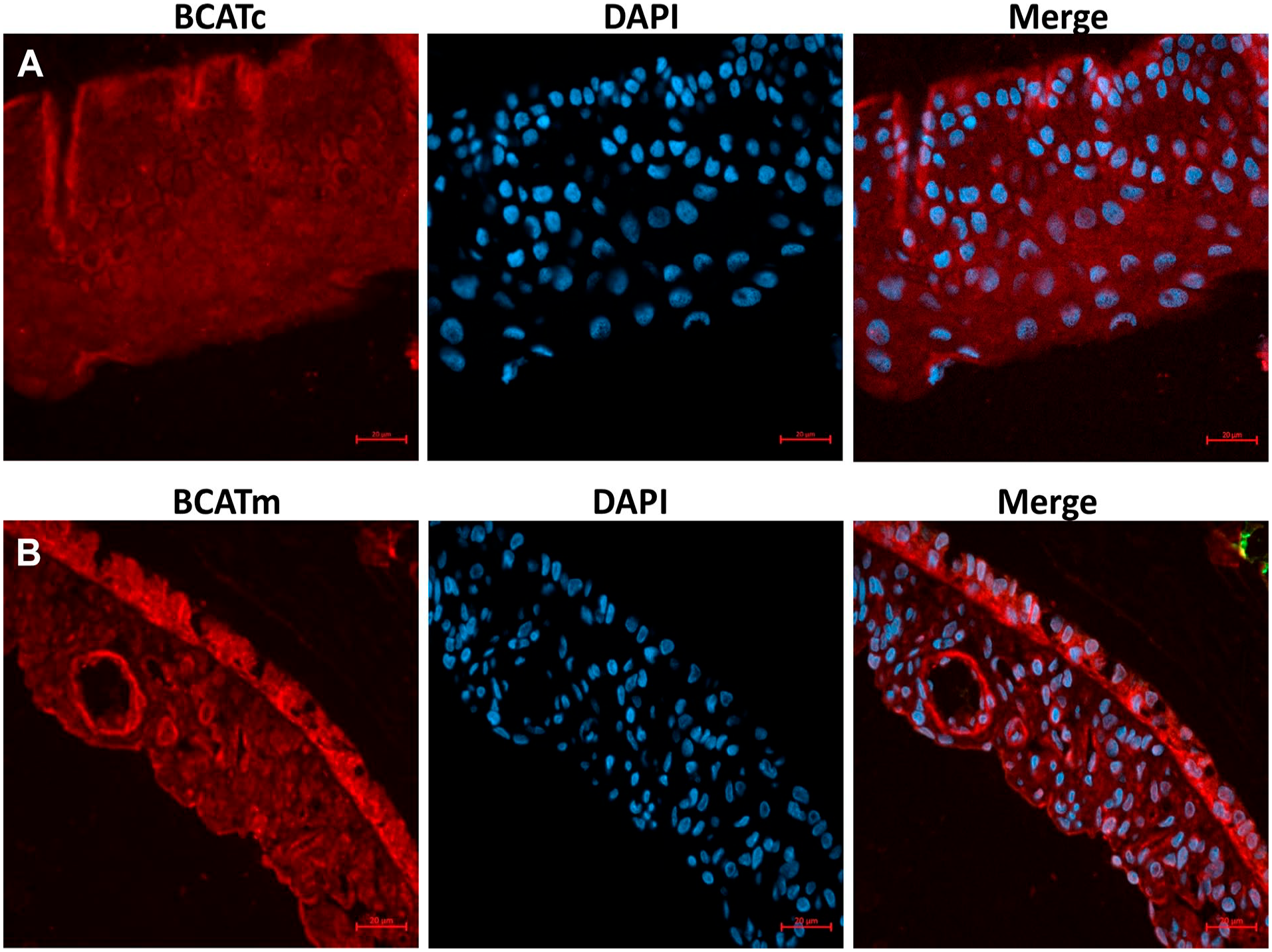
Immunofluorescence staining of branched-chain aminotransferase isoforms of the section of rat iris layers. (A) Immunofluorescence stained for cytosolic branched-chain aminotransferase (BACTc). (B) Immunofluorescence stained for mitochondrial branched-chain aminotransferase (BACTm). DAPI was used as a nuclear stain.
BCAT Expression in the Corneal Layers and Lens
In the cornea, all epithelium layers were expressed BCAT enzymes (Fig. 8). The BCATc staining appeared concentrated in the epithelium’s basal and intermediate cell layers, and a weak stain was observed in the superficial epithelial cells (Fig. 8B). Some staining was also observed within the stroma layer. BCATm was also detected throughout the epithelium cornea cells, and intense labeling was also found in the endothelium cell layers (Fig. 8C). The immunostaining of BCATc was not detected in the whole lens (Fig. 9C and D). Only BCATm labeling was strong in the anterior part of the lens (Fig. 9E, A11, 12) but absent in the posterior pole of the lens (Fig. 9F).
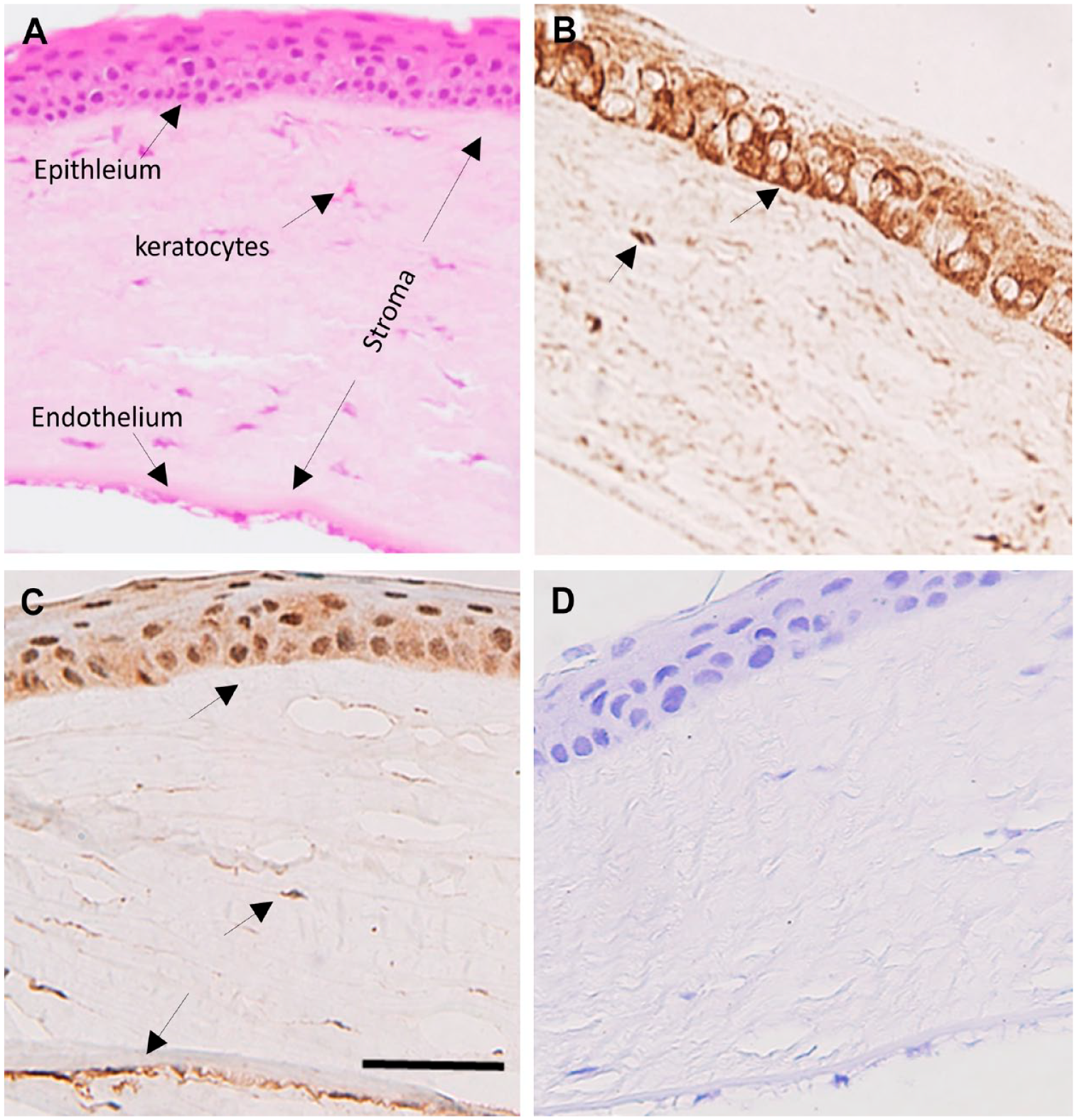
Expression of cytosolic branched-chain aminotransferase (BCATc) and mitochondrial branched-chain aminotransferase (BCATm) in the section of normal rat cornea. (A) H&E staining of the corneal layers. (B) BCATc staining of the corneal layers. (C) BCATm staining of the cornea layers. (D) Negative control. Scale bar 20 µm, 40× magnification.
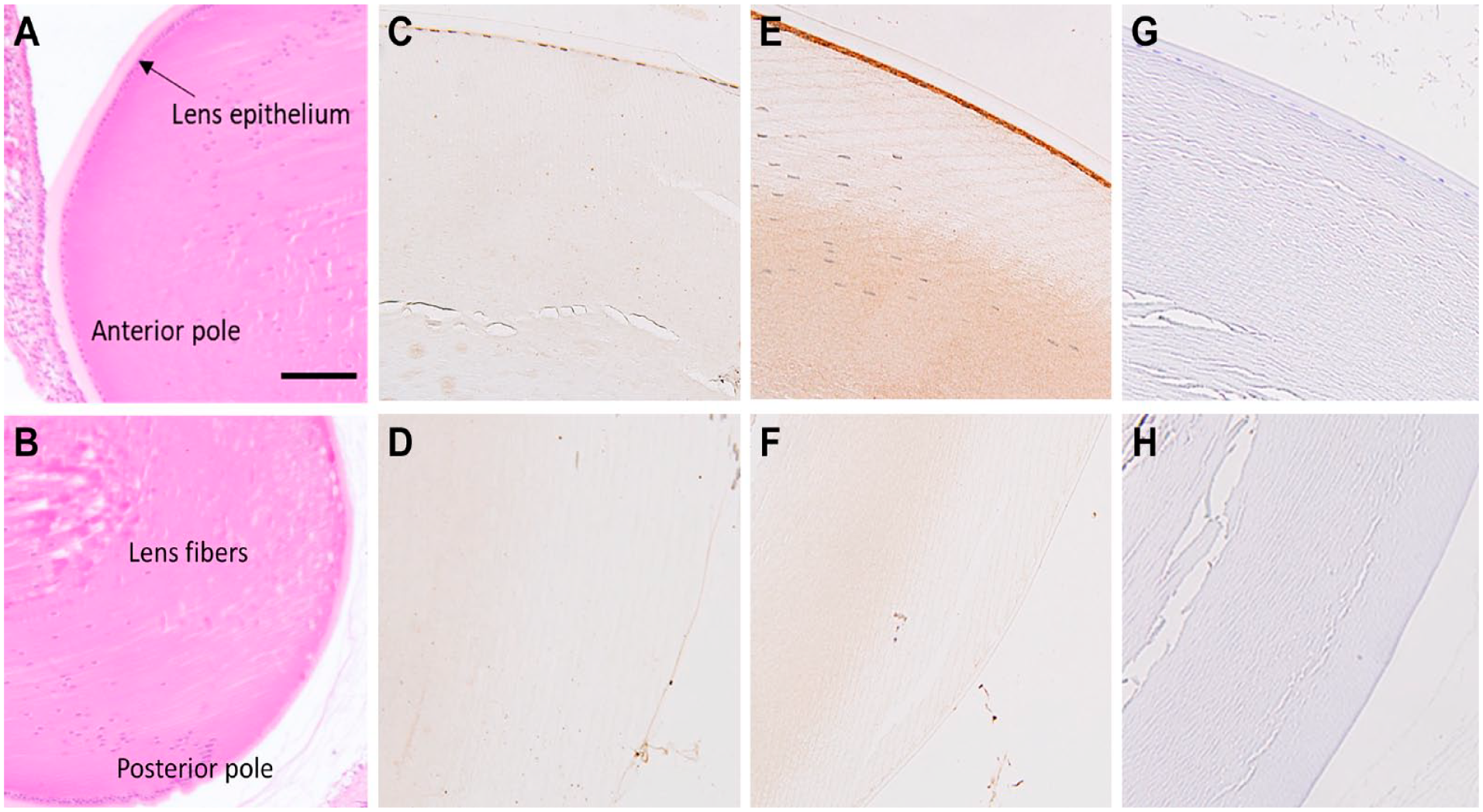
Expression and distribution of cytosolic branched-chain aminotransferase (BCATc) and mitochondrial branched-chain aminotransferase (BCATm) in the sections of rat lens. (A&B) H&E staining showing the anterior and posterior pole of the lens. (C&D) BCATc in anterior and posterior lens. (E) The epithelium layer of the anterior pole of the lens shows the positive staining of BCATm. (F) BCATm in the posterior pole of the lens. (G&H) Negative control. Scale bar 40 µm, 20× magnification.
Discussion
Branched-chain aminotransferase (BCAT) isozymes are essential metabolic enzymes with tissue and cell-specific expression that convert BCAAs to keto acids to maintain cellular energy levels and glutamate pools. The reversible transamination BCAA by BCAT enzymes provides a mechanism for transferring nitrogen between organs, tissues, and even different cell types within a tissue and exchanging metabolites. 3 In this study, we used IHC and immunofluorescence methods to examine the expression and distribution of BCAT isozymes in the neural retina and other intact rat ocular tissues. The histochemical analysis data indicate that BCAT enzymes are expressed in the retina cell layers and some other ocular tissues, suggesting an essential role of BCAT in glutamate synthesis and energy hemostasis in the eye. Besides energy metabolism, glutamate is a major neurotransmitter and a precursor of glutamine, GABA in the retina and brain, and an integral component of antioxidant GSH. 12 Previous studies in rat brains, retina, and cell cultures have demonstrated that BCATs are involved in glutamate metabolism in neuronal cells.10,13 Furthermore, retinal studies using leucine tracing and BCATc inhibitor described that a significant portion of glutamate was produced by BCATc,10,14 implying a relevance of cell-specific distribution and localization of the enzyme in retinal cells and tissues.
The immunolabeling of BCATc in the intact adult rat retina shows that the enzyme is localized in glutamatergic and GABAergic neurons, similar to those previously observed in brain studies.15,16 Moreover, we observed a distinct pattern of BCATc localization in the retinal neuronal cells. An intense immunoreactive BCATc was observed in the cell body of ganglion cells, reflecting the possibility of maintaining glutamate storage rather than excitation. We also found the strong immunoreactivity of BCATc in the outer and inner segments of INL, which reflects the position of the cell body of horizontal and amacrine cells. 17 This further suggests that BCATc localization, like that shown in brain cells, may also be important in regulating the GABA pool in the GABAergic neurons of the retina.7,15 However, further studies are required to confirm these findings using other cell-specific markers. Moreover, the inner plexiform layer (IPL), which contains the synapses of bipolar, amacrine, and ganglion cells, and the outer plexiform layer (OPL), which has the synapses of bipolar, horizontal, and photoreceptors, showed low staining of BCATc, indicating an additional role of BCATc in glutamate production during excitation as observed in brain studies.8,16 All these results support the significant roles of BCATc in glutamate homeostasis, both in glutamatergic and GABAergic neurons in the retina.
To determine the cell-specific localization of BCATc and BCATm within Muller glial cells of the retina, we performed immuno-colocalization studies using a glutamine synthetase antibody, a specific marker of Muller cells. We found little or no expression of BCATc within Müller cells, as confirmed by overlay image (Fig. 2A). In contrast, the BCATm was primarily expressed within Muller glial cells of the retina, as reported previously, 11 besides its moderate expression in RGC and the inner segment of photoreceptor cells. As observed in the brain studies, the cell’s specific expression of BCATc in neuronal cells and BCATm in glial cells are essential in providing nitrogen for the Glu/Gln cycle, the predominant pathway that regulates glutamate levels between neurons and glial cells in the retina and brain (Muller in the retina or astrocyte in the brain).3,12 Thus, the differential expression of BCATc and BCATm in the retina cells parallels the observed expression in the brain. These findings validate the concept of BCAA/BCKA shuttle between neuron and glial cells to recycle and regenerate glutamate in the retina, similar to that observed in the brain.18,19
Most metabolites are filtered through RPE cells, a monolayer between the retina and choroid. The RPE and choroid work together to provide crucial nutritional support to the retina. BCATc is expressed in the single layer of RPE and both BCAT enzymes are expressed in the choroid layers. The expression of BCATc and BCATm in the choroid is correlated well with the expression patterns observed in previous studies by others in peripheral nerves, macrophages, and endothelial cells.8,16,20 Recent studies of energy metabolism have revealed that RPE and retina are metabolically coupled and maintain metabolic homeostasis by bidirectional filtering of metabolites. 21 Photoreceptors shed 10% of their outer segment mass daily and need to synthesize new membranes and proteins daily. For this reason, photoreceptors have high metabolites and energy demands that RPE provides. 22 [ 15 N]-Leucine tracing studies in RPE/choroid showed that leucine is incorporated into many other amino acids, including glutamate, glutamine, and alanine, 14 which the retina may take up. 23 This indicates that the BCAA metabolism supports the retina by producing other amino acids for energy, neurotransmission, and protein synthesis. 14 Besides their role in the anabolic (metabolites synthesis) and energy production, another well-characterized function of BCAT is to regulate and activate the mechanistic target of the rapamycin (mTOR) pathway, which is involved in the control of the protein synthesis, cell growth, and proliferation. 24 Previous studies found that mTOR contributes to the RPE function and facilitates retinal regeneration. 25 Our findings are consistent with these studies, suggesting that BCAA transamination could contribute to the metabolic coupling between the retina and RPE cells. The choroid, RPE, ciliary body, and iris are pigmented tissues and contain pigment melanin. Few studies found that BCAAs inhibit melanogenesis in melanoma cells, which may explain the high expression of BCAT enzymes in these tissues. Langford and coworkers 26 reported that glutamate levels were elevated in both the iris and ciliary bodies, indicating the role of BCAT isozymes in increasing the concentration of Glu in these tissues.
Furthermore, we found low histochemical staining of BCATc in the corneal stroma, which indicates that this protein may be expressed in the main stromal nerve bundle in the cornea, as reported by Murfurt et al. 27 Conversely, the basal epithelial cells’ subnuclear region showed intense BCATc staining, comprising the densest part of the corneal innervation. 28 The positive staining of BCATm was also noted within these layers but was strong within the endothelium cells. Corneal endothelial cells are highly metabolically active, with the highest mitochondrial density to supply the energy necessary for maintaining corneal hydration and transparency. This may explain the increased expression of BCATm in the endothelium layer.29,30 In addition, the cornea is always vulnerable to oxidative stress due to its position in the eye and elevated metabolic rate; therefore, it possesses a high amount of GSH to protect itself from oxidative damage.29,31,32 The synthesis of GSH requires a high amount of glutamate. This increased demand for glutamate for the cornea might be met through BCAT enzymes. Previously, Snytnikova et al. 31 reported that the levels of BCAA metabolites glutamate and GSH were found in high concentration in the cornea along with a lowered ratio of valine, leucine, and isoleucine, which strongly supports our findings of the increased expression of BCAT enzymes in these layers, indicating the vital role of BCAA catabolism in the tissue. To our knowledge, this is the first report of the expression of isozymes in the cornea. However, further studies are required to determine the cell types expressing the BCATs isozymes in the cornea.
The ocular lens is a unique avascular organ with only a single layer of epithelial cells on its anterior surface. 33 The epithelial cells of the anterior surface are the only part of the lens that contains mitochondria, carry out aerobic metabolism, and play an essential role in maintaining the metabolic homeostasis and transparency of the lens. 34 Therefore, it is anticipated that the anterior part of the lens with a monolayer of epithelium is more specific for BCAA metabolism than the posterior. Several independent studies reported leucine and valine metabolism in the lens during the last decades.35–37 This study found that only the mitochondrial BCAT form was expressed in the anterior part of the lens. BCAA metabolism in the lens provides a critical nitrogen source for other amino acid synthesis, such as glutamate, glutamine, and proline. Several studies found that lenses contain high concentrations of GSH, essential in maintaining lens transparency by protecting the thiols of the structural lens crystalline protein from oxidation by reactive oxygen species. 38
Taken together, we have found that BCAT isozymes are expressed in various ocular tissues. BCATc was highly expressed in neurons of the retina, and BCATm, specifically in Muller cells. This suggests that the enzyme may play a vital role in BCAA/BCKA shuttling to maintain glutamate hemostasis in the retina, as suggested in the brain between neurons and astrocytes. 19 The cellular compartmentalization of BCAT isozymes in ocular tissues may contribute to the metabolites shuttling between eye tissues and may be involved in energy production, antioxidant, and glutamate hemostasis. The study provides insight into the metabolism of BCAA within ocular tissues, especially the retina, and their association with glutamate homeostasis and metabolite shuttling. Further studies are required to understand better the role of BCAT in the eye and retina, especially under pathological conditions, as in the case of diabetic retinopathy.
Footnotes
Appendix
Acknowledgements
The authors thank Professor Mohammad Al-Shabrawey from OUWB School of Medicine, Eye Research Institute, Oakland University, for his help in anatomical analyses of eye sections. They thank the Department of Biochemistry and the Graduate Research Program, College of Science, KSU, to support the study. Authors also thank the funding of this research from KCAST-NPST grant (grant number 2-17-03-001-0041), KSU, Riyadh, KSA.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
D.I.A. and M.S.O. conceived and designed the experiments; D.I.A. and Y.A.A. performed the experiments; M.S.O., I.O.A., and A.S.A. contributed reagents/materials tools; D.I.A. wrote the first draft. M.S.O. revised and edited the manuscript. All authors contributed to the article and approved the submitted version.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors thank the KCAST-NPST grant (grant number 2-17-03-001-0041), King Saud University, for funding this work.
