Abstract
During investigating the role of peptidylarginine deiminase (PAD) enzymes in dilated cardiomyopathy (DCM), we observed unique spheroid formation in DCM-myofibroblasts that distinguished them from normal cardiac myofibroblasts. The present study aimed to assess the presence of PADs, the extracellular matrix (ECM), and citrullination in DCM spheroids using immunofluorescence staining and imaging techniques. The results revealed that spheroids derived from DCM-myofibroblasts displayed a more distinctive, tightly packed structure compared with those derived from human cardiac fibroblasts. DCM spheroids showed abundant protein expression of the PAD 2, 3, and 4 enzymes. Notably, increased Ki67 protein expression was associated with increased proliferation in DCM spheroids. Cytoskeletal proteins such as Col-1A, vimentin, α-SMA, and F-actin were highly abundant in DCM spheroids. Furthermore, DCM spheroids contained citrullinated cytoskeletal proteins, mainly citrullinated vimentin and citrullinated fibronectin. These observations supported the occurrence of PAD-mediated citrullination of ECM proteins in DCM spheroids. Collectively, these findings describe the distinctive features of DCM spheroids, representing the cellular characteristics of DCM myofibroblasts. Therefore, DCM spheroids can serve as an in vitro model for further investigations of disease morphology and therapeutic efficacy.
Keywords
Introduction
Spheroid formation generally refers to a cluster of cells that are organized into three-dimensional (3D) spherical morphologies (spheroid morphs) and influenced by various factors, including cellular phenotype and underlying disease state. 1 There have been active efforts to develop 3D models of human cardiac tissue to mimic the intricacies of the human heart. These tissue models not only sidestep ethical concerns often associated with traditional animal models but also bridge gaps that exist between different species. 2
Dilated cardiomyopathy (DCM) is the most common form of heart muscle disease. This makes the muscle walls thin and weaker, and the heart chambers enlarge, causing inefficient contraction. The heart cannot pump as much blood as it normally would. 3 DCM has a poor prognosis, with 1-year mortality rates ranging from 25% to 30% and 5-year mortality rates of approximately 50%. 4 Despite the high prevalence of this disease, numerous aspects of its pathogenesis remain unclear. As a result, there are still no curative or adequate preventive measures available for this disease.
Researchers have used cardiomyocytes, induced pluripotent stem cell (iPSC)-derived cardiac fibroblasts, and coronary artery endothelial cells to develop multicellular 3D cardiac spheroids as alternative models to investigate disease biology. 5 However, iPSC-derived spheroid models do not accurately recapitulate the mature disease phenotype. A significant drawback associated with human iPSC-derived cells is their underdeveloped characteristics. These cells exhibit distinct biological and physiological features that differ from those of mature cardiac cells. 6 Tissue engineering-based approaches emphasize the utilization of cells with inherent capabilities to organize into 3D aggregates without external cues. 7
Disease-derived cell type-specific spheroids closely mimic the in vivo microenvironment and are often used in research to study cell behavior, disease modeling, and drug testing.5,8 Hence, we aimed to assess the ability of DCM-driven human cardiac fibroblasts (HCFs) to generate spheroids and determine the detailed morphological and cytoskeletal characteristics of these cells.
Ex vivo and in vitro cardiac spheroid models generated from stem cell-derived cardiomyocytes and fibroblasts showed extracellular matrix (ECM) deposition. 5 Peptidylarginine deiminase (PAD) family members play vital roles in ECM remodeling in several heart diseases, particularly during fibrosis development.9–12 Identification of citrullinated myofilament proteins in the cardiac proteome indicated that PAD enzymes mediate citrullination as unique posttranslational modifications in heart failure. 9 Given recent evidence that PADs and citrullinated proteins are involved in remodeling and fibrosis during chronic cardiac fibrosis, we further tested whether PADs might mitigate spheroid features in fibroblasts derived from DCM patients. We evaluated the following parameters by immunofluorescence (IF) staining: spheroid shape, proliferation, cytoskeletal marker (Col-1A, α-SMA, or vimentin), and citrullinated ECM (citrullinated fibrinogen and citrullinated vimentin) protein expression.
Materials and Methods
Cell Culture
Primary human cardiac myofibroblasts were derived from DCM patients (Lot. No. 486Z019.1) and healthy participants (HCF; Lot. No. 486Z019.1), purchased from PromoCell GmbH (Heiderberg, Germany). The cells were cultured in complete growth media (Fibroblast Growth Medium 3 containing FBS Supplement Mix [C-23025, PromoCell GmbH, Heiderberg, Germany] and 1% antibiotic [REF: 15240-062, Gibco, USA]) and incubated under standard culture conditions (37C, 5% CO2), after which the cellular growth and morphology were monitored for 1 week.
IF Staining and Microscopy
Actively proliferating cells were seeded on 8-well glass chamber slides (Corning, 354118). Spheroid formation was identified and observed during cell culture. At 72 hr post-spheroid culture, the cells were harvested to assess the expression of the markers via IF staining.
The culture media was aspirated, and the cells were washed twice with ice-cold PBS (1X). Subsequently, cell fixation was performed by adding 200 µL of 4% paraformaldehyde (Cat: J61899.AP, Thermoscientific, USA) for PAD 2, 3, 4, Cit-Fibrinogen, α-SMA, Col-1A, Ki67 protein markers. For Vimentin and Cit-vimentin, the cells were fixed in ice-cold 80% methanol (Fisher Scientific, CAS: 67-56-1) for 15 min at room temperature (RT).
After fixation, the cells were washed with ice-cold PBS (1×) and proceeded for permeabilization (10 mins, RT) using PBS (1X) containing 0.5% Triton X-100 (Cat: X100-100ML, Sigma, USA) for nuclear protein markers (PDA-4 and Ki67), 0.25% Triton X-100 for cytoplasmic markers (PAD 2, 3, α-SMA); PBS (1X), 0.1% Triton X-100 for cytoskeleton membrane associated markers (Vimentin, Cit-vimentin) and 90% methanol for Cit-Fibrinogen, and Col-1A protein markers. The cells were washed in PBS (1×) twice for 5 min and subsequently blocked with 1% bovine serum albumin (Cas: 9048-46-8, Tocris Bioscience, Minneapolis, USA) in PBS/Tween 20 (PBST) for 1 hr at RT. After blocking, the cells were incubated in a mixture of 200 µl/well of solution consisting of PBS (1×) / 1% BSA + 0.1% Tween 20 (Cat: P7949—500ML, Sigma, USA ) containing primary antibodies; PAD 2 (Rabbit, Conc: 1/500, Cat: ab16478, Abcam, USA); PAD 3 (Rabbit, Conc: 1/500, Cat: HPA043739, Sigma-Aldrich, USA); PAD 4 (Mouse, Conc: 1/500, Cat: ab128086, Abcam, USA); Cit-Fibrinogen (Mouse, Conc: 1/250, Cat: 17088, Cayman, USA); α-SMA (Mouse, Conc: 1/500, Cat: 14-9760-82, Invitrogen, USA); Col-1A (Rabbit, Conc: 1/500, Cat: ab260043, Abcam, USA); Ki67 (Rabbit, 1/500, Cat: NBP2-54791, NovusBiologics, Centennial, CO, USA); Vimentin (Rabbit, Conc: 1/500, Cat: ab92547, Abcam, USA); and Cit-vimentin (Mouse, Conc: 1/250, Cat: 22054, Cayman, Ann Arbor, Michigan, USA) separately overnight. After the incubation solution was removed, the cells were washed twice with PBS (1×) and incubated with secondary antibodies (1/1000, Anti-Rabbit Alexa Fluor Plus 594, Cat: A32740; Goat, Anti-Mouse, Alexa Fluor 488, Cat: A11001, Invitrogen, USA) in 1% BSA for 1 hr at RT in the dark. The mixture was removed, and the cells were washed in the dark with PBS (1×) to remove unbound secondary antibodies. Afterward, the cells were counterstained with 200 µl/well of phalloidin (Cat: A12379, Invitrogen, USA) and incubated for 30 min at RT in the dark. The cells were washed in the dark with PBS and processed for nuclear labeling by adding 200 µl/well of Hoechst (0.1 µl/1000 µl) and incubating for 10 min at RT. After completing the staining, the cells were washed with PBS, and the slides were mounted. The glass chambers were removed, and appropriate amounts of ProLong Glass Antifade (REF: P36984, Invitrogen, USA) were added and mounted with coverslips. The coverslips were sealed to prevent drying and movement under a microscope. The slides were subsequently stored in the dark at 4C for image processing.
Image Acquisition
The stained cells were examined using a fluorescence microscope (EVOS M5000 Imaging System, ThermoFisher Scientific, USA) equipped with suitable filters for the fluorophores used in secondary antibodies at suitable magnification (10×, 20×). The image for each channel was acquired individually and used as merged images for the multichannel images. Representative images were captured and saved for analysis. 13
Post-acquisition Image Processing
The original individual channel images and merged images were compiled for figures (non-quantitative analysis). Qualitative aspects such as adjusted brightness and contrasts were applied equally to the final images (Figs. 1–6) to allow increased visibility, by using an inbuilt picture format and correction features in PowerPoint software v2302. 14
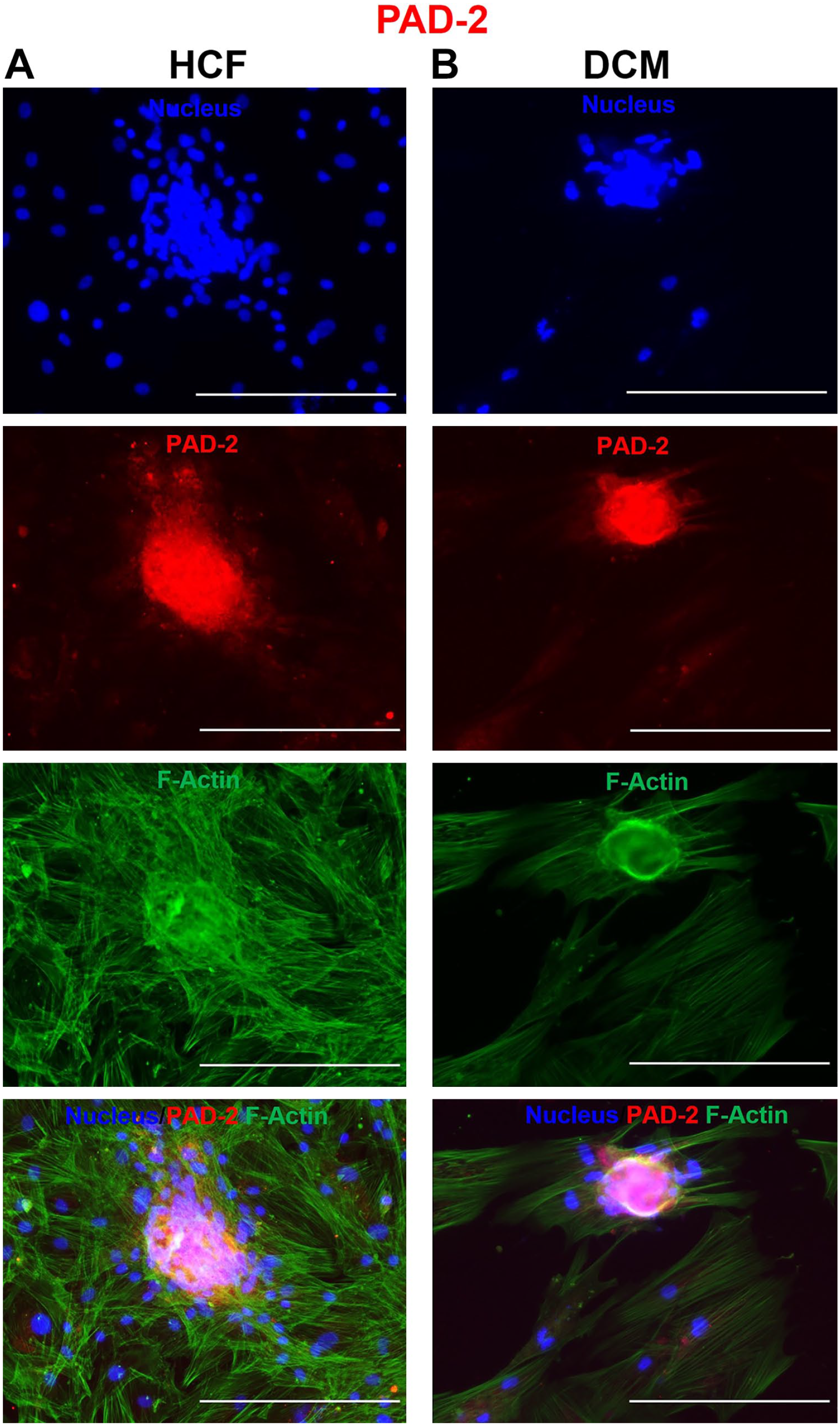
Immunofluorescence microscopy showing the protein expression of the peptidylarginine deiminase 2 (PAD 2) enzyme isoform in (A) human cardiac fibroblasts (HCFs) and (B) dilated cardiomyopathy (DCM)-spheroid morphs. The cells were immunofluorescently labeled with anti-rabbit Alexa Fluor Plus 594 for PAD 2 (red), counterstained with Hoechst (nucleus, blue) and phalloidin (cytoplasmic, F-actin, green), and imaged with a 10× objective. Bars A and B = 300 µm.
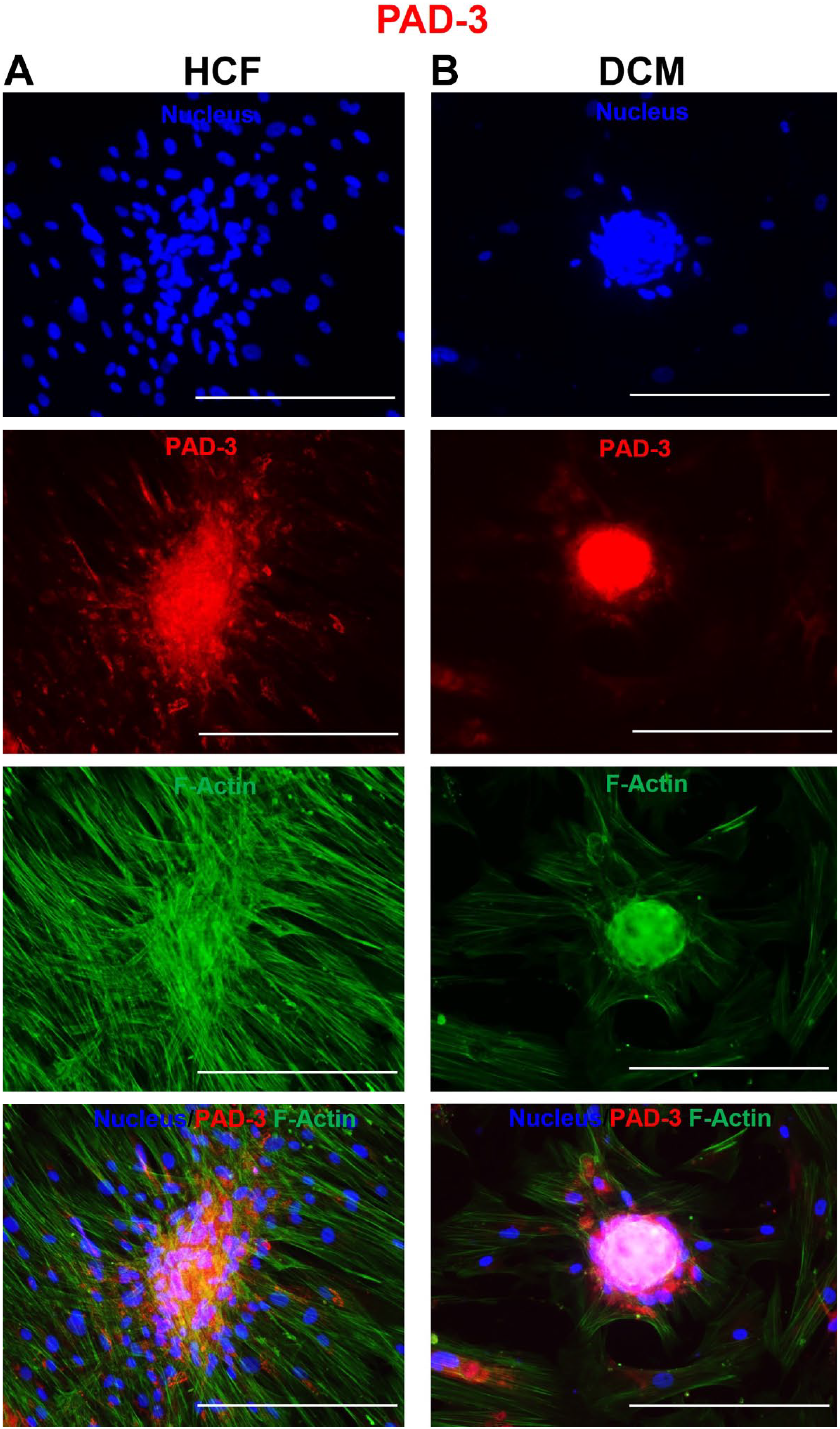
Immunofluorescence microscopy showing the protein expression of the peptidylarginine deiminase 3 (PAD 3) enzyme isoform in (A) human cardiac fibroblasts (HCFs) and (B) dilated cardiomyopathy (DCM)-spheroid morphs. The cells were immunofluorescently labeled with anti-rabbit Alexa Fluor Plus 594 for PAD 3 (red), counterstained with Hoechst (nucleus, blue) and phalloidin (cytoplasmic, F-actin, green), and imaged with a 10× objective. Bars A and B = 300 µm.
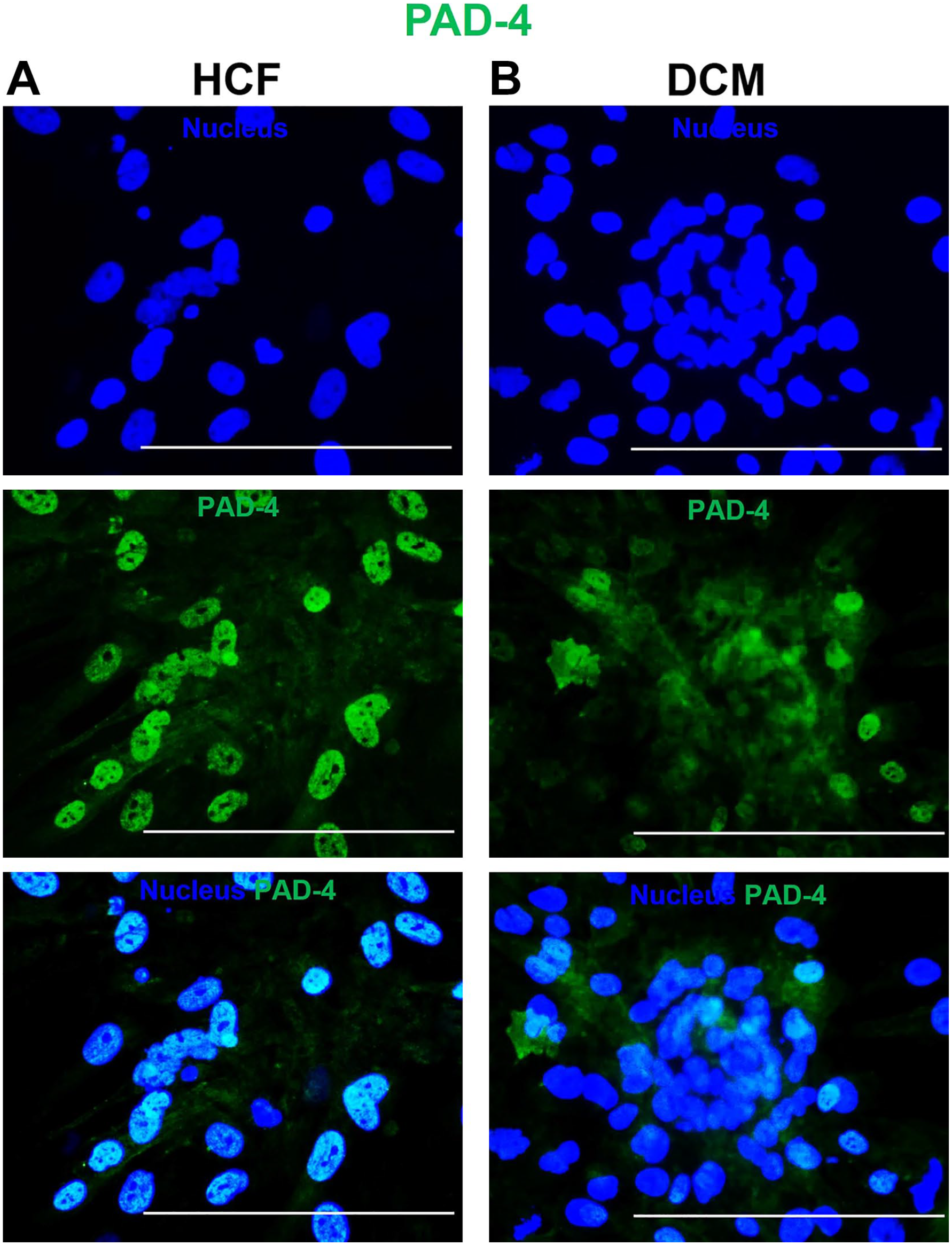
Immunofluorescence microscopy showing nuclear protein expression of the peptidylarginine deiminase 4 (PAD 4) enzyme isoform in (A) human cardiac fibroblasts (HCFs) and (B) dilated cardiomyopathy (DCM)-spheroid morphs. The cells were immunofluorescently labeled with anti-mouse Alexa Fluor 488 for PAD 4 (green), counterstained with Hoechst (nucleus, blue), and imaged with a 20× objective. Bars A and B = 150 µm.
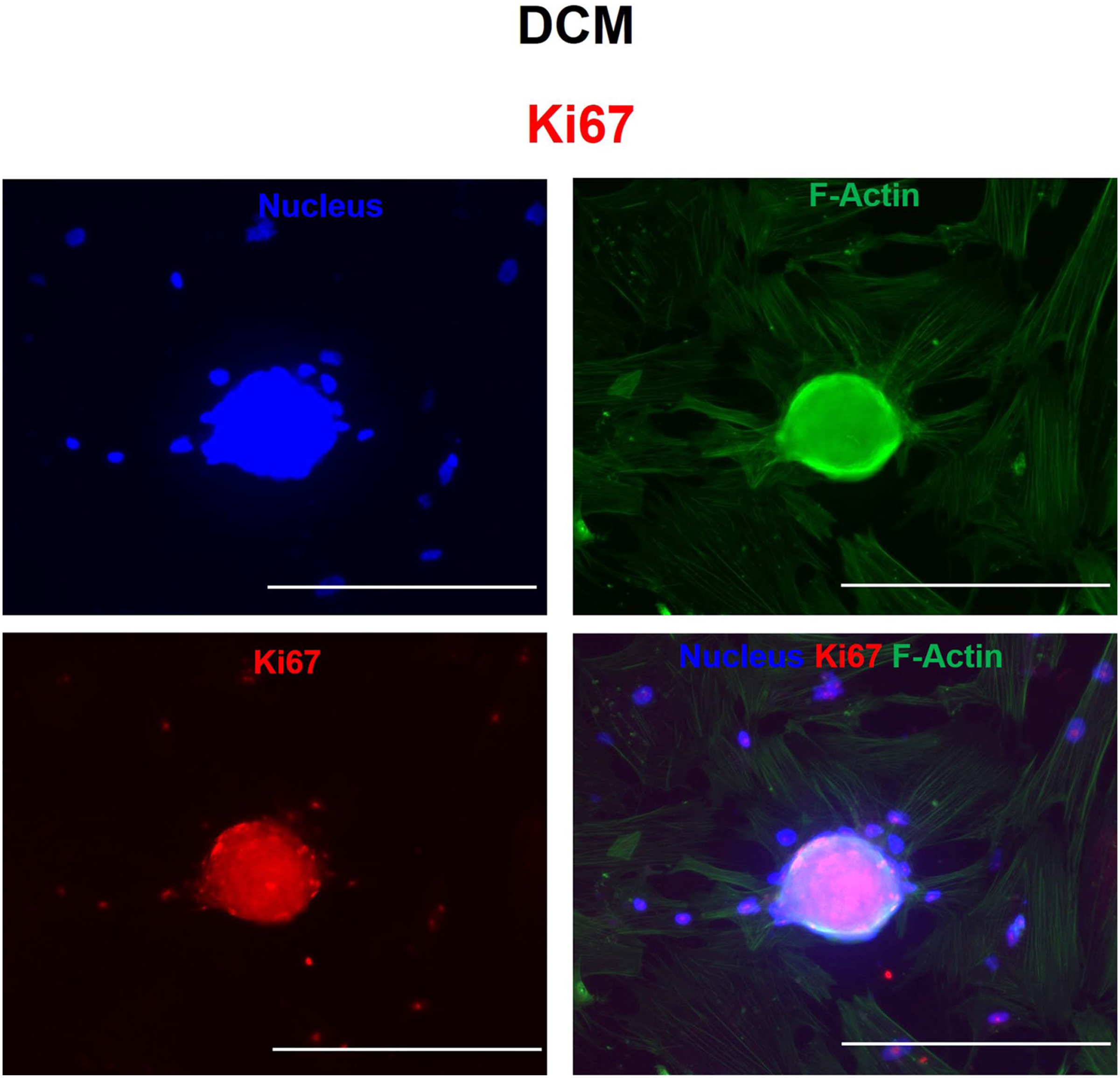
Immunofluorescence microscopy showing nuclear protein expression of the proliferation marker Ki67 in (A) human cardiac fibroblasts and (B) dilated cardiomyopathy (DCM)-spheroid morphs. The cells were immunofluorescently labeled with anti-rabbit Alexa Fluor Plus 594 for Ki67 (red), counterstained with Hoechst (nucleus, blue) and phalloidin (cytoplasmic, F-actin, green), and imaged with a 10× objective. Bars A and B = 300 µm.
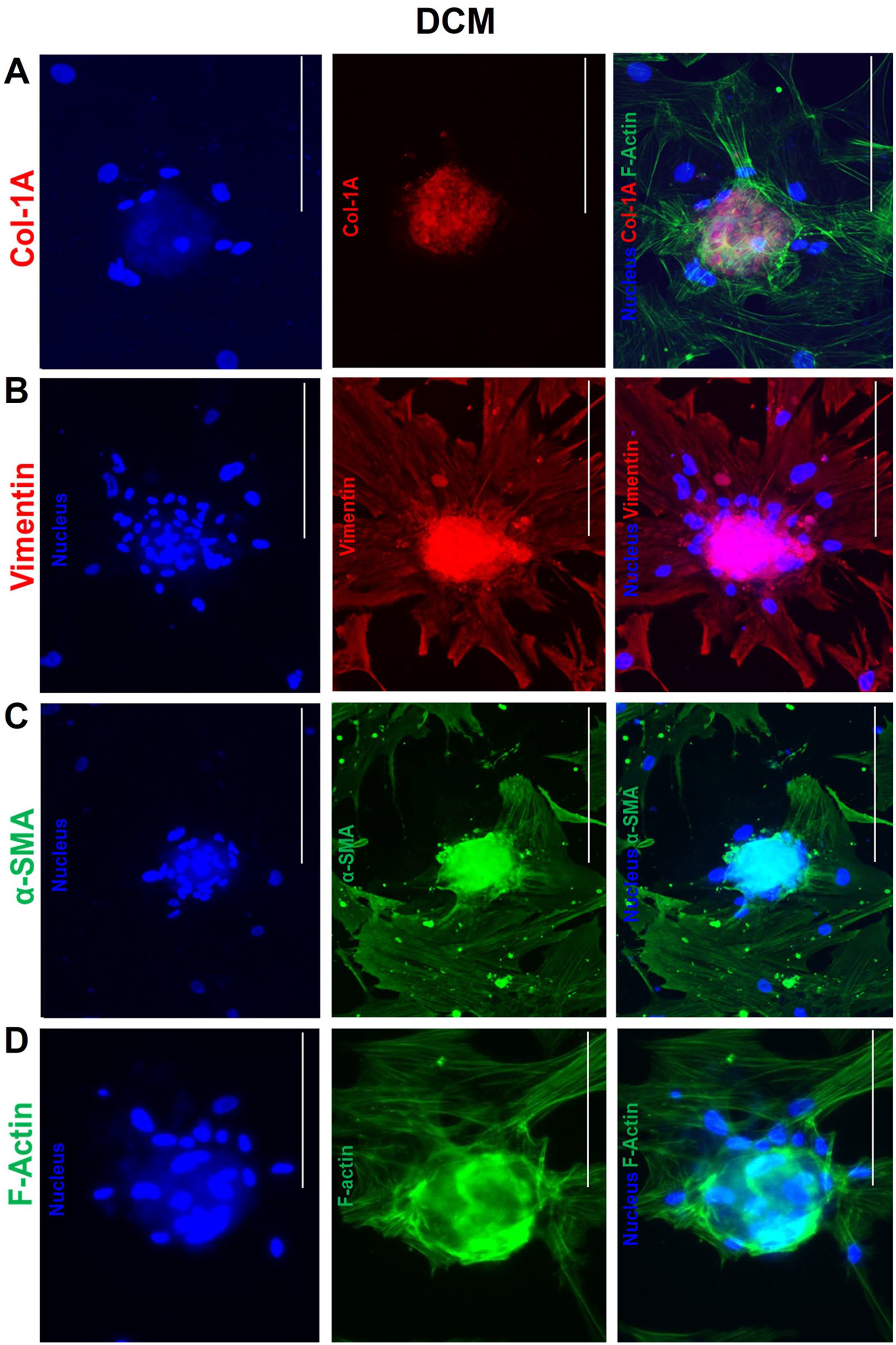
Immunofluorescence microscopy showing cytoskeletal protein expression of (A) Col-1A, (B) vimentin, (C) α-SMA, and (D) F-actin in dilated cardiomyopathy (DCM)-spheroid morphs. The cells were immunofluorescently labeled with anti-rabbit Alexa Fluor Plus 594 and with anti-mouse Alexa Fluor 488 for α-SMA, counterstained with Hoechst (nucleus, blue) and phalloidin (cytoplasmic, F-Actin, green), and imaged with a 10× objective. Bars A–D = 300 µm.
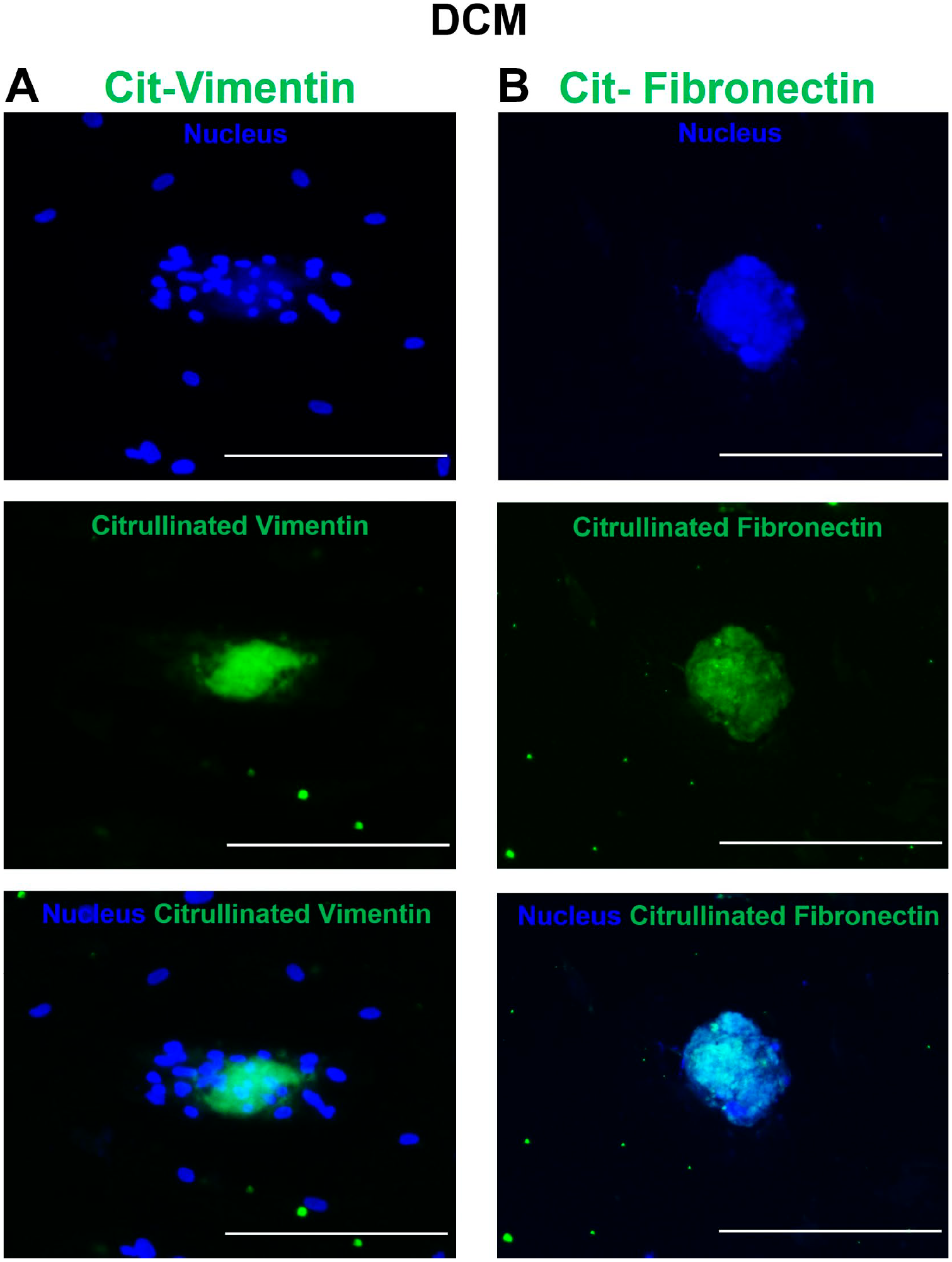
Immunofluorescence microscopy showing cytoskeletal protein expression of (A) citrullinated vimentin and (B) citrullinated fibronectin in dilated cardiomyopathy (DCM)-spheroid morphs. The cells were immunofluorescently labeled with anti-mouse Alexa Fluor 488, counterstained with Hoechst (nucleus, blue), and imaged with a 10× objective. Bars A and B = 300 µm.
Results
DCM-Derived Spheroids Exhibit Different Morphological Characteristics
During routine cell culture, we noticed a unique inherent tendency to form spherical structures in DCM-myofibroblast culture. Multicellular spherical formation was observed after 48 hr of seeding the cells on glass chamber slides without the use of hanging drops or agarose-coated U bottom plates. We performed morphological analysis of 3D cluster formation by IF staining following microscopic evaluation. Images revealed that the HCF group exhibited loose arrangements similar to those of cellular aggregates or clusters, but the DCM group exhibited more defined 3D organization, potentially indicating more compact transformation; these cells were identified as DCM spheroids than normal HCF myofibroblasts (Figs. 1 and 2).
Spheroids Express PAD Enzyme Isoforms 2, 3, and 4
Citrullinated proteins have been reported in 3D spheroids of fibroblasts and cancer cells; however, the expression of PAD enzyme isoforms has not been reported (Pekka et al., 2022). 15 Therefore, we investigated the presence of PADs in DCM spheroids. IF images showed that the spheroids abundantly expressed PAD enzyme isoforms. We detected the PAD isoforms PAD 2 and PAD 3 in the cytoplasm, whereas PAD 4 was expressed in the nucleus in HCF and DCM spheroids (Figs. 1–3).
DCM Spheroids Are Highly Proliferative
To evaluate the cellular state (quiescent or proliferative) in spheroids, after 72 hr, we assessed the expression of the Ki67 marker via IF. The images showed an even distribution of Ki67-related nuclear proteins in the central and peripheral areas of DCM spheroids. Ki67 expression showed that the cells in the spheroids actively proliferated and did not decompose after 72 hr, which indicated fibrosis (Fig. 4).
DCM Spheroids Express Cytoskeletal Proteins
3D spheroid formation in DCM was found to increase the expression of Col-1A, vimentin, α-SMA and F-actin (Fig. 5).
DCM Spheroids Exhibit Citrullinated Vimentin and Citrullinated Fibronectin
The expression of PAD enzyme isoforms (2, 3, and 4) and ECM proteins (acting as substrates for PAD enzymes) in DCM spheroids prompted us to analyze citrullinated ECM proteins. We performed IF staining to detect the presence of citrullinated proteins. Images of DCM spheroids revealed citrullination of cytoskeletal proteins, mainly citrullinated vimentin and citrullinated fibronectin (Fig. 6).
Discussion
Spheroids are typically created in a scaffold-free environment by placing cells in suspension colonies without the aid of ECMs or other physical support. 16 Spheroids offer the opportunity to create more complex 3D models with the power to recreate in vivo more accurately, such as tissue conditions. 17 The mechanical properties of spheroids can differ from those of two-dimensional cell culture conditions. Spheroid formation might expose cells to more physiologically relevant mechanical forces, affecting their behavior. Therefore, spheroids derived from human primary cells offer a more representative model of the stromal microenvironment than established cell lines. 18
Cardiac myofibroblasts can transition from a quiescent state to an actively proliferating state and are known to be involved in tissue repair and fibrosis. 19 Here, for the first time, we aimed to demonstrate that DCM patient-derived ts create spheroids that exhibit high expression and organization of PAD enzymes. Our results revealed strong proliferation in DCM spheroids based on the central and peripheral distributions of Ki67, and these findings are consistent with those of previous studies conducted on skin-derived spheroid models. 18
The formation of spheroids could indicate increased cell‒cell interactions among myofibroblasts. These interactions could be due to altered cell signaling pathways, changes in cell adhesion molecules, or a response to changes in the ECM composition. 20 Cardiac myofibroblasts are responsible for ECM remodeling in the heart. Spheroid formation might reflect changes in the ECM composition and organization. Abnormal ECM remodeling is a hallmark of DCM and can contribute to impaired cardiac function. 10 Our results revealed that DCM-treated spheroids expressed various cytoskeletal proteins, such as Col-1A, vimentin, α-SMA, and F-actin. As in our study, a previous study developed human adult multicellular cardiac spheroids, and iPSC-derived cardiac spheroid models also showed positive expression of vimentin. 5 In another study, spheroids generated from skin also expressed vimentin intermediate filaments and stress fibers (α-SMA), which are cytoskeletal markers of the phenotypic transition (proto-myofibroblasts) of myofibroblasts. 18
Our results showed the expression of citrullinated vimentin and citrullinated fibronectin in DCM-spheroid morphs. PAD enzyme-mediated citrullination has been identified as a unique posttranslational modification in the cardiac proteome, particularly for citrullinated myofilament proteins found in heart failure. 9 This posttranslational modification plays a role in various biological processes, including proliferation and ECM remodeling during fibrosis. 11
Although preliminary, our work on DCM spheroids revealed that PAD expression and the proliferative features of spheroids could contribute to the cellular ECM microenvironment associated with DCM fibrosis. These findings can be valuable in various research areas, particularly for evaluating gene expression profiles, the ECM composition, cell signaling pathways, and functional assays to evaluate the impact of spheroid formation on cardiac tissue remodeling and function, as well as for developing drug testing models.
In conclusion, the unique inherent characteristics of DCM-spheroid morphs, coupled with the expression of specific PAD enzymes (isoforms 2, 3, and 4), proliferative features (Ki67), cytoskeletal proteins, and citrullination, provide a comprehensive picture of the cellular changes occurring in DCM. This model is representative of further investigations of disease morphology and therapeutic efficacy.
Footnotes
Acknowledgements
The authors thank Dr. Emily Barruet from the Children Health Institute, Cedars-Sinai Medical Center, for sharing the immunofluorescence image facility.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: conceptualization (AS), data curation and formal analysis (AS), funding acquisition (JFB), investigation and methodology (AS), project administration and supervision (JFB), writing—original draft (AS), writing—review and editing (AS, JFB).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Cedars Sinai Institution.
