Abstract
Cisplatin (cPt) is a commonly used treatment for solid tumors. The main target of its cytotoxicity is the DNA molecule, which makes the DNA damage response (DDR) crucial for cPt-based chemotherapy. Therefore, it is essential to identify biomarkers that can accurately predict the individual clinical response and prognosis. Our goal was to assess the usefulness of alkaline comet assay and immunocytochemical staining of phosphorylated Hsp90α (p-Hsp90α), γH2AX, and 53BP1 as predictive/prognostic markers. Pre-chemotherapy peripheral blood leukocytes were exposed to cPt in vitro and collected at 0, 24 (T24), and 48 (T48) hr post-drug removal. Healthy subjects were also included. Baseline DNA damage was elevated in cancer patients (variability between individuals was observed). After cPt, patients showed increased γH2AX foci/nucleus (T24 and T48). Both in healthy persons and patients, the nuclear p-Hsp90α and N/C (nuclear/cytoplasmic) ratio augmented (T24), decreasing at T48. Favorable clinical response was associated with high DNA damage and p-Hsp90α N/C ratio following cPt. For the first time, p-Hsp90α significance as a predictive marker is highlighted. Post-cPt-DNA damage was associated with longer disease-free survival and overall survival. Our findings indicate that comet assay and p-Hsp90α (a marker of DDR) would be promising prognostic/predictive tools in cP-treated cancer patients.
Keywords
Introduction
The search for effective antineoplastic drugs in cancer treatment led to the study and discovery of alkylating agents, chemical compounds whose molecular target is the DNA and ultimately conduce to the expected tumor cell death. Cisplatin [(cis-diamminedichloroplatinum(II)] (cPt), the first anticancer agent belonging to this group, was approved by the Food and Drug Administration in 1978. Until today, this drug is commonly used in cancer chemotherapy because it became the backbone of different antitumor regimens for several types of solid tumors, including lung, testicular, bladder, ovarian, and breast cancers, lymphomas, and melanomas. 1
cPt cytotoxic effects are caused by its interaction with DNA. In the nucleus, cPt forms cross-links at N7 reactive center on residues of purine, mainly intrastrand-crosslinks, which represent approximately 90% of the cPt-induced DNA adducts. These adducts can distort the DNA structure, DNA replication, and transcription processes, activating apoptotic cell death signaling pathways, if the DNA repair mechanisms fail to correct the DNA damage. 2 Unfortunately, some cancer patients who initially respond to cPt-based chemotherapy can relapse and the development of drug resistance occurs. Several mechanisms have been proposed to be involved with cPt resistance, among them are the reduction in the intracellular accumulation of the drug or alterations in the DNA repair mechanisms. 3 Numerous DNA-damaging agents produce a range of DNA lesions, including base lesions and strand breaks, which promote alterations to the DNA double helix. 4 Once DNA damage occurs, various cellular responses are triggered to allow the repair of the DNA damage through a variety of mechanisms. DNA repair systems constitute one of the hallmarks of cancer that are involved in genomic integrity maintenance. 5 The main DNA repair mechanisms implicated in cPt toxicity are nucleotide excision repair (NER) and mismatch repair (MMR). The resistance to cPt has been related to increased NER system activity or deficient MMR function in tumor cells. 6
One of the challenges in cancer research has been to identify molecular markers able to predict individual clinical response and outcome. Therefore, the evaluation of DNA damage would be important for predicting treatment response and prognosis of patients, highlighting its role as a biomarker in cancer. Diverse methods can be used to evaluate DNA damage and repair. 7 For many years, the single-cell gel electrophoresis or comet assay has been used to measure DNA damage in individual cells. Over the years, the comet assay gained popularity as a highly sensitive, simple, and low-cost method getting consistent and reliable results. 8 The comet assay has been recognized as a useful method for biomonitoring DNA damage induced by a wide range of DNA-damaging agents in white blood cells. 9 Several studies have shown comet assay’s utility in assessing cancer patients’ progression and treatment. 10 Some years ago, our research group reported a pilot study indicating the alkaline comet assay’s usefulness in predicting the response to doxorubicin or cPt -based chemotherapy in peripheral blood lymphocytes from cancer patients. 11 The potential of alkaline comet assay as a predictive or prognostic marker has not yet been completely explored.
Double-strand breaks (DSBs) constitute one of the most lethal DNA lesions because they induce genome instability and even malignant transformations. The study of γH2AX (histone H2A variant H2AX phosphorylated at Ser139) helps to detect and quantify DNA DSBs. 12 Briefly, after DSBs arise, H2AX is rapidly phosphorylated by DNA damage-activated kinases (ATM, ATR, and DNA-PK), to locate in chromatin foci at the sites of DNA damage. The γH2AX molecules also act as a scaffold for other DNA repair components such as 53BP1 protein (p53-binding protein 1), which recruits other responsive proteins to the damage sites. Both γH2AX and 53BP1 have been proposed as molecular biomarkers of response and prognosis in cancer. 13
Heat shock proteins (HSP) are known to be induced by heat shock and other chemical and physical stresses in a wide range of species. They maintain the integrity of the proteome and promote cell survival, tumorigenesis, invasion, and metastasis. Then, they were characterized as molecular chaperones, which have the property of interacting with other proteins and maintaining proteostasis. HSPs have been correlated with tumorigenesis and implicated in the apoptosis process regulation. 14 Heat shock protein 90 (Hsp90) is a highly conserved molecular chaperone implicated in the stability and function of more than 200 proteins, including protein kinases, chromatin remodeling factors, transcription factors, and others. The Hsp90 family includes the following members: Hsp90α (Hsp90AA1), Hsp90β (Hsp90AB1), glucose-regulated protein 94 (GRP94) (Hsp90B1), and tumor necrosis factor receptor–associated protein 1 (TRAP1). Hsp90α and Hsp90β present high homology, nevertheless, Hsp90α, unlike Hsp90β, contains two threonine residues (Thr5 and Thr7). Both proteins are predominantly cytoplasmic, but a small fraction can also be found in the nucleus. 15 It has been shown that during DNA damage response (DDR), the site on threonine 7 (Thr7) of Hsp90α can be phosphorylated by DNA-PK and the phosphorylated form of Hsp90α (p-Hsp90α) accumulates at the site of the DNA damage, that is important for the maintenance of γH2AX foci. For this reason, p-Hsp90α (Thr7) has been proposed as a surrogate biomarker for genetic instability. 16
In the present study, we investigated whether DNA damage evaluation using comet assay, p-Hsp90α, and if other common DNA damage markers detected by immunostaining such as γH2AX and 53BP1 can predict clinical response and follow-up. We used easily accessible cells from cancer patients such as peripheral blood leukocytes (PBL) in vitro cultured and treated with cPt. The Thr5/7 phosphorylation of the protein Hsp90α in PBL and its utility as a biomarker to predict response to cPt-based chemotherapy and patients’ outcome has not yet been studied. The hypothesis is supported by our previous reports showing that comet assay can inform about the DNA repair status and response to chemotherapy in PBL from cancer patients. On the other hand, p-Hsp90α evaluation can provide information for patient management and treatment.11,17 The associations of the studied markers with clinical res-ponse, disease-free survival (DFS), and overall survival (OS) were also determined.
Materials and Methods
Patients
Fifty-three cancer patients from the Regional Integrati-on Cancer Center (COIR) in Mendoza, Argentina were enrolled in the study. According to the Declaration of Helsinki, all subjects gave written informed consent in agreement with an approved protocol by the COIR’s Ethics Committee in Research and Health (CEIS). Pa-tients’ eligibility criteria included histologic and/or cytologic diagnosis of cancer suitable for platinum-based chemotherapy as their first-line treatment, no prior chemotherapy or radiotherapy, and normal hepatic and kidney function. After diagnosis or surgery, the patients received one to six cycles of treatment with cPt-based chemotherapy. Table 1 shows the main characteristics of the 53 patients. Ten healthy non-smoker persons, five men and five women, aged from 24 to 54 years (mean age = 32 years), and with no family history of cancer were also included in the study as control subjects.
Main Characteristics of the Patients.

Abbreviation: NE, not evaluable.
Cell Culture and Experimental Design
Ten milliliters of peripheral blood obtained by venipuncture from cancer patients and control subjects was collected with heparin. White blood cells (leukocytes) were collected as described previously.
11
Cells were plated in T25 flasks containing RPMI 1640 supplemented with 5 µg/ml of phytohemagglutinin, 2 mM
Immunocytochemistry
Cell suspensions were fixed in 4% formaldehyde (Merck, Darmstadt, Germany) in 1× phosphate buffer saline (PBS, pH 7.4) for 20 min, then washed with 1× PBS and seeded onto 3-aminopropyltriethoxysilane (Sigma)-coated slides. For cell permeabilization, the slides were immersed in 0.1% TritonX-100 in 1× PBS for 5 min. After washing, the slides were incubated in 3% hydrogen peroxide (Merck Darmstadt, Germany) in 1× PBS for 30 min. Antigen retrieval was performed for 30 min in boiling 0.01 M citrate buffer pH 6. The slides were blocked in 10% non-fat dry milk for 1 hr at room temperature followed by overnight incubation at 4°C in a humidity chamber with specific primary antibodies at the following dilutions: rabbit monoclonal antibody (mAb) against γH2AX Ser 139 (1:450; #9718 Cell Signaling Technology, Danvers, MA), rabbit polyclonal Ab (pAb) against 53BP1 (1:350, #4937 Cell Signaling), and rabbit pAb against p-Hsp90α Thr 5/7 (1:200, #3488 Cell Signaling). The slides were wash-ed and then incubated with biotinylated anti-rabbit and anti-mouse IgG secondary antibodies (whole molecule, BA-1400; Vector Laboratories, Burlingame, CA) followed by avidin-biotin HRP system (PK-6100; Vector) according to manufacturer’s instructions. Dia-minobenzidine-peroxidase kit (ImmPACT DAB; Vector) was used as substrate chromogen. Slides were lightly counterstained with 0.5% methyl green in sodium acetate (0.1 M, pH 4.6) and dehydrated. The immunostaining was evaluated according to the number of foci per nucleus and the percentage of positive cells counting 200 cells per sample under double blindness with a Nikon Eclipse E200 microscope (Japan). The p-Hsp90α antibody specificity was determined by Western blot (Fig. A1). Nonspecific rabbit IgG antibody (Cell Signaling) was used as isotype-negative control. Tumor tissues known to express γH2AX, 53BP1, and p-Hsp90α Thr 5/7 were included as positive controls as described previously. 17
Alkaline Comet Assay
The alkaline version of the comet assay was performed at pH >12 to detect in the DNA molecule, the presence of single-strand breaks (SSBs), DSBs, and alkaline-labile sites. 18 Briefly, frozen PBL were thawed at 37°C, washed in 1× PBS, and resuspended in cold 1× PBS to a final concentration of 1 × 106 cells/ml. All the steps were carried out in the dark and at 4°C to prevent additional DNA damage. Alkaline comet assay was performed as published elsewhere. 11 Treatment with hy-drogen peroxide (H2O2), which originates DNA breaks, was used to evaluate Pt-induced DNA crosslinks. For this step, cPt-treated cells were incubated with 6 × 10−1 μg/ml H2O2 for 1 hr. As a positive control, a group of untreated cells was exposed to 6 × 10−1 μg/ml H2O2 for 1 hr. After electrophoresis, the agarose gels were placed on microscope slides and silver stained as previously reported. 19 Comets were evaluated under the ×40 objective of a Nikon Eclipse E200 microscope in 40 randomly selected cells per slide (i.e., 80 cells/each sample). A visual score based on the extent of migration was used: 0, very low migration; 1, 5–10% of migrated DNA; 2, 11–30% of migrated DNA; 3, 31–60% of migrated DNA; 4, 61–95% of migrated DNA; and 5, >95% of migrated DNA.19,20 To facilitate the management of the data, an average of DNA migration was calculated as: [(% of cells with score 1) ×1+ (% of cells with score 2) ×2+ (% of cells with score 3) ×3+ (% of cells with score 4) ×4+ (% of cells with score 5) ×5]/100. 21 The assay was done in duplicate.
Clinical Response
Response to chemotherapy was assessed according to the Response Evaluation Criteria in Solid Tumors (RECIST 1.1) which are defined as complete response (CR; the disappearance of all target lesions), partial response (PR; at least a 30% decrease in the sum of diameters of the lesions), progressive disease (PD; at least a 20% increase in the sum of diameters of the lesions), or stable disease (SD; neither sufficient shrinkage to qualify for PR nor sufficient increase to qualify for PD).17,22
Follow-Up
After chemotherapy, cancer patients were regularly assessed by clinical evaluation and image studies. The mean follow-up of the patients (from March 2016 to March 2022) was of 37.57 months (range, 1–72 months). DFS was calculated as the time to progression after first-line chemotherapy and OS as the period from diagnosis until the date of death.
Statistical Analysis
The Kolmogorov–Smirnov and Shapiro–Wilk tests were used to assess the normality of the data distribution. Comparisons between cancer patients and healthy in-dividuals were done using the Mann–Whitney test. Comparisons between experimental conditions in pa-tients and healthy individuals were performed using the Kruskal–Wallis test. Associations between γH2AX, 53BP1, nuclear p-Hsp90α, cytoplasmic p-Hsp90α expr-ession, DNA migration mean, percentage of cells with score 5, and clinical response were examined using the χ2 test or Fisher’s exact test according to the number of frequencies for 2 × 2 contingency tables. Fisher’s exact test was performed using the healthy subjects’ mean value as the cut-off. Correlations were assessed by Spearman’s ρ coefficient. Survival analyses were performed by the Kaplan–Meier method. The difference between curves was evaluated with the log-rank test. For all calculations, two-sided p<0.05 was considered to indicate a statistically significant difference. Statistical analysis was performed using SPSS software version 25.0.
Results
Basal DNA Damage and Immunocytochemical Expression of γH2AX, 53BP1, and p-Hsp90α in PBL From Healthy Subjects and Cancer Patients
DNA damage was determined as DNA migration mean by alkaline comet assay. Despite higher basal DNA damage observed in PBL from cancer patients (approximately 27.7% higher than healthy controls), no statistically significant differences were found between healthy persons and cancer patients (Fig. 1A). Although patients’ PBL displayed higher total DNA damage (score 5) than healthy persons (approximately 62.4% higher in patients), the difference was not statistically significant (Fig. 1B). γH2AX (foci/nuclei or % of positive cells) levels were found to be very similar between healthy individuals and cancer patients (Fig. 1C and D). Nuclear and cytoplasmic p-Hsp90α expression or N/C ratio of the protein were also comparable among healthy subjects and patients at basal conditions (Fig. 1E to G). However, the number of 53BP1 foci/nuclei, although not statistically significant, was 46% higher in cancer patients than in healthy persons (p=0.0662), Fig. 1H. Interestingly, high interindividual variability between patients was particularly noted.
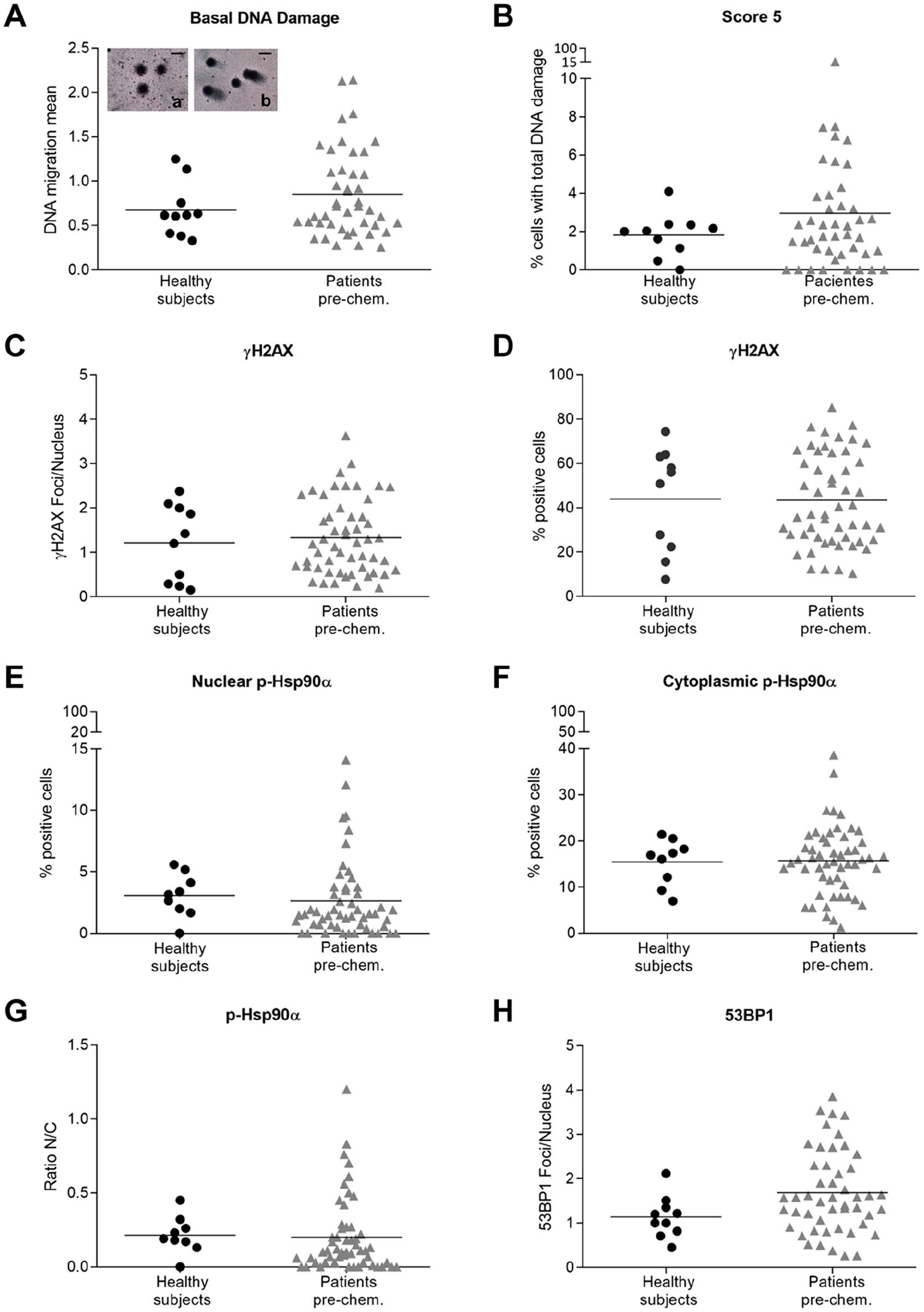
DNA damage and γH2AX, 53BP1, and p-Hsp90α expressions in PBL from healthy subjects and cancer patients at basal conditions. (A) Scatterplot for DNA migration mean differences between healthy subjects and cancer patients. Representative comet images were embedded into the graph: (a) from a healthy volunteer sample and (b) from a cancer patient. (B) Percentage of cells with total DNA damage by comet assay (score 5) in healthy subjects and patients. γH2AX immunoexpression evaluated as foci/nucleus (C) and percentage of positive PBL (D) in healthy controls and patients. Percentage of nuclear positive (E) and cytoplasmic (D) immunostained PBL for p-Hsp90α. (F) Scatterplot showing the nuclear/cytoplasmic ratio (N/C) for p-Hsp90α. (G) The number of 53BP1 foci/nucleus. Each point in the scatter graphs represents the value for each case-control or patient. The center lines represent the mean value. Comparisons were done with the Mann–Whitney test. Photographs were captured using Nikon Eclipse 80i microscope. A scale bar was included in each microphotograph (30 µm).
DNA Migration Mean and Total DNA Damage in PBL From Healthy Individuals and Patients Treated With Cisplatin. Comparisons Between Experimental Groups Across Recovery Times
PBL from healthy persons and pre-chemotherapy PBL from patients were exposed to cPt followed by 24 or 48 hr of recovery. Because cPt cytotoxicity involves the formation of DNA adducts that compact the genome, we used hydrogen peroxide (H2O2) as a second agent to induce DNA breaks. 9 No statistically significant differences were found between experimental conditions and the positive control for DNA migration mean in healthy individuals (Fig. 2A). In contrast, statistically significant differences were observed in PBL from patients at 0, 24, and 48 hr post-cPt treatment (p<0.01), Fig. 2B. The percentage of cells with total DNA damage (score 5) 48 hr post-cPt treatment was significantly higher in healthy individuals than in the positive control (Fig. 2C). In addition, the percentage of score 5 cells was higher at T48 than at T0 (Fig. 2C). However, in cancer patients, the percentage of score 5 cells was significantly decreased in all experimental conditions concerning the positive control (Fig. 2D). No statistically significant differences were found between healthy volunteers and cancer patients for DNA migration mean and levels of total DNA damage in the different experimental groups (data not shown).
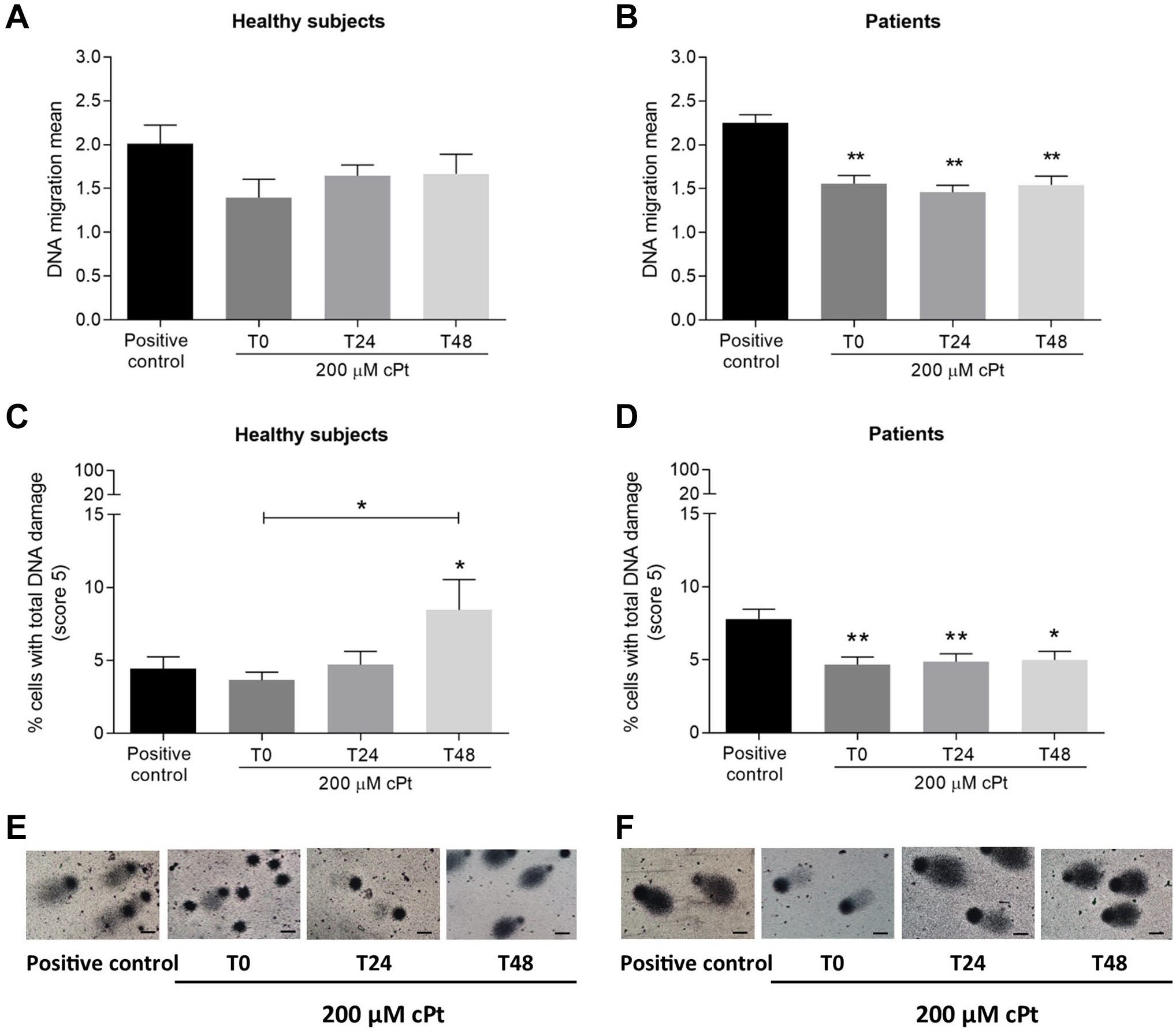
Comet DNA damage measurements in healthy subjects and patients PBL within 0 to 48 hr after cPt treatment. Healthy subjects (A) and cancer patients (B) DNA migration mean after experimental treatments. Percentage of PBL with total DNA damage (score 5) in healthy persons (C) and patients (D) post-cPt exposure. Comet microphotographs of representative cases for healthy persons (E) and patients (F) at experimental conditions. Bars represent the mean±SEM. Positive control: PBL exposed to 60 µM H2O2 for 1 hr. T0: PBL collected immediately after 200 µM cPt treatment. T24 and T48: PBL collected 24 or 48 hr post-cPt, respectively. Comparisons were done with Kruskal–Wallis with post-Dunn’s test. *p<0.05. **p<0.01. Photographs were captured using Nikon Eclipse 80i microscope. A scale bar was included in each microphotograph (20 µm).
Immunocytochemical Expression of γH2AX, 53BP1, and p-Hsp90α in PBL From Healthy Subjects and Cancer Patients In Vitro Exposed to cPt Over Recovery Time
The induction and persistence of the DNA damage biomarkers, γH2AX and 53BP1 (foci/nuclei), were also studied. In PBL from healthy persons, γH2AX foci increased 24 hr after cPt (p<0.05), this behavior was also observed in 53BP1 levels, although the differences were not statistically significant (Fig. 3A and C). Surprisingly, patients showed a significant increase of γH2AX foci at 24 hr (p<0.001) that persisted 48 hr post-cPt (p<0.05), but this was not observed in 53BP1 foci levels (Fig. 3B and D).
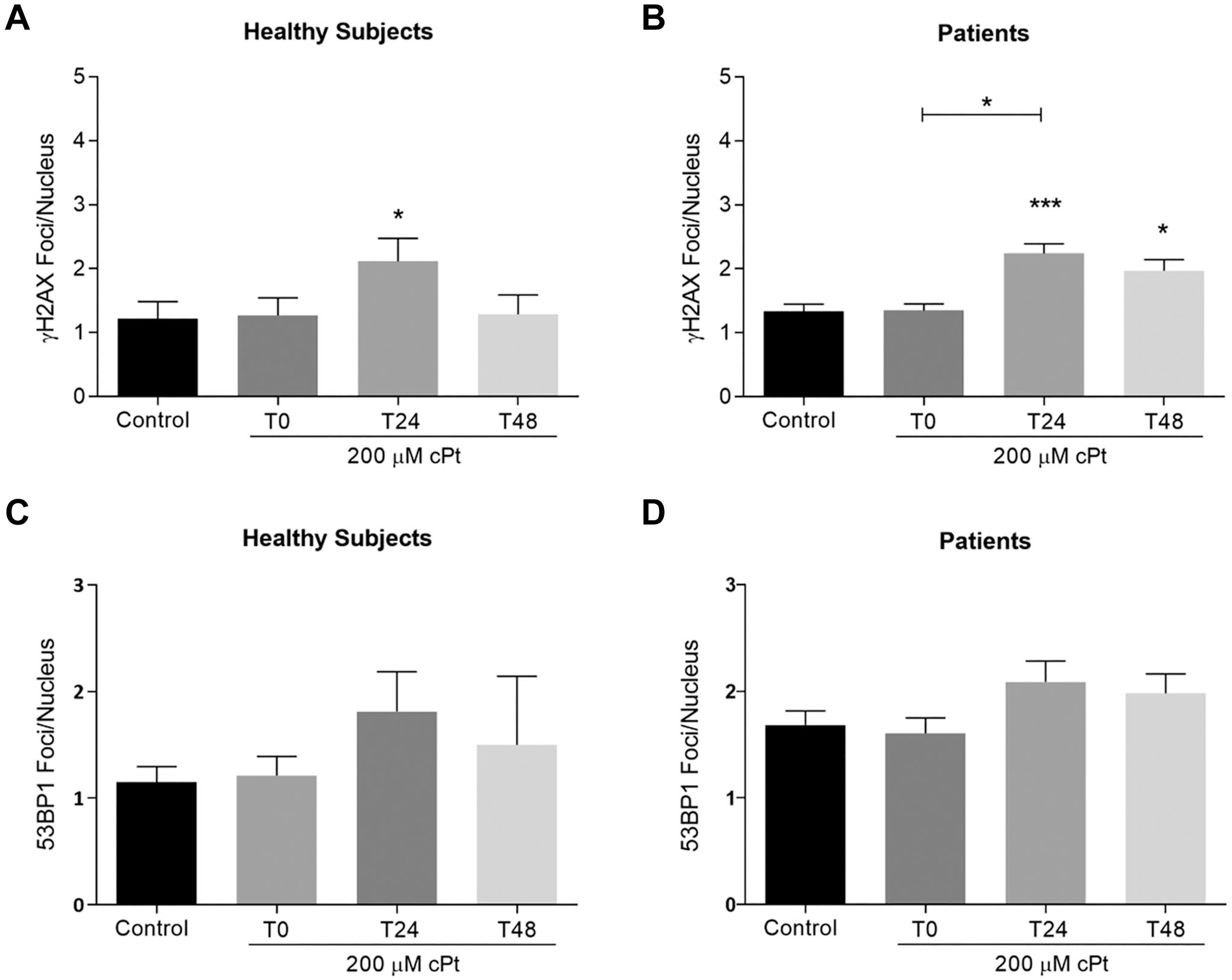
Positive immunoexpressions for γH2AX and 53BP1 as foci/nucleus in PBL from healthy persons and cancer patients in vitro exposed to cPt and across recovery periods. γH2AX foci/nucleus in healthy subjects (A) and patients PBL (B) under experimental treatments. 53BP1 foci/nucleus in healthy persons (C) and patients (D). Bars represent the mean±SEM. Control: untreated PBL. T0: PBL collected immediately after 200 µM cPt treatment. T24 and T48: PBL collected 24 or 48 hr post-cPt, respectively. Comparisons were done using Kruskal–Wallis with post-Dunn’s test. *p<0.05. ***p<0.001.
Phosphorylated-Hsp90α was evaluated in the PBL cytoplasm and nuclei. The treatment with cPt immediately increased cytoplasmic levels of p-Hsp90α in PBL from healthy subjects but did not affect the levels of the protein in the nucleus (Fig. 4A and C). After recovery, particularly at 24 hr post-cPt, the expression of the protein significantly increased in the nuclei (p<0.05), subsequently decreasing at 48 hr post-treatment (Fig. 4C). While the cytoplasmic expression of the protein showed no changes in patients (Fig. 4B), the nuclear levels significantly increased 24 hr after cPt (p<0.001) and remained at higher levels than control at 48 hr post-treatment (p<0.01, Fig. 4D). In both healthy individuals and patients, the N/C ratio increased at 24 and 48 hr after cPt treatment, but it was statistically significant only in cancer patients (p<0.001 and p<0.05, respectively) (Fig. 4E and F).
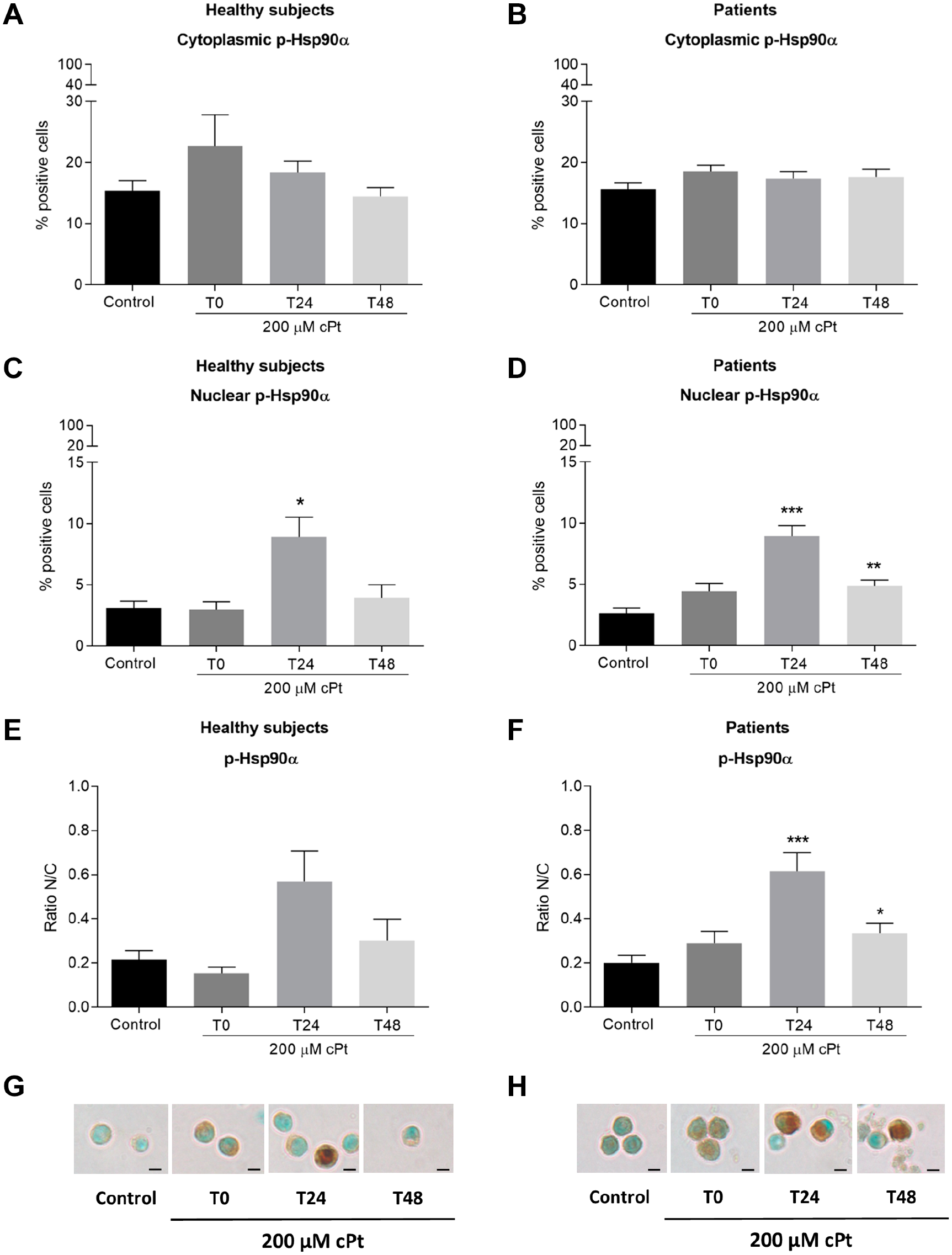
Immunoexpression of p-Hsp90α in healthy persons and patients PBL in vitro exposed to cPt and after 24 and 48 hr of recovery. Percentage of cytoplasmic positive immunoexpression for p-Hsp90α in healthy subjects (A) and patients (B) PBL in vitro treated with cPt. Percentage of nuclear positive PBL for p-Hsp90α expression in healthy subjects (C) and patients (D) after cPt exposure. Nuclear/cytoplasmic ratio (N/C) for p-Hsp90α in healthy subjects (E) and patients (F). Representative microphotographs for p-Hsp90α immunoexpression in PBL from healthy subjects (G) and patients (H). Note the cytoplasmic and the nuclear expression of the protein, the latter, particularly at T24 and T48. Bars represent the mean±SEM. Control: untreated PBL. T0: PBL collected immediately after 200 µM cPt treatment. T24 and T48: PBL collected 24 or 48 hr post-cPt, respectively. Comparisons were done using Kruskal–Wallis with post-Dunn’s test. *p<0.05. ***p<0.001. Photographs were captured using Nikon Eclipse 80i microscope. A scale bar was included in each microphotograph (10 µm).
DNA Damage and γH2AX, 53BP1, and p-Hsp90α Expressions Associations With Patient Response to cPt Treatment
The associations of clinical response with comet assay DNA migration mean and total DNA damage (score 5) were also evaluated. PBL from responder’s patients (CR/PR) that were exposed to cPt displayed higher levels of total DNA damage at T0 than non-responder’s patients (p<0.05, Fig. 5A). Fisher’s exact test comparisons using the healthy subjects’ mean value as cut-off showed that 48 hr after cPt treatment a higher percentage of cells with total DNA damage was associated with better clinical response (p=0.033). On the other hand, the DNA migration mean did not show differences between responders and non-responders patients (data not shown).
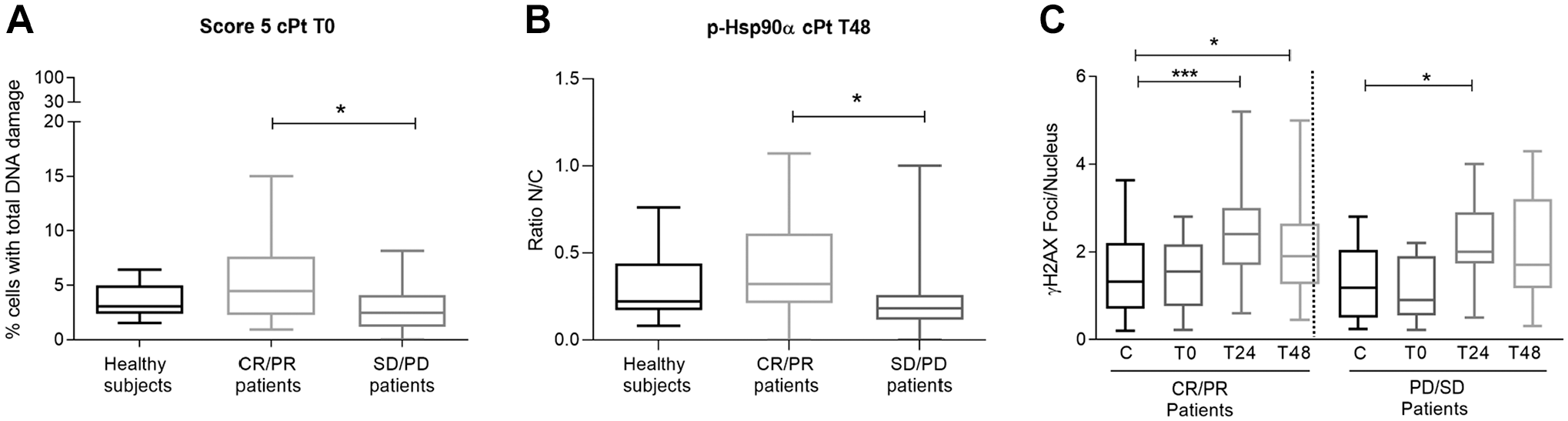
Comet DNA damage and expression of p-Hsp90α and γH2AX in cancer patients according to clinical response. (A) Percentage of cells with total DNA damage (score 5) immediately after cPt treatment (T0). (B) N/C ratio for p-Hsp90α at 48 hr post-cPt treatment (T48). (C) Patients’ kinetics of γH2AX foci/nucleus according to their clinical response to cPt-based chemotherapy. CR (complete response), PR (partial response), SD (stable disease), and PD (progressive disease). Comparisons between patients with CR or PR versus patients with SD or PD are shown. Box plots indicate mean±SEM. The p values were calculated using the Mann–Whitney test. *p<0.05. ***p<0.001.
A high N/C p-Hsp90α ratio at 48 hr post-cPt (T48) was interestingly found in responder’s patients (CR/PR) (p<0.05, Fig. 5B). Fisher’s analysis also showed that a high N/C p-Hsp90α ratio was associated with better clinical response to cPt-based chemotherapy (p=0.013).
The kinetics of γH2AX phosphorylation revealed increased levels of foci/nuclei particularly at 24 hr post-cPt treatment in both responders and non-responder’s patients (p<0.001 and p<0.05, respectively) (Fig. 5C). After 48 hr of cPt exposure, the levels of γH2AX foci/nucleus in the responder’s patients remained elevated unlike the untreated control group (p<0.05) (Fig. 5C).
No statistically significant differences were observed for 53BP1 status between experimental conditions and clinical response.
DNA Damage Biomarkers and Survival Among cPt-Treated Cancer Patients
A total of 32 patients (60.4%) died during the follow-up. Kaplan–Meier survival analysis showed that a high percentage of cells with total DNA damage (score 5) after cPt incubation (time 0, cPt0) was significantly associated with longer DFS of the patients (p=0.008; Fig. 6A). In addition, patients having high levels of total DNA damage at this time condition (cPt0) displayed longer OS, but the results trend toward statistical significance (p=0.0573; Fig. 6B). Cancer patients’ leukocytes with high DNA migration mean after 24 and 48 hr of cPt treatment had longer DFS (p=0.039 and p=0.038, respectively, Fig. 6C and E) and higher OS after 48 hr of cPt exposure (p=0.047) (Fig. 6F). No statistically significant associations were found between DNA migration mean and OS at T24 (Fig. 6D) and among γH2AX and 53BP1 with OS or DFS in cancer patients (data not shown).
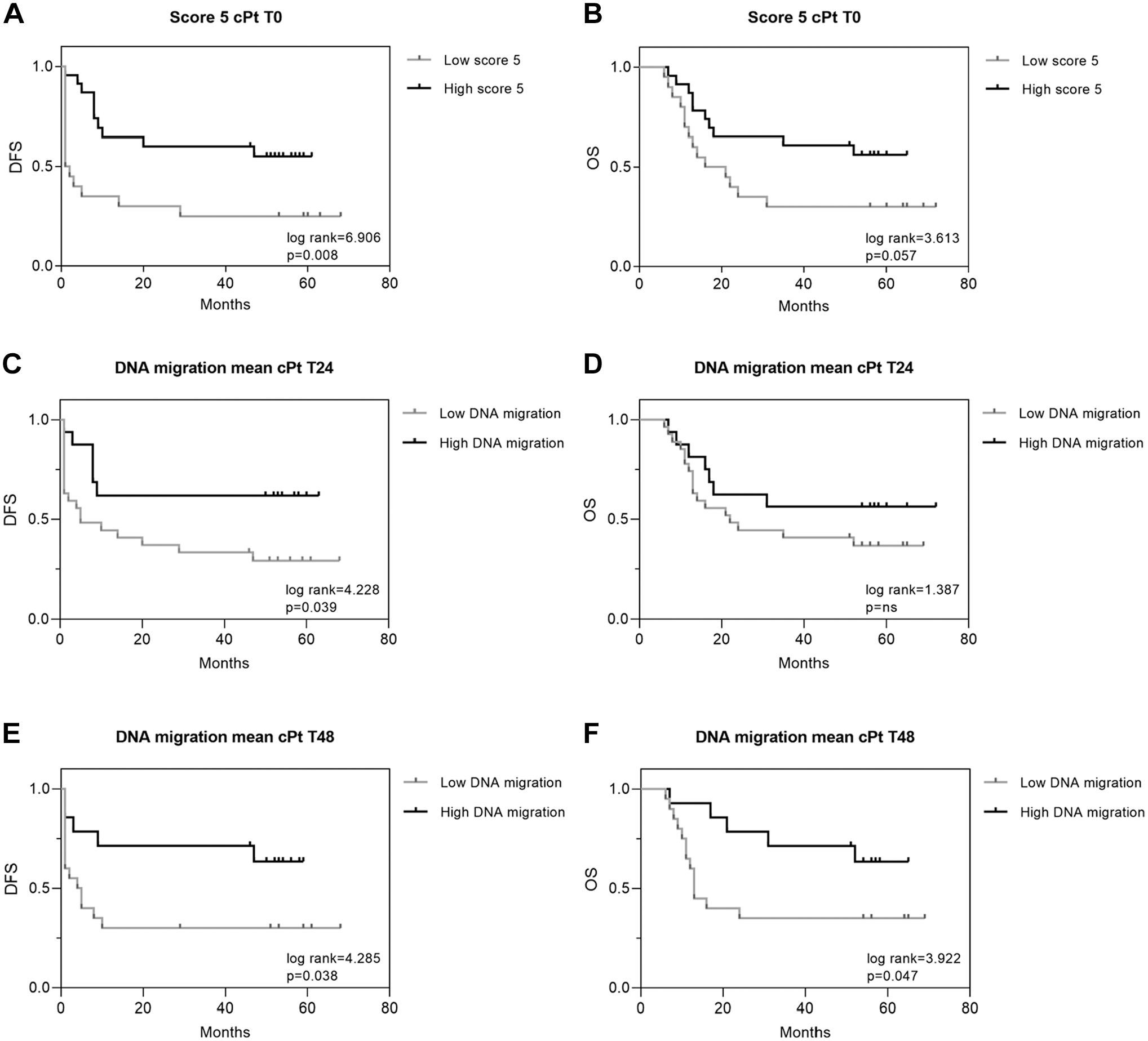
Kaplan–Meier curves of disease-free survival (DFS) and overall survival (OS) for comet assay total DNA damage and DNA migration mean. DFS (A) and OS (B) curve for the % of total DNA damage cells (score 5). DFS (C) and OS (D) curve for DNA migration mean after 24 hr recovery post-cPt (T24). DFS (E) and OS (F) curve for DNA migration mean at T48 (48 hr recovery post-cPt). The exact p-value is indicated inside each graph. *p<0.05. **p<0.01.
Discussion
For decades, cPt has been widely established for solid tumors treatment. The cytotoxic action of cPt is mediated by its interaction with DNA to generate DNA adducts, although the DNA from non-cancer cells is also damaged by the drug. A common approach to predict response drug sensitivity involves the use of PBL to measure the effect of antineoplastic drugs. PBL have been used to determine the DNA damage and DNA repair efficiency in various types of cancer at basal conditions and in response to antineoplastic drugs.10,11,23–25 However, the exploration of new practical biomarkers and cost-effective techniques for clinical response and prognosis evaluation is still needed. Many research studies have been published in this regard, but none has investigated the effects of cPt in PBL from cancer patients by testing conventional, such as γH2AX with new potential biomarkers of DNA damage. Biomonitoring studies in circulating PBL may contribute to precise and personalized chemotherapy choices assessment because they contain all the components of DNA repair machinery, which are specific for each person. Therefore, PBL can be employed as indicators of individual DNA damage repair response from exposure to chemotherapeutic drugs. 26
Here, the basal DNA damage was evaluated using pre-chemotherapy PBL from cancer patients under cPt-based therapy through the measurement of recognized DNA damage biomarkers. The baseline DNA damage was used as a control to establish comparisons in healthy subjects and cancer patients and between them (after cPt administration and post-cPt recovery). Comet assay evaluations demonstrated higher levels of basal DNA damage in cancer patients than in normal subjects. Nevertheless, the lack of statistically significant differences may be attributed to interindividual variations particularly detected among patients. Interindividual variability can be the result of many external factors (diet, air pollution, exercise, among others) that exceed the inclusion criteria taken for the recruitment of the patients in our study. Although this situation has been reported by other research groups, to improve the reproducibility of the assay, the method conditions need to be further standardized. 27 Previously, following other groups, our research team has shown that PBL from cancer patients are characterized by high background levels of DNA damage.11,28–30 These variations could also be attributed to genetic differences such as polymorphisms in DNA repair genes and changes in the expression of transcription factors and/or epigenetic factors.30,31 Despite that, to date, there is no convincing explanation for this finding in cancer patients. The levels of other DNA damage markers such as endogenous γH2AX and 53BP1 foci were also tested. In PBL from healthy persons, these molecule levels did not significantly differ from those in our cancer patients, and high interindividual variations were also noted. Some researchers are in line with our results, that is, breast cancer patients undergoing radiotherapy displayed large interindividual variability for these molecular markers. 32 In contrast, other research groups reported that cancer patients had higher γH2AX and 53BP1 levels than healthy persons, but it is important to consider that these studies were performed in cancer patients’ PBL exposed to ionizing radiation.33,34 One of the possible reasons for these variations in the background DNA damage levels may be related to genetic instability and alterations or polymorphisms in DNA repair among cancer patients as mentioned above. 31
The cPt-induced DNA damage and the repair kinetics in healthy subjects PBL using comet assay showed that 48 hr after cPt treatment, the levels of total DNA damage significantly increased, and they were higher than at T0 (immediately after cPt). Despite this, in PBL from cancer patients, the DNA migration mean levels post-cPt were like those of healthy individuals practically at all recovery times. The alkaline comet assay has shown similar sensitivity to detect DNA damage as the γH2AX biomarker. 35 After 24 and 48 hr of cPt exposure, the PBL from cancer patients had significantly elevated levels of γH2AX foci/nucleus (and not significantly high levels of 53BP1 foci/nucleus), suggesting that DNA damage in patients, in contrast with healthy subjects, remained unrepaired. Years ago, we demonstrated in a pilot study that the alkaline comet assay can be useful in predicting the response to chemotherapy. In this study, the DNA migration in PBL after cPt treatment at T0 and T24 was higher in cancer patients than in healthy individuals, showing a low rate of adducts repair in persons with cancer. 11 The reduction of the DNA repair capacity in patients has been proposed to be caused by gene alterations in the DNA repair genes and/or epigenetic factors, one of the hallmarks of cancer and the main determinants of DNA-damaging agents’ efficacy.5,31,36 Nevertheless, the interindividual variations observed through these methodologies highlight the importance of considering all these tools together to assess DNA damage in the progress of personalized protocols in cancer treatment.
It is well-known importance of using predictive and prognostic markers on providing information about clinical response to chemotherapy and the evolution of the cancer patient. In our study, the patients were divided according to their clinical chemotherapy response into two groups: those responders (CR or PR) and those non-responders (SD or PD). Patients who had a low percentage of PBL with total DNA damage (score 5) after cPt treatment (T0) exhibited an unfavorable clinical response. The levels of score 5 cells were very similar between healthy controls and responders’ patients. In a previous pilot study, we reported that the number of PBL with a score of 5 increased with chemotherapy and that cancer patients with clinical CR to chemotherapy showed higher levels of cells with total DNA damage than healthy individuals. 11 On the other hand, it is worth mentioning that alkaline comet assay detects DNA strand breaks (single and double), alkali-labile sites, and crosslinks; it has also been reported to be useful in response to therapy and drug resistance evaluation. 37 Other studies have reported the use of comet assay to predict chemotherapy response in cancer patients.38–40 Nonetheless, in these reports, the levels of other conventional or non-conventional DNA damage biomarkers at individual cell level were not determined. The measurements of the percentage of cells with total DNA damage and DNA migration mean in PBL from cancer patients showed that higher levels of cPt-induced DNA damage were associated with longer DFS and OS. Previously, we reported that PBL from deceased cancer patients showed lower levels of DNA damage than total cancer patients mean particularly 24 hr after cPt treatment. 11
Here, in addition to the comet assay, we also measured cPt-induced DNA damage and the DNA repair kinetics in patients PBL testing other molecular biomarkers by immunocytochemistry such as γH2AX and 53BP1. γH2AX, a marker of genomic instability, could also be used to follow cancer progression. 41 The phosphorylation of H2AX (γH2AX) is caused by cPt-induced interstrand crosslinks that originate replication fork arrest. The presence of γH2AX foci 24 hr after cPt treatment was shown to be a useful marker of cPt-induced DNA damage in human cancer cell lines. 42 We observed that responders like non-responders’ patients showed the same behavior in the DNA repair kinetics of cPt lesions (γH2AX foci), and no statistically signific-ant differences with healthy subjects were found. Nevertheless, γH2AX and 53BP1 determinations were not associated with the clinical response and outcome of the patients. In ovarian cancer patients, the DNA damage measured by γH2AX foci and comet assay in PBL was significantly higher in platinum-sensitive than in platinum-resistant individuals; moreover, patients were also characterized by a reduction of the DNA repair efficiency. 43 A diminished γH2AX response has been associated with increased radiosensitivity or chemosensitivity and poor outcome, so the identification of non-responders’ patients may contribute to optimizing and personalizing cancer treatments.44,45 Another study found no predictive utility for γH2AX and 53BP1 foci determinations regarding the radiotherapy outco-me. 33 The measurement of γH2AX and 53BP1 foci in PBL of patients receiving radioligand therapy may contribute to predicting patients with gastroenteropancreatic neuroendocrine tumors at high risk of early progression. 46 In summary, comet assay can be a useful tool for the follow-up of oncologic disease, but the prognostic role of γH2AX and 53BP1 using PBL from cancer patients was not found in our study. It is important to note that studies of these DNA damage biomarkers in chemotherapy-treated patients such as cPt are very scarce and our present work on patients’ PBL makes an important contribution in this field, being relevant for the clinic-al management of the cancer patients. Therefore, despite the promising predictive and/or prognostic value of these markers, they need larger studies to further establish their utility in clinical oncology.
Hsp90 is a phosphoprotein whose changes in phosphorylation status affect its chaperone activities. Hsp90 expressions have been correlated with prognosis and response to certain treatments. 47 The inducible form of Hsp90 has also been associated with DSB repair and multiple components of the DNA repair machinery, such as DNA-PK. 48 DNA-dependent protein kinase (DNA-PK) is a component of the DDR that phosphorylates Hsp90α on Thr-5 and Thr-7 during early apoptosis (p-Hsp90α). The p-Hsp90α rapidly localizes into the apoptotic ring, which participates in the apoptotic process induced by DNA dama-ge and enables the stabilization and activation of DNA-PK.16,49 Hsp90α phosphorylation is also crucial for γH2AX formation at sites of DNA breaks during the DDR. The key role of Hsp90α in the DDR, supported by the number of client proteins, has led to the hypothesis that genome instability is related to excessive demand for the protein that overcomes its capacity. 50 Furthermore, Thr-7 phosphorylation of Hsp90α correlates with γH2AX in tumors, suggesting its usefulness as a potential DNA damage biomarker.16,51 Because of p-Hsp90α implications in DDR, the study of the protein in cancer patients became the focus of our research. Recently, we reported that nuclear p-Hsp90α in tumor tissues correlated with good clinical response in patients treated with platinum-based chemotherapy. 17 However, the clinical implications of p-Hsp90α in PBL from cancer patients remain still unknown. For the first time, we evaluated by immunocytochemistry the nuclear and cytoplasmic expression of p-Hsp90α in pre-chemotherapy PBL from cancer patients at basal conditions and after cPt exposure. The basal levels of nuclear and cytoplasmic p-Hsp90α were not different between healthy subjects and patients, even the N/C ratio. Following cPt recovery, the cytoplasmic expression of p-Hsp90α practically did not change in either healthy subjec-ts or patients. However, it is important to mention that nuclear levels of p-Hsp90α and the N/C ratio increased within 24 hr in healthy persons and patients, subsequently decreasing 48 hr after cPt, pointing to the value of p-Hsp90α as a DNA damage biomarker. Moreover, those patients with better clinical response to cPt-based chemotherapy showed a high p-Hsp90α N/C ratio 48 hr after cPt treatment, highlighting for the first time in clinical research the importance of measuring p-Hsp90α in the nucleus and cytoplasm and its potential as a predictive marker of cPt response until now. Therefore, our results could explain p-Hsp90α role in DDR or apoptosis because the protein phosphorylation on Thr-5/7 has not been implicated in its chaperone activities. 51
Our results stress the relevance of studying the DNA damage associated with the cytotoxicity of platinum-based drugs due to its implications in therapy response and disease evolution. The use of DNA damage-related biomarkers in easily accessible PBL from cancer patients may reflect the individual chemotherapy response. Predicting the individual res-ponse to chemotherapy would be a great advance beyond personalized cancer treatment. The comet assay can rapidly measure cPt-DNA damage with high sensitivity, but it needs validation and standardization to be regularly applied to cancer patients. Early biomarkers for identifying DNA damage and repair status are important in the clinic to overcome cancer resistance and tailor therapy to achieve the goal of personalized therapy and avoid overtreatment with cytotoxic drugs. In conclusion, we believe that the study of DNA damage and p-Hsp90α subcellular localization in clinical oncology would be useful to determine the more adequate therapy for the cancer patients.
Footnotes
Appendix
Acknowledgements
We thank all the staff of the Regional Integration Cancer Center (COIR) for their invaluable contribution to this work.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SBN designed the study, performed an alkaline comet assay, and wrote the manuscript. MLS performed cell culture, immunocytochemistry, analysis, and interpretation of data, and contributed to discussing results. AR performed immunocytochemistry. LCG contributed to data statistical analysis. JI, MBG, and LG collected blood samples and performed clinical and therapeutic management of the patients and their follow-ups. LMVR revised the clinical and technical aspects of the manuscript. All authors have read and given their approval for the submission.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Agency for Scientific and Technological Promotion, Argentina (grant number PICT 2015-1171); the Alberto J. Roemmers Foundation, Argentina; and the National University of Cuyo, Mendoza, Argentina (grant number SIIP tipo 1 06/J525).
