Abstract
Preeclampsia is a complication of pregnancy that affects 3–5% of pregnancies and is one of the major causes of maternal/neonatal mortality and morbidities worldwide. We aimed to investigate the distribution of Foxp3+ regulatory T-cells and CD68+ Hofbauer cells in the placenta of preeclamptic and healthy pregnant women with a special focus on correlating these findings with placental histology. Decidua and chorionic villi of the placenta obtained from healthy and preeclamptic pregnancies were evaluated in full-thickness sections. Sections were stained with hematoxylin and eosin and Masson’s trichrome and immunostained for Foxp3 and CD68 for histological analyses. The total histomorphological score for placentas was found to be higher in preeclamptic placentas than that in the controls. The CD68 immunoreactivity was higher in the chorionic villi of preeclamptic placentas than that in the controls. The immunoreactivity of Foxp3 was found widely distributed within the decidua in both the groups and did not differ significantly. Interestingly, Foxp3 immunoreactivity in the chorionic villi was found mainly in the villous core and, to a lesser extent, in the syncytiotrophoblasts. We found no significant relation between Foxp3 expressions and morphological changes observed in preeclamptic placentas. Although extensive research is being carried out regarding the pathophysiology of preeclampsia, the findings are still controversial.
Introduction
Preeclampsia is considered a complication of pregnancy that affects 3–5% of pregnancies and is one of the major causes of maternal and neonatal mortality and morbidities worldwide. 1
The diagnostic criteria for preeclampsia have been revised as de novo hypertension occurring after 20 weeks of gestation and proteinuria (>300 mg/day) or other end-organ dysfunction such as maternal acute kidney injury, liver dysfunction, neurological or hematological complications, uteroplacental dysfunction, or fetal growth restriction (FGR).2,3 Although there are interventions to control blood pressure and prevent seizures, timed delivery remains the only treatment option for preeclampsia, which mostly leads to preterm birth compromising the health of the newborn.
It has been well known that preeclampsia resolves once the placenta is removed, and thus, disorders in placentation involving both maternal and fetal factors have become a focus in identifying the pathophysiology of the syndrome. Despite numerous research and extensive knowledge on structural, genetic, angiogenic, immunological, and metabolic pathways involved in preeclampsia, the pathophysiology remains poorly understood, limiting the prediction of and the interventions to prevent and treat preeclampsia.
Common pathological findings in the preeclamptic placenta are acute atherosis characterized by fibrinoid necrosis and accumulation of lipid-laden intimal macrophages, sclerotic narrowing of arteries and arterioles, fibrin deposition, and infarcts, which all reflect placental hypoperfusion and ischemia.4,5 Although these findings are not specific to preeclampsia, they have been found to be more common in preeclamptic pregnancies than in normotensive controls. 6 It has been shown that there is defective remodeling/physiological transformation of the spiral arteries that supply to the placenta by the extravillous trophoblasts in preeclampsia. 7 Moreover, in preeclampsia, the physiological transformation of the spiral arteries was limited to the decidua, whereas it extended into the myometrium in normal pregnancies. 8
Immunological pathways play a crucial role not only in a normal pregnancy but also in the pathophysiology of preeclampsia. 9 Maternal innate immunity usually provides protection from pathogens throughout pregnancy; however, an immune tolerance is displayed toward the semi-allogeneic fetus and placenta.10,11 A timely shift and a balance between the proinflammatory and antiinflammatory responses are essential for successful pregnancy.12 –15 Alterations in maternal immune tolerance have been found to be tightly related to the development of preeclampsia.16–18
Regulatory T-cells (Tregs), a subset of CD4+ T-cells, are the main cells involved in regulating immune tolerance. Tregs are characterized by CD4, CD25, and Foxp3 (Forkhead family transcription factor forkhead box p3). 19 Tregs have been found to be decreased significantly in the human endometrium of women with infertility and in cases of miscarriages, indicating their importance in establishing and maintaining a healthy pregnancy.20–22 Foxp3 expression is required for Treg development and function. Immune suppression is a major function of Tregs, and deficiency in Foxp3 expression impairs their function.23,24 Unlike other forkhead transcription factor family members, it is mainly expressed in a subset of CD4+ T-cells that play a suppressive role in the immune system.23,24 It acts through the suppression of the function of nuclear factor of activated T-cells and nuclear factor κB, which in turn leads to the suppression or expression of many genes. Tregs use Foxp3 as a lineage specification factor. It is known that the deficiency of Treg cells is related to the loss-of-function mutations of the Foxp3 gene causing severe systemic immune-mediated inflammatory lesions. 25
There is mounting evidence suggesting a possible contribution of Foxp3 polymorphisms in susceptibility to preeclampsia in different populations.26–30 Moreover, it has been observed that CD4+ Foxp3+ cell counts were significantly lower in maternal circulation of patients with preeclampsia than those in healthy pregnant women and nonpregnant controls.9,31 On the contrary, Rahimzadeh et al. 32 have concluded that changes in Treg cell number and function in preeclampsia remain a controversial issue in a review where they compiled the associations of Tregs and preeclampsia risk.
Macrophages are another type of immune cell that perform crucial immunological functions during pregnancy. They are involved in promoting tolerance toward the semi-allogeneic fetus and maintaining the homeostatic environment needed for normal fetal development.18,33–35 They comprise a heterogeneous population of immune cells, and among these are the macrophages of the placenta, the Hofbauer cells, which are found in the stroma of placental villi throughout pregnancy. 36 These macrophages play important roles in placental development, morphogenesis, 37 vasculogenesis, and angiogenesis.38,39 Hofbauer cells are involved in the pathophysiology of pregnancy complications such as chorioamnionitis and villitis of unknown etiology. 40 Moreover, the number of CD163-positive Hofbauer cells was found to have significantly decreased in preeclamptic pregnancies compared with that in normal pregnancies. 18 Another study also reported that Hofbauer cell count decreased in preeclampsia cases, and it is suggested that these cells have essential immune regulatory roles in maternal-fetal tolerance. 33 In contrast, Zelinka-Khobzey et al. 41 evaluated and reported that the stroma of the terminal villi of the placenta had a higher expression for CD68+ macrophages in preeclampsia than that in the control group. There is limited information on the role of these cells in placental physiology and in the pathophysiology of preeclampsia.
Considering the important roles and the limited and inconsistent information about the functions of Tregs and Hofbauer cells in the pathogenesis of preeclampsia, the present study was carried out to investigate the distribution of Foxp3+ Treg cells and CD68+ Hofbauer cells in the placenta of preeclamptic women and healthy pregnant women with a special focus on correlating these findings with placental histology.
Materials and Methods
Study Groups and Tissue Collection
Placental tissues from 43 women were studied in the following groups: preeclampsia (PE;
Human maternal peripheral blood, urine, and placental tissues were obtained from participants recruited from the Department of Obstetrics and Gynecology of Zeynep Kamil Women and Children Diseases Training and Research Hospital (Uskudar, Istanbul, Turkey). There was no significant difference in the age of the healthy and preeclamptic mothers. The average age of the participants in the healthy pregnant women group was 29.79 ± 1.25 years, and in the preeclamptic pregnant women group, the average age was 30.94 ± 1.34 years. The ethnic background of both the groups was Caucasian (100%). Gestational age was established based on the menstrual date confirmed by sonographic examination before 20 weeks of gestation. The demographics and other characteristics of the mothers are indicated in Table 1.
Clinical and Laboratory Findings in Control and Preeclampsia Groups.
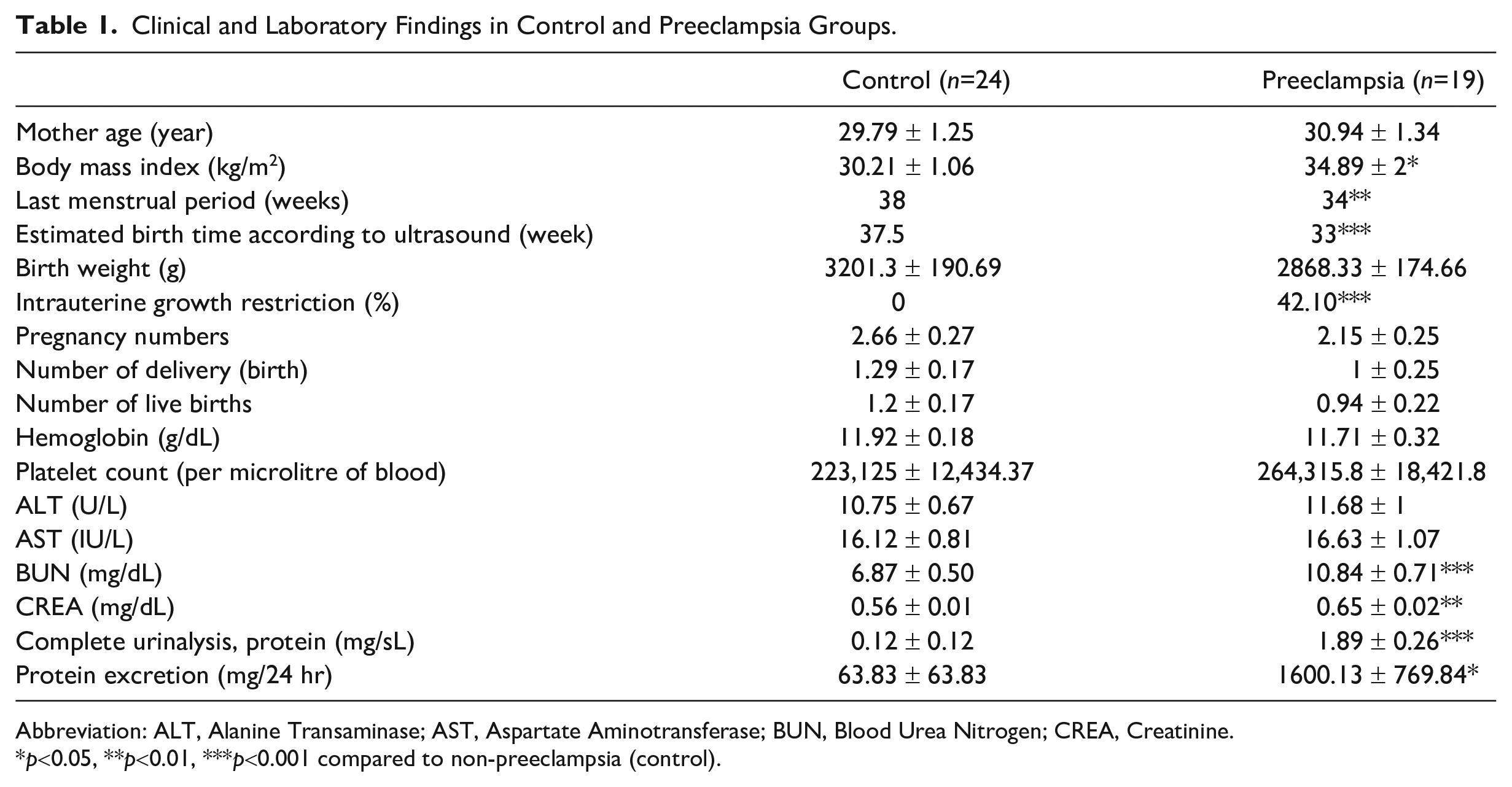
Abbreviation: ALT, Alanine Transaminase; AST, Aspartate Aminotransferase; BUN, Blood Urea Nitrogen; CREA, Creatinine.
All preeclampsia patients were diagnosed using the following criteria: (1) gestational proteinuric hypertension after >20 weeks of gestation, (2) systolic blood pressure of ≥140 mmHg, (3) diastolic blood pressure of ≥90 mmHg on two occasions at least 6 hr apart, (4) proteinuria ≥300 mg in a 24-hr urine specimen or ≥3+ on two dipstick urine samples collected at least 4 hr apart, (6) oliguria (≤500 ml/24 hr), and (7) thrombocytopenia (<100.000 mm3). Control normotensive pregnant women had a normal range of protein levels for pregnancy (6.1–7.9 mg/dl). Patients with a history of diabetes or diagnosis of gestational diabetes and patients that have received any kind of fertility treatment were excluded from the study.
Placentas were collected, evaluated macroscopically, weighed, and measured by the obstetrician immediately after cesarean section or vaginal delivery. Bilobed/multilobed placentas and placentas with velamentous cord insertion or abnormal appearances/shapes were omitted from the study. For the standardization of the collected samples, placentas with a thickness between 2.5 and 3.5 cm, diameter between 15 and 25 mm, and weight between 500 and 650 g were included in the study. To assess the organ from the basal plate to the chorionic plate, full-thickness tissue biopsies were collected from the edge of the placenta in addition to villous tree and decidua samples obtained from different cotyledons such as the ones closer to the umbilical cord. Tissue samples were excised from areas without visible damage, hematomas, or infarctions. To remove maternal blood, samples were washed in ice-cold phosphate-buffered saline (PBS). Placental tissue samples were fixed in 10% neutral-buffered formalin. After washing under tap water, they were dehydrated with a series of ethanol immersion, cleared in xylene, and embedded in paraffin for immunohistochemical and histomorphological analyses. Ten tissue biopsies were performed for each of the subjects; therefore, there were 10 paraffin-embedded tissue blocks for each subject. Four sections from each block were taken for the hematoxylin and eosin (H&E) staining, Masson’s trichrome staining, immunohistochemistry (IHC), and immunofluorescence (IF) experiments. For the histomorphological analysis using H&E and Masson’s trichrome, 10 fetal and 10 maternal areas were scored. For the IHC analysis, five areas in each section were scored.
Immunohistochemistry
Five-μm-thick serial sections were collected on poly-l-lysine–coated slides (Sigma, St. Louis, MO) and incubated overnight at 56C. Tissue sections were deparaffinized in xylene and rehydrated in a graded series of ethanol. Sections were then boiled in a microwave oven in 10-mM citrate buffer, pH 6.0, for 15 min. Thereafter, the slides were left to cool for 20 min. Endogenous peroxidase activity was quenched by 3% hydrogen peroxide in methanol for 20 min. Afterward, sections were incubated in a blocking serum (Ultra V Block, TP-060-HL; NeoMarker, Fremont, CA) for 7 min to block nonspecific binding. Without the washing step, sections were subsequently incubated overnight at 4C with either mouse monoclonal antihuman Foxp3 (Abcam-Ab20034/dilution 1:100) to identify the Tregs or mouse monoclonal antihuman CD68 (Thermo Scientific CD68/Macrophage Marker Ab-3/dilution 1:100) to identify the placental macrophages. Sections were incubated with the biotinylated secondary antibody (TP-060-HL; Lab Vision, Fremont, CA) and a streptavidin-peroxidase complex (TP-060-HL; Lab Vision) for 15 min, respectively. The resulting signal was developed with diaminobenzidine (Dako, CA, USA; K3466). Each step was followed by three washes in PBS unless otherwise stated. Sections were counterstained with Mayer’s hematoxylin, dehydrated, and mounted with CytoSeal 60, PSA, USA. Human tonsil tissue sections were used as the positive controls, whereas appropriate mouse immunoglobulin G (IgG) diluted to the same final protein concentration as the primary antibody was used for negative controls. All samples for each antibody were exposed to the same protocol at the same time and were stained using the same incubation periods. Photomicrographs were taken using an Olympus BX61 microscope (Olympus, Japan).
Immunofluorescence
Five-µm-thick sections were collected on positively charged slides and incubated overnight, deparaffinized in xylene, and rehydrated in a graded series of ethanol. Sections were then boiled in a microwave oven in 10-mM citrate buffer, pH 6.0, for 15 min. Thereafter, the slides were left to cool for 20 min. Afterward, sections were incubated in a 5% BSA solution for 1 hr to block nonspecific binding. Without the washing step, sections were subsequently incubated for 1 hour with both mouse monoclonal antihuman Foxp3 (Abcam, UK; Ab20034/dilution 1:100) and rabbit monoclonal antihuman keratin 7 (KRT7) (Boster, CA, USA; M02416-1/dilution 1:200) for double staining to identify the Foxp3-positive cells and trophoblasts, respectively. Sections were incubated with fluorescent-labeled goat antimouse IgG (Abcam, UK; Ab175471) and goat antirabbit IgG (Santa Cruz, TX, USA, sc-2010). Each step was followed by three washes in PBS unless otherwise stated. Sections were mounted by using a mounting medium with DAPI (4,6-diamidino-2-phenylindole) (Abcam, UK; 104139). Appropriate mouse IgG diluted to the same final protein concentration as the primary antibody and normal rabbit serum was used as negative controls. Photomicrographs were taken using an Olympus BX61 microscope with a fluorescence attachment (Tokyo, Olympus, Japan).
H&E Staining
For H&E stainings, sections were first immersed in Ehrlich’s hematoxylin. After confirming that the nuclei were stained blue, excessive dye in the cytoplasm was washed with acid alcohol. Tap water was used as the mordant, and then the sections were immersed in eosin dye to stain the cytoplasm pink.
Masson’s Trichrome Staining
To analyze connective tissue elements, Masson’s trichrome staining was used. The sections were immersed in Weigert’s iron hematoxylin until the nuclei were stained, and then tap water was used as a mordant. Afterward, sections were stained with xylidine ponceau and methyl blue to highlight the cytoplasm and connective tissue, respectively.
Histomorphological Analysis and Image Data Analysis
For histomorphological analyses, tissues stained with H&E and Masson’s trichrome were examined under a light microscope (Olympus BX61, Japan) by two histologists blinded to the groups. A specific histological scoring method was used by modification of previous data because of the limited microscopic morphological studies and the lack of consistency of these studies on human preeclamptic placentas.42–44 We have used the terminology and definitions of placental lesions according to a workshop consensus published by Khong et al. 45 Both decidua and chorionic villi of the placenta of preeclamptic and control placentas were evaluated, and a score of 0 (no pathological finding), 1 (mild pathological findings), 2 (moderate pathological findings), or 3 (severe pathological findings) was given for the following criteria: calcifications, syncytial knots, fibrinoids, infarct, and intravillous hemorrhage. Representative photomicrographs with the aforementioned pathologies are given in Fig. 1.
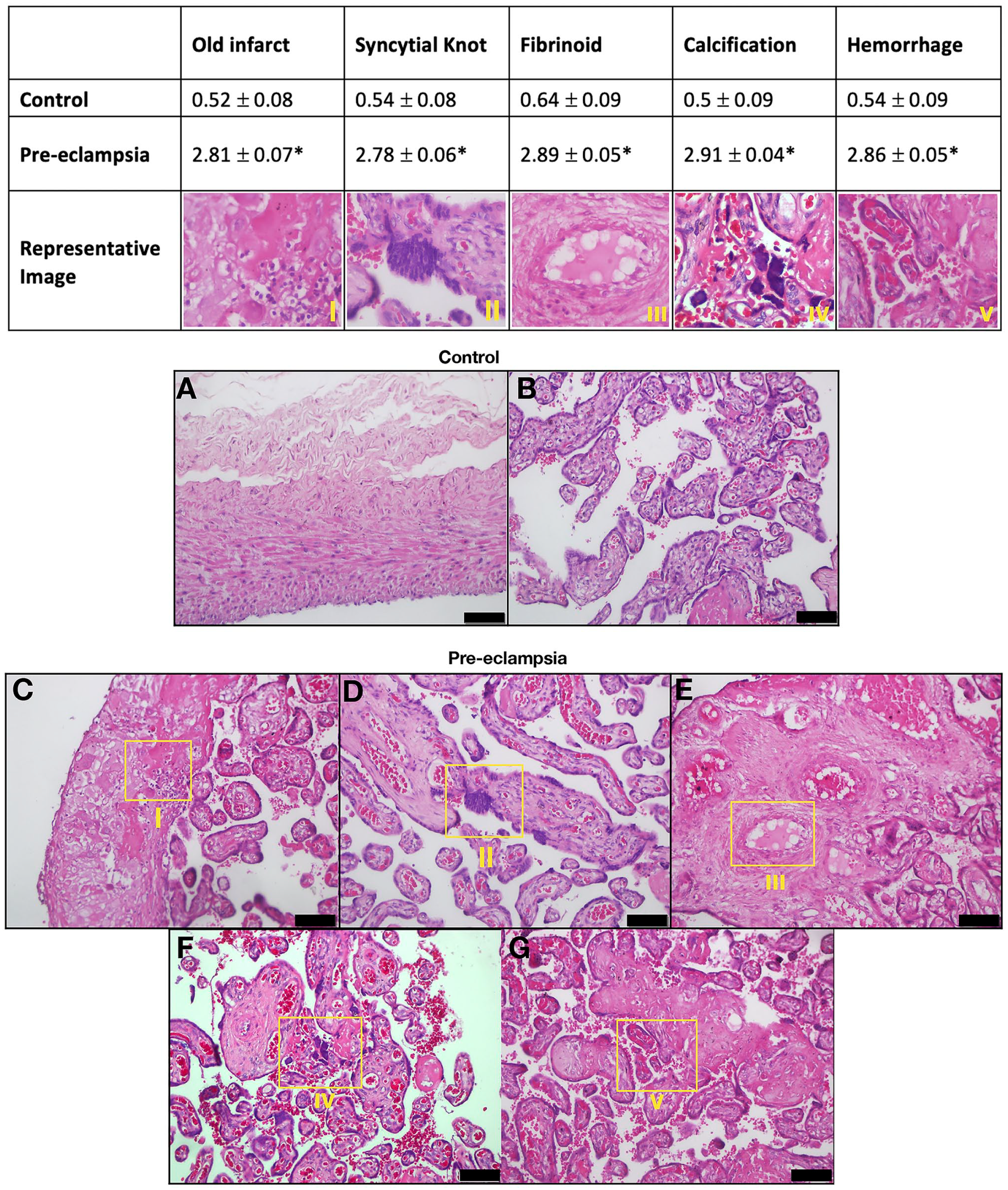
Histomorphological score for each criterion in control and preeclampsia groups. (A) Decidua and (B) chorionic villi of control; (C–G) preeclempsia groups show low magnification for each criteria. Scale bars = 50 µm. *
In addition, the Masson’s trichrome stain was used to measure connective tissue by using ImageJ, an open-source digital-image-processing software program. 46 Therefore, three random images were captured from either decidua basalis or stem or anchoring stem villi from decidua and chorionic villi sides of the placenta with a 20× objective. We performed a two-step evaluation for the analysis of Masson’s trichrome–stained connective tissue micrographs. In the first step, only connective tissue areas were measured, and this was done via the following procedures: (1) The image was opened in the software, and color deconvolution was applied to divide the image into its color channels, (2) all color channel windows were closed except the blue one which represents the stained connective tissue areas, (3) the threshold was applied to detect all blue staining areas, and (4) the measure command was used to calculate only detected areas. In the second step, all the stained tissue components of the same image were measured via the following procedures: (1) The image was opened in the software and converted to the 8-bit type, (2) the threshold was applied to detect all tissue components, (3) the measure command was used to calculate only the detected area. We calculated the final connective tissue area percentage by using the following formula: (the measured area from the first step)/(the measured area from the second step) × 100.
A H-score analysis was carried out for immunohistochemical stainings. The intensity of immunoreactivity was semi-quantitatively evaluated as follows. Positively stained cells were grouped according to the following categories: (no staining) 1+ (weak but detectable), 2+ (moderate or distinct), 3+ (intense). For each tissue, an H-score value was calculated by summing the percentages of cells grouped in one intensity category and multiplying this number with the weighted intensity of the staining, using the formula [HSCORE = Pi(i + l)], where i represents the intensity scores, and Pi is the corresponding percentage of the stained cells. In each slide, five different areas were evaluated under a microscope at 200× original magnification. The percentage of cells for each intensity within these areas was determined by two investigators who were blinded to the groups, and the average score was used. 38
Statistical Analysis
Statistical analyses were performed using the R.
47
For independent samples, a
Results
Clinical Findings
Placental samples from 24 normal and 19 preeclamptic women were collected. The demographic and clinical characteristics and laboratory findings of these pregnant women are given in Table 1. The median ages of the pregnant women were similar between the healthy controls (29.79 ± 1.25) and the preeclamptic women (30.94 ± 1.34). The body mass index of women with preeclampsia was higher than that of the controls (
Histomorphological Findings
Prominent areas of calcification were observed particularly in the preeclamptic placentas. These calcification areas were more abundant than the control placentas (2.91 ± 0.04 in PE vs 0.5 ± 0.09 in C,
The total histomorphological score calculated for placentas was found to be significantly higher in preeclamptic placentas than that in the controls (
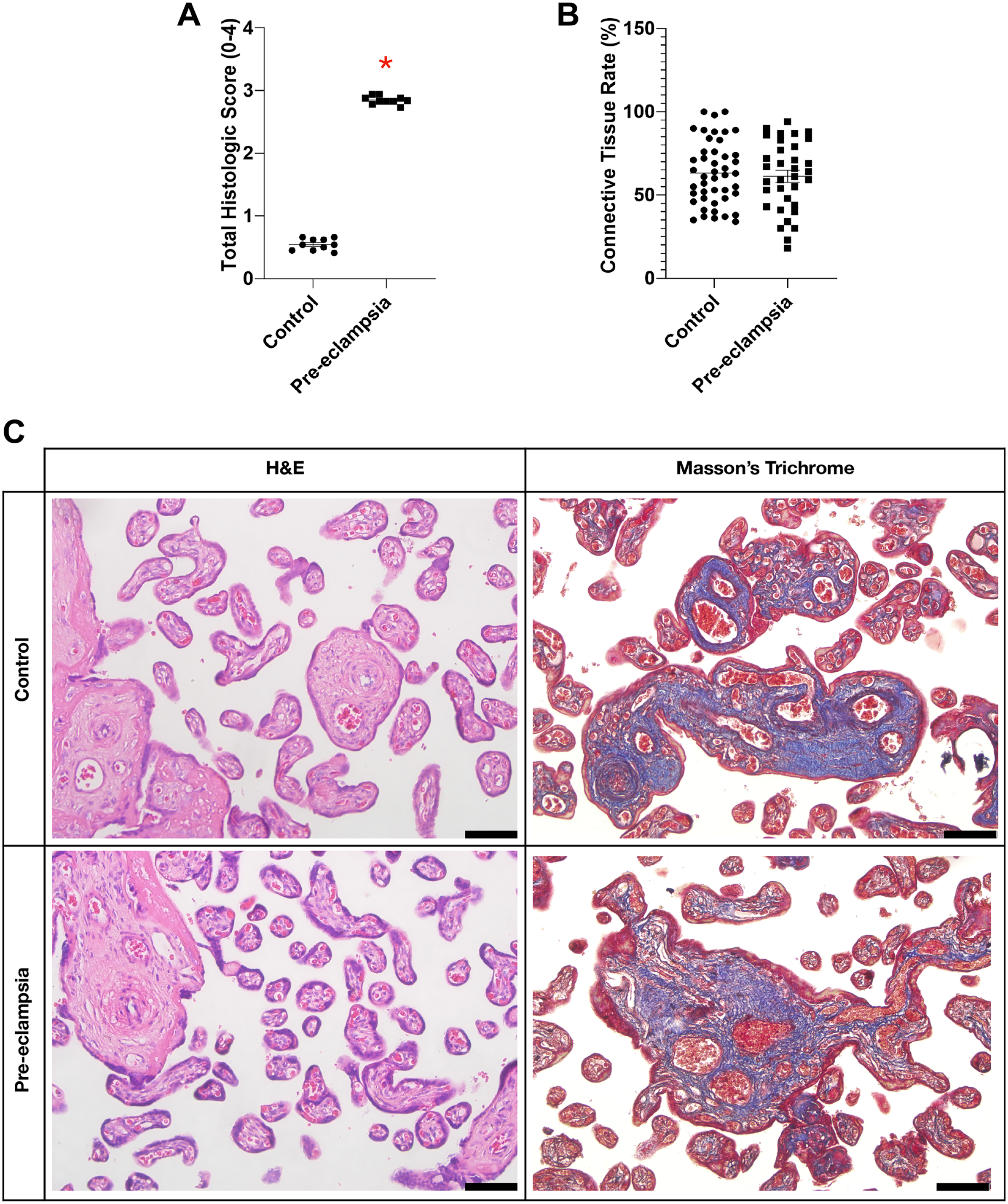
Graphs of (A) total histomorphological score, (B) connective tissue area rate, and (C) hematoxylin & eosin and Masson’s trichrome staining of the placenta in control and preeclampsia groups. Scale bars = 50 µm. *
The connective tissue rate detected with Masson’s trichrome staining and analyzed by ImageJ showed no differences among the groups (Fig. 2B).
Immunohistochemical Findings
Sections belonging to both the decidua and chorionic villi of the preeclamptic and control placentas were stained immunohistochemically with the CD68 antibody to identify Hofbauer cells and with Foxp3 antibody to identify Treg cells. According to the H-SCORE analysis, CD68 immunoreactivity was lower in the decidua, in both the control and preeclamptic placentas, than that in their chorionic villi. When the CD68 immunoreactivity was compared between the decidua of control and preeclamptic groups, there was a trend of increase in the preeclamptic decidua, but this increase was insignificant. The CD68 immunoreactivity was significantly higher in the chorionic villi of preeclamptic placentas than that in the controls (
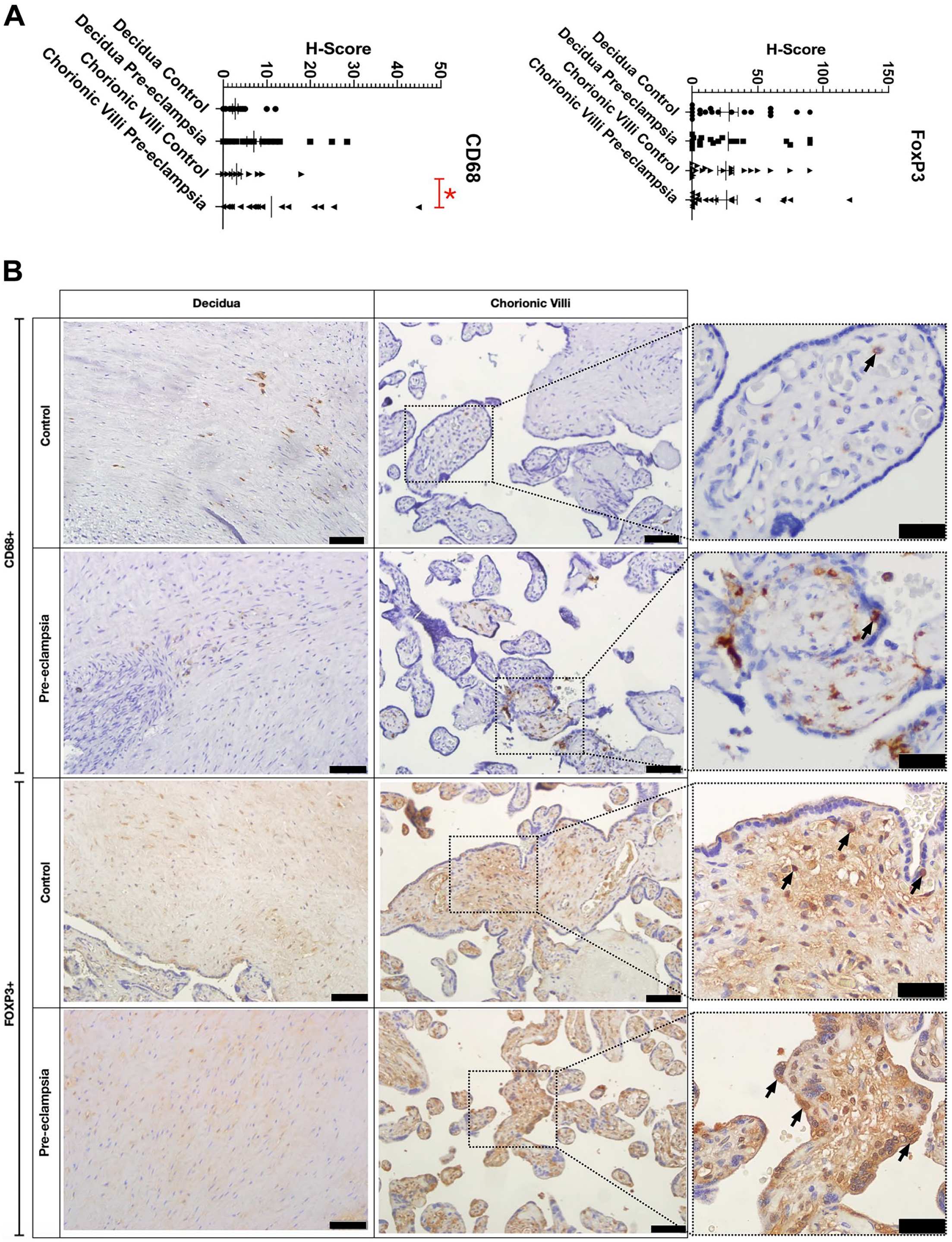
(A) Graphs of H-score for Foxp3 and CD68. (B) Micrographs of Foxp3 and CD68 immunostaining in control and preeclampsia groups. Scale bars = 50 µm and 20 µm, respectively. *
The immunoreactivity of Foxp3 was found widely distributed within the decidua in both the groups and did not differ significantly. Foxp3 immunoreactivity in the chorionic villi was found mainly in the villous core and, to a lesser extent, in the syncytiotrophoblasts, which did not exert any significant difference among the two groups (Fig. 3A and B). Negative controls did not show any reaction, and positive control showed reaction (Supplemental Fig. 1S).
IF Findings
Immunofluorescent staining was performed with CD68 and Foxp3 antibodies on sections belonging to both preeclamptic and control placentas. The KRT7 immunostaining was observed solely in trophoblasts (Fig. 4, KRT7, red). Foxp3 expression was localized in some of these KRT7-positive cells in the control group, whereas Foxp3 (Fig. 4, Foxp3-Green) immunolocalization was found in most of the KRT7-positive cells in the preeclamptic group. When the images were merged, it was observed that more cells showed colocalization in the preeclamptic group than in the control group (Fig. 4, merge, yellow). Negative controls did not show any reaction (Supplemental Fig. 2S).
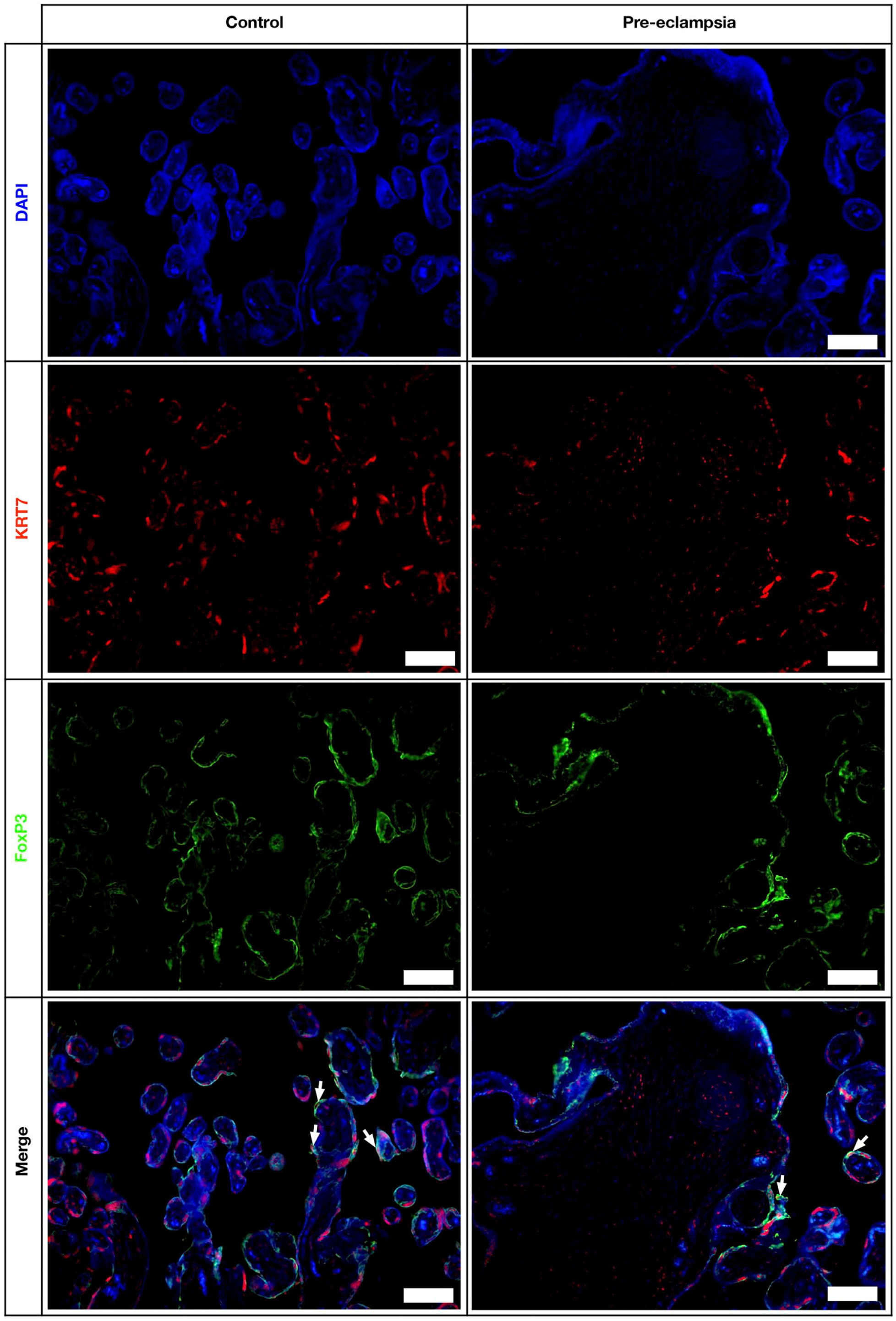
Immunofluorescent staining; chorionic villi are seen in the section. DAPI (blue), KRT7 (red), Foxp3 (green), and merged images. Scale bars = 50 µm.
Discussion
Although the etiology of preeclampsia remains undefined, it has been associated with insufficient invasion of spiral arteries by the trophoblasts, endothelial cell dysfunction, poor placental perfusion,5,48 and to some extent, immune system dysregulation.9,49
In normal pregnancy, the maternal immune system develops tolerance toward the allogeneic fetus. Macrophages and immunosuppressive Treg cells play roles in the development of this tolerance favoring successful pregnancies.9,35 The placental immune system comprises natural killer (NK) cells, macrophages (~20%), T-cells (~10–20%), and rarer cell types, such as dendritic cells, B-cells, NK T-cells, and mast cells. 50
In this study, we investigated placentas from normal and preeclamptic pregnancies for the identification of histomorphological changes and the distribution of Foxp3-positive cells and CD68-positive macrophages.
Most of the studies regarding Treg cells in preeclampsia studied these cells in the peripheral blood and, to a lesser extent, in the decidua. Moreover, the results of these studies are still controversial. Some studies examining peripheral blood indicate a decrease in Treg cell numbers9,51,52 in preeclampsia, whereas some show no change in Treg cells in preeclampsia compared with normal subjects.53,54 There are fewer numbers of studies like ours that examine Tregs in preeclamptic decidua basalis of the basal plate. Tilburgs et al. 55 have shown that Treg cells are abundant in decidual tissues at the maternofetal interface, the decidua basalis, and predominantly in the decidua parietalis.
Contrary to the results of studies by Chen et al. 30 and Quinn et al., 49 which have suggested a significant decrease in Foxp3 expression in the decidua of preeclamptic pregnancies, we found no significant change in Foxp3 expression in the decidua of preeclamptic pregnancies compared with normal placentas. Interestingly, besides the expected expression of Foxp3 in Tregs, in our study, we detected Foxp3 expression in chorionic villi, trophoblasts, some fetal blood cells, and mesenchymal cells. We used appropriate isotype-negative and isotype-positive control tissues for our antibody and did not observe staining in the negative controls. To test whether the stainings were specific, we further performed double immunofluorescent staining with KRT7 (cytokeratin-7) and Foxp3. Consistent with our immunohistochemical findings, the trophoblasts exerted positivity with Foxp3. These findings are also consistent with the findings of Chen et al. 30 who have also shown the same pattern of staining in the control and preeclamptic villi. Our study is the first to emphasize the staining of Foxp3 in the placenta. The functions of Foxp3 in these cells need further investigation.
The exact role of macrophages in the pathogenesis of preeclampsia is still to be elucidated. It has been shown that these cells regulate pregnancy, 56 facilitate tolerance toward the allogeneic fetus, 35 and play important roles in placental vascularization and angiogenesis. 39
The numbers of these cells have been studied by several groups, and similar to many aspects of preeclampsia, the findings of these studies are also controversial. While some studies indicate an increase in their numbers in decidua from preeclamptic women compared with controls, 57 some indicate a decrease, 58 and some even indicate that there is no change. 59
In the present study, we have examined CD68-positive macrophages both in the decidua basalis and chorionic villi in control group, consistent with the study by Bezemer et al. 60 Our finding that there is no difference in the distribution of decidual macrophages in preeclampsia is in accordance with the results of previous studies indicating that the number of CD68 immunopositive cells in the basal plate and decidua did not show any difference in preeclamptic women.59,61 However, we observed a significant increase in CD68-positive macrophages, so-called Hofbauer cells, in the chorionic villi of preeclamptic placentas. According to our histomorphological findings, we found more intravillous hemorrhage in chorionic villi, which may be related with this CD68 increment.
There are two main subgroups of macrophages: M1, or classically activated macrophages, and M2, or alternatively activated macrophages. M1 macrophages mediate resistance to pathogens and are proinflammatory, and M2 macrophages are immunomodulatory and secrete anti-inflammatory cytokines.62,63 The local cytokine and growth factor milieu in the tissue microenvironment determines the polarity of macrophages. Appropriate temporal regulation of M1–M2 switch is vital for successful pregnancy. 64 Disturbances in the balance between M1 and M2 and inappropriate macrophage polarization may result in complications, such as preeclampsia, FGR, and preterm delivery.65–67 It has been suggested that preeclamptic women have fewer M2 macrophages but more classically activated M1 macrophages, suggesting that polarization of macrophages toward the M1 phenotype may be responsible for the inflammatory environment in these pregnancies.64,68 However, in a study by Schonkeren et al., 57 CD163, a marker used for the detection of M2 cells, was significantly upregulated in the decidua basalis of preterm preeclamptic pregnancies compared with that in preterm control pregnancies. In our study, inconsistent with some other studies, we did not find any significant changes in the decidual macrophages of the preeclamptic women.59,61 This discrepancy might have occurred because we used the pan-macrophage marker CD68 and have not distinguished between the M1 and M2 phenotypes. We have found an increase in Hofbauer cells in the chorionic villi of preeclamptic pregnancies compared with controls. Our results are consistent with a study by Reister et al. 69 Because preeclampsia is associated with excess inflammation, it is possible that alteration of macrophage phenotype from M2 to M1 and the subsequent altered behavior might be responsible for the decrease in trophoblast invasion observed in preeclampsia. 70 It has been shown that peripheral blood macrophages treated with lipopolysaccharide, a potent macrophage inducer of type-1 cytokines, can significantly reduce extravillous trophoblast invasiveness in vitro. 71 With regard to this finding, the secretions of M1 macrophages could also lead to a similar effect on trophoblast invasiveness, contributing to inadequate invasion of spiral arteries in preeclampsia. In the light of the aforementined information, one can say that the type of macrophages rather than the number is important in the pathogenesis of preeclampsia.
Maternal obesity is one of the risk factors associated with the development of preeclampsia, particularly late-onset preeclampsia.5,72,73 It is suggested that obesity-associated low-grade chronic inflammation might be the influencing factor leading to complications in pregnancies, but the underlying mechanism is yet to be elucidated. 72 A limitation of our study is that we did not exclude obese patients. Therefore, it must be kept in mind that the observed changes in the distribution of the immune cells might also be affected by the maternal obesity-related changes although not yet proven. Therefore, further studies investigating the immune cell composition and activities in obese pregnant women and obese preeclamptic women are needed. 72
Studies have shown that there is an increased level of placental apoptosis in preeclamptic pregnancies.74,75 Ishihara et al. 75 have shown that syncytiotrophoblasts exerted a decreased level of Bcl-2 and thus a higher rate of apoptosis in severe preeclamptic and intrauterine growth retardation–term placentas than that in normal-term placentas. It should be kept in mind that both the decidual macrophages and Hofbauer cells of the chorionic villi are phagocytes that are responsible for the efficient clearance of the apoptotic bodies to prevent aberrant inflammatory responses. Therefore, an increase in Hofbauer cell numbers in preeclamptic placentas found in our study might be related to the increase in apoptosis observed in preeclampsia.
In the present study, the total histomorphological score for placentas was calculated and found to be significantly higher in preeclamptic placentas. Placental abnormalities such as calcifications, syncytial knots, intravillous hemorrhage, and infarcts were more frequently detected in the preeclamptic placentas. Our findings are consistent with the study by Sankar et al. 76 who found that syncytial knot density is significantly higher in preeclamptic placentas than that in healthy controls. However, Lager et al. 44 have found no substantial difference in calcifications and fibrin prevalence between complicated and healthy pregnancies.
In conclusion, we have shown that Foxp3 expression is not correlated with histomorphological abnormalities observed in preeclamptic placentas. Studies on lesions observed in preeclampsia decidua and chorionic villi tissues and their frequency and the numbers as well as distribution of immune cell populations in these tissues are quite contradictory. Therefore, more studies are needed to assess the changes and elucidate the underlying mechanisms in the pathophysiology of this disease with Foxp3+ and CD68+ cells.
Supplemental Material
sj-tif-1-jhc-10.1369_00221554231170662 – Supplemental material for The Distribution of Foxp3 and CD68 in Preeclamptic and Healthy Placentas: A Histomorphological Evaluation
Supplemental material, sj-tif-1-jhc-10.1369_00221554231170662 for The Distribution of Foxp3 and CD68 in Preeclamptic and Healthy Placentas: A Histomorphological Evaluation by Yasemin Ersoy Canillioglu, Gozde Erkanli Senturk, Hakan Sahin, Sadik Sahin and Yasemin Seval-Celik in Journal of Histochemistry & Cytochemistry
Supplemental Material
sj-tif-2-jhc-10.1369_00221554231170662 – Supplemental material for The Distribution of Foxp3 and CD68 in Preeclamptic and Healthy Placentas: A Histomorphological Evaluation
Supplemental material, sj-tif-2-jhc-10.1369_00221554231170662 for The Distribution of Foxp3 and CD68 in Preeclamptic and Healthy Placentas: A Histomorphological Evaluation by Yasemin Ersoy Canillioglu, Gozde Erkanli Senturk, Hakan Sahin, Sadik Sahin and Yasemin Seval-Celik in Journal of Histochemistry & Cytochemistry
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: conception of and planning the experiments (GES, YEC), performing histological methods (HS under the supervision of GES, YEC, and YS-C, and all authors contributed to the interpretation of the results and writing the manuscript, provided critical feedback, and helped shape the research, analysis, and manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
