Abstract
Herein, we aimed to use double-labeling immunofluorescence to describe the expression pattern of Calbindin-D28K (CaBP28K) in the mouse cochlea from late embryonic (E) stages to the adulthood. CaBP28K was expressed in the inner hair cells (IHCs) and the greater epithelial ridge (GER) at E17. In addition, its expression was observed in the interdental cells. On postnatal day 1 (P1), CaBP28K immunoreactivity was observed in the IHCs and outer hair cells (OHCs) and was also specifically expressed in the nucleus and the cytoplasm of spiral ganglion neurons (SGNs). At P8, CaBP28K labeling disappeared from the interdental cells, and the CaBP28K-positive domain within the GER shifted from the entire cytoplasm to only the apical and basal regions. At P14, CaBP28K immunoreactivity was lost from the GER; however, its expression in the IHCs and OHCs, as well as the SGNs, persisted into adulthood. The identification of CaBP28K in the hair cells (HCs) and cuticular plates, as well as SGNs, was confirmed by its colocalization with several markers for Sox2, Myosin VIIa, Phalloidin, and Tuj1. We also detected colocalization with calmodulin in the cytoplasm of both HCs and SGNs. Western blot revealed an increase in CaBP28K postnatal expression in the mouse cochlea.
Introduction
Calbindin-D28K (CaBP28K) is a Ca2+-binding protein that belongs to the calmodulin superfamily. 1 Dysfunction in Ca2+-binding protein-mediated voltage-gated calcium channels has been associated with various human diseases. 2 It has been suggested that CaBP28K has several roles in hearing, including cochlear micromechanics, auditory transduction, and inner ear ontogeny. 3 Alternations of CaBP28K expression in the cochlear cells were reported to be involved in hearing impairment caused by ototoxic drugs, 4 aging, and thyroid hormone deficiency.5,6 Furthermore, changes in the distribution of CaBP28K-labeled neuronal subpopulations in the central auditory system were linked to hearing loss in circling mice as well as hearing loss after acoustic overstimulation.7–9 Although CaBP28K is an extensively studied Ca2+-binding protein in the cochlea, the exact physiological function of CaBP28K remains unclear. The comprehensive description of CaBP28K immunolocalization before and after the onset of hearing further elucidates its role in cochlear function. Previous immuno-peroxidase immunohistochemistry studies revealed that it was widely expressed in the developing cochleas of various species, including humans.6,10,11 It was also reported to be mainly present in auditory sensory hair cells (HCs) and cochlear neuronal structures. However, these studies have poorly identified the nature of the CaBP28K-immunolabeled cells at early developmental stages, and some earlier reports have observed its preferential localization to the cuticular plate region of neonatal HCs 12 ; yet, this intracellular distribution has to be confirmed. In addition, there is some discrepancy in the reported immunohistochemical studies of CaBP28K in the cochlea. For example, Buckiová and Syka 13 found that in the mouse organ of Corti, CaBP28K was expressed in supporting cells, which seemed contradictory to the widely held view of CaBP28K as a marker of inner and outer hair cells (IHCs and OHCs). 14 In addition, Pack and Slepecky 15 reported that in both gerbil and guinea pig, it was present in some, but not all, of the HCs.
The aim of the present study was to re-investigate the expression pattern of CaBP28K on mouse cochlear cross-sections at different developmental stages. We colabeled CaBP28K with Myosin VIIa (HCs marker), Sox2 (supporting cells marker), Phalloidin (F-actin marker), and Tuj1 (beta-III tubulin, neuronal marker) in combination with confocal laser microscopy to determine the precise identity of CaBP28K-immunopositive cells and obtain more details on its localization in specific cell types. In addition, dual-staining with calmodulin, a ubiquitously expressed Ca2+-binding protein in a eukaryotic cell, 16 was performed on cryostat sections of the mouse cochlea to further investigate the expression pattern of CaBP28K and to study its possible colocalization with other Ca2+-binding proteins in the cochlea. Moreover, Western blot analysis was used for a temporal study of CaBP28K expression in mouse cochlea during postnatal development. The findings of the present study could help researchers better understand the role of CaBP28K in the cochlea.
Materials and Methods
Animals
All animal studies (including the mice euthanasia procedure) were done in compliance with the regulations and guidelines of Southeast University institutional animal care and conducted according to the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) and the Institutional Animal Care and Use Committee (IACUC) guidelines. After mating, female BALB/c mice were checked daily for a vaginal plug; the first day a plug was seen was defined as embryonic (E) day 1. Individual embryos were staged according to standard mouse development tables.
Immunofluorescence
Intraperitoneal injection of 10% chloral hydrate (0.2 mL/100 g) was used to anesthetize pregnant (gestational days 17–19) and postnatal (P1–P28) BALB/c mice. Normal saline and ice-cold 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4) were given to postnatal mice via transcardial injection. Embryonic mice were quickly decapitated, and their cochleae were dissociated after being removed from the uterus. The detailed methods and procedures of immunofluorescent staining have been described in previous publications. 17 Briefly, the round and oval windows of the cochlea were opened, and a small hole was made in the bony apex. After perilymphatic perfusion with the above fixative, cochlea was postfixed in the same fixative for 35 min at room temperature. The cochlea of mice older than P5 was decalcified in 10% EDTA at pH 7.4. The duration of decalcification of mouse cochlear tissues is shorter than that of rat cochlea, so on average, 2 hr of decalcification at 4C were enough for the P8 mouse cochlea to obtain the fine structural details of the cochlear tissues. The decalcified mouse cochleae were soaked in the 0.01 M PBS for 5 min to minimize background. After that, the cochleae were immersed in a sucrose gradient of 15% for 3 hr and 30% overnight. Next, the cochlear tissue was embedded in an optimum cutting temperature compound at 4C for 2.5 hr, rapidly frozen at –20C, and cryosection was performed using a Leica cryostat microtome generating 6 μm sections. Cochlear cross-sections were treated for 30 min with 10% donkey serum and 0.3% Triton X-100 in PBS at room temperature to make cell membranes more permeable to antibodies. Then sections were incubated overnight at 4C with rat anti-CaBP28K monoclonal antibody (dilution 1:200, #13176, Cell Signaling Technology, USA). In double-labeling experiments, anti-CaBP28K monoclonal antibody was incubated with the primary antibodies raised in different species, respectively: goat anti-Sox2 polyclonal antibody (1:200, #sc-17320, Santa Cruz Biochemicals, USA), mouse anti-Myosin VIIA antibody (1:200; Hybridoma Bank, Iowa City, IA, USA), and mouse anti-Tuj1 monoclonal antibody (1:250, #4466, Cell Signaling Technology, USA). Two monoclonal anti-calmodulin antibodies (namely, 1:20, #GTX22860, GeneTex, USA, and 1:20, #MA3-917, Invitrogen, USA) were used in double-labeling experiments. After rinsing three times for 15 min in the 0.01 M PBS, they were put for 1 hr at 37C in the mixture of the following secondary antibodies: donkey anti-rat IgG conjugated with Alexa 488 (1:250, Yeasen, China), Cy3-labeled donkey anti-goat IgG (1:250, Beyotime, China), donkey anti-mouse IgG conjugated with Alexa 555 (1:400, Beyotime, China), and Coralite594-conjugated Phalloidin antibody (1:250, #PF00003, Proteintech, China). The control sections were incubated with 0.01 M PBS, omitting the primary antibodies. In addition, rabbit (DA1E) monoclonal antibody IgG XP Isotype control (#3900, Cell Signaling Technology, USA) was used as a negative control, instead of CaBP28K antibody. 18 Then, the sections were washed and counterstained with 4′,6-diamidino-2-phenylindole (DAPI; 1:600, Biyuntian, China) for 5 min to visualize cell nuclei. After washing with PBS, fluorescence was preserved by sealing specimens with an antifade mounting medium (Biyuntian, China). Cryostat sections were viewed with a Zeiss (LSM900) laser scanning confocal microscope with ×10 [numerical aperture (NA) = 0.45], ×20 (NA = 0.8), ×40 (NA = 0.95) and ×63 oil (NA = 1.4) objectives at 1024 × 1024 pixels. Zen3.0 acquisition software was used. Immunostaining presented in figures was representative of three individual experiments. Images were cropped and resized using Adobe Photoshop CC 2019.
Western Blotting
The mouse cochlear tissues at P1, P7, P14, and P28 were microdissected, and the cochlear tissues were homogenized with RIPA Lysis Buffer containing 1 mM PMSF. After centrifuging (12,000 rpm, 4C, 30 min), the supernatant was stored at –80C until use. Protein concentrations were determined with a BCA Protein Assay Kit (P0012S, Biyuntian, China). After electrophoresis, the proteins were transferred onto polyvinylidene fluoride membranes (IPVH00010, Millipore, USA), which were blocked with 5% non-fat dried milk for 1 hr at room temperature, and then incubated in monoclonal rabbit anti-CaBP28K antibody (1:1000, #13176, Cell Signaling Technology, USA) and anti-GAPDH (glyceraldehyde-3-phosphate dehydrogenase) antibody (1:1000, #10494-I-AP, Yeasen, China) and were used as an internal loading control. After rinsing three times (5 min each) with TBST, the membrane was incubated with horseradish peroxidase-conjugated anti-rabbit and anti-mouse IgG (1:5000, #SE134 and SE131, Solarbio, China) for 1 hr at room temperature. Following washout with TBST, the membrane was developed using enhanced chemiluminescence reagents (P10300, NCM Biotech, China) and visualized using a chemiluminescence system (Clinx, Shanghai, China). Reaction product levels were quantified by the Quantity One System (Bio-Rad, Hercules, CA, USA).
Statistical Analysis
All statistical analyses were performed using SPSS (version 25.0 IBM). Statistical significance was determined using a one-way analysis of variance, and the Dunnett post hoc test was used to evaluate the statistical differences between groups and P1.
Results
The Expression Patterns of CaBP28K in the Mouse Cochlea During Development by Double-Labeling Immunofluorescence
The greater epithelial ridge (GER) resulted as a significant component of the overall CaBP28K expression in the mouse cochlea during the late embryonic and early postnatal stages of development. At E17 (the earliest stage studied), double-immunofluorescence analysis with Sox2 indicated the position of CaBP28K in the GER and IHCs. The Sox2 is known to label both HCs and support cell nuclei at this stage of development. 19 Except for Sox2-positive expression domain close to the IHCs, CaBP28K was expressed throughout the entire cell of the GER. The GER is a temporary structure in the development of the cochlea that is degraded by the second postnatal week. 20 CaBP28K immunoreactivity in the IHCs was observed in the basal and middle cochlear turn but not in the apical cochlear turn (Fig. 1A and B). There was no detectable immunolabeling in the OHCs in all cochlear turns (Fig. 1C and D). CaBP28K was also not observed in other cochlear structures, except for the interdental cells within the inner sulcus (Fig. 1E). Although immunolabeling for CaBP28K was still absent from IHCs in the apical turn at E19 (Fig. 1F), CaBP28K-stained IHCs in the middle turn could be clearly observed from its colocalization with Myosin VIIa identified CaBP28K in the IHCs (Fig. 1H), CaBP28K was found throughout the IHC body, including basal and apical regions, in a consistent pattern. As previously reported, nonspecific staining, which occurs in anti-mouse IgG antibodies, was observed in the basilar membrane and the spiral vessel underneath the basilar membrane (Fig. 1H). 21 At this age, a nuclear localization in IHCs was particularly obvious. The GER and the interdental cells continued to express CaBP28K, and the OHC remained CaBP28K-unlabeled (Fig. 1G and I). CaBP28K immunoreactivity was found in both IHCs and three rows of OHCs in all cochlear turns, with the cytoplasm and nucleus immunolabeled until P1 (Fig. 2A to C). Costaining with phalloidin confirmed that it was preferentially localized in the cuticular plates of HCs (Fig. 2D to F). Phalloidin is a well-known marker for F-actin-rich cellular structures, including stereocilia and cuticular plates. 22 The immunodistribution of CaBP28K in the GER and the interdental cells was retained. CaBP28K immunostaining with nuclear labeling was found in a subpopulation of primary sensory neurons within Rosenthal’s canal, while the other neurons were not immunoreactive, as confirmed by double immunofluorescence with Tuj1 (Fig. 2G to I). Tuj1 is a well-established afferent marker for both type I and type II spiral ganglion neurons (SGNs) during the early stages of cochlear development. 23 Double-labeling results showed that CaBP28K was colocalized with Tuj1 in a subpopulation of SGN somata, and no colocalization of CaBP28K and Tuj1 was detected in the nucleus of SGNs. At P5, there was no evidence of an expression gradient. Our results demonstrated the distribution of CaBP28K in the cochlear middle turn and represented all turns. CaBP28K continued to be expressed in the GER, HCs, and cuticular plates of the HCs, whereas labeling for CaBP28K was absent from the HC nuclei (Fig. 3A to C). Most of the somas of CaBP28K-immunopositive SGNs were double-labeled for Tuj1, and nuclei were stained (Fig. 3D to F). At P8, when the GER started to regress, changes in the CaBP28K-immune distribution were found in the GER, particularly in the medial portion close to the spiral limbus. Immunolabeling for CaBP28K was concentrated in the apical and basal surface of the GER and extended to the inner sulcus. CaBP28K expression disappeared from the interdental cells (Fig. 3G and H). CaBP28K immunolabeling was retained in the cytosol and the nucleus of SGNs (Fig. 3I). CaBP28K was only found in the apical surface of the remnant GER cells at P10 (Fig. 4A), but it was widely distributed throughout the HCs body. In the cuticular plates, CaBP28K almost completely overlapped with the F-actin cytoskeleton with phalloidin (Fig. 4B and C). At the onset of hearing (P14), the GER simultaneously disappeared with the loss of CaBP28K expression from this structure. The immunostaining pattern was similar to that seen in the cochlea of the adult mouse. CaBP28K immunostaining was limited to the IHCs and OHCs, as well as the cuticular plates (Fig. 4D to F). Dual-staining of the two calcium-binding proteins in the mouse cochlea, CaBP28K and calmodulin, was also carried out in the present study. Using two monoclonal anti-calmodulin antibodies, we achieved the same pattern of immunoreactivity in the mouse cochlear cross-sections. During the prehearing developmental phase, we failed to yield specific staining using both antibodies with an optimized immunofluorescence procedure. Not until P14 did anti-calmodulin antibodies give satisfactory fluorescent staining in the mouse organ of Corti (Fig. 5A to C), possibly due to the decrease in artifactual staining that occurred with mice antibodies in later development. 22 Specific labeling for calmodulin was demonstrated in the stereociliary bundles of P14 and P21 HCs (Fig. 5D to F), which is consistent with the notion that hair bundles rely on mobile Ca2+ buffers to regulate Ca2+ levels in stereocilia. 24 An overlapping distribution of CaBP28K and calmodulin was found in the cytoplasm of HCs and persisted into adulthood. In the cochlea of adult (P28) mice, there were no changes in staining patterns in HCs and SGNs. Coexpression of CaBP28K with Myosin VIIa and F-actin was maintained in the HCs cytoplasm (Fig. 6A to C) and cuticular plates (Fig. 6D to F). Given that only Tuj1 is strongly expressed in Type I SGNs in the adult mouse, 25 many (but by far not all) of the cytoplasm of CaBP28K-positive SGNs were double-stained for Tuj1, where some scattered SGNs appeared to express only CaBP28K, indicating that possibly both type I and type II SGNs were labeled for CaBP28K (Fig. 6G to I). Similarly, calmodulin expression was restricted to the HCs of the adult organ of Corti and SGNs, and no immunoreactivity of calmodulin was detected in the supporting cells and other regions (Fig. 7A and B). A control for nonspecific binding of anti-rabbit and anti-mouse IgG antibodies in a double-labeling experiment showed no staining (Fig. 7C), and the rabbit monoclonal antibody IgG isotype control used to replace the primary antibody showed negative results (data not shown). Calmodulin immunoreactivity persisted in the adult stereociliary bundle (Fig. 7D to F). Costaining of CaBP28K and calmodulin revealed that while most or all SGNs were immunoreactive for calmodulin, only a subpopulation of calmodulin-labeled SGN somata was coexpressed with CaBP28K, and there was no colocalization in the nucleus of SGNs (Fig. 7G to I).
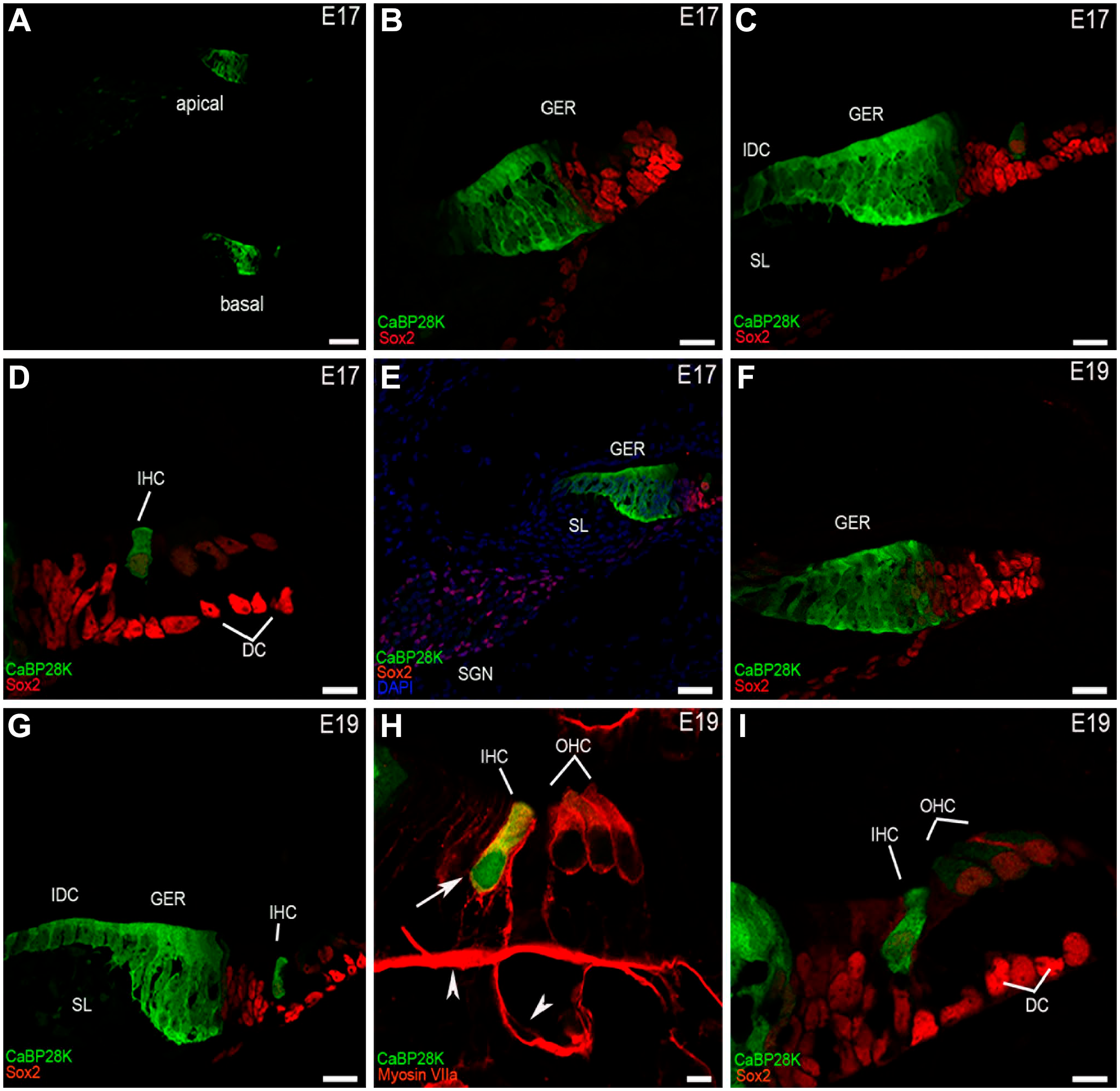
CaBP28K immunolabeling in the mouse cochlea at E17 and E19. (A) A low-magnification view of the cross-sections of the mouse cochlea labeled with CaBP28K (green) at E17. CaBP28K labeling was mainly present in the GER and IHCs, no other structure was labeled in the mouse cochlea. Scale bar, 50 μm. (B) CaBP28K (green) and Sox2 (red) labeling in the apical turn of the mouse cochlea at E17. CaBP28K was localized in the cytosol of columnar epithelial cells of the GER, a portion of the GER that exhibited the nuclear staining pattern of Sox2 was not stained. Scale bar, 20 μm. (C) CaBP28K (green) and Sox2 (red) labeling in the middle turn of E17. The expression of CaBP28K emerged in the IHCs and the interdental cells in the spiral limbus, in addition to CaBP28K-labeled GER. Sox2-expressing cells localized in the most external part of the GER were immunonegative for CaBP28K. Scale bar, 20 μm. (D) Detail of CaBP28K labeling in the organ of Corti in the middle turn of E17. Sox2 labeled hair cell and supporting cell nuclei, Sox2-stained nucleus indicated the position of hair cells. CaBP28K was observed in the cytosol of IHC, the OHCs above three rows of the Deiters’ cell were not stained. Scale bar, 10 μm. (E) A low-magnification view of CaBP28K labeling in the medial turn of E17. No CaBP28K expression was observed in the SGNs and Sox2-labeled glial cells of Rosenthal’s canal. (F) CaBP28K (green) and Sox2 (red) labeling in the apical turn of the mouse cochlea at E19. IHCs remained unlabelled. Scale bar, 20 μm. (G) CaBP28K (green) and Sox2 (red) labeling in the middle turn of E19. CaBP28K immunostaining was observed in the GER, the IHCs, and the interdental cells. Scale bar, 20 μm. (H) Detail of E19 mouse organ of Corti double-labeled by CaBP28K (green) and Myosin VIIa (red). Colocalization of CaBP28K with Myosin VIIa in the IHC cytosol (yellow) could be detected in the merged image. The nucleus of IHCs (large arrow) showed labeling for CaBP28K. Non-specific staining was observed in the basilar membrane and the spiral vessel underneath the basilar membrane (arrowheads). Scale bar, 5 μm. (I) Detail of CaBP28K labeling in the organ of Corti in the basal turn at E19. The expression of CaBP28K in three rows of OHCs was marginally above the background level. Scale bar, 10 μm.Abbreviations: GER, greater epithelial ridge; IHC, inner hair cell; SGN, spiral ganglion neuron; SL, spiral limbus; IDC, the interdental cells; DC, Deiters cells; OHC, outer hair cell.
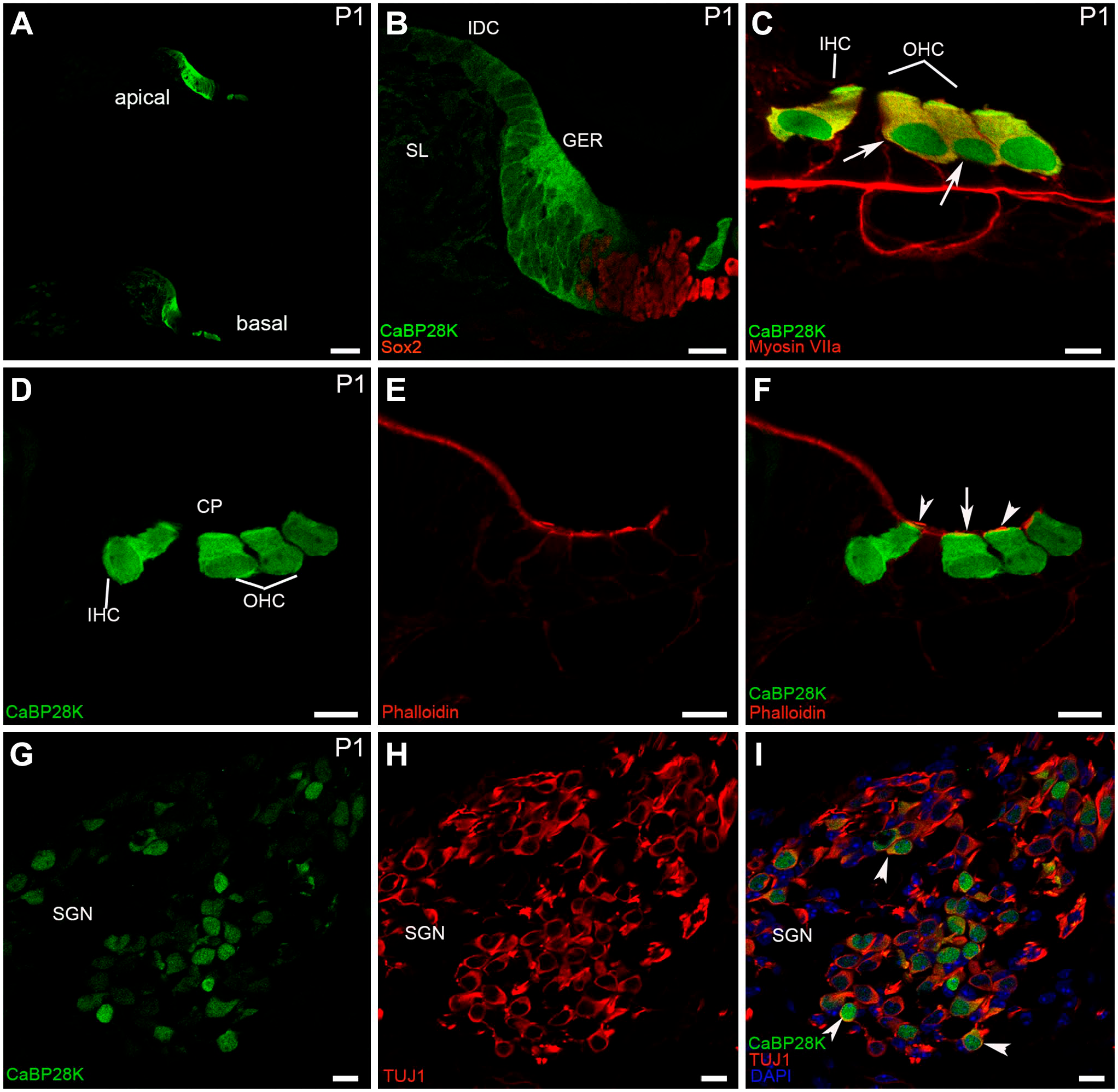
CaBP28K immunolabeling in the mouse cochlea at P1. (A) A low-magnification view of the cross-sections of the mouse cochlea labeled with CaBP28K (green) at P1. The expression of CaBP28K emerged in the IHCs and three rows of OHCs in the apical turn of the P1 mouse cochlea. Scale bar, 50 μm. (B) Detail of CaBP28K (green) labeling in the GER in the middle turn of P1. CaBP28K was not detectable in a few columnar inner supporting cells located medially to the IHCs. CaBP28K immunoreactivity was maintained in the interdental cells. Scale bar, 20 μm. (C) Detail of CaBP28K (green) labeling in the P1 organ of Corti. CaBP28K immunoreactivity was present in both the cytoplasm (yellow) and the nucleus of IHC and OHCs (large arrow), as demonstrated by co-labeling with Myosin VIIa (red). Scale bar, 10 μm. (D, E, F) Detail of CaBP28K (green) and phalloidin (red) expression in the P1 organ of Corti. Colocalization of CaBP28K with phalloidin was found in the cuticular plates (large arrow) of HCs, CaBP28K was not observed in the phalloidin-positive stereociliary bundle (red) of HCs (arrowheads). Scale bar, 10 μm. (G, H, I) Detail of CaBP28K (green) and Tuj1 (red) expression in the P1 SGNs and the merged image+DAPI. CaBP28K immunolabeling was present in a subpopulation of scattered SGNs. Both the cytoplasm and the nucleus of SGNs were stained. In CaBP28K-Tuj1 double-positive SGNs, CaBP28K labeling was colocalized with Tuj1 in the cytoplasm (arrowheads). Abbreviations: GER, greater epithelial ridge; IHC, inner hair cell; OHC, outer hair cell; SGN, spiral ganglion neuron; CP, cuticular plates; SL, spiral limbus; IDC, the interdental cells.
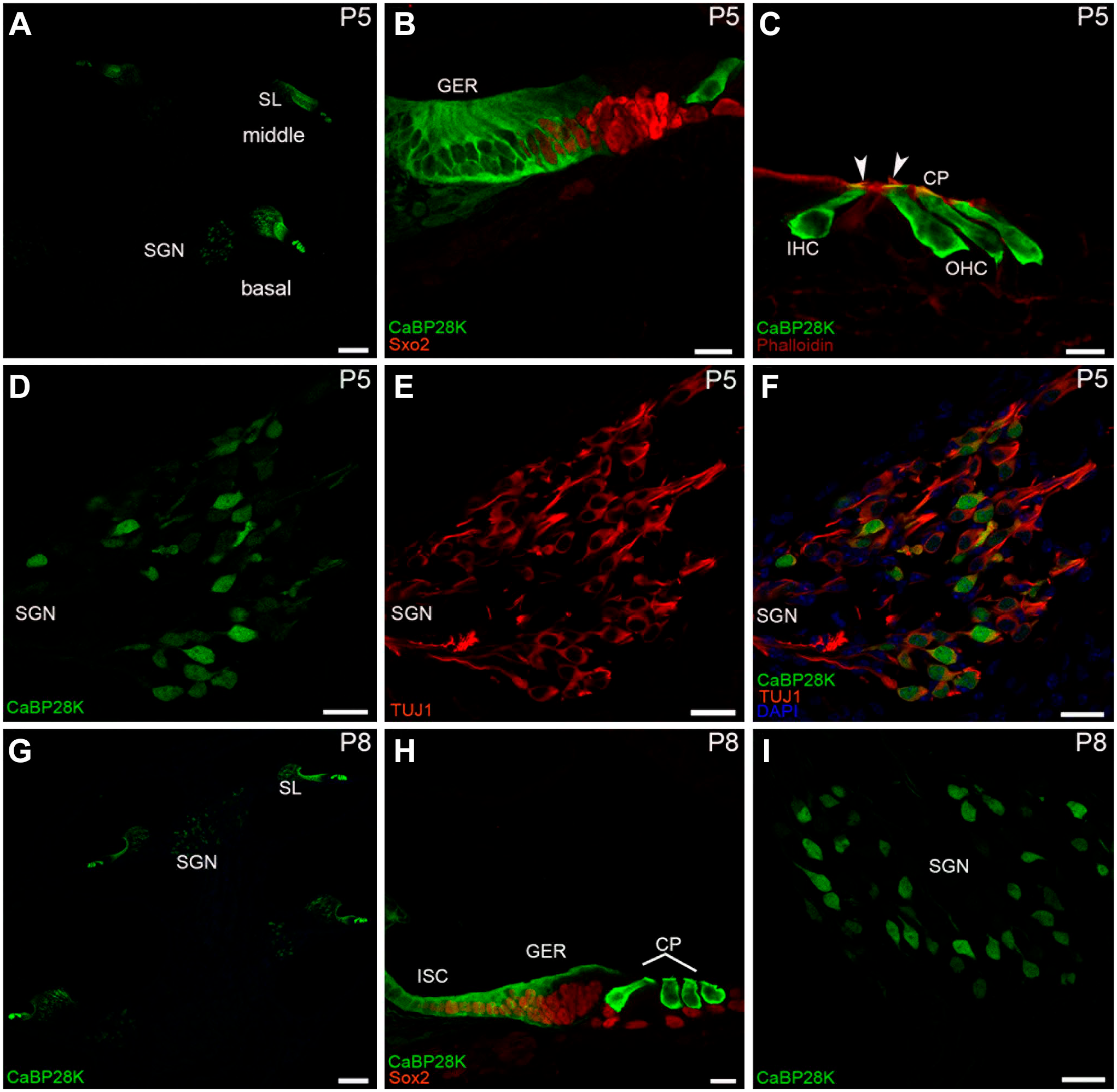
CaBP28K immunolabeling in the mouse cochlea at P5 and P8. (A) A low-magnification view of CaBP28K (green) labeling in the P5 cochlear section. Scale bar, 100 μm. (B) Detail of CaBP28K (green) labeling in the GER of P5. Sox2 (red) expression domain extended medially into the medial portion of the GER. Scale bar, 20 μm. (C) Detail of CaBP28K (green) and phalloidin (red) expression in the P5 organ of Corti. CaBP28K was co-labeled with phalloidin in the cuticular plates (yellow) of HCs. Phalloidin was clearly visible in the stereociliary bundles (arrowheads) of HCs. CaBP28K immunoreactivity was lost from hair cell nuclei. Scale bar, 10 μm. (D, E, F) Detail of CaBP28K (green) and Tuj1 (red) expression in the P5 SGNs and the merged image+DAPI. Most of the CaBP28K-stained SGN somata were colabeled with Tuj1, and the nucleus of SGNs expressed only CaBP28K (green). Scale bar, 10 μm. (G) An overview of CaBP28K (green) labeling in the cochlear section at P8. Labeling for CaBP28K was absent from the interdental cells. Scale bar, 100 μm. (H) Detail of CaBP28K (green) labeling in the P8 organ of Corti. Note that the thickened ridge of the GER has regressed, CaBP28K expression was restricted to the apical and basal parts of the GER, the inner sulcus started expressing CaBP28K. Scale bar, 10 μm. (I) Detail of CaBP28K (green) labeling in the P8 SGNs. CaBP28K expression remained in SGN somata and nuclei. Scale bar, 10 μm. Abbreviations: SGN, spiral ganglion neuron; GER, greater epithelial ridge; IHC, inner hair cell; OHC, outer hair cell; CP, cuticular plates.
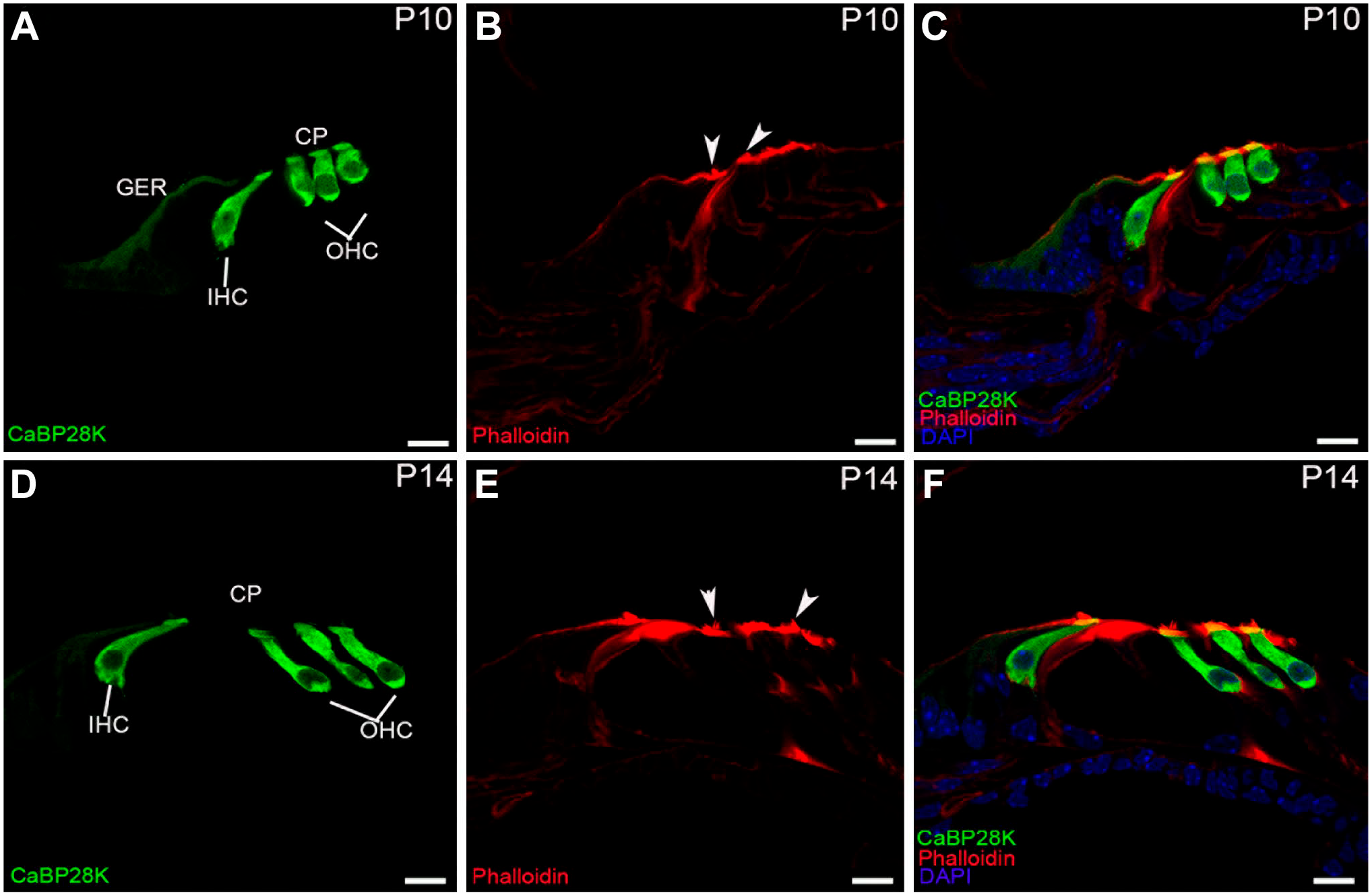
Detail of CaBP28K (green) and phalloidin (red) expression in the P10 and P14 mouse organ of Corti. (A, B, C) Colocalization of CaBP28K with phalloidin in the cuticular plates (yellow) of P10 HCs, CaBP28K immunostaining was limited to the apical surface of the residual GER cells. CaBP28K and phalloidin were clearly colocalized in the cuticular plates (yellow) of P10 HCs. No immunoreactivity for CaBP28K was observed in the stereociliary bundles (arrowheads). (D, E, F) Colocalization of CaBP28K with phalloidin in the cuticular plates (yellow) of P14 HCs. CaBP28K completely disappeared from the GER, the stereociliary bundles (arrowheads) were still unstained. Scale bar, 10 μm. Abbreviations: GER, greater epithelial ridge; IHC, inner hair cell; OHC, outer hair cell; CP, cuticular plates
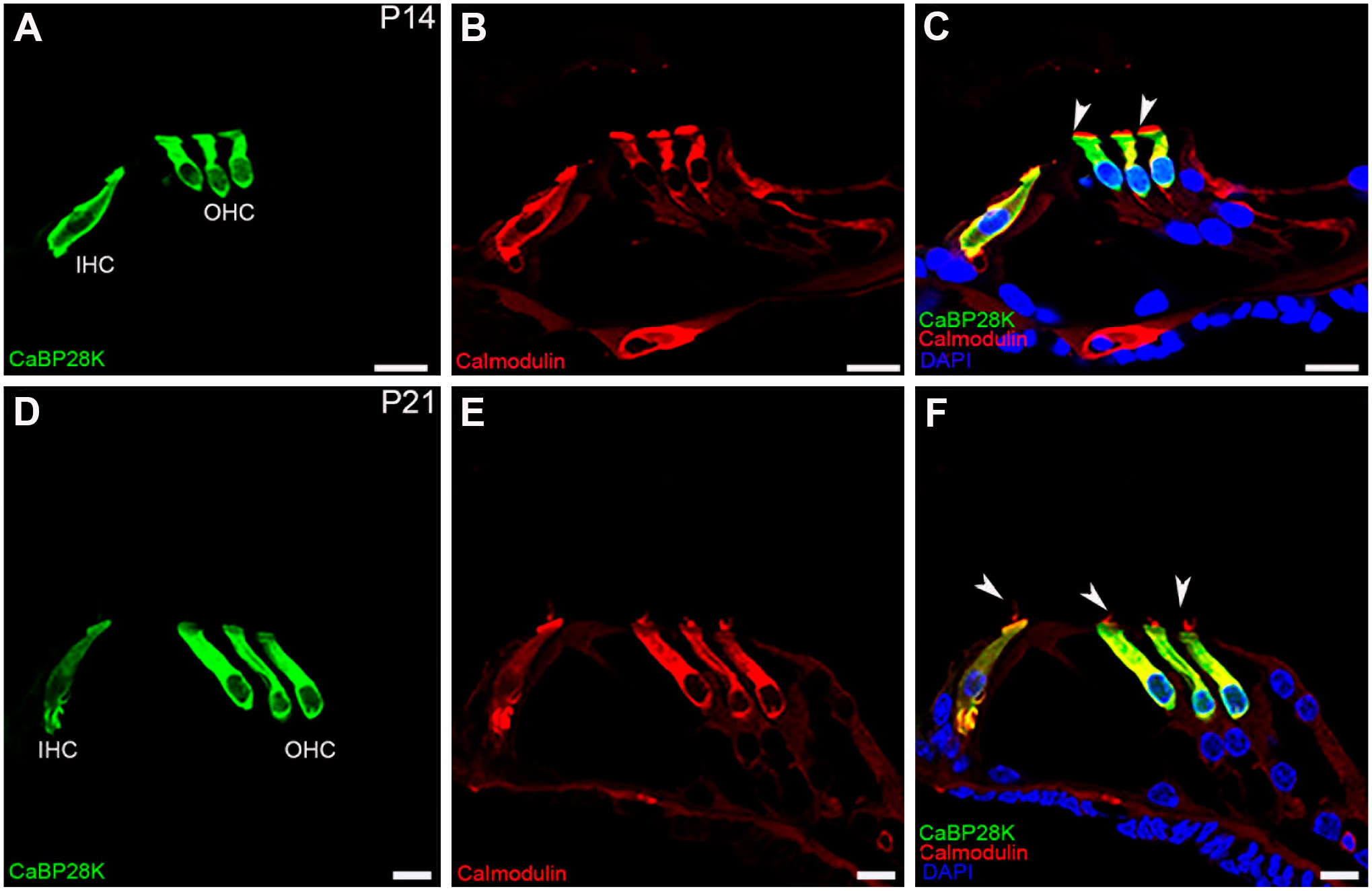
Double-immunofluorescence staining with CaBP28K (green) and calmodulin (red) in P14 and P21 mouse organ of Corti. (A, B, C) Co-expression of CaBP28K and calmodulin was detected in the cytoplasm (yellow) of P14 HCs. Note the stereociliary bundles (arrowheads) were labeled with calmodulin (red) but not with CaBP28K (green). (D, E, F) At P21, CaBP28K was colocalized with calmodulin in the cytoplasm (yellow) of HCs but not in the stereociliary bundles (arrowheads). Scale bar, 10 μm. Abbreviations: IHC, inner hair cell; OHC, outer hair cell
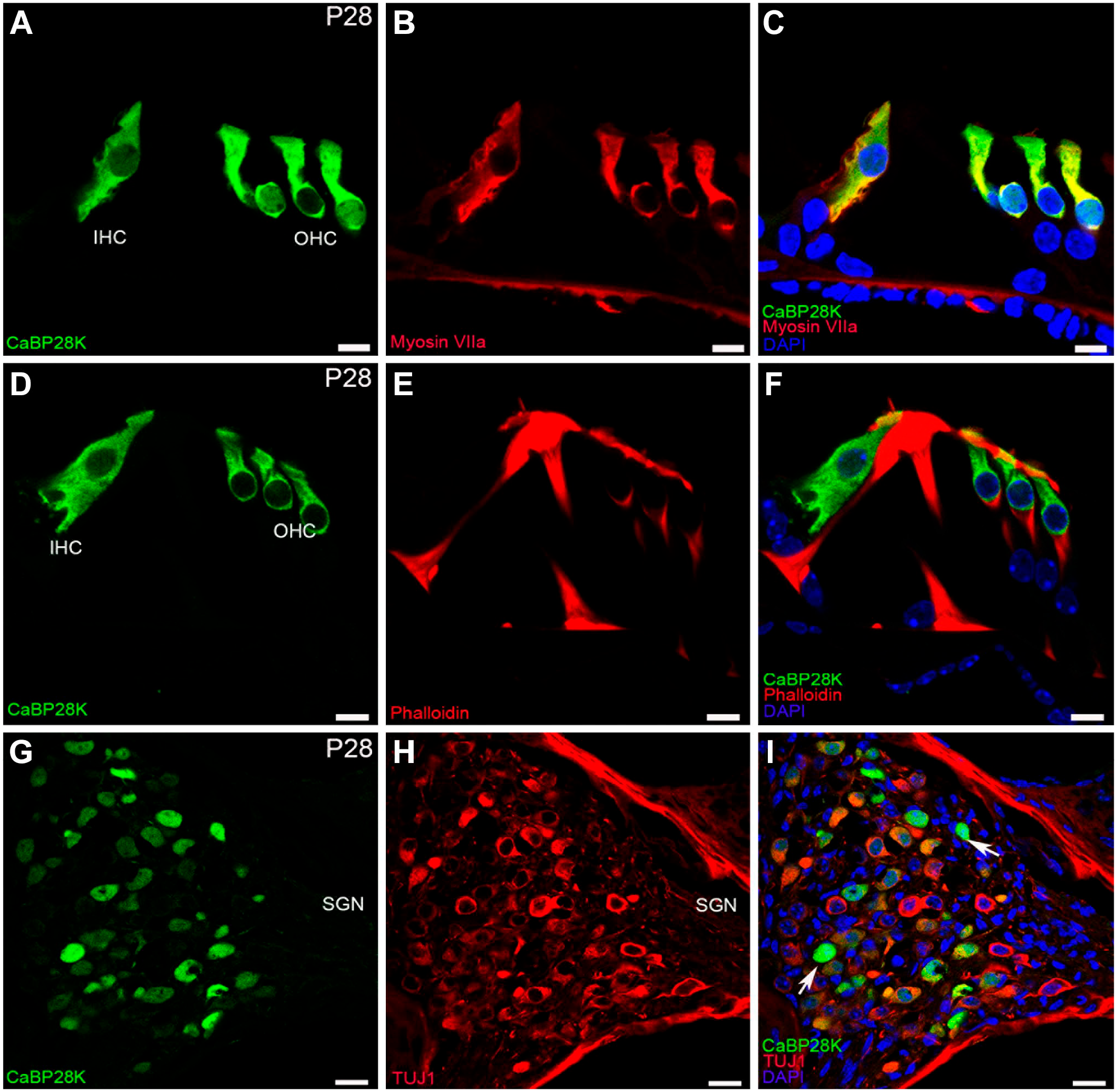
CaBP28K immunolabeling in the mouse cochlea at P28. (A, B, C) Colocalization of CaBP28K (green) with Myosin VIIa (red) in the cytoplasm (yellow) of P28 HCs. Scale bar, 10 μm. (D, E, F) Colocalization of CaBP28K (green) with phalloidin (red) in the cuticular plates (yellow) of P28 HCs. Scale bar, 10 μm. (G, H, I) Detail of CaBP28K (green) and Tuj1 (red) expression in the P28 SGNs and the merged image+DAPI. Many, but not all, CaBP28K-positive the cytoplasm of SGNs were double-labeled for Tuj1, and a few CaBP28K-positive SGNs somata (arrows) were not co-labeled with Tuj1.Abbreviations: IHC, inner hair cell; OHC, outer hair cell; SGN, spiral ganglion neuron.
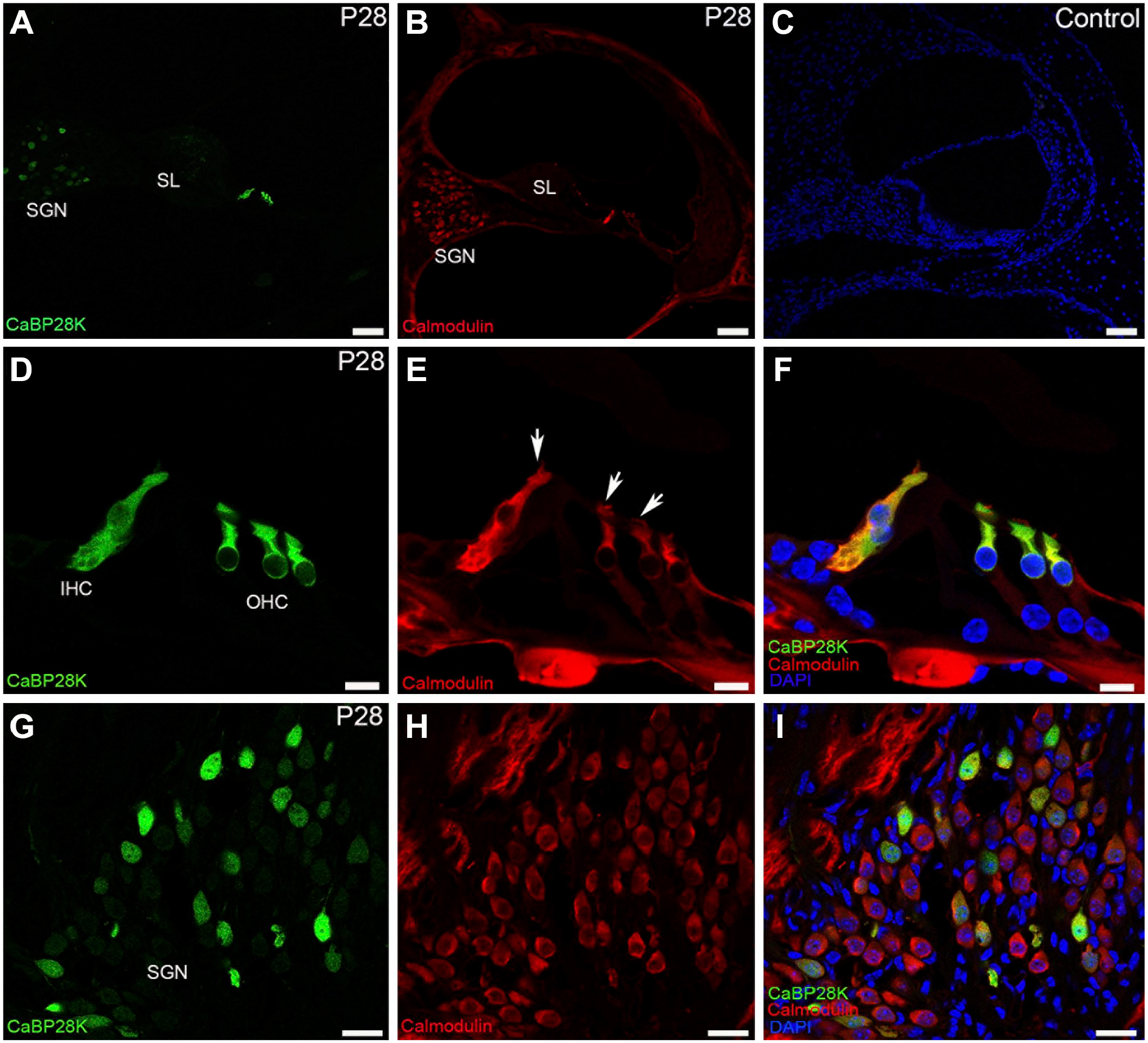
Double-immunofluorescence staining with CaBP28K (green) and calmodulin (red) in adult (P28) mouse organ of Corti. (A, B) An overview of CaBP28K (green) and calmodulin (red) labeling in the middle turn of the adult cochlea. Scale bar, 50 μm. (C) The secondary antibodies showed no non-specific binding. Scale bar, 50 μm. (D, E, F) Co-expression of CaBP28K (green) and calmodulin (red) was detected in the cytoplasm of adult HCs. Scale bar, 10 μm. (G, H, I) Detail of calmodulin expression in the adult SGNs. Among the calmodulin-positive SGNs, only a subpopulation of calmodulin-positive SGN somata was dually stained (yellow), SGN nuclei were unstained. Scale bar, 20 μm.Abbreviations: SGN, spiral ganglion neuron; IHC, inner hair cell; OHC, outer hair cell;SL, spiral limbus.
The Protein Expression of CaBP28K in the Mouse Cochlea by Western Blot Analysis
The monoclonal anti-CaBP28K antibody recognized a band in mouse cochlea with a molecular weight of about 28 kDa. These findings demonstrated that this antibody was suitable for immunohistochemistry. Statistical analysis of CaBP28K protein levels in the mouse cochlea at P1, P7, P14, and P28 (Fig. 8) revealed differences in the expression of CaBP28K between P1 compared with P7, P14, and P28 (all
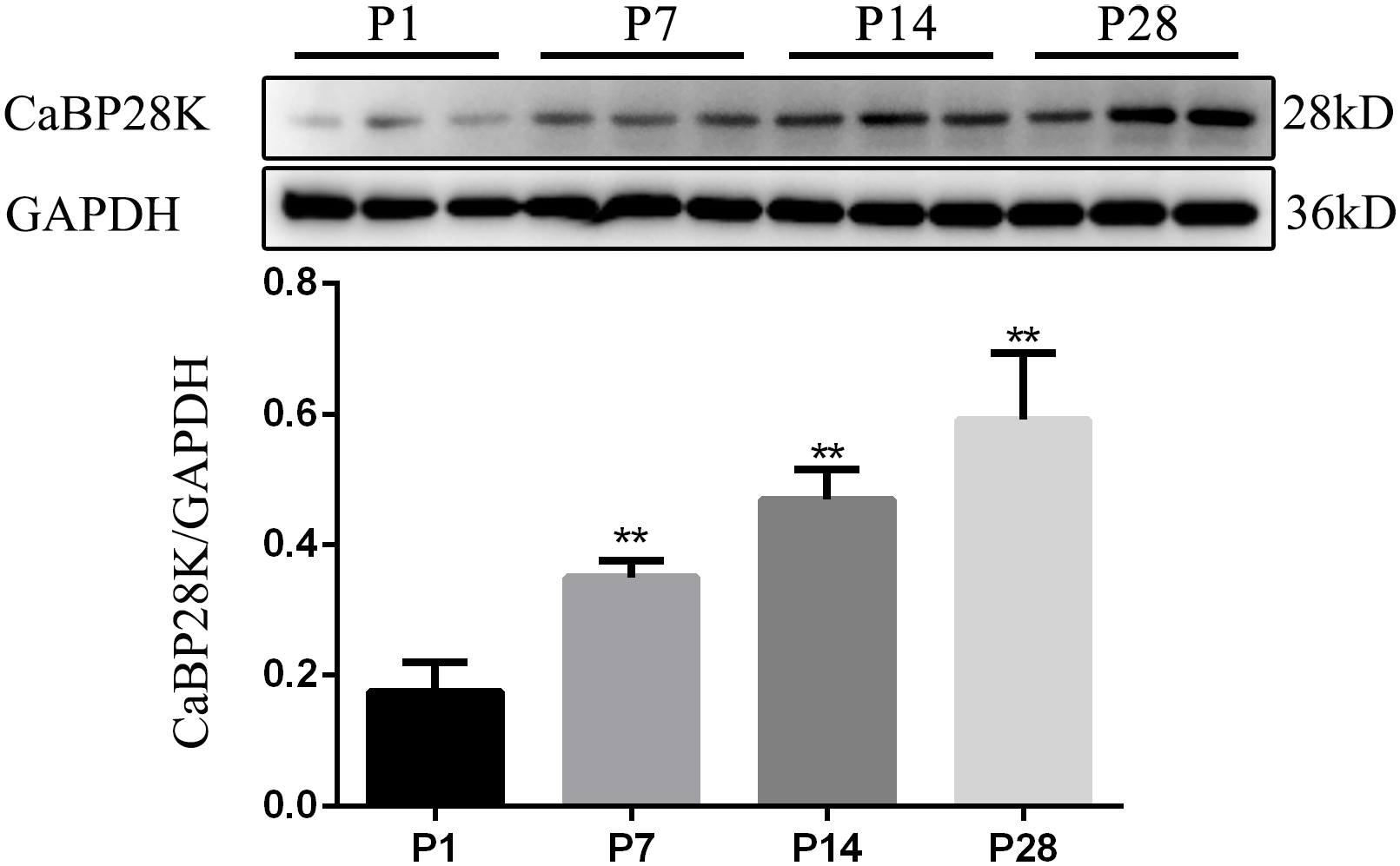
Statistical analysis of CaBP28K protein levels in the mouse cochlea at P1, P7, P14 and P28. GAPDH, used as an internal control, was detected at a position corresponding to a molecular weight of 36 kDa. There were difference in expression of CaBP28K at the various developmental stages (
Discussion
In the present study, we confirmed and extended previous reports of the spatiotemporal distribution of CaBP28K in the cochlea of developing mice by double immunofluorescence. As previously described, in the rodents (mouse, rat), CaBP28K immunoreactivity occurred in late embryonic mouse cochlea and showed age-related changes during the early stage of cochlear development. Before the onset of hearing, our results showed transient expression in the interdental cells of the spiral limbus and that spatiotemporal-specific changes in the expression of CaBP28K in the GER were parallel to the development and maturation of the mouse cochlea. It is well known that the morphological changes within the GER that eventually disappear and form the inner sulcus characterize prehearing mammalian cochlear development. 26 To the best of our knowledge, this is the first study that performed double-labeling immunofluorescence to further clarify CaBP28K localization in the GER. In particular, at P5, immunolocalization in this structure was detected for the first time in the present study. It has been proposed that the GER and the interdental cells of the spiral limbus may participate in the secretion of the tectorial membrane that is essential for auditory transduction during development.27,28 It has been suggested that prolonged survival of the GER and resultant malformations of the tectorial membrane in the hypothyroid rat model are associated with persistent CaBP28K immunoreactivity. In addition, among the four major Ca2+-binding proteins with EF-hand, including calmodulin, caldendrin, and parvalbumin,29,30 previous studies have identified CaBP28K as the only one found in the GER, thus implying it may have a crucial and independent role during cochlear development. Our Western blot analysis revealed CaBP28K expression for the first time at protein levels, which increased during postnatal development, thus further favoring its involvement in cochlear development. Consistent with the notion that CaBP28K is specific HCs marker, 31 we demonstrated its expression in the HCs in neonatal and mature mouse organ of Corti, as revealed by double-labeling with either Myosin VIIa or phalloidin staining of F-actin. The findings of the current study are also consistent with previous reports of a basal-apical immunostaining gradient in late prenatal IHCs and a relatively late start of CaBP28K expression in the neonatal OHCs,6,10 except for certain stages, that is, between E19 and P1, when CaBP28K showed nuclear and cytoplasmic labeling. The general pattern of CaBP28K immunostaining in HCs showed in this study was cytoplasmic, including the apical and basal cytoplasm, confirming further the notion that CaBP28K was a cytosolic protein. 32 Herein, the onset of CaBP28K immunoreactivity in late embryonic IHCs was verified by costaining with Sox2 and Myosin VIIa, early markers of HC differentiation, 33 indicating a possible association between CaBP28K with the differentiation and development of HCs. The role of CaBP28K in regulating Ca2+ influx and exocytosis in HCs was described, and it was demonstrated that changes in CaBP28K immunoreactivity occurred in the mouse HCs as early as P5,34,35 acting as an endogenous compensatory mechanism, and participating in the otoprotective effect of pituitary adenylate cyclase-activating polypeptide. We found CaBP28K expression in the HCs at P8 when motility of the OHCs appeared, and the HCs became functional. Furthermore, our findings of CaBP28K expression in the cuticular plates of HCs may facilitate the investigation of its physiological functions. Various cytoskeletal contractile proteins, such as actin and myosin, are known to be present in the cuticular plates.36,37 Under the control of Ca2+-binding proteins, actin has been proposed to participate in regulating the stiffness of cuticular plates. 38 It was reported that noise could cause a decrease in F-actin labeling in the cuticular plate and the resultant decreased mechanical rigidity of the organ of Corti. Our confocal data revealed for the first time that CaBP28K and actin colocalized in the cuticular plates of both HCs in all studied postnatal stages, implying a possible interaction between the two proteins in hearing processes. Cuticular plate immunostaining has been reported to be used to evaluate HC loss, 39 and loss of HCs was proposed to be involved in the pathogenesis of hearing loss associated with traumatic lesions, such as acoustic trauma, 40 ototoxicity, 41 low-dose gamma irradiation, and endolymphatic hydrops.42,43 Accordingly, it is tempting to suggest that immunostaining for CaBP28K could serve as a useful tool for the study of early-onset changes in cytochemistry of sensory HCs under pathological conditions. The mammalian cochlea has been reported to colocalize CaBP28K with other Ca2+-binding proteins, 44 To the best of our knowledge, the present study is the first that investigated the possible colocalization of CaBP28K and calmodulin on the cochlear cross-sections; we also detected calmodulin’s presence in the stereocilia bundles of HCs, where no CaBP28K staining was detected at any time point by our method, suggesting the two proteins have different functions in HCs. The coexpression of CaBP28K and calmodulin in the cytoplasm of HCs likely reflects their overlapping roles.
We provided the first consistent morphological evidence for its presence in many, but not all, of SGNs and nuclear and cytoplasmic CaBP28K labeling by costaining with the neural marker in the neuronal structures of the mouse cochlea. CaBP28K colabeling with calmodulin in the SGN somata, but not the nucleus, revealed that CaBP28K is present in the nucleus. Moreover, CaBP28K has been found in the nucleus and cytoplasm of different neuronal subpopulations in the rodent brain.45,46 Both cytosolic and nuclear Ca2+ were involved in neuronal differentiation and development, particularly its nuclear localization within the nerve cells, which is an important functional region for CaBP28K, relevant to the possible calcium-regulated gene expression. 47 A neuroprotective role has been suggested for CaBP28K-containing neurons in the brain.48,49 It has also been postulated that upregulation of CaBP28K expression by SGNs of aging mice protected neurons against age-related hearing loss. Although the exact function of CaBP28K in the SGNs remains unknown at this time, based on its roles in other types of neurons, early and consistent expression of CaBP28K in the SGNs, and its specific distribution in cell nuclei, it is reasonable to believe that CaBP28K may have similar important developmental roles in the SGNs as also suggested by previous studies in the central nervous system.
This colocalization study used specific cell markers and confirmed previously reported specific expression of CaBP28K in the developing and adult HCs and SGN populations, where its preferential intracellular localization was shown in the cuticular plates and the nucleus of SGNs. The distribution of CaBP28K and calmodulin in HCs and SGNs showed a partial overlap. The GER and spiral limbus showed developmental changes in CaBP28K expression and cellular distribution during the early postnatal period. Western blots also revealed temporal variation in the expression of CaBP28K in the mouse cochlea. Our findings may further the understanding of the role of CaBP28K in auditory development and function.
Footnotes
Acknowledgements
We thank Dr. Renjie Chai and Bin Wang for their support of this work.
Author Contributions
WL conceived, carried out the study, and wrote the article. HC and XZ aided in analyzing data. XZ and HY helped to edit and revise the manuscript. All authors read and approved the final manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (No. 82000987) and Natural Science Foundation of Jiangsu Province (No. BK20200394).
