Abstract
The liver has a unique ability to recover from injury unlike any other organ. A poorly understood aspect of liver regeneration is the role of hepatocellular polarization. Neighbor of Punc E11 (Nope) is an oncofetal stem/progenitor cell marker, which is expressed by depolarized adult hepatocytes after cholestatic liver injury and in hepatocellular carcinoma. Liver injury induced by a choline-deficient and ethionine-supplemented diet is reversible if followed by an additional dietary stop interval and enabled us to study the expression of Nope during the induction of chronic liver injury and during subsequent liver regeneration. We could show by quantitative RT-PCR, Western blotting, and immunohistochemistry that the expression of Nope is induced in depolarized adult hepatocytes during injury. However, after another 2 weeks of a normal diet, the polarization of hepatocytes was almost completely restored and the expression of Nope remained limited to bile ducts and oval cells. Using an inducible CK19-lineage tracing model, we could demonstrate that oval cell–mediated hepatocyte regeneration is rare and was preceded by repolarization of hepatocytes. In conclusion, polarization of hepatocytes is an important part of liver regeneration and precedes oval cell–mediated regeneration of the liver. This process can be visualized by a characteristic expression pattern of Nope:
Introduction
The liver has a unique ability to recover from injury unlike any other organ. This process of liver regeneration is orchestrated by a complex network of growth factors, cytokines, and metabolic pathways.1,2 A poorly understood aspect of liver regeneration is the role of hepatocellular polarization. 3 The polarization of hepatocytes is an absolute prerequisite of a fully functioning mature liver. As accepted mouse models of severe human liver diseases such as bile duct ligation (BDL) show, liver dysfunction is often accompanied by loss or irritation of hepatic polarization. 4
Neighbor of Punc E11 (Nope) is a stem/progenitor cell marker that is highly expressed in the liver during fetal development. During the final maturation and hepatocellular polarization of the developing liver within the first 2 weeks after birth, Nope is relocalized to the sinusoidal membrane of hepatocytes and decreases rapidly thereafter. In the adult liver (AL), the expression of Nope is reduced to a minimum level and can only be found in cholangiocytes. 5
In earlier studies, we described the reappearance of Nope during liver regeneration: First, we identified Nope as a marker of oval cells in the 3,5-diethoxycarbonyl-1,4-dihydro-Collidine (DDC) mouse model. 5 More recently, we identified Nope as a marker of hepatocellular depolarization after BDL, showing an inverse correlation to the gap junction protein connexin 26 (Cx26). 6 Although Cx26 loses its expression pattern and its surrounding hepatocytes after this cholestatic liver injury, Nope reappears and is stably expressed on the sinusoidal membrane of hepatocytes. 6 Mislocalization of Cx26 has also been described in neoplastic hepatocytes,7,8 which are known to lose their original polarization. 9 In fact, we have shown that Nope is expressed in murine and human hepatocellular carcinoma (HCC) and may be a useful biomarker.10,11
However, the BDL model and oncogenic mouse models are irreversible and therefore pose a limitation to investigate the role of Nope in the regenerative process after cessation of the liver injury any further.
In this study, we therefore used the well-established mouse model of a choline-deficient and ethionine-supplemented (CDE) diet 12 for induction of chronic liver injury that is reversible if followed by an additional dietary stop interval. As the CDE diet model is also commonly used to analyze liver regeneration by oval cell proliferation, we used an inducible lineage tracing model to coinvestigate whether cytokeratin 19 (CK19)-positive oval cells give rise to Nope-positive hepatocytes.
The aim of our study is to assign a place for the biological process of hepatocellular depolarization and repolarization in the complex system of liver regeneration.
Materials and Methods
Mice
Animals were housed in individually ventilated cages in the mouse facility at the Institute of Pharmacology, University of Cologne, kept under a 12-hr light cycle, and given a regular chow diet (Harlan, diet no.2918) and water ad libitum. Experiments were approved by the State Agency for Nature, Environment and Consumer Protection (LANUV), North Rhine-Westphalia, Recklinghausen, Germany (Protocol No. 84-02.04.2012.A392) and were in accordance with the German Animal Welfare Act as well as the German Regulation for the protection of animals used for experimental purposes or other scientific purposes. The experiments were planned and performed according to the 3Rs concept of reduction, refinement, and replacement.
CDE diet
The CDE diet was applied as chowing pellets (5000 mg DL-ethionine/kg diet in 10 mm sized pellets; Altrom Special feed, Lage, Germany) and replaced the normal food during the time-lapse completely (100%). In the experimental design, two groups (C57BL/6N strain) were compared with adult control mice. Both groups each of at least three animals with mixed sex were fed with the CDE diet for 3 weeks. In the second group, the CDE diet was followed by 2 weeks of a normal diet (Fig. 1A). In addition, one group was fed with the CDE diet for 4 weeks.
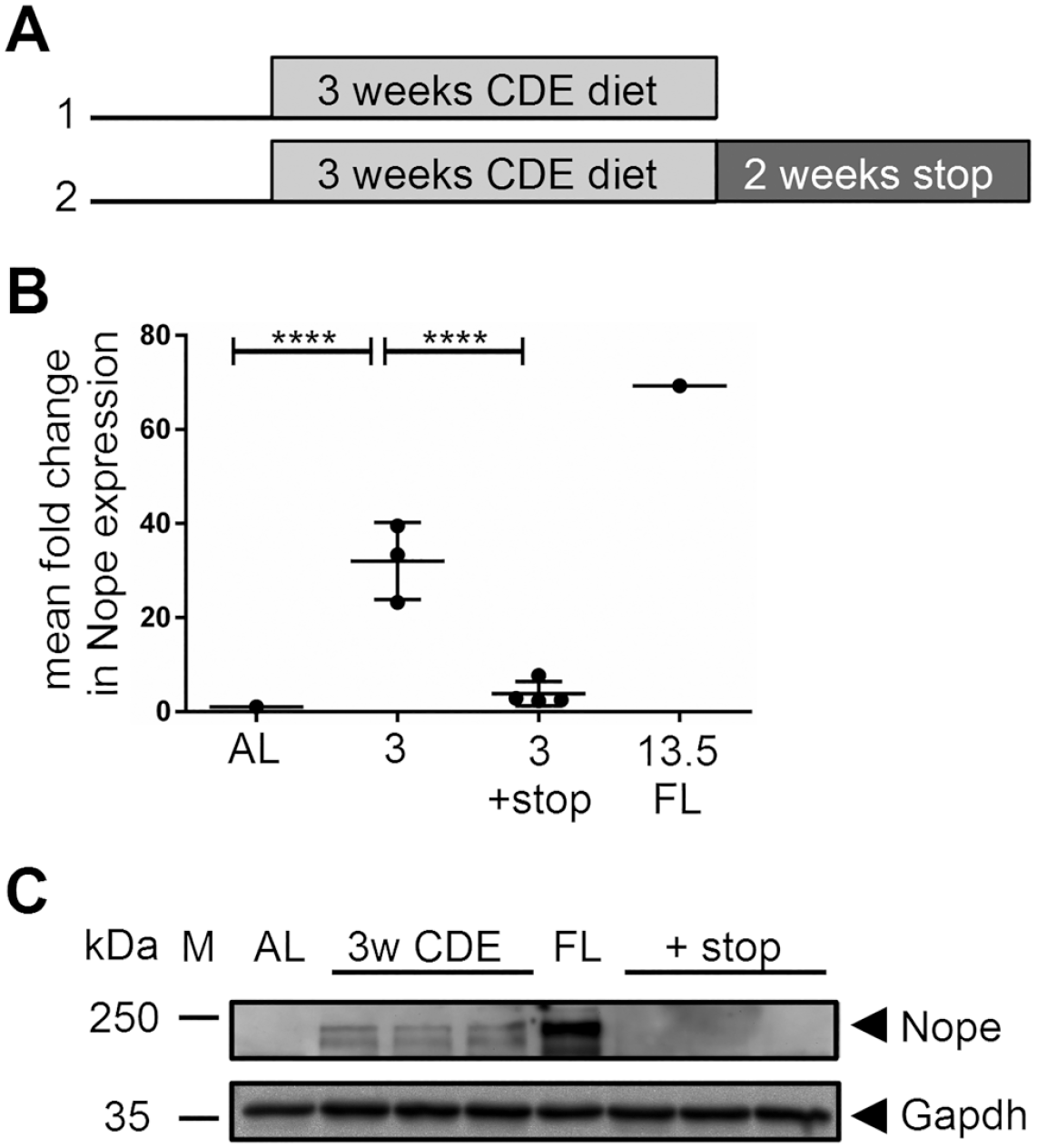
Expression of Nope in the CDE diet mouse model. (A) Study design: Group 1: Mice were fed with CDE diet for 3 weeks. Group 2: Mice were fed with CDE diet for 3 weeks, followed by 2 weeks of normal chow. Control groups were equally old, but untreated. (B) Expression of Nope was analyzed by qPCR. Three weeks of CDE diet induced a significant increase in the expression of Nope (p<0.0001). After another 2 weeks of a normal diet, the expression of Nope was significantly reduced to 3.8 (p<0.0001). AL is set as 1, and fetal liver is used as positive control. (C) Western blot analysis of Nope confirmed the qPCR results for group 1 (lanes 3–5) and group 2 (lanes 7–9) compared with AL (lane 2) and FL (lane 6). First lane M = marker; Nope, 170 kDa; Gapdh, 37 kDa. Abbreviations: CDE, choline-deficient and ethionine-supplemented; AL, adult liver; FL, fetal liver; qPCR, quantitative PCR; Gapdh, glyceraldehyde-3-phosphate dehydrogenase; Nope, Neighbor of Punc E11.
Lineage Tracing of CK19-CreERT Mice
CK19CreERT mice were a gift from Dr. David Scholten. 13 Gt(ROSA)26Sortm1Sormice were obtained from The Jackson Laboratory (Bar Harbor, ME). The CK19CreERT mice were crossed with Gt(ROSA)26Sortm1Sor reporter mice to obtain a CK19-lineage tracing mouse model (CKL_creERT strain) in which all CK19-derived cells express the LacZ gene [beta-Galactosidase (b-Gal)] and can be detected by fluorescence. The expression of cre recombinase was induced by intraperitoneal tamoxifen injection (20 mg/ml; 9 mg/40 g body weight). Tamoxifen injection started with four injections in a row in the week before the CDE diet started. During the CDE diet, tamoxifen was injected once per week (Fig. 4B).
Protein Extraction and Western Blotting
Total protein was extracted from all specimens using sodium dodecyl sulfate (SDS) lysis buffer containing 0.5% SDS, 15 mM Tris-HCl (pH 6.8), 2.5% glycerol, 1 mM ethylenediaminetetraacetic acid (EDTA), and proteinase inhibitor (Roche; Mannheim, Germany). After SDS-PAGE with equal amounts of protein (40 μg per lane), proteins were transferred onto nitrocellulose membranes (Whatman; Dassel, Germany) using a semidry blot system (Bio-Rad Laboratories; Munich, Germany). The membrane was blocked overnight at 4C in phosphate-buffered saline (PBS), pH 7.4, containing 5% dry milk powder. Primary antibodies, including goat anti-mouse Nope (R&D Systems; Minneapolis, MN) and rabbit anti-mouse glyceraldehyde-3-phosphate dehydrogenase (Gapdh) (Santa Cruz Biotechnology; Inc., Dallas, TX), were applied for 2 hr at room temperature. After extensive washing, membranes were incubated for 1 hr at room temperature with peroxidase-conjugated anti-goat (Sigma-Aldrich) or anti-rabbit (Sigma-Aldrich), respectively. Immune complexes were detected using ECL advance Western blot substrate (Amersham; Brunswick, Germany) according to the manufacturer’s instructions. After detection, membranes were stripped using 0.2 N NaOH for 10 min before application of the next primary antibody.
RNA Extraction and Reverse Transcription of Isolated RNA to cDNA
The total RNA from all samples was extracted with RNeasy Mini Kits (RNeasy Minikit (50)-Set Qiagen Shredder and RNAse-Free Dnase; Qiagen; cat. no. 74104) according to the manufacturer’s instructions. The amount of RNA was measured with a Nanodrop ND-1000 spectrophotometer (NanoDrop Technologies; Wilmington, DE).
The reverse transcription of the isolated RNA into cDNA was performed by Super ScriptTm III First Strand Synthesis for RT-PCR kit (Invitrogen; cat. no.18080-051) according to the manufacturer’s instructions. For each sample, 5 µg RNA per assay was used.
Quantitative Real-time Reverse Transcriptase Polymerase Chain Reaction
The differential expression of Nope and Cx26 was analyzed using quantitative real-time reverse transcriptase polymerase chain reaction (qRT-PCR). We performed a two-step qRT-PCR with SYBR Green Master Mix and the iQ5 System (BioRad Laboratories; Munich, Germany). Gapdh was used as a reference gene. Oligonucleotide primers for Gapdh, Nope, and Cx26 are listed in Supplementary Table 1. The amplification protocol was as follows: 95C for 3 min, followed by 40 cycles of 95C for 15 sec and 1 min at 60C, supplemented by a dissociation curve to identify false positive amplicons. The relative expression level of each gene was calculated using formula 2(−DD CT). 14 The AL and fetal liver were used as the standard control, and all samples were tested in triplicates. For each condition, the mean fold change of the expression level of at least three different liver specimens was calculated.
Immunohistochemistry
Immunohistochemical detection was performed on cryosections of liver tissue fixed in ice-cold acetone.
The following primary antibodies were used: rat anti-mouse E-cadherin (TaKaRa; Otsu, Shiga, Japan), rat anti-mouse CK19 (TROMA, DSHB, University of Iowa), goat anti-mouse Nope (R&D Systems), rat anti-mouse A6 (generous gift from V.M. Factor), and rabbit anti-mouse Cx26 antibodies (Invitrogen); for details, see Supplementary Table 2. These primary antibodies were detected through APC-conjugated donkey anti-rat, Cy3-conjugated donkey anti-goat, and Cy2-conjugated donkey anti-rabbit IgGs, respectively. Dipeptidyl peptidase IV (DPPIV) was detected using a fluorescein isothiocyanate (FITC)-conjugated rat anti-mouse antibody (BD Bioscience). Isotype controls were performed routinely to control for false-positive reactions and background level. Nuclei were stained with 4′,6-diamidino-2-phenylindole (DAPI; Sigma-Aldrich). Fluorescence images were acquired using fluorescent microscope IX 81 (Olympus; Hamburg, Germany) and Cell P Analysis Software (AnalySIS; Soft Imaging System GmbH, Muenster, Germany).
Hematoxylin and Eosin Stain
Liver cryosections of all groups were stained by hematoxylin and eosin (H&E) according to standard procedures.
Statistical Analysis
The differential gene expression was analyzed by unpaired t-test analysis using GraphPad Prism (version 6.07 for Windows; GraphPad, La Jolla, CA; www.graphpad.com). The p values shown are uncorrected and two-tailed, and α < 0.05 was considered as statistically significant (indicated as *, α < 0.01 indicated as **, α < 0.001 indicated as ***, and α < 0.0001 indicated as ****). The correlations were analyzed using Microsoft Office Excel (2014).
Results
CDE Diet and Stop Model
After the CDE diet, indicators of liver injury such as alanine aminotransferase (ALT) and bilirubin were measured in serum samples. Here, mice on a 3-week CDE diet showed an average value of 46.94 U/l ALT compared with 12 U/l ALT after normal diet. If the CDE diet was followed by an additional 2 weeks of a normal diet, the value decreased to an average of 10.67 U/l ALT (Supplemental Fig. S1). The maximum bilirubin level measured after 3 weeks of CDE diet was 24.49 mg/dl and after another 2 weeks of a normal diet was 3.3 mg/dl compared with the average value of <0.5 mg/dl with normal diet. Liver damage and recovery were confirmed by H&E staining (Supplemental Fig. S2): Compared with AL (Supplemental Fig. S2A), hepatic steatosis and inflammation can be observed after 3 weeks of CDE diet (Supplemental Fig. S2B). After additional 2 weeks of a normal diet, both steatosis and inflammation have resolved (Supplemental Fig. S2C).
Nope Expression in the CDE Diet and Stop Model
After 3 weeks of CDE diet, we detected a 33-fold increase in Nope expression compared with levels in AL by qPCR, reaching almost half of the expression level of Nope observed in fetal liver (3 weeks of CDE: cycle threshold (CT) = 33 vs adult liver: CT = 1; p<0.0001 CT). After another 2 weeks of a normal diet, the expression level of Nope diminished by more than 90%, thereby reaching almost levels as in normal adult liver (3 weeks of CDE + stop: CT = 3.8 vs 3 weeks of CDE diet = 33; p<0.0001 (CT= cycle threshold)) (Fig. 1B).
These qPCR results were confirmed by Western blot analysis, which is shown with a representative selection of protein lysates of the corresponding liver samples (Fig. 1C). In the AL, Nope was not detectable (lane 2), whereas the fetal liver was used as positive control (lane 6). After 3 weeks of CDE diet, Nope expression was detectable in all samples (170 kDa) (lanes 3–5). After another 2 weeks of a normal diet, Nope was no longer detectable in any of the samples by Western blot analysis (lanes 7–9).
Expression of Nope on Hepatocytes, Cholangiocytes and Oval Cells in Normal and Regenerating Liver
We used immunohistochemistry to investigate the expression of Nope by different cell types and at different stages of physiological liver development. At 1 week postnatal (wpn), Nope is detected throughout the majority of hepatocytes as well as in cholangiocytes (Fig. 2A). In the AL, Nope is reduced to a minimum expression level that is restricted to cholangiocytes, whereas hepatocytes showed no staining above background level (Fig. 2B), which was confirmed by isotype control staining (Supplemental Fig. S3). After 3 weeks of CDE diet, Nope is again detectable on the majority of hepatocytes as well as on cholangiocytes comparable to the levels at 1 wpn (Fig. 2C). In addition, Nope was detectable on oval cells as identified by costaining with A6, highlighting the ductular structures indicative of liver regeneration (Fig. 2C). After another 2 weeks of a normal diet, Nope-positive staining is only detectable on some residual hepatocytes (Fig. 2D, red), whereas oval cells are still frequently detectable by the coexpression of Nope and A6 (Fig. 2D).
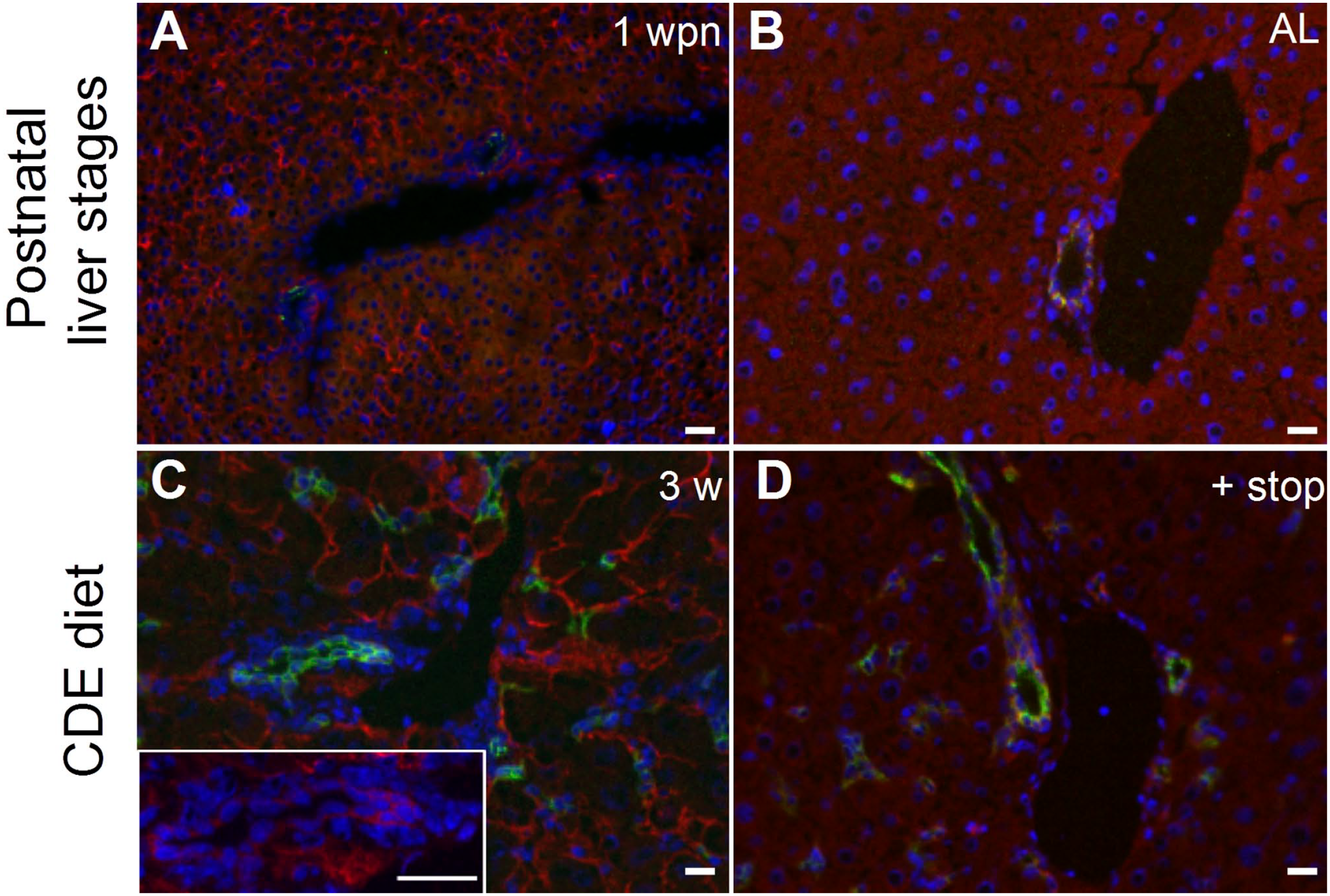
Detection of depolarized hepatocytes and oval cells. Cryosections of postnatal and adult livers (ALs) during and after CDE diet–induced liver injury. (A) At 1 wpn, Nope (red) is expressed by early postnatal hepatocytes. At this stage, a few biliary epithelial cells are detected by A6 (green). Nuclei were stained with DAPI (blue). (B) In AL, differentiated adult hepatocytes show no expression of Nope and A6 detects biliary epithelial cells. (C) After 3 weeks of CDE diet, hepatocytes show high expression of Nope. Oval cell activation was confirmed by staining for A6. The inset shows a higher magnification of a ductular structure to underline the expression of Nope. (D) After 3 weeks of CDE diet followed by a 2-week period of normal chow, hepatocytes showed no expression of Nope, whereas the expression of A6 in ductular structures and oval cells remained detectable. Abbreviations: CDE, choline-deficient and ethionine-supplemented; 1wpn, 1 week postnatal; AL, adult liver. DAPI, 6-diamidino-2-phenylindole; Nope, Neighbor of Punc E11. Scale bar: 50 µm.
Depolarization and Repolarization of Hepatocytes in the CDE Diet Mouse Model
After administration of a CDE diet for 3 weeks, the expression of the polarization marker Cx26 was reduced by factor 3.7 (AL: CT = 36.2 vs 3 weeks of CDE: CT = 9.8) as measured by qPCR (p<0.001). After pausing the diet for another 2 weeks, Cx26 expression increased approximately 2.5-fold (3 weeks of CDE: CT = 9.8 vs 3 weeks of CDE + stop: CT = 24.3) (p<0.05), thereby reaching almost levels as in normal AL (Fig. 3A). Immunohistochemical costaining of Cx26 with the bile canalicular marker protein DPPIV illustrated an overlapping expression pattern in AL, showing that Cx26 and DPPIV are restricted to the canalicular membrane (Fig. 3B; AL). After CDE-induced liver injury, Cx26 staining surrounds the hepatocytes including the basolateral membrane in contrast to DPPIV, which remains restricted to the bile canalicular site of hepatocytes (Fig.3B; 3 w CDE diet). Interestingly, after another 2 weeks of a normal diet, the Cx26 pattern is completely restored (Fig. 3B; + stop). The insets illustrate the change of Cx26 distribution on the membrane of individual hepatocytes (Fig. 3B, Merge insets).
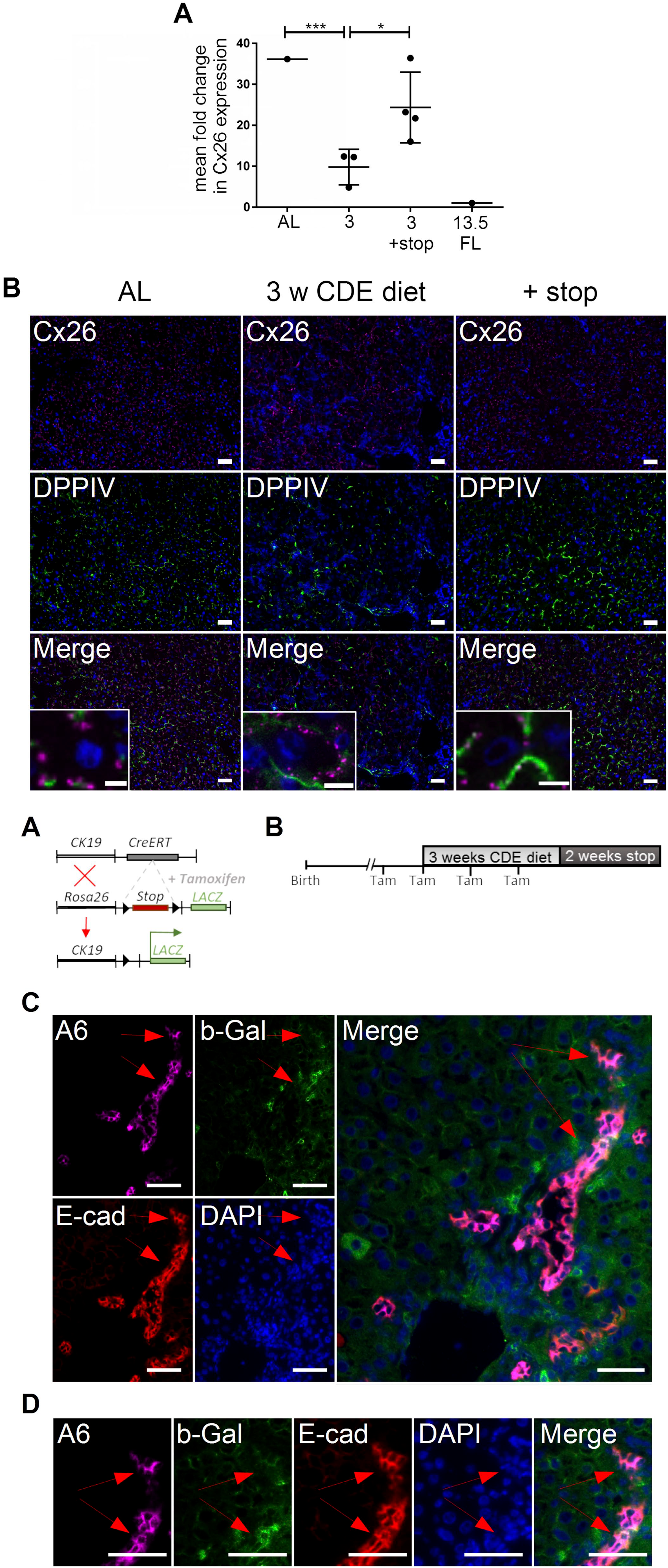
Expression pattern of Cx26 in adult liver (AL) with and without CDE diet and after stop diet. (A) mRNA expression of Cx26 was more than 3-fold reduced after 3 weeks of CDE diet (p<0.001). After another 2 weeks of a normal diet, the expression level of Cx26 showed a 2.5-fold increase (p<0.05). (B) Costaining of Cx26 (violet) as a marker of hepatocyte polarization with DPPIV (green) as a marker of the canalicular side of hepatocytes in AL, after 3 weeks of CDE diet (3 w CDE diet) and after another 2 weeks of a normal diet (+ stop). Nuclei were stained with DAPI (blue). In the merged pictures (insets showing single hepatocytes), the hepatocytic status of polarization is visualized by the distribution pattern of Cx26. In AL, Cx26 is strictly visualized in line with DPPIV at the canalicular membrane of hepatocytes (Merge). After 3 weeks of CDE diet, Cx26 has lost its expression pattern and can be visualized in the cellular membrane of hepatocytes including the basolateral side (Merge). After another 2 weeks of a stop diet, localization of Cx26 is back to the canalicular membrane (Merge). Abbreviations: CDE, choline-deficient and ethionine-supplemented; Cx26, connexin 26; DPPIV, dipeptidyl peptidase IV; DAPI, 6-diamidino-2-phenylindole. Scale bar: 50 µm. Scale bar of insets: 6.25 µm.
Oval Cell–mediated Hepatocytic Regeneration Begins After Repolarization of Hepatocytes
To further investigate the process of liver regeneration and whether Nope-positive hepatocytes originate from oval cells, we used a tamoxifen-inducible lineage tracing model with expression of b-Gal under the CK19 promoter (Fig. 4A). Using this lineage tracing model, we were able to detect b-Gal in E-cadherin-positive and A6-positive ductular structures and oval cells after the CDE diet, which persisted after another 2 weeks of a normal diet (Fig. 4C and D, red arrow).
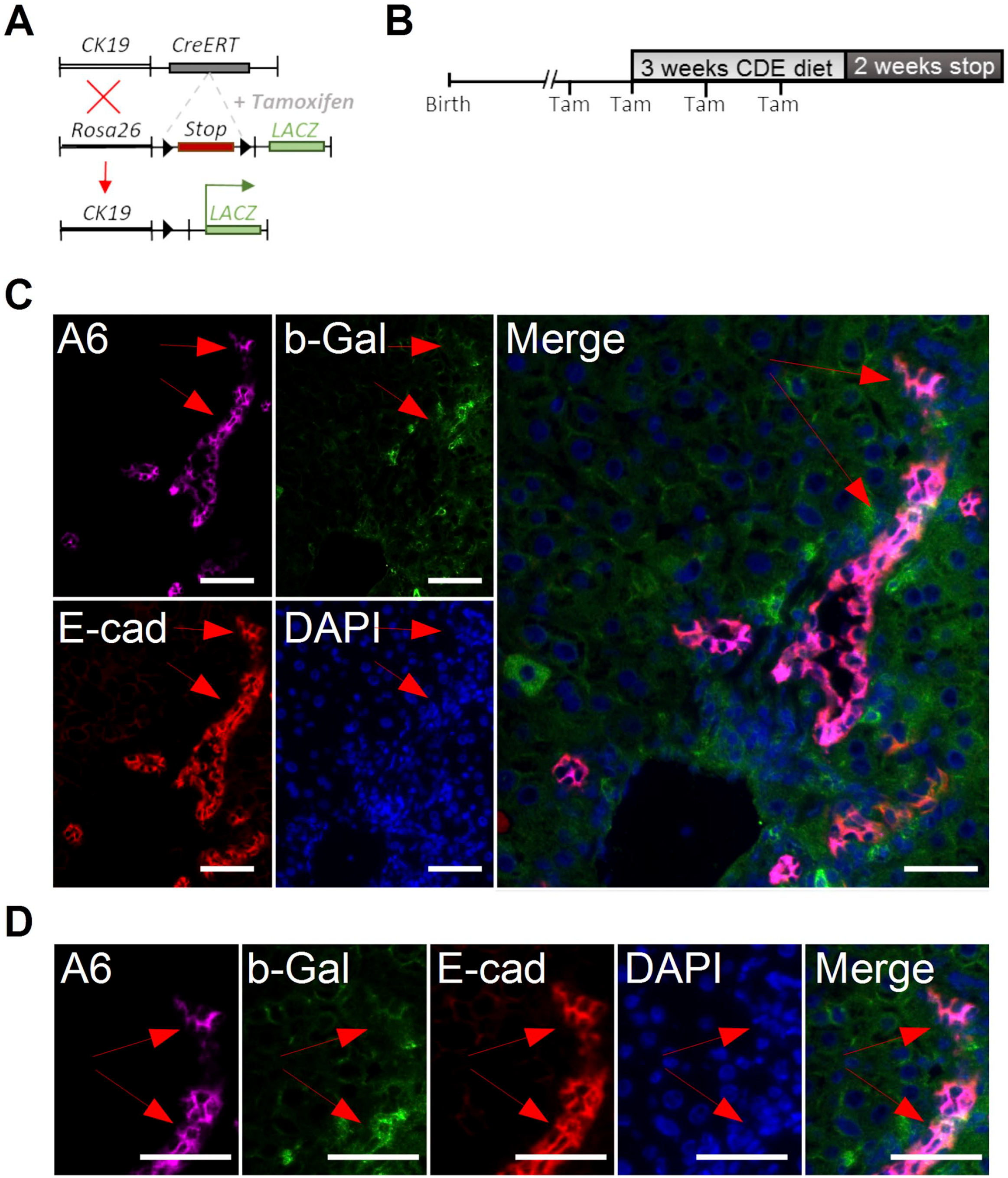
Ductular reaction was studied using a CK19-lineage tracing model in response to liver injury by a CDE diet. (A) Mating scheme for the generation of inducible b-Gal mice. (B) Study design: The expression of the reporter gene was induced by intraperitoneal administration of tamoxifen before and during the injury. (C + D) Immunohistochemistry of A6 (violet), E-cadherin (red), and b-Gal (green) in regenerating liver after 3 weeks of CDE diet followed by 2 weeks of a normal diet. Nuclei were stained with DAPI (blue). (D) Enlarged version to visualize the coexpression of E-cadherin, A6, and b-Gal in ductular structures. Abbreviations: CDE, choline-deficient and ethionine-supplemented; Tam, tamoxifen; A6, oval cell marker; b-Gal, beta-Galactosidase; E-cad, E-cadherin; DAPI, 6-diamidino-2-phenylindole. Scale bar C: 20 µm and C (Merge): 10 µm and D: 20 µm. Red arrows mark b-Gal-positive ductular cells.
However, we did not detect a single hepatocyte staining positive for b-Gal after the CDE diet (Fig. 5A). Only after another 2 weeks of a normal diet, we detected rare b-Gal-positive hepatocytes, which was confirmed by membranous coexpression of E-cadherin and no expression of A6 (Fig. 5B). However, none of these b-Gal-positive hepatocytes stained positive for Nope.
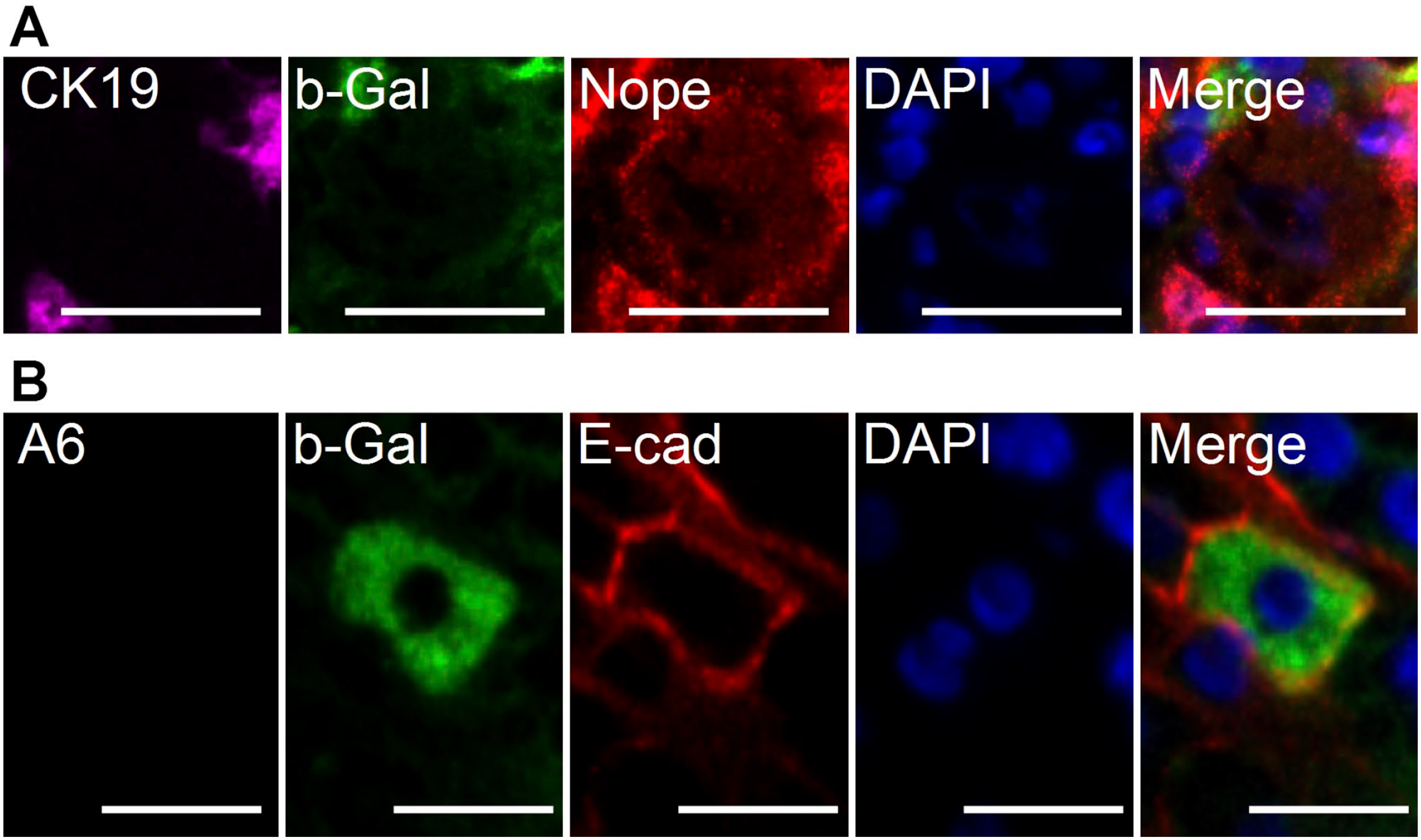
Expression of Nope by hepatocytes in response to liver injury by CDE diet and during liver regeneration using a CK19-lineage tracing model. (A) Immunohistochemistry of CK19 (violet), b-Gal (green), and Nope (red) during CDE diet–induced liver injury. Nuclei were stained with DAPI (blue). Hepatocytes express Nope but show no costaining with CK19 or b-Gal, excluding their origin from CK19-positive cells. (B) Immunohistochemistry of A6 (violet), b-Gal (green), and E-cadherin (red) in regenerating liver after 2 weeks of a normal diet. Newly generated mature hepatocytes rarely originate from oval cells as shown by the coexpression of b-Gal in E-cadherin-positive hepatocytes. Abbreviations: CDE, choline-deficient and ethionine-supplemented; A6, oval cell marker; b-Gal, beta-Galactosidase; E-cad, E-cadherin; DAPI, 6-diamidino-2-phenylindole; Nope, Neighbor of Punc E11. Scale bar (A) 20 µm, (B) 50 µm.
Discussion
In our study, the CDE diet mouse model proved useful to analyze the biological process of hepatocellular depolarization and repolarization in the complex system of liver regeneration. We could show for the first time that a CDE diet–based liver injury induces the expression of the oncofetal stem/progenitor cell marker Nope in adult hepatocytes, which is accompanied by a loss of hepatocyte polarization. After another 2 weeks of a normal diet, polarization of hepatocytes was almost completely restored, whereas the expression of Nope spared hepatocytes and remained limited to bile ducts and oval cells. Importantly, regenerating hepatocytes only rarely originated from CK19-positive oval cells, and this oval cell–mediated hepatocyte regeneration was preceded by repolarization of hepatocytes.
In our previous work, we had been able to show that Nope is an oncofetal biomarker5,10,15 that is also expressed in depolarized adult hepatocytes after BDL. 6 However, the BDL model is irreversible and was therefore not useful to analyze any further functional implication. The CDE diet, however, is of special value because we were able to demonstrate that the expression of Nope on depolarized hepatocytes is completely reversible after 2 weeks of a stop diet accompanied by hepatocytic repolarization.
Changes in hepatocytic polarity due to cholestatic liver injury have been reported before, 16 but have not been described for chronic liver injury models like CDE diet, yet. 17 To the best of our knowledge, this is the first work showing that the CDE/stop model can also be used as a hepatocellular depolarization and repolarization model. Hepatocellular polarity not only plays an essential role in liver development and regeneration but is also important for maintaining liver function. 3 Polarity is a kind of prerequisite for all functional fields of the liver, such as detoxification, metabolism, and protein synthesis. 3 Polarity ensures that the important separation between the bile ductular and the sinusoidal side is maintained. Various gap junctions, tight junctions, and connexins form a network that maintains polarity.18,19 In this context, the expression of Nope is complementary to other known hepatocellular polarization markers such as Cx26 and bile salt export pump.20–22 This phenomenon is in line with our previous finding after cholestatic liver injury by BDL. 6
The functional relevance between hepatocyte polarity and the expression of Nope remains not fully understood. During the final maturation stage of physiological development after birth, the polarity of single hepatocytes is not yet established and Nope switches its expression pattern on hepatocytes from circular to strictly sinusoidal. 5 Indeed, the expression pattern of Nope on depolarized hepatocytes in the CDE diet–injured liver is quite similar to the expression of Nope on early postnatal hepatocytes. Furthermore, comparison with other proteins of the same family, such as Punc, Dcc, or Neogenin, suggests that it may be important in the context of cellular differentiation and axonal guidance. 23
When the regeneration of the liver had taken place for at least 2 weeks, repolarized hepatocytes did not express Nope any longer. Interestingly, the CDE diet–induced oval cells were not only detectable at 3 weeks of CDE diet but also remained detectable after an additional 2 weeks of a normal diet. 21 These cells were A6- and Nope-positive. The characterization of AL progenitor cells (oval cells) in the rat model as Alb+/Afp+/CK19+ and their expression of Dlk1 24 have long suggested a developmental relationship with fetal liver stem/progenitor cells. Recent work has also shown for the first time, using Sox9-based lineage tracing, a link from ductal plate cells to adult progenitor cells. 25 The Sox9-positive progenitor cells described in this article were the origin of periportal but not pericentral hepatocytes after DDC/CDE diet. However, this was also shown by another research group using Sox9-based lineage tracing in various liver injury models. 26 In another lineage tracing analysis, the transcription factor Foxl1 was detected in adult progenitor cells in the DDC model with bipotent differentiation into cholangiocytes and periportal hepatocytes. 27 However, Foxl1 only identifies a part of Sox9-positive cells and, in contrast to Sox9, is not detectable in the fetal liver or AL. Recent work suggested that new hepatocytes only originate from stem cells, when the regeneration of the liver through preexisting hepatocytes is blocked.28,29
Due to the spatial proximity of Nope-positive hepatocytes and oval cells in zones 1 and 2, we wanted to clarify the hypothesis whether these hepatocytes have newly emerged from oval cells. For this purpose, we used the CK19-lineage tracing model and fed the mice with CDE and CDE-stop diets. The chronic liver injury model impaired regeneration via hepatocyte proliferation and induced proliferation of oval cells. 30 Nope-expressing hepatocytes were found in zones 1 and 2 after 3 and 4 weeks of CDE diet. We did not find any hepatocytes arising from oval cells at this stage, which is in accordance with preexisting literature. 31 Regenerating hepatocytes only rarely originated from CK19-positive oval cells, and this oval cell–mediated hepatocyte regeneration did not occur before 2 weeks of stop diet, that is, was preceded by repolarization of hepatocytes.
These findings indicated that repolarization of hepatocytes precedes the onset of ductular regeneration of liver mass. Our findings complement the published model of Nope and Cx26 expression patterns during liver development and AL with the stage of injured liver and recovery 6 (Schematic Fig. 6): In the fetal liver, hepatoblasts are not yet polarized and show high levels of Nope (red) expression, whereas Cx26 (white circles) is not yet detectable (Fig. 6A). Postnatally, the polarization of hepatocytes is initiated and Nope expression is high. The protein is located in the sinusoidal membrane of the hepatocytes. The expression of Cx26 is increasing with a patchy distribution pattern over the whole membrane (canalicular domain is depicted in green) (Fig. 6B). In the AL, polarization is completed, Nope expression is limited to cholangiocytes, and the expression pattern of Cx26 shows a restriction to the bile canalicular domain (Fig. 6C). Challenged with CDE diet or BDL, adult hepatocytes lose their polarization and show expression levels and patterns of Nope and Cx26 that are comparable to postnatal levels showing initialization of polarization (Fig. 6D). Interestingly, when the CDE diet is stopped, repolarization takes place, the expression of Nope is decreased again, and the expression and pattern of Cx26 correspond to the normal AL again (Fig. 6E). This repolarization of hepatocytes precedes an oval cell–mediated hepatocyte regeneration.
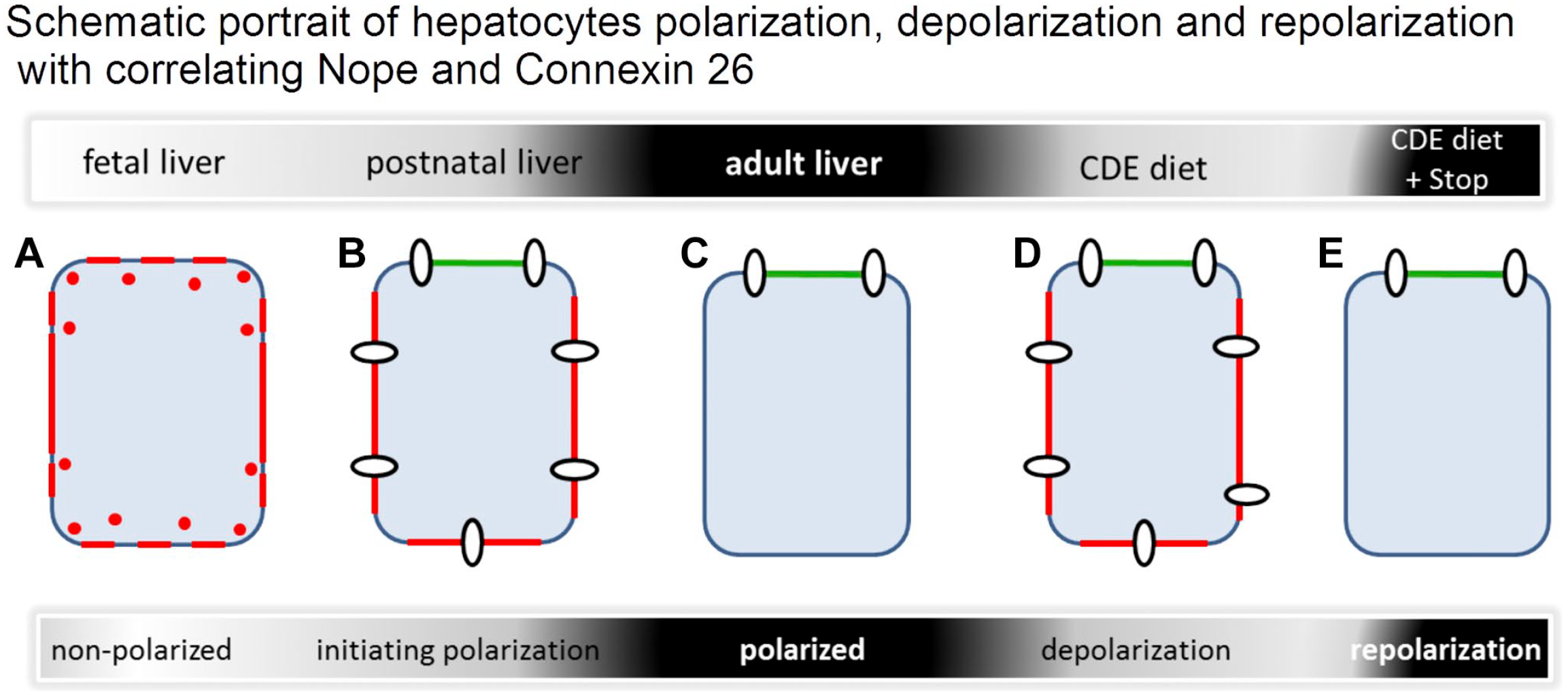
Schematic representation of hepatocytes at different stages of polarization. In the early fetal liver, hepatocytes show a circular membranous expression pattern of Nope (red) while Cx26 (white circles) is not yet expressed (A). Initiating polarization postnatally with Nope at the sinusoidal sites and connexin distributed to all membranes (canalicular domain is depicted in green) (B). Polarized hepatocyte without any Nope and Connexin distributed to the typical localization at the sinusoidal–canalicular transition (C). After CDE diet–induced liver injury, the expression level and pattern of Nope and Cx26 resemble that of early postnatal hepatocytes that are initiating polarization (D). If the CDE diet is followed by normal diet, repolarization can be observed with Cx26 localized at the canalicular membrane and no expression of Nope as in normal AL allowing liver regeneration (E). Abbreviations: CDE, choline-deficient and ethionine-supplemented; AL, adult liver; Cx26, connexin 26; Nope, Neighbor of Punc E11.
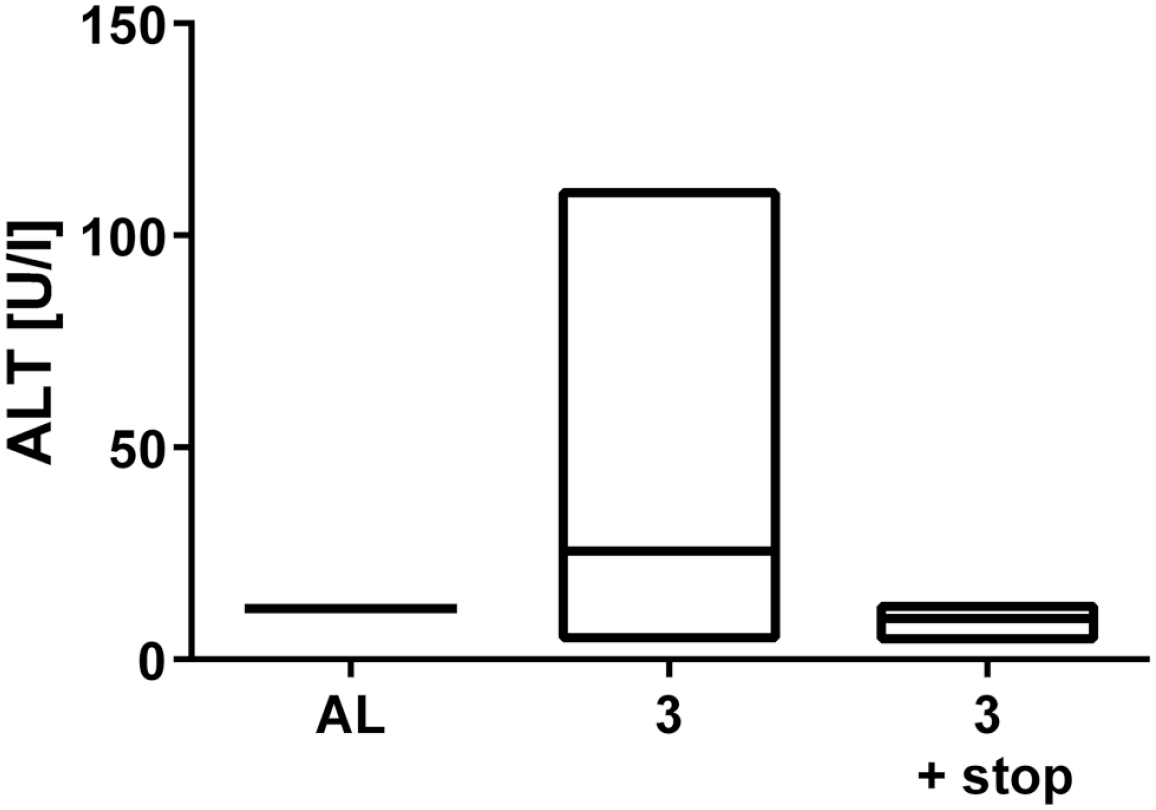
Serum analysis of alanin-aminotransferase (ALT) after CDE diet. Mice on a 3 week CDE diet showed an average value of 46.94 U/l ALT compared to 12 U/l ALT after normal diet. After an additional 2 weeks on normal diet, the value decreased to an average of 10.67 U/l ALT.
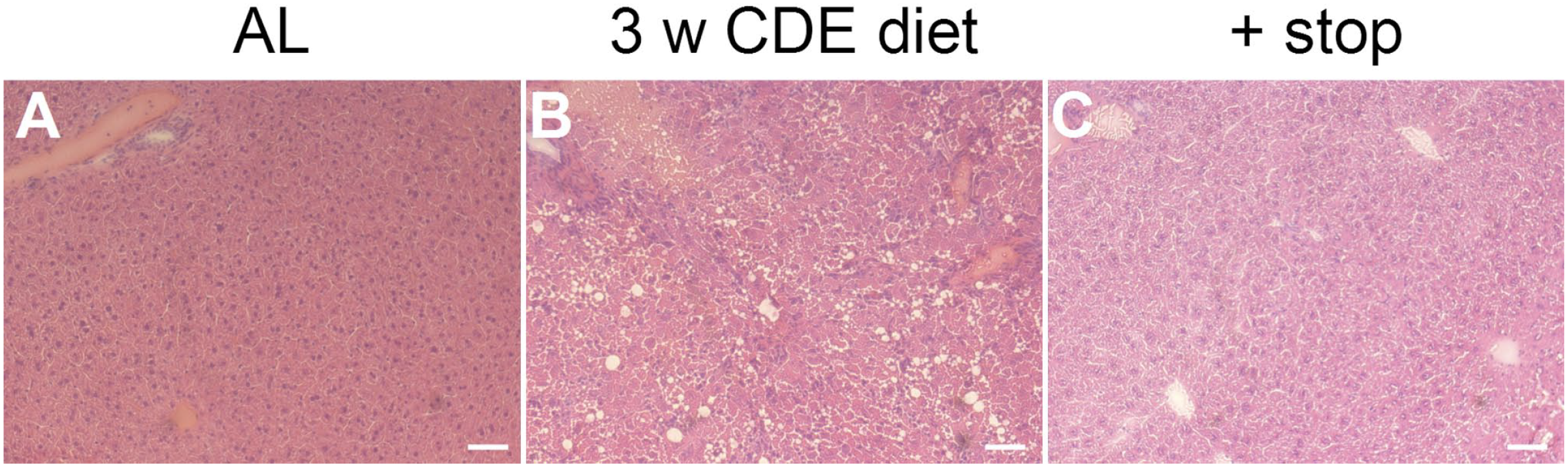
H&E staining of untreated, CDE diet and CDE diet followed by two weeks of normal diet treated mice. A) Adult liver showing a normal structure with a portal field and a central vene. B) After three weeks of CDE diet, hepatic steatosis and inflammation are visible. C) After another two weeks of normal diet, both steatosis and inflammation have resolved. H&E staining: Hematoxylin and eosin staining; scale bar 50 μm.
Cryosections of adult livers were stained using IgG control for Nope (IgG goat), Cx26 (IgG rabbit), E-cadherin, CK19 and A6 (IgG rat). Nuclei were stained with DAPI. DAPI, 6-diamidio-2-phenylindole. Scale bar 50 μm.
In summary, polarization of hepatocytes is an important part of liver regeneration and precedes oval cell–mediated regeneration of the liver. This process can be visualized by a characteristic expression pattern of Nope.
Supplemental Material
sj-docx-4-jhc-10.1369_00221554221084665 – Supplemental material for Repolarization Precedes Oval Cell–mediated Hepatocytic Regeneration in the CDE Diet Mouse Model
Supplemental material, sj-docx-4-jhc-10.1369_00221554221084665 for Repolarization Precedes Oval Cell–mediated Hepatocytic Regeneration in the CDE Diet Mouse Model by Susanne Zweerink, Vera Mueck, Laurenz P. Kraemer, Eva Tischler, Sigrid Schulte, Tobias Goeser and Dirk Nierhoff in Journal of Histochemistry & Cytochemistry
Supplemental Material
sj-tif-1-jhc-10.1369_00221554221084665 – Supplemental material for Repolarization Precedes Oval Cell–mediated Hepatocytic Regeneration in the CDE Diet Mouse Model
Supplemental material, sj-tif-1-jhc-10.1369_00221554221084665 for Repolarization Precedes Oval Cell–mediated Hepatocytic Regeneration in the CDE Diet Mouse Model by Susanne Zweerink, Vera Mueck, Laurenz P. Kraemer, Eva Tischler, Sigrid Schulte, Tobias Goeser and Dirk Nierhoff in Journal of Histochemistry & Cytochemistry
Supplemental Material
sj-tif-2-jhc-10.1369_00221554221084665 – Supplemental material for Repolarization Precedes Oval Cell–mediated Hepatocytic Regeneration in the CDE Diet Mouse Model
Supplemental material, sj-tif-2-jhc-10.1369_00221554221084665 for Repolarization Precedes Oval Cell–mediated Hepatocytic Regeneration in the CDE Diet Mouse Model by Susanne Zweerink, Vera Mueck, Laurenz P. Kraemer, Eva Tischler, Sigrid Schulte, Tobias Goeser and Dirk Nierhoff in Journal of Histochemistry & Cytochemistry
Supplemental Material
sj-tif-3-jhc-10.1369_00221554221084665 – Supplemental material for Repolarization Precedes Oval Cell–mediated Hepatocytic Regeneration in the CDE Diet Mouse Model
Supplemental material, sj-tif-3-jhc-10.1369_00221554221084665 for Repolarization Precedes Oval Cell–mediated Hepatocytic Regeneration in the CDE Diet Mouse Model by Susanne Zweerink, Vera Mueck, Laurenz P. Kraemer, Eva Tischler, Sigrid Schulte, Tobias Goeser and Dirk Nierhoff in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
We are grateful to Gisela Holz, Gudrun Suckau, Birgitta Jakob, and Elisabeth Konze for excellent technical assistance.
Author’s Note
Eva Tischler is now affiliated to Department of Gynecology and Obstetrics, University Tuebingen, Tuebingen, Germany.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors Contributions
All authors have contributed to this article as follows: study of concept and design (VM, DN), CDE diet application (VM, ET), acquisition of data (SZ, VM, LPK, ET), analysis and interpretation of data (VM, SZ, LPK, ET, DN), statistical analysis (SZ), drafting of the manuscript (SZ, LPK, DN), critical revision of the manuscript for important intellectual content (VM, SS), and acquisition of funding (TG, DN).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was supported by the Wilhelm-Doerenkamp Foundation, Cologne, and the Medical Faculty of the University of Cologne.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
