Abstract
Interleukin (IL)-1 plays a key role in carcinogenesis, tumor progression, and metastasis. Although IL-1 may enhance the expansion of CD8+ T-cells, the pathological contribution of IL-1-activated CD8+ T-cells to tumor metastasis remains unclear. This study used a liver metastasis model of the EL4 T-cell lymphoma cells transplanted into human IL (hIL)-1α conditional transgenic (hIL-1α cTg) mice. Overproduction of hIL-1α suppressed both macroscopic and histological liver metastasis of EL4 T-cell lymphoma. The hIL-1α-induced inflammatory state increased the number of CD8+ T-cells both within and around metastatic tumors. Moreover, larger numbers of CD8+ T-cells showed greater infiltration of liver blood vessels in hIL-1α cTg mice than in control wild-type mice. Terminal deoxynucleotidyl transferase dUTP nick-end labeling staining of liver tissue from hIL-1α cTg mice indicated increased apoptosis of cells in the tumor. Localization of apoptosis cells resembled that of CD8+ T-cells. In addition, cytotoxicity assay showed that CD8+ T-cell counts from tumor-bearing hIL-1α cTg mice correlated with cytotoxicity against EL4. In summary, IL-1α suppresses lymphoma metastasis, and IL-1α-activated CD8+ T-cells may play important roles in inhibiting both tumor metastasis and metastatic tumor growth:
Introduction
Interleukin (IL)-1 is a central mediator of innate immunity and inflammation. 1 IL-1α and IL-1β are major agonist molecules of the IL-1 family.2–5 Both are synthesized as precursor proteins. Because of its powerful inflammation-promoting capacity, the bioactivity of IL-1β is regulated by IL-1-converting enzyme, designated as “caspase-1,” which cleaves the IL-1β precursor and results in an active soluble form of IL-1β. 6 In contrast, IL-1α precursor is capable of acting in a membrane-bound form and is biologically active.7,8 While IL-1β has been shown to play an important role in carcinogenesis and tumor growth, 9 membrane IL-1α paradoxically promotes antitumor effects.10–12
CD8+ T-cells are associated with innate and adaptive immunity against viral infection and tumor generation, and have the ability to induce cell death via secretion of tumor necrosis factor-α and interferon-γ, cytolytic granule release, and induction of Fas-mediated apoptosis. IL-1 enhances in vitro expansion of CD8+ T-cells responding to polyclonal stimulants.13,14 A previous experimental study showed that IL-1 exerted a marked enhancing effect on antigen-specific CD8+ T-cell expansion, differentiation, migration to the periphery, and memory T-cell development and suppressed tumor progression promoted by the tumor cell line. 15 However, no studies have determined whether CD8+ T-cells induced by IL-1 suppress tumor generation according to histological findings.
To assess the suppressive effects of IL-1-induced CD8+ T-cells on metastasis of lymphoma, this study used a liver metastasis model of EL4 T-cell lymphoma cells transplanted into human IL (hIL)-1α conditional transgenic (hIL-1α cTg) mice. We hypothesized that metastasis of EL4 to the liver would be suppressed and infiltration of CD8+ T-cells would be stimulated in hIL-1α cTg mice. The purpose of this study was to assess the effects of IL-1α on lymphoma metastasis by focusing on histopathological findings of CD8+ T-cells both before and after EL4 liver metastasis in hIL-1α cTg mice.
Materials and Methods
Animals
Wild-type C57BL/6 (WT) mice were purchased from Sankyo Labo Service (Tokyo, Japan). Mice were kept under specific pathogen-free conditions in animal facilities certified by the animal care committee at Keio University.
Generation and Induction of hIL-1α cTg Mice
An hIL-1α cTg construct was generated using the loxP-neomycin resistance gene (Neo)-polyA-loxP-hIL1alpha sequence under a β-actin promotor in a C57BL/6 background. Offspring harboring the hIL-1α cTg transgene were crossed with Mx Cre Tg mice to establish Mx Cre/hIL-1α cTg mice, designated as hIL-1α cTg mice. Human IL-1α expression was induced in male hIL-1α cTg mice by injecting 200 μl of a solution containing 250 μg of PolyI-PolyC (Sigma-Aldrich; St. Louis, MO) intraperitoneally for three consecutive days. Some hIL-1α cTg mice were not treated with PolyI-PolyC. 16
Tumor Cell Line and Tumor Cell Injection
This study used EL4 T-cell lymphoma (EL4) cells of C57BL/6 (H-2b) origin, which are highly metastatic to the liver. Tumor cells were propagated in RPMI 1640 medium supplemented with 10% heat-inactivated fetal calf serum, 2 mM
ELISA Analysis
Livers were collected from WT and hIL-1α cTg mice at 1 week after PolyI-PolyC injection. Liver cell lysates were prepared from the liver of each mouse using RIPA buffer (1% Tween 20, 0.1% SDS, 150 mM NaCl, 10 mM Tris-HCl [pH 7.4], 0.25 mM phenylmethylsulfonylfluoride, 10 μg/ml aprotinin, 10 μg/ml leupeptin, 1 mM Na3VO4, and 5 mM NaF [Sigma-Aldrich Co.]). ELISA for hIL-1α in cell lysate was performed in accordance with the instructions from the manufacturer (R&D Systems; Minneapolis, MN).
Histological Analysis
For histological analysis of the liver, liver samples were collected 3 weeks after PolyI-PolyC injection, or 30 days after EL4 cell transplantation, fixed with formalin, cut into 11 pieces, and then embedded in paraffin. Each paraffin-embedded liver was cut into 4-μm-thick sections, stained with hematoxylin and eosin (H&E), and subjected to quantitative analyses.
Immunostaining
Each section was deparaffinized, rehydrated, heated in citrate buffer (0.01 mol/L, pH 6.0), and treated with endogenous peroxidase at room temperature. After blocking in 0.3% hydrogen peroxide methanol, immunostaining for CD8+ T-cells was performed using anti-CD8 antibody (ab203035; Abcam, Cambridge, UK) as the primary antibody, and sections were incubated overnight at 4C. Immunoreactivity was visualized by the avidin–biotin complex method (Avidin–Biotin Complex kits; Vector Laboratories, Burlingame, CA), 17 and sections were developed in diaminobenzidine (DAB) reagent (SK-4100; Vector Laboratories). For immunofluorescence staining, Streptavidin Dylight 488 (SA-5488; Vector Laboratories) was used, and sections were observed under fluorescent biological microscopy.
Terminal Deoxynucleotidyl Transferase dUTP Nick-end Labeling (TUNEL) Staining
TUNEL staining was performed using an In Situ Apoptosis Detection Kit (Takara Bio; Shiga, Japan). After deparaffinization and proteinase K treatment (10–20 μg/ml, 15 min), EL4 sections were washed with PBS. After blocking in 0.3% hydrogen peroxide methanol, sections were washed with PBS. Fifty microliters of labeling reactor (5 μl TdT enzyme + 45 μl Labeling Safe Buffer) was applied on the slide, and sections were reacted for 60–90 min at 37C in a wet box (using a cover slip to prevent drying) and then washed 3× with PBS for 5 min to stop the reaction. Observation under fluorescence microscopy was then performed.
Quantification of Tumor Area
Tumor colonies were divided into two size groups using a cutoff value of 1 mm and counted under microscopy. In the analysis of metastatic tumor area, five sections containing metastatic tumor were randomly chosen from the liver of each mouse. Metastatic tumors were traced and tumor area was calculated using Image J version 1.48 (National Institutes of Health; Bethesda, MD). The number of tumor colonies and tumor area were compared between WT mice and hIL-1α cTg mice.
Quantification of CD8+ T-cells and Apoptotic Cells
Liver sections from each mouse were divided into three groups: non-tumor area, tumor area, and area within liver blood vessels. Five locations in each group were chosen randomly. The number of CD8+ T-cells was counted in each location and compared between WT and hIL-1α cTg mice. In addition, the ratio of CD8+ T-cells to vessel area (mm2) was calculated in each location and compared between WT and hIL-1α cTg mice. To assess apoptotic cells in the tumor, six locations containing metastatic tumor were randomly chosen from each mouse. The number of TUNEL-positive cells was then counted manually in each location, and tumor area in each location was also analyzed using Image J version 1.48 software (National Institutes of Health). The ratio of TUNEL-positive cells to tumor area (mm2) was then determined in each location and compared between WT and hIL-1α cTg mice.
Cytotoxicity Assay
Splenocytes were harvested from tumor-bearing WT mice and hIL-1α cTg mice at 7 days after tumor inoculation and stimulated with EL4 cell lysates with rhIL2 (1 μg/ml) for 4 days. CD8+ T-cells were isolated from cultured splenocytes by negative selection using a CD8+ T-cell isolation kit in accordance with the protocol from the manufacturer (Miltenyi Biotec; Auburn, CA). CD8+ T-cell activity was assayed in a 4-hr cytotoxicity assay using EL4 cells as target cells. Effector CD8+ T-cells were added to target cells in different effector:target (E/T) ratios and incubated for 4 hr each. Cytotoxic activity was determined by the CytoTox 96 non-radioactive cytotoxicity assay (Promega; Madison, WI).
Statistical Analysis
Results are expressed as mean ± SD. The statistical significance of differences between groups was evaluated using the Mann–Whitney U-test. For the testing of all hypotheses, a two-sided α threshold of 0.05 was considered statistically significant. All statistical analyses were conducted using SPSS Statistics ver. 24.0 software (IBM; Armonk, NY).
Study Approval
All mouse protocols were approved by the Animal Care Committee at Keio University, and all experiments were carried out based on committee guidelines.
Results
CD8+ T-cell Counts and hIL-1α Levels in the Liver Were Elevated in IL-1α cTg Mice
To assess CD8+ T-cells in the liver, we injected PolyI-PolyC into 8-week-old WT mice and hIL-1α cTg mice. Three weeks after PolyI-PolyC injection, we retrieved the liver and determined the number of CD8+ T-cells in the liver from WT mice (Fig. 1A) and hIL-1α cTg mice (Fig. 1B). In the retrieved liver, the number of CD8+ T-cells was significantly larger in hIL-1α cTg mice than in WT mice (Fig. 1C). We next isolated liver cells and determined intracellular hIL-1α levels by ELISA with hIL-1α cTg mice 1 week after PolyI-PolyC injection. As shown in Fig. 1D, hIL-1α protein was specifically detected in liver cells from hIL-1α cTg mice.
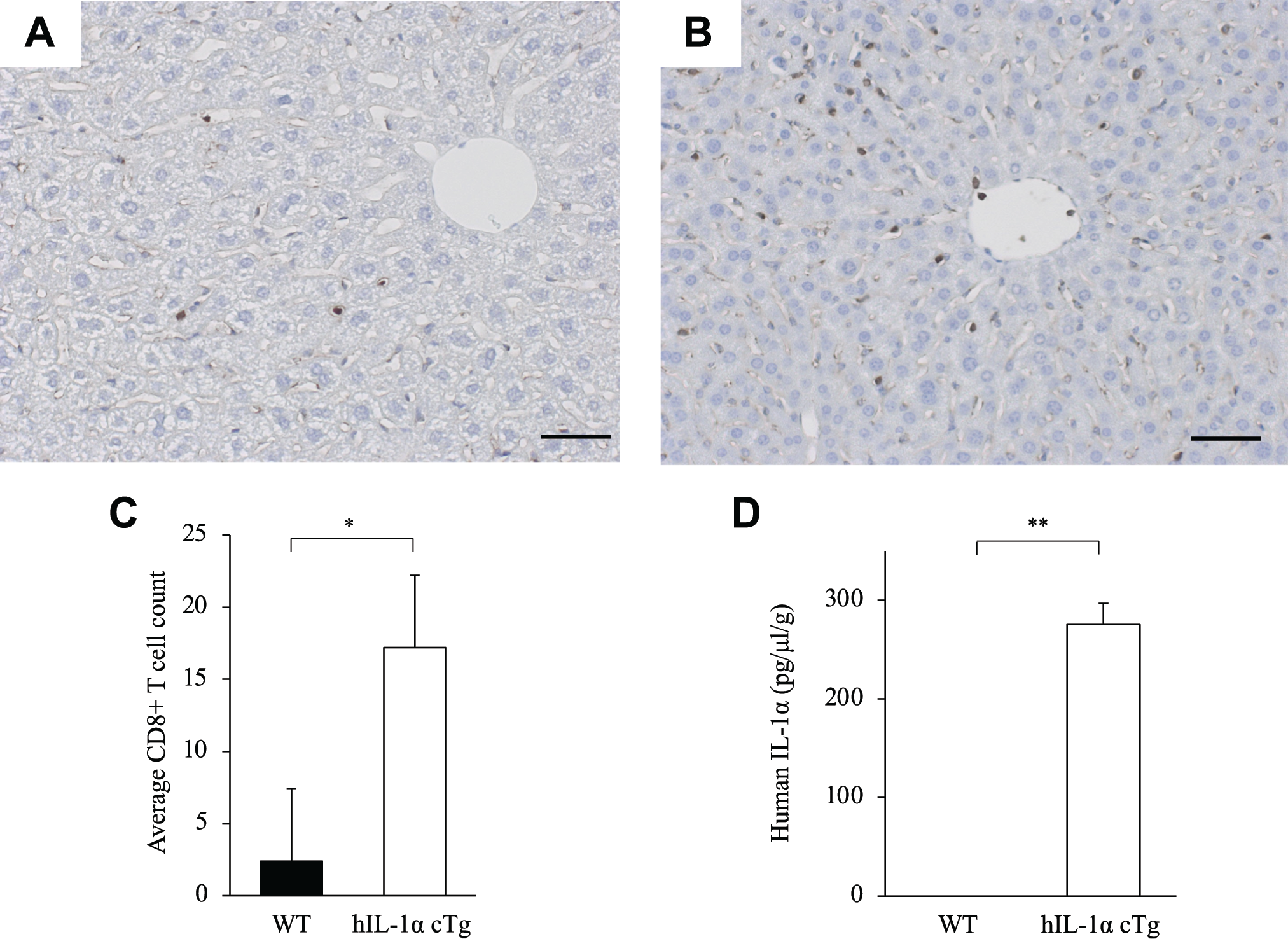
Immunohistochemistry and ELISA of CD8+ T-cells in liver. Anti-CD8 antibody (ab203035; Abcam) is used to stain CD8+ T-cells in liver sections in WT mice (A) and human IL-1α conditional transgenic (hIL-1α cTg) mice (B). The graph shows the number of CD8+ T-cells (C) and hIL-1α protein levels as determined by ELISA in livers of WT mice and hIL-1α cTg mice (D). *p<0.05. **p<0.01. Bar = 50 μm. Abbreviation: WT, Wild-type C57BL/6.
Tumor Metastasis Was Suppressed in hIL-1α cTg Mice
We collected liver samples from WT and hIL-1α cTg mice at 30 days after EL4 injection (n=4). All four WT mice exhibited gross tumor metastasis in the liver (Fig. 2A), but three of four hIL-1α cTg mice did not (Fig. 2B). Histological analysis revealed that WT mice exhibited numerous tumor colonies ≥1 mm in diameter in the liver (Fig. 2C), whereas hIL-1α cTg mice exhibited a much lower number of tumor colonies at low magnification (Fig. 2D). Tumor colonies were confirmed in all WT (Fig. 2E) and hIL-1α cTg mice (Fig. 2F) at high magnification. The number of total colonies, colonies ≥1 mm, and colonies <1 mm were significantly lower in hIL-1α cTg mice than in WT mice (Fig. 2G). The average area of metastatic tumor was significantly smaller in hIL-1α cTg mice than in WT mice (Fig. 2H).
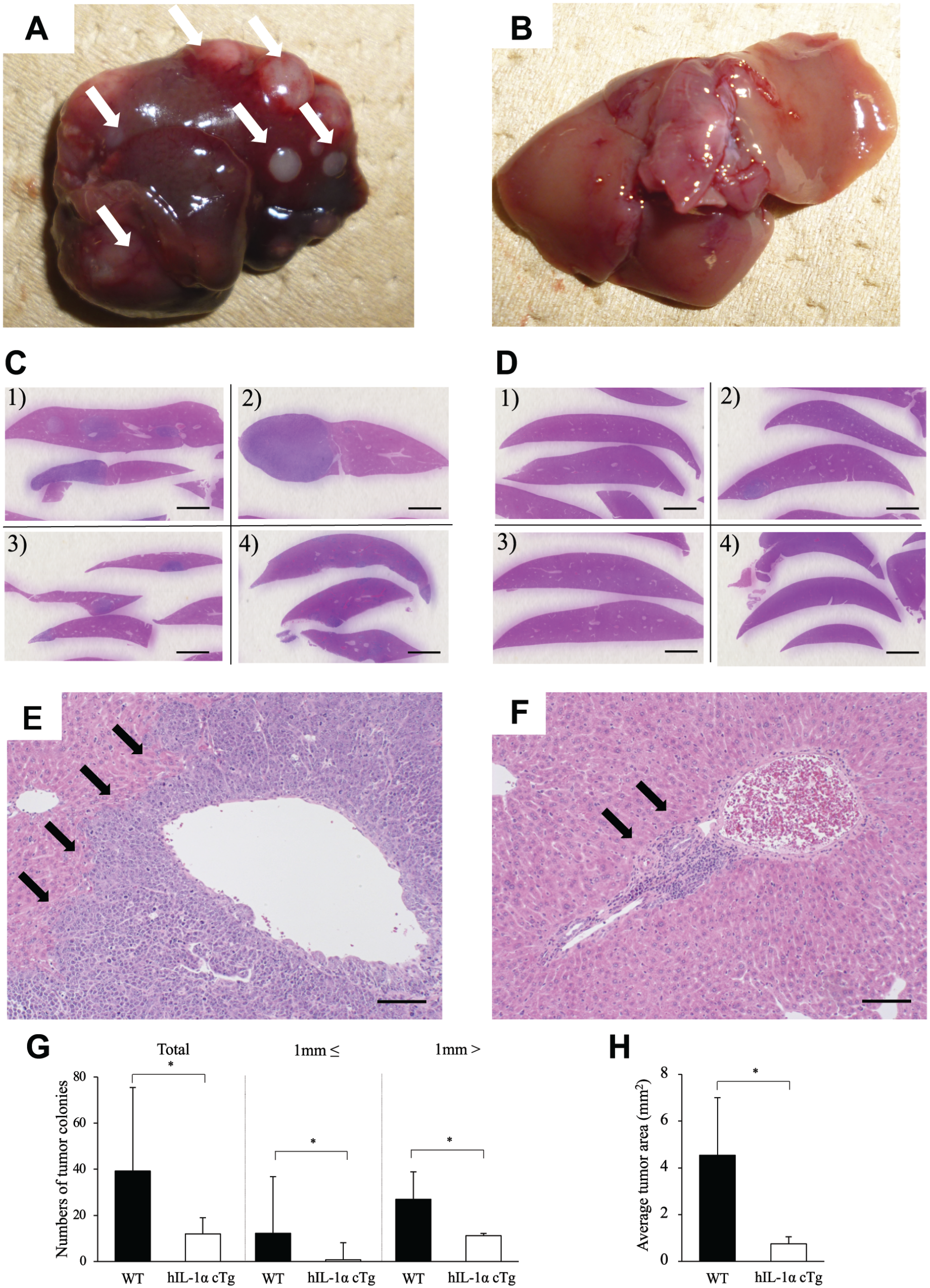
Macroscopic and histological findings of EL4 metastasis. EL4 cells are transplanted into WT mice and hIL-1α cTg mice (n=4). Macroscopic liver appearance (A, B) and microscopic liver appearance (C, D) at low magnification are shown, and white arrows indicate tumor colonies (A). Histological findings in high-magnification views reveal liver metastasis (E, F: black arrows). Tumor colonies are divided into two groups based on size at a cutoff of 1 mm, and the number of tumor colonies in each group are counted (total group, ≥ 1 mm group, and < 1 mm group) (G). Average tumor area in WT mice and hIL-1α cTg mice (H). *p<0.05. A, C, E: WT mice; B, D, F: hIL-1α cTg mice. Bar = 2 mm (C, D); Bar = 100 μm (E, F). Abbreviation: WT, wild-type C57BL/6.
CD8+ T-cells Were Abundant Around and Within the Tumor in hIL-1α cTg Mice
To assess the localization of CD8+ T-cells in the liver from WT and hIL-1α cTg mice at 30 days after EL4 injection, immunostaining for CD8+ T-cells was performed. The total number of CD8+ T-cells was significantly larger in hIL-1α cTg mice than in WT mice. Regardless of the non-tumor or tumor area, the number of CD8+ T-cells was significantly larger in hIL-1α cTg mice than in WT mice (Fig. 3A). CD8+ T-cells were hardly seen in WT non-tumor areas (Fig. 3B), but scattered in hIL-1α cTg non-tumor areas (Fig. 3C). In WT tumor areas, CD8+ T-cells were scattered only at the tumor edge and few were present within the tumor (Fig. 3D), whereas in the hIL-1α cTg tumor area, CD8+ T-cells were found not only at the tumor margin but also within the tumor (Fig. 3E). To evaluate the number of CD8+ T-cells within blood vessels in WT (Fig. 4A) and hIL-1α cTg mice (Fig. 4B), blood vessels were divided from hepatocytes or tumors (black dotted line). The ratio of CD8+ T-cells to blood vessel area (mm2) was significantly higher in hIL-1α cTg mice than in WT mice (Fig. 4C).
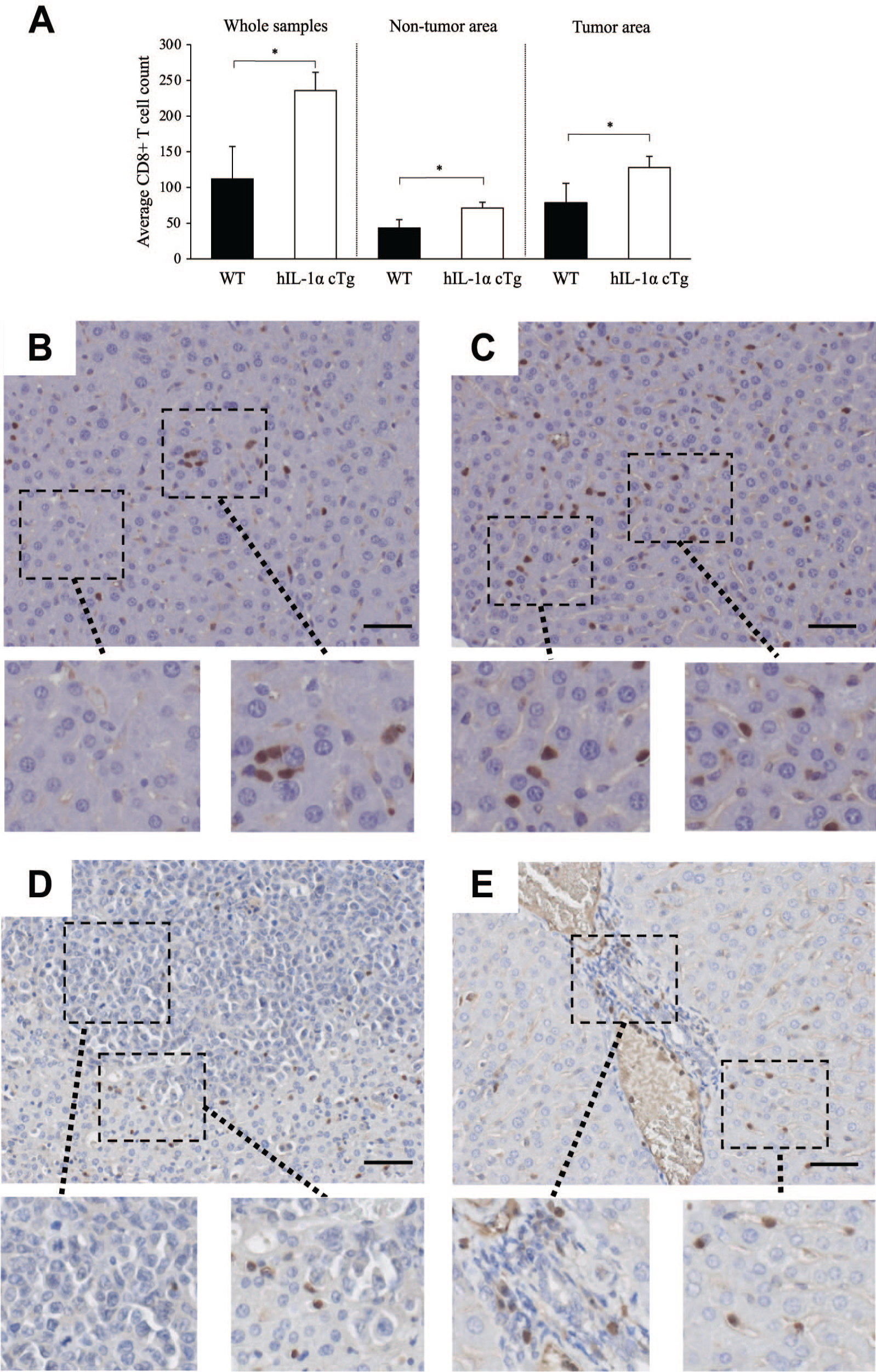
Localization of CD8+ T-cells after injection of EL4. The number of CD8+ T-cells in whole samples, tumor area, and non-tumor area (A). Immunostaining with anti-CD8 antibody (ab203035; Abcam) is used to identify the localization of CD8+ T-cells in non-tumor and tumor areas from WT mice (B, D) and hIL-1α cTg mice (C, E). B, C: non-tumor area; D, E: tumor area. *p<0.05. Bar=50 μm. Abbreviation: WT, wild-type C57BL/6.
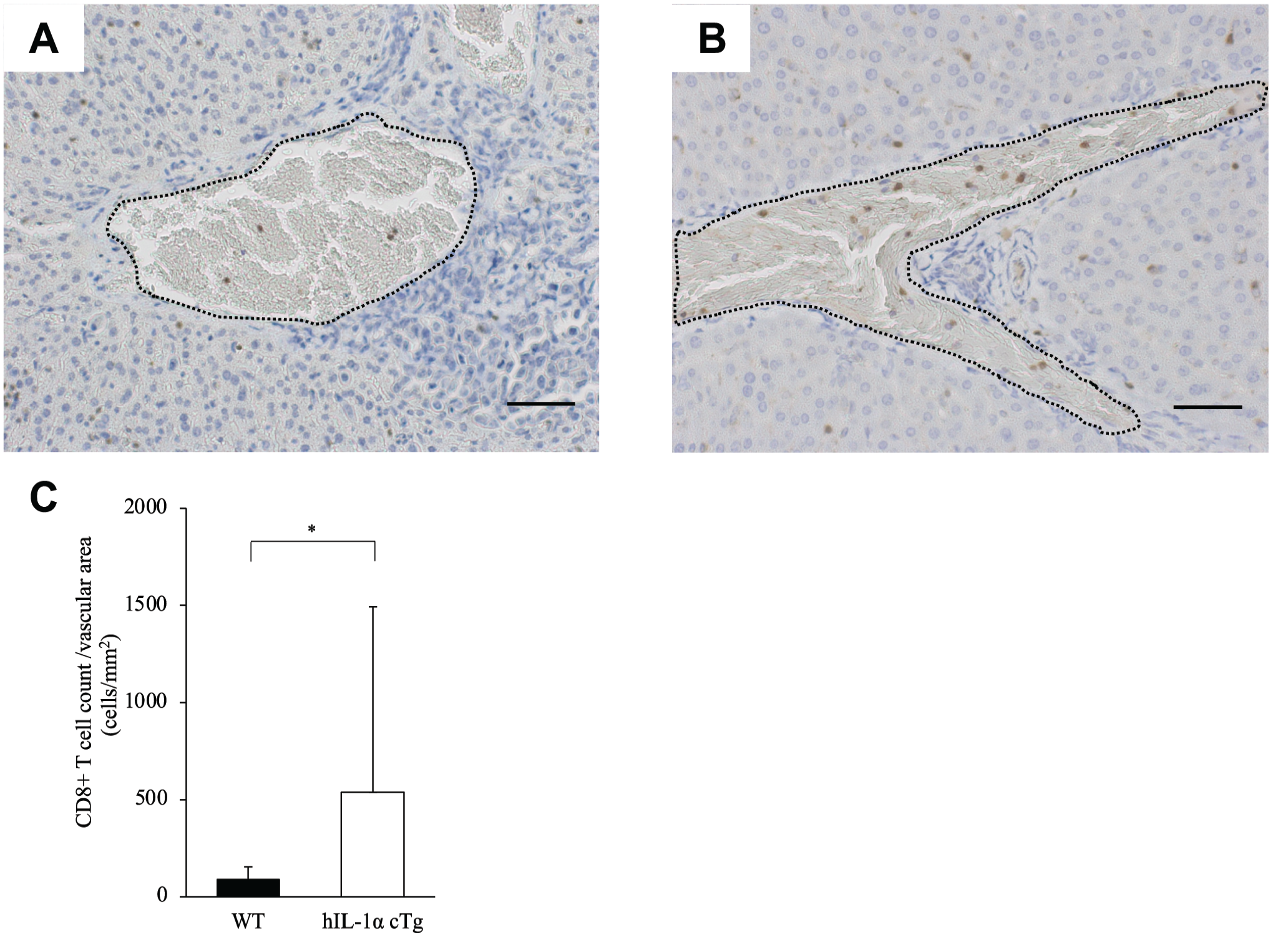
CD8+ T-cells in liver vessels. Immunolocalizations of CD8+ T-cells in liver vessels around tumor are shown in WT mice (A) and hIL-1α cTg mice (B). Ratio of CD8+ T-cell count to vascular area (mm2) in WT mice and hIL-1α cTg mice (C). *p<0.05. Bar = 50 μm. Abbreviation: WT, wild-type C57BL/6.
Abundant Apoptotic Cells Were Confirmed in Early-stage Metastasis in hIL-1α cTg Mice
Next, TUNEL staining of livers from WT and hIL-1α cTg mice was conducted at 30 days after EL4 injection. TUNEL staining indicated that the number of apoptotic cells in the tumor was quite low in WT mice (Fig. 5A), whereas in hIL-1α cTg mice abundant apoptotic cells existed in the early stage of tumor metastases recognized around the liver vessels (Fig. 5B). We then calculated the tumor area in the corresponding areas stained with H&E in WT mice (Fig. 5C) and hIL-1α cTg mice (Fig. 5D). The ratio of TUNEL-positive cells to tumor area (mm2) was significantly higher in hIL-1α cTg mice than in WT mice (Fig. 5E).
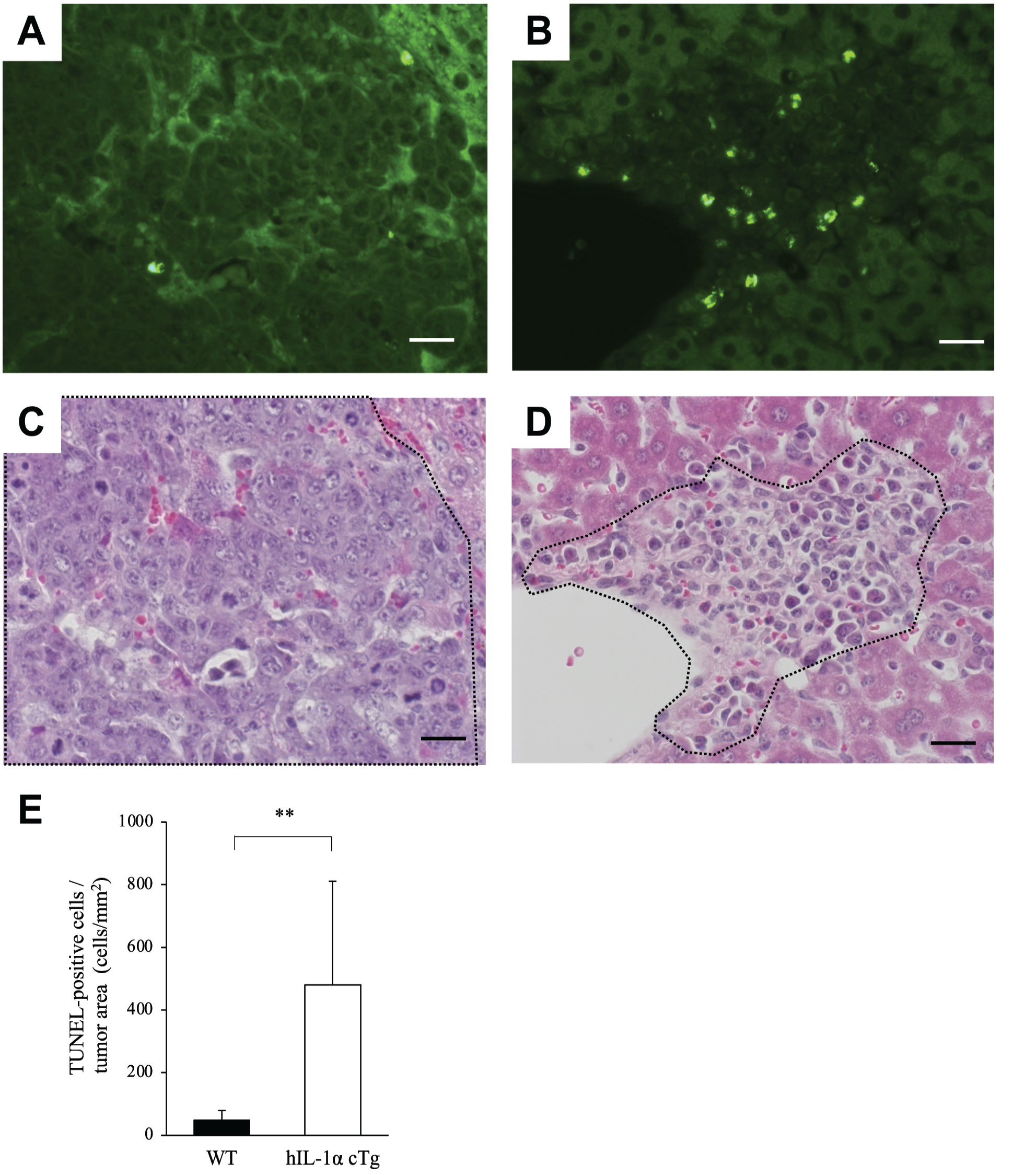
Apoptotic cells in metastatic tumor. TUNEL staining shows apoptotic cells in metastatic tumor in WT mice (A) and hIL-1α cTg mice (B). Hematoxylin and eosin staining in the corresponding areas in WT mice (C) and hIL-1α cTg mice (D) is shown, and the black dotted line is used to divide the tumor area and normal area. TUNEL-positive cell to tumor area (mm2) ratio in WT mice and hIL-1α cTg mice (E). **p<0.01. Bar = 20 μm. Abbreviations: TUNEL, terminal deoxynucleotidyl transferase dUTP Nick-end labeling; WT, wild-type C57BL/6.
CD8+ T-cells From hIL-1α cTg Mice Exhibited Cytotoxicity Against EL4
To assess the cytotoxicity of CD8+ T-cells against EL4, we isolated CD8+ T-cells from tumor-bearing hIL-1α cTg mice and WT mice (n=4). A cytotoxicity assay for CD8+ T-cells against EL4 was then performed. The results showed that tumor cytotoxicity by CD8+ T-cells from tumor-bearing hIL-1α cTg mice with an E/T ratio of 80:1 was significantly higher than that of mice with an E/T ratio of 20:1 (Fig. 6A), whereas such trend was lacking in CD8+ T-cells from WT mice (Fig. 6B). However, there was no significant difference in cytotoxicity against EL4 by CD8+ T-cells between tumor-bearing hIL-1α cTg mice and tumor-bearing WT mice.
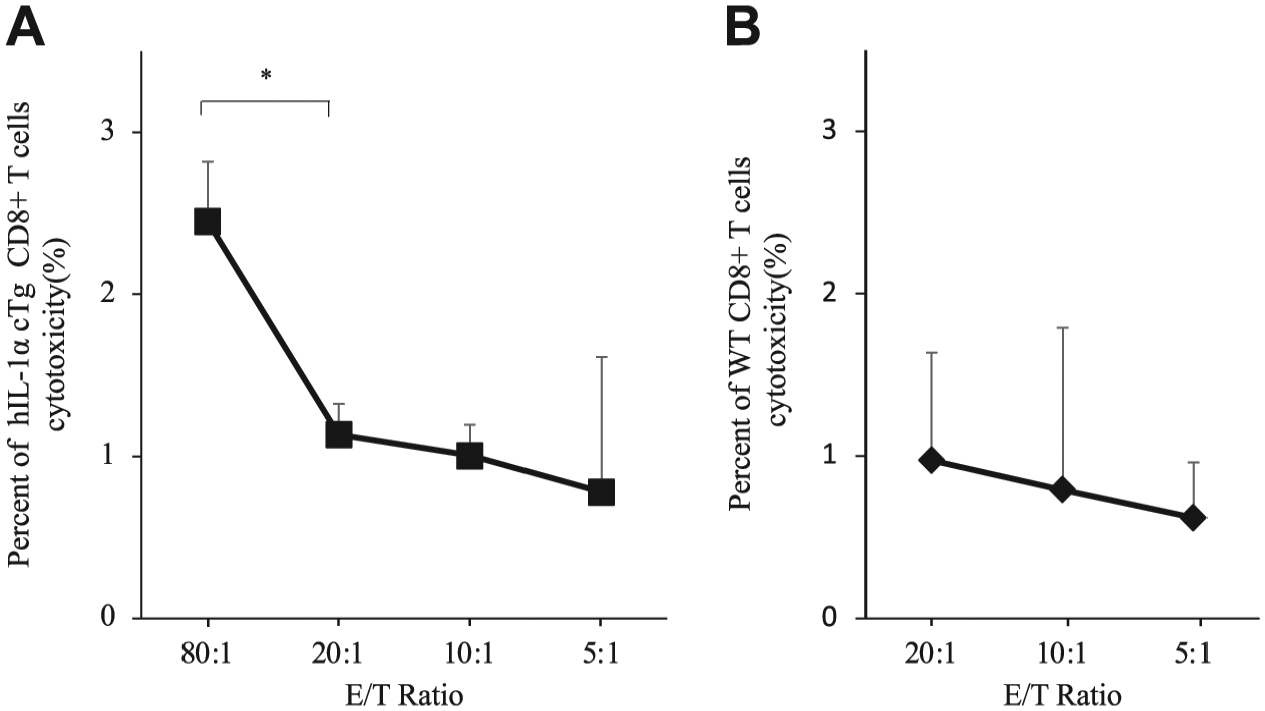
Cytotoxic assay of CD8 T-cells against EL4. Tumor cytotoxicity against EL4 of CD8+ T-cells, from tumor-bearing hIL-1α cTg mice at E/T ratios of 80:1, 20:1, 10:1, and 5:1 (A) and from WT mice at E/T ratios of 20:1, 10:1, and 5:1 (B). Data represent mean CD8 T-cell tumor cytotoxicity ± SE. *p< 0.05. Abbreviation: E/T, effector:target ratio; WT, wild-type C57BL/6.
Discussion
This study showed that IL-1α overexpression in mice suppressed tumor in the EL4 lymphoma model, and the number of CD8+ T-cells in both non-tumor area and tumor area was higher in hIL-1α cTg mice than in WT mice. These findings suggested that IL-1α exerted antitumor effects and CD8+ T-cells appeared to play an important role in tumor suppression. In addition, the IL-1α-induced inflammatory state increased the number of CD8+ T-cells in liver before and after EL4 cell transplantation, and abundant CD8+ T-cells infiltrated blood vessels. Moreover, hIL-1α protein was specifically detected in liver cells from hIL-1α cTg mice, and the increase in the number of CD8+ T-cells from tumor-bearing hIL-1α cTg mice correlated with higher cytotoxicity against EL4. Accordingly, hIL-1α-induced CD8+ T-cells are speculated to suppress not only tumor metastasis but also the growth of metastatic tumor. In clinical settings, patients treated with IL-1 receptor antagonist showed a higher incidence of lymphoma compared with the expected rate in the general population, which may support our findings. 18
Generally, inflammation plays two contrasting roles in the development of cancer. Chronic inflammation has been perceived as a predisposing factor for cancer development. Approximately 25% of cancers are associated with chronic inflammation caused by infection or physicochemical agents. 19 In contrast, a certain degree of inflammation is considered to arise as a defensive response by the body and has been reported to promote anticancer effects. Currently, Mycobacterium bovis bacillus Calmette-Guérin (BCG)-induced acute inflammation is used in the therapy of squamous cell cancer of the bladder. 20 These contrasting roles of inflammation in cancer development correspond to the effects of proinflammatory cytokines such as IL-1. IL-1α has been reported to form a key component of feed-forward signaling amplification loops during malignant transformation, tumor progression, and metastasis,21–23 whereas IL-1α in lymphoma and fibrosarcoma is expressed in the cytosol in concordance with the expression of membrane-associated IL-1α, and IL-1α-expressing tumor cells fail to induce tumor progression in intact mice or manifest initial tumor growth followed by regression.10,24 Membrane-associated IL-1α with limited secretion into the tumor microenvironment favors the induction of antitumor cell immunity,12,25 which may be the case with our model of liver lymphoma metastasis in hIL-1α cTg mice, as membrane-associated IL-1α has previously been reported to be overexpressed in hIL-1α cTg mice, contributing to the progression of arthritis. 26 In this study, CD8+ T-cells could be activated by contact with membrane-associated IL-1α, and activated CD8+ T-cells could suppress metastatic tumors.
CD8+ T-cells are key players in adaptive immune responses and have been shown to be crucial for protective immune responses against a wide range of tumors through cytokine expression and cytolytic activity. IL-1 enhances adhesion and transmigration of CD8+ T-cells into various tissues by controlling chemokine production and possibly adhesion molecule expression.27–30 Moreover, IL-1 stimulates antigen-specific CD8+ T-cell expansion, differentiation, and migration to the periphery in the spleen, lymph node, and liver. 15 Tumor regression of IL-1α-positive fibrosarcoma cells reportedly depends on activated CD8+ T-cells. 10 In this study, the number of CD8+ T-cells in the liver was significantly increased in hIL-1α cTg mice compared with WT mice, which may have contributed to the increased ratio of TUNEL-positive cells to tumor area. The hIL-1α protein was specifically detected in liver cells from hIL-1α cTg mice, and the number of CD8+ T-cells from tumor-bearing hIL-1α cTg mice correlated with the cytotoxicity to EL4. These findings suggest that the increase in CD8+ T-cells in the liver from hIL-1α cTg mice would correlate with the overexpression of hIL-1α, and the CD8+ T-cells may play immunosuppressive roles for EL4.
In fact, increment of CD8+ T-cell density was recognized not only at the tumor periphery but also within the tumor in hIL-1α cTg mice. The number of CD8+ T-cells was also increased in the vessels of hIL-1α cTg mice, suggesting that CD8+ T-cells were recruited from outside the liver. Chen et al. 31 revealed that the density of CD8+ T-cell infiltration around the tumor was important, and a high density of CD8+ T-cells in a tumor may predict better therapeutic outcome. In addition, apoptotic cells existed around liver vessels in both WT and hIL-1α cTg mice. Although apoptotic cells were identified in tumors around vessels in WT mice, those around liver vessels in hIL-1α cTg mice were observed where tumor was not clearly apparent. Moreover, apoptotic cells existed mainly at the edge of large tumors and could not be identified in large tumors in WT mice. However, in hIL-1α cTg mice, many apoptotic cells were seen not only around the tumor but also within the tumor (Supplemental Fig. 1). The localization of CD8+ T-cells around the tumor was similar to that of apoptosis cells. Combined with the histological findings of the liver in hIL-1α cTg mice after PolyI-PolyC injection, CD8+ T-cells were constitutively activated in the liver before transplantation of EL4 cells and could play critical roles in suppressing tumor metastasis.
Finally, some limitations must be taken into consideration when interpreting the results of this study. First, the mechanisms underlying IL-1α-induced CD8+ T-cell infiltration in metastatic tumor remain to be clarified. To date, toll-like receptor signal, CCR5 receptor, and CCR5 ligands have been associated with the expansion of CD8+ T-cells32,33 and differentiation into cytotoxic CD8+ T-cells.34,35 Second, due to the small number of CD8+ T-cells from WT mice, we could not perform cytotoxicity assays with sufficiently large numbers of CD8+ T-cells. Third, the error bars for tumor colonies of WT mice were very large. As we identified statistical significance using non-parametric tests, the differences between WT and hIL-1α cTg mice were still considered significant. In addition, the large error bars for WT mice were mainly caused by large (≤1 mm) tumor colonies and may reflect tumor immunity in each WT mouse. In fact, apoptotic cells were identified not in large tumors, but in small tumors around liver vessels in WT mice (Supplemental Fig. 1). Moreover, we did not perform analysis with IL-1 antagonism to confirm the immunosuppressive effects on IL-1. Due to the constant overexpression of hIL-1α in peripheral blood and the lack of information about the appropriate dose of IL-1 antagonism for hIL-1α cTg mice, inhibiting the effects on hIL-1α using IL-1 antagonism would be hard. Finally, previous reports have shown that IL-1α activated not only CD8+ T-cells but also other immune cells such as natural killer cells and macrophages,24,36 and correlated with G0–G1 cell cycle arrest in tumor cells.37,38 In this study, we detected the correlation between the number of CD8+ T-cells and high tumor cytotoxicity to EL4, and confirmed higher number of CD8+ T-cells in the liver from hIL-1α cTg mice than those from WT mice. From this perspective, we believe that CD8+ T-cells activated by hIL-1α might play an immunological role in suppressing EL4. However, the effect of other immune cells would also impact our results. Further experimental studies of hIL-1α cTg mice are warranted to gain a better understanding of the mechanisms underlying IL-1-induced CD8+ T-cell activation and subsequent tumor regression.
In conclusion, IL-1α suppresses EL4 metastasis, and IL-1α-activated CD8+ T-cells may play an important role in inhibiting both tumor metastasis and metastatic tumor growth.
Supplemental Material
sj-pdf-1-jhc-10.1369_0022155421991634 – Supplemental material for Overexpression of Interleukin-1α Suppresses Liver Metastasis of Lymphoma: Implications for Antitumor Effects of CD8+ T-cells
Supplemental material, sj-pdf-1-jhc-10.1369_0022155421991634 for Overexpression of Interleukin-1α Suppresses Liver Metastasis of Lymphoma: Implications for Antitumor Effects of CD8+ T-cells by Kazuhiko Udagawa, Yasuo Niki, Toshiyuki Kikuchi, Yusuke Fukuhara, Yuki Takeda, Takeshi Miyamoto, Morio Matsumoto and Masaya Nakamura in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
We thank Dr. Tami Kobayashi and Dr. Yukiko Sato in preparing animals for experiments. We are also grateful to Hiroya Kanagawa, Kana Miyamoto, Atsuhiro Fujie, Akihito Oya, and Go Itakura for technical assistance with the experiments.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
KU, YN, and TK performed the research. KU, YN, TK, and TM conceived and designed the research. YF, YT, MM, and MN edited and reviewed the article. KU and TK analyzed the data. KU and YN wrote the article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
