Abstract
In the vertebrate cochlea, the reticular lamina seals the organ of Corti against the endolymph filled scala media. After noise exposure, fast alterations in the endothelial nitric oxide synthase (eNOS) expression level were identified in this cochlear structure. Minor amounts of nitric oxide (NO) produced by eNOS or applied by NO donors such as S-nitroso-N-acetyl-penicillamine (SNAP) might protect this vulnerable part of the organ of Corti, on the line of gap junctions of supporting cells and cochlear microcirculation. In n=5 anesthetized guinea pigs, SNAP was intravenously applied in two concentrations. Six untreated animals served as controls. The cochleae were removed and prepared for immunoelectron microscopy using specific gold-labeled anti-eNOS antibodies. The density of the gold particles was quantified for seven cellular regions in the reticular lamina at the ultrastructural level. Following SNAP application, a significant increase in eNOS expression (+176%) was detected compared with controls (p=0.012). The increase occurred mainly in actin-rich cuticular structures and the prominent microtubules bundles. Correlation analysis revealed three clear and five moderate cellular associations for controls, whereas only one clear and one moderate after SNAP application. Thus, application of the NO donor SNAP resulted in an increase in eNOS expression in distinct regions of the reticular lamina:
Introduction
The small signal molecule nitric oxide (NO) is formed enzymatically by three nitric oxide synthase (NOS) isoforms. The endothelial nitric oxide synthase (eNOS) and the neuronal nitric oxide synthase (nNOS) are constitutively expressed in the inner ear, whereas the inducible nitric oxide synthase (iNOS) has only been found so far in pathological conditions. 1 The constitutively expressed eNOS isoform produces low concentrations of NO, whereas iNOS induction results in significantly higher NO concentrations. 1
Within the mammalian cochlea, three different NO-dependent regulatory systems might substantially influence inner ear homeostasis: (1) the epithelial gap junctions, (2) blood vessel perfusion, and (3) neural synaptic interactions. Gap junctions are channels within the cell membrane which are formed by two semi-channels from neighboring cells, each with a pore diameter of 1–1.5 nm and which enable communication between cells. An increase in NO concentration resulted in a fast uncoupling of the gap junctions in Deiters’ cells (DC). 2 The regulatory capacity of gap junctions by NO was also demonstrated for neurons in the central nervous system. 3
In cochlear microcirculation, the pressure conditions within the vessels are influenced locally by the NO formation in endothelial cells and pericytes. The NO-producing eNOS isoform was identified among others in the endothelial cells of the strial capillaries and in the blood vessels of the spiral ligament.4,5 Furthermore, it was shown that pericytes also contributed to the local regulation of microcirculation by producing large amounts of NO.6,7
In relation to synaptic signaling, a low concentration of NO played a protective role in glutamate neurotoxicity by closing the NMDA receptor-gated ion channels8,9 and by influencing the release of dopamine. 10
In the inner ear of adult chinchillas, direct SNAP application into the cochlea via the round window membrane resulted in a significant ABR threshold elevation after 5 hr and elevated nitrate/nitrite levels in the perilymph. 11 These findings imply that an increased concentration of an NO donor will result in severe sensorineural hearing loss and otitis media. 11 However, during intracochlear perfusion or intravenous application of SNAP by our group, it was shown that the cochlear potentials were not altered in the guinea pig model, but significant alterations in the systemic blood pressure were detected after intravenous injection. 12 At the electron microscopic level, SNAP application resulted in an increase in anti-eNOS immunoreactions at the apical side of the organ of Corti. 12 In addition, it was shown that SNAP enhanced the ATP-induced Ca2+ increase of the intracellular concentrations in outer hair cells (OHC) 13 and in spiral ganglion neurons of the guinea pig. 14 In addition, the protective effect of low NO concentrations was discussed after intense noise exposure for the reticular lamina in guinea pigs. 15 This cochlear region is strongly exposed to oscillating forces in the context of sound perception and signal transduction.16,17
Anatomically, the reticular lamina consists of the apical regions of hair cells and neighboring non-sensory supporting cells, including DC, pillar cells (PC), and inner phalangeal cells, which are tightly connected by junctional proteins, thus forming a physical barrier separating the differing ionic contents of the cochlea, endolymph, and perilymph. 18
To obtain information about possible intercellular communication pathways and interactions between SNAP application and cellular eNOS expression, we determined alterations in eNOS immunostaining intensities in seven areas of the reticular lamina at the electron microscopic level by quantifying gold-labeled anti-eNOS antibodies. The results are discussed in view of NO-dependent mechanisms in tissue protection.
Materials and Methods
Subjects
Eleven healthy pigmented male guinea pigs (Tricolor; Charles River, Sulzfeld, Germany) weighing 175–260 g with good Preyer’s reflexes and no evidence of middle ear disease were used in this study. All experiments were conducted following the German Prevention of Cruelty to Animals Act and were approved by the supervising authorities. Animals were kept on a 12 hr:12 hr light:dark cycle in the university’s animal facility.
Cochlea Preparation
All animals were anesthetized with chlorprothixene 30 mg/kg i.p. and pentobarbital (Narcoren; Hallbergmoos, Germany) 25 mg/kg i.p., tracheotomized, and given artificial respiration. SNAP concentrations of 1 mmol/l (n=3) and 10 mmol/l (n=2) were given intravenously.
Thirty minutes after SNAP application, the animals were killed by pentobarbital (448 mg/kg body weight). All cochleae were completely removed from the bullae and transferred into a solution that consisted of 0.2% picric acid, 4% para-formaldehyde, and 0.1% glutardialdehyde and embedded in LR White. We used this short time period between application and fixation, as we were interested in the fast processes in the cochlea. Comparable fast cellular responses were already detected after noise exposure for eNOS expression, 19 COX-2 expression, 20 and Akt expression. 21
As the human ear is most sensitive between 2 and 5 kHz corresponding to the second cochlea turn, 22 we used this turn for our analyses.
Postembedding Immunolabeling
Postembedding immunolabeling was performed using ultrathin tissue sections of the cochleae as previously described. 23 Briefly, the sections were incubated overnight at 4C with the primary monoclonal mouse IgG1 eNOS antibody (Transduction Laboratories; Lexington, KY). The antigenicity of the tissue was tested by labeling ultrathin sections with an antitubulin (monoclonal anti-β-tubulin antibody; Sigma Aldrich, St. Louis, MO, diluted 1:500, data not shown).
First, we tested different solutions of the anti-eNOS antibody. At dilutions of 1:750 and 1:1000, similar reaction patterns were obtained with no statistically significant difference in the number of gold-labeled anti-eNOS antibodies for untreated control ears and after SNAP injection. Therefore, the results of both SNAP concentrations were considered together. In addition, no distinction was made between first and repeated labeling procedures as they led to comparable staining patterns (see statistical approaches below).
Positive and Negative Incubation Controls
In addition, control experiments were performed by incubating the ultrathin sections with an isotype IgG antibody (Cymbus Biotechnology; Chandlers Ford, UK) and by labeling ultrathin sections of the neocortex of the prenatal mouse (positive control) and vitellogenic follicles of the fruit fly Drosophila (negative controls) with the primary antibody (data not shown). The immunoelectron microscopic analysis revealed that the eNOS labeling of the ooplasma in vitellogenic follicles (0.125 gold particles/µm2) was in the range of cochlea background labeling. It is widely accepted in the quantitative immunoelectron microscopic analysis that the background labeling must be below 1 gold particle/µm2. In the cells of the neocortex, a density of 2.31 gold-labeled anti-eNOS antibodies/µm2 was quantified that corresponded to unstimulated OHC eNOS immunoreactivity. For all staining protocols, negative controls were included (i.e., no primary antibody). An additional control for the specificity of cell labeling is inherent in the method itself, as the gold particles present in the embedding resin or acellular structures of the organ of Corti, such as the basilar and tectorial membrane, represent nonspecific background labeling.
Quantification of Gold Particles
The density of gold particles was determined on various ultrathin sections in a frame of 1 µm2 (see Fig. 1). At higher magnification, the electron-dense gold particles can be identified on the unstained ultrathin section (see Fig. 1B). The number of gold particles was counted in seven different cell areas or cell types: cuticular plates of inner and outer PC (grouped together as P cu), the microtubule-rich area of inner PC (IP MT), the cytoplasm of apical DC (DA pl), cuticular structures of apical DC (D cu), the cytoplasm of inner hair cells (IHC pl), the cytoplasm of OHC (OHC pl), and cuticular plates of OHC (OHC cu). The distribution of gold-labeled anti-eNOS antibodies was determined on ultrathin sections of six control animals, three animals which were treated with 1-mM SNAP, and two animals which had received 10-mM SNAP. In total, gold particles in 9928 arbitrarily selected areas were evaluated.
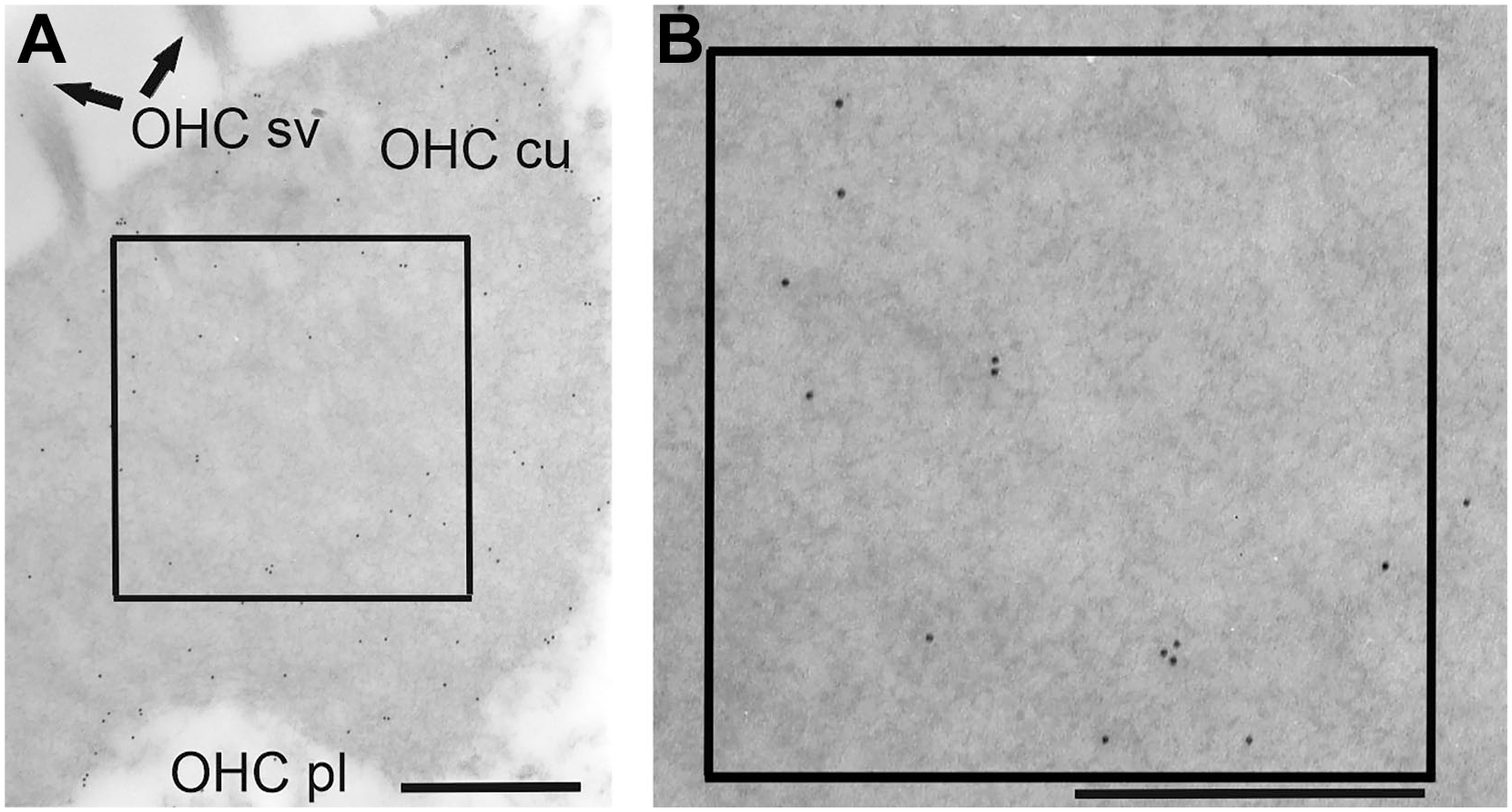
Electron microscopic pictures of the apical side of an outer hair cell in the second turn of the cochlea demonstrating the method of quantification of gold-labeled anti-eNOS antibodies. (A) Cuticular plate of an outer hair cell. (B) Enlargement of the inlets visualizing gold particles in a 1-µm2 area. Scale bar = 500 nm. Abbreviations: eNOS, endothelial nitric oxide synthase; OHC cu, cuticular plate of the outer hair cell; OHC pl, cytoplasm of the outer hair cell; OHC sv, stereovilli of the outer hair cell.
Statistics
Analyzing the amount of gold-labeled anti-eNOS antibodies in different cell regions of the reticular lamina, we applied a negative binomial mixed model. For this model, we used treatment, region, and dilution plus an interaction term between treatment and region as fixed effects and animal as a random effect. The variable treatment had two levels (control or SNAP), the variable region had seven levels, and the variable dilution had two levels (1:750 or 1:1000). The mean values and the standard deviations were computed per treatment, region as well as animal and visualized in graphs. The corresponding p values were derived from an analysis of variance and post hoc tests with adjustment for multiple comparisons via Bonferroni–Holm correction. To determine cellular associations, the amount of gold-labeled anti-eNOS antibodies was averaged over a specific animal, dilution, and experiment (first or repeated) for each region. The associations were described by Pearson correlation coefficients (Pearson’s r) and related p values, testing whether the correlation is different from zero. These p values were not adjusted for multiple comparisons and should be regarded as descriptive. Statistical analysis was performed using R, a language and environment for statistical computing. 24
Results
Localization of Gold-labeled eNOS Antibodies
An increase in the amount of gold-labeled anti-eNOS antibodies was detected after SNAP application in all analyzed areas. Gold particles were seen in the microtubule-rich area of IP MT and the underlying cuticular plate (IP cu) in untreated controls (see Fig. 2A). After SNAP application, the number of gold particles was higher in both regions when compared with control (see Fig. 2B).
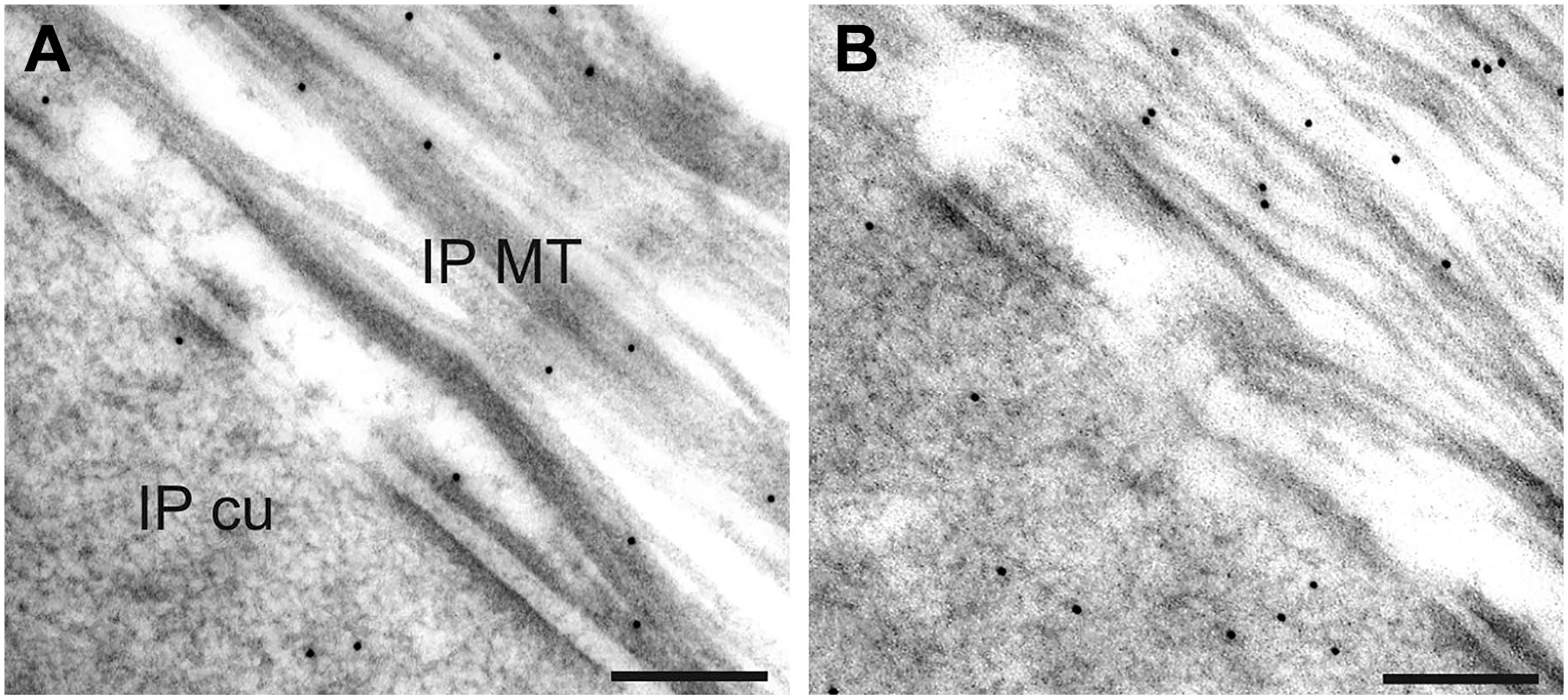
Immunoreactions in cuticular plates and the microtubule-rich area of inner pillar cells. Gold-labeled anti-eNOS antibodies were found in both areas (A). After SNAP application, the number of immunoreactions was increased in both areas (B). Abbreviations: eNOS, endothelial nitric oxide synthase; IP cu, cuticular plates of the inner pillar cell; IP MT, microtubule-rich area of the inner pillar cell; SNAP, S-nitroso-N-acetyl-penicillamine. Scale bar = 200 nm.
Individual Differences in Anti-eNOS Immunoreaction
Quantifying the number of gold-labeled anti-eNOS antibodies at the level of the individual ear, differences in the amount of eNOS immunoreaction were identified in control ears and SNAP-treated ears (see Fig. 3). In control ears, the mean individual variability in eNOS expression was between 2 and 4 gold particles/µm2. After SNAP injection, individual differences were found between 3 and 6 gold particles/µm2. In the SNAP-treated group, the experimental group can be classified in responders (S1, S2, S11, S12) and non-responders (S3) which was in the range of controls. Comparing both groups, the number of the gold-labeled anti-eNOS antibodies was 1.76× higher after SNAP treatment (p=0.012).
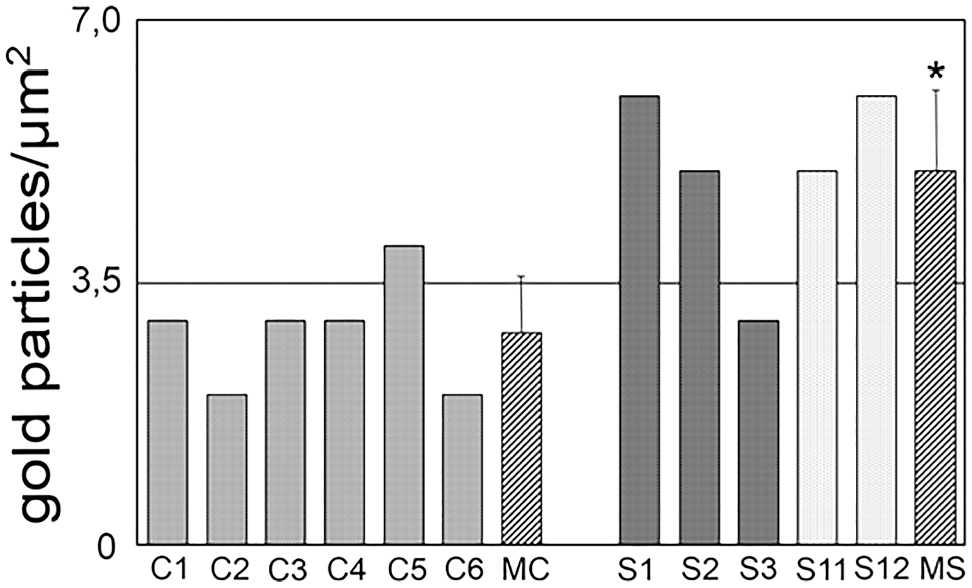
Averaged number of gold particles at the level of the individual ear in six controls (C1–C6), in three animals after application of 1-mM SNAP, and in two animals after application of 10-mM SNAP. The mean density of gold particles was 1.76× higher in the grouped SNAP-treated ears (MS) compared with controls (MC). Abbreviation: SNAP, S-nitroso-N-acetyl-penicillamine. *p<0.05.
Cellular Alterations of Gold-labeled Anti-eNOS Antibodies
Analyzing the differences of gold-labeled anti-eNOS antibodies between controls and SNAP-treated ears for the seven different areas, a clear increase was detected after SNAP treatment in all analyzed areas. For a better overview, the important cell regions of the reticular lamina where gold-labeled anti-eNOS antibodies were quantified are shown in a schematic drawing (see Fig. 4A). The actin-containing structures are marked by gray shades in the figure and are localized in IHC and OHC, DA pl, OP cu, and IP cu (see Fig. 4A).
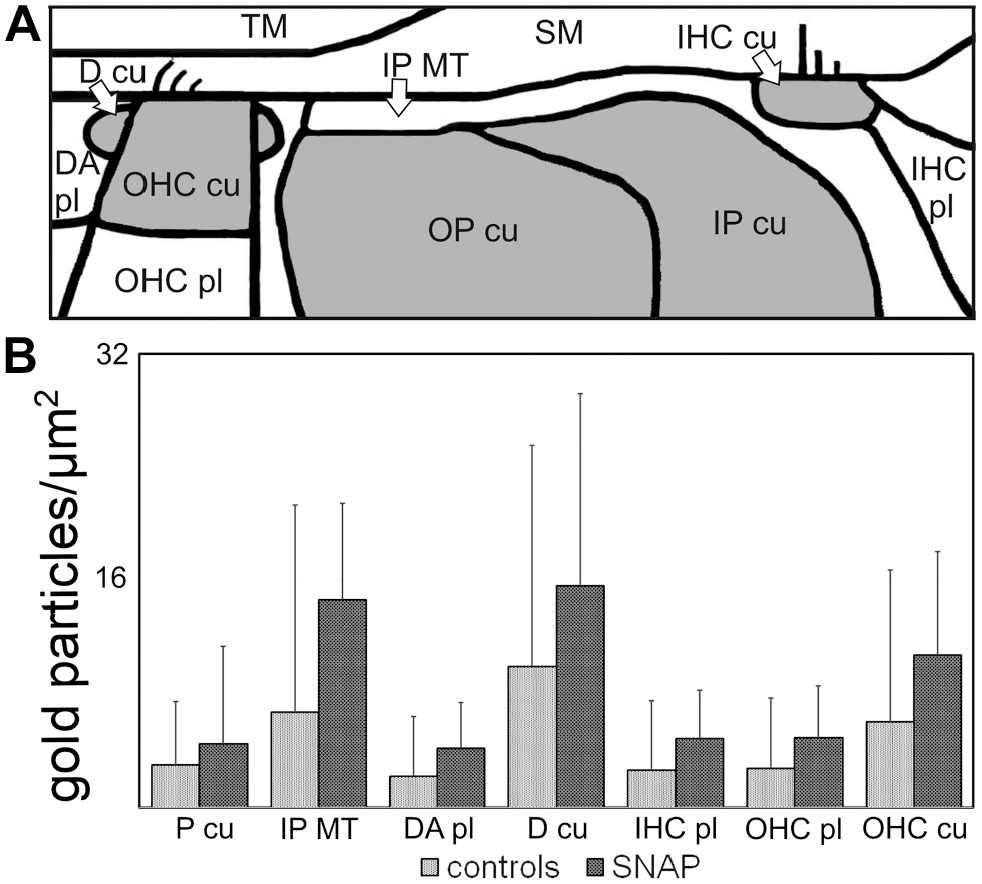
Quantification of gold-labeled anti-eNOS antibodies in seven regions of the reticular lamina. Schematic drawing of the reticular lamina with the analyzed areas (A) and quantification of gold-labeled eNOS antibodies in controls and after SNAP application (B). Abbreviations: eNOS, endothelial nitric oxide synthase; D cu, cuticular structure of the apical Deiters’ cell; DA pl, cytoplasm of the apical Deiters’ cell; IHC cu, cuticular plate of the inner hair cell; IHC pl, cytoplasm of the inner hair cell; IP cu, cuticular plate of the inner pillar cell; IP MT, microtubule-rich area of the inner pillar cell; OHC cu, cuticular plate of the outer hair cell; OHC pl, cytoplasm of the outer hair cell; OP cu, cuticular plate of the outer pillar cell; P cu, cuticular plates of pillar cells (values of the cuticular plates of inner and outer pillar cells were grouped together); SM scala media; TM, tectorial membrane; SNAP, S-nitroso-N-acetyl-penicillamine. *p<0.05, ***p˂0.001.
The highest values of SNAP-induced eNOS expression were identified in D cu and in the microtubule bundles of IP MT which are found between P cu and the scala media (see Fig. 4B). In P cu and D cu, the increase was slightly significant (p=0.041), but for all other regions clearly significant (p˂0.001).
Cellular Associations of eNOS Immunostaining Intensity
To obtain information about any interplay of cellular eNOS expression between the seven analyzed areas, the associations were determined at the level of the individual ear. In controls, the statistical analysis revealed three clear and five moderate positive associations of eNOS expression in controls (Fig. 5). Clear associations were seen between P cu, IHC pl, and OHC cu. In SNAP-treated animals, only one strong association and one moderate association were detected. The strong cellular association was found between P cu and D cu which was a moderate association in controls. The moderate association after SNAP was seen between OHC cu and PC cu which was a clear association in controls. Thus, there were quantitative and qualitative alterations in the cellular association pattern of eNOS expression.
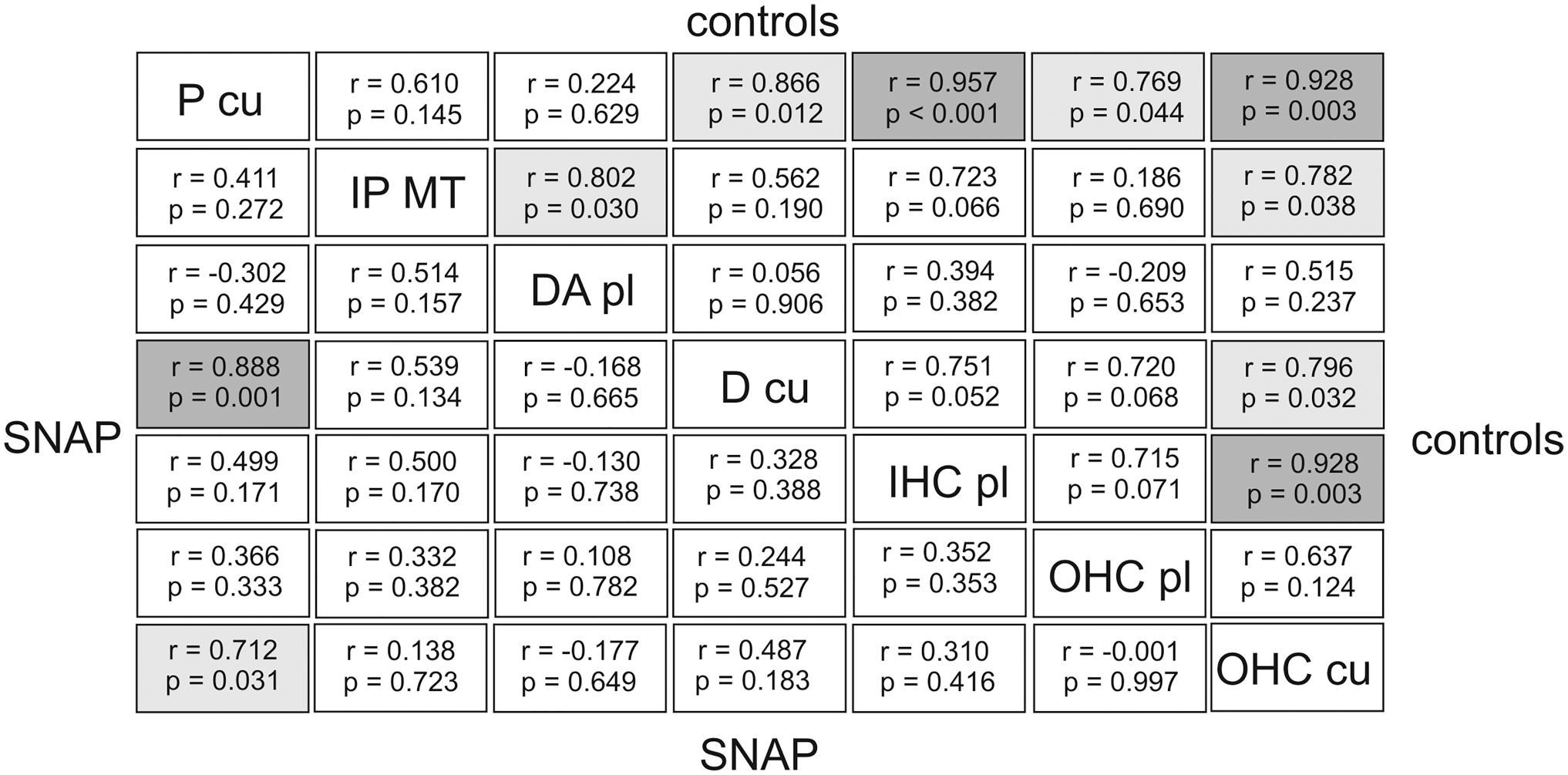
Cellular associations(Pearson’s r and p values) of gold-labeled anti-eNOS antibodies in control ears and after SNAP application. Comparing the seven selected areas of the reticular lamina in controls and after SNAP application clear associations were detected revealing a well- coordinated interplay in eNOS expression between distinct areas. In controls there was clear interaction between cuticular plates of pillar cells, cytoplasm of inner hair cells, and cuticular plates of outer hair cells. After SNAP application an interplay in eNOS expression was observed between cuticular structures of Deiters cells and cuticular plates of pillar cells. Abbreviations: eNOS, endothelial nitric oxide synthase; D cu, cuticular structure of the apical Deiters’ cell; DA pl, cytoplasm of the apical Deiters’ cell; IHC pl, cytoplasm of the inner hair cell; IP MT, microtubule-rich area of the inner pillar cell; OHC cu, cuticular plate of the outer hair cell; OHC pl, cytoplasm of the outer hair cell; P cu, cuticular plate of the inner and outer pillar cell; SNAP, S-nitroso-N-acetyl-penicillamine.
Discussion
In this study, we were able to demonstrate that SNAP application resulted in an increase in eNOS immunostaining intensity in all analyzed areas of the reticular lamina. In control ears, three clear cellular associations of eNOS expression were identified, for example, between P cu, IHC pl, and OHC cu, revealing potential communication pathways between these areas. In SNAP-treated ears, two of these clear associations were absent, while one association was changed from a clear to a moderate one. After SNAP application, another clear association was found between cuticular plates of PC and D cu. These observations point to an important function of the actin-containing cuticular structures in controlling eNOS expression in the cochlea which can be influenced by an NO donor.
The Importance of the Reticular Lamina
The reticular lamina serves as an ion barrier mainly formed by tight junctions, 25 whereas adherens junctions and the cytoskeleton are responsible for the integrity and mechanical resistance of tissues. 18 The reticular lamina is characterized particularly by the actin-containing structures, which were identified in OHC and IHC, in PC, and in the apical DC where they form a circumferential belt. Furthermore, numerous microtubule bundles were seen in the apical cytoplasm of the IP MT between the cuticular plates and the scala media.
Focusing on a possible role of the reticular lamina role in eNOS regulation, it was demonstrated that 60 hr after noise trauma, the eNOS staining intensity had increased in the actin-rich cell structures concomitant with an intracellular Ca2+ increase in all cell types except in OHC pl. 15 These findings might be interpreted as adaptation processes in noisy conditions, where eNOS-regulated NO production might be protective. 15 In this study, a comparable increase in eNOS immunostaining intensity was found in all cellular regions of the reticular lamina, especially in the actin-rich regions after SNAP application.
Actin-dependent eNOS Regulation
In the different cell types of the mammalian cochlea, various actin isoforms were identified. Sensory and supporting cells of the mammalian organ of Corti have cytoskeletons that are formed by β- and γ-actin isoforms with clear differences between sensory and supporting cells with respect to cell type and along the length of the cochlea from the base to the apex. 26 Besides the various isoforms, the polymerization rate and the local concentration of actin monomers are an important factor which can influence actin cytoskeleton. 27 The organization of the actin cytoskeleton was found to play an important role in the post-transcriptional regulation of eNOS during cell growth. Especially the relationship between the state of actin polymerization and eNOS expression was identified as a key player in enzyme control. 27 Pharmacological perturbation of the G/F-actin ratio led to changes in eNOS expression that were consistent with the finding that monomeric actin was part of a destabilization mechanism for eNOS mRNA. 27
In human umbilical vein endothelial cells, the association of globular actin with eNOS was found to play an essential and necessary role in agonist-induced eNOS activation, through enabling its phosphorylation by Akt at serine residue 1177. 28 In addition, in the pulmonary artery endothelial cells model, it was proven that the incubation of purified eNOS with purified F-actin and G-actin resulted in an increase in eNOS activity. 29 The increase in eNOS activity caused by G-actin was much higher than that caused by F-actin, demonstrating that the polymerization state of actin plays an important role in the regulation of eNOS activity. 29
In this study, clear cellular associations between the P cu and OHC cu and the IHC pl were identified in control ears in view of the eNOS immunostaining intensity. In addition, there were clear associations between OHC cu cells and the IHC pl. The findings revealed potential communication routes of a cellular cross talk between these three areas in respect of securing a controlled eNOS expression pattern and level. However, the nature of the underlying molecular communicator is still unknown.
After SNAP application, these three clear associations had disappeared, but a new association was detected. There was a clear association between P cu and apical D cu. Consequently, the polymerization state of actin may potentially be involved in the regulation of eNOS expression.
In addition to actin, other cytoskeletal elements might also influence the eNOS expression level. Su et al.30,31 demonstrated that tubule-active agents were able to modify NO production in pulmonary artery endothelial cells. In this study, an increase in eNOS expression was identified in the IP MT which are located between the cuticular plate and the apical cell membrane facing the scala media.
SNAP Influences Gene Expression
Molloy et al. 32 were able to show by microarray analysis that the NO donor SNAP affects the expression of a wide range of genes that are important to the structure of human connective in a dose-dependent manner. Furthermore, it was shown that exogenous generation of NO by the application of the NO donor SNAP modulated iNOS gene expression and activity in isolated rat hepatocytes. 33 Thus, SNAP blocked the cytokine-induced increase in nuclear factor kappa B (NF-κB) activation, iNOS promoter activity, nuclear translocation of cytosolic NF-κB p65 subunit, and inhibitor of κB (IκB) α (IκBα) degradation. Therefore, SNAP-dependent NO supply revealed a novel negative feedback mechanism whereby NO downregulates iNOS gene expression. 33 In bovine pulmonary artery endothelial cells, exogenous NO led to a biphasic response, an early increase in intracellular calcium followed by an increase in NOS activity, and an increase in eNOS gene expression, whereas iNOS induction was not observed. 34 It was assumed that in contrast to other techniques, SNAP application results in a relatively constant concentration of NO (detected as nitrite) over the entire volume of the cells and surrounding media. 32 After preconditioning human cardiac stem cells by the NO donor diethylenetriamine NO adduct, cell survival was promoted. 35 Concomitant upregulation of several cell signaling molecules was observed, including elevated phosphorylation of NRF2, NFκB, STAT3, ERK, and Akt, as well as increased protein expression of HO-1 and COX-2. 35 It was demonstrated that NO induces three distinct waves of gene activity by using several independent signaling pathways to induce gene expression. 36
Protection After SNAP Application
A valuable effect of SNAP was demonstrated in the therapy of different diseases. A concentration-dependent protection of exogenous NO against simulated ischemia-/reperfusion-induced injury was detected in mouse embryonic stem cell-derived cardiomyocytes. 37 In this context, a soluble guanylate-cyclase, protein kinase G and KATP channels were supposed to play an important role in the downstream pathway of SNAP-induced cytoprotection.37,38 Furthermore, NO was found to protect isolated perfused hearts of mice against I/R injury by preventing the hyperdynamic response during early reperfusion. 39 In addition, administration of SNAP at 2 mM has been shown to mimic preconditioning protection in mouse hearts. 40 In cardiac H9c2 cells, SNAP-induced protection was supposed to be carried out by the induction of heat shock protein 75 expression in mitochondria. 41 Furthermore, it was shown that pretreatment with SNAP prevented H2O2-induced apoptosis in SK-N-MC human neuroblastoma cells and induced phosphorylation of Akt and Bcl-2-antagonist of cell death. 42 Thus, the application of exogenous NO offers a new therapeutic tool to support cellular protection mechanisms by maintaining microcirculation.
Systemic application of the NO donor SNAP in minor concentrations results in an increased eNOS expression in specific cell types in the reticular lamina. Future studies must examine the influence of such modulation in other cochlear regions and the benefit of NO administration on cochlear function and protection.
Footnotes
Acknowledgements
The authors thank Mrs. K. Benz for technical assistance.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
U-RH, RHS, and SS helped in conception and design of the study. U-RH, RM, BPE, IS, RHS, and SS helped in acquisition, analysis, and interpretation of data for the work. DD helped in critical revision for important intellectual content. Each author has read and approved the article before submission.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was realized by funds of the Deutsche Forschungsgemeinschaft (DFG grant STR 1014), the research initiative of the Government of Rhineland-Palatinate “Impulsfonds” 2017/2018), and by the intramural BiomaTiCSprogram (“Biomaterials, Tissues and Cells in Science”) of the University Medical Center Mainz, Germany.
