Abstract
Angioimmunoblastic T-cell lymphoma (AITL) is a T-cell lymphoma of follicular helper T-cell origin. Histologically, neoplastic T-cells proliferate to form clusters adjacent to or between arborizing high endothelial venules (HEVs). HEVs in normal lymph nodes express sulfated glycans called peripheral lymph node addressin (PNAd); however, it remains unclear whether PNAd is also expressed on HEVs in AITL. Furthermore, although it is widely accepted that HEVs are conspicuous in AITL due to their proliferation, quantitative histological support for this concept is lacking. To investigate these issues, we employed monoclonal antibodies recognizing PNAd, namely, MECA-79, HECA-452, and 297-11A, and performed quantitative immunohistochemical analysis of HEVs in 36 AITL-affected and 67 normal lymph nodes. Staining with all three antibodies confirmed that AITL HEVs express PNAd. Moreover, AITL HEVs were bound calcium-dependently by L-selectin-IgM fusion proteins, indicating that they function in the recruitment of L-selectin-expressing lymphocytes. Unexpectedly, HEV distribution density was not increased but rather decreased in AITL compared with normal lymph nodes, but HEV cross-sectional area in AITL was significantly greater than that seen in normal lymph nodes. Overall, these results indicate that the prominence of AITL HEVs is likely due to increased cross-sectional area rather than increased distribution density.
Keywords
Introduction
Angioimmunoblastic T-cell lymphoma (AITL) is one of the most common subtypes of peripheral T-cell lymphoma (PTCL), accounting for roughly 20% of PTCL. 1 Currently, neoplastic T-cells are thought to originate from follicular helper T-cells, which, under normal conditions, play pivotal roles in the formation and maintenance of germinal centers and B-cell differentiation. 2 In AITL, neoplastic T-cells proliferate against a background consisting of reactive host immune cells, including B-cells (often with a blast form), plasma cells, eosinophils, and macrophages, as well as an irregularly expanded meshwork of follicular dendritic cells (FDCs) and arborizing high endothelial venules (HEVs).3,4
HEVs are specialized venules composed of tall and plump endothelial cells located mainly in T-cell zones, such as paracortical and interfollicular areas; however, some HEVs are also present in B-cell zones, particularly in the periphery of lymphoid follicles. 5 Under physiological conditions, HEVs function to recruit lymphocytes from the circulation to the lymphoid parenchyma. HEVs express a series of glycoproteins, including CD34 decorated with sulfated sialyl Lewis x (sLex) glycans, collectively called peripheral lymph node addressin (PNAd).6–8 Expression of PNAd in HEV-like vessels is reportedly induced in various chronic inflammatory diseases, 9 and we also reported such PNAd expression in chronic Helicobacter pylori gastritis, 10 ulcerative colitis, 11 autoimmune pancreatitis, 12 chronic prostatitis associated with benign prostatic hyperplasia, 13 eosinophilic chronic rhinosinusitis (ECRS), 14 and, most recently, oral lichen planus. 15 The relatively weak, transient adhesive interaction between PNAd on HEVs (as well as HEV-like vessels) and L-selectin on circulating lymphocytes results in lymphocyte rolling along the luminal face of HEVs, followed by chemokine-dependent lymphocyte activation, integrin-mediated lymphocyte firm attachment to endothelium, and platelet endothelial cell adhesion molecule 1 (PECAM-1)-mediated lymphocyte transmigration across blood vessels.8,16
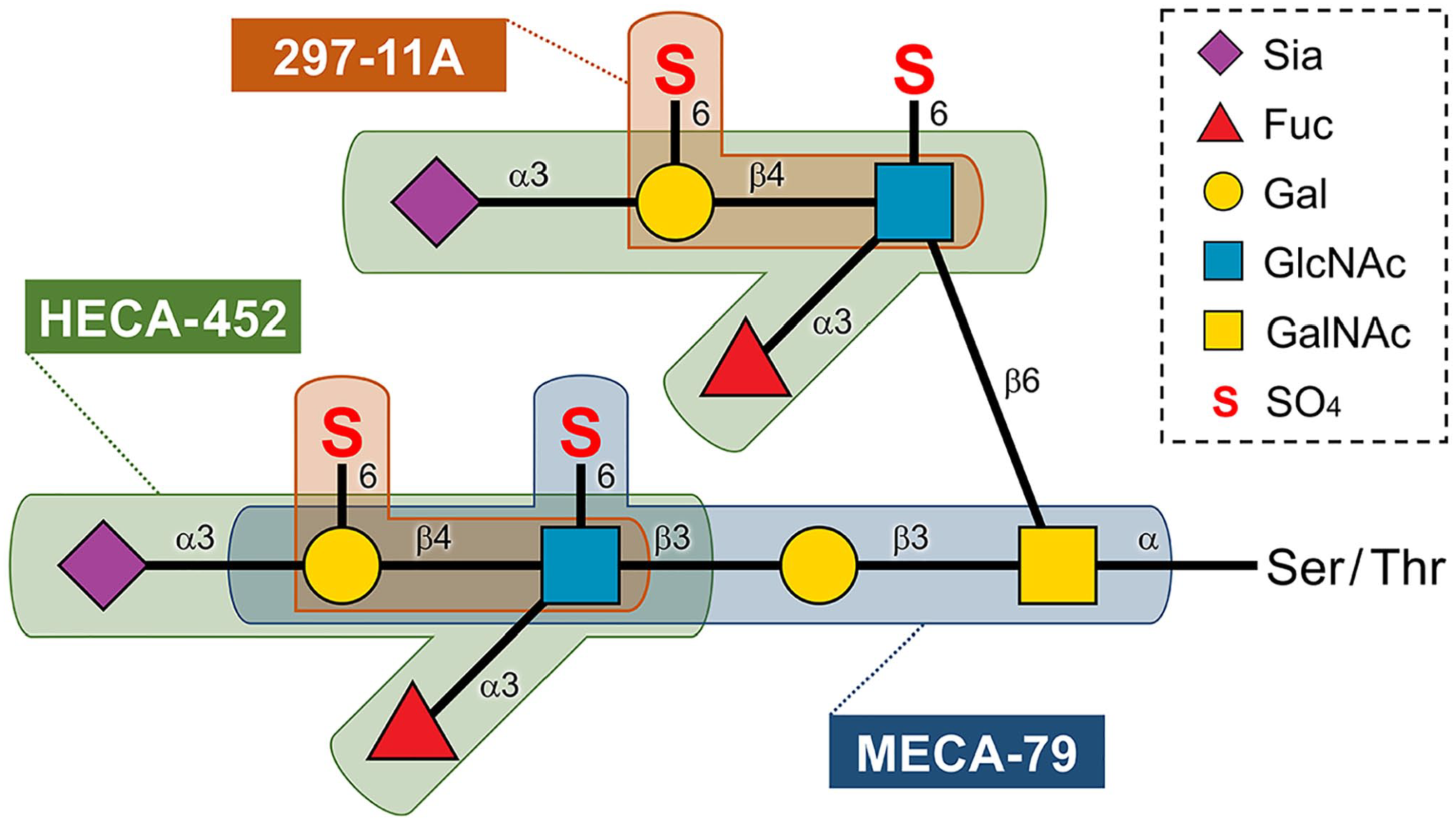
PNAd is often detected using the monoclonal antibody MECA-79, 17 whose epitope has been shown to be 6-sulfo [i.e. N-acetylglucosamine (GlcNAc)-6-O-sulfated] N-acetyllactosamine (LacNAc) attached to extended core 1 O-glycans, Galβ1→4(SO4→6)GlcNAcβ1→3Galβ1→3GalNAcα1→Ser/Thr (in which Gal is galactose and GalNAc is N-acetylgalactosamine) (Fig. 1). 18 This epitope overlaps with 6-sulfo sLex, Siaα2→3Galβ1→4[Fucα1→3(SO4→6)]GlcNAcβ1→R (in which Sia is sialic acid and Fuc is fucose), the L-selectin recognition determinant. This determinant can be also detected by the monoclonal antibody HECA-452, whose epitope has been shown to be sLex tetrasaccharide, Siaα2→3Galβ1→4(Fucα1→3)GlcNAcβ1→R, regardless of GlcNAc- and/or Gal-6-O-sulfation (Fig. 1).19–21 GlcNAc-6-O-sulfation of sLex moieties is required for optimal L-selectin binding and is catalyzed cooperatively by GlcNAc-6-O-sulfotransferase 1 (GlcNAc6ST-1) and GlcNAc-6-O-sulfotransferase 2 (GlcNAc6ST-2).22,23 On the contrary, HEVs in peripheral lymph nodes also express sialyl 6′-sulfo (i.e., Gal-6-O-sulfated) LacNAc-capped O-glycans, Siaα2→3(SO4→6)Galβ1→4GlcNAcβ1→R, which can be detected with the antibody 297-11A (Fig. 1), which we recently developed. 24 Gal-6-O-sulfation of sLex moieties also comprises an L-selectin-binding determinant in vitro 25 and is catalyzed by keratan sulfate Gal-6-O-sulfotransferase (KSGal6ST). 26 However, it remains to be determined whether these sulfated sLex-related glycans are also expressed in HEVs formed in AITL. Furthermore, although it is widely accepted that HEV proliferation is a histological hallmark of AITL, to our knowledge, there are no reports of quantitative histological analysis of HEVs to confirm this idea.
This study consists of two parts: functional analysis of the carbohydrate moiety of PNAd expressed in HEVs, and quantitative and morphometric analysis of HEVs. First, we conducted immunohistochemical staining with a battery of anti-PNAd monoclonal antibodies and confirmed that sulfated sLex, which is expressed in normal lymph node HEVs, is also expressed in HEVs formed in AITL. Then, we used selectin-IgM fusion proteins to determine whether sulfated sLex expressed in these HEVs potentially functions as an L-selectin ligand. In the second part of our analysis, we asked whether HEV distribution density (specifically, the number per unit area) in AITL differed from that seen in normal lymph nodes and how HEV cross-sectional area in AITL compared with that seen in normal lymph nodes. Our analysis indicated overall that HEVs formed in AITL show increased cross-sectional area, rather than increased distribution density.
Materials and Methods
Human Tissue Samples
Formalin-fixed, paraffin-embedded (FFPE) tissue blocks of biopsied lymph nodes with a diagnosis of AITL were obtained from the pathology archives of University of Fukui Hospital (Eiheiji, Japan), National Hospital Organization Mito Medical Center (Ibaraki, Japan), Omachi Municipal General Hospital (Omachi, Japan), and the Japanese Red Cross Fukui Hospital (Fukui, Japan). Using hematoxylin and eosin (H&E) staining with adjunctive immunohistochemical staining for CD3 (rabbit polyclonal, cat. no. A0452; Dako, Glostrup, Denmark), CD4 (mouse monoclonal IgG1, clone 1F6, cat. no. NCL-CD4-1F6; Leica Biosystems, Newcastle Upon Tyne, UK), CD10 (mouse monoclonal IgG1, clone 56C6, cat. no. 413261; Nichirei Biosciences, Tokyo, Japan), CD20 (mouse monoclonal IgG2a, clone L26, cat. no. M0755; Dako), CD21 (mouse monoclonal IgG1, clone 1F8, cat. no. M0784; Dako), BCL6 (mouse monoclonal IgG1, clone P1F6, cat. no. 413761; Nichirei Biosciences), CXCL13 (goat polyclonal, cat. no. AF801; R&D Systems, Minneapolis, MN), and CXCR5 (rabbit polyclonal, cat. no. GTX100351; GeneTex, Irvine, CA), histological diagnosis of AITL was re-evaluated by certified pathologists (AK, MF, HO and MK) according to the latest edition of the World Health Organization (WHO) classification (revised 4th edition). 4 From that analysis, we selected specimens with histological findings in best agreement with AITL (n=36 from 19 patients; mean age, 73.7 ± 12.2 years; male:female = 12:7). FFPE tissue blocks of surgically resected axillary lymph nodes negative for metastasis (n=67 from 19 patients; mean age, 59.2 ± 12.8 years; male:female = 1:18) were collected as controls. Analysis of human lymph node tissues was approved by the Ethics Committee of the Faculty of Medical Sciences, University of Fukui (reference number 20160100, approved on December 7, 2016; reference number 20200193, approved on April 14, 2021).
Monoclonal Antibodies
The following monoclonal antibodies were used as primary antibodies: QBEND10 (mouse IgG1, cat. no. IM0786; Immunotech, Marseille, France) recognizing human CD34, a marker of vascular endothelial cells 27 ; MECA-79 (rat IgM, cat. no. 553863; BD Biosciences, Franklin Lakes, NJ) 17 ; HECA-452 (rat IgM, cat. no. 550407; BD Biosciences) 19 ; and 297-11A (mouse IgM). 24 Epitopes for MECA-79, HECA-452, and 297-11A are shown in Fig. 1.
Expression of MECA-79 and 297-11A Epitopes in Lec2 Cells
To express the minimum epitope of MECA-79, Lec2 cells (obtained from American Type Culture Collection, Manassas, VA), a mutant CHO line that lacks sialic acid modification due to inactivation of the Golgi CMP-sialic acid transporter, 28 were co-transfected with pcDNA1-GlcNAc6ST-229,30 and pcDNA3-β1,3-N-acetylglucosaminyltransferase 3 (β3GlcNAcT-3) 18 (the latter harboring a neomycin resistance gene) at a 9:1 ratio using Lipofectamine Plus (Thermo Fisher Scientific; Waltham, MA) according to the manufacturer’s instructions. We then selected stable transformants in 400 µg/ml of Geneticin (Sigma-Aldrich; St. Louis, MO). Cells positive for MECA-79 were cloned by limited dilution, resulting in Lec2/β3GlcNAcT-3/GlcNAc6ST-2 cells. Cells were then transiently transfected with either pcDNA3-KSGal6ST 26 or empty pcDNA3 (mock). After 36 hr, cells were dissociated into monodispersed cells using phosphate-buffered saline (PBS) containing 0.5 mM ethylenediaminetetraacetic acid (EDTA) and then doubly immunostained with MECA-79 and 297-11A. Expression of both glycoepitopes was assayed simultaneously by flow cytometry using an FACSCanto II (BD Biosciences) with FlowJo software (Tree Star; Ashland, OR).
Immunohistochemistry
Immunohistochemical staining for MECA-79 and HECA-452 was conducted using an indirect method as described, 14 and staining for CD34 and 297-11A was carried out using the Histofine system (Nichirei Biosciences), as per the manufacturer’s instructions. For 297-11A staining, to remove sialic acid, which inhibits 297-11A binding, sections were preincubated at 37C for 90 min in a humidified chamber with 0.2 U/ml neuraminidase from Arthrobacter ureafaciens (Nacalai Tesque; Kyoto, Japan) dissolved in 50 mM sodium acetate (pH 5.2). 31 Double immunofluorescence staining for MECA-79 and 297-11A was performed essentially as described. 32
L- and E-Selectin-IgM Chimera In Situ Binding Assays
To obtain L- or E-selectin-IgM chimeric proteins, COS-1 cells were transiently transfected with pcDNA1.1-L-selectin•IgM or pcDNA1.1-E-selectin•IgM, respectively. 10 Transfected cells were cultured 3 days and conditioned media were recovered. L- and E-selectin-IgM binding to CHO cells expressing non-sulfated sLex attached to core 2-branched O-glycans (named CHO/CD34/F7/C2) and to those expressing its GlcNAc-6-O-sulfated form (designated CHO/CD34/F7/C2/LSST) 31 was assayed by flow cytometry, as described.21,33 L- and E-selectin-IgM chimera in situ binding assays on FFPE tissue sections were carried out as described, 10 with modification. In brief, after quenching endogenous peroxidase activity and blocking nonspecific protein binding, sections were incubated with conditioned media containing L- or E-selectin-IgM chimeras for 30 min and washed with Dulbecco’s modified Eagle medium (DMEM; Thermo Fisher Scientific). Sections were then incubated 30 min with horseradish peroxidase (HRP)-conjugated goat anti-human IgM antibody (Millipore; Billerica, MA) and washed with DMEM. The color reaction was developed using Betazoid DAB Chromogen Kit (Biocare Medical; Pacheco, CA). Sections were briefly counterstained with hematoxylin. Negative controls were washed using DMEM supplemented with 5 mM EDTA to chelate calcium ions. In the E-selectin-IgM chimera in situ binding assay, sections were preincubated with CSLEX1 monoclonal antibody (mouse IgM, cat. no. 551344; BD Biosciences), which binds exclusively to non-sulfated sLex,20,21 as a means to detect 6-sulfo sLex only.
Quantification of HEVs
Immunostained slides were scanned with NanoZoomer RS (Hamamatsu Photonics; Hamamatsu, Japan) to obtain whole slide images. The area of each lymph node (mm2) and the number of vessels positive for CD34, MECA-79, HECA-452, or 297-11A in the entire area of each lymph node were determined using NDP.view2 Plus software (Hamamatsu Photonics). We then divided the number of vessels positive for CD34, MECA-79, HECA-452, or 297-11A in each lymph node by the area of corresponding lymph node to calculate the distribution density of vessels of interest. In addition, the number of HEVs positive for MECA-79, HECA-452, or 297-11A was divided by the number of CD34-positive vessels to determine their proportion in total vessels. To determine the mean cross-sectional area of one HEV, as well as the total cross-sectional area of HEVs present in a unit area (1 mm2), regions in H&E-stained sections with densely distributed HEVs were selected from five cases each of AITL-affected or normal lymph nodes, and for each HEV present in a region, we determined the inner area surrounded by the basement membrane.
Statistical Analysis
Data are expressed as mean ± SD. Differences between groups were statistically analyzed by two-tailed unpaired t-test using GraphPad Prism 7 software (GraphPad Software; La Jolla, CA). P values less than 0.05 were considered significant.
Results
HEVs Formed in AITL Express Sulfated sLex That Constitutes PNAd
AITL cases examined here showed histopathological features typical of AITL, 4 in which lymphoma cells proliferate throughout the lymph node, and normal lymph node architecture is effaced (Fig. 2A). Against a background of reactive host immunoblasts, plasma cells, and eosinophils, small to medium-sized lymphocytes with clear-to-pale cytoplasm (so-called clear cells) proliferate to form clusters adjacent to or between arborizing HEVs (Fig. 2B). Normal lymph node HEVs reportedly constitutively express a series of glycoproteins, including CD34 decorated with sulfated sLex, which constitutes PNAd.6,7 To determine whether HEVs formed in AITL are decorated with sulfated sLex, we conducted immunohistochemical staining for CD34, MECA-79, HECA-452, and 297-11A. In addition to CD34 (Fig. 2C), HEVs formed in AITL were positive for MECA-79 (Fig. 2D), and a proportion were also positive for HECA-452 (Fig. 2E) or 297-11A (Fig. 2F) (see “The Proportion of HEVs in Total Blood Vessels Does Not Increase in AITL” below for detailed quantitative data). Given the glycoepitopes recognized by these antibodies (see Fig. 1), this result indicates that as in normal lymph node, HEVs formed in AITL express sulfated sLex.
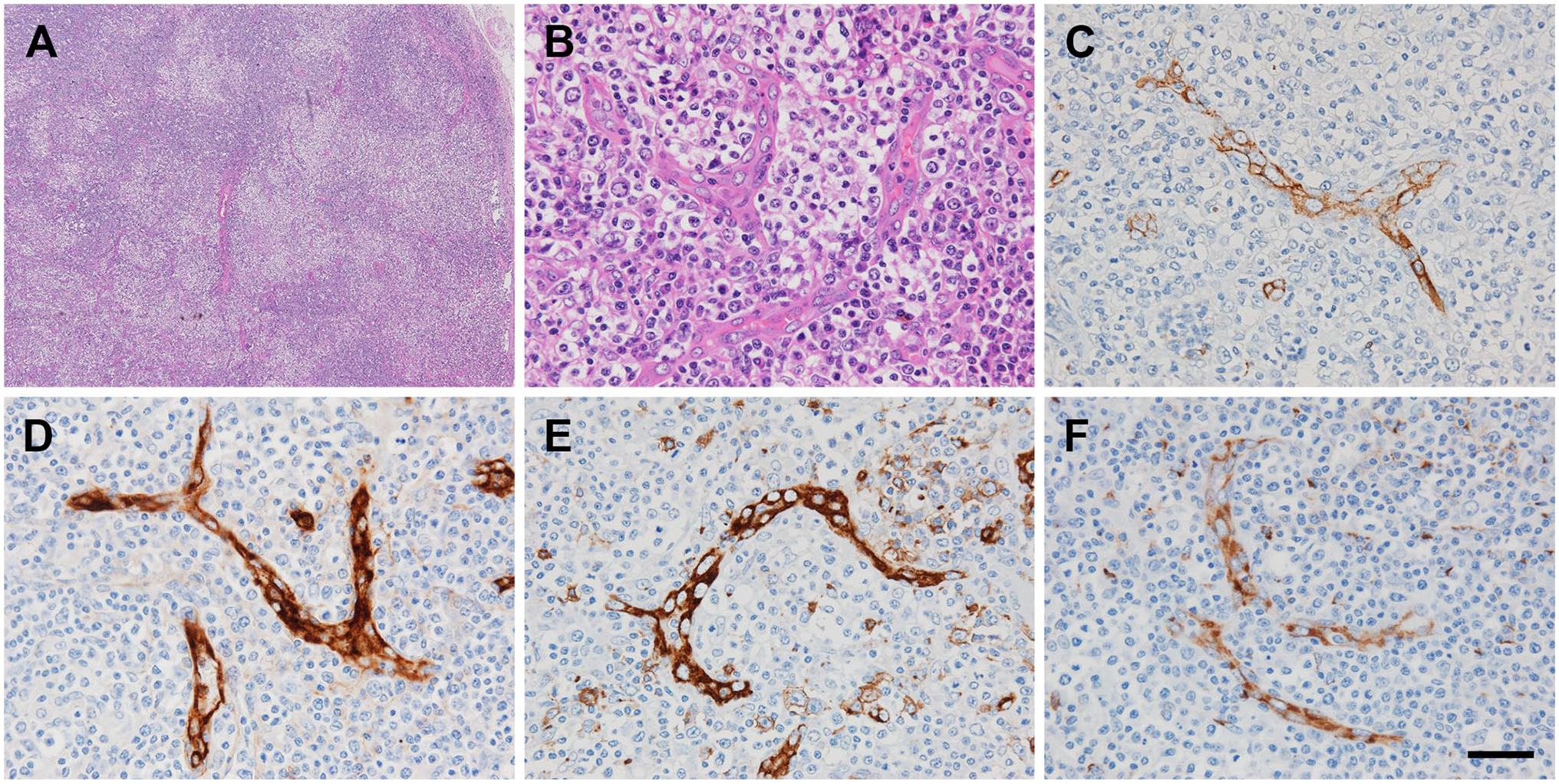
Immunohistochemical profiles of HEVs formed in AITL. (A, B) Histology of AITL. Normal lymph node architecture is effaced due to proliferation of lymphoma cells throughout the lymph node (A). Lymphoma cells with “clear cell” features proliferate to form clusters adjacent to or between arborizing HEVs (B). H&E staining. (C–F) HEVs formed in AITL stain positively for CD34 (C), MECA-79 (D), HECA-452 (E), and 297-11A (F). Signals were visualized with 3,3’-diaminobenzidine (DAB) (brown), and tissues were counterstained with hematoxylin. Bar = 400 µm for panel A and 40 µm for the rest. Abbreviations: HEVs, high endothelial venules; AITL, angioimmunoblastic T-cell lymphoma; H&E, hematoxylin and eosin.
Gal-6-O-sulfation of LacNAc in the MECA-79 Epitope Does Not Inhibit Reactivity to MECA-79
We then conducted double immunofluorescence staining of AITL tissues with MECA-79 and 297-11A anti-bodies and found that, overall, 297-11A and MECA-79 signals on HEVs colocalized (Fig. 3), a pattern described in normal lymph node HEVs. 24 The minimum MECA-79 epitope is reportedly 6-sulfo LacNAc attached to extended core 1 O-glycans, and it is known that α2,3-sialylated and/or α1,3-fucosylated forms of this structure are also recognized by MECA-79 antibody (see Fig. 1). 18 To determine whether 6′-O-sulfation (Gal-6-O-sulfation) of LacNAc in the MECA-79 epitope alters its reactivity to MECA-79, we first established Lec2/β3GlcNAcT-3/GlcNAc6ST-2 cells, which express the minimum MECA-79 epitope (Fig. 4A). We then transfected these cells with either KSGal6ST cDNA to express the 297-11A epitope in addition to the MECA-79 epitope or the empty vector (mock) and then evaluated the expression levels of both epitopes simultaneously by flow cytometry. As shown in Fig. 4B (right upper panel), 29.8% (29.7% + 0.1%) of mock-transfected cells were MECA-79-positive, whereas in cells transfected with KSGal6ST cDNA, 33.3% (28.1% + 5.2%) of cells were MECA-79-positive (Fig. 4B, right lower panel). Importantly, expression levels of the MECA-79 epitope in the 297-11A-positive cell population were not attenuated relative to those seen in the 297-11A-negative cell population (Fig. 4B, right lower panel). This result indicates that Gal-6-O-sulfation of LacNAc—that is, formation of the 297-11A epitope—in the MECA-79 epitope does not inhibit reactivity to MECA-79.
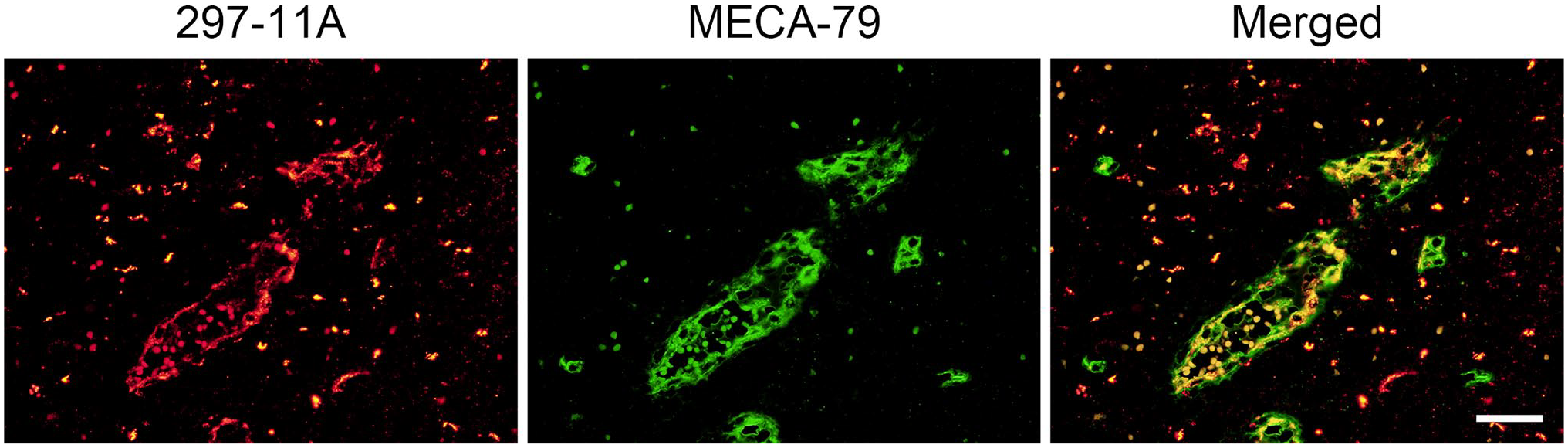
Dual immunofluorescence of HEVs formed in AITL for 297-11A (red) and MECA-79 (green). Yellow signals in Merged indicate colocalization of both carbohydrate antigens. Bar = 40 µm. Abbreviations: HEVs, high endothelial venules; AITL, angioimmunoblastic T-cell lymphoma.
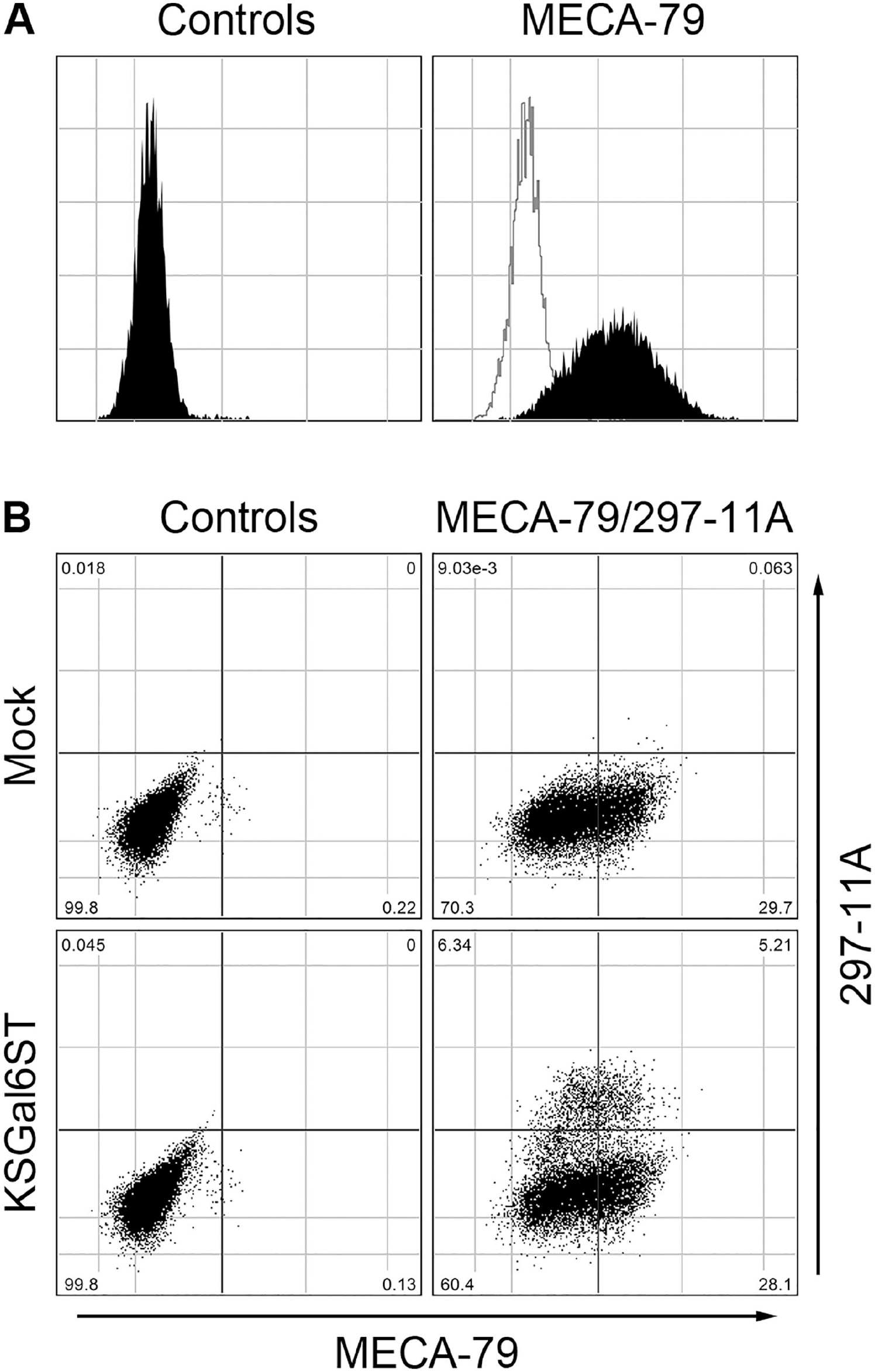
Gal-6-O-sulfation of LacNAc in the MECA-79 epitope does not inhibit reactivity to MECA-79. (A) Stable expression of the MECA-79 minimum epitope in Lec2/β3GlcNAcT3/GlcNAc6ST-2 cells (filled histogram in right panel). Filled histogram in the left panel and open histogram in the right panel represent negative controls in which the primary antibody was replaced with isotype-matched immunoglobulin. X- and Y-axes indicate fluorescence intensity and number of events, respectively. Note that cells expressing the MECA-79 epitope are distributed normally, indicating that they are of a single clone. (B) Lec2/β3GlcNAcT-3/GlcNAc6ST-2 cells were transiently transfected with empty vector (mock; upper panels) or KSGal6ST cDNA (lower panels), and then doubly immunostained with MECA-79 and 297-11A (right panels). Left panels represent negative controls replacing primary antibodies with isotype-matched immunoglobulins. X- and Y-axes indicate fluorescence intensity originating from MECA-79 and 297-11A, respectively. Abbreviations: LacNAc, N-acetyllactosamine; cDNA, complementary DNA.
Sulfated sLex Expressed in AITL HEVs Potentially Functions as an L-Selectin Ligand
Although both L- and E-selectin reportedly bind to 6-sulfo sLex,33,34 binding affinity to 6-sulfo sLex of IgM fusion proteins of these two selectins has not been fully evaluated. Thus, we first assayed their binding affinity to 6-sulfo sLex by flow cytometry using CHO cells expressing 6-sulfo sLex, as well as control CHO cells expressing non-sulfated sLex. As shown in Fig. 5A (blue histograms), L-selectin-IgM chimeras did not bind to CHO cells expressing non-sulfated sLex (upper panel) but did bind to CHO cells expressing 6-sulfo sLex, although the signal intensity was weak (lower panel). On the contrary, E-selectin-IgM chimeras showed comparable binding to CHO cells expressing either non-sulfated sLex or 6-sulfo sLex (Fig. 5A, red histograms). It should be noted that E-selectin-IgM chimeras bound to 6-sulfo sLex more readily than did L-selectin-IgM chimeras (Fig. 5A, lower panel). This finding indicates that, at least in vitro, E-selectin-IgM chimeras possess higher capacity to bind to 6-sulfo sLex than do L-selectin-IgM chimeras and thus could serve as a sensitive functional probe for 6-sulfo sLex.
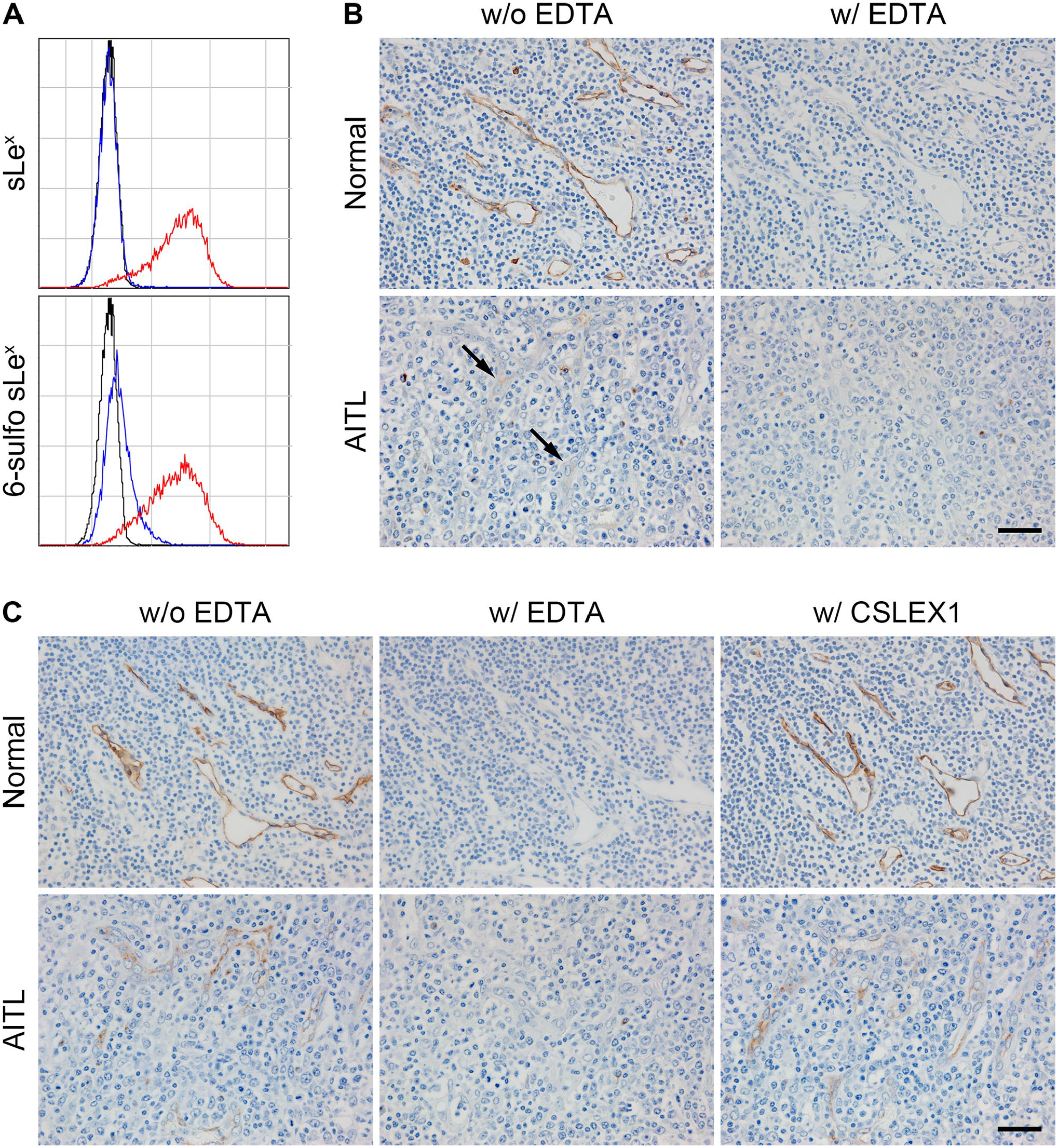
L- and E-selectin-IgM chimera binding assays. (A) Flow cytometric analysis of L-selectin-IgM (blue histograms) and E-selectin-IgM (red histograms) chimera binding to CHO cells expressing non-sulfated sLex (upper panel) or 6-sulfo sLex (lower panel). Black histograms represent negative controls performed using conditioned medium from untransfected COS-1 cells. X- and Y-axes indicate fluorescence intensity and number of events, respectively. (B, C) L-selectin-IgM (B) and E-selectin-IgM (C) in situ binding assays on FFPE tissue sections of normal (upper panels) and AITL-affected (lower panels) lymph nodes. Both L- and E-selectin-IgM chimeras bind to HEVs in the presence of calcium ions (without EDTA), but binding is completely abrogated in the absence of calcium ions (with EDTA). Note very weak L-selectin-IgM binding to HEVs formed in AITL (arrows). E-selectin-IgM chimera binding to HEVs is maintained even after incubating sections with CSLEX1 antibody, which binds exclusively to non-sulfated sLex (with CSLEX1). Signals were visualized with DAB (brown), and tissues were counterstained with hematoxylin. Bar = 40 µm. Abbreviations: FFPE, formalin-fixed, paraffin-embedded; AITL, angioimmunoblastic T-cell lymphoma; HEVs, high endothelial venules; EDTA, ethylenediaminetetraacetic acid.
We then conducted L- and E-selectin-IgM chimera in situ binding assays to investigate whether sulfated sLex expressed in AITL HEVs functions as an L-selectin ligand. Both L-selectin-IgM (Fig. 5B) and E-selectin-IgM (Fig. 5C) bound calcium-dependently not only to HEVs in normal lymph nodes but also to those formed in AITL, although the signal intensity of the L-selectin-IgM chimera in AITL HEVs (Fig. 5B, arrows) was much less than that seen in normal lymph node HEVs. It is noteworthy that E-selectin-IgM chimera binding to these HEVs was preserved even after incubation of tissue sections with anti-non-sulfated sLex antibody CSLEX1 (Fig. 5C, with CSLEX1). These findings collectively indicate that HEVs formed in AITL possess some selectin-binding ability, albeit limited.
HEV Distribution Density in AITL Decreases Rather Than Increases
Given reports that vascularity in AITL often increases as does HEV arborization, 4 we investigated these changes by performing quantitative immunohistochemical analysis of HEVs using 36 AITL-affected lymph nodes and 67 normal axillary lymph nodes as controls. To calculate vessel distribution density, specifically, the number of vessels per unit area, we first determined the area of each lymph node. Not surprisingly, the area of AITL-affected lymph nodes (52.2 ± 59.7 mm2) was greater than that of normal lymph nodes (19.3 ± 19.8 mm2) with high statistical significance (p<0.001) (Fig. 6A). Consequently, the distribution density of total vessels in AITL (62.5 ± 20.6/mm2) was less than that of normal lymph nodes (110.8 ± 35.4/mm2) with high statistical significance (p<0.001) (Fig. 6B). As for HEVs, the distribution density of MECA-79-positive HEVs in AITL (31.0 ± 16.1/mm2) was less than that of normal lymph nodes (47.8 ± 14.9/mm2) with high statistical significance (p<0.001) (Fig. 6C). Similarly, the distribution density of both HECA-452-positive and 297-11A-positive HEVs in AITL (14.2 ± 8.9/mm2 and 7.8 ± 12.4/mm2, respectively) was less than that seen in normal lymph nodes (30.2 ± 13.1/mm2 and 17.3 ± 19.9/mm2, respectively) with statistical significance (p<0.001 and p=0.011, respectively) (Fig. 6D and E, respectively). These findings indicate that rather than increasing, vascularity in AITL decreases relative to normal lymph nodes, as does HEV distribution density.
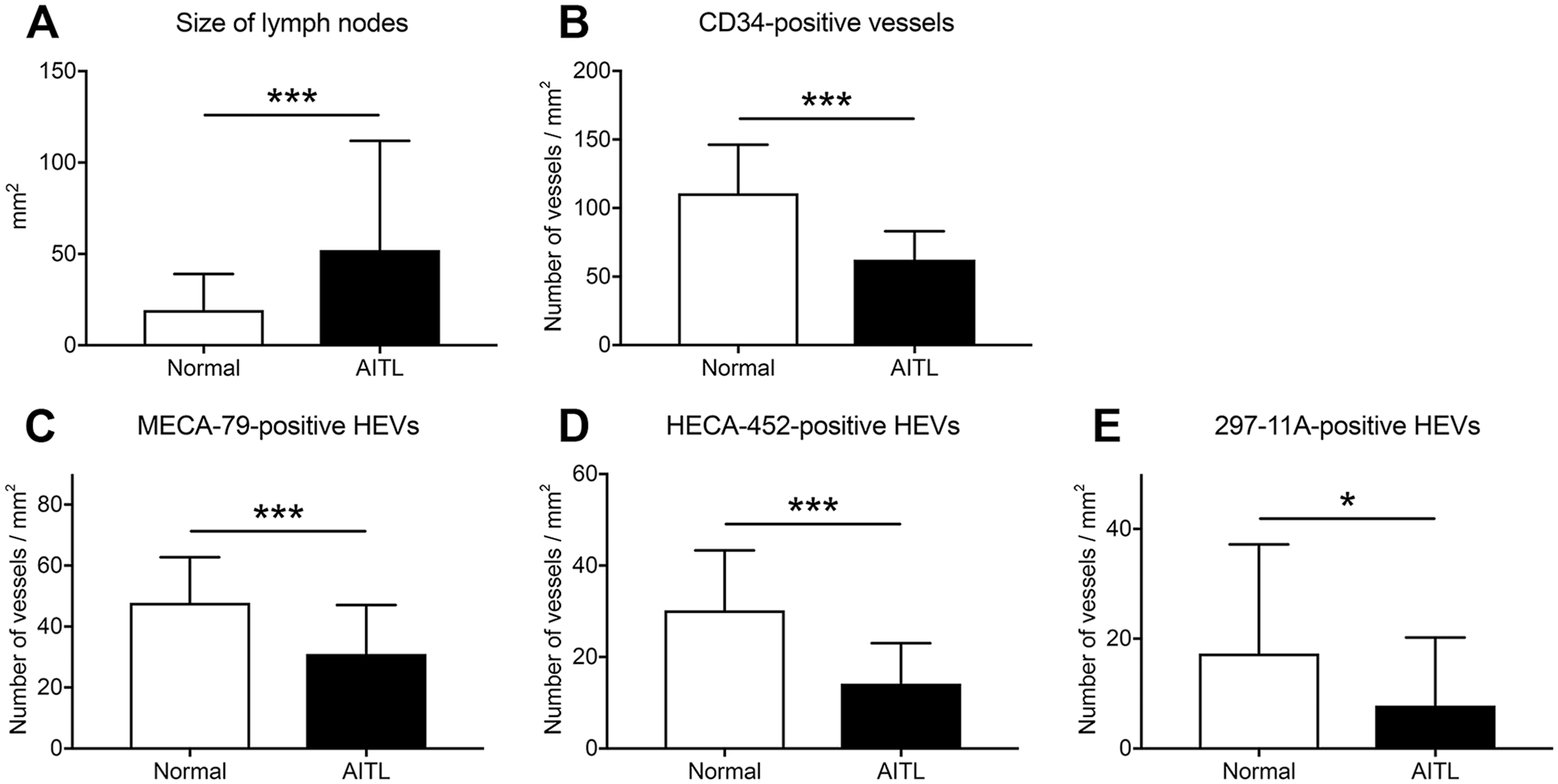
Distribution density of HEVs. (A) The size (area) of normal (n=67; open boxes) and AITL-affected (n=36; filled boxes) lymph nodes. (B–E) Distribution density of vessels immunoreactive for CD34 (B), MECA-79 (C), HECA-452 (D), or 297-11A (E) in normal and AITL-affected lymph nodes. Data are presented as means with SD. Abbreviations: HEVs, high endothelial venules; AITL, angioimmunoblastic T-cell lymphoma. *p < 0.05; ***p < 0.001.
The Proportion of HEVs in Total Blood Vessels Does Not Increase in AITL
We next assessed the proportion of HEVs in total vessels. As shown in Fig. 7A, the proportion of MECA-79-positive HEVs among CD34-positive total vessels in AITL (50.2% ± 22.2%) and in normal lymph nodes (46.2% ± 17.1%) was comparable (p=0.320). Similarly, the proportion of 297-11A-positive HEVs among total vessels in AITL (11.9% ± 17.3%) and in normal lymph nodes (15.6% ± 14.7%) did not differ significantly (p=0.249) (Fig. 7C). These findings indicate that among total vessels, the proportion of HEVs is comparable in AITL and normal lymph nodes. Interestingly, however, the proportion of HECA-452-positive HEVs among total vessels in AITL (22.1% ± 10.9%) was smaller than that seen in normal lymph nodes (28.7% ± 12.2%) with statistical significance (p=0.008) (Fig. 7B).
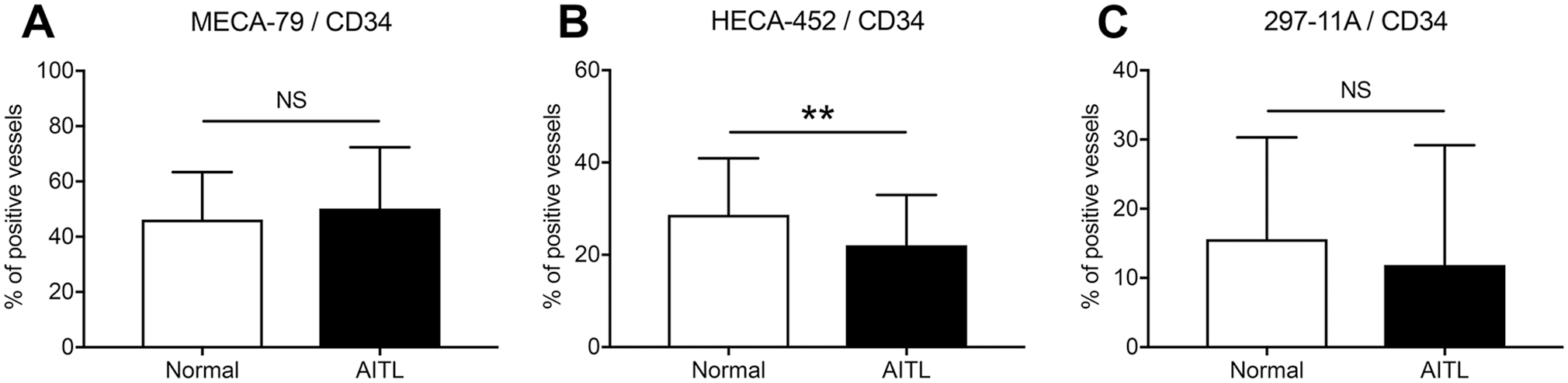
The proportion of HEVs immunoreactive for MECA-79 (A), HECA-452 (B), or 297-11A (C) among CD34-positive total vessels in normal (n=67; open boxes) and AITL-affected (n=36; filled boxes) lymph nodes. Data are presented as means with SD. Abbreviations: HEVs, high endothelial venules; AITL, angioimmunoblastic T-cell lymphoma; NS, not significant. **p<0.01.
HEV Cross-sectional Area Increases in AITL
We next performed a careful review of H&E-stained tissue specimens and found that HEVs formed in AITL (Fig. 8B) were composed of higher endothelial cells with more plump cytoplasm and a larger cross-sectional area than those seen in normal lymph nodes (Fig. 8A). Also, it is noteworthy that in some AITL HEVs, neoplastic “clear cell” T-cells infiltrate the space between endothelial cells and basement membrane and appear to push the basement membrane outward, resulting in increased cross-sectional area of HEVs (Fig. 8C). Thus, we first measured the cross-sectional area of every single HEV in AITL-affected and normal lymph nodes. As shown in Fig. 8D, the mean cross-sectional area of an HEV in AITL (0.0014 ± 0.0012 mm2) was greater than that seen in normal lymph nodes (0.0008 ± 0.0010 mm2) with high statistical significance (p<0.001). We then calculated the total cross-sectional area of HEVs present in a unit area (1 mm2) of lymph nodes. As shown in Fig. 8E, that area in AITL (0.062 ± 0.015 mm2) was greater than that seen in normal lymph nodes (0.035 ± 0.015 mm2) with statistical significance (p=0.019), whereas the number of HEVs present in the same region did not differ between normal and AITL-affected lymph nodes (41.6 ± 8.1/mm2 and 44.8 ± 8.4/mm2, respectively; p=0.558) (Fig. 8F).
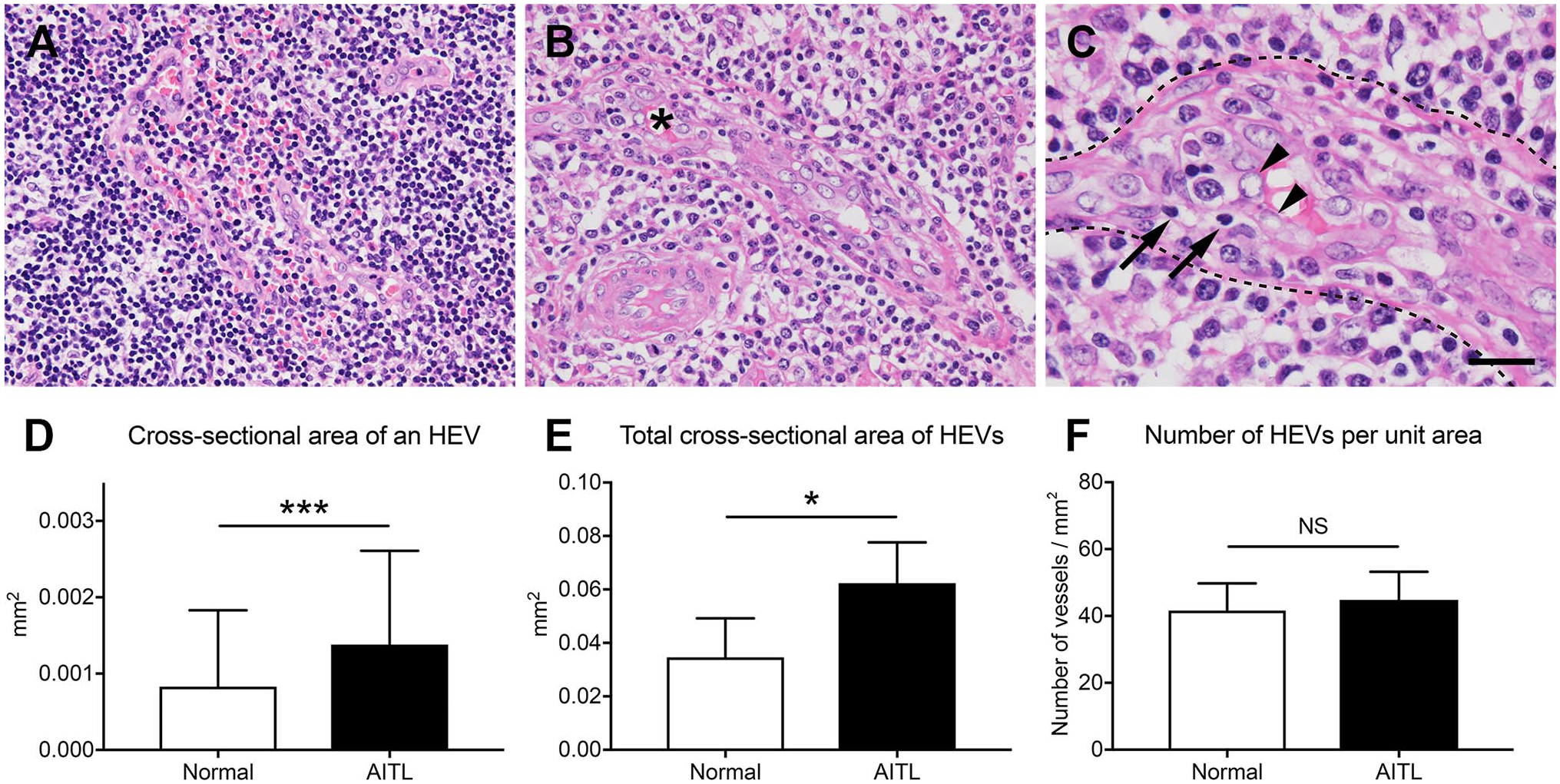
Increased cross-sectional area of HEVs in AITL-affected lymph nodes. (A–C) Photomicrographs of HEVs in normal (A) and AITL-affected (B and C) lymph nodes. Panel C is an enlarged view of the region indicated by asterisk in panel B. Note that in AITL, neoplastic T-cells (arrows) and reactive immunoblasts infiltrate the space between endothelial cells (arrowheads) and the basement membrane (dotted lines) likely pushing the basement membrane outward and resulting in increased cross-sectional area of HEVs. H&E staining. Bar = 40 µm for panels A and B and 20 µm for panel C. (D) The mean cross-sectional area of one HEV in normal (n=5; open box) and AITL-affected (n=5; filled box) lymph nodes. (E) The total cross-sectional area of HEVs present in a unit area (1 mm2) of normal (n=5) and AITL-affected (n=5) lymph nodes. (F) The number of HEVs per unit area in the region assessed in (E) in normal (n=5) and AITL-affected (n=5) lymph nodes. Data are presented as means with SD. Abbreviations: HEVs, high endothelial venules; AITL, angioimmunoblastic T-cell lymphoma; NS, not significant. *p<0.05, ***p<0.001.
Discussion
In this study, we demonstrate that HEVs formed in AITL express sulfated sLex that constitutes PNAd and potentially functions as an L-selectin ligand. Moreover, we demonstrate that the distribution density of HEVs does not increase but rather decrease in AITL compared with normal lymph nodes, whereas the proportion of HEVs in total blood vessels is essentially comparable between AITL and normal lymph nodes. On the contrary, we found that the cross-sectional area of HEVs in AITL was significantly greater than that seen in normal lymph nodes. These results overall indicate that the apparent increase in PNAd-expressing functional HEVs in AITL is likely due to increased cross-sectional area, not to increased distribution density.
HEV proliferation is a hallmark of AITL, as is described in the latest edition of the WHO classification. 4 Strikingly, however, here we found that the distribution density of not only total vessels but also HEVs in AITL was significantly less than that seen in normal lymph nodes. These findings suggest that AITL lymph nodes become enlarged due to proliferation of neoplastic T-cells and recruitment and/or proliferation of reactive host immune cells, possibly in response to humoral factors secreted by neoplastic T-cells, 3 whereas stromal components including HEVs do not increase, or even if they do, that increase is not to the same extent as the tumor parenchyma. Nevertheless, some pathologists report that HEVs apparently increase in number in AITL, 1 a perception that we attribute to increased HEV cross-sectional area. This phenomenon could be due to increased expression of lymphotoxin αβ (LT-αβ) secreted by neoplastic T-cells or reactive immunoblastic B-cells. 35 As LT-β receptor signaling is important for the formation and maintenance of HEV morphology, including endothelial cell cuboidal shape and vessel arborization, 36 increased signaling in this pathway may contribute to the unusual morphology of HEVs seen in AITL. Alternatively, in AITL, neoplastic “clear cell” T-cells often proliferate to form clusters surrounding HEVs, and in some HEVs infiltrate the space between endothelial cells and basement membrane to expand the area surrounded by the basement membrane.
Overall, we feel our findings could be helpful in diagnosing AITL. Occasionally, tissue specimens that have a strong probability of being AITL lack prominent HEVs. In such cases, it is important to understand that it is not the number of HEVs that increases in AITL but rather their cross-sectional area. A consideration of this finding might prevent pathologists from erroneously ruling out a diagnosis of AITL in these circumstances.
Muramatsu 37 has reported that the number and proportion of HEVs, which were identified morphologically based on H&E and silver staining, were significantly greater in AITL than in normal lymph nodes. This result is inconsistent with our results reported here, potentially due to different methods used to identify HEVs. In the Muramatsu study, vessels with a diameter of less than 15 µm, even if they were composed of high endothelial cells, were classified as capillaries rather than HEVs. Thus, a substantial portion of small-sized PNAd-expressing vessels that we categorize as HEVs here might not have been classified as HEVs in the other study. As these vessels are frequently found in normal lymph nodes, it is plausible that the number of HEVs in normal lymph nodes was underestimated, resulting in an apparent relative increase in the number of HEVs in AITL.
We also demonstrate that HEVs formed in AITL express sulfated sLex recognized by MECA-79, and some are also positive for HECA-452 and 297-11A. This is an immunohistochemical profile similar to that seen in normal peripheral lymph node HEVs. 24 We previously demonstrated that sulfated sLex is also expressed in HEVs formed in lymphoid stroma of salivary Warthin’s tumor, 38 testicular seminoma, 39 and a diffuse sclerosing variant of papillary thyroid carcinoma. 40 These HEVs presumably function in the histogenesis of distinct lymphoid stroma formed in these tumors. However, HEV function in AITL histogenesis remains uncharacterized. As AITL is composed not only of neoplastic T-cells but also of a variety of reactive host immune cells, it is tempting to speculate that HEVs formed in AITL may function to recruit these reactive host immune cells, particularly those expressing high levels of L-selectin such as naïve T- and B-cells. 5 Further studies are, however, required to test this hypothesis.
We also show for the first time that a Gal-6-O-sulfated form of the MECA-79 epitope is recognized by this antibody. Double immunofluorescence staining revealed that 297-11A signals on HEVs formed in AITL colocalized with MECA-79 signals, as is seen in HEVs in normal peripheral lymph nodes. 24 Furthermore, flow cytometric analysis revealed that Gal-6-O-sulfation of LacNAc, that is, formation of the 297-11A epitope, in the MECA-79 epitope does not attenuate reactivity to MECA-79. In addition, we previously demonstrated by glycan array analysis that GlcNAc-6-O-sulfation of Gal-6-O-sulfated LacNAc does not alter reactivity to 297-11A. 24 These findings overall indicate that KSGal6ST catalyzes Gal-6-O-sulfation of 6-sulfo LacNAc attached to extended core 1 O-glycans to form 6,6′-disulfo (both GlcNAc- and Gal-6-O-sulfated) LacNAc attached to extended core 1 O-glycans, which can be detected by both MECA-79 and 297-11A.
In in situ binding assay performed here, normal lymph node HEVs were readily bound by L-selectin-IgM chimeras. By contrast, HEVs formed in AITL were decorated very weakly with this chimera, indicating substantially diminished L-selectin-binding capacity of HEVs formed in AITL. On the contrary, E-selectin-IgM chimeras bound readily to HEVs in normal lymph nodes and relatively well to those formed in AITL. As E-selectin-IgM chimera binding was not abolished by pretreatment of tissue sections with CSLEX1 antibody, it is reasonable to conclude that E-selectin-IgM chimera binding to HEVs is mediated by 6-sulfo sLex. Thus, we propose that HEVs formed in AITL likely possess potential L-selectin-binding properties, albeit to a limited extent.
In this study, although the proportion of MECA-79-positive and 297-11A-positive HEVs in total vessels did not differ significantly between AITL-affected and normal lymph nodes, the proportion of HECA-452-positive HEVs in total vessels was significantly smaller in AITL compared with normal lymph nodes. Given the glycoepitopes recognized by MECA-79, 297-11A, and HECA-452 antibodies (see Fig. 1), this result suggests that GlcNAc-α1,3-fucosylation in sLex moieties is downregulated in AITL compared with normal lymph nodes. Hiraoka et al. 41 previously demonstrated that KSGal6ST competes with α1,3-fucosyltransferase 7 (FucT-7) for the same acceptor substrate, namely, LacNAc, and downregulates synthesis of 6-sulfo sLex by inhibiting α1,3-fucosylation. As GlcNAc-α1,3-fucosylation of 6-sulfo sLex moieties is a prerequisite for selectin binding, 42 L-selectin-binding capacity of HEVs may be decreased in AITL. This hypothesis is supported by the finding that both L- and E-selectin-IgM chimera binding to HEVs decreased in AITL, although the significance of this finding remains to be determined.
Footnotes
Acknowledgements
The authors thank Hisataka Kato, Takuya Suga, Shinichi Fujii, and Moriyasu Hattori for preparation of tissue sections; Maiko Yamanaka for immunohistochemical staining; Junko Yamamoto for assisting with flow cytometry; Dr. Akifumi Muramoto for helpful discussion on statistical analysis; and Dr. Elise Lamar for critical reading of the manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: AK designed and performed the research, analyzed the data, and wrote the manuscript; MF, HH, TK, and TO performed the research; MM wrote the manuscript; TOA and JM performed the research; HO analyzed the data and wrote the manuscript; and MK conceived of and designed the research, analyzed the data, and wrote the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a Grant-in-Aid for Scientific Research (B) 21H02702 from the Japan Society for the Promotion of Science (to M.K.).
