Abstract
Radiation therapy–mediated salivary gland destruction is characterized by increased inflammatory cell infiltration and fibrosis, both of which ultimately lead to salivary gland hypofunction. However, current treatments (e.g., artificial saliva and sialagogues) only promote temporary relief of symptoms. As such, developing alternative measures against radiation damage is critical for restoring salivary gland structure and function. One promising option for managing radiation therapy–mediated damage in salivary glands is by activation of specialized proresolving lipid mediator receptors due to their demonstrated role in resolution of inflammation and fibrosis in many tissues. Nonetheless, little is known about the presence and function of these receptors in healthy and/or irradiated salivary glands. Therefore, the goal of this study was to detect whether these specialized proresolving lipid mediator receptors are expressed in healthy salivary glands and, if so, if they are maintained after radiation therapy–mediated damage. Our results indicate that specialized proresolving lipid mediator receptors are heterogeneously expressed in inflammatory as well as in acinar and ductal cells within human submandibular glands and that their expression persists after radiation therapy. These findings suggest that epithelial cells as well as resident immune cells represent potential targets for modulation of resolution of inflammation and fibrosis in irradiated salivary glands.
Introduction
According to the American Cancer Society, more than 80,000 people will develop head and neck cancer each year and those patients commonly receive radiation therapy (RT) as a part of their treatment course. 1 RT regularly destroys the salivary glands and leads to a loss of secretory function commonly referred as hyposalivation.2,3 This condition is usually accompanied by an altered sense of taste as well as difficulty in speech and poor swallowing ability4,5 among other problems (e.g., bacterial and fungal infections).6,7 Nonetheless, mechanisms underlying the loss of salivary gland function after head and neck cancer RT are not clearly understood. 8 Previous studies indicate that RT-induced hyposalivation is the result of diverse events, such as DNA damage, apoptosis, senescence, tight junction disruption, epithelial to mesenchymal transition, exarcerbated inflammatory infiltrate, fibrosis, and alteration of parasympathetic innervation.8–11 In addition, current treatments remain largely ineffective, with therapeutic interventions being limited to the use of saliva substitutes with modest effectiveness12–14 and medications that provide only temporary relief.12,15–17 In light of the high degree of need and the limitations of current therapies, the development of alternative treatments to restore salivary gland functioning is essential.
One promising option for treating RT-damaged salivary gland is through activation of specialized proresolving lipid mediator receptors (SPMRs) by their ligands (e.g., resolvins, lipoxins, and maresins). These receptors belong to the G protein–coupled receptor family, and diverse members have been identified to date, such as lipoxin A4 receptor/formyl peptide receptor 2 (ALX/FPR2), G protein–coupled receptor 32 (GPR32; also known as DRV1), chemerin chemokine-like receptor 1 (CMKLR1, also known as ERV1 or ChemR23), and leukotriene B4 receptor 1 (BLT1).18–21 As reported first by Serhan, 22 SPMRs are key players in resolution of inflammation as they modulate immune response while increasing tissue repair. Given that RT induces inflammation followed by fibroblast recruitment and exacerbated extracellular matrix deposition (i.e., fibrosis),23,24 targeting SPMR activation may provide an alternative option to mitigate this process in irradiated glands. For this reason, the objective of this article is to detect whether these SPMRs are expressed in healthy salivary glands and if their organization is maintained after RT damage with the ultimate goal of using them as potential therapeutic targets.
Materials and Methods
Materials
Hematoxylin and eosin Y solution were purchased from MilliporeSigma (Burlington, MA). Rabbit anti-FPR2 was obtained from Alomone Labs (Jerusalem, Israel). Rabbit anti-GPR32, rabbit anti-BLT1, rabbit anti-CMKLR1, Alexa fluor 488 conjugated anti-rabbit IgG, phosphate buffered saline (PBS), 4′,6-diamidino-2-phenylindole (DAPI), triton X-100, sodium citrate, xylene and ethanol were purchased from Thermo Fisher Scientific (Waltham, MA).
Human Subjects
Specimens were obtained from the University of Utah, Department of Otolaryngology, Head and Neck Surgery. Analyses of these specimens were conducted under the guidelines and with the approval of the University of Utah Health Sciences Institutional Review Board, with informed consent obtained for each patient and data on age, sex, and radiation doses collected from their medical records.
Tissue Section Processing
Human submandibular glands (hSMGs) were fixed in 4% paraformaldehyde for 24 h and later transferred to 70% ethanol. They were then dehydrated through a series of graded ethanol baths, embedded in paraffin and cut into 3 µm sections.
Hematoxylin and Eosin Staining
Tissue sections were washed three times at room temperature for 5 min in xylene, once in xylene:ethanol (1:1), twice for 5 min in 100% ethanol, followed by single washes with 95%, 80%, 70%, and 50% ethanol for 5 min each, and rinsed twice with distilled water. Later, rehydrated sections were stained with Harris hematoxylin for 6 min and washed with distilled water for 2 min, washed once with 0.5% lithium carbonate (Li2CO3; w/v) for 1 min, and rinsed once with distilled water. Then, sections were washed with 95% ethanol for 1 min followed by incubation with eosin for 1 min and washed with 95% ethanol for 1 min. Subsequently, samples were rinsed thrice with 100% ethanol cleared in xylene and mounted with a xylene-based mounting medium. Finally, samples were examined using a Leica DMI6000B inverted microscope (Leica Microsystems; Wetzlar, Germany).
Immunofluorescence Staining
Deparaffinized tissue sections were incubated with sodium citrate buffer (10 mM sodium citrate, 0.05% Tween 20, pH 6.0) at 95C for 30 min for antigen retrieval. Then, samples were rinsed twice with distilled water and permeabilized with 0.1% Triton X-100 in PBS at room temperature for 45 min. Sections were blocked with 5% goat serum in PBS at room temperature for 1 hr and incubated with primary antibodies at 4C overnight, as detailed in Table 1 (below). Next, specimens were washed three times with PBS and incubated with secondary antibodies as detailed in Table 2 (below) at room temperature for 1 hr. Likewise, sections were again washed three times with PBS and counterstained with DAPI at room temperature for 15 min (1:1000 dilution). Later, Z-stack sections (10 sections per sample) were captured and analyzed using a confocal DMi8 Stellaris Leica microscope with 20× and 63× objectives. Finally, negative control (Appendix Fig. 1) and internal positive controls (e.g., inflammatory cells; see below) validated our findings, thereby ruling out non-specific staining and false positive results.
Primary Antibodies Used in This Study.
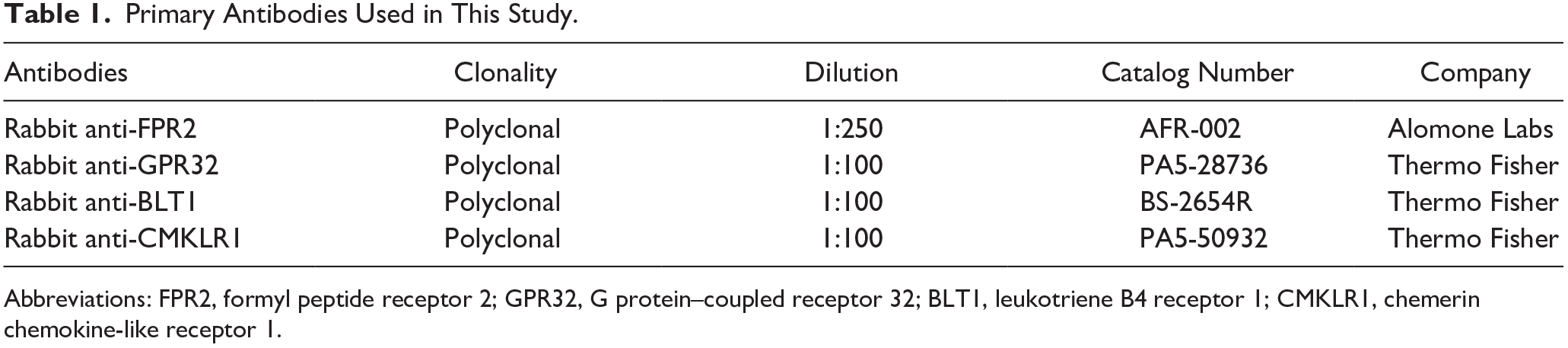
Abbreviations: FPR2, formyl peptide receptor 2; GPR32, G protein–coupled receptor 32; BLT1, leukotriene B4 receptor 1; CMKLR1, chemerin chemokine-like receptor 1.
Secondary Antibodies Used in This Study.

Clinical Data.

Results
Radiation Therapy Disrupts Tissue Morphology
Before detecting SPMRs, we first corroborated whether RT caused alteration of architecture and morphology in the specimens studied using hematoxylin and eosin staining (as described in Materials and Methods section). As shown in Table 3, each group involved two females and one male with an average age of 68 years, both with and without RT. Non-irradiated glands displayed lobules divided by thin connective tissue septa (Fig. 1A) as well as mixed serous and mucous acinar units accompanied by branching intercalated and striated ducts (Fig. 1B). In contrast, irradiated glands showed disruption of lobular architecture due to loss of parenchymal tissue (Fig. 1C) featured by destruction of both acinar and ductal structures (Fig. 1D), nuclear atypia (Fig. 1D), extensive fibrosis (Fib. 1D), as well as inflammatory infiltrates (Fig. 1D). Together, these results indicate that RT caused alteration of architecture and morphology in the specimens studied.
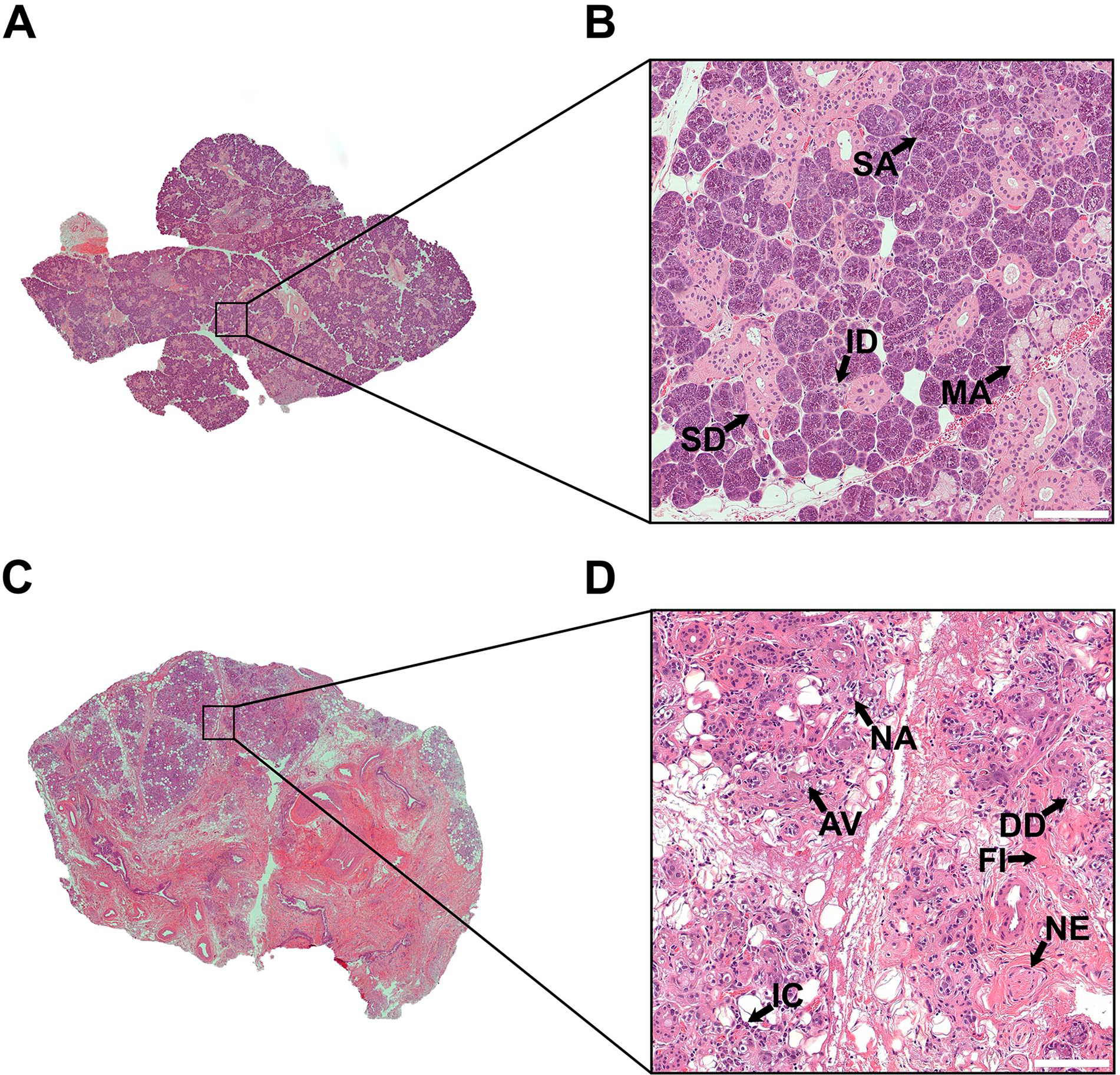
Histological features of non-irradiated and irradiated human submandibular glands. (A) Non-irradiated human submandibular glands (hSMG) displayed lobular architecture. (B) Non-irradiated hSMG showed mixed serous and mucous units accompanied by ductal structures and thin connective strands. (C) Irradiated hSMG displayed disruption of lobular architecture. (D) Irradiated hSMG showed acinar vacuolization, ductal degeneration, nuclear atypia and alterations in the stroma. A representative image from n = 3 per experimental group is shown. Scale bars = 100 µm. Abbreviations: ID, intercalated duct; SA, serous acini; MA, mucous acini; SD, striated duct; AV, acinar vacuolization; DD, degenerated duct; NA, nuclear atypia; NE, nerve; IC, inflammatory cells; FI, fibrosis.
ALX/FPR2 Is Expressed in Mucous Acini and Intercalated Ducts
We observed that non-irradiated hSMG expressed ALX/FPR2 in cytoplasmic droplets within mucous acini (Fig. 2, white arrow) and intercalated ducts (Fig. 2, brown arrows). Moreover, a similar pattern was observed in irradiated hSMG, with the exception that a weaker staining was observed in the mucous cells given that this cell type is diminished after RT (Fig. 2, brown arrows). Note that ALX/FPR2 was absent in both serous acini and striated ductal cells but was detected within the cytoplasm of resident mononuclear cells (Fig. 2, red arrow), endothelial cells (Fig. 2, purple arrow) and adipocytes (Fig. 2, yellow arrows). Taken together, these results indicate that ALX/FPR2 is expressed in mucous acini and intercalated ducts from hSMG.
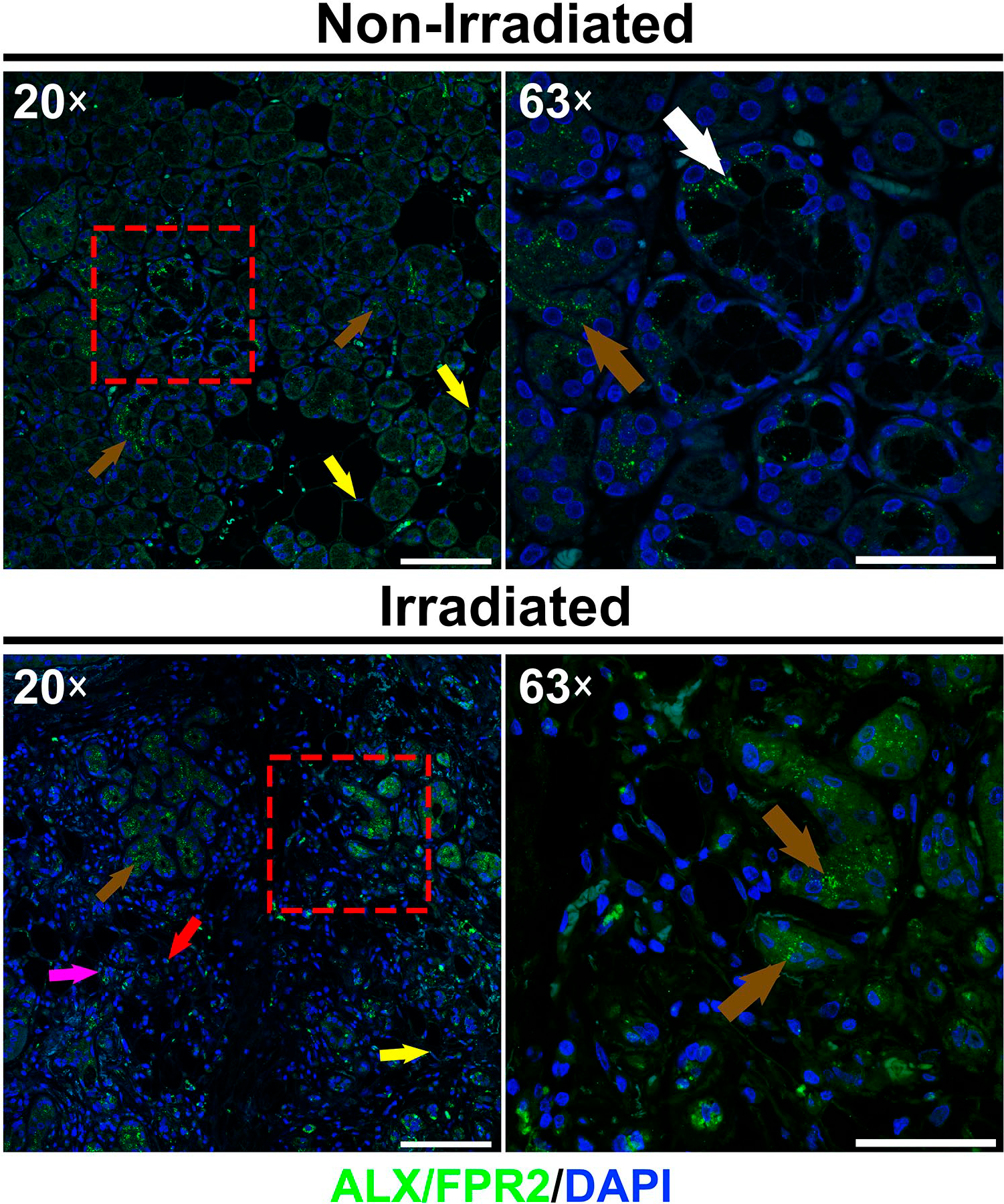
Distribution of ALX/FPR2 in non-irradiated and irradiated human submandibular glands. Image shows the presence of ALX/FPR2 as cytoplasmic droplets in mucous acini, intercalated ducts, resident mononuclear cells, endothelial cells and adipocytes. White arrows indicate mucous cells, brown arrows indicate ductal cells, red arrow indicates a resident mononuclear cell, purple arrow indicates endothelial cells and blue arrows indicate adipocytes. Note that 20× and 63× images of each group were obtained to show parenchymal/stromal interface and detailed epithelial structures, respectively. A representative image from n = 3 per experimental group is shown. Scale bars in 20× and 63× magnifications represent 100 and 50 µm, respectively.
GPR32 Is Expressed in Mucous Acini, Serous Acini, and Striated Ducts
Our results show that both non-irradiated and irradiated hSMG express GPR32 at the perinuclear region of serous and serous demilune cells (crescent- shaped aggregation of serous cells at the periphery of mucous cells) (Fig. 3, white arrows). Additionally, this receptor was detected at the apical membrane of striated ducts (Fig. 3, brown arrows) but absent in mucous cells and intercalated ducts. Finally, GPR32 was identified across the cytoplasm of resident mononuclear cells (Fig. 3, red arrow), endothelial cells (Fig. 3, purple arrow) and adipocytes (Fig. 3, blue arrow). Together, these results indicate that GPR32 is present in mucous and serous acini as well as in striated ducts of hSMG.
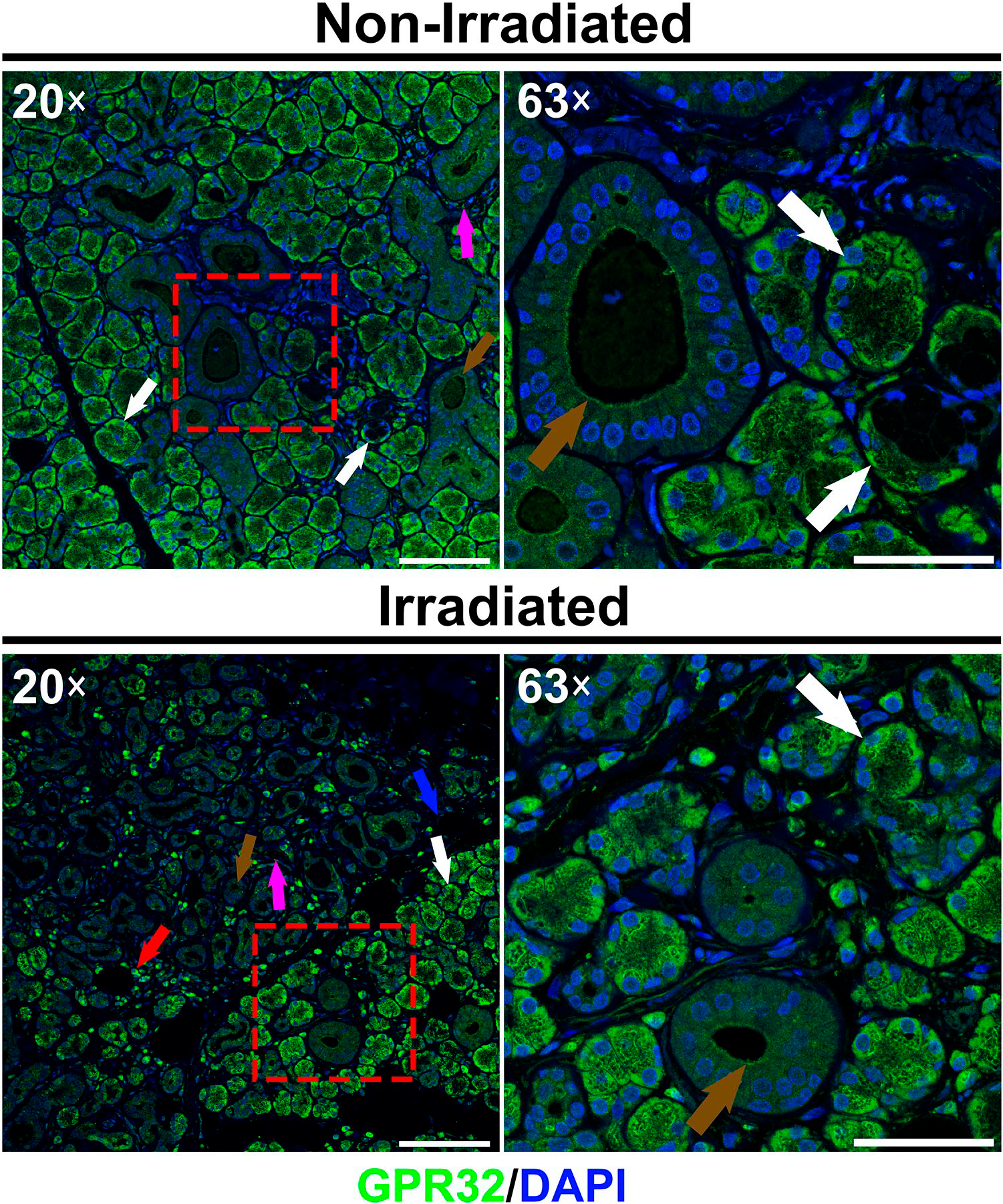
Distribution of GPR32 in non-irradiated and irradiated human submandibular glands. Image shows the presence of GPR32 in serous acini and serous demilunes as well as in striated ducts, resident mononuclear cells, endothelial cells and adipocytes. White arrows indicate perinuclear expression in serous acinar cells, brown arrows indicate apical location in striated ducts, red arrow indicates resident mononuclear cells, purple arrow indicates endothelial cells and blue arrow indicates adipocytes. Note that 20× and 63× images of each group were obtained to show parenchymal/stromal interface and detailed epithelial structures, respectively. A representative image from n = 3 per experimental group is shown. Scale bars in 20× and 63× magnifications represent 100 and 50 µm, respectively.
CMKLR1 Is Expressed in All Cell Types
We observed that non-irradiated and irradiated hSMG express CMKLR1 throughout the plasma membrane (i.e., basolateral and apical surfaces) of serous and mucous acini (Fig. 4, white arrows) as well as of intercalated and striated ducts (Fig. 4, brown arrows). Additionally, CMKLR1 was detected across the cytoplasm of mononuclear cells within the glandular tissue (Fig. 4, red arrow) and endothelial cells (Fig. 4, purple arrows). Results thus indicate CMKLR1 is present in all reviewed cell types within hSMG.
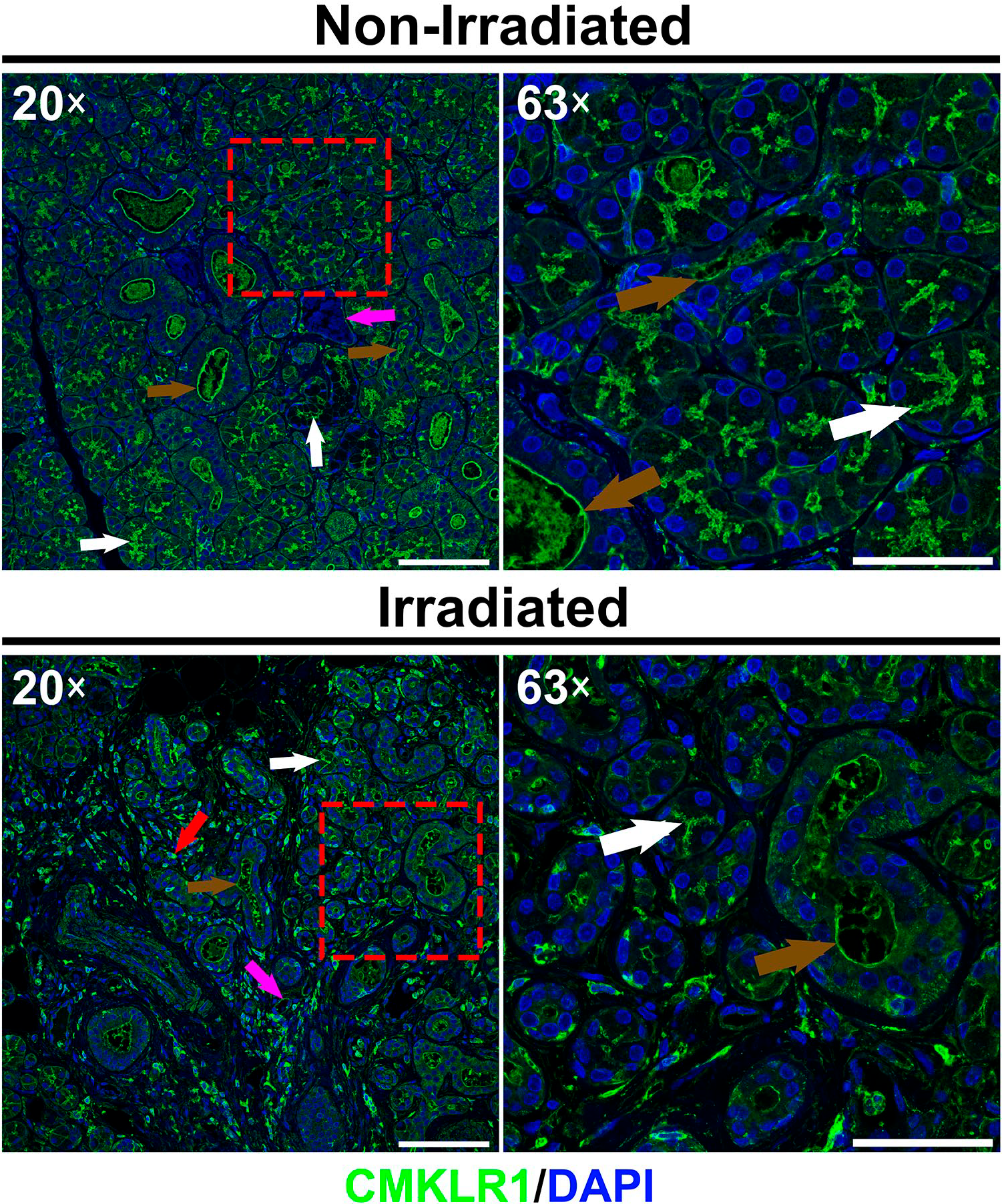
Distribution of CMKLR1 in non-irradiated and irradiated human submandibular glands. Image shows the presence of CMKLR1 in serous and mucous acini as well as in ducts, resident mononuclear cells and endothelial cells. White arrows indicate plasma membrane expression in acini, brown arrows indicate plasma membrane expression in ducts, red arrow indicates resident mononuclear cell and purple arrows indicate endothelial cells. Note that 20× and 63× images of each group were obtained to show parenchymal/stromal interface and detailed epithelial structures, respectively. A representative image from n = 3 per experimental group is shown. Scale bars in 20× and 63× magnifications represent 100 and 50 µm, respectively.
BLT1 Is Expressed in All Cell Types
We observed that non-irradiated and irradiated hSMGs express BLT1 in cytoplasmic droplets within serous acini (Fig. 5, white arrows). Likewise, BLT1 was detected at the basolateral membrane of mucous acini (Fig. 5, green arrows) and at the plasma membrane (i.e., basolateral and apical surfaces) of intercalated and striated ducts (Fig. 5, purple arrows). Finally, BLT1 was identified across the cytoplasm of resident mononuclear cells (Fig. 5, red arrow). As such, these results indicate that BLT1 is expressed in all cell types reviewed within the glandular tissue.
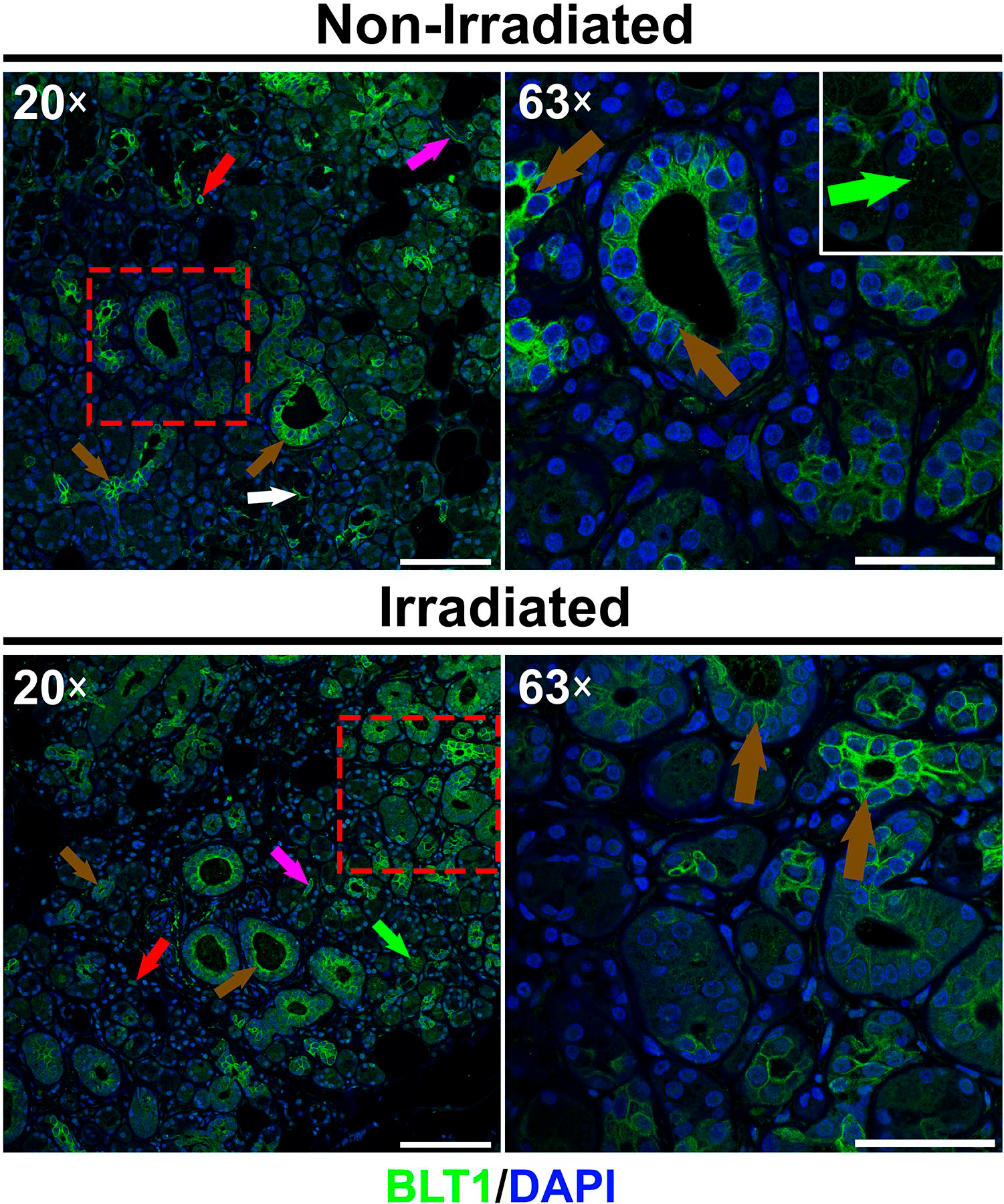
Distribution of BLT1 in non-irradiated and irradiated human submandibular glands. Image shows the presence of BLT1 in acinar and ductal structures as well as in mononuclear cells and endothelial cells. Green arrows indicate cytoplasmic droplets expression in serous acini (inset), white arrow indicates basolateral expression in mucous acini, brown arrows indicate plasma membrane expression in intercalated and striated ducts, red arrow indicates a resident mononuclear cell and purple arrows indicate endothelial cells. Note that 20× and 63× images of each group were obtained to show parenchymal/stromal interface and detailed epithelial structures, respectively. A representative image from n = 3 per experimental group is shown. Scale bars in 20× and 63× magnifications represent 100 and 50 µm, respectively.
Discussion
This study provides a description of the major SPMRs in hSMGs. Using a confocal analysis approach, we demonstrated that these receptors are expressed in both non-irradiated (i.e., healthy) and irradiated glands. Our previous studies demonstrated that ALX/FPR2 is expressed in salivary glands25 and its activation with resolvin D1 (RvD1) stimulates intracellular calcium increases and phosphatidylinositol 3‑kinase (PI3K-Akt) signaling, both of which lead to tight junction integrity, thus improving salivary cell viability and tissue functionality.25,26 Moreover, activation of ALX/FPR2 decreased Sjögren’s syndrome-like features in a mouse model,27,28 indicating control of inflammation in salivary glands. These striking proresolving effects in salivary epithelium led us to investigate whether other SPMRs are expressed in salivary glands and to extend these findings to irradiated salivary glands, where proresolution receptors have never been studied. Particularly, while head and neck RT and Sjögren’s syndrome have vastly different causes (i.e., ionizing radiation 8 vs. an as-yet poorly understood complex of genetic predisposition and environmental triggers), 29 they nonetheless share a comparable clinical presentation (i.e., they both involve an exaggerated and sustained inflammatory response that results in glandular destruction and fibrosis),8,29 thus supporting their comparison for symptom alleviation.
Inflammation due to RT is characterized by an increased production of histamine and prostaglandins that cause neutrophil chemotaxis 30 and lead to monocyte recruitment as well as an increased amount of resident macrophages. 31 These events further augment inflammatory cytokine secretion, thereby promoting local tissue destruction. 31 Specifically, transforming growth factor beta 1 (TGFβ1) mediates the migration of fibroblasts into damaged areas followed by their transdifferentiation into myofibroblasts.32,33 These cells secrete exaggerated amounts of extracellular matrix components, such as collagen and fibronectin,23,24 resulting in replacement of functional units (i.e., acini and ducts).34,35 In addition, TGFβ1 induces fibrosis in salivary epithelium by increasing the rate of conversion of epithelial cells to fibroblasts, a process known as epithelial–mesenchymal transition.36,37 This fibrotic phenomenon is irreversible, and its progression is observed as salivary gland hypofunction.8,38 As such, identifying SPMRs in non-irradiated and irradiated salivary glands is the first step to unveil a new treatment to attenuate RT-mediated hyposalivation, which is currently limited to palliative care. 39 For these purposes, activation of SPMRs offers a potential treatment for controlling inflammation and subsequent fibrosis in salivary glands, as detailed below.
Most of the proresolution studies have focused on ALX/FPR2, which is detected in humans and rodents, where it is widely expressed in leukocytes (e.g., neutrophils and macrophages), 40 epithelial 41 and mesenchymal cells. 42 This protein was first categorized as an FPR receptor in light of its activation by N-formyl methionyl peptide (fMLP). 43 Later, it was proved to have a higher binding affinity for lipoxin A4 (LXA4) than fMLP, which rendered the acronym ALX. 44 Furthermore, ALX/FPR2 exerts either proresolution or pro-inflammatory signals depending on the ligand to which it is exposed. Specifically, previous studies showed that proresolution activation of ALX/FPR2 by LXA4 leads to reduction of tumor necrosis factor α (TNFα)-mediated leukocyte trafficking in a dorsal air pouch mouse model 45 and increased neutrophil efferocytosis by bone marrow–derived macrophages isolated from mice. 44 In contrast, pro-inflammatory activation of this receptor by serum amyloid A leads to increased chemotaxis of neutrophils harvested from the peritoneal cavity of mice. 46 In addition, ALX/FPR2 has been shown to reduce fibrosis in various organs. For instance, activation of this receptor with LXA4 suppressed immune cell recruitment and downregulated TGFβ1, thereby leading to reduced fibrosis and improved function in an irradiated lung mouse model. 47 Moreover, activation of ALX/FPR2 with a synthetic agonist reduced both collagen density and hydroxyproline levels in a scleroderma mouse model, thus indicating reduced fibrosis. 48 Given that ALX/FPR2 is expressed in healthy mucous acini and intercalated duct cells within hSMGs and is likewise maintained after RT, agonists for this receptor could be used to both resolve inflammation and reduce fibrosis in irradiated glands.
Another pertinent receptor is GPR32, which is primarily expressed on neutrophils, 49 monocytes, 20 adipose tissue, 50 and vascular endothelial cells. 51 GPR32 is involved in resolution of inflammation, with previous studies demonstrating that its activation leads to reduced neutrophil recruitment 49 as well as enhanced phagocytosis and clearance of cellular debris in human phagocytes. 20 Regarding the role of GPR32 in fibrosis prevention, previous studies demonstrated that activation of this receptor with RvD1 blocked TGFβ1-induced epithelial–mesenchymal transition in a human lung cell line, thereby reducing expression of profibroblast mesenchymal stem cell markers and restoring expression of epithelial cell markers. 52 Likewise, RvD1 activation of GPR32 reduced expression of N-cadherin and vimentin while also increasing expression of ZO-1 and E-cadherin in a fibrosis-like epithelial–mesenchymal transition nasopharyngeal carcinoma cell line. 53 Given that GPR32 survives RT (as evidenced by its continued presence in irradiated glands), we believe this receptor may be a viable therapeutic target for resolving inflammation and diminishing fibrosis following RT.
CMKLR1, which is primarily expressed in leukocytes 54 as well as in epithelial 55 and mesenchymal cells, 56 is another potentially promising SPMR, given its role in resolving inflammation. Specifically, previous studies demonstrated its proresolving properties in a dorsal air pouch mouse model, where activation of CMKLR1 with resolvin E1 (RvE1) resulted in attenuated interleukin 12 production and decreased TNFα-mediated nuclear factor kappa B signaling. 18 Likewise, this treatment enhanced phagocytosis in a mouse model of diabetes. 57 Particularly, previous studies indicated that activation of CMKLR1 enhances TGFβ1 expression and promotes fibroblast activation with enhanced collagen deposition. 58 Furthermore, this receptor was gradually upregulated and caused excessive production of TGFβ1 in a diabetic rat, which facilitated endothelial to mesenchymal transition and finally resulted in kidney fibrosis. 59 Nonetheless, CMKLR1 is a profibrotic mediator in various organs,58,59 suggesting that it may induce fibrosis and thus render it unsuitable for use in irradiated glands, an issue that warrants further investigation before attempts are made to take advantage of the above noted anti-inflammatory properties.
Regarding BLT1, previous studies have shown that it is widely expressed in neutrophils, macrophages, and eosinophils 60 as well as in non-inflammatory cells61,62 (e.g., including minor salivary glands) 63 and that activation of this receptor by specific lipid mediators regulates inflammation. Specifically, previous studies showed that stimulation of BLT1 with leukotriene B4 induces leukocyte infiltration in a myocardial infarction mouse model. 64 Moreover, activation with RvE1 leads to neutrophil apoptosis and efferocytosis by macrophages in an acute lung injury model. 65 However, similar to CMKLR1 as mentioned above, this receptor has been noted to cause fibrosis in various organs. For instance, BLT1−/− mice showed fewer fibroblasts and decreased collagen I accumulation as compared with wild-type mice in a unilateral ureteral obstruction kidney fibrosis mouse model. 66 Another study showed that BLT1 blockade reduced inflammatory cells, TGFβ1, and fibrosis in a bleomycin-mediated lung fibrosis mouse model. 67 Again, further investigation of potential fibrotic effects would be needed for this receptor to be applied in the current context.
In summary, our results demonstrate heterogenous distribution of various SPMRs in hSMG. Note that the expression of these SPMRs is preserved after being irradiated, indicating high resistance against RT damage. For this reason, we speculate that both ALX/FPR2 and GPR32 could be potentially used to regulate inflammation and subsequent fibrosis, while CMKLR1 and BLT1 show promise for inflammation management but require further investigation to rule out fibrotic contributions.
Supplemental Material
sj-pdf-1-jhc-10.1369_00221554211031678 – Supplemental material for SPM Receptor Expression and Localization in Irradiated Salivary Glands
Supplemental material, sj-pdf-1-jhc-10.1369_00221554211031678 for SPM Receptor Expression and Localization in Irradiated Salivary Glands by Harim Tavares dos Santos, Kihoon Nam, Jason P. Hunt, Luke O. Buchmann, Marcus M. Monroe and Olga J. Baker in Journal of Histochemistry & Cytochemistry
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this article as follows: planning (HTdS, KN, and OJB), acquisition of samples (JPH, MMM, and LOB), execution of experiments (HTdS), analysis of data (HTdS, KN, and OJB), writing of manuscript (HTdS, KN, and OJB), and all authors have read and approved the manuscript as submitted.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Institutes of Health-National Institute of Dental and Craniofacial Research Grant R01DE027884 (to OJB).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
