Abstract
We hypothesized that tenascin expression is increased in pleural inflammatory and fibrotic diseases and that its expression can be used as a marker of active pleural involvement. For this purpose we analyzed 71 histological samples of inflammatory and fibrotic pleura from patients with asbestos-induced pleural reaction (n = 6), postcardiac injury syndrome (n = 6), parapneumonic infection and/or empyema (n = 23), tuberculosis (n = 5, rheumatoid disease (n = 1), and fibrosis with inflammation of unknown etiology (n = 30). All 71 cases were studied by immunohistochemistry for tenascin. In 19 selected cases tenascin mRNA in situ hybridization was also performed. In every case, tenascin was increased by immunohistochemistry. Most prominent immunoreactivity was detected in areas of newly formed fibrosis. Increased tenascin mRNA expression by in situ hybridization was detected in the individual cells of the newly formed fibrosis underneath the fibrinous exudate. The tenascin mRNA-positive cells localized in areas in which by immunohistochemical studies the cells were positive for α-smooth muscle actin, desmin, and vimentin, suggesting a myofibroblast phenotype. Tenascin mRNA expression was also seen less frequently in areas in which some cells were positive for cytokeratin. These cells might represent mesothelial cells entrapped in the inflammatory lesion. Alternatively, they might represent fibroblast-type cells with aberrant cytokeratin expression. We conclude that in pleural inflammatory and fibrotic diseases tenascin immunoreactivity is increased and tenascin mRNA-positive cells localized mainly in the areas of myofibroblast- and, less often, mesothelial-type cells, suggesting that mainly myofibroblasts and, less commonly, also mesothelial cells might be responsible for tenascin expression in pleural inflammatory and fibrotic diseases.
N
Histological findings according to the type of reaction of each diagnosis group a
aPMNs, polymorphonuclear leukocytes; ∗, granulomatous inflammation; Total no., total number of cases in various groups.
Tenascin was discovered in the 1980s (Chiquet and Fambrough 1984). It is an oligomeric extracellular matrix glycoprotein consisting of isoforms tenascin-C, -X, -R, -Y, and -W (Erickson 1993; Hagios et al. 1996; Weber et al. 1998). Tenascin has been suggested to play a structural role and to modulate adhesive and migratory functions of the cells (Chiquet–Ehrismann 1990). It is expressed in several nonmalignant and malignant diseases, in inflammation, and in wound healing (Erickson 1993; Chiquet–Ehrismann 1995).
Tenascin expression is low in normal human lung (Koukoulis et al. 1991). Increased tenascin expression has been detected during lung morphogenesis, in fetal and newborn rat lung (Young et al. 1994), in benign and malignant lung tumors (Koukoulis et al. 1991; Soini et al. 1993), and in various fibrotic lung disorders (Wallace et al. 1995; Kaarteenaho–Wiik et al. 1996). Because tenascin is induced by transforming growth factor β (TGFβ) and by tumor necrosis factor α (TNFα) in cultured human mesothelial cells (Kinnula et al. 1998), it might also be expressed in inflammatory pleural diseases in vivo. In a recent study, tenascin concentration was shown to be increased in malignant pleural effusions studied by dot immunobinding (dot-blot) assay using a monoclonal tenascin antibody (Lawniczak et al. 1992). However, to date no studies have been done on tenascin expression in inflammatory or fibrotic diseases of the pleura.
(
Increased tenascin immunoreactivity in the granulation tissue-like area of the newly formed fibrosis in a pleural biopsy from a patient with empyema. (
(
(
This study was undertaken to investigate whether tenascin-C (later called tenascin) expression, as demonstrated by immunohistochemistry and in situ hybridization (ISH), is increased in inflammatory and fibrotic pleura, and to describe the immunohistochemical findings of tenascin distribution in these disorders. ISH was used to elucidate which cells are responsible for the expression of tenascin mRNA in pleural inflammatory and fibrotic diseases. One of our hypotheses was that tenascin might be upregulated in inflammatory but not necessarily in chronic fibrotic pleural disorders.
Materials and Methods
Patients and Handling of Specimens
Histopathologically typical cases of pleural inflammatory and fibrotic diseases were retrieved from the files of the Department of Pathology, Oulu University Hospital, by reevaluating pleural material taken as thoracoscopic or open pleural biopsies (n = 25), as decortication and/or pleurectomy samples (n = 40), or as percutaneous biopsies (n = 6) between 1992 and 1998. A total of 71 patients (10 women and 61 men) with asbestos-induced pleural fibrotic reaction (n = 6, Group 1), postcardiac injury syndrome (n = 6, Group 2; five cases that had occurred after coronary bypass operation and one case after myocardial infarct), parapneumonic infection and/or empyema (n = 23, Group 3), tuberculosis (n = 5, Group 4), rheumatoid disease (n = 1, Group 5), and of unknown etiology (n = 30, Group 6) were included in the study. Postcardiac injury occurs after a variety of myocardial or periocardial injuries. Pleural effusion is a common manifestation of postcardiac injury syndrome, and the histological changes are nonspecific. The clinical follow-up information was obtained from the patient records at the University Hospital and at the nearby Central Hospitals. Of the 71 patients, 59 were men (mean age 55.6 years, SD ± 13.1 years) and 12 women (mean age 68.0 years, SD ± 11.4 years). Mean number of pack-years (i.e., the number of years multiplied by the number of packs of cigarettes smoked daily) for men was 29.3 (SD ± 20.2), and for women 9.9 (SD ± 9.9). Information on smoking habits was not available in 13 cases.
The pleural material was fixed in 10% formalin and then dehydrated and embedded in paraffin. Sections 4 μm thick were stained with hematoxylin–eosin and van Gieson. Staining for acid-fast bacilli was carried out in selected cases. Uninvolved pleural tissue, used as a control, was obtained from five patients who underwent surgery for lung carcinoma. All the material was reevaluated and one representative tissue block in each case was selected for tenascin immunohistochemical studies. In addition, one tissue block was selected for tenascin mRNA ISH in 19 cases. To identify the phenotype of the tenascin-expressing cells, the serial sections of those 19 cases were stained with commercially available antibodies against α-smooth muscle actin (Clone 1A4; Sigma Bio-Sciences, St Louis, MO), human cytokeratin (Clone MNF116; Dako, Glostrup, Denmark), desmin (Clone D33; Dako), vimentin (Clone V9; Dako), human muscle actin (Clone HHF35; Dako, Carpinteria, CA), CD34 (Clone QBEnd/10; Novocastra, Newcastle-upon-time, UK), and FVIII-related antigen (Dako; Glostrup, Denmark) at dilutions of 1:1000, 1:100, 1:50, 1:50, 1:100, 1:25, and 1:50, respectively.
Scoring of Tenascin Immunoreactivity and Statistical Methods
The extent of tenascin immunoreactivity was scored as absent (0), low (+), moderate (+ +), and strong (+ + +) in controls and study groups. The significance of the associations was determined using Fisher's exact probability test designed for small sample groups.
Anti-tenascin Antibodies and Immunohistochemical Staining
A monoclonal antibody (MAb) 143DB7 was used that reacts with the two major isoforms of tenascin-C. MAb 143DB7 was developed to detect tenascin in formaldehyde-fixed tissue and has been characterized in detail elsewhere (Soini et al. 1993). It is available from Locus–Genex (Helsinki, Finland).
Sections 4 μm thick were deparaffinized in xylene and rehydrated in graded ethanols. Endogenous peroxidase was abolished by incubating the sections in 0.1% hydrogen peroxide in absolute methanol for 20 min. Before immunostaining, the sections were treated with 0.4% pepsin (Merck; Darmstadt, Germany) at 37C for 30 min. For the immunostaining, the avidin–biotin–peroxidase complex method was used as described (Kaarteenaho–Wiik et al. 1996).
MAb 143DB7 with a dilution of 1:1000 of supernatant was used as the primary antibody. The sections were incubated with the primary antibody at 4C overnight, followed by a biotinylated rabbit anti-mouse secondary antibody (at a dilution of 1:300 for 30 min) and the avidin–biotin–peroxidase complex (both from Dakopatts; Glostrup, Denmark). The color was developed with diaminobenzidine. Sections were counterstained with a light hematoxylin stain and mounted with Eukitt (Kindler; Freiburg, Germany). The negative control consisted of substituting the primary antibody with PBS (at pH 7.2) or normal mouse serum.
(
(
Preparation of Tissue Sections for ISH
Four-μm-thick sections from paraffin-embedded pleural biopsies were collected on clean Superfrost Plus glass slides, paraffin was removed by xylene, and tissues rehydrated through a graded ethanol series. After three immersions in pH 7.2, the sections were treated in 0.2 M HCl for 20 min, twice in PBS for 3 min each, followed by proteinase K (100 μg/ml in PBS) treatment for 15 min at 37C. Then tissue sections were transferred into 0.025 M glycine for 30 sec. Tissues were postfixed in 4% paraformaldehyde (Fluka; Buchs, Switzerland) in PBS, pH 7.2 (all solutions made with 0.1% diethyl pyrocarbonate-treated water). After postfixation, tissue sections were transferred into 0.025 M glycine in PBS for 3 min and then acetylated in 0.25% acetic anhydride in 0.1 M triethanolamine (pH 8.0) for 10 min (Rosenfeld et al. 1991). After that, tissue sections were rinsed in 2 × SSC, dehydrated through a graded series of ethanol, and air-dried.
Preparation of RNA Probes
A cDNA fragment (bases 814–1316) of the full-length human tenascin-C cDNA (Siri et al. 1991) was synthesized by PCR from the HT-11 subclone kindly provided by Dr. Luciano Zardi (Instituto Nazionale Per la Ricerca Sul Cancro, Genova, Italy) by using the following primers: 5′ CCC TGC AGT GAG GAG CAC GGC ACA 3′ and 5′ TGC CCA TTG ACA CAG CGG CCA TGG 3′. The 503-bp PCR product containing a specific sequence for tenascin-C was subcloned into a TA vector (TA cloning Kit; Invitrogen, San Francisco, CA). Sense and antisense RNA probes were generated from a linearized template by using a riboprobe transcription kit (Promega; Madison, WI) and the probes were labeled with [35S]-UTP (Amersham; Little Chalfont, UK). The radioactively labeled RNA probes were purified by centrifugation through Bio-Gel P-30 columns (Bio-Spin 30; Bio-Rad, Richmond, CA). Each in vitro transcription reaction yielded RNA probes of high specific activity (typically 4.5–6 × 108 dpm/1 μg DNA template and 50–70% incorporation).
In Situ Hybridization
The hybridization mixture contained the 35S-labeled RNA probe (1.2 × 105 dpm/μl), 50% deionized formamide (BRL; Rockville, MD), 5 mM dithiothreitol, 500 μg/ml yeast tRNA (BRL), 2 mg/ml bovine serum albumin (BRL), and 4 × SSC. The samples were hybridized overnight at 50C while covered with Parafilm (Pääkkö et al. 1992) and then washed three times in 2 × SSC/50% formamide at 52C and 2 × SSC at room temperature (RT), followed by incubation in an RNase solution (100 μg/ml RNase A; Boehringer Mannheim, Mannheim, Germany) in 2 × SSC at 37C for 30 min. The tissue sections were then washed in 2 × SSC/50% formamide at 52C for 5 min and in three changes of 2 × SSC at RT, dehydrated sequentially in 70%, 80%, and 95% ethanol for 1 min each with agitation, and air-dried.
Autoradiography was performed by dipping the sections in Kodak NTB-2 nuclear track emulsion diluted 1:1 with sterile distilled water at 42C. After a week's exposure in the dark at 4C, the slides were developed in Kodak D-19 developer for 5 min, rinsed in 1% acetic acid in distilled water for 30 sec, fixed in Kodak Agefix for 5 min, rinsed in distilled water, and stained with hematoxylin–eosin.
To evaluate the specificity of the 35S-labeled antisense tenascin probes to the tissue sections of pleural specimens, control experiments were performed using 35S-labeled sense tenascin probes separately for each sample.
The hybridized tissue sections of pleural samples were examined by light microscopy and the number of grains over the cells was evaluated in general and especially at the locations where tenascin immunoreactivity was located. Cells or cell groups hybridized with the 35S-labeled antisense tenascin probe were considered positive if they contained more grains than corresponding cells and tissue areas that had been hybridized with the 35S-labeled sense tenascin probe.
Results
Histological Findings
Granulation-like tissue corresponding to newly formed fibrosis (i.e., loose extracellular matrix with leukocytes, proliferation of fibroblasts, and formation of capillaries), was seen in 34 of the 71 cases. Dense fibrosis corresponding to old fibrosis (i.e., sparsely cellular dense tissue with a few leukocytes) was seen in 31 cases. Granulomatous inflammation could be detected in five cases with tuberculosis and one case with rheumatoid disease. Fibrinous exudate overlying the detached mesothelium was observed in 59 and polymorphonuclear leukocytes (PMNs) in 31 of the 71 cases; mononuclear leukocytes were detectable in every case. The histological findings in each disease group are shown in Table 1. Aggregation of PMNs was found significantly more often in pleural samples from patients with infective etiology (i.e., parapneumonia, empyema, and tuberculosis) than in other patients (p < 0.00001 by Fisher's exact probability test).
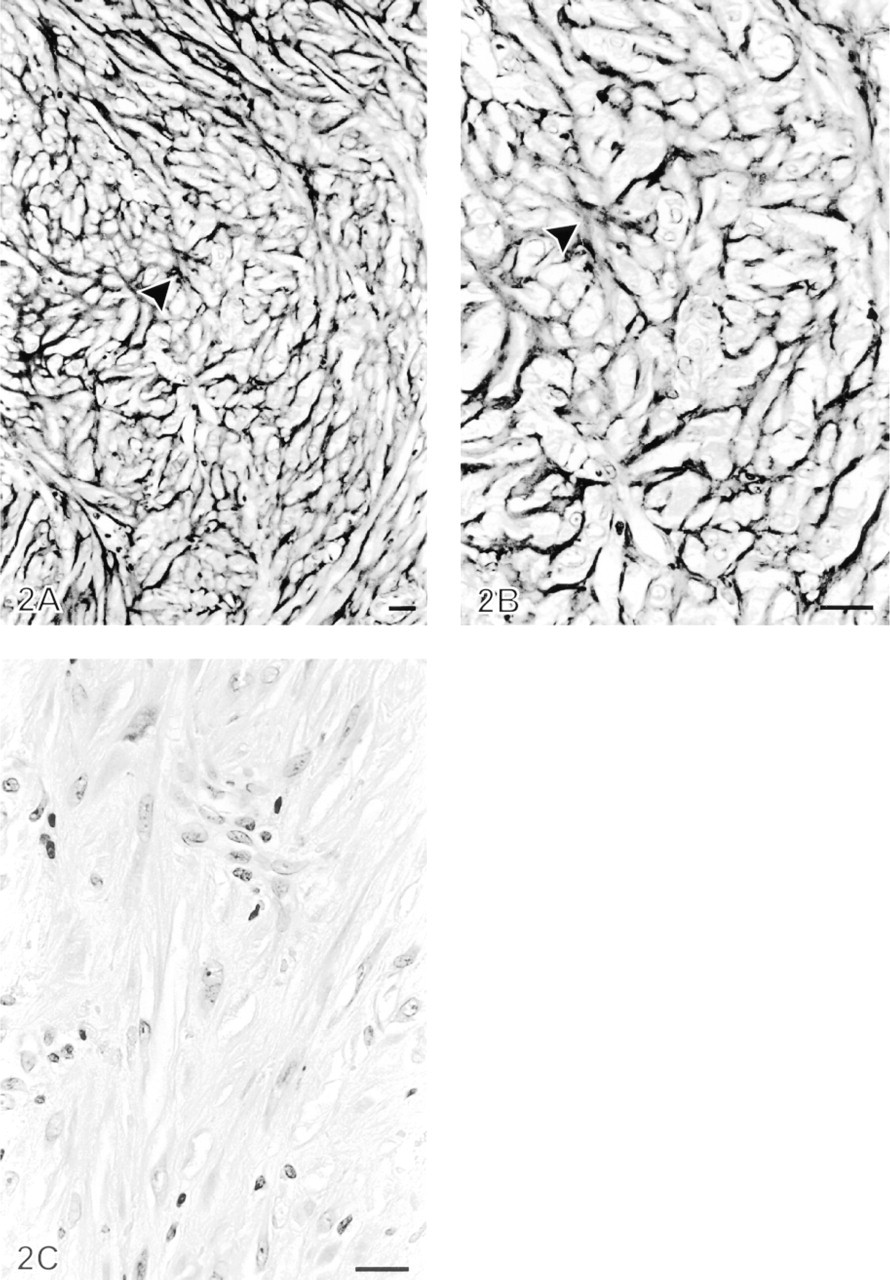
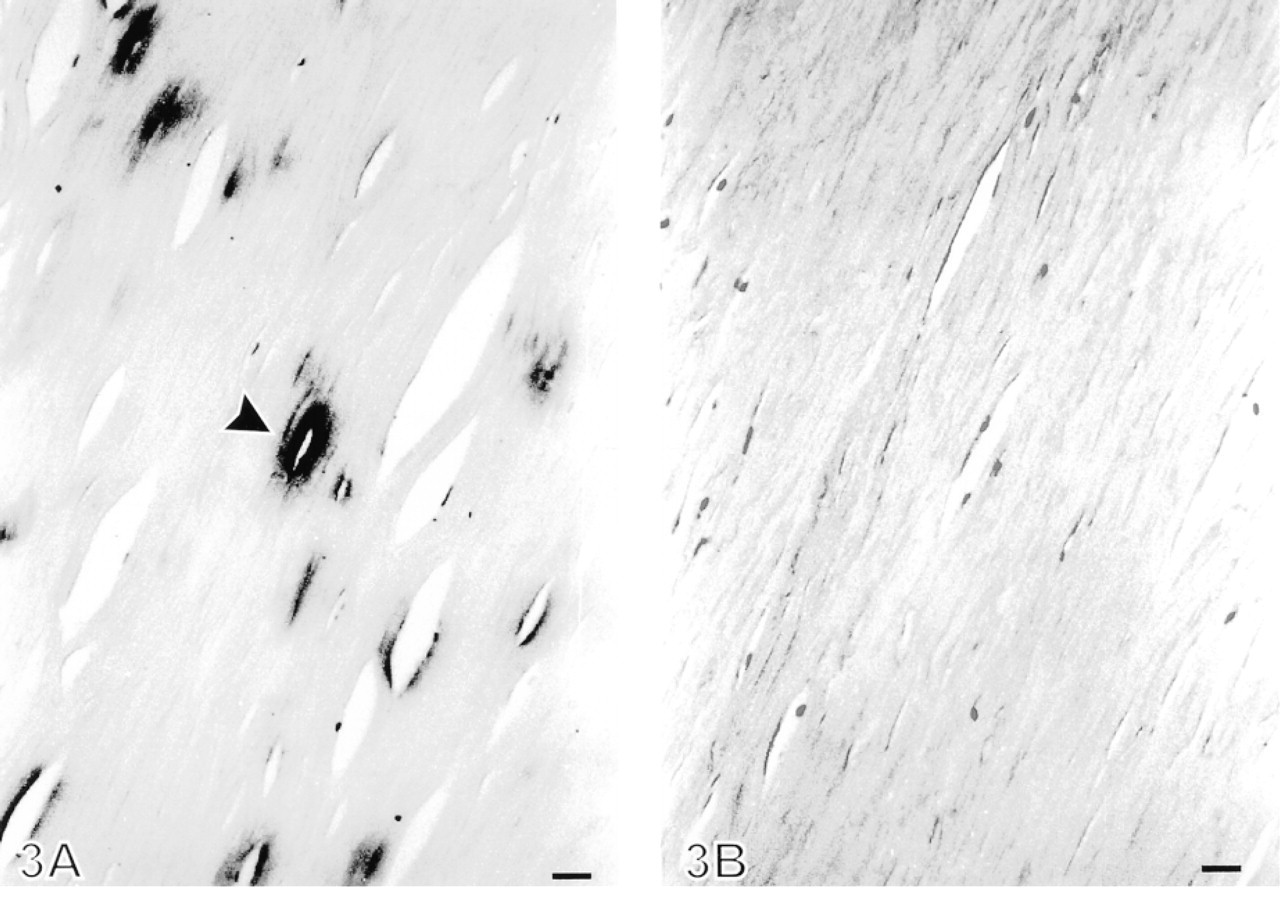
Diffuse tenascin mRNA expression in inflammatory pleura from a patient with empyema. (
Tenascin Immunohistochemistry
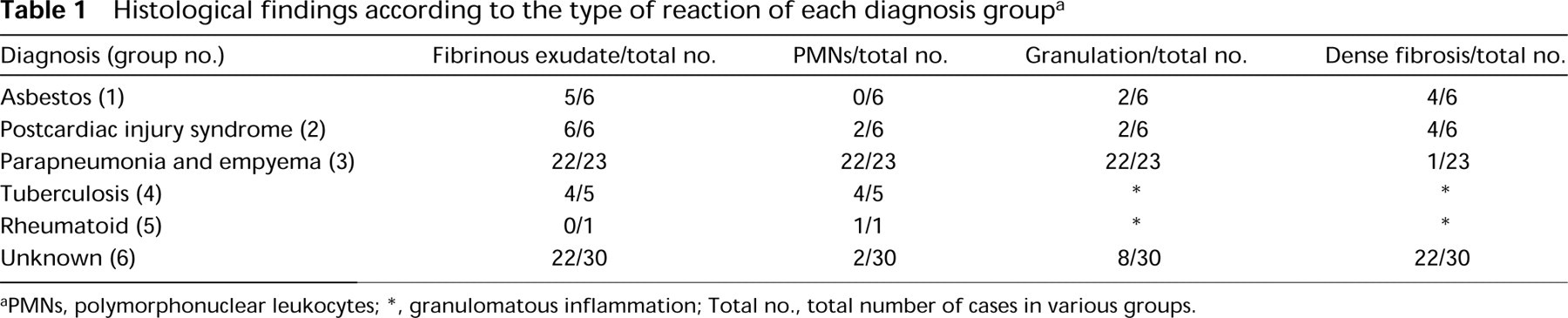
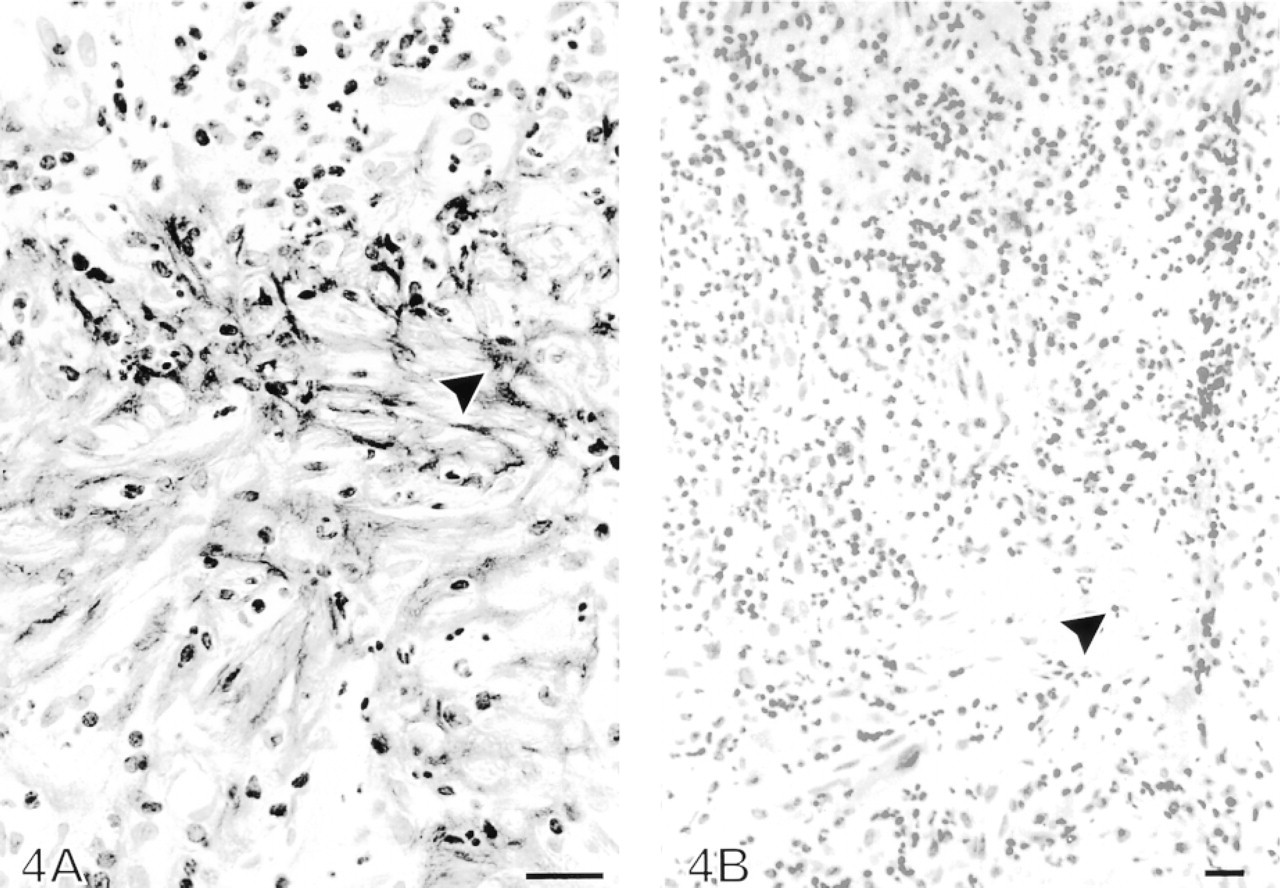
Tenascin was not detectable in normal pleura, in which the immunoreactivity for tenascin was scored as zero (0) (Figure 1). In contrast, in all cases with inflammatory and fibrotic lesions of the pleura, clearly positive immunoreactivity for tenascin was observed. In general, the immunoreactivity was more intense and extensive in active, newly formed granulation-like fibrosis than in old, dense fibrosis (Figures 2, 3, and 4). The immunoreactivity for tenascin was scored as strong (+ + +) in 34/71 cases, i.e., in every case with newly formed fibrosis. The intensity and localization of tenascin were similar in every disease group except for the five cases with tuberculosis and one case with rheumatoid disease in which the inflammatory reactions were granulomatous. The immunoreactivity for tenascin was scored as strong (+ + +) in granulomatous cases.
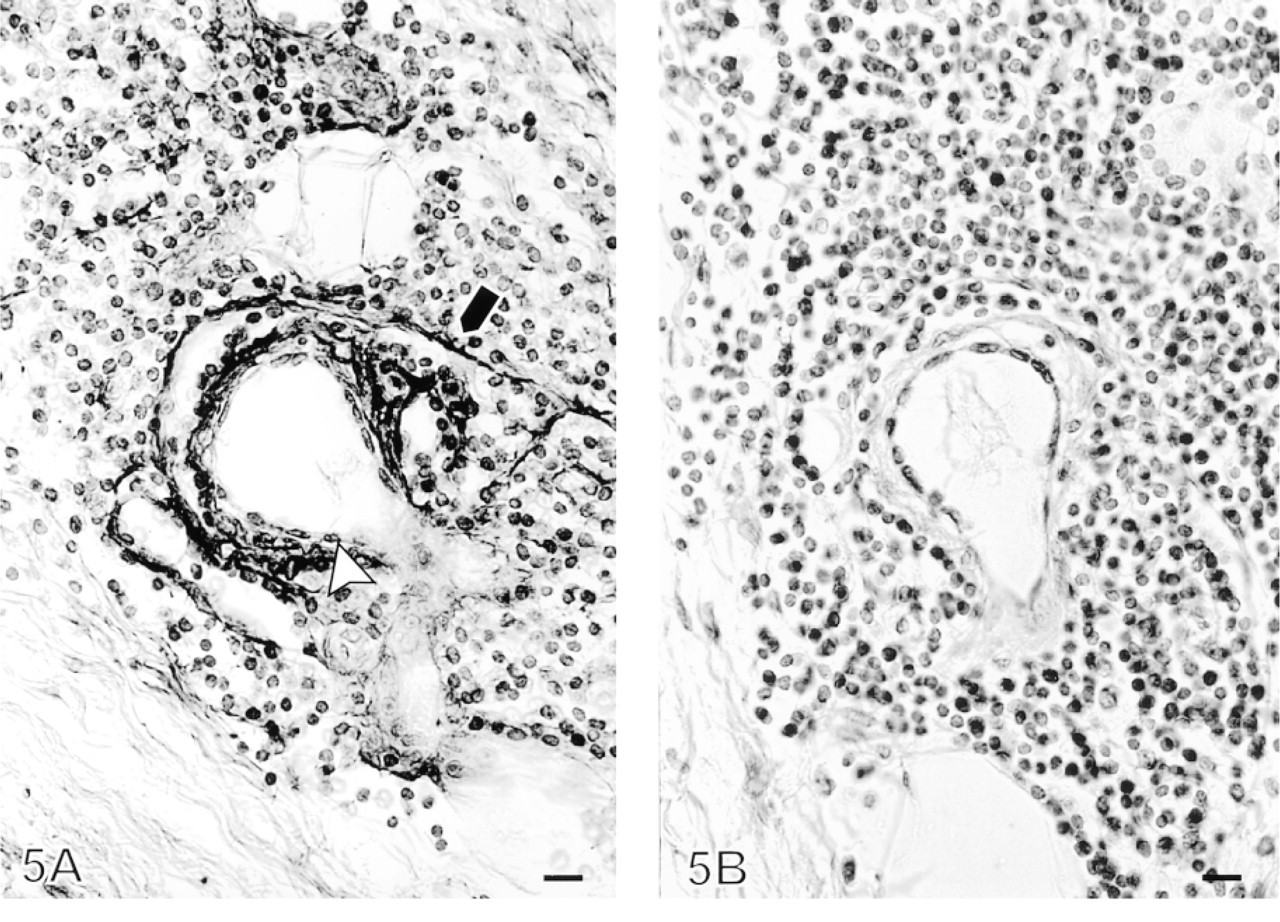
Tenascin immunoreactivity in old fibrosis was most intense in superficial areas of the pleura in zones underlying detached mesothelial cells. Immunopositive stripes could also be detected in deeper, sparsely cellular areas, where they were often localized around slitlike openings (Figure 3). The immunoreactivity for tenascin was scored as moderate (
Compared to all tenascin-negative control samples (five cases), all our study samples of different diseases (71 cases) were strongly or moderately positive for tenascin (p < 0.00001 by Fisher's exact probability test).
In the active, newly formed fibrosis, linear and occasionally coiled immunopositive fibers of tenascin were seen at a wider area than in the old fibrosis (Figures 2 and 3). Intense tenascin immunopositivity was localized on a fibrinous exudate overlying detached mesothelial cells. Tenascin immunopositivity could also be observed in the interstitial area of pleural extracellular matrix, but not within the cells.
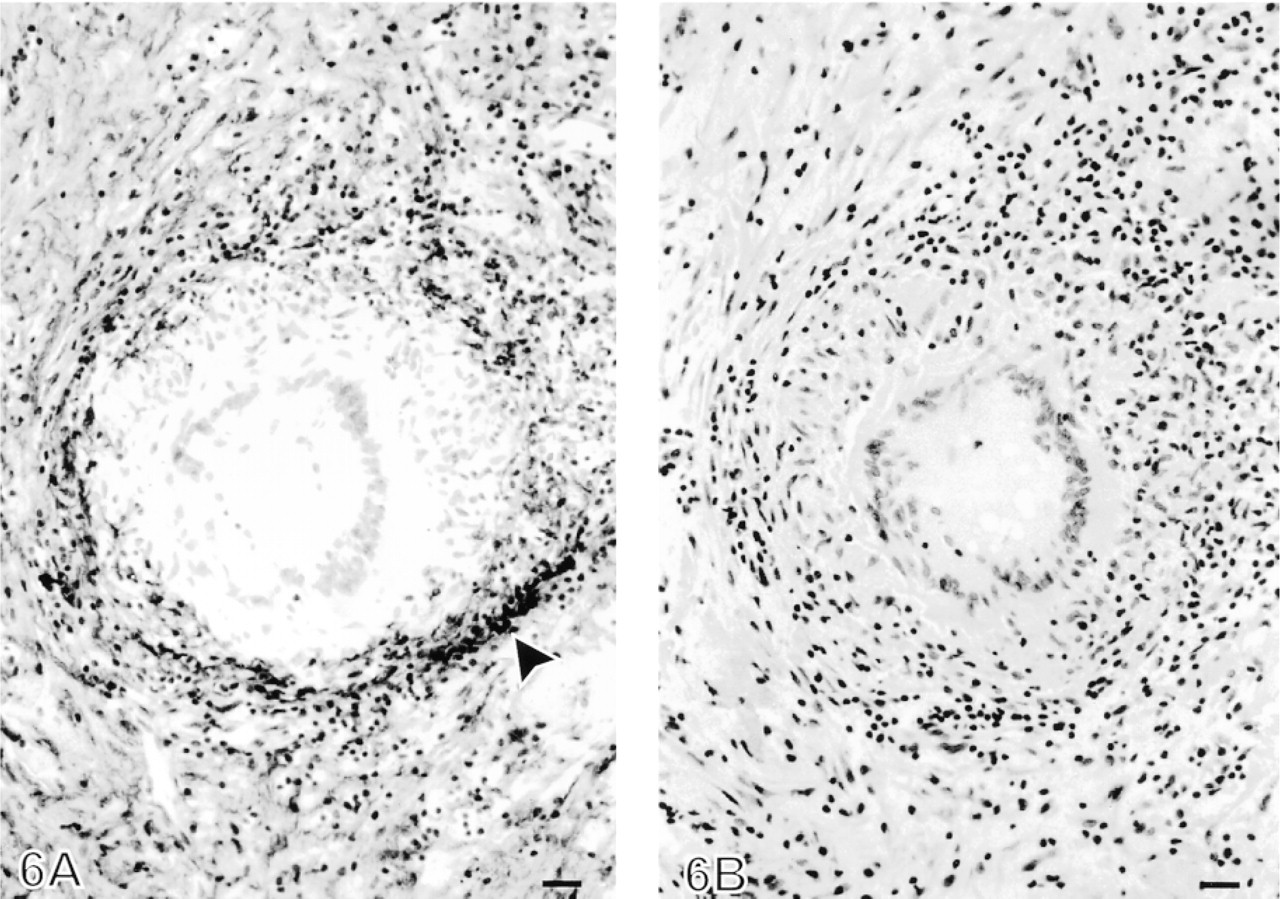
Pleural tuberculosis contained a very intense and extensive area of immunopositive linear fibers and network-type pattern for tenascin in fibrosis around the granulomas (Figure 6). Very few if any faintly immunopositive tenascin fibers were seen in the granulomas of pleural tuberculosis. Strong tenascin immunopositivity was also noted in fibrotic areas between the granulomas.
(
Tenascin mRNA ISH
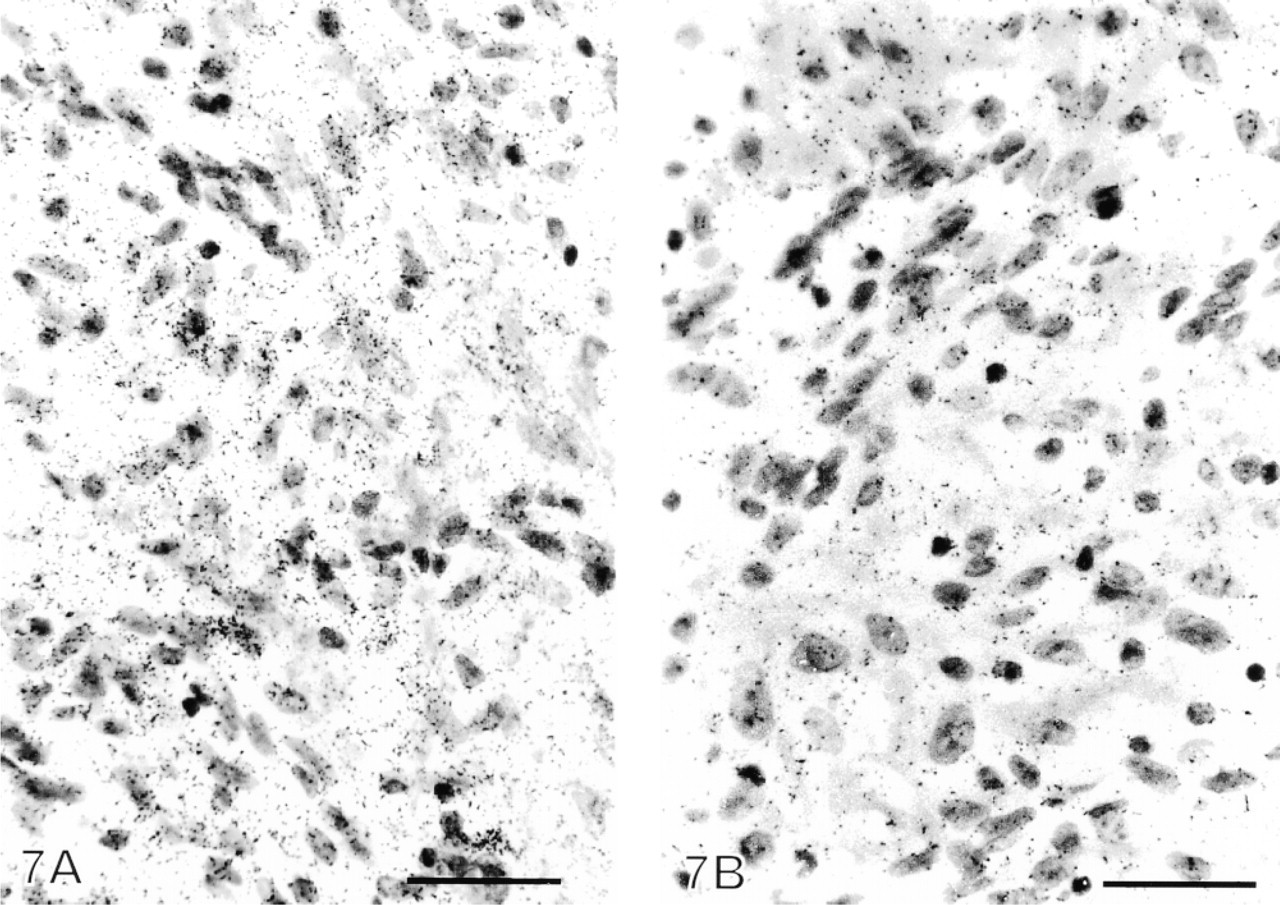
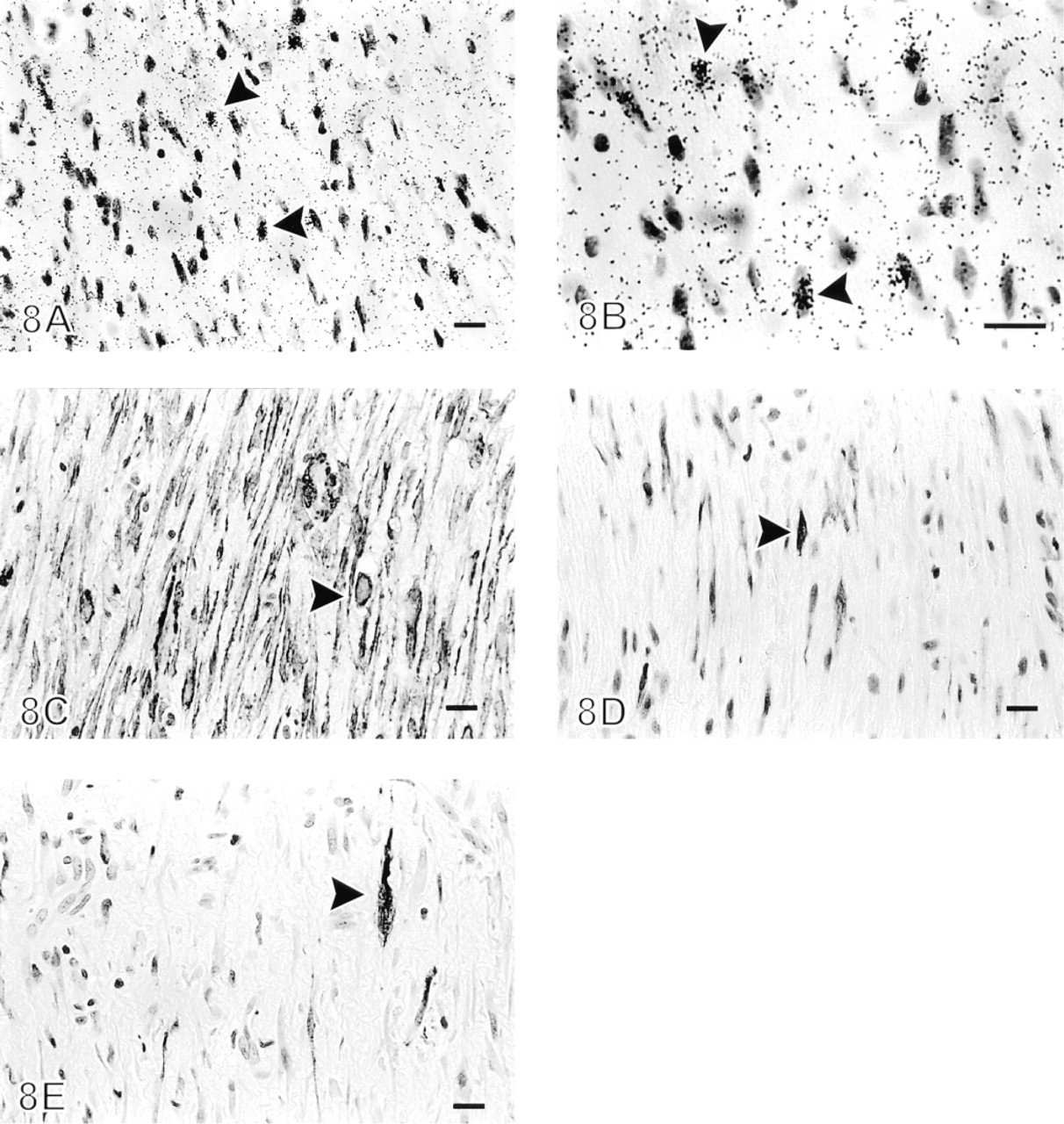
The numbers of mRNA grains were diffusely increased in 16 of the 19 cases in tissue hybridized with 35S-labeled antisense tenascin RNA probe compared to those hybridized with 35S-labeled sense RNA probe (Figure 7). The majority of the label for tenascin mRNA was localized in the granulation-like tissue beneath the fibrinous exudate, whereas in the dense and old fibrosis the label was sparse.
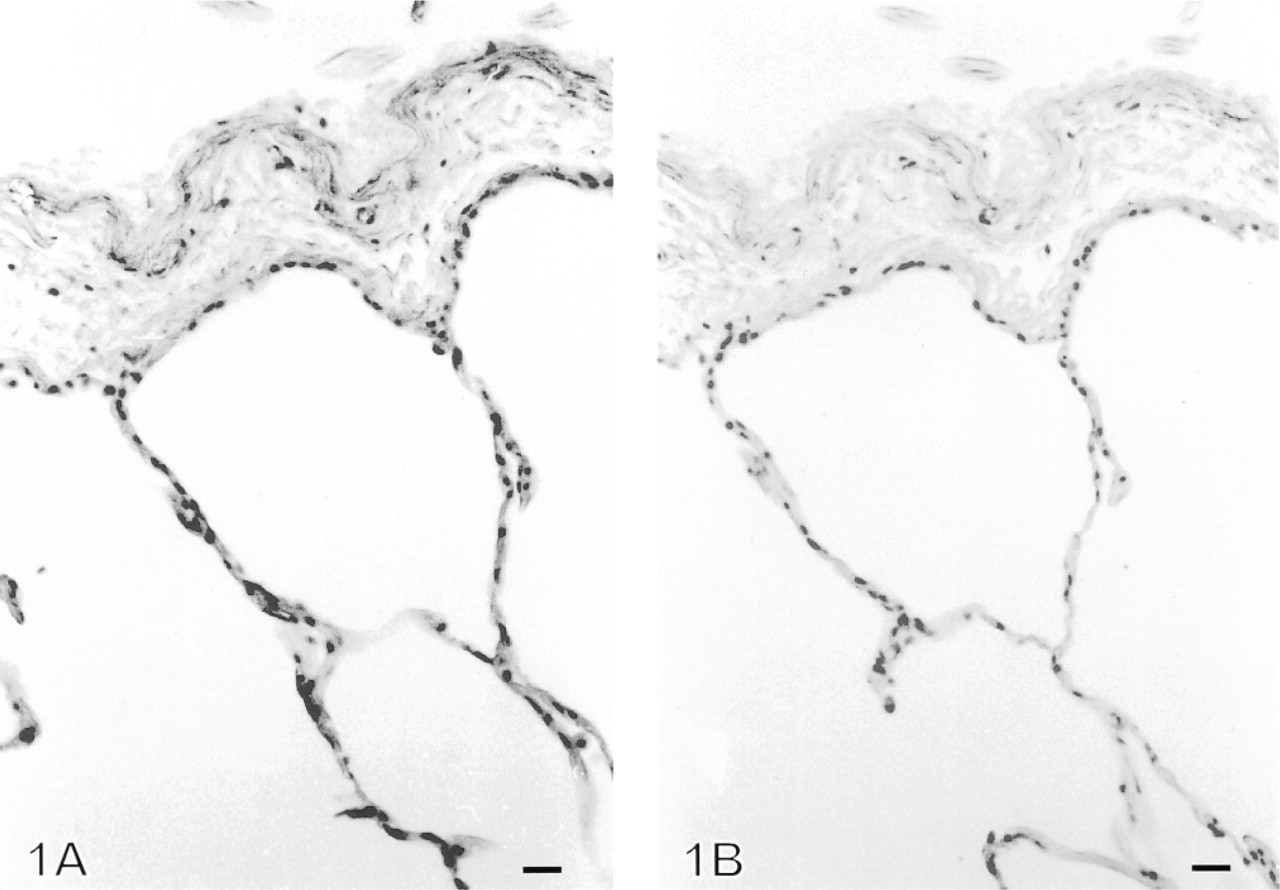
Large and oval mesenchymal cells of the matrix, especially in the granulation tissue underneath the fibrinous exudate, showed increased tenascin expression in 11 of 19 cases (Figure 8). The number of cells expressing tenascin mRNA varied from six to over 100 in one section (roughly 1 cm2) of pleural tissue (Table 2). Immunohistochemical staining on serial sections demonstrated that the majority of these cells were positive for α-smooth muscle actin, vimentin, desmin, and human muscle actin, suggesting a myofibroblastic phenotype (Schürch et al. 1998) (Figure 8). The cells were less frequently positive for cytokeratin, indicating either mesothelial cells and/or fibroblastictype cell differentiation with aberrant cytokeratin expression (Figure 8). The cells expressing tenascin mRNA were negative for FVIII-related antigen and CD34.
Individual cells showing strong tenascin mRNA expression could be detected significantly more often in the cells of pleural samples from patients with infective etiology (i.e., parapneumonia, empyema, and tuberculosis) than in those taken from other patients (p = 0.037 by Fisher's exact probability test). Individual cells showing strong tenascin mRNA expression could also be observed significantly more often in pleural samples showing granulation tissue than in those from samples showing only dense fibrosis (p = 0.0001 by Fisher's exact probability test).
Discussion
This is the first study showing increased tenascin expression by immunohistochemistry in pleural fibrotic and inflammatory diseases, such as asbestos-induced reactions, postcardiac injury syndrome, parapneumonic infection and/or empyema, tuberculosis, and rheumatoid disease. Strong immunopositivity for tenascin was detected especially in the newly formed fibrosis under detached mesothelial cells in every case, which is in keeping with our previous findings of increased tenascin immunopositivity in different types of fibrotic lung disorders, particularly in cases with usual interstitial pneumonia UIP, where tenascin was localized especially in the loose, newly formed fibrosis at the sites of recent alveolar epithelial injury (Kaarteenaho–Wiik et al. 1996).
ISH for tenascin mRNA expression as shown by number of individual cells expressing tenascin mRNA in one tissue section compared with the diagnosis and the reaction type of the pleural disease a

aDiagnosis group no: 1, asbestos; 2, postpericardiac syndrome; 3, parapneumonia and empyema; 5, unknown.
Tenascin immunoreactivity was intense in fibrinous exudate overlying the active granulation tissue-like lesions of fibrosis. In a recent study, we found increased tenascin concentrations in the serum and epithelial lining fluid (ELF) of the patients with UIP, sarcoidosis, and extrinsic allergic alveolitis (Kaarteenaho–Wiik et al. 1998). In addition, one recent study showed tenascin in malignant pleural effusions (Lawniczak et al. 1992). On the basis of this and previous findings, tenascin might possibly be detected in pleural effusions and might play a role as a marker for acute, ongoing inflammatory reaction in these disorders.
By ISH, foci of pleural interstitial matrix cells were shown to express tenascin mRNA. The tenascin mRNA-positive cells localized in the same areas that were positive for α-smooth muscle actin, vimentin, desmin, and human muscle actin, suggesting that they might be myofibroblasts (Schürch et al. 1998). However, our study has not been able to co-localize the expression signals in serial sections with 100% certainty. Further confirmative studies on this aspect are warranted. The finding that tenascin mRNA-positive cells in pleural matrix might be myofibroblasts is in line with the investigation by Hanamura and co-workers (Hanamura et al. 1997) demonstrating tenascin-C mRNA expression in myofibroblasts and in vascular and epithelial cells in human colon adenomas and carcinomas. A recent study by immunofluorescence and Western blotting with anti-tenascin antibodies and Northern blotting with a cDNA probe for tenascin showed that mesothelial cells in vitro are also capable of producing tenascin (Kinnula et al. 1998). Unfortunately, in fibrotic and inflammatory pleural lesions mesothelial cells usually detach, as they have also done in our study material, leading to difficulties in studying mesothelial cells in such disorders. Some cells in the granulation tissue and the fibrinous exudate, however, were positive also for cytokeratin, suggesting a mesothelial origin of tenascin mRNA expression in the minority of the cells.
A special feature of this study is the predominance of male patients; 59 of the 71 cases were men (83.1%). A large number of patients with a definite diagnosis as well as those with unknown etiology (30 of the 71) were heavy smokers. Smoking might increase the risk for prolonged and complicated pleural disease independent of the etiology of the disorder. Possible reasons for pleural disorders with unknown etiology could be viral infections, as previous studies have shown (Alptekin 1958; Fine et al. 1970). Another reason might be nondetectable (low-level) exposure to asbestos fibers. Cigarette smoke itself is known to cause activation of inflammatory cells and recruitment of neutrophils and to enhance oxidant-related cell damage and inflammation. Inflammatory cytokines induce tenascin expression both in airway epithelial cells and in human mesothelial cells, at least in vitro (Härkönen et al. 1995; Kinnula et al. 1998).
In conclusion, our results demonstrate that tenascin is increased in etiologically varied inflammatory and fibrotic pleural diseases. Tenascin is actively synthesized in the granulation tissue-like areas of the newly formed fibrosis. Therefore, tenascin expression in pleural lesions may serve as a marker for an acute and ongoing reaction. The tenascin mRNA-positive cells localized in areas in which the cells might be mainly myofibroblasts, and less frequently in the mesothelial cells and/or fibroblasts expressing aberrant cytokeratin.
Footnotes
Acknowledgements
Supported by the Finnish Cancer Societies, the Finnish Anti-Tuberculosis Association Foundation, the Cancer Society of Northern Finland, the Paulo Foundation, and Suomen Astra Oy.
The technical assistance of Ms Mirja Vahera, Ms Erja Tomperi, Ms Riitta Vuento, Ms Marjo Kaarteenaho, and Mr Hannu Wäänänen and Mr Tapio Leinonen is gratefully acknowledged.
