Abstract
Colorectal cancer can develop through molecular, chromosomal, and epigenetic cumulative changes that transform the normal intestinal epithelium into the colorectal polyps, called conventional adenomas (CAs) or serrated polyps (SPs), recognized as precursors of invasive colorectal neoplasia. These benign lesions need to explore the morphology, histological diagnosis, and biomarkers profile to accurately characterize lesions with potential for evolution to cancer. This study aimed to correlate the immunohistochemical expression of Parkin and Adenomatous Polyposis Coli (APC; tumor suppressors), Human Apurinic/Apyrimidinic endonuclease 1 (APE1), and B-cell lymphoma-extra-large (Bcl-xL; oncogenic proteins) in sporadic colorectal polyps with clinical, endoscopic, and diagnostic data. Immunohistochemical analysis was performed on tissue microarray samples of 306 polyps. Based on the Allred score, the expressions were graduated in the cytoplasm and nucleus of superficial and cryptic cells. There was higher Parkin nuclear expression (p=0.006 and 0.010) and APC cytoplasmic expression in cryptic cells (p<0.001) in SPs. CAs, APE1 (p<0.001) and Bcl-xL (p<0.001) were more expressed in the nuclei and cytoplasms, respectively. These results are related to the biological role proposed for these proteins in cellular functions. They can contribute to the diagnosis criteria for polyps and improve the knowledge of biomarkers that could predict cancer development:
Introduction
Colorectal cancer (CRC) is the third most commonly occurring cancer in men and the second in women worldwide. 1 The life risk is around 5% in the Western world 2 and is correlated to physical, chemical, and biological agents, closely linked to eating habits.3,4 The transformation of the colorectal epithelium into neoplasia involves a succession of cumulative changes.5,6 Initially, benign lesions called conventional adenomas (CAs) or serrated polyps (SPs) are formed, which are the most common precursor lesions of the CRC.2,4 Afterward, changes happen on the mechanisms of proliferation like blood vessel formation, and the cell gets the capacity to invade tissues and form distant metastasis of the original tumor.5,7
The World Health Organization (WHO) subdivides the SPs, until 2019, into three groups: hyperplastic polyps (HPs), sessile serrated adenomas/polyps (SSA/Ps) with or without cytological dysplasia, and traditional serrated adenomas (TSAs) with or without conventional dysplasia.8–12 It was proposed that SSA/Ps are considered an evolution of HPs in their progression pathway to CRC. 13 Currently, the classification is subdivided into five groups: HPs, sessile serrated lesions (SSLs), sessile serrated lesions with dysplasia (SSLs-D), TSAs, and unclassified serrated adenomas. 14
Two thirds of sporadic CRCs were reported to arise from CAs, which are subdivided into advanced conventional adenomas (AAs) and non-advanced conventional adenomas (NAAs), and up to 35% of sporadic CRCs arise from SPs.10,15 CAs are characterized by having some degree of dysplasia, when compared with SPs that cannot have nuclear dysplasia. 16 Regarding the risk of malignancy, SSLs, especially those from the right colon, were recognized as the primary precursor lesions that lead to a faster tumor progression between the subgroups of the SP pathway, and also in comparison with CAs. 10 HPs, previously considered benign, did not recommend patient follow-up. Endoscopists did not bother to resect small HP-type lesions due to the absence of a risk of malignancy.17–19 Currently, however, due to the knowledge of the risk of malignancy of SSLs and the difficulty in differentiating between the SPs through colonoscopy, it is described, and it is mandatory to remove them for histological examination.17–19
A summary of these epithelial colonic polyp classifications is shown in Table 1.
Summary Classification of Serrated Polyps and Conventional Adenomas of the Colon.

Conventional adenomas with size ≥ 10 mm in diameter or with prominent villous or with high-grade dysplasia.
In this way, there is a need for more in-depth knowledge about the morphology of polyps among pathologists to improve the histological diagnosis and define that the immunohistochemical (IHC) biomarker profile characterizes lesions with potential for evolution to CRC. The evaluation of the IHC biomarkers in premalignant lesions has helped predict the malignant transformation and has provided a better understanding of these polyp’s role in different pathways of carcinogenesis and the biological behavior of these lesions, contributing to refining the diagnosis, increasing the reliability of results, and allowing better CRC prevention and treatment strategies. 20
The Parkin RBR E3 ubiquitin protein ligase (PRKN) gene encodes Parkin, an E3 ubiquitin ligase protein, that ubiquitinates proteins for control of intracellular traffic or induces proteasomal degradation of those unfolded and damaged21,22 and also regulates mitochondrial homeostasis 23 and biogenesis (fusion, fission, DNA repairs, and destruction). 24 PRKN is a tumor suppressor gene whose haploinsufficiency collaborates with the mutant Adenomatous Polyposis Coli (APC), other tumor suppressor genes, in colorectal alterations, accelerating adenoma progression and increasing polyp multiplicity in mutant mice. 25 PRKN probably is also involved in negative feedback on the epidermal growth factor proliferation-signaling pathway in CRC 26 and can decrease cell cycle progression by proteasomal degradation of cyclins D and E.27,28 Parkin also contributes to the proteasomal degradation of Human Apurinic/Apyrimidinic endonuclease 1 (APE1), a DNA repair and positive redox (reduction/oxidation) regulator of transcriptional factors, that maintains the transcription factors involved in promoting and progressing cancer in a reduced activity state. 29 Parkin regulates apoptosis, controlling the stability of B-cell lymphoma-extra-large (Bcl-xL) protein by its ubiquitination to proteasomal degradation 30 and seems to induce mitophagy, control cell proliferation, and apoptosis.27,31
This is the first study that describes the expression of Parkin, APC, APE1, and Bcl-xL proteins together in colorectal polyps. Furthermore, it aims to analyze these possible biomarkers to collaborate in the characterization of polyps and increase the quality of diagnosis and prevention of CRC. These markers were chosen because they have interactions with each other and can be in the same cancer pathway.
Materials and Methods
Study Population and Sample Collection
The Ethics Research Committee of the Hospital de Clínicas da Universidade Federal do Paraná (HC-UFPR) approved this study, with registration number 820,432. In total, 306 epithelial polyps from sequential patients undergoing colonoscopies in 2008 diagnosed as CAs or SPs that had adequate histological material for analysis were included. These samples were obtained from the Pathology Service of Hospital Santa Cruz in Curitiba, Paraná, Brazil, and were used in previous studies.32,33 Reanalysis of these samples was performed by physicians specialized in gastrointestinal pathology to confirm the anatomopathological diagnosis. 32 Inflammatory and hamartomatous polyps or that presented diagnostic disagreement in the reanalysis were excluded. The patient population included both men and women (n=177 and 129, respectively), aged >18 years.
Histopathological and clinical variables were obtained from patient files stored in the hospital database. The following clinicopathological variables were considered: gender, age, colon polyp location (right, left, transverse, or rectosigmoid regions, and proximal or distal to the splenic angle), number of polyps, polyp size in the endoscopic examination, and appearance of polyps (sessile or pedunculated).
Tissue Microarray (TMA) and Immunohistochemistry
TMA and IHC analyses were conducted at the Laboratory of Experimental Pathology of the Pontifícia Universidade Católica do Paraná (PUCPR). Hematoxylin and eosin slides were prepared from original paraffin blocks of polyps to select areas for TMA construction. The selected polyps’ areas were isolated using a semi-automated method, and samples were spotted along with normal matched control tissues on the TMAs. Seventeen TMAs were constructed with all the 306 samples. For IHC, the TMA slides were subjected to antigen retrieval with Target Retrieval Solution (Dako; Glostrup, Denmark) and then incubated with the following monoclonal antibodies: anti-Parkin antibody (mouse, 1:100; Abcam, Cambridge, UK), anti-APC antibody (rabbit, 1:200; Abcam), anti-APE1 antibody (rabbit, 1:600; Abcam), anti-Bcl-xL antibody (rabbit, 1:100; Abcam), Subsequently, disclosure polymer (Spring Bioscience Corp, Pleasanton, CA) was used as a secondary antibody. Slides were incubated with diaminobenzidine complex and substrate and then counterstained with Harris hematoxylin. Positive and negative controls were included in the IHC analysis for each antibody. The positive controls for Parkin, APC, APE1, and Bcl-xL were human colon cancer samples, with known positivity and intensity. Negative controls were made by omitting the primary antibodies.
Allred Score Analysis
The graduation method of IHC expression was the Allred semi-quantitative score that analyzed the nuclear and cytoplasmic expressions of proteins in the superficial and cryptic epithelium. This score consists of two values: one of proportion and another of the intensity of staining. The proportion score is subdivided into 0 (0% positive nuclei or cytoplasm), 1 (>0–1%), 2 (>1–10%), 3 (>10–33%), 4 (>33–66%), and 5 (>66–100%). The intensity graduation is subdivided into 0 (negative), 1 (weak), 2 (moderate), to 3 (strong). The total score is calculated with the addition of the proportion and intensity values and ranges from 0 to 8. 34
For the intensity evaluation, the positive and negative controls were used as calibration. The images of the human protein atlas were consulted to compare the protein distribution in normal and abnormal tissues. 35
Statistical Analysis
Statistical analysis was performed using the SPSS Statistics v.20 software, the Canva for Fig. 4, and the JMP program to develop the graphs. To compare clinicopathological and quantitative variables, the non-parametric Mann–Whitney and Kruskal–Wallis tests were used to identify significant differences between two or more groups, respectively. The Chi-square test evaluated the association between qualitative variables. The values of p<0.05 were considered statistically significant.
Results
The clinicopathological characterization of the patient and polyps are shown in Table 2. Patients had a median age of 60 years (range, 19–91). The sample consists of polyps located in the proximal and distal colon (before and after splenic flexure, respectively) in the right, transverse, left, and rectosigmoid regions. There are more patients (63.7%) with multiple than single and with sessile (90.5%) than pedunculated polyps. Regarding the size, 77.1% were 2 to 8 mm and 22.9% more than 10 mm; polyps smaller than 2 mm were not removed from the patients. Polyps were diagnosed in CAs and SPs: NAAs (41.5%) and AAs (33.3%) were diagnosed in CAs, and HPs (15%), SSLs (8.8%), and TSAs (1.3%) were diagnosed in SPs.
Clinicopathological Characteristics of Patients and Polyps.
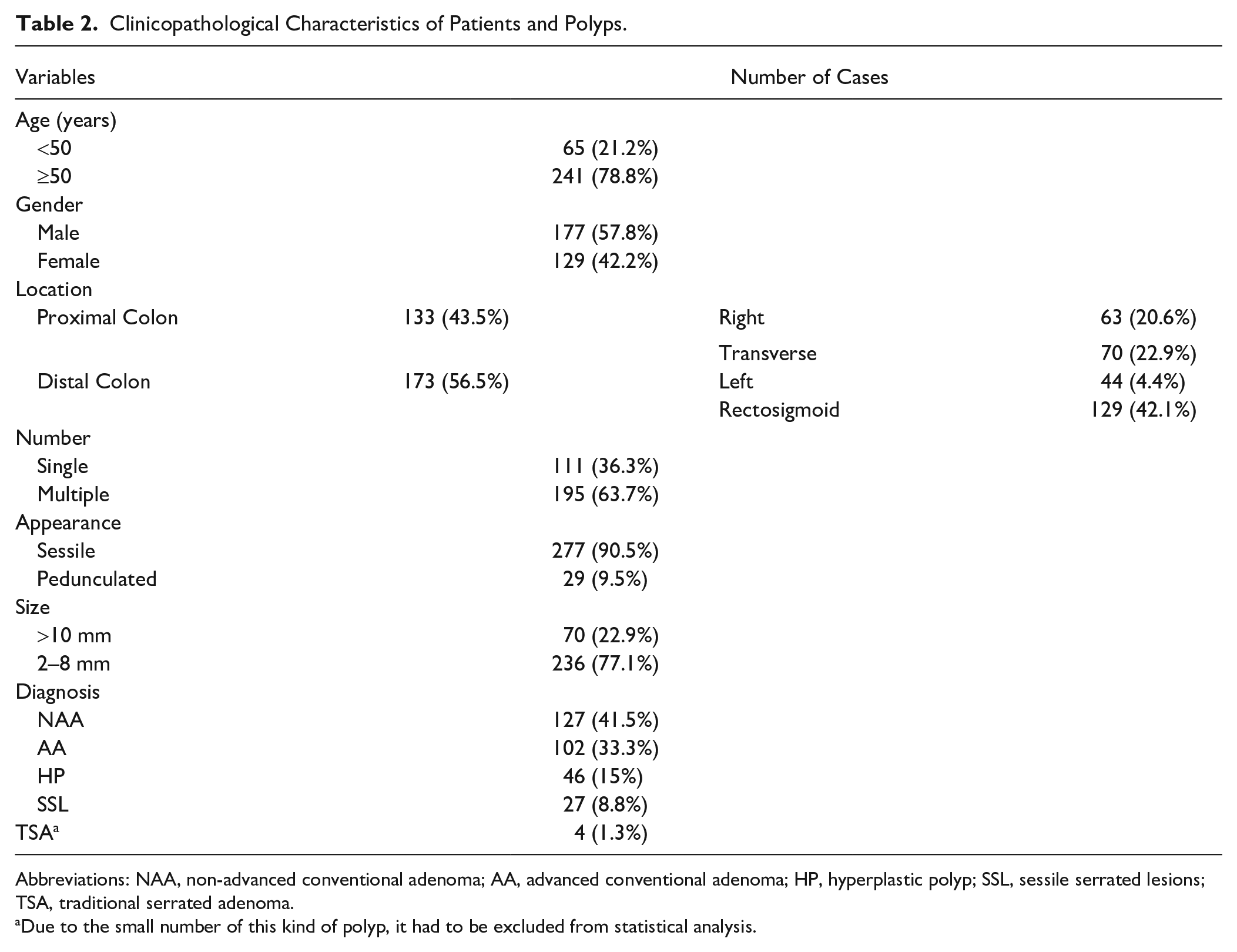
Abbreviations: NAA, non-advanced conventional adenoma; AA, advanced conventional adenoma; HP, hyperplastic polyp; SSL, sessile serrated lesions; TSA, traditional serrated adenoma.
Due to the small number of this kind of polyp, it had to be excluded from statistical analysis.
The results reported in this research were those that had statistical significance in our analyses and correlations. Therefore, all the data listed in Table 2 were correlated with the expression of each one of the evaluated proteins. Non-significant results have not been reported.
Parkin immunoreactivity in nucleus and cytoplasm showed higher expression in cytoplasm, independent of the polyps’ cell regions, superficial or cryptic epithelium. On the contrary, APC showed expression in the cytoplasm, which was higher in the superficial epithelium and negative in the nucleus. APE1 showed little difference in expression between cell and polyps’ regions. Moreover, Bcl-xL showed higher expression in cytoplasm independent of polyps’ regions (Table 3). Figure 1 demonstrates Parkin nuclear expression in HP and CA. Figure 2 shows the antagonism of APC cytoplasmic expression in one HP and one CA.
Parkin, APC, APE1, and Bcl-xL Expression in Different Parts of the Cells and Regions of the Polyps.
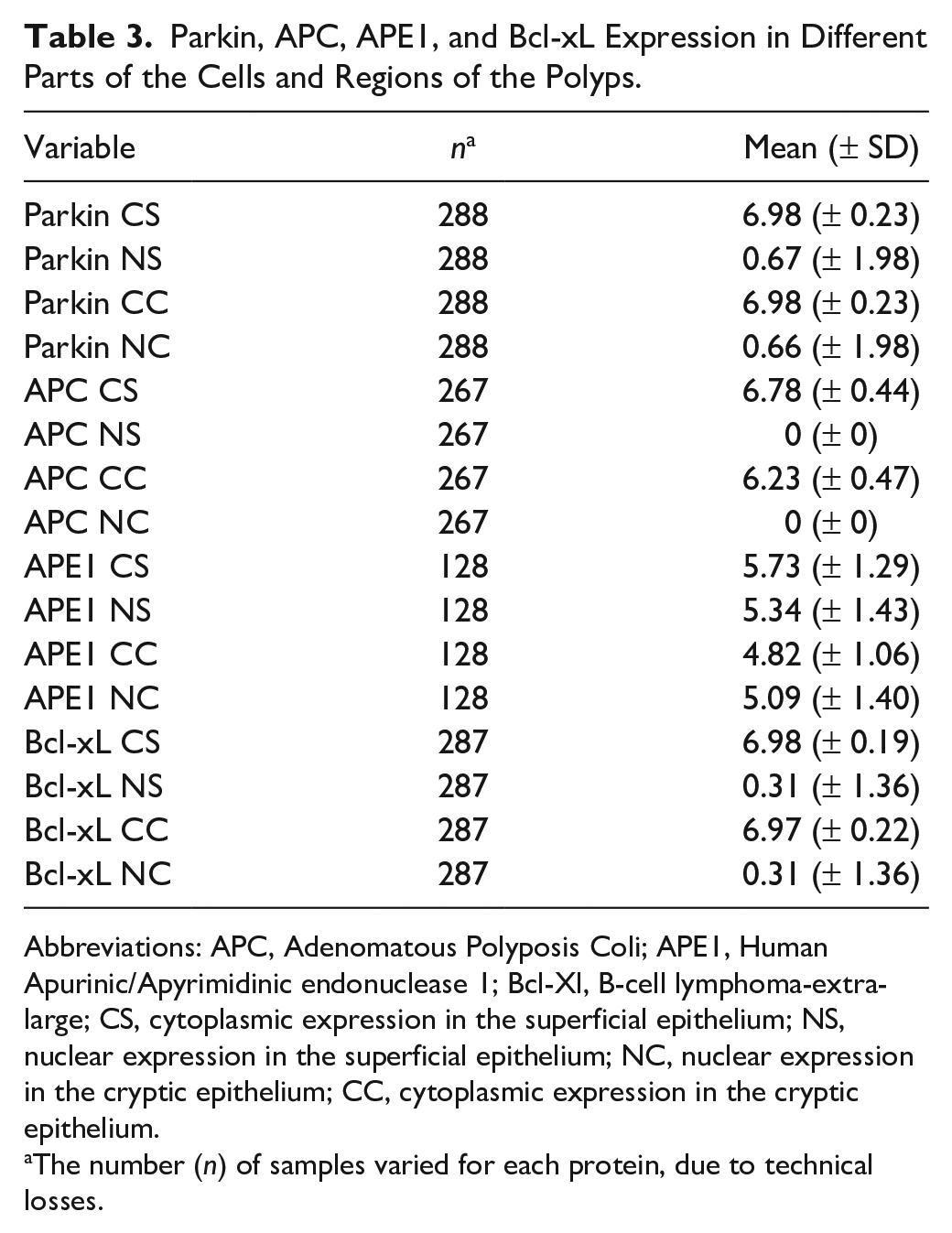
Abbreviations: APC, Adenomatous Polyposis Coli; APE1, Human Apurinic/Apyrimidinic endonuclease 1; Bcl-Xl, B-cell lymphoma-extra-large; CS, cytoplasmic expression in the superficial epithelium; NS, nuclear expression in the superficial epithelium; NC, nuclear expression in the cryptic epithelium; CC, cytoplasmic expression in the cryptic epithelium.
The number (n) of samples varied for each protein, due to technical losses.
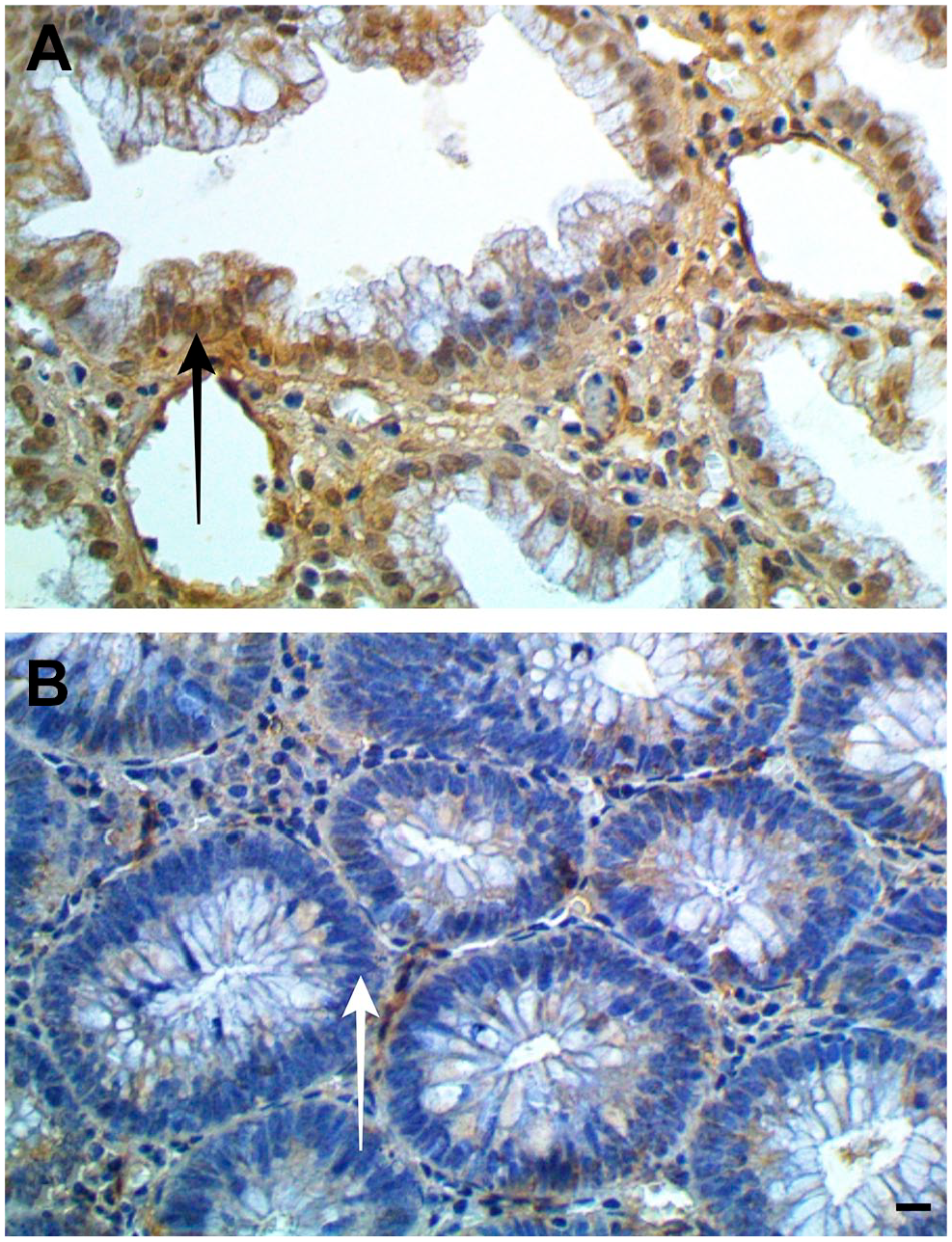
Nuclei are positive (black arrow) for Parkin in the hyperplastic polyp (A) and negative (white arrow) in the conventional adenoma (B). Scale bar indicates 20 µm. Anti-Parkin Antibody.
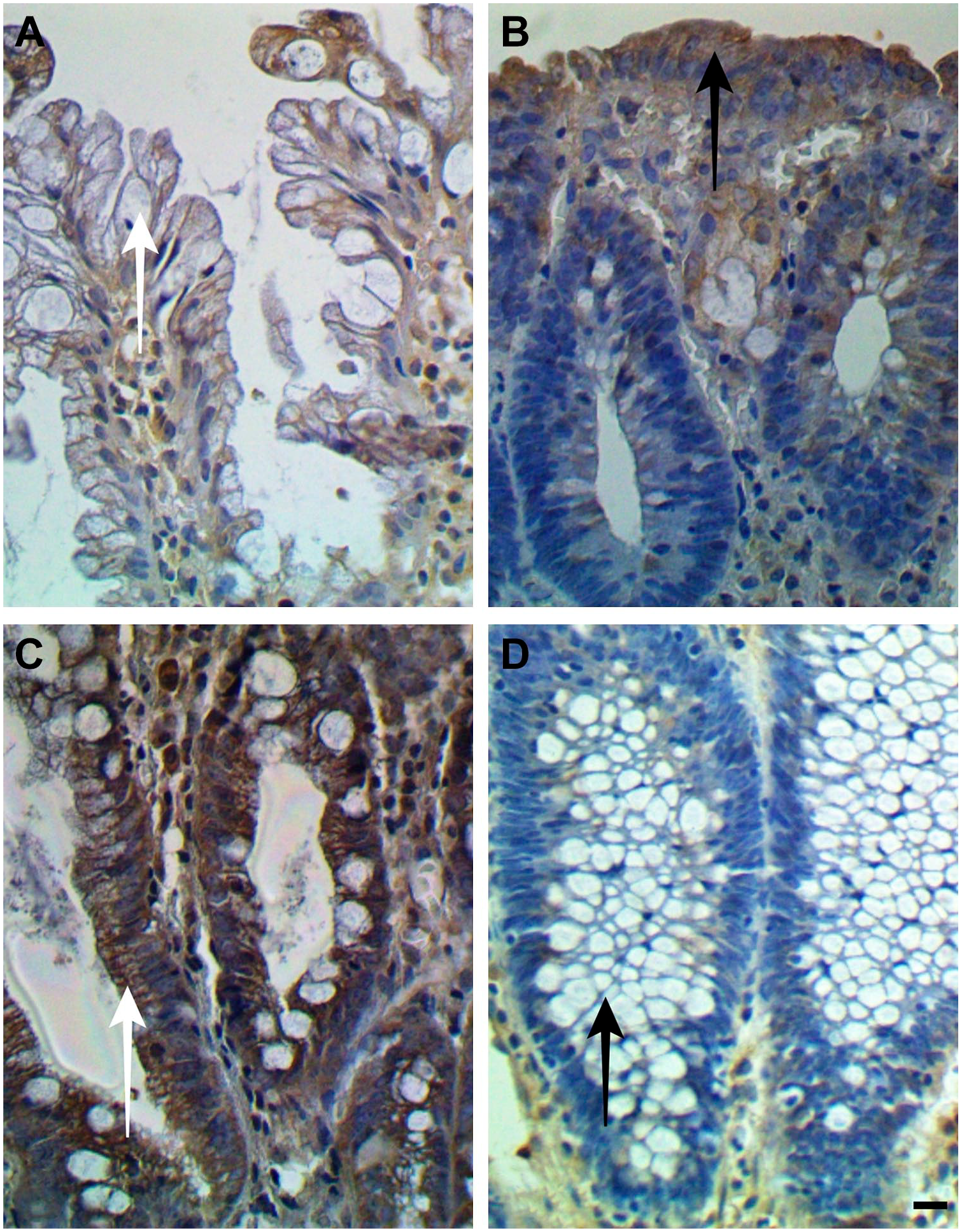
The cytoplasm of the superficial epithelium is more marked in B than in A (arrows). The cytoplasm of the cryptic epithelium is more marked in C than in D (arrows). Photos of hyperplastic polyp in A and C and conventional adenoma in B and D. Scale bar indicates 20 µm. Anti-APC Antibody. Abbreviation: APC, Adenomatous Polyposis Coli.
IHC analysis of proteins immunoreactivity in different polyp’s diagnosis showed statistically significant differences (Table 4 and Fig. 3). Parkin nuclear superficial (Parkin NS) and cryptic (Parkin NC) showed higher expression in SPs than in CAs in the superficial and cryptic epithelium. APC showed lower cytoplasmic expression in SPs than in CAs in the superficial epithelium (APC CS), and higher expression in SPs than in CAs was observed when analyzing the cryptic epithelium (APC CC). In the superficial and cryptic epithelium, the nuclear expression of APE1 (APE1 NS and NC) and cytoplasmic expression (Bcl-xL CS and CC) were higher in CAs than in SPs. These results are detailed in Table 5, where polyps’ diagnosis was grouped two by two and analyzed related to proteins immunoreactivity. No association was observed between polyps’ diagnosis and cytoplasmic Parkin and APE1 and nuclear APC and Bcl-xL. Parkin NC also demonstrated a significant difference regarding the polyps’ size, with higher expression in larger polyps (10–30 mm) as shown in Table 6. Similar to Parkin, APE1 NS and NC also demonstrated higher expression in larger polyps. Concerning polyps aspect, higher expression of APE1 NC was observed in pedicellate (Table 6). APC CS showed higher expression in the right and transverse colon polyps, and APC CC was higher in rectosigmoid. No association was observed between polyps localization, size, aspect, risk, and Bcl-xL, and no association was observed concerning all proteins and the gender and number of polyps.
Parkin, APC, APE1, and Bcl-xL Expression by Diagnosis in Parts of the Cells and Regions of the Polyps.
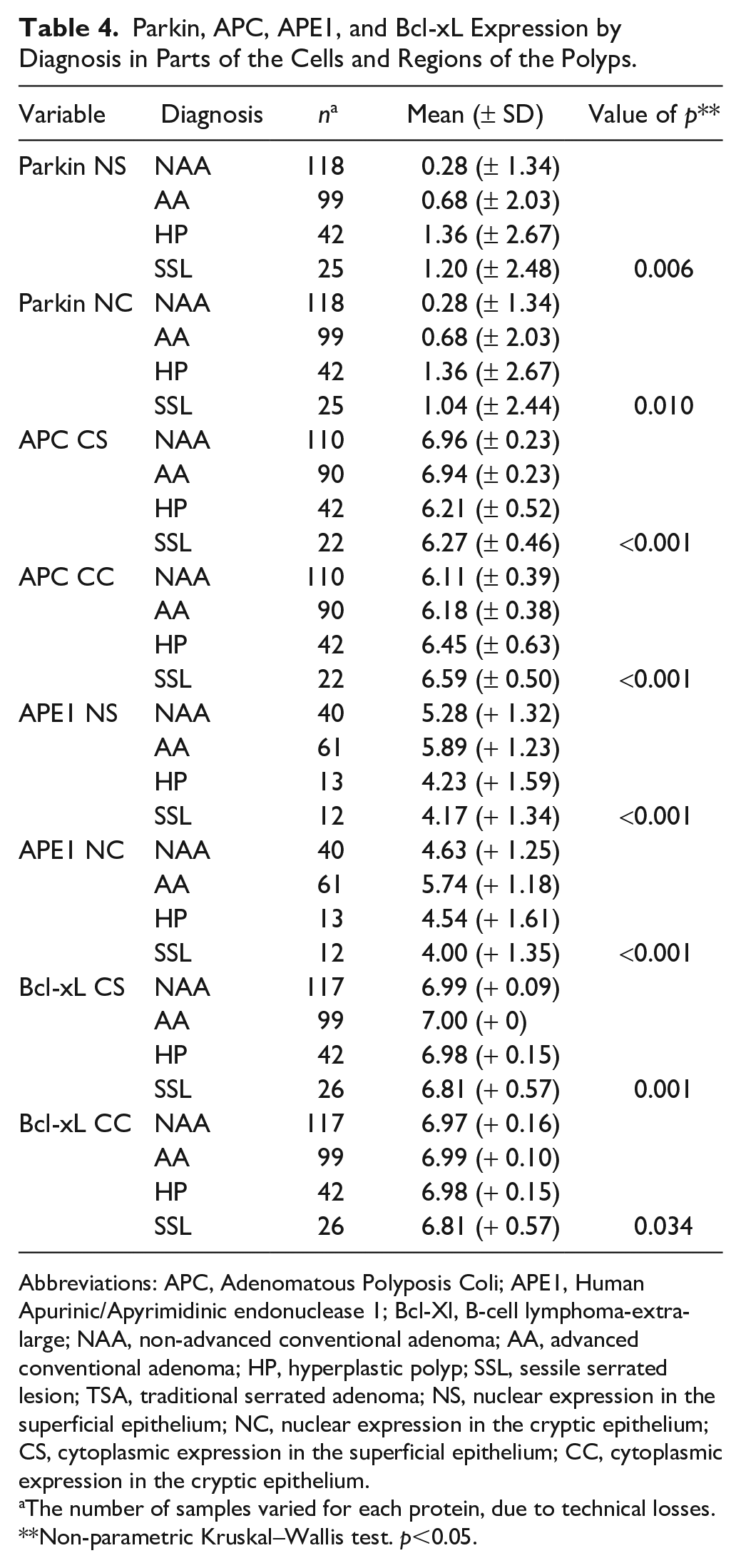
Abbreviations: APC, Adenomatous Polyposis Coli; APE1, Human Apurinic/Apyrimidinic endonuclease 1; Bcl-Xl, B-cell lymphoma-extra-large; NAA, non-advanced conventional adenoma; AA, advanced conventional adenoma; HP, hyperplastic polyp; SSL, sessile serrated lesion; TSA, traditional serrated adenoma; NS, nuclear expression in the superficial epithelium; NC, nuclear expression in the cryptic epithelium; CS, cytoplasmic expression in the superficial epithelium; CC, cytoplasmic expression in the cryptic epithelium.
The number of samples varied for each protein, due to technical losses.
Non-parametric Kruskal–Wallis test. p<0.05.
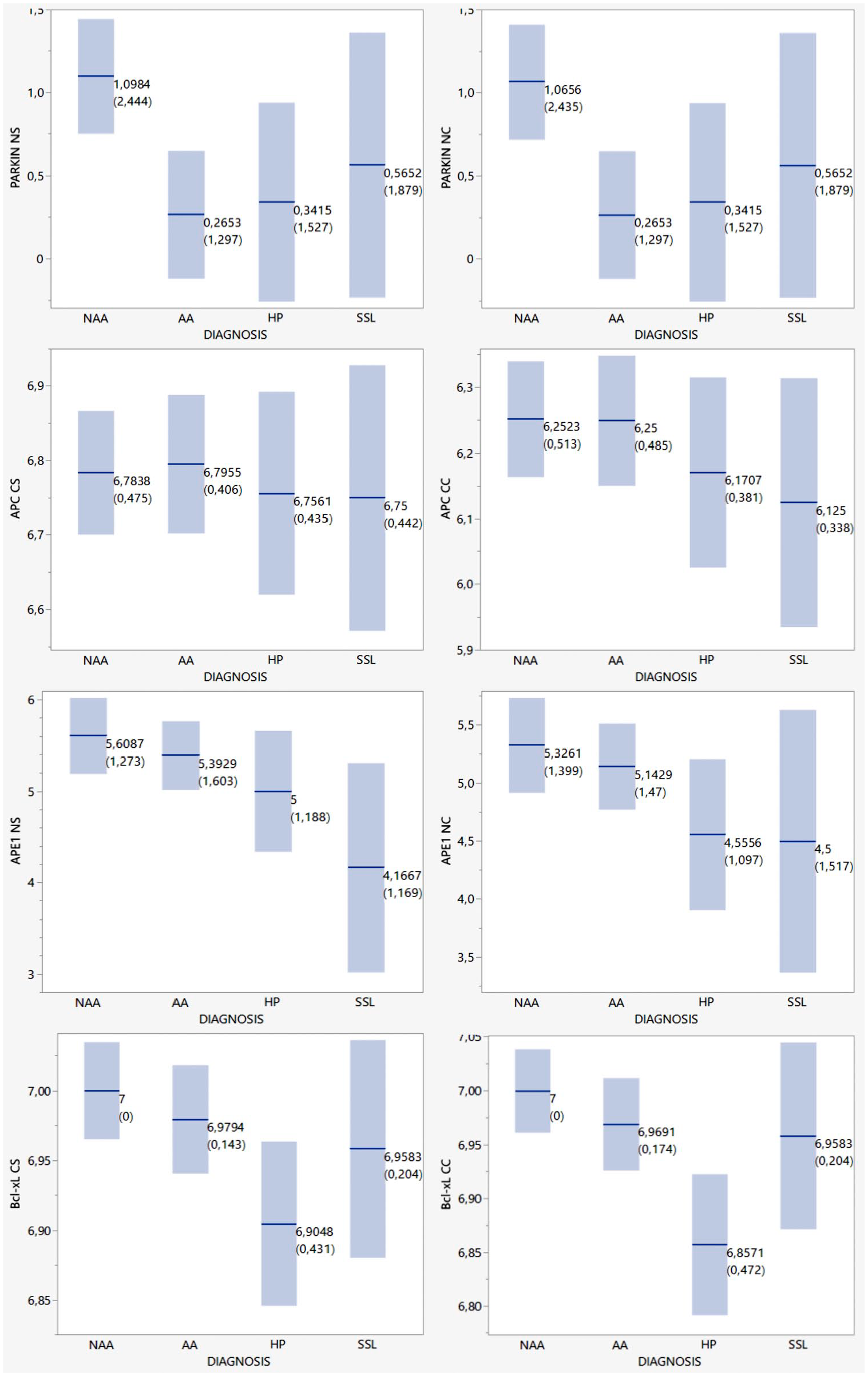
Main differences between IHC expression by polyp type. Abbreviations: NS, nuclear expression in the superficial epithelium; NC, nuclear expression in the cryptic epithelium; CS, cytoplasmic expression in the superficial epithelium; CC, cytoplasmic expression in the cryptic epithelium. NAA, non-advanced conventional adenoma; AA, advanced conventional adenoma; HP, hyperplastic polyp; SSL, sessile serrated lesion. The means and standard deviations are demarcated in the graphics.
P Values Related to the Expression of Parkin, APC, APE1, and Bcl-xL When Comparing Diagnoses Two by Two.

Abbreviations: APC, Adenomatous Polyposis Coli; APE1, Human Apurinic/Apyrimidinic endonuclease 1; Bcl-Xl, B-cell lymphoma-extra-large; NAA, non-advanced conventional adenoma; AA, advanced conventional adenoma; HP, hyperplastic polyp; SSL, sessile serrated lesion; NS, nuclear expression in the superficial epithelium; NC, nuclear expression in the cryptic epithelium; CS, cytoplasmic expression in the superficial epithelium; CC, cytoplasmic expression in the cryptic epithelium.
Expression of Parkin, APC, and APE1 Concerning the Size, Location, and Aspect of the Polyps.
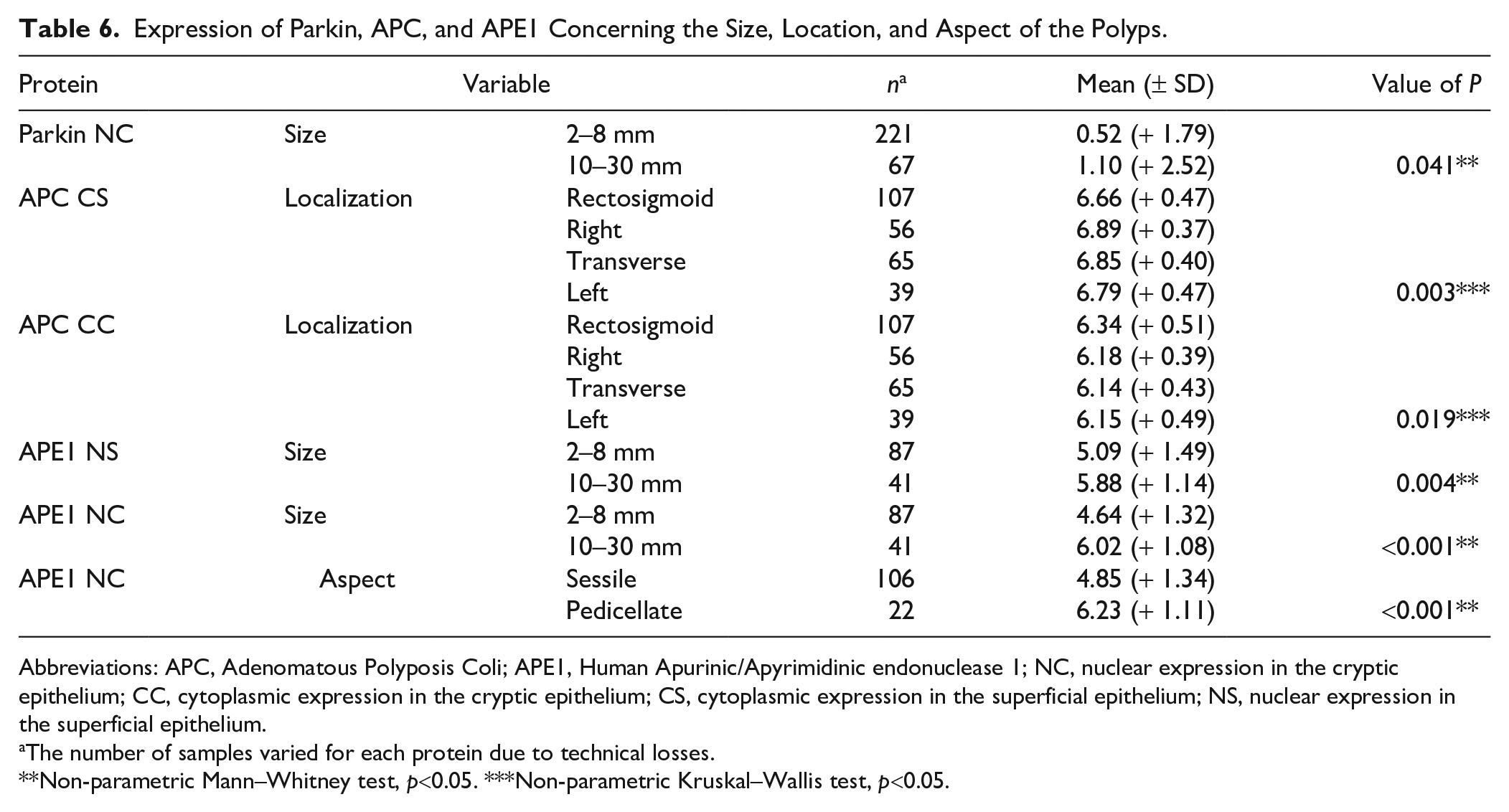
Abbreviations: APC, Adenomatous Polyposis Coli; APE1, Human Apurinic/Apyrimidinic endonuclease 1; NC, nuclear expression in the cryptic epithelium; CC, cytoplasmic expression in the cryptic epithelium; CS, cytoplasmic expression in the superficial epithelium; NS, nuclear expression in the superficial epithelium.
The number of samples varied for each protein due to technical losses.
Non-parametric Mann–Whitney test, p<0.05. ***Non-parametric Kruskal–Wallis test, p<0.05.
Discussion
We demonstrated Parkin expression in the nucleus and cytoplasm with higher expression in the cytoplasm in the present study. In addition to acting as a ubiquitin ligase E3 protein and in mitochondrial homeostasis, Parkin also functions as a transcription factor, regulating the expression of proteins related to various cellular functions. 36 The variety of functions attributed to this protein explains its expression in several cell portions. The APC expression was observed in the cytoplasm, higher in the superficial epithelium, like the distribution found in the normal colon, in CAs. It is observed in the cytoplasm of enterocytes in the upper crypt portions of the intestinal mucosa and is virtually absent at the crypt base. 37 Here, although its expression was higher in the superficial epithelium, this was also observed in the cryptic epithelium. APC protein negatively regulates the transcription factor β-catenin by proteasome degradation 38 and prevents excessive cell proliferation 39 ; it can explain APC higher expression in superficial epithelium where less proliferation is observed. APE1 showed little difference in expression in the nucleus and cytoplasm in polyps’ regions. APE1 is a multifunctional protein that acts as a transcriptional coactivator in apoptosis, proliferation, and differentiation. Its subcellular expression is prominently nuclear, but different conditions change the APE1 subcellular localization, and cytoplasmic localization has been described as associated with different functional processes in highly metabolic or proliferative non-neoplastic cells.40,41 The cytoplasmic APE1 location has also been reported to be associated with aggressiveness in some cancers.41,42 On the contrary, nuclear and cytoplasmic expression levels of APE1 are essential indicators of poor prognosis in solid tumors.40,41 Bcl-xL showed higher expression in the cytoplasm. This protein belongs to the Bcl-2 (B2-cell lymphoma) family and regulates cell death by protecting apoptosis. Its expression is described in the cytoplasm, and Bcl-xL is involved in the electrical and osmotic homeostasis of mitochondria. 43
The nuclear Parkin expression was higher at the SPs than in the CAs. Higher nuclear and cytoplasmic Parkin expressions were observed in both normal tissue and CRC, respectively. 44 In CRC, it was observed that patients with higher Parkin expression in more advanced tumors had a better prognosis. 44 The SPs are associated with less probability of cancer development and, characterized by frequently not having nuclear dysplasia, CAs have some degree of dysplasia and are more associated with the CRC development. 16 The reduction of nuclear Parkin expression in CAs could be correlated with the molecular changes that would alter the PRKN gene expression in the group of the most dysplastic polyps. This can lead to the expression of alternatively spliced isoforms of Parkin, which were not detected by the marker. These altered isoforms can contribute to increased cell proliferation through attenuation of regulation mediated by cyclin E proteolysis. 26
The APC cytoplasmic expression observed in CAs is like the distribution seen in the normal colon. In the anatomopathological analysis of the CAs, the superficial maturation of the epithelium is lost. The superficial epithelium of these lesions is immature and proliferated in appearance, similar to the cryptic area. However, the opposite was observed concerning SPs, which seem to develop an abnormal or different APC expression. In SPs, especially in HPs, the superficial maturation of the epithelium is observed. In SSLs, epithelial maturation changes in its distribution with deviation and lateralization of the proliferative zone, leaving the crypt region different. The architecture of these polyps changes the location of their proliferative area. 10 This change could explain the different distribution of APC, but the proliferative zone remained in the cryptic area evaluated in this work. APC gene mutation is less common in these polyps and, when it occurs, its spectrum is different. 45 In CAs, APC gene mutations are reported in 63% of the cases. 46 APC gene mutations occur early in carcinogenesis and are associated with the classical pathway, influencing the progression and growth of the adenoma. 47
APE1 nuclear expression was significantly higher in the CAs than in the SPs, demonstrating a progressive increase of this protein in the AAs. A progressively higher expression of APE1 is observed in normal mucosa, adenomas, and carcinomas. 41 APE1 is proposed to be a central protein of cellular regulation. Its overexpression accelerates proliferation and growth factors and may stimulate the generation of reactive oxygen species (ROS). Many reports are showing high levels of ROS in various tumor cells. The redox-dependent regulation of Wnt-β-catenin signaling likely contributes to stress-induced cell proliferation. 48 Overexpression of APE1 has been positively correlated with cancer progression in several tumors and is associated with a worse prognosis. 41
The cytoplasmic expression of Bcl-xL is higher in CAs and HPs in SSL polyps. Its higher expression was previously observed in the CAs and the CRC.31,49,50 The higher expression of Bcl-xL in HPs may explain late apoptosis in these lesions, 10 due to its inhibitory action of apoptosis. 43 The increased cytoplasmic expression of Bcl-xL in CAs may correlate with the KRAS mutation, which is more common in CAs than in SPs.17,51,52 Mutated KRAS leads to positive regulation of Bcl-xL and is a crucial mechanism of resistance to apoptosis in CRC.53,54
There were less nuclear Parkin expression and higher cytoplasmic expression of Bcl-xL in the CAs. However, in the SPs, the opposite occurred in this research. These results are consistent with one study where inactivation of the Parkin led to the accumulation of Bcl-xL both in vivo and in vitro. 30
The association of higher Parkin NC expression with larger polyps (10–30 mm) could be related to a possible mechanism of control of cell proliferation through the transcriptional control of genes related to proliferation. The patients with higher Parkin were related to better survival in CRC, 44 and here Parkin could be working in the same way. The higher expression of APE1 in larger polyps agrees with demonstrating that its expression is higher as the lesion progresses in volume to tumor formation. 41 There was also a significant increase in the expression of APE1 NC in pedicellate polyps, possibly correlated to the fact that all pedicled lesions are CAs, and these are more prone to tumor development.10,15 Regarding higher APC CS expression in right and transverse polyps and higher APC CC in the rectosigmoid, the rectosigmoid and right colons are associated with a higher incidence of CRC, and the transverse colon is associated with lower incidence. 55 The higher expression of APC in these regions can collaborate to control tumor proliferation and development.
In conclusion, our Parkin, APC, APE1, and Bcl-xL expression results demonstrated that CAs compared with SPs expressed less Parkin (suppressor tumor) and more APE1 and Bcl-xL (oncogenic proteins). APC was more expressed on the surface of the CAs, and the reverse occurred to the SPs. More markers should be studied in colorectal polyps to improve their histological diagnosis and define their biomarkers profile. The accuracy in characterizing these premalignant lesions can help to prevent their potential evolution to CRC. A summary of the interaction mechanisms between the markers and their different results in CAs and SPs can be seen in Fig. 4.
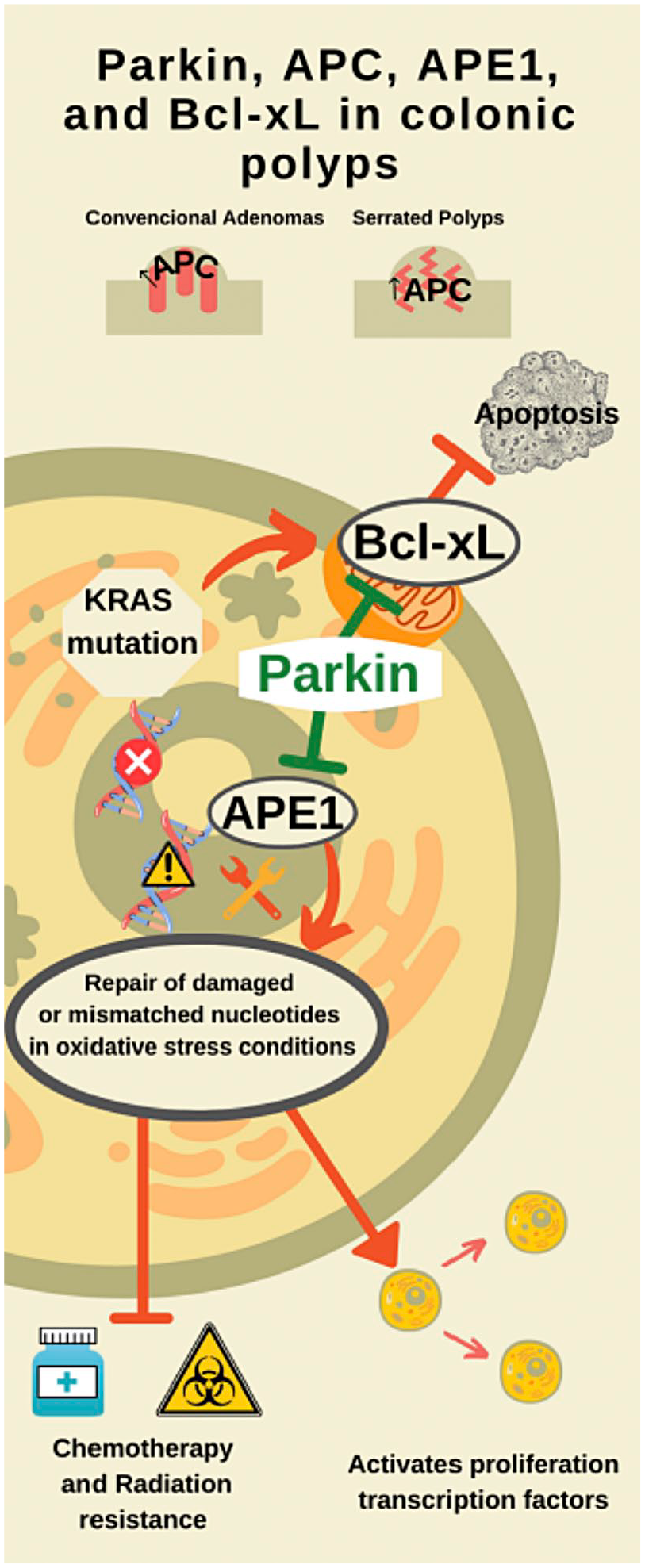
The scheme shows mechanisms of interaction between the markers and their different results in CAs and SPs. The opposite expression of APC. CAs expressed more APC in the cytoplasm of surface cells, similar to normal colonic epithelium. In the crypt region, the SPs scored more APC. The Parkin was less marked in the CA nuclei. Such a fact could explain the greater expression of APE1 and Bcl-xL in these polyps because the Parkin inhibits them. Abbreviations: APC, Adenomatous Polyposis Coli; APE1, Human Apurinic/Apyrimidinic endonuclease 1; Bcl-Xl, B-cell lymphoma-extra-large; CA, conventional adenoma; SP, serrated polyp.
Footnotes
Acknowledgements
The authors thank the institutions (Universities or Faculties) they are affiliated with for encouraging the publication of this manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
RKSB, CAPMR, LN, and APVC conceived, designed, and revised the manuscript. CCVS-C conducted the confection of tissue microarrays. VSS supervised the immunohistochemistry. APMS and MRMN analyzed and revised the cases to include in the work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the institutions the authors are affiliated with (Universities or Faculties).
