Abstract
Our previous studies indicated that YIGSR-A99 peptides chemically conjugated to fibrin hydrogel (FH) and applied to wounded submandibular gland (SMG) in vivo, formed new organized salivary tissue, whereas wounded SMG treated with FH alone or in the absence of a scaffold showed disorganized collagen formation and poor tissue healing. While these studies indicated that damaged SMG grow and differentiate when treated with FH containing L1 peptide, they were performed only in female mice. However, there is a well-established sexual dimorphism present in mouse SMG (e.g., males develop well-differentiated granular convoluted tubules, but these structures are poorly developed in females) and little is known about how these sex differences influence wound healing events. Therefore, the goal of this study was to conduct comparative analyses of regeneration patterns in male and female mice using L1p-FH in a wounded SMG mouse model. Particularly, we focused on sex-dependent wound healing events such as macrophage polarization, vascularization, tissue organization, and collagen deposition, and how these events affect salivary gland functioning:
Introduction
Our previous studies showed that Laminin-111 peptides YIGSR and A99 chemically conjugated to fibrin hydrogels (L1p-FH) and applied to wounded female mouse submandibular glands (SMG) in vivo formed new organized salivary tissue. In contrast, wounded SMG treated with FH alone or left untreated showed disorganized collagen formation and poor tissue healing. Together, these results indicate that L1p-FH promotes SMG regeneration in vivo. 1 While these studies are highly significant as they could offer a viable path forward for advancing the treatment of hyposalivation, they have not yet been performed in male mice.
Mouse SMG development occurs rapidly in males and slower in females. 2 Additionally, the acini occupy a greater volume of the total glandular space in females as compared to males (e.g., with 57% and 30%, respectively). 3 Moreover, male SMG display well-differentiated granular convoluted tubules, but these structures are poorly developed in females. 4 Additionally, the rate of SMG cell proliferation during aging appears to be considerably faster in males as compared to females resulting in a clear sexual dimorphism in early adulthood (e.g., at only 6 weeks of age). 5 Furthermore, there is a differential sexual gene expression of critical mouse saliva components including epidermal growth factor (EGF) and nerve growth factor (NGF). 2 In fact, the sole Nobel Prize in the area of salivary gland research was awarded for seminal work relying on sexual dimorphism, in which the discovery of the existence of growth factors (EGF and NGF) and their role in cell growth and differentiation was greatly facilitated by detection of extremely high levels of these molecules in male SMG as compared to females. 6 While these studies show evidence of a clear sexual dimorphism in mouse SMG, little is known about how sex differences influence the wound healing process in this organ. Therefore, the goal of this study was to conduct comparative analyses of regeneration patterns in both male and female mice using L1p-FH in a wounded SMG mouse model. Particularly, we focused on wound healing events such as macrophage polarization, vascularization, tissue organization, and collagen deposition, and how they affect salivary gland functioning.
Materials and Methods
Materials
Lyophilized fibrinogen from human plasma was purchased from EMD Millipore (Billerica, MA). Spectra/Por 7 dialysis membrane (MWCO = 3.5 kDa) was purchased from Spectrum Laboratories (Rancho Dominguez, CA). A Whatman syringe filter (0.8 μm) was purchased from GE Healthcare Life Sciences (Pittsburgh, PA). A Millex syringe filter (0.22 μm) was purchased from Merck Millipore (Billerica, MA). Insulin-transferrin-sodium selenite media supplement, retinoic acid, hydrocortisone, gentamicin, EGF from murine submaxillary gland, DL-dithiothreitol (DTT), and ε-aminocaproic acid (εACA) were purchased from Sigma-Aldrich (St. Louis, MO). DyLight 680 NHS-ester were purchased from Thermo Fisher Scientific (Newington, NH). Laminin-111 peptides A99 and YIGSR were synthesized by University of Utah DNA/Peptide synthesis core facility. Rabbit anti- iNOS, rabbit anti-Arg-1, TO-PRO-3 iodide, Alexa Fluor 488 conjugated anti-rabbit IgG secondary antibody, Alexa Fluor 568 conjugated anti-rabbit IgG secondary antibody, and Alexa Fluor 568 conjugated anti-mouse IgG secondary antibody were purchased from Invitrogen (Carlsbad, CA). Mouse anti-ICAM and rabbit anti-VCAM were purchased from Abcam (Cambridge, MA). Masson’s trichrome stain kit was purchased from Polysciences, Inc (Washington, PA).
Animals
Male and female C57BL/6J mice at 5–6 weeks of age, and weighing 22–30 g and 17–22 g, respectively, were purchased from the Jackson Laboratory. Mice were then randomly assigned into one the following groups: untreated, treated with L1p-FH, or sham surgery controls. All animal management and procedures were approved by the Institutional Animal Care and Use Committee at the University of Utah.
Hydrogel Preparation
Hydrogels were prepared as previously described. 1 Briefly, L1p-FH was generated by mixing YIGSR-conjugated fibrinogen (1.25 mg/ml), A99-conjugated fibrinogen (1.25 mg/ml), and thrombin (2.5 U/ml) in Tris-buffered saline (TBS) with CaCl2 (2.5 mM) and εACA (2 mg/ml).
Surgical Procedure
Mice were anesthetized with 2% isoflurane with an oxygen flow rate set at 2.0 L/min as previously described. 1 Subsequently, a skin incision of approximately 3–5 mm in length was made along the anterior surface of the neck. Then, SMG were exposed, and surgical wounds were created using biopsy punches of 3.5-mm diameter in males and 3.0-mm in females. Note in Appendix Fig. 1 that males require bigger size punches to achieve the same percentage of glandular loss as compared to females.
Treatment
To each wound, 20 µl of the L1p-FH scaffold was added with forceps placed underneath to prevent leakage as previously described. 1 Then, once the hydrogel attached to the wound, the skin incision was sutured and post-surgical studies were performed at days 1, 3, 8, and 20.
Hydrogel Stability
To monitor scaffold stability in vivo, mice were treated with FH680, which was generated by mixing fibrinogen (2 mg/ml) and DyLight 680 labeled fibrinogen (0.5 mg/ml), and thrombin (2.5 U/ml) in TBS with CaCl2 (2.5 mM) and εACA (2 mg/ml). Fluorescent intensity of the scaffold was monitored at post-surgery days 1, 3, 8, and 20 using a BioRad Chemi-DocTM MP imaging system (Bio-Rad, Hercules, CA).
Confocal Microscopy
SMG were removed and immersed in 10% formalin at room temperature (RT) for 24 hr, dehydrated in ethanol, and embedded in paraffin wax as previously described. 1 Then, SMG were cut into 3 μm sections and mounted on glass slides. Sections within the center of each group were deparaffinized with xylene, and rehydrated with serial ethanol (100%, 95%, 80%, 70%, and 50%) and distilled water. Deparaffinized sections were incubated in sodium citrate buffer (10 mM sodium citrate, pH 6.0 with 0.05% Tween 20) at 95C for 30 min, then cooled down for 20 min at RT, washed with distilled water, and permeabilized with 0.1% Triton X-100 in PBS at RT for 45 min. Sections were blocked in 5% goat serum in PBS with 0.05% Tween 20 (PBST) for 1 hr at RT and incubated overnight at 4C with a primary antibody solution as described in Appendix Table 1. The following day, tissue sections were washed three times with PBST and incubated with the corresponding secondary antibodies for 1 hr at RT. Sections were then washed three times with PBS and counter-stained with TO-PRO-3 iodide at RT for 15 min (1:1000 dilution). Finally, tissue samples were analyzed using a 10× objective confocal Zeiss LSM 700 microscope (Carl Zeiss, Obercochen, Germany).
Macrophage Ratio
Macrophage counts were determined using ImageJ. Specifically, the number of M1 and M2 macrophages were determined by color thresholding and counting positive cells. The color threshold was set to isolate the colocalized signal of nuclei and macrophage positive cells (i.e., overlap of red and blue signal = magenta; overlap of green and blue signal = cyan) and counting was restricted to the regenerated area. Additionally, the regenerated area was measured to normalize cell counts between samples. Statistical significance was assessed in GraphPad Prism by one-way ANOVA (*P < 0.05) and Tukey’s honestly significant differences post-hoc test. Data is representative of n = 3 specimens.
Histological Studies
Deparaffinized sections were stained with Hematoxylin and Eosin (H&E) or Masson’s trichrome and visualized using a 20x objective Leica DMI6000B inverted microscope (Leica Microsystems, Wentzler, Germany). To examine overall wound healing at post-surgery day 20, five equidistant tissue sections from each SMG (21 mm apart) were stained and observed. The images shown are the sections we determined were most representative (middle section) of the healing pattern throughout the entire SMG.
Salivary Flow Rates
Mice were anesthetized with ketamine (100 mg/kg) and xylazine (5 mg/kg), and saliva secretion was stimulated with pilocarpine (50 mg/kg) and isoproterenol (0.5 mg/kg) via intraperitoneal injection as previously described 1 at post-surgery day 20. Then, whole saliva was collected for 5 min and saliva was collected using a 200 μl pipette and transferred to a tube on ice. The volume of saliva was determined by weighing the Eppendorf tube before and after adding the saliva to the tube. Statistical significance was assessed in GraphPad Prism by one-way ANOVA and Tukey’s honestly significant differences post-hoc test. Specifically, one-way ANOVA was used to compare the means of the salivary secretion rates across the untreated, L1p-FH-treated, and sham controls by sex where significant results indicated that at least one pair of groups have different means. Additionally, Tukey’s honestly significant post-hoc test was used to compare all pairs of means and determine which of them were different without inflating familywise type I error rate.
Results
Scaffolds Were Successfully Implanted and Monitored In Vivo
To determine whether sex affected scaffold stability in mouse SMG (e.g., differences in proteases-mediated degradation patterns), we used a dye-conjugated FH scaffold (i.e., FH680) and compared degradation rates between males and females. As shown in Fig. 1, we observed that fluorescence intensity was approximately 50% lower on post-surgery day 8 as compared to post-surgery day 3 in both males and females with the signal completely disappearing at post-surgery day 20. These results indicate that the scaffold was successfully implanted and degraded over time in vivo at a similar rate in both sexes. Since no sex differences were observed, further protease-mediated studies were not performed.
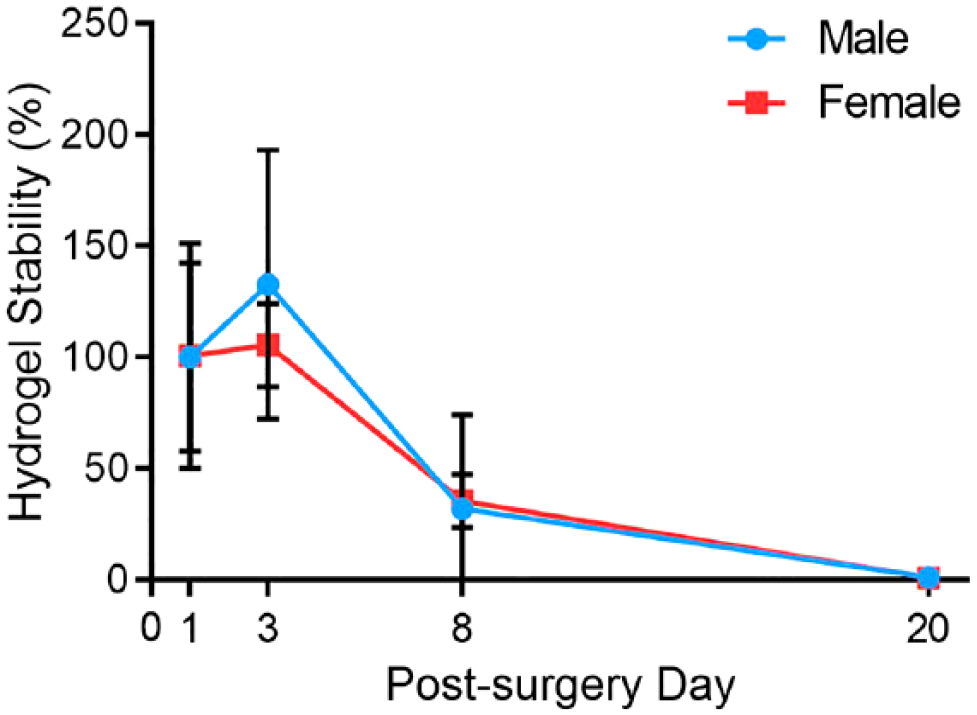
Scaffolds were successfully implanted and displayed similar biostability in vivo in both sexes. Hydrogel stability was assessed in male and female submandibular gland at post-surgery days 1, 3, 8, and 20. Data represent the means ± SD of n=4 specimens per condition.
L1p-FH Promotes Macrophage Polarization in Female Mice Only
Macrophages can be classified as pro-inflammatory (M1) or pro-resolving (M2) subtypes. 7 Specifically, M1 macrophages are observed in early tissue injury responses where they exhibit increased phagocytosis and pro-inflammatory cytokine production, thereby promoting removal of tissue debris. 8 M1 macrophages also promote pathogen-killing by producing increased amounts of reactive oxygen species (ROS) and nitrogen radicals, caused by upregulation of inducible NO synthase (iNOS). 9 In contrast, M2 macrophages appear at a later stage during tissue repair where they produce anti-inflammatory cytokines such as IL-10 to promote the resolution of inflammation. 10 Moreover, M2 macrophages promote wound healing by secreting IL-4 and upregulate arginase-1 (Arg-1) activity, which is the enzyme enrolling the production of polyamines and collagen, thus regenerating the damaged tissue. 11 Since macrophage polarization is necessary for promoting wound healing, we determined the presence of M1 and M2 subtypes within the SMG from untreated and L1p-FH treated groups at post-surgery days 1 and 3 using the specific markers iNOS for M1 macrophages and Arg-1 for M2 macrophages. 12 As shown in Fig. 2, males from both untreated and L1p-FH treated groups expressed iNOS-positive cells with approximately 10–20 macrophages per 100,000 µm2 (Fig. 2A and B, summarized in Fig. 2E). Moreover, males expressed Arg-1-positive cells with approximately 40–50 macrophages per 100,000 µm2 (Fig. 2C and D, summarized in Fig. 2G) with the macrophage numbers being similar at days 1 and 3 (Fig. 2E and G). Regarding females, both untreated and L1p-FH-treated groups expressed iNOS-positive cells with approximately 40–50 macrophages per 100,000 µm2 at post-surgery day 1; however, this number was significantly reduced in the L1p-FH-treated group at post-surgery day 3 (Fig. 2A and B, summarized in Fig. 2F). Moreover, females expressed Arg-1-positive cells with approximately 15–20 macrophages per 100,000 µm2 (Fig. 2C and D, summarized in Fig. 2H) with the macrophage number being similar at days 1 and 3 (Fig. 2F and H). Together, these results indicate that L1p-FH causes a decrease in M1 macrophages in SMG only in female mice, while it does not affect M1 macrophages in males or M2 macrophages in either male or females.
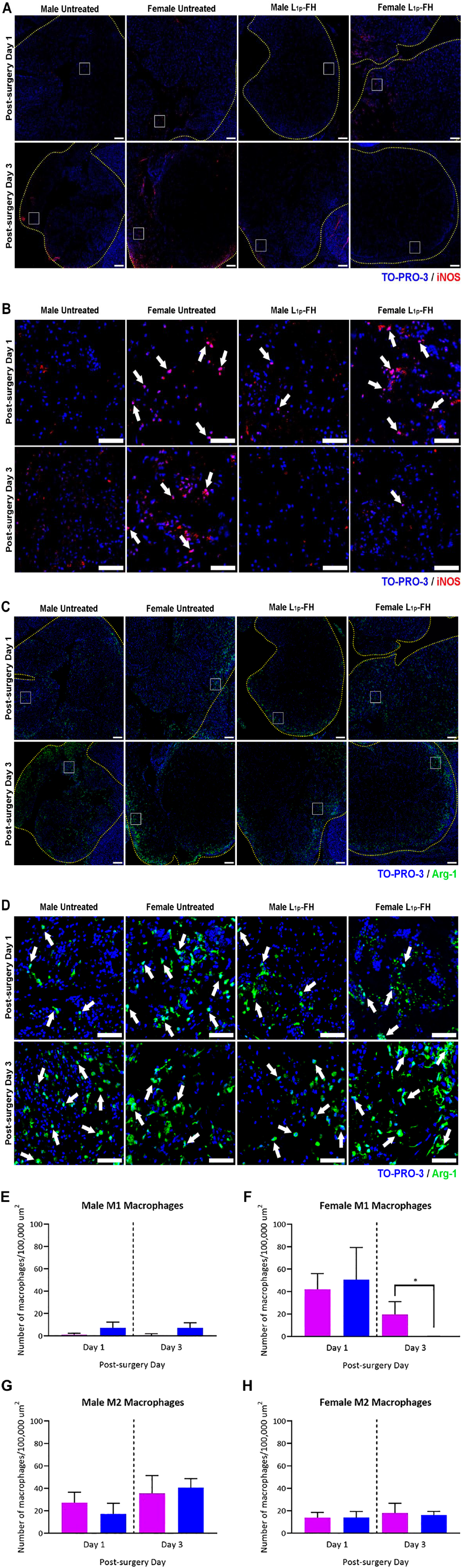
L1p-FH promotes macrophage polarization in female mice only. Immunostaining for macrophage markers (iNOS, red; Arg-1, green) in untreated and L1p-FH groups in males and females at post-surgery day 1 and post-surgery day 3 (A-D). Yellow dotted lines indicate wounded area. Representative of n=3 specimens. Scale bars = 200 µm. Inset white boxes shown to identify areas of interest, with white arrows indicating iNOS+ cells (B) and Arg-1+ cells (D). Scale bars = 50 µm. Specimens were analyzed using a confocal Zeiss LSM 700 microscope at 10× magnifications. The number of iNOS+ and Arg-1+ cells were analyzed in untreated (purple) and L1p-FH (blue) groups using ImageJ and the M1 to M2 ratio was calculated (E-H). Data represent the means ± SD of n=3 mice per condition and statistical significance was assessed by one-way ANOVA (*p<0.05) and Tukey’s honestly significant differences post-hoc test. Abbreviations: FH, fibrin hydrogel; iNOS, inducible NO synthase.
L1p-FH Promotes Blood Vessel Formation in Females Only
During wound healing, M2 macrophages express high amounts of vascular endothelial growth factor (VEGF), thereby promoting cellular proliferation, blood-vessel development and deposition of collagen.13,14 As such, we investigated whether vasculature was affected in male and female SMG during wound healing. As shown in Fig. 3, males in untreated and L1p-FH-treated groups showed areas with VCAM-1 (green) and ICAM1 (red) expression (see white arrows indicating blood vessel formation at post-surgery day 8 (Fig. 3A and B)). Regarding females, we also observed areas with VCAM-1 (green) and ICAM1 (red) expression at post-surgery day 8; however, the staining pattern was discontinuous when compared to males or female sham group. At post-surgery day 20, males staining pattern was similar to day 8 (e.g., continuous green and red fluorescent signals for VCAM-1 and ICAM1 respectively). Regarding females at post-surgery day 20, we observed a continuous staining patterns similar to those observed in males only in the L1p-FH-treated group, while the untreated group showed the same discontinuous staining pattern observed at post-surgery day 8. Together, these results indicate that L1p-FH treatment promotes blood vessel formation in female mice, while L1p-FH treatment has no effect in male mice.
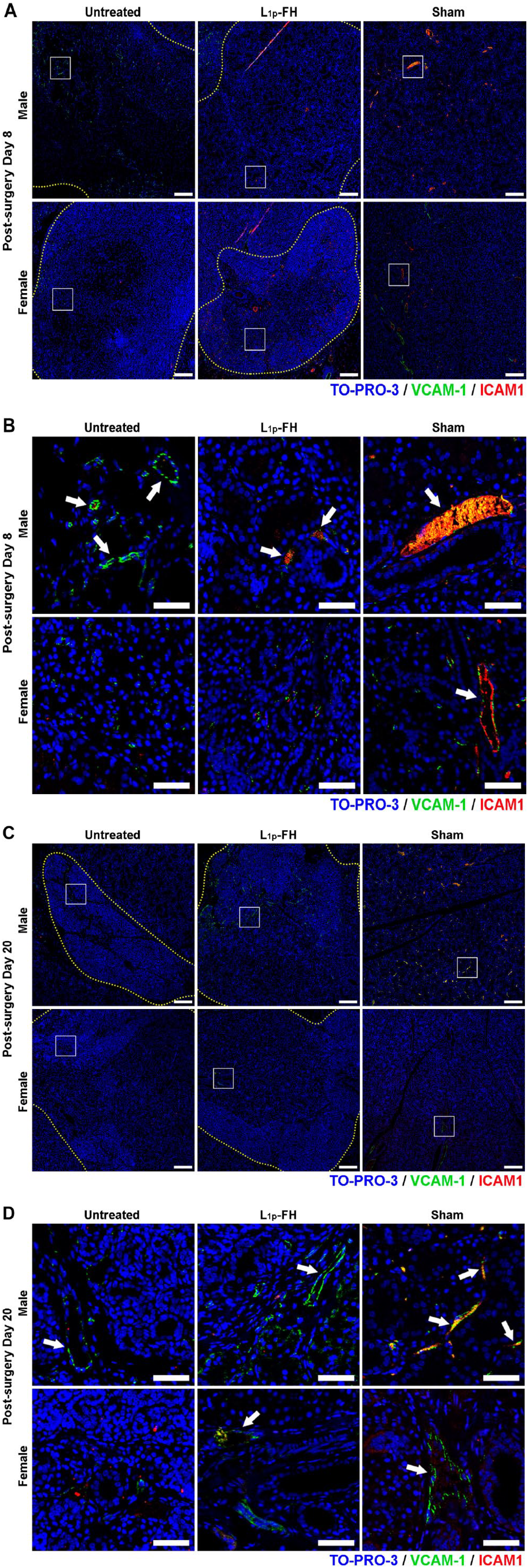
L1p-FH promotes blood vessel formation in females only. Immunostaining for blood vessels markers (VCAM-1, green; ICAM1, red) in untreated and L1p-FH groups in males and females at post-surgery day 8 (A, B) and post-surgery day 20 (C, D). Yellow dotted lines indicate wounded area. Representative of n=3 specimens. Scale bars = 200 µm. Inset white boxes shown to identify areas of interest, with white arrows indicating blood vessels at post-surgery day 8 (B) and post-surgery day 20 (D). Scale bars = 50 µm. Specimens were analyzed using a confocal Zeiss LSM 700 microscope at 10× magnifications. Representative of n=3 specimens. Abbreviation: FH, fibrin hydrogel.
L1p-FH Induces Wound Healing in Female Mice Only
To determine whether sex differences affect SMG wound healing in vivo, we stained tissue sections with H&E at post-surgery day 20. As shown in Fig. 4, males from untreated, L1p-FH-treated and sham control groups displayed intact lobules as well as acinar and ductal structures, indicating a rapid recovery with almost completely regenerated areas (blue dotted lines, Fig. 4A). In contrast, females from the untreated group displayed dense fibrotic tissue throughout the wounded area (blue dotted lines, Fig. 4B). Interestingly, the extent of recovery in females from the L1p-FH-treated group showed some fibrotic areas but selected wounded areas (inset black boxes) revealed the presence of acinar and ductal structures (green and yellow arrows, respectively) with blood vessels (black arrows) and well-organized collagen fibers (inset Masson’s trichrome staining, Fig. 4A and B). These healing patterns were not observed in the female untreated group as there was a lack of acinar and ductal structures in the wounded areas, high fibrotic areas, and incomplete wound healing (inset Masson’s trichrome staining, red arrows, Fig. 4B). Together these results indicate that L1p-FH treatment partially induces wound healing in female mice, whereas L1p-FH treatment does not have an effect in males.
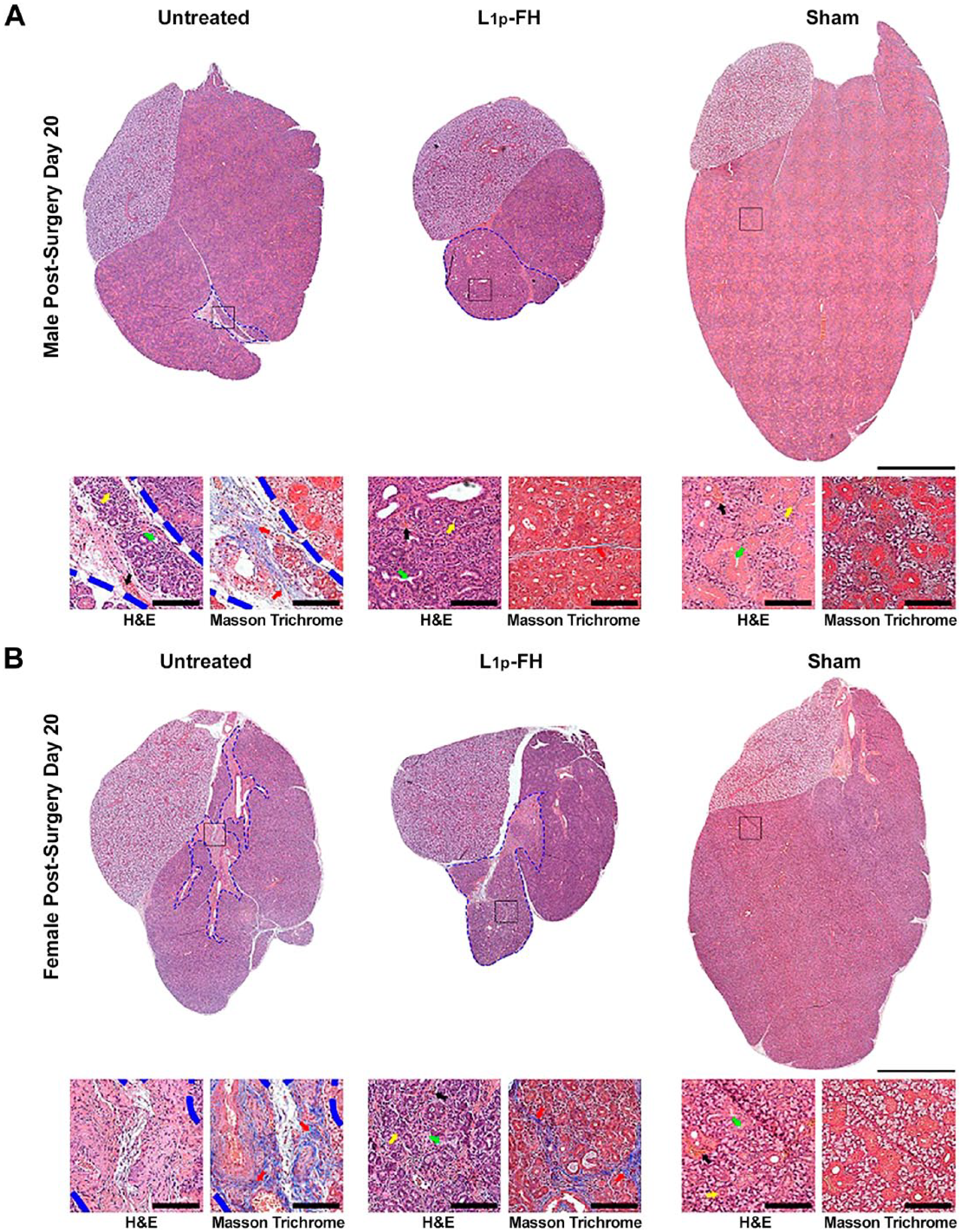
L1p-FH induces wound healing in female mice only. Histology sections of whole SMG in untreated, L1p-FH, and sham groups in males (A) and females (B) at post-surgery day 20. Whole gland images show extent of wounded area (blue dotted lines). Scale bar = 1 mm. Inset black boxes shown to identify structures of interest, specifically acini (yellow arrows, H&E), ducts (green arrows, H&E), blood vessels (black arrows, H&E), and areas of dense collagen deposition (red arrows, Masson trichrome). Specimens were analyzed using a Leica microscope at 20× magnifications. Experiments are representative of n=3 specimens. Scale bars = 200 µm. Abbreviations: FH, fibrin hydrogel; H&E, Hematoxylin and Eosin; SMG, submandibular gland.
L1p-FH Promotes Saliva Secretion in Females Only
As shown in Fig. 5, both male and female mice from untreated groups showed significant reduction in saliva flow rates as compared to sham controls indicating that the SMG wounded model was effective to elicit salivary gland dysfunction. Regarding male mice, the L1p-FH-treated group showed comparable saliva flow rates to the untreated controls, which was significantly lower than saliva flow rates observed in sham controls (Fig. 5A). Regarding female mice, the L1p-FH-treated group showed a significant increase in saliva flow rates as compared to untreated controls; however, it was still significantly lower than saliva secretion levels observed in sham controls (Fig. 5B and Appendix Fig. 2). These results indicate that L1p-FH treatment partially induces restores saliva secretion in female mice, whereas L1p-FH treatment effects were not observed in males.
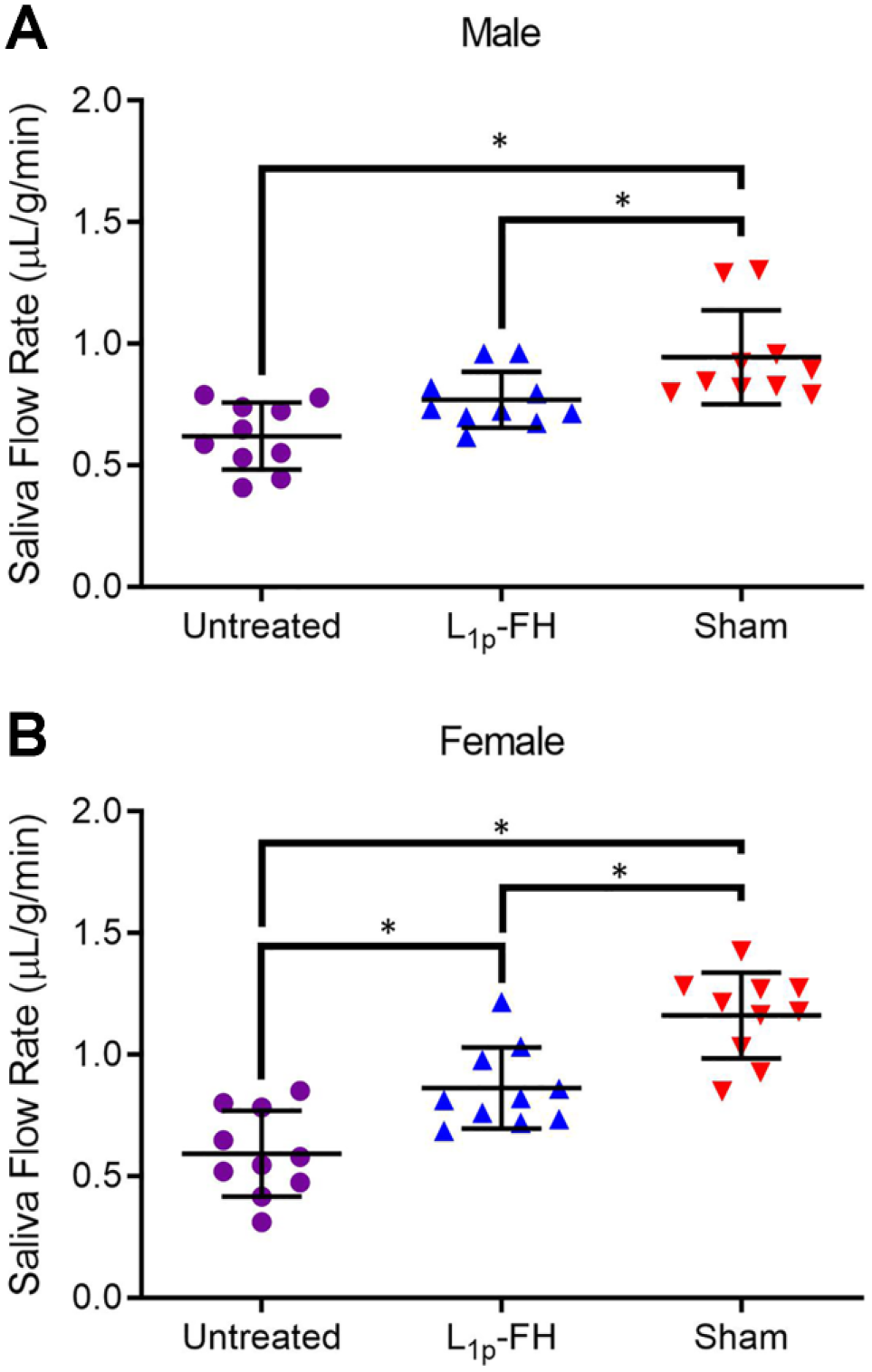
L1p-FH promote saliva secretion in females only. Mice were anesthetized and stimulated with pilocarpine (50 mg/kg) and isoproterenol (0.5 mg/kg) in untreated (purple), L1p-FH (blue), and sham groups (red) at post-surgery day 20. Then, whole saliva was collected for 5 min in male (A) and female (B) mice. Data represent the means ± SD of n=10 mice per condition and statistical significance was assessed by one-way ANOVA (*P < 0.05) and Tukey’s Honestly Significant Differences post-hoc test. Abbreviation: FH, fibrin hydrogel.
Discussion
Our previous work demonstrated that L1p-FH promotes tissue regeneration in a wounded SMG mouse model; however, the experiments were performed in female mice only. In this study, we used male mice to establish sex-dependent differences in healing patterns in mouse SMG when using L1p-FH. Our results showed that in the wounded SMG model, L1p-FH effects were observed only in females. The effects observed in females treated with L1p-FH include a decrease of M1 macrophages, formation of continuous blood vessels as well as acinar and ductal structures and a significant increase in saliva secretion as compared to untreated controls. In contrast, males regardless the treatment showed no changes in macrophage populations, formation of continuous blood vessels as well as acinar, and ductal structures and no changes in saliva secretion. These results indicate sex-attributed differences in wound healing patterns between males and female mouse SMG. These results are consistent with the sexual dimorphism observed in mouse SMG, where a higher number of convoluted tubules rich in EGF and NGF (involved in cell proliferation and neurogenesis, respectively) present in males as compared to females.15,16 Interestingly, EGF and NGF are elevated by treatment with androgens or by ovariectomy,17 –19 indicating their regulation by sex hormones. Moreover, a transcriptional profiling study demonstrated that mouse SMG expressed sexually dimorphic genes with a relatively high expression of several kallikreins genes, 20 a gene family involved in bradykinin production and activation of NGF, EGF, and TGFβ. 21 While SMG sex dimorphism has not been reported in humans or in other mouse salivary glands such as parotid and sublingual, 22 such sex-related differences observed in mice as reported above may help explain why salivary gland diseases tend to affect females at a far higher prevalence than males (i.e., approximately 9:1). 23 Consequently, this raises the intriguing hypothesis that if the male protective and/or healing mechanisms is enacted in the far more vulnerable female population, then functional loss associated with salivary gland diseases may be greatly mitigated if not virtually eliminated.
Regarding pertinent healing mechanisms, M1 macrophages become activated to promote inflammatory responses by releasing pro-inflammatory cytokines and chemokines (e.g., IL-1β, IL-6, IL-12, and iNOS) 24 in early stages of inflammation while switching to an M2 phenotype and secreting anti-inflammatory mediators in late stages (e.g., Arg-1, IL-4, and IL-10). 25 Consequently, the equilibrium between M1 and M2 macrophages is essential for wound healing and tissue homeostasis. 26 Our results indicate that both M1 and M2 macrophages were present during SMG wound healing in both sexes (Fig. 2A to D). While M1 macrophages were significantly higher in females at post-surgery day 1 and significantly decreased in the L1p-FH-treated group at post-surgery day 3 (Fig. 2F), in males we observed steady macrophage levels across the treatment groups, meaning that no effects from L1p-FH were observed. These results are noteworthy, given that macrophages are at the center of the inflammatory response and aid in the vascularization of dysfunctional salivary glands. 27 Previous studies on salivary gland regeneration have focused largely on the role of macrophages on scaffold degradation and have not widely discussed their importance for tissue regeneration.28,29 Specifically, macrophages were long believed to be solely pro-inflammatory and destructive. 30 However, with the new understanding of resolution of inflammation, it is clear that the transition of macrophages from the M1 to M2 is critical for tissue regeneration in vivo.
As alluded to above, macrophages and blood vessels work together in response to inflammation during tissue repair. Specifically, macrophages are drawn to areas of inflammation and blood vessels extend toward the macrophages, thus allowing new blood vessels to sprout. 31 We observed blood vessel formation during wound healing in male untreated and L1p-FH-treated groups at post-surgery day 8, which achieved a high level of organization at post-surgery day 20 (Fig. 3A to D). In females, we observed continuous staining indicative of organized blood vessel formation only in the L1p-FH group while it was absent in the untreated group (Fig. 3E to H). These results are parallel to the macrophage polarization studies and suggest that during SMG wound healing in females, L1p-FH may improve tissue regeneration by causing macrophage polarization as well as by improving blood vessel formation. In fact, when we supplemented L1p-FH with VEGF and FGF9 in females, they achieved blood vessel levels similar to sham controls, 1 suggesting that males have intrinsic factors that may contribute to the blood vessel formation observed in this study. Likewise, previous studies in a mouse model of hind limb ischemia demonstrated that females display decreased blood flow recovery as compared to males, thereby suggesting the possibility of generalized differences in vasculature by sex. 32 Specifically, diminished blood flow recovery in females is accompanied by a higher vascular resistance and is likely attributable to a reduced capacity to remodel and/or to an absence of preexisting collaterals. 32 Furthermore, female vessels have diminished smooth muscle sensitivity to nitric oxide–induced vasodilation and a greater sensitivity to the vasoconstrictor PE as compared with males. 33 Also, endogenous androgens are involved in the coordination and enhancement of neovascularization in the context of ischemic injury in males but not females. 34 In light of the sex differences noted above, the need to further investigate vascularization processes and related macrophage mechanisms in order to better understand the impact of dimorphism on tissue regeneration, both in salivary glands and throughout the body, is indicated.
After inflammation and vascularization, the next step in wound healing is collagen deposition as well as granulation tissue formation. 35 These processes are important for salivary gland epithelialization as they provide a frame for cells to proliferate and differentiate. 36 In our studies, we observed that untreated and L1p-FH-treated males showed an organized pattern in both collagen deposition and granulation tissues at post-surgery day 20 (Fig. 4A). In contrast, untreated and L1p-FH-treated female mice had areas of disorganized collagen as well as abundant granulation tissue with these defects more pronounced in the untreated group (Fig. 4B). However, females treated with L1p-FH had more organized collagen deposition similar to that of sham controls (Fig. 4B). These results are consistent with our previous studies showing similar effects in females. 1
Saliva production from functional glands is essential for food chewing as well as maintaining oral health. Our results show that saliva flow rates recovered to near normal values in untreated and L1p-FH male groups after 20 days post-surgery (Fig. 5A). Interestingly, only females treated with L1p-FH showed a significant recovery in saliva secretion as compared to untreated controls (Fig. 5B). A limitation of this study is that total saliva measurements were used while the surgical punch and treatments were only performed in SMG. This may lead to underestimation of the degree of damages in SMG as well as the effectiveness of L1p-FH treatment. However, SMG is the major contributor of stimulated saliva secretion in mice, and there were differences observed in the untreated versus L1p-FH treated female mice.
In summary, the above studies demonstrate sex-dependent wound healing events (e.g., macrophage polarization, vascularization, collagen deposition and tissue organization) and indicate how they affect salivary gland functioning. Future studies will expand this model to other mouse salivary glands, as well as a head and neck irradiated mouse model, thereby providing further insights into sex-dependent regeneration patterns within SMG and in the salivary glands as a whole.
Supplemental Material
2020-00019R1_Production_Supplemental_Data_online_supp_rev.pdf – Supplemental material for Sex-dependent Regeneration Patterns in Mouse Submandibular Glands
Supplemental material, 2020-00019R1_Production_Supplemental_Data_online_supp_rev.pdf for Sex-dependent Regeneration Patterns in Mouse Submandibular Glands by Callie T. Brown, Kihoon Nam, Yue Zhang, Yuqing Qiu, Spencer M. Dean, Harim T. Dos Santos, Pedro Lei, Stelios T. Andreadis and Olga J. Baker in Journal of Histochemistry & Cytochemistry
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors have contributed to this manuscript as follows: planning (CB, KN, PL, SA, OB), acquisition of samples (CB, SD, and KN), execution of experiments (CB, SD, KN, and HS), analysis of data (CB, KN, HS, YZ, YQ, and OB), writing of manuscript (CB, KN, and OB), and all authors have read and approved the manuscript as submitted.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health (DE02169707A1 and DE02297107S1 to OB and DE02297107 to OB and SA).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
