Abstract
The aim of this study was to investigate carnitine action against negative effects of etoposide on stem/progenitor spermatogonia and on sperm production. Carnitine (250 mg/kg body weight/day) and etoposide (5 mg/kg body weight/day) were administered from 25-days postpartum to 32-days postpartum. Testes were collected at 32-days postpartum, 64-days postpartum, and 127-days postpartum, and submitted to the immuno-labeling of UTF1, SOX2, and PLZF proteins to identify undifferentiated spermatogonia populations. At 127-days postpartum, sperm were collected for analysis. Carnitine+etoposide group showed a higher numerical density of spermatogonia labeled for all studied proteins at 64-days postpartum (critical age) compared to the etoposide group. Moreover, there was an improvement of spermatic parameters and sperm DNA integrity in rats of the carnitine+etoposide group in comparison with rats of the etoposide group. The results suggest that carnitine improves the self-renewal of undifferentiated spermatogonia and promotes a partial protection on them, alleviating the etoposide harmful late effects and leading to an enhancement of the sperm parameters in adulthood:
Keywords
Introduction
Etoposide is a potent chemotherapeutic agent used in different types of cancer.1,2 Despite its efficiency against the cancer cells, previous studies of our group showed that etoposide administration to prepubertal rats leads to a reduction of testicular morphometric parameters3–5 and sperm concentration, and to germ cell apoptosis.4,5 The most known mechanism of action of etoposide is through its interaction with the enzyme topoisomerase II. In the testis, high levels of this enzyme are present in intermediary and type B spermatogonia. 1 However, the observation of reproductive damages in adult rats exposed to etoposide during prepuberty suggests that topoisomerase II inhibition is not the only mechanism of action by which etoposide causes damages to the testes. 4
The stem/progenitor spermatogonia are responsible for the constant repopulation of the seminiferous epithelium and consequently, for maintaining sperm production. Damages of these cells can impair spermatogenesis, leading to germ cell death, production of defective sperm, or even to a reduction of sperm number.6,7 Although the existence of the stem/progenitor spermatogonia has been known for a long time, their exact identification has been widely debated, basically because they are a highly dynamic cell population. The subtypes of undifferentiated spermatogonia, for example, are determined by the asymmetric protein distribution in their cytoplasm during their division, leading to the production of spermatogonia committed to differentiation or to the maintenance of the stem spermatogonia pool.8,9 Thus, it is fundamental to identify them not only by morphological but also by molecular criteria.
The stem cells are usually considered the most resistant type of spermatogonia, probably due to the low proliferation rate when compared to other spermatogonia. 10 Even so, they are sensitive to toxicants such as mono-(2-ethylhexyl)-phthalate (MEHP), WR-2721, etoposide6,11,12 and other xenobiotic agents. 13 Because stem/progenitor spermatogonia are responsible for maintaining spermatogenesis, any damage to these cells can result in reduction of fertility or in increase of the production of abnormal sperm. Thus, tools to protect stem/progenitor spermatogonia from the damages caused by chemotherapy can reduce the reproductive side effects, improving the sperm parameters, including the integrity of sperm DNA.
Carnitine is a quaternary amine present in the male genital system and contributes to sperm maturation in the epididymis. 14 High concentration of carnitine has been found in Sertoli cells, sperm, seminal vesicle, and epididymis. 15 Carnitine has been known to enhance the energy generation in mitochondria and to reduce the availability of free unsaturated fatty acids for lipid peroxidation. It may also reduce the reactive oxidative species (ROS) in infertile patients promoting improvement in sperm vitality, sperm motility, and increased pregnancy rate. 16 Carnitine has shown to reduce the apoptosis in germ cells and to improve integrity of sperm DNA, acrosome integrity, and the reproductive competence in adult rats, which were doxorubicin-treated at prepuberty,17,18 a chemotherapeutic drug whose mechanism of action is similar to that of etoposide. Besides, exogenous carnitine can act as a cell protector due to its anti-inflammatory and antioxidant properties,19–21 maintaining DNA integrity, 17 and promoting DNA repair when needed. 22
Peroxidative damage is considered the most significant cause of testicular functional impairment, as consequence of a variety of conditions including chemotherapeutic drugs. 23 One of the early effects of the chemotherapeutics in the testis is the induction of oxidative stress, which leads to the upregulation of pro-apoptotic proteins and cell death. Considering the increasing incidence of cancer observed in the last few years, chemotherapy can be pointed as a relevant element regarding male fertility aspects, 24 especially because the incidence of cure is also rising, 25 making the fertility a future concern in the quality of life of the patient.
In a previous study, our group showed that carnitine administration in prepubertal rats, prior to the etoposide treatment, reduced, at adult age, the rate of apoptotic germ cells and the concentration of abnormal forms of spermatozoa. 4 Thus, we hypothesized that etoposide might have also acted on the stem/progenitor spermatogonia and carnitine can probably have conferred a substantial level of protection to these cells. For this purpose, the labeling of UTF1, SOX2, and PLZF proteins was performed. These markers were used because they have been referred as typical indicatives of stemness in different cell types including primordial germ cells, 26 embryonic stem cells, 27 and spermatogonia themselves28–32 in rats and many other species.
In the current study, we studied the short-, mid-, and long-term effects of carnitine and etoposide administration during prepuberty on the spermatogonial lineages, as well as the results of these treatments on sperm quantitative parameters and on the sperm DNA integrity.
Materials and Methods
Animals and Groups
Twenty-five-day-old male Wistar rats (Rattus norvegicus albinus, n=100), weighing approximately 60 g, were distributed into four groups (25 animals per group) according to the treatment applied: sham-control (S), treated with 0.9% saline solution; carnitine (C), treated with 2 g/kg of
Treatment began at 25 days of age. The total doses of carnitine and etoposide were distributed into 8 consecutive days, so that the daily doses were 250 mg/kg and 5 mg/kg, respectively. The doses were established according to the literature33–36 and to previous data obtained by our group.3–5,37,38 This etoposide dose was chosen by the harmful on germ cells, however, maintaining the quality of life of animals.
For the CE group, carnitine was administered 1 hr before etoposide. Importantly, carnitine did not interfere with the antitumor activity of doxorubicin in patients. 33
The etoposide is commercialized as flasks containing a volume of 5 ml; in each milliliter of this medicine there are, as vehicle of etoposide, 20 mg of etoposide, as well as 650 mg of macrogol 300, 2 mg of citric acid, 80 mg of polysorbate 80, and 1 ml of ethanol. However, the vehicle group was not included because in a previous study it did not reveal to cause abnormalities on spermatogenesis and to alter the health of rats. 5
The S group received 0.9% saline solution in the same schedule and volume applied to E, CE, and C groups. The treatments were performed via intraperitoneal route. The chemicals handling, and dilution followed the supplier recommendations.
The animals were housed under controlled conditions (12/12 hr light/dark cycles at 23C to 25C) and water and food were provided ad libitum until the euthanasia day.
Each group (S, C, E, and CE) was divided into 3 subgroups, according to the day of euthanasia, which were as follows: 32 (prepuberty, 12 hr after the end of the treatment), 64 (late puberty, 32 days after the end of the treatment), and 127 (adulthood, 95 days after the end of the treatment). The subgroups were as follows: S32 (n=5), S64 (n=5), S127 (n=15), C32 (n=5), C64 (n=5), C127 (n=15), E32 (n=5), E64 (n=5), E127 (n=15), CE32 (n=5), CE64 (n=5), and CE127 (n=15).
The experiments were carried out under the Guide for the Care and Use of Laboratory of National Institutes of Health and performed according to the rules of the Ethical Committee for Animal Research of the Federal University of Sao Paulo (Protocol number 1610/10).
Testicular Volume and Total Volume of the Seminiferous Epithelium
Just after the collection, the testes had their volume (V) measured by the Scherle’s method. 39 The testes were then fixed in Bouin’s liquid, processed and embedded in paraffin. The testicular sections were used to obtain the total volume of the seminiferous epithelium (Vt). The volume density (Vv) of a component of an organ or tissue can be calculated through a system of point count, using an integrating eyepiece; here, a 25-point integrating eyepiece attached to a light microscope was used under 100x magnification. 40 The volume density of the seminiferous epithelium (Vvt) was systematically and randomly sampled in 30 fields of testicular sections, per each testis (right and left respectively), totaling 60 fields per animal; thus, to obtain the final Vvt in each subgroup 750 test-points were counted by testis; thus, a total of 1500 test-points were analyzed per animal. The planimetric fraction of a section occupied by sections of a given component corresponds to the fraction of the tissue volume occupied by this component. 41 Thus, there is a similar interpretation between volume density (Vv) and the density of area of the profiles (AA) regarding the space.41–43 Consequently, in the current study, Vvt represents the proportion of seminiferous epithelium in the whole testis. 44 So, since we got the total testicular volume (V) and the volume density of the seminiferous epithelium (Vvt), the total volume of the seminiferous epithelium (Vt) was obtained using the formula: Vt = Vvt (%) x V/100.
Immunohistochemistry
With the aim to quantify the undifferentiated spermatogonia, the left testis was collected and fixed in Bouin’s liquid for 24 hr (for immunolabeling of the UTF1 and SOX2 proteins). Immediately after the collection of the left testis, the right testis was perfusion-fixed with 4% formaldehyde (n=5 per subgroup), postfixed by immersion in 4% formaldehyde for 24 hr, processed and embedded in paraffin (for immunolabeling of the PLZF protein). From each paraffinized testicular fragment, two 6-µm-thick non-consecutive cross-sections per rat (10 cross-sections of same thickness apart from each) were obtained and submitted to the immunolabeling of the following proteins: UTF1 (polyclonal/rabbit/AB3383; Millipore, Billerica, MA; 1:150), SOX2 (monoclonal/mouse/ab75485; Abcam, Cambridge, MA; 1:200), and PLZF (monoclonal/mouse/sc28319; Santa Cruz, Santa Cruz, CA; 1:200). Antigen retrieval was dependent on the target protein: for PLZF, heat treatment (95C) in citrate buffer (1 mM citric acid, pH 6.0) for 40 min in water bath was used, whereas for UTF1, microwave treatment in citrate buffer for 10 min was used. No antigen retrieval was used for SOX2. The sections were incubated in 3% hydrogen peroxide for 15 min followed by 5% bovine serum albumin (BSA, A4503; Sigma Aldrich) incubation. The antibodies were incubated overnight at 4C. The sections were washed in phosphate buffered saline (PBS, 0.05M, pH 7.4) and incubated with the secondary biotinylated antibody (KP500; Diagnostic Bio Systems, Pleasanton, CA) and then with streptavidin (KP500; Diagnostic Bio Systems), both for 15 min. The reaction was revealed with 3,3’-diaminobenzidine (DAB-125; Spring, Pleasanton, CA) and the nuclei were stained by Harris’ hematoxylin. The sections were analyzed using the Leica Qwin-V3 image analysis system (Leica-Cambridge®, UK) coupled to a light microscope (BX50; Olympus, Sao Paulo, SP, Brazil), under 20x and 40x objective lenses. The images were captured using a digital camera connected to the light microscope.
Control incubations were performed in the absence of primary antibodies and the presence of the IgG antibody (ab46540; Abcam; 1:150 and 1:200, the same dilution of UTF1 and SOX2/PLZF antibodies, respectively) to determine the effect of serum on non-specific background staining.
The numerical densities (Nv) of UTF1+, SOX2+, and PLZF+ spermatogonia were obtained by the ratio between the number of positive spermatogonia counted for a specific protein and the volume of seminiferous epithelium analyzed (Fig. 1B), as already it has been done with other proteins in previous study. 5 The seminiferous tubule sections could not be staged due to asynchronized cell associations and loss of germ cells caused by etoposide (Figs. 2E, F, 3E, F, H, 4E, F and H).
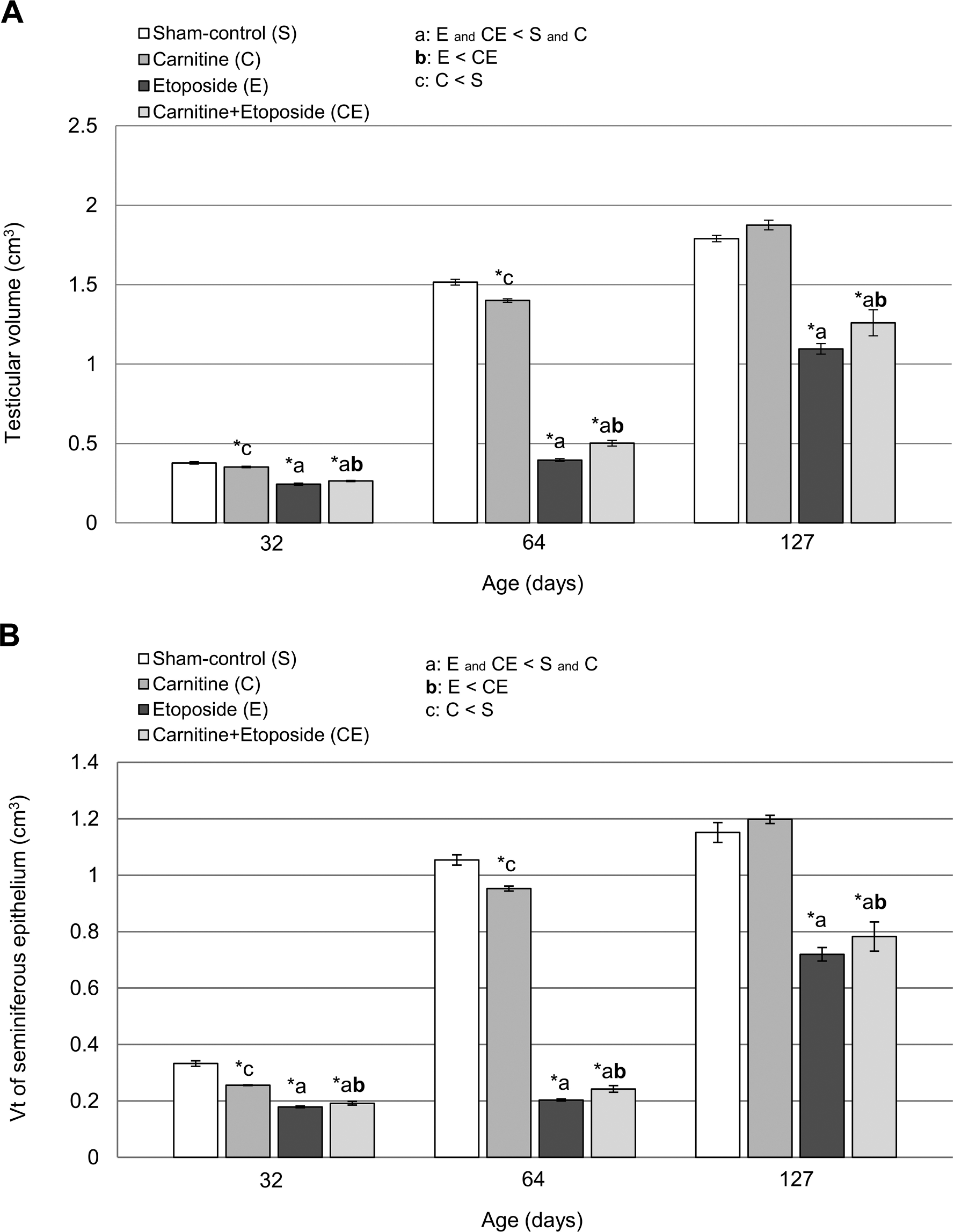
Testicular volume (A) and total volume (Vt) of the seminiferous epithelium (B) in 32-, 64-, and 127-day-old rats of sham-control (S), carnitine (C), etoposide (E), and carnitine+etoposide (CE) groups. Mean ± standard error of mean; Analysis of Variance test (n=5). *p≤0.05
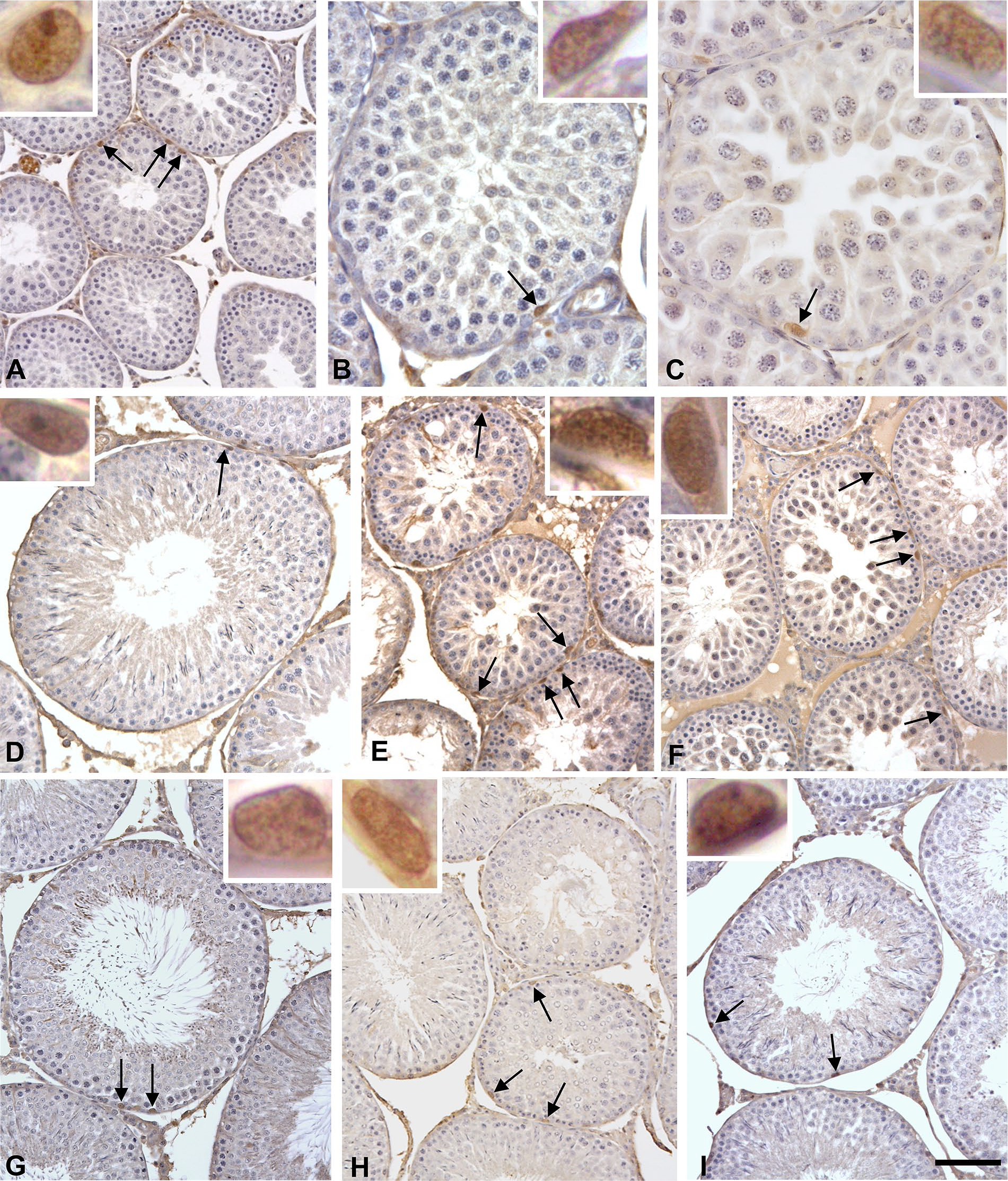
Photomicrographs of testicular cross-sections of 32dpp (A to C), 64dpp (D to F), and 127dpp (G to I) rats submitted to UTF1 immunolabeling. Nuclei of type A spermatogonia (arrows) are labeled in control (A, D, and G), etoposide (B, E, and H), and carnitine+etoposide (C, F, and I) rats. At 32dpp, more than one labeled nucleus (arrows) per tubular section is observed in control rats whereas in etoposide and carnitine+etoposide rats, only one labeled nucleus is observed per tubular section. On the other hand, in 64dpp, etoposide and carnitine+etoposide rats show more than one labeled nucleus (arrows) per tubular section whereas the control rats show only one labeled nucleus per tubular section. Insets: details of type A spermatogonia nuclear labeling. Scale bars: (A) 93 µm, inset: 2 µm; (B) 35 µm, inset: 2 µm; (C) 23 µm, inset: 4µm; (D) 93 µm, inset: 2 µm; (E) 70 µm, inset: 2 µm; (F) 70 µm, inset: 2 µm; (G) 93 µm, inset: 2 µm; (H) 70 µm, inset: 3 µm; and (I) 70 µm, inset: 3 µm.
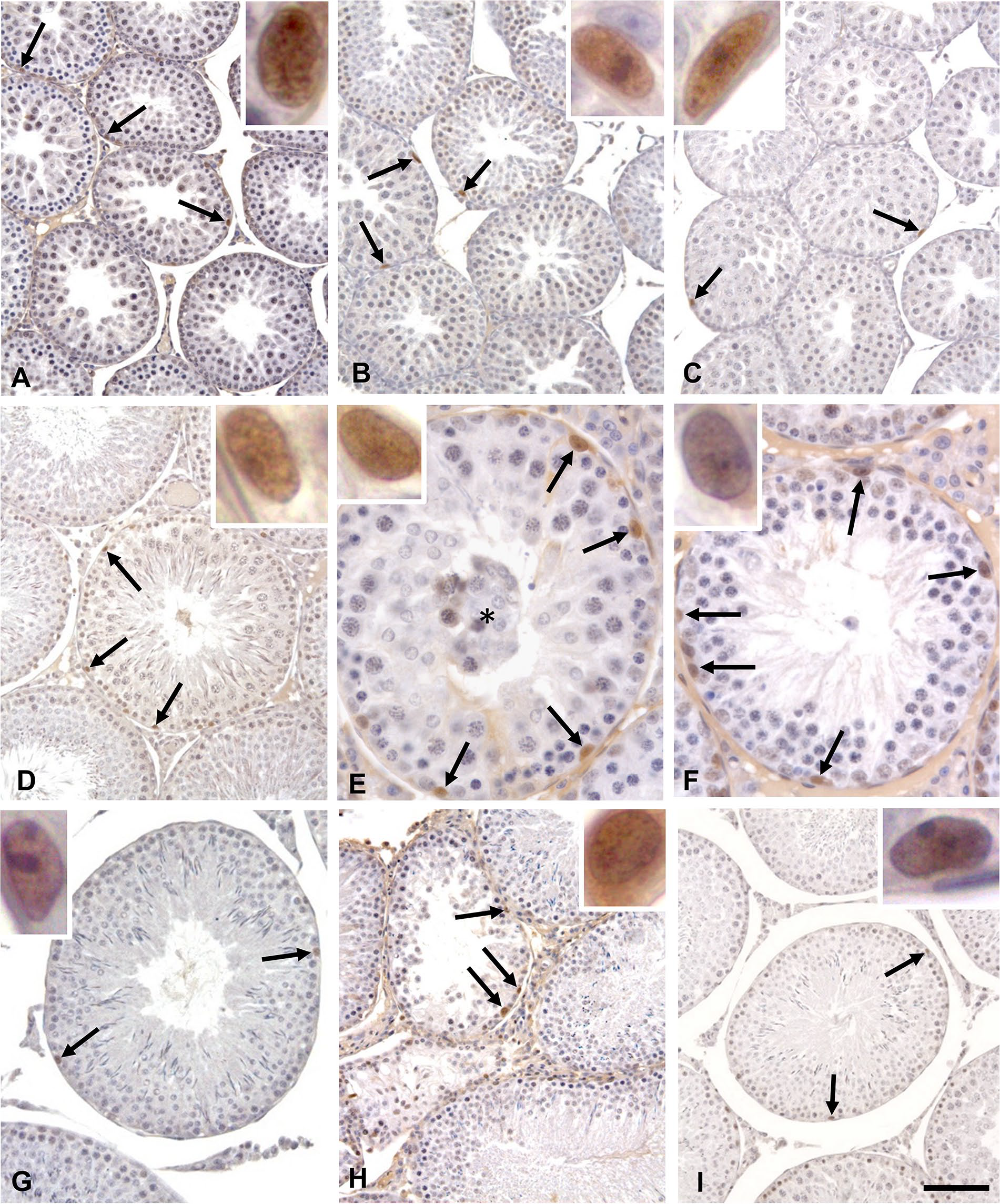
Photomicrographs of testicular cross-sections of 32dpp (A to C), 64dpp (D to F), and 127dpp (G to I) rats submitted to SOX2 immunolabeling. Nuclei of type A spermatogonia (arrows) are labeled in control (A, D, and G), etoposide (B, E, and H), and carnitine+etoposide (C, F, and I) rats. Asterisk: Detached germ cells. Insets: details of type A spermatogonia nuclear labeling. Scale bars: (A) 70 µm, inset: 5 µm; (B) 70 µm, inset: 3µm; (C) 70 µm, inset: 2µm; (D) 70µm, inset: 3 µm; (E) 28 µm, inset: 2 µm; (F) 35 µm, inset: 3 µm; (G) 70 µm, inset: 2 µm; (H) 93 µm, inset: 2 µm; and (I) 93 µm, inset: 2 µm.
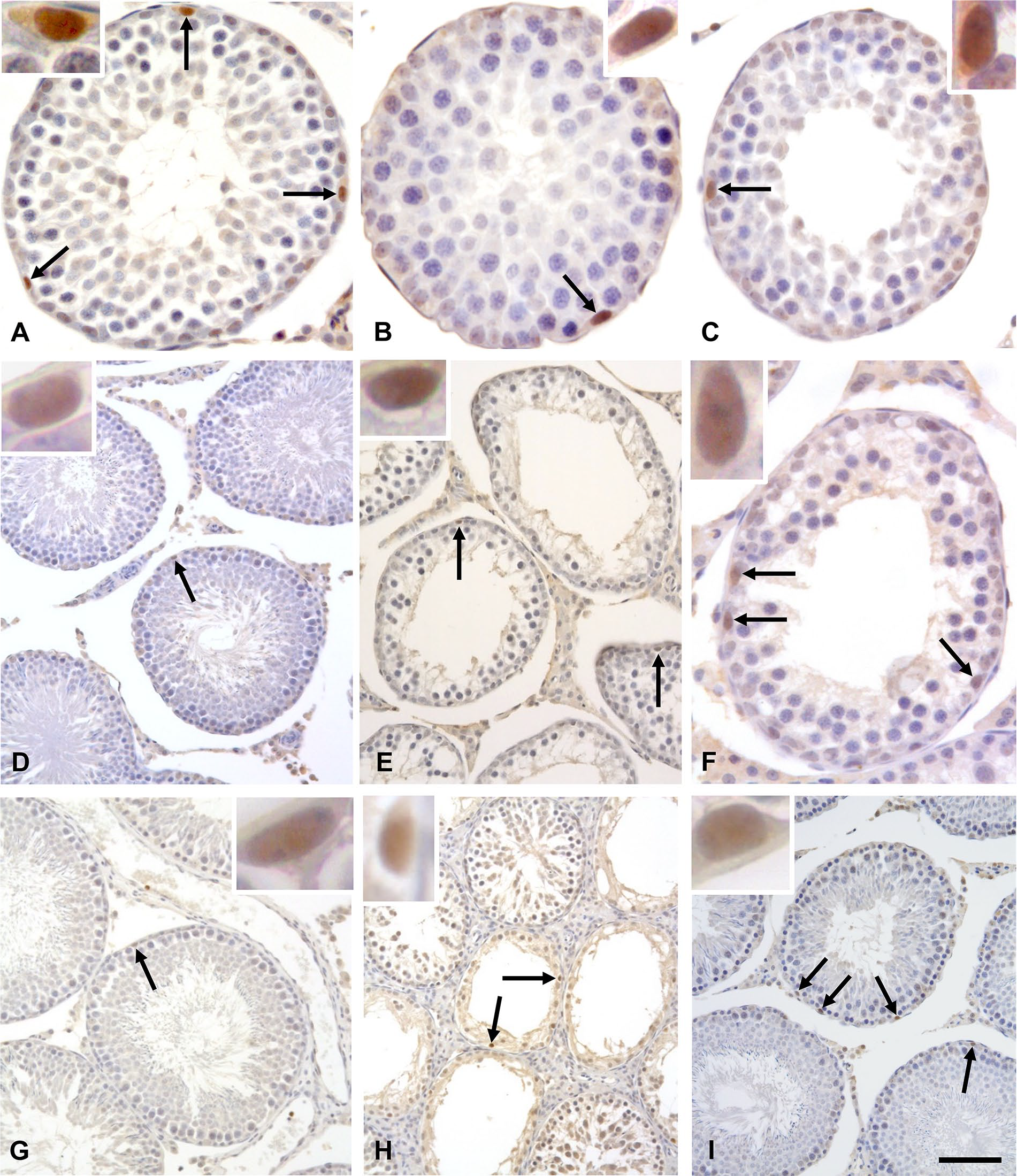
Photomicrographs of testicular cross-sections of 32dpp (A to C), 64dpp (D to F), and 127dpp (G to I) rats submitted to PLZF immunolabeling. Nuclei of type A spermatogonia (arrows) are labeled in control (A, D, and G), etoposide (B, E, and H), and carnitine+etoposide (C, F, and I) rats. At 32dpp, more than one labeled nucleus (arrows) per tubular section is observed in control rats whereas in etoposide and carnitine+etoposide rats, only one labeled nucleus is observed per tubular section. On the other hand, in 64dpp and in 127dpp, etoposide and carnitine+etoposide rats show more than one labeled nucleus (arrows) per tubular section, whereas the control rats show only one labeled nucleus per tubular section. Insets: details of type A spermatogonia nuclear labeling. Scale bars: (A) 35µm, inset: 2 µm; (B) 23 µm, inset: 2 µm; (C) 35 µm, inset: 2 µm; (D) 70 µm, inset: 3 µm; (E) 70 µm, inset: 3µm; (F) 35 µm, inset: 3 µm; (G) 70 µm, inset: 3 µm; (H) 93 µm, inset: 2 µm, and (I) 70 µm, inset: 3µm and negative control, 93 µm.
The number of UTF1+, SOX2+, and PLZF+ spermatogonia per seminiferous tubule section was also obtained by the ratio between the total number of positive spermatogonia for each specific protein investigated and the total number of seminiferous tubule sections analyzed.
Step 19 Spermatid Number, Epididymal Sperm Count, Sperm Daily Production and Spermatic Transit Time
The right testes and epididymides from the S127, C127, E127, and CE127 subgroups (10 rats per subgroup) were collected. Each epididymis was divided in two portions: caput+corpus and cauda. Both epididymal segments were weighed and homogenized according with the methodology previously described.43,45,46 Subsequently, the samples were counted using a hemocytometer chamber. The absolute and relative numbers of homogenization-resistant spermatids (respectively step19 spermatid number x106 per testis and per gram of testis) were then determined. The daily sperm production (DSP) was also calculated in order to determine the efficiency of the process. For this goal, the number of step 19 spermatids was divided by 6.1 which is the number of days of the seminiferous cycle in which these spermatids are present in the rat seminiferous epithelium. The number of sperms was also determined in the epididymal segments, caput+corpus and cauda, both the absolute (106/epididymal segment) and relative (106/g of epididymal segment) numbers. The absolute results were used to obtain the sperm transit time through each epididymis segment (in number of days) by dividing the sperm number per epididymal segment/DSP. 46
Analysis of Sperm DNA Fragmentation by Alkaline Comet Assay
Sperms of the epididymis cauda (left) were collected from rats of the S127, C127, E127, and CE127 subgroups (10 rats per subgroup and the same animals used to evaluate sperm parameters).
Sperm DNA fragmentation was evaluated using the alkaline comet assay.18,47,48 The epididymis cauda was incised and placed in Petri dish with PBS, for 10 min, to allow the dispersion of the sperms. The solution was filtered in a 100 µm nylon cell strainer and centrifuged at 700 g for 10 min and washed in hypotonic solution (0.45% saline) for blood cell cytolysis. Aliquots of 200 µL were stored at -80C.
Frozen sperm samples were thawed at 37C for 2 min in a water bath and diluted in 0.5% low melting point agarose (LGL Laboratories, Sao Paulo, SP, Brazil) at 37C to a concentration of 1 x 105 cells/ml. The solution of agarose and sperm was placed on slide precovered with 1.5% normal melting point agarose (LGL Laboratories). In all subsequent steps, the slides were protected from light to prevent artificial DNA damage. The slides were cooled at 4C for 10 min and covered with 1 ml of lysis buffer (2.5M NaCl, 100 mM EDTA and 10 mM Tris-HCl; pH 10) containing: 10% dimethylsulfoxide (D8418; Sigma Aldrich), 1% Triton X-100 (T9284; Sigma Aldrich) and 40 mM dithiothreitol (DTT; Bio Basic Inc, Ontorio, Canada) at 4C for 1 hr. The slides were washed in ice-cold distilled water and covered with 1 ml of lysis buffer with proteinase K (0.1 mg/ml) at 37C for 2.5 hr. Then, they were washed in ice-cold distilled water and immersed in alkaline solution (1 mM EDTA and 0.05M NaOH, pH 12.1) for 45 min. Then, they were washed twice in Tris-Borate-EDTA buffer (TBE, 89 mM Tris, 89 mM boric acid, and 0.025 mM EDTA, pH 8) for 5 min and immersed in ice-cold TBE in a horizontal electrophoresis chamber at 30 V (1.3 v/cm) for 20 min. The slides were fixed in 70% ethanol for 5 min and stained with ethidium bromide (20 µg/ml). One hundred spermatozoa were analyzed per rat. Percent of tail DNA, tail length and tail extent moment (tail length/fraction of tail DNA) were measured using the KOMET 6.0.1 Software (Andor Technology, Ulster, UK) and analyzed with image system (Kinetic Imaging, Ltda., Sao Paulo, Brazil) coupled to an epifluorescence microscope (Olympus BX51), under 40x objective lens.
Statistical Analysis
Statistical analyses were performed using the Sigma Plot 12.0 program. The data were submitted to the One-Way Analysis of Variance (ANOVA) test. When the values did not show normal distribution and variance similarity, the non-parametric Variance Analysis of Kruskal-Wallis test was used. When differences were statistically significant (p≤0.05), the data were submitted to the Student-Newman-Keuls (SNK) test.
Results
Carnitine Administration Previously to Etoposide Reduced the Loss of Testicular and Seminiferous Epithelium Volumes
The testicular volume (V) and the total volume of seminiferous epithelium (Vt) of E and CE groups were significantly lower in comparison with those from the Sham-Control and carnitine groups; this was observed at all ages analyzed (Fig. 1). However, there was an evident improvement of these parameters in the CE group when compared to the E group; this fact was detected at all studied ages (Fig. 1).
UTF1, SOX2, and PLZF Was Restricted to Type A Spermatogonia
Undifferentiated spermatogonia were identified morphologically and by the presence of UTF1, SOX2, and PLZF. In all groups (S, C, E, and CE), UTF1, SOX2, and PLZF expression was detected in the nuclei of spermatogonia at all ages analyzed (Figs. 2 to 4; Supplemental Fig. S1). Besides, the immunolabeling of these proteins was rare and restricted to type A spermatogonia (Figs. 2 to 4). PLZF+ spermatogonia were more abundant that UTF1+ and SOX2+ spermatogonia at all ages (Supplemental Fig. S2). In all tested conditions, control staining in the absence of the primary antibodies and presence of the IgG antibody was negative in spermatogonia (Supplemental Fig. S3).
Etoposide Causes an Age-dependent Imbalance of the Populations of Type A Spermatogonia and Carnitine Seems to Improve Their Self-renewal
The numerical density of PLZF+ spermatogonia was higher in all groups and at all ages studied, when compared with the numerical densities of UTF1+ and SOX2+ spermatogonia (Supplemental Fig. S2), except for E127 subgroup (Supplemental Fig. S2C). Even as, the numerical density of SOX2+ spermatogonia in all groups and at all ages studied, that was higher when compared with numerical density of UTF1+ spermatogonia (Supplemental Fig. S2), except for CE127 subgroup (Supplemental Fig. S2D).
E32 and CE32 subgroups showed a reduction in the numerical density of SOX2+ spermatogonia when compared to the S32 subgroup (Supplemental Fig. 5B). No significant differences were observed between E32 and CE32 subgroups for all analyzed proteins (Fig. 5). On the other hand, the number of UTF1+ and SOX2+ spermatogonia per seminiferous tubule section was increased in the CE32 subgroup when compared to the E32 subgroup (Supplemental Fig. S4). This increase was also observed at 64 days (Supplemental Fig. S4).
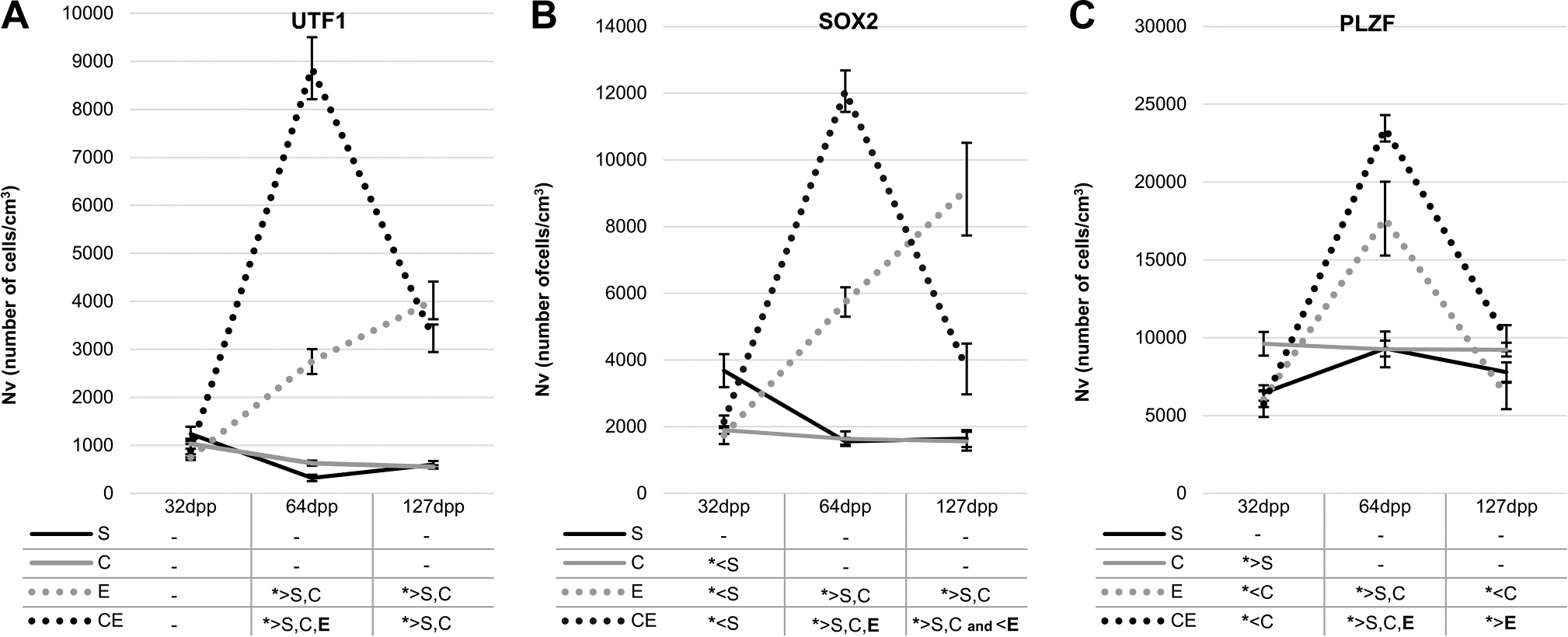
Numerical densities (Nv) of UTF1+(A), SOX2+ (B), and PLZF+ (C) spermatogonia in 32-, 64-, and 127-day-old rats of sham-control (S), carnitine (C), etoposide (E), and carnitine+etoposide (CE) groups. Mean ± standard error of mean; analysis of variance test (n=5); *p≤0.05.
The numerical densities of UTF1+, SOX2+, and PLZF+ spermatogonia increased in the E64 and CE64 subgroups when compared to the S64 and C64 subgroups (Fig. 5). On the other hand, the preadministration of carnitine (CE64 subgroup) lead to an increase in the numerical density of labeled spermatogonia for all studied proteins when compared to the E64 subgroup (Fig. 5).
At 127dpp, the numerical densities of UTF1+ and SOX2+ spermatogonia were higher in the E127 subgroup than in the S127 and C127 subgroups (Fig. 5). Interestingly, the numerical density of SOX2+ spermatogonia in the E127 subgroup was significantly higher than in the CE127 subgroup (Fig. 5B), whereas the numerical density of PLZF+ spermatogonia was higher in the CE127 subgroup when compared to the E127 subgroup (Fig. 5C). Similarly, there was an increase of the number of PLZF+ spermatogonia per seminiferous tubule section in the CE127 subgroup in comparison with the E127 (Supplemental Fig. S4C).
Carnitine Prevented the Loss of Step 19 Spermatids Caused by Etoposide
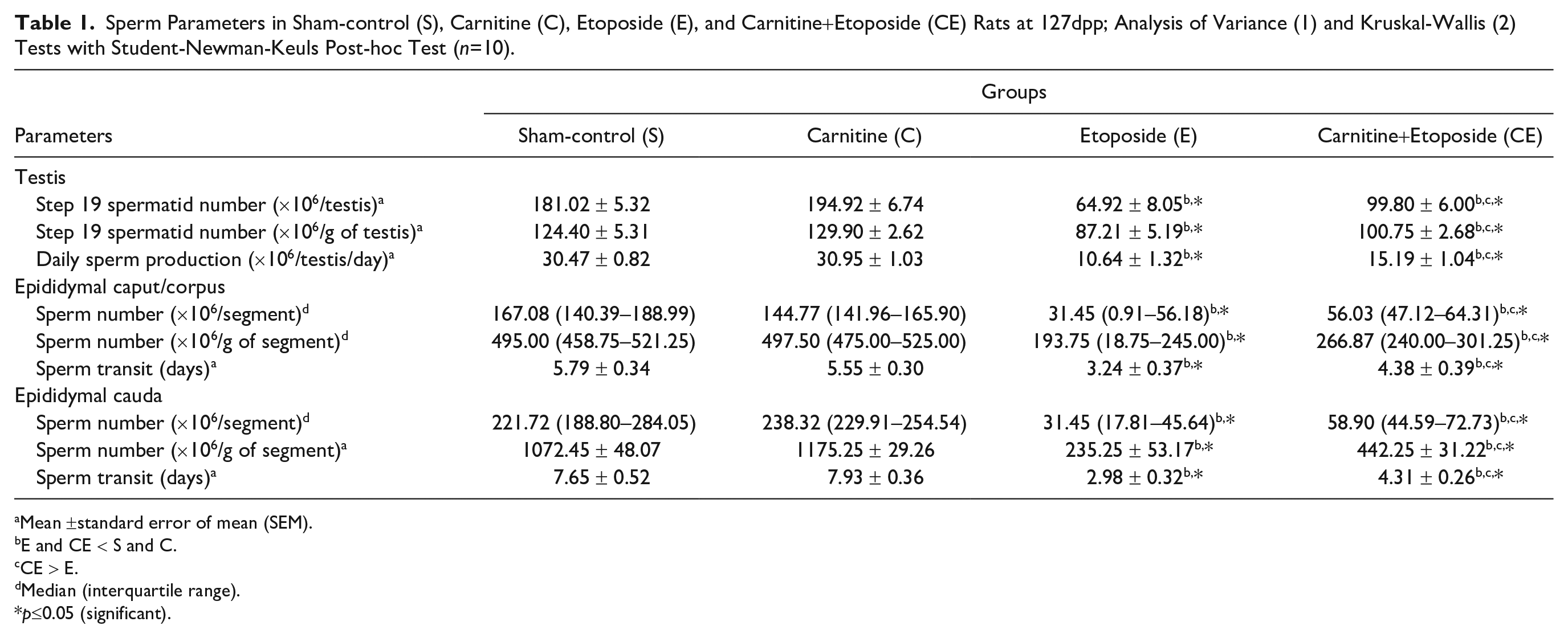
There was a reduction in the number of step 19 spermatids per testis and per gram of testicular tissue in the E127 subgroup when compared with S127 and C127 subgroups. The administration of carnitine before etoposide (CE127 subgroup) led to a significant increase in the number of step 19 spermatids per testis and per gram of testicular tissue when compared with the E127 subgroup, although this number was still lower than that observed in the S127 and C127 subgroups (Table 1).
Sperm Parameters in Sham-control (S), Carnitine (C), Etoposide (E), and Carnitine+Etoposide (CE) Rats at 127dpp; Analysis of Variance (1) and Kruskal-Wallis (2) Tests with Student-Newman-Keuls Post-hoc Test (n=10).
Mean ±standard error of mean (SEM).
E and CE < S and C.
CE > E.
Median (interquartile range).
p≤0.05 (significant).
Carnitine Preadministration Moderated the Reduction of the DSP and of Sperm Number and Transit in the Different Epididymal Segments Caused by Etoposide
The E127 and CE127 subgroups showed a decrease of sperm number, DSP, and sperm transit time in the different epididymal segments when compared to the S127 and C127 subgroups. However, in the CE127 subgroup, there was an evident improvement of these parameters in comparison with the E127 subgroup, suggesting that the previous carnitine administration stimulated the self-renewal of undifferentiated spermatogonia, promoting the partial protection of the spermatogonial lineage and conducing to an enhancement of the spermatogenesis and consequently of the sperm production and its quality (Table 1).
Carnitine Reduced the Damage Caused by the Etoposide in the Sperm DNA
The tail length and the tail extent moment were higher in the E127 subgroup when compared with the S127, C127, and CE127 subgroups (Fig. 6). No significant differences were observed among the S127, C127, and CE127 subgroups regarding to the percentage of tail DNA, the tail length, and the tail extent moment (Fig. 6).
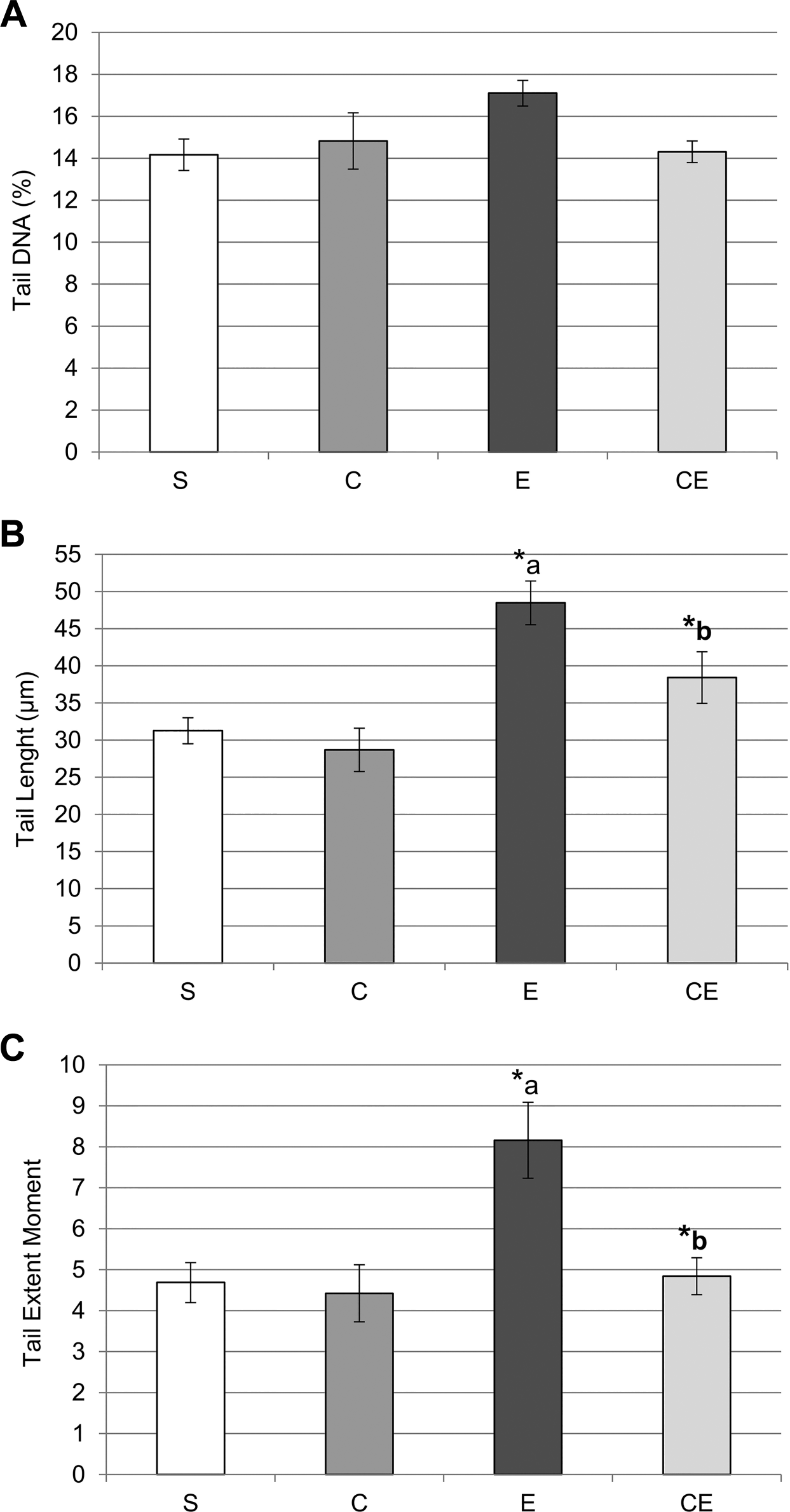
Percentage of tail DNA (A), tail length (B), and tail extent moment (C) determined by COMET assay in 32-, 64-, and 127-day-old rats of sham-control (S), carnitine (C), etoposide (E), and carnitine+etoposide (CE) groups. Values are shown as mean±standard error of mean. Analysis of variance test (n=10). *p≤0.05; a: E > S and C;
Discussion
Etoposide is a chemotherapeutic agent that interferes with male fertility through germ cell and Sertoli cell damage when administered to prepubertal rats4,5,37,38; this harm is especially observed in intermediary and type B spermatogonia that are known to contain high levels of the enzyme topoisomerase II. In a previous study, our group showed that carnitine, when administered to prepubertal rats previously to etoposide, improved the spermatogenesis in adulthood, 4 suggesting that carnitine protected other germ cell types, at least in part, and through a mechanism of action that does not involve topoisomerase II. Thus, considering the prior findings, the aim of the present study was to investigate whether such protection occurred via undifferentiated spermatogonia.
Although spermatogonial stem cells (SSC) have been an important subject of study in the field of Reproductive Biology, the identity of the male germ stem cell has not been categorically defined yet, especially in rats. Another point to be considered is the term “very small embryonic-like cell (VSELs)” that might be an alternative nomenclature to refer to the subtype of the spermatogonial stem cell line that is believed to be the actual stem cell of the spermatogenic lineage. The term has been used in different studies about germ cells that survived anti-cancer treatments. 49 The characteristics of the VSEL are their relatively quiescent state and the presence of pluripotency markers such as OCT-4 and NANOG. Considering that these characteristics are also attributed to the spermatogonial stem cells, we strongly believe that these terms are used to refer the same cell type in the testis.
So in order to identify SSC in the seminiferous epithelium, some markers in subpopulations of undifferentiated spermatogonia have been used8,9,28,29,32,50 as well as their morphological characterization.51–53 Thus, in the present study, we used the previously described spermatogonia markers UTF1, SOX2, and PLZF and morphological characteristics to identify the undifferentiated spermatogonia. Interestingly, the population of PLZF+ spermatogonia has been significantly wider in all studied groups. This observation indicates that the PLZF+ spermatogonia could either contain the UTF1+ and SOX2+ populations or could be a distinct population of spermatogonia. The expression of Utf1 and Plzf genes has been detected in gonocytes and type A spermatogonia in pre- and postnatal rat testes, although the pattern of expression of these markers was not the same. 29 According to van Bragt et al., 29 in rats, all spermatogonia that expressed Utf1 also expressed Plzf. On the other hand, among the spermatogonia that expressed Plzf, few also expressed Utf1. 29 This suggests that the PLZF+ spermatogonia indeed represent a wider population in which the UTF1+spermatogonia are included. We also detected the presence of SOX2 in type A spermatogonia. This finding agrees to those reported by van Bragt et al. 29 that similarly described the expression of Sox2 in type A spermatogonia. Importantly, SOX2 is a transcription factor that controls Utf1 expression. 27
In the present study, the numerical densities of UTF1+, SOX2+, and PLZF+ spermatogonia were similar inside each subgroup, where the CE64 subgroup showed the highest density, followed by the E64 subgroup and by the S64 and C64 subgroups. This indicates that etoposide administration seems to affect the spermatogonia expressing these proteins even when carnitine is administrated.
Since these spermatogonia are responsible for the repopulation of the seminiferous epithelium, this increase in the numerical density could be a compensatory mechanism in attempt to re-establish spermatogenesis. Corroborating with this, an improvement of the testicular volume and of the total volume of seminiferous epithelium was observed in the CE group in comparison with E group in all ages analyzed (32, 64, and 127 days).
It is important to emphasize that, although the rats treated only with etoposide showed an increase of the numerical density of UTF1+, SOX2+, and PLZF+ spermatogonia populations, spermatogenesis reestablishment seems to have taken longer in these rats than in those pretreated with carnitine, as observed in the mid (64dpp) and long (127dpp) term analyses. Similar results concerning the number of UTF1+, SOX2+, and PLZF+ spermatogonia per seminiferous tubule section also indicate that a late spermatogenesis reestablishment occurred in etoposide only treated rats.
At 127dpp, the numerical density of UTF1+ and SOX2+ spermatogonia was higher in the rats treated only with etoposide than in the control rats. Thus, it is possible that these two populations of cells are still working on a compensation process to maintain spermatogenesis, since at this age the seminiferous epithelium is still compromised.
It has been suggested that A single spermatogonia (As) undergoes mitosis without completing cytokinesis to generate chains of progenitor (or transit-amplifying) spermatogonia that are called A aligned (Aal) spermatogonia. 53 Among these Aal cells, some continue to differentiate along spermatogenesis and other get back to a self-renewing state.50,54,55 However, in case of loss of As, progenitor (Aal) spermatogonia are able to self-renew. 55 This reinforces the hypothesis that the increase of the numerical density of UTF1+, SOX2+, and PLZF+ spermatogonia in the etoposide treated rats (E and CE groups) is part of a compensatory mechanism, in response to SSC damage, to maintain spermatogenesis in adulthood. It is important to emphasize that it has been suggested that the undifferentiated spermatogonia are a particularly dynamic population of cells that controls the production of their subpopulations according to the demand for proliferation, differentiation, and stem cell maintenance. 56 And still, according to Luo et al. 8 and Suzuki et al., 9 during the cellular division of the spermatogonia, there is no equal distribution of the proteins in the cytoplasm of daughter cells. In the current study, the effects of two substances, etoposide and carnitine, may have caused functional differences in daughter cells leading to maintenance of the stemness in one cell and differentiation in another cell.
Etoposide toxicity is caused by its interaction with topoisomerase II 1 and by the generation of oxidative stress. 57 The negative effects of etoposide on undifferentiated spermatogonia may have occurred through both mechanisms. On the other hand, the long-term negative effects of etoposide on spermatogenesis might be a result of DNA damage. In the current study, etoposide caused injury of sperm DNA in adult rats; since etoposide was administered during prepuberty, it is reasonable to conclude that this occurred through the damage of SSC DNA during the treatment. It is important to emphasize that, in Wistar rats, the period of the spermatogenesis duration is around 53 days, 58 and in the study, sperm was analyzed after 95 days of the end of the treatment.
In addition, the frequency of morphologically normal sperm was reduced 4 and the sperm transit time along the epididymis was also affected, as observed in the current study. On the other hand, when carnitine is administrated before the etoposide, there is an improvement of the frequency of morphologically normal sperm, 4 as also noted in a protocol using doxorubicin-treatment. 18
Carnitine is also an important cofactor to mitochondrial membrane and is responsible for oxidation, facilitating energy production.59,60 It is also possible that the daily administration of carnitine during prepuberty, for 8 days, caused an increase of antioxidant enzymes such as superoxide dismutase (SOD) and catalase 21 protecting the spermatogonia stem cells against the oxidative damage caused by etoposide and leading to an increase in sperm counts as well as to an improvement of sperm motility and viability.21,61 Likewise, Cabral et al. 17 noted that carnitine previously administrated to prepubertal rats reduced the lipid peroxidation caused by doxorubicin and attenuated the long-term alterations caused by this drug in the germ cells, improving male reproductive capacity. 17 Thus, carnitine antioxidant property as well as its action as a free radical scavenger20,62 was probably important for the reestablishment of SSC and, consequently, for seminiferous epithelium recovery observed here. Besides, as carnitine has antioxidant properties19,21 and promotes DNA repair, 22 it is possible that it might have protected the SSC against etoposide damage during the treatment in the prepupertal phase, leading to an improvement of sperm parameters and DNA integrity in the adulthood, as here observed. Cabral et al.17,18 also observed an improvement of the integrity of the sperm DNA in adult rats that were exposed to a single dose of doxorubicin at prepuberty, but that had been previously treated with carnitine.17,18
It must be also considered that etoposide administration can be associated with increased urinary loss of carnitines. 63 So, in this case, carnitine exogenous supplementation before the administration of etoposide could restore carnitine homeostasis that is altered by this anticancer drug.17,62,63 Thus, carnitine has been suggested as a foremost adjuvant in different chemotherapeutic treatments since it reduces their multiple organ toxicities. 36
Damage caused by doxorubicin, for example, can involve NADPH oxidase activation, which is a critical apoptotic trigger. 64 The NADPH oxidase activation, in its turn, can be inhibited by previous administration of carnitine, reducing the damage in cardiomyocytes. 65 In addition, NADPH oxidase activation is an important ROS-producing source in testis. 66 Although physiological level of ROS is required by spermatogonial cells, allowing the stem cell self-renewal, 67 its increased production can promote apoptosis and cell death. 66 Thus, it is probable that etoposide can have caused a deregulation of this process, resulting in late damage of the spermatogenesis. On the other hand, its feasible regulation by carnitine, as previously observed, 65 could indirectly contribute to a spermatogonial stem cell self-renewal.
In summary, in the present study, the administration of carnitine previously to etoposide improved sperm parameters and seminiferous epithelium recovery, probably due to an attenuation of etoposide negative effects on some spermatogonium subpopulations.
In the present study, we have analyzed undifferentiated spermatogonia through morphology and immunolocalization of UTF1, PLZF, and SOX2. By using these methodologies, we were able to identify the undifferentiated spermatogonia, to detected alterations in their number in etoposide-treated rats and to suggest that carnitine acts positively on SSC and male fertility when administered previously to etoposide. Additional analysis by western blotting and in situ hybridization would be important in a future study to confirm whether these markers and these approaches are, indeed, valuable to address SSC toxicity.
Supplemental Material
2019-00146R2_Production_Supplemental_Data_online_supp – Supplemental material for Carnitine Diminishes Etoposide Toxic Action on Spermatogonial Self-renewal and Sperm Production in Adult Rats Treated in the Prepubertal Phase
Supplemental material, 2019-00146R2_Production_Supplemental_Data_online_supp for Carnitine Diminishes Etoposide Toxic Action on Spermatogonial Self-renewal and Sperm Production in Adult Rats Treated in the Prepubertal Phase by Fatima Kazue Okada, Taiza Stumpp and Sandra Maria Miraglia in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
We would like to thank Prof. Ricardo Pimenta Bertolla (Urological Research Center—Federal University of Sao Paulo) for helping in the image system coupled to the epifluorescence microscope and the KOMET 6.0.1 software.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
FKO has carried out the experimental work; TS and SMM have designed the research. All authors have analyzed interpreted the data, written and revised critically the content, and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We would like to express our gratitude to Foundation of Support to Research of Sao Paulo State (Fundação de Amparo à Pesquisa do Estado de São Paulo—FAPESP—2011/12371-8) for the financial support and to Coordination for the Improvement of Higher Education Personnel (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—CAPES) for the scholarship for Okada F.K.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
