Abstract
Oocyte cell death is a normal process in the mammalian ovary during follicular growth. Recent reports have demonstrated the presence of pro-apoptotic and pro-autophagic proteins during oocyte elimination. The goal of this study was to identify the interactions between proteins involved in different types of programmed cell death in the same oocyte during follicular atresia. We evaluated the presence of Beclin 1 and its interaction with the pro-apoptotic proteins active caspase-3, Bax, and Bak by means of histochemical observations, ultrastructural immunodetection, and immunoprecipitation techniques in ovaries from prepubertal (28- and 33-day-old), juvenile (40-day-old), and young adult (90-day-old) rats. In this study, we identified that oocyte elimination occurred with a high quantity of pro-autophagic protein Beclin 1 and increased the presence of the pro-apoptotic proteins active caspase-3, Bax, and Bak. Conversely, the antiapoptotic protein Bcl-2 was reduced in oocytes from atretic follicles. In addition, Beclin 1 was shown to interact with active caspase-3 and Bax. Our results suggest that the increase in Beclin 1 is directly related to the rise of pro-apoptotic proteins, which could promote the apoptotic process during oocyte elimination.
Introduction
In the mammalian ovary, follicular atresia is a normal process that involves germinal cell elimination to select the oocytes capable of being fertilized. 1 Several studies have shown that apoptosis is the route of elimination of somatic cells inside the follicular structure.2–7 Diverse evidence has also shown the autophagy-associated cell death as another important route to eliminate germ cells in the rat ovary.8–12 Recent published reviews expose the complex routes inside the ovary to underpin oocyte elimination, with necrosis and necroptosis present during this process.13,14 Necrosis is a non-regulated cell death process that leads to organelle and cellular swelling conducive to a loss of cytoplasmic membrane. 15 The term necroptosis refers to a type of cell death with similar morphology to necrosis, but this process is induced by death receptor ligation. 16 Apoptosis is morphologically defined by cytoplasmic shrinking, nuclear compaction, membrane blebbing, 17 and, finally, the formation of apoptotic bodies, which in turn are engulfed by macrophages or other phagocytic cells. 18 A hallmark of apoptosis is the conserved integrity of the plasma membrane to avoid an inflammatory reaction, as the engulfing cells do not produce antiinflammatory cytokines.19,20 Cells undergoing apoptosis are biochemically characterized by the participation of diverse proteins, including caspases, which are proteases expressed as inactive proenzymes. Once activated into their catalytic form, caspases act on diverse intracellular substrates, including other procaspases. These proteases are classified as initiator caspases (caspases-10, -9, and -8) and executioner caspases (caspases-7, -6, and -3).21,22 Apoptosis regulation involves the participation of Bcl-2 family members, including antiapoptotic prosurvival factors (Bcl-XL, Bcl-2, Bcl-W, A1, and Mcl-1) 23 and pro-apoptotic multidomain factors and BH3-only proteins (Bax, Bak, Bim, Bik, Bad, Noxa, and Puma). The Bcl-2 family possesses up to four conserved Bcl-2 homology domains (BH1-4), which are the major regulators of the apoptotic process. 24
Autophagy constitutes a cytoprotective mechanism under certain stress conditions. It is a homeostatic catabolic process in which cytoplasmic material is sequestered for elimination via lysosomal degradation. 25 During the autophagic process, both proteins and organelles can be degraded.26,27 Under certain circumstances, autophagy may lead to cell death. Autophagic cell death is characterized by the absence of chromatin condensation and the large-scale production of autophagic vesicles. 28 Autophagosome formation involves the participation of autophagy-related proteins (ATG), many of which have mammalian orthologues.29,30 Beclin 1, the mammalian orthologue of Atg6, was the first identified mammalian gene with a role in mediating autophagy.31,32 Increased levels of Beclin 1 have been associated with autophagy activation. 33 In addition, Beclin 1 has a functional BH3 domain 34 that allows for its classification as a Bcl-2 family member. Several Bcl-2 family members such as Bcl-2, Bcl-XL, Bcl-W, and Mcl-1 bind to Beclin 1 to inhibit autophagy.34,35 The interaction of Beclin 1 with antiapoptotic members is disrupted by BH3-only proteins, which affect the autophagic process.34,36
Our previous observations have allowed us to recognize the presence of both apoptotic and autophagic proteins during oocyte elimination.8–10 However, how cells activate caspases in the presence of an increased quantity of pro-autophagic proteins, which other pro-apoptotic proteins become activated, and ultimately which route of cell death predominates in altered oocytes remain poorly understood. Considering that high levels of the pro-autophagic protein Beclin 1 have been identified in granulosa cells of atretic follicles, 37 and the properties of this pro-autophagic protein as a Bcl-2 family member, 38 it is important to clarify the role of this protein in the oocytes of atretic follicles. In this study, we assessed the presence of Beclin 1 in the oocytes of atretic follicles in prepubertal, juvenile, and adult Wistar rats to define its interactions with proteins of the apoptotic route during oocyte elimination.
Materials and Methods
Experimental Animals
Nine female Wistar rats for each group aged 28 (prepubertal), 33 (juvenile), 40 (juvenile), or 90 days (adult) were used in this study. They were kept under a 12-hr light–dark cycle with water and food ad libitum. All animals were handled in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals 39 and were approved by the ethical committee under the protocol number PI_2019_02_010.
Histology
Ovaries were fixed in 4% paraformaldehyde in phosphate-buffered saline (PBS) at pH 7.2 for 24 hr at room temperature, embedded in paraffin, and serially sectioned. Five-micrometer-thick sections from each ovary were aligned on glass microscope slides covered with poly-
Immunohistochemistry and Morphometry
Ovaries were fixed in 4% paraformaldehyde in PBS at pH 7.2 for 24 hr at room temperature, embedded in paraffin, and serially sectioned. Five-micrometer-thick sections from each ovary were aligned on glass microscope slides covered with poly-
Antibodies Used for IHC, IEM, IP, and WB.
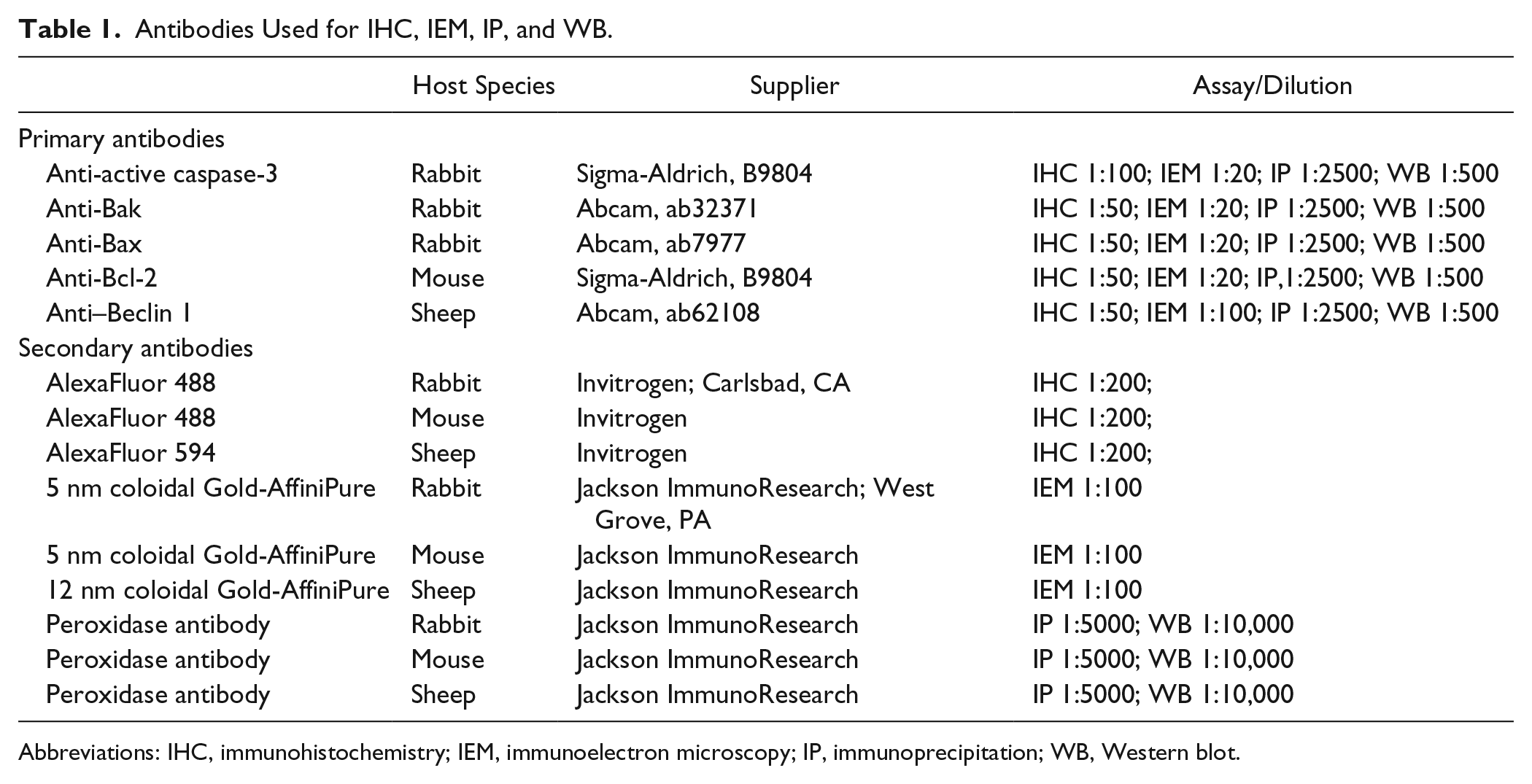
Abbreviations: IHC, immunohistochemistry; IEM, immunoelectron microscopy; IP, immunoprecipitation; WB, Western blot.
Beclin 1 and Apoptotic Protein Colocalization
Ovaries were fixed in 4% paraformaldehyde in PBS at pH 7.2 for 24 hr at room temperature, embedded in paraffin, and serially sectioned. Five-micrometer-thick sections from each ovary were aligned on glass microscope slides and covered with poly-
Transmission Electron Microscopy
Epon Resin Embedding
Ovaries were sectioned in 1 mm3 (approximately) slices and fixed in 2.5% glutaraldehyde–4% formaldehyde in PBS at pH 7.2 for 90 min at room temperature. After rinsing in the same buffer, tissues were postfixed in 1% osmium tetroxide (OsO4) in a PBS buffer at pH 7.2 for 1 hr. Tissues were then dehydrated using a graded ethanol series and embedded in Epon (Embed-812; Electron Microscopy Science, Hatfield, PA). Semi-thin sections were stained with toluidine blue. Selected areas were thin-sectioned and mounted on grids, which were counterstained with uranyl acetate and lead citrate. Sections were examined under a JEOL 1010 electron microscope operated at 60 kV. Digital images were taken with a Hamamatsu camera (Hamamatsu Photonics K. K., Japan).
Immunoelectron Microscopy
Ovaries were sectioned in 1 mm3 slices (approximately) and fixed in 4% formaldehyde in PBS at pH 7.2 for 90 min at 4C. After rinsing in the same buffer, tissues were dehydrated using a graded ethanol series and embedded in Lowicryl K4M acrylic resin (Polysciences, Inc.; Warrington, PA). The whole process was carried out at −20C. Semi-thin sections were stained with toluidine blue. Selected areas were thin-sectioned and mounted in single-slot nickel grids. Ultrathin sections of Lowicryl K4M were mounted on formvar-coated nickel grids and incubated in PBS for 3 min. Subsequently, they were blocked with normal goat serum (NGS, 1:50 in PBS) for 30 min at room temperature. The grids were then incubated overnight at 4C with mixed primary anti–Beclin 1 and anti-active-caspase-3 or anti-Bak, or anti-Bax, or anti-Blc-2. Anti–Beclin 1 antibody was diluted 1:100 in PBS–BSA–Tween (PBS–bovine serum albumin 0.001%–Tween 20 0.05%), and all the other antibodies were diluted 1:20 in PBS–BSA–Tween. Preparations were then washed with PBS–Tween at 0.05%, and subsequently in PBS. The preparations were incubated for 10 min with NGS (1:50 in PBS). The grids were then incubated for 1 hr with secondary antibodies: goat anti-rabbit immunoglobulin coupled to 5 nm gold micelles to detect active-caspase-3, Bak, and Bax. Bcl-2 was detected with goat anti-mouse immunoglobulin conjugated to 5 nm gold particles. Beclin 1 was detected using an anti-sheep immunoglobulin coupled to 12 nm gold micelles. All the antibodies were diluted 1:100 in PBS. Finally, the grids were rinsed with PBS, then with H2O, and air dried. Sections were stained with 0.04% uranyl acetate for 40 sec. Ultrathin sections were examined under a JEOL 1010 electron microscope operated at 60 kV. Digital images were taken with a Hamamatsu camera.
Western Blot
Isolated oocytes from adult rats were used to determine the specificity of primary antibodies. Isolation of oocytes was conducted as previously reported. 8 The cells were placed in lysis buffer for 15 min (50 mM Tris-Cl, pH 7.5; 150 mM NaCl, 0.1% SDS, 1 mM PMSF, 0.5% sodium deoxycholate, and 1% Nonidet P-40), supplemented with complete protease inhibitor cocktail (Roche; Mannheim, Germany). Fifty micrograms of total protein was loaded onto a 12% SDS-PAGE gel. Proteins were transferred to polyvinylidene fluoride membranes and incubated for 1 hr at room temperature in blocking buffer. Then, the membranes were incubated with rabbit anti-active caspase-3, rabbit anti-Bak, rabbit anti-Bax, mouse anti-Bcl-2, and sheep anti–Beclin 1 antibodies, at a dilution of 1:500. Next, the proteins were tagged by incubation with a secondary anti-mouse peroxidase antibody (Jackson ImmunoResearch; West Grove, PA) to identify anti-Bcl-2; anti-rabbit peroxidase antibody (Jackson ImmunoResearch) to detect anti-active caspase-3, anti-Bak, and anti-Bax; and anti-sheep peroxidase antibody (Jackson ImmunoResearch) to detect anti–Beclin 1 at 1:10,000 in blocking buffer for 1 hr at room temperature. Horseradish peroxidase was used as the substrate (Immobilon Western; Millipore Corporation, Billerica, MA). Specific labeling was detected by chemiluminescence. The film (Hyperfilm; Amersham Biosciences, Piscataway, NJ) was exposed to the membranes to detect chemiluminescence.
Immunoprecipitation Assay
For detection of endogenous interaction between Beclin 1 and apoptotic proteins active caspase-3, Bak, Bax, or Bcl-2, immunoprecipitation was carried out using isolated oocytes of different ages. Isolation of oocytes was conducted as previously reported. 9 Briefly, the oocytes were washed with PBS and lysed in immunoprecipitation lysis buffer 0.5% TNTE (50 mM Tris, 150 mM NaCl, 0.5% Triton X100, and 1 mM EDTA) supplemented with protease inhibitors and phosphatase inhibitors. Cell lysates were cleared by centrifugation at 14,000 × g for 5 min, and the supernatant was incubated with 5 µl of anti–Beclin 1. The mixture was incubated under constant rotation at 4C for 2 hr. Then, protein A–coupled Sepharose beads were added to the mixture and incubated for 2 hr at 4C and rinsed three times with wash buffer 0.1% TNTE. Samples were resuspended in Laemmli buffer, boiled for 5 min, and analyzed by SDS-PAGE/immunoblot analysis to test for the levels of active caspase-3, Bak, Bax, and Bcl-2.
Statistical Analyses
Immunodetection was quantified using the Analyze tools of the ImageJ software. The measured area of each oocyte was limited to compare the relative intensities of fluorescence between normal and altered oocytes. At least 60 oocytes from each group (normal, initial atresia, and advanced atresia) of three rats from each age were recorded with a digital camera to quantify fluorescence intensity. A t-test was used to compare the differences between the two groups: basal labeling and increased-altered labeling from each immunodetected protein. Significance was set at p<0.05.
Results
Morphological Description
To characterize the general morphology of the ovaries of 28-, 33-, 40-, and 90-day-old rats, sections of ovaries were stained with hematoxylin and eosin. At all the ages evaluated, follicles were identified in different developmental phases. Normal follicles were recognized by the conserved shape of the follicle and the oocytes, and granulosa cells were located among them. On the contrary, atretic follicles were recognizable because they had an irregular shape, and the granulosa cells and oocytes were altered. In 28-day-old rats, the corpus luteum was not present, showing that ovulation had not yet occurred (Fig. 1A). Follicles at different degrees of atresia advance were identified. Follicles in early atresia were distinguishable because the oocyte began to lose its regular form, with the granulosa cells starting to detach from each other and losing their cellular relationship (Fig. 1A2). The more advanced follicular phase observed at this age represented the preovulatory stage (Fig. 1A3). In the ovaries from 33-, 40-, and 90-day-old rats, the presence of corpus luteum indicated the occurrence of ovulation (Fig. 1B–D). It was possible to identify diverse atretic follicles at all the ages evaluated; some of them presented advanced cell death. At all the ages evaluated, both normal and atretic follicles were present (Fig. 1).
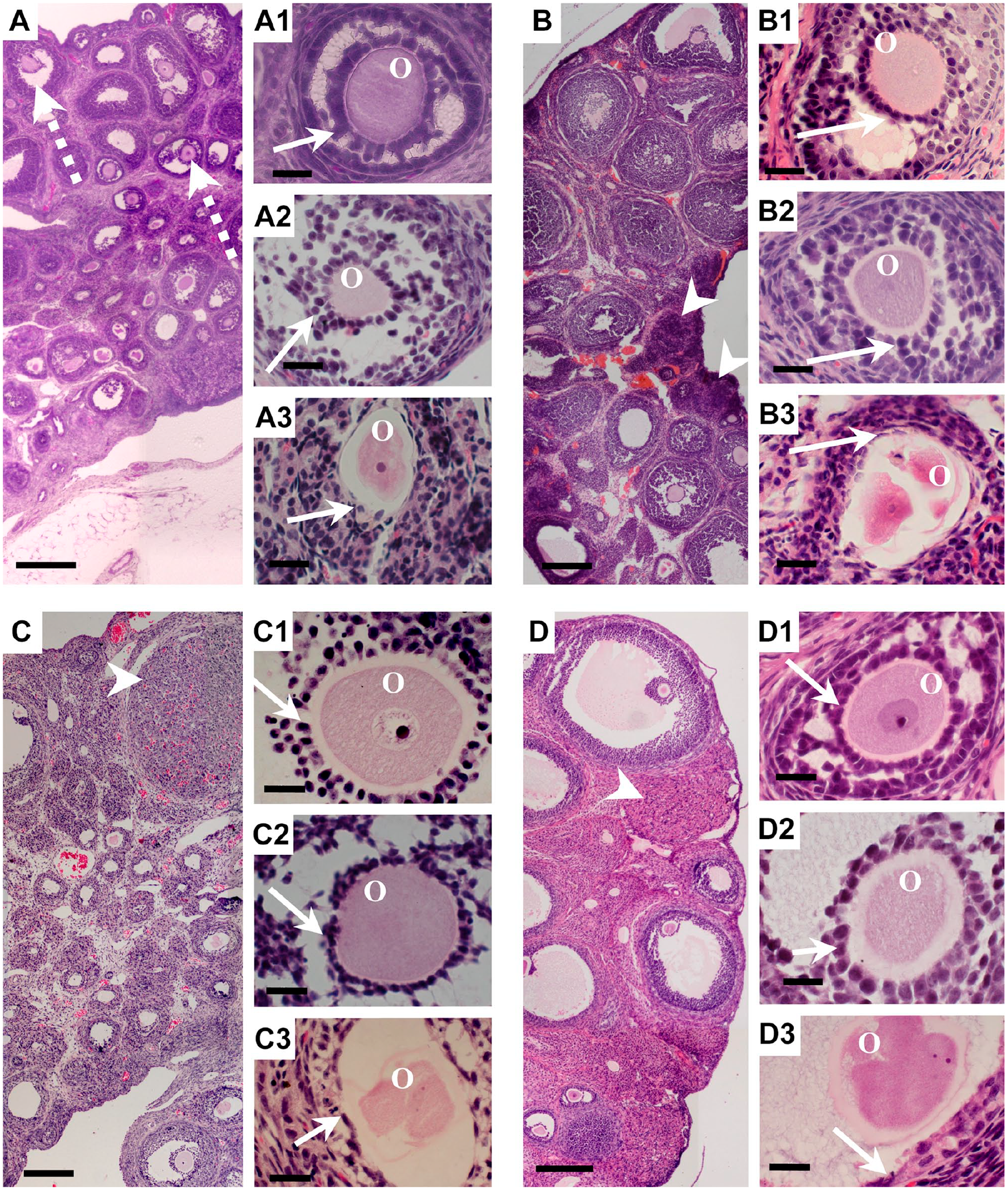
Histological morphology of ovaries from rats of different ages: hematoxylin and eosin staining. (A) Twenty-eight-day-old ovary showing follicles in different phases of development; some follicles were at the preovulatory stage (dotted arrows): (A1) early-antral non-atretic follicle showing granulosa cells (arrow) around an oocyte; (A2) antral follicle in early atresia, in which the granulosa cells are detached from each other (arrow) and the oocyte has an irregular shape; (A3) advanced atretic follicle, in which the granulosa cells (arrow) are altered and the oocyte is highly compacted with an irregular shape. (B) Thirty-three-day-old ovary showing follicles in different phases, with arrow heads pointing to the corpus luteum: (B1) antral non-atretic follicle with arrows indicating granulosa cells surrounding the oocyte; (B2) early-antral follicle in early atresia; (B3) highly advanced atretic segmented oocyte. (C) Forty-day-old ovary, with the presence of the corpus luteum (arrow heads) showing gonadal maturation at this age: (C1) non-atretic antral follicle rounded by granulosa cells (arrow); (C2) antral follicle in early atresia; (C3) highly altered follicle with a segmented oocyte and highly altered granulosa cells (arrow). (D) Ninety-day-old ovary, in which the antral and preovulatory follicles are more evident than in younger ovaries, with the corpus luteum present (arrow head): (D1) early antral normal follicle with a conserved shape; (D2) antral follicle in initial atresia; (D3) highly altered oocyte. Abbreviation: O, oocyte. Scale bars: A, B, C and D, 200 μm; all the others, 20 μm.
Identification of Apoptotic Proteins and Beclin 1
To identify the presence of the apoptotic process, the proteins active caspase-3, Bak, and Bax were immunodetected in follicles from the different ages studied (Fig. 2). Caspase-3 in its active form (Fig. 2K) was present only in the oocytes undergoing apoptosis. Conversely, the pro-apoptotic proteins Bak and Bax were detected at a basal level (Fig. 2D and F), and both were increased during apoptosis (Fig. 2O and Q). Antiapoptotic Bcl-2 protein was present at high quantities in non-altered oocytes (Fig. 2H) but was detected at a lower level in altered oocytes (Fig. 2S). In addition, the pro-autophagic protein Beclin 1 was present at a basal level in normal oocytes (Fig. 2J) but at increased levels in altered oocytes (Fig. 2U).

Immunodetection of the apoptotic proteins active caspase-3, Bak, Bax, and Bcl-2 and the autophagic protein Beclin 1. (A, C, E, G, H, K, M, P, R, T) Phase contrast images. Active caspase-3 was detected only in apoptotic oocytes (L). Bak (D, O) and Bax (F, Q) proteins were present at different quantities, as determined by immunofluorescence, and were increased in apoptotic oocytes. Bcl-2 was present at a basal level in altered oocytes (S) and in increased non-altered oocytes (H). The autophagy-related protein Beclin 1 was increased in altered oocytes (U). Scale bar: 20 μm.
Although the difference in labeling was evident by immunofluorescence, it was necessary to determine significant differences between basal levels and the increased levels. To determine this difference, fluorescence was quantified in each oocyte using the ImageJ program (Fig. 3A). Our results indicate that, in altered oocytes, the pro-apoptotic proteins Bak and Bax were significantly increased (p<0.05). This was observed at all the ages studied. An important aspect was the presence of Bax in oocytes from juvenile rats (33- to 40-day-old), as this protein was increased more than the pro-apoptotic protein Bak. In adult rats, both Bak and Bax increased in altered oocytes in a similar manner (p<0.05). Beclin 1 was increased at all the ages evaluated and was more evident in prepubertal animals (28-day-old). The antiapoptotic protein Bcl-2 was increased in non-altered oocytes at all the ages analyzed (Fig. 3A; p<0.05). Otherwise, in altered oocytes, the level of Bcl-2 was significantly reduced (p<0.05).
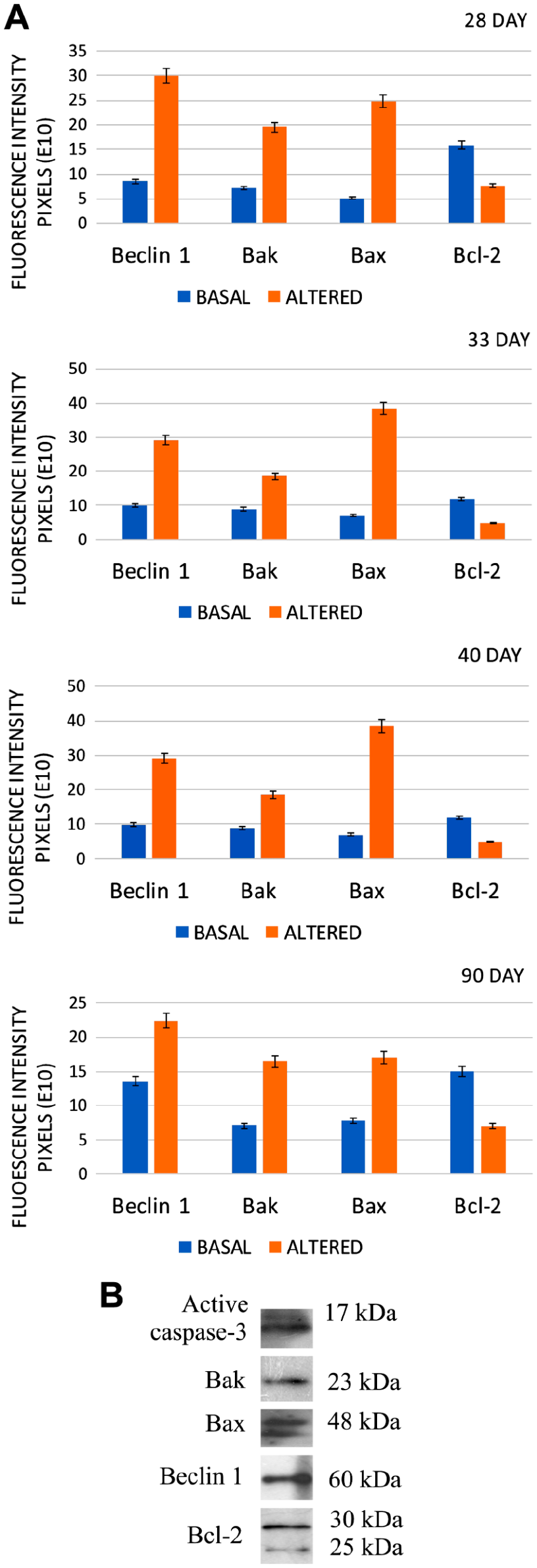
(A) Graphical representation of the levels of fluorescence in non-altered and altered oocytes from different ages. The differences between the levels of labeling show that each protein was significantly different between normal (basal) oocytes and altered oocytes. (B) Western blot to determine the specificity of the primary antibodies.
Beclin 1 Colocalizes With the Pro-apoptotic Proteins Bax and Active Caspase-3
Beclin 1 protein was increased in altered oocytes; similarly, the pro-apoptotic Bak, Bax, and effector caspase-3 were increased in its catalytic form, indicating an active apoptotic process. The oocytes were classified into initial and advanced atresia. A low intensity of fluorescence of pro-apoptotic active caspase-3 labeling was considered to define initial atresia; similarly, increased labeling was considered as an advanced phase of atresia. In addition to the levels of active caspase-3 present in the oocytes, the irregular shape of the cell was considered as an additional parameter to define the advance of atresia. To determine the behavior of Beclin 1 in oocytes undergoing apoptosis, we identified the presence of active caspase-3 simultaneously with the pro-autophagy protein Beclin 1. In addition, the simultaneous presence of Bak and Bax with Beclin 1 was identified.
Our results show that oocytes in initial and advanced atresia simultaneously expressed apoptotic and autophagic proteins. In fact, active caspase-3 and Bax colocalized with Beclin 1; this colocalization was increased in more advanced altered oocytes. In oocytes from the ovaries of 28-day-old rats (Fig. 4), the colocalization of active caspase-3 and Bax with Beclin 1 (Fig. 4C, I, O, and U) was more evident than with Bak and Bcl-2 (Fig. 4F, L, R, and X). In oocytes at this age, there was no colocalization of Beclin 1 with Bcl-2. Bak colocalized with Beclin 1 in oocytes in advanced atresia (Fig. 4U).
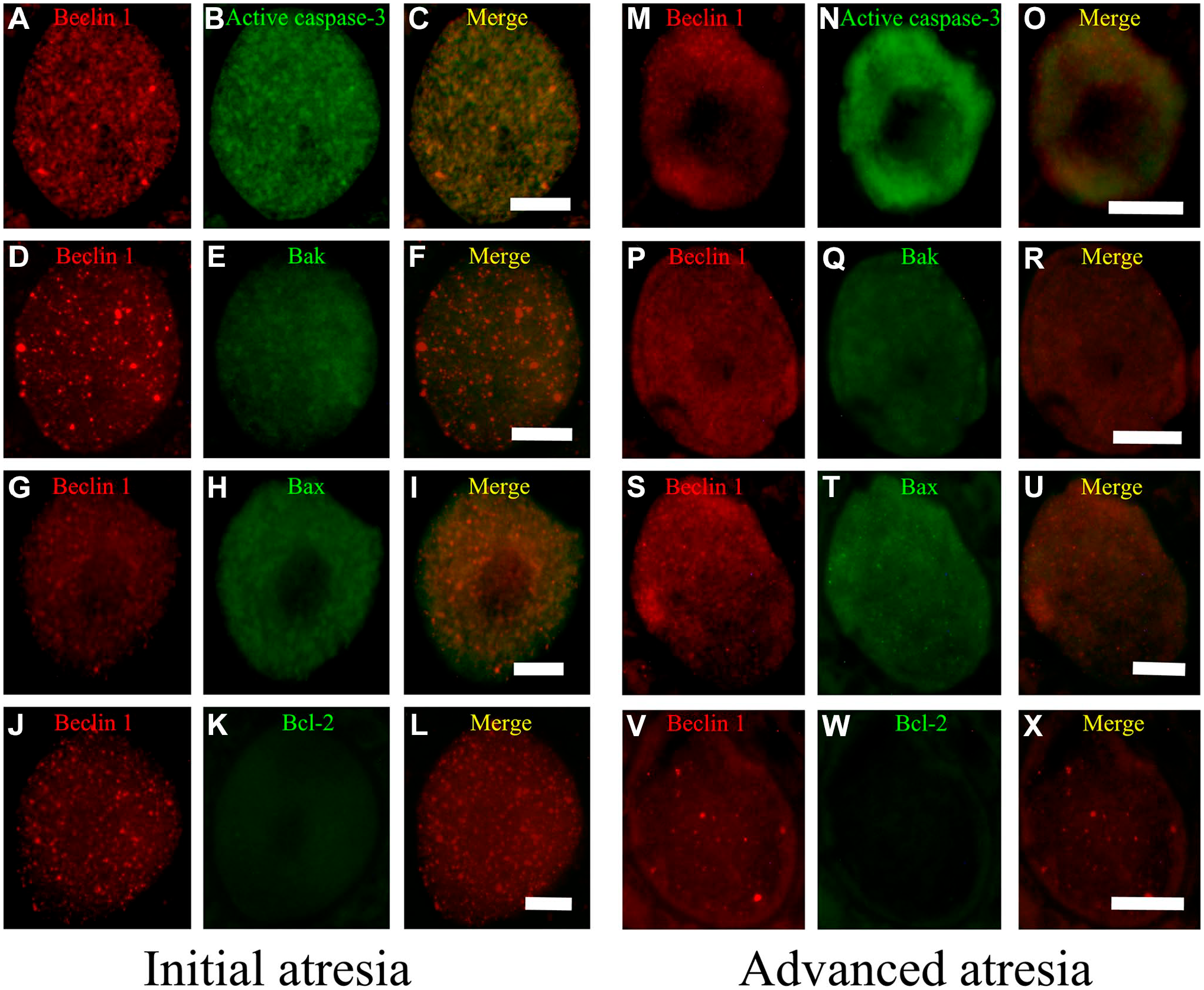
Oocytes from the ovaries of 28-day-old rats. Simultaneous immunodetection of Beclin 1 (red) with apoptotic proteins (green). (A–L) Initial atresia; (M–X) advanced atresia; (A, B, D, G, H, M, P, S, V) Beclin 1; (B, N) active caspase-3; (E, Q) Bak; (H, T) Bax; and (K, W) Bcl-2. (C, F, I, L, O, R, U, X) Merge Beclin 1 with apoptotic proteins. Merged images indicate the colocalization (yellow) of Beclin 1 with another pro-apoptotic protein; this colocalization was increased in the oocytes in advanced atresia. Beclin 1 did not colocalize with Bcl-2 (L, X). Scale bar: 20 μm.
In the oocytes from 33-day-old rats (Fig. 5), there was an important change in the distribution of the proteins in initial and advanced atresia, as the antiapoptotic protein Bcl-2 colocalized with Beclin 1 (Fig. 5L). The merged images allowed us to observe the presence and colocalization of Beclin 1 with apoptotic proteins, which increased with the progression of the atretic process (Fig. 5O, R, and U).
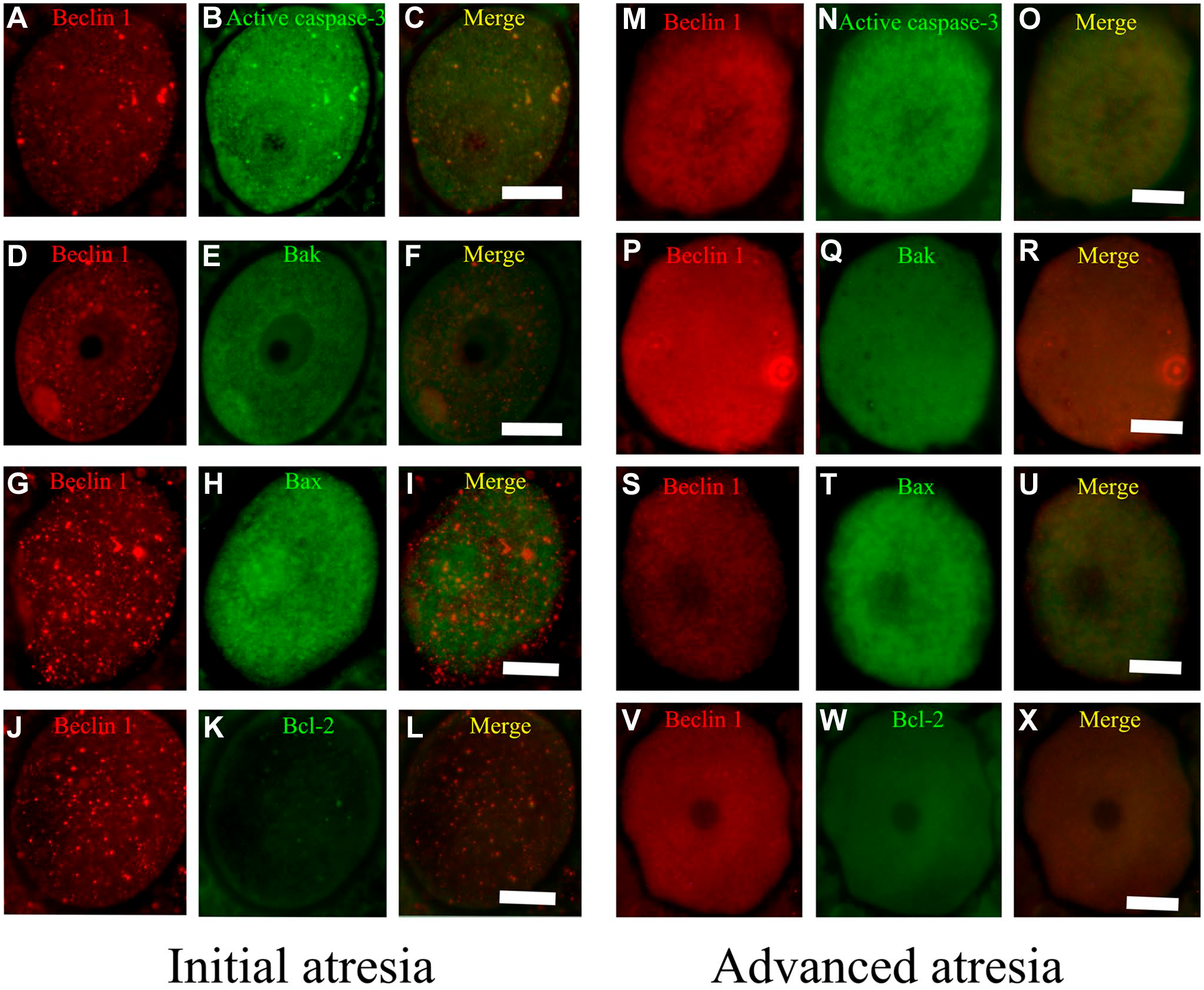
Oocytes from the ovaries of 33-day-old rats. (A, B, D, G, H, M, P, S, V) Beclin 1; (B, N) active caspase-3; (E, Q) Bak; (H, T) Bax; and (K, W) Bcl-2. (C, F, I, L, O, R, U, X) Merge Beclin 1 with apoptotic proteins. In oocytes from follicles in initial atresia (A–L), the major colocalization was between Beclin 1 and active caspase-3 (C). In advanced atresia (M–X), Beclin 1 colocalized with active caspase-3 (O) and Bax (U). Bcl-2 colocalized with Beclin 1 in oocytes in advanced atresia (X). Scale bar: 20 μm.
In 40- and 90-day-old rats (Figs. 6 and 7), the distribution of these proteins was similar in altered oocytes. Beclin 1 colocalized with active caspase-3 in oocytes in initial (Figs. 6C and 7C) and advanced atresia (Figs. 6O and 7O); this colocalization was more evident in oocytes in advanced atresia. Beclin 1 strongly colocalized with active caspase-3 and Bax (Figs. 6I and 7I). Bcl-2 colocalized with Beclin 1 in some cytoplasmic sections of oocytes in advanced atresia (Figs. 6X and 7X).
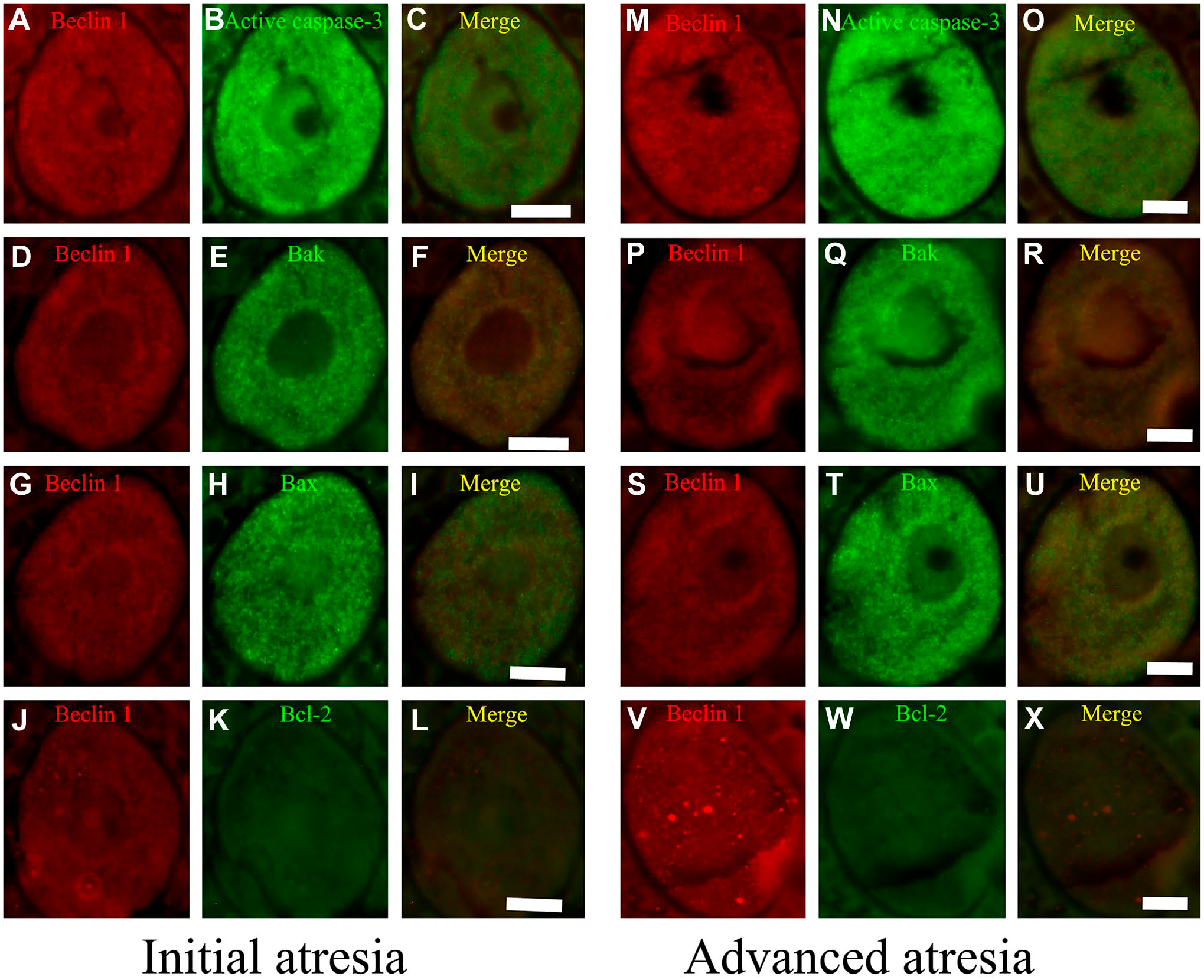
Double immunolocalization of Beclin 1 with apoptotic proteins in oocytes from the ovaries of 40-day-old rats. (A–L) Initial atresia; (M–X) advanced atresia; (A, B, D, G, H, M, P, S, V) Beclin 1; (B, N) active caspase-3; (E, Q) Bak; (H, T) Bax; (K, W); and Bcl-2. (C, F, I, L, O, R, U, X) Merge Beclin 1 with apoptotic proteins. Beclin 1 colocalized with active caspase-3 (C) and Bax (I). This colocalization increased in oocytes in advanced atresia (O, U). Bcl-2 colocalized with Beclin 1 in some areas of the cytoplasmic space of oocytes in advanced atresia (X). Scale bar: 20 μm.
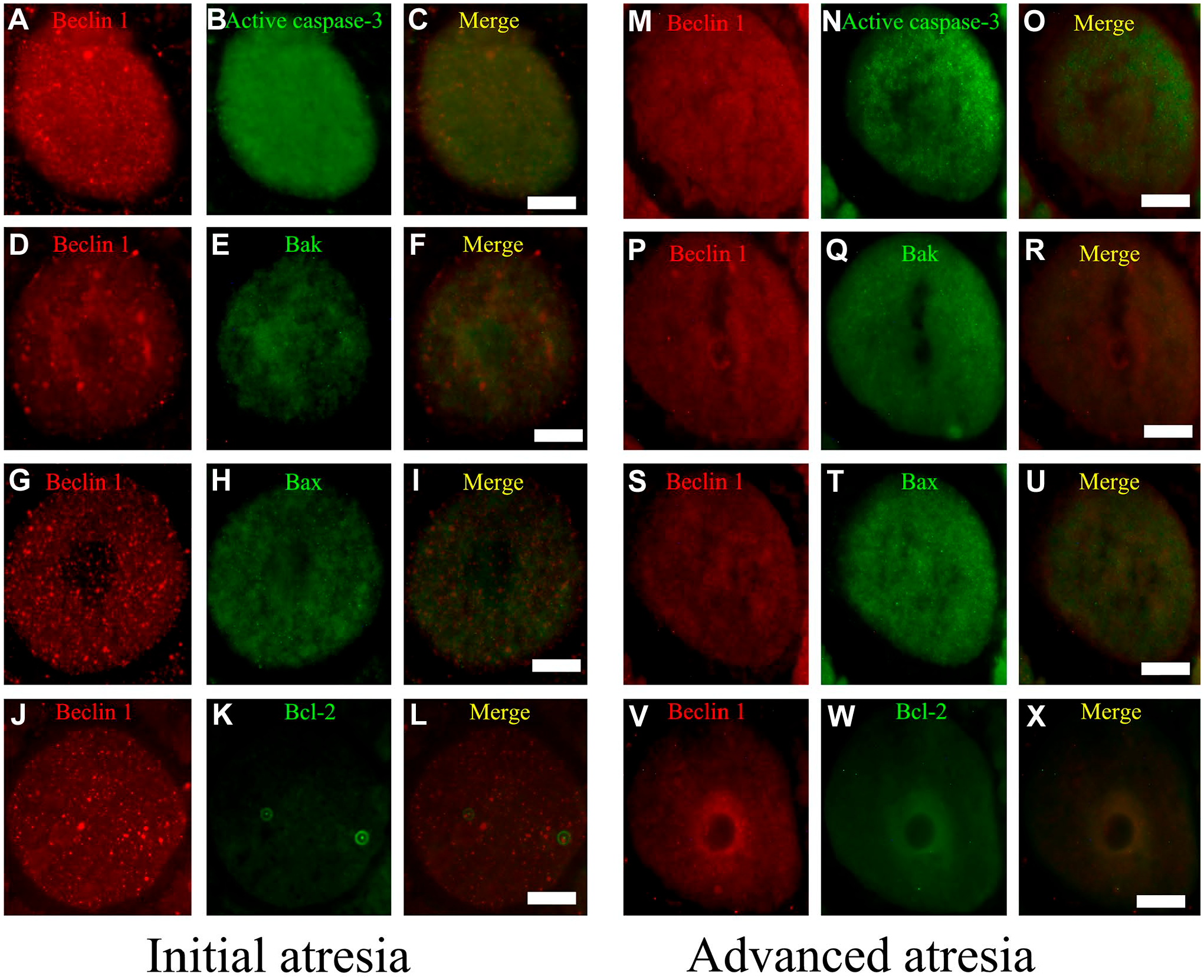
Simultaneous detection of Beclin 1 and apoptotic proteins in the oocytes of 90-day-old rats. (A–L) Initial atresia; (M–X) advanced atresia; (A, B, D, G, H, M, P, S, V) Beclin 1; (B, N) active caspase-3; (E, Q) Bak; (H, T) Bax; and (K, W) Bcl-2. (C, F, I, L, O, R, U, X) Merge Beclin 1 with apoptotic proteins. The colocalization of Beclin 1 with the pro-apoptotic proteins active caspase-3 (C, O) and Bax (I, U) was present in oocytes undergoing initial and advanced atresia. In some areas of the cytoplasm of oocyte in advanced atresia, Beclin 1 colocalized with Bcl-2 (X). Scale bar: 20 μm.
Ultrastructural Characteristics of Oocytes of 28-, 33-, 40-, and 90-day-old Rats
Ultrastructural observations allowed us to identify oocytes inside follicles in different phases of development. Some of them were normal (non-altered) follicles, and others were undergoing atresia.
Normal follicles were characterized by a regular shape (Fig. 8A–D), as granulosa cells conserve their cytoplasmic contact with neighboring cells. The oocyte is surrounded by a regular zona pellucida, which is crossed by the cytoplasmic prolongations of granulosa cells to reach the cytoplasm of the oocyte. The cytoplasmic environment of the oocyte showed conserved mitochondria (Fig. 8A1, B1, C1, and D1). Altered oocytes were identified by their irregular shape (Fig. 8E–H), as the zona pellucida was detached from the oocyte and the granulosa cells had completely lost their contact with the cytoplasm of the oocyte. Oocytes undergoing atresia had an altered cytoplasm (Fig. 8E–H), altered mitochondria, regions with a high quantity of autophagic vesicles, and a compacted cytoplasm, and the cytoplasmic prolongations were altered or lost (Fig. 8E1, F1, G1, and H1). These ultrastructural characteristics were observed at all the ages evaluated.
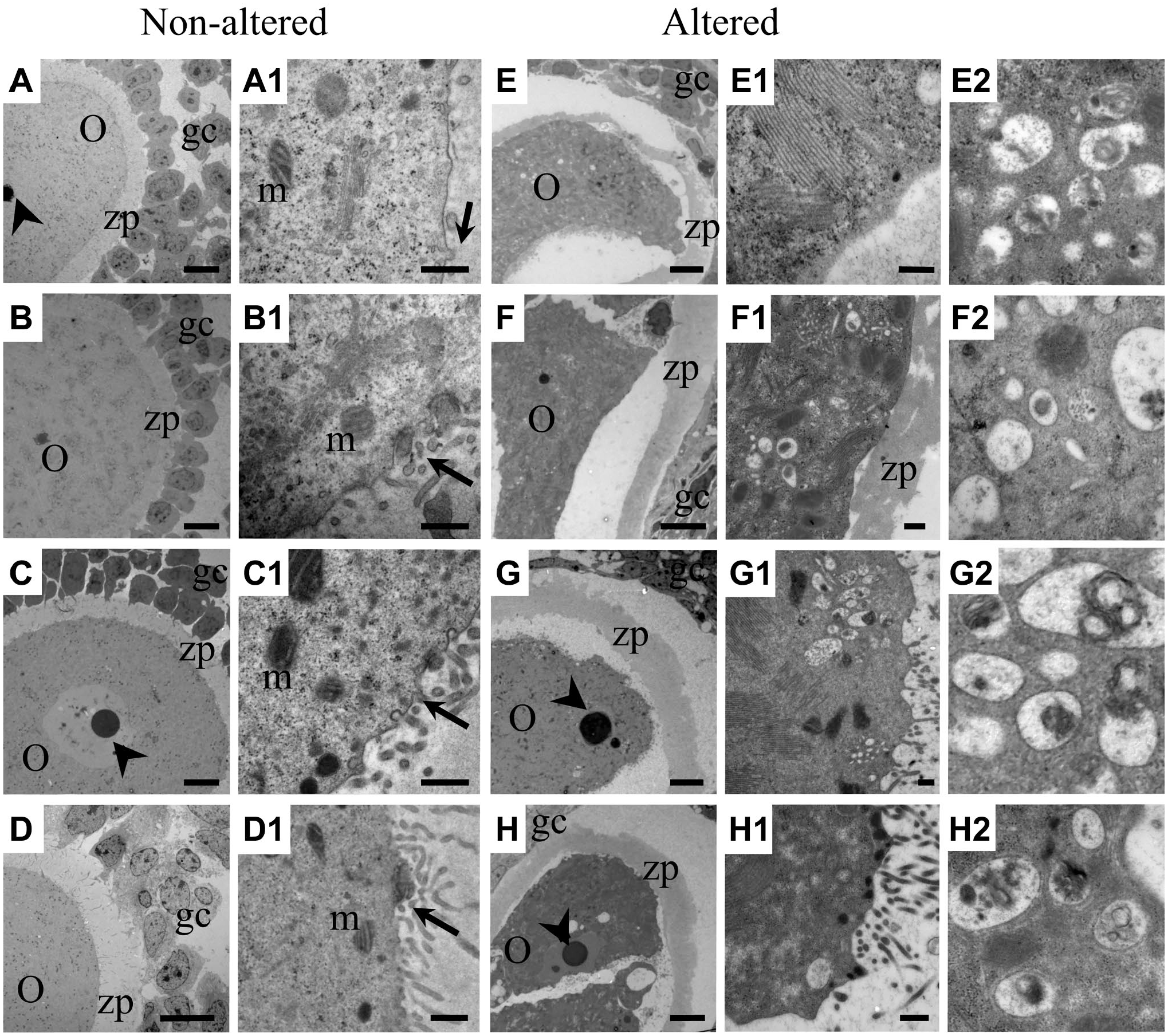
Ultrastructural view of oocytes in normal and atretic follicles. (A, B, C, D) Normal oocytes surrounded by granulosa cells that maintain their relationship with the cytoplasm of the oocyte. The zona pellucida has a regular shape and maintains a close relationship with the oocyte. (A1, B1, C1, D1) Higher magnifications show cytoplasmic prolongations of the granulosa cells with conserved gap junctions (arrow) and healthy mitochondria. (E, F, G, H) Atretic follicles were highly altered, as the oocytes had lost their rounded shape, the zona pellucida had detached, the cytoplasm was highly compacted, and the quantity of granulosa cells was lower. (E1, F1, G1, H1) Higher magnifications show highly altered cytoplasmic elements and the loss of cytoplasmic prolongations into the oocytes. (E2, F2, G2, H2) High magnifications of autophagic vesicles with cytoplasmic content in different degrees of degradation. Abbreviations: O, oocytes; gc, granulosa cells; zp, zona pellucida; m, mitochondria. Low-magnification scale bars: 10 µm; high-magnification scale bars: 500 nm.
Ultrastructural Interaction of Beclin 1 With Apoptotic Proteins
Our optical observations allowed us to determine that active caspase-3 and Beclin 1 were colocalized in altered oocytes. To validate these observations, immunoelectron transmission microscopy was performed to investigate in detail the presence of these proteins, using double-labeling postembedding immunogold for Beclin 1 and active caspase-3, Bak, Bax, or Bcl-2. To identify the presence of Beclin 1, 12 nm gold particles were used, and to identify apoptotic proteins, 5 nm gold particles were used.
Our observations allowed us to identify the presence of Beclin 1 in the cytoplasmic space of altered oocytes; the protein colocalized with Bcl-2 (Fig. 9A–D), Bax (Fig. 9I–L), and active caspase-3 (Fig. 9M–P) in a close manner, which indicates an interaction between the autophagic protein Beclin 1 and these proteins. Bak protein was found to be localized in the mitochondria, but the relationship with Beclin 1 is not closer than with the other proteins (Fig. 9E–H).
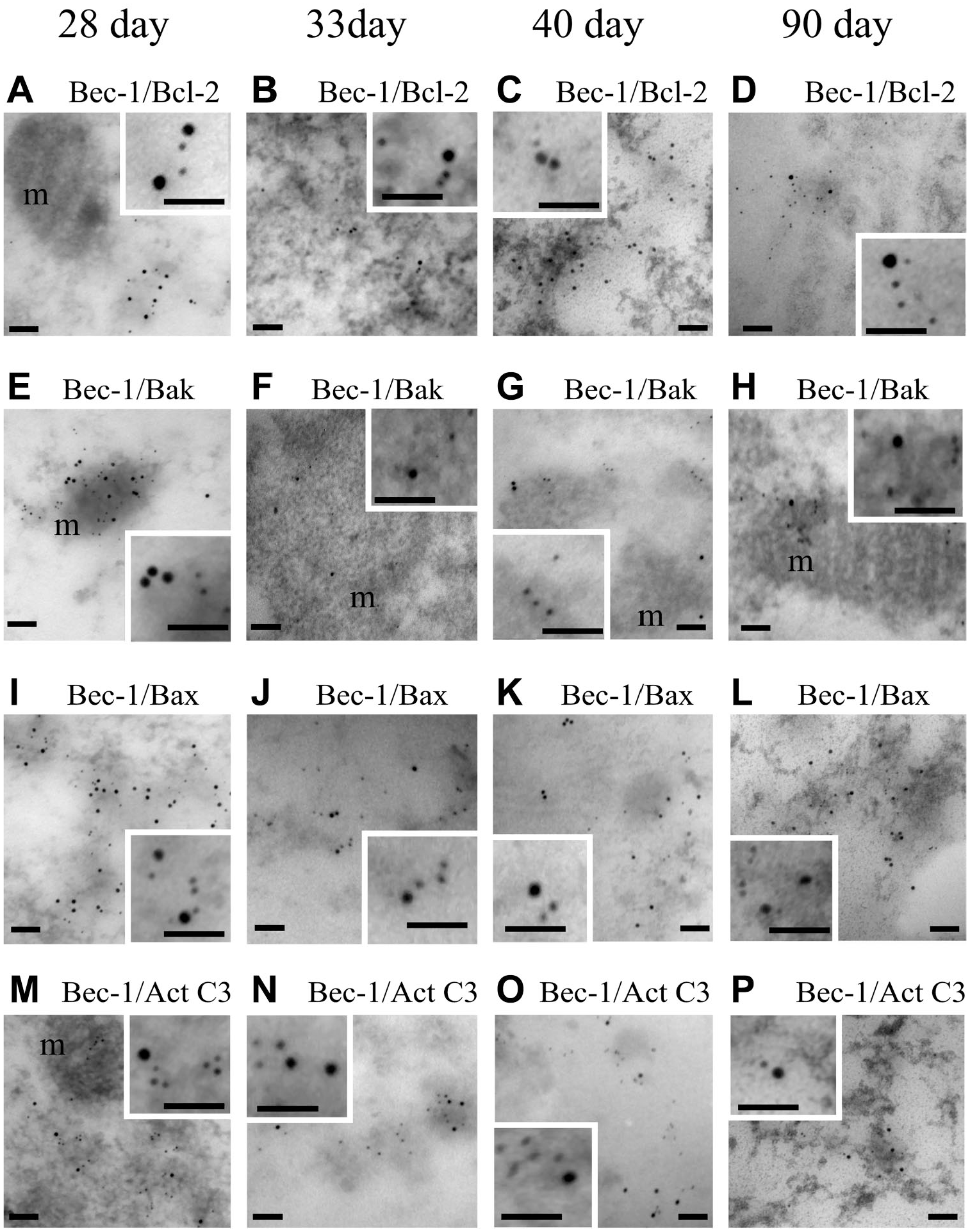
Double-labeling postembedding immune gold for Beclin 1 and apoptotic proteins in oocytes of rats of different ages. Beclin 1 was identified with 12 nm gold particles. All the other proteins were identified by 5 nm gold particles. Higher magnifications of altered oocytes show the presence of gold particles inside the cytoplasm. Bak was present in the mitochondria (m), but the relationship with Beclin 1 was scarce (E, F, G, H). The 5 nm gold particles labeling active caspase-3 (M, N, O, P), Bax (I, J, K, L), and Bcl-2 (A, B, C, D) were close to the gold particles labeling Beclin 1. Inset shows the colocalization of the immunodetected proteins. Scale bars: 100 nm. Scale bars of insets: 50 nm.
Our ultrastructural immunodetection evidenced the presence of Beclin 1 colocalized with active caspase-3, Bax, and Bcl-2 proteins. The near localization of gold particles allowed us to propose that Beclin 1 interacts with the apoptotic proteins.
Beclin 1 Interacts With Active Caspase-3, Bax, and Bcl-2
As our optical and ultrastructural immunodetection indicated that Beclin 1 colocalized with active caspase-3, Bax, and Bcl-2, suggesting that Beclin 1 also interacted with these proteins, we performed immunoprecipitation for Beclin 1 in cellular extracts of purified cell populations of oocytes to determine whether Beclin 1 interacts with the pro-apoptotic proteins active caspase-3, Bax, and Bak or with the antiapoptotic protein Bcl-2.
Immunoprecipitation showed that Beclin 1 interacted with active caspase-3, Bax, and Bcl-2. Conversely, Bak did not interact with Beclin 1 (Fig. 10). This relationship was observed in the oocytes of 28-, 33-, 40-, and 90-day-old rats. These results allowed us to confirm the interaction between Beclin 1 and the pro-apoptotic proteins active caspase-3, Bax, and Bak.
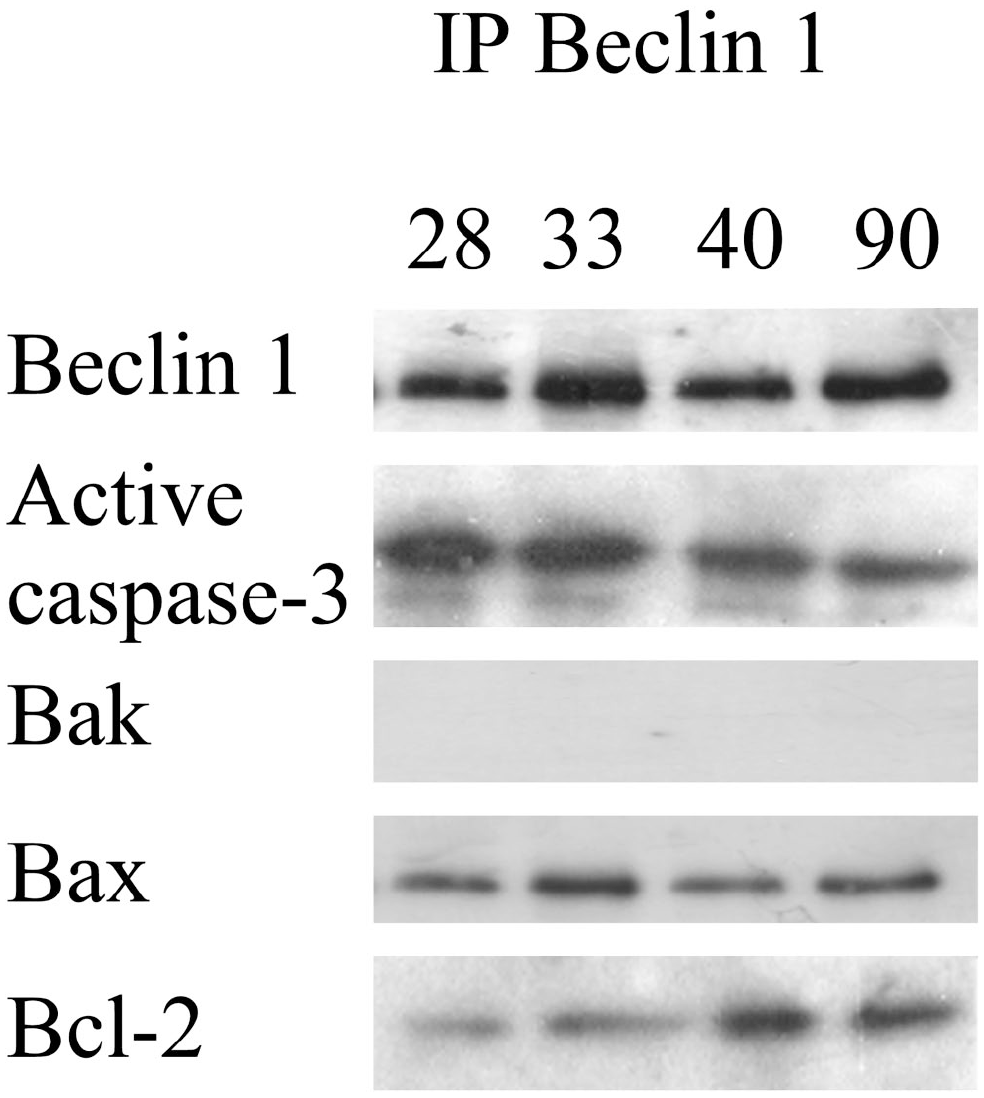
Immunoprecipitation of Beclin 1. Active caspase-3, Bax, and Bcl-2 proteins interacted with Beclin 1. Conversely, Bak protein did not interact with Beclin 1.
Discussion
Follicular atresia in the mammalian ovary is a normal process that helps maintain tissue homeostasis, and apoptotic cell death is an important player during follicular atresia in mammals. 2 Through this process, both somatic cells and oocytes are eliminated. Our work has focused on oocyte cell death during prepubertal and adult ovary atresia in Wistar rats. We previously showed that oocyte cell death is orchestrated by apoptotic and autophagic proteins.8–10 In addition, we identified the presence of the pro-autophagic protein Beclin 1 in granulosa cells 37 ; however, its role during oocyte elimination had not been defined. Beclin 1, the mammalian orthologue of yeast Atg6, is an important player during autophagy. Furthermore, the initiation of autophagosome formation requires Beclin 1. 40 In this study, we investigated the influence of this autophagic protein during oocyte elimination in prepubertal, juvenile, and adult Wistar rats.
Our morphological analyses allowed us to define the absence or presence of corpora lutea in the analyzed organisms. The ovaries from 28-day-old rats are in the prepubertal stage, which was confirmed by the absence of corpora lutea. A corpus luteum was observed in juvenile rats at 33 and 40 days; however, it was smaller in size than that observed in adult 90-day-old rats, showing that 33- and 40-day-old rats had achieved at least two ovulation periods. 41 These observations warranted an evaluation of the presence of the pro-autophagic protein Beclin 1, as well as for proteins involved in apoptosis under different physiological conditions. In the same manner, the morphological analyses in combination with the immunohistochemical characteristics indicated the presence of atretic follicles that were clearly distinguishable at all the ages studied.
Oocytes of atretic follicles were undergoing apoptosis, demonstrated by the presence of active caspase-3; this protease is considered the most important of the executioner caspases and is activated by the initiator caspases (caspase-8, -9, or -10). 18 The effector caspases are responsible for the morphological characteristics of apoptosis, such as membrane blebbing, chromatin condensation, nuclear fragmentation, and engulfment of the apoptotic cell. 42 Active caspase-3 positive oocytes in follicles undergoing initial atresia had an altered shape; this altered morphology was more evident in advanced atresia with more intense active caspase-3 labeling.
As the pro-autophagic protein Beclin 1 has been shown to be present inside granulosa cells in atretic follicles in the ovaries of adult rats, 37 in this work, the presence of this protein was immunodetected in oocytes not only in adult rats but also in prepubertal and juvenile organisms. This study allowed us to show that oocytes from follicles in initial atresia were more positive for Beclin 1 in comparison with non-altered oocytes. In addition, Beclin 1 was increased in altered oocytes with cytoplasmic shrinking and detached granulosa cells. In our previous reports on prepubertal rats, we found a high incidence of oocytes being eliminated by a process involving pro-apoptotic and pro-autophagic proteins. 8 However, the initial mechanism activating both routes of cell death during the elimination of oocytes was not determined. Our present results identify the pro-autophagic Beclin 1 protein as being present at the beginning of the atretic event; moreover, levels were increased with progression of apoptotic characteristics in oocytes.
Very interesting data were obtained by means of the simultaneous immunodetection of Beclin 1 with pro-apoptotic and antiapoptotic proteins. Our results show a correspondence in the levels of Beclin 1 and the pro-apoptotic proteins Bak, Bax, and active caspase-3. We found that increased levels of Beclin 1 during oocyte elimination were correlated with an increased presence of the pro-apoptotic BH3-only proteins Bax and Bak, in which homodimerization is necessary for the intrinsic apoptotic route. 43 This triggers caspase activation, responsible for the proteolytic cleavage of a wide spectrum of cellular targets, including nuclear and cytoskeletal proteins. 39
Furthermore, we identified that the levels of the antiapoptotic protein Bcl-2 were decreased in oocytes with increased labeling of Beclin 1 and pro-apoptotic proteins; in fact, Bcl-2 and Beclin 1 colocalized in some cytoplasmic spaces of oocytes in initial and advanced atresia. This is an interesting phenomenon because Beclin 1 is a Bcl-2-member family and has a functional BH3 domain. 34 Beclin 1 can be inhibited by some Bcl-2 family members 44 such as Bcl-2, Bcl-XL, Bcl-W, and Mcl-1 to inhibit autophagy.34,35 In this sense, during oocyte elimination with morphological characteristics corresponding to apoptosis, Bcl-2 could inhibit Beclin 1, resulting in the release of the pro-apoptotic proteins Bak and/or Bax to promote apoptosis via the activation of active caspase-3.
Our results revealed that Beclin 1 directly interacts with the antiapoptotic protein Bcl-2 and with the pro-apoptotic proteins Bax and active caspase-3. Initially, increased levels of Beclin 1 appear to be controlled by their interaction with Bcl-2 to inhibit the autophagy process. However, this first response induces release of the pro-apoptotic protein Bax from Bcl-2, which allows Bak and Bax to breach the mitochondrial outer membrane, leading to cytochrome c release and caspase activation to promote cell death in oocytes. These tight interactions were evidenced by means of their ultrastructural immunolocalization, demonstrating that the pro-apoptotic proteins active caspase-3 and Bax colocalized with Beclin 1, in the same manner that Bcl-2 interacts with Beclin 1, indicating its regulatory role in apoptosis by direct physical interaction. These interactions of Beclin1 with pro-apoptotic proteins could be an indicator that Beclin 1 is playing different roles in both autophagy and apoptosis.
Increased levels of Beclin 1 suggest that initially autophagy contributes to the survival of oocytes; however, Beclin 1 can be captured by Bcl-2 to control the autophagic process. This impairs the ability of Bcl-2 to inhibit the pro-apoptotic proteins Bax and Bak, which in turn could promote the apoptotic cell elimination. These results provide new insights into oocyte elimination, suggesting that the pro-autophagic Beclin 1 protein could promote apoptosis in a synergistic fashion.
Footnotes
Acknowledgements
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MLE carried out immunohistochemistry; SP-M performed the histological technique; SJ-C performed the ultrastructural immunohistochemistry; LS-S carried out the immunoprecipitation studies; and MLE, OME, and GHV-N designed the study and drafted the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants PAPIIT IN225117, PAPIIT IN227919, and PAPIIT IN216718.
