Abstract
Summary
Intermittent administration of human parathyroid hormone (1-34) (hPTH(1-34)) promotes anabolic action in bone by stimulating bone remodeling, while eldecalcitol, an analog of active vitamin D3, suppresses osteoclastic bone resorption, and forms new bone by minimodeling. We have examined the biological effects of combined administration of eldecalcitol and hPTH(1-34) on 9-week-old Wistar rats that underwent an ovariectomy (OVX) or Sham operation. They were divided into a Sham group, OVX with vehicle (OVX group), OVX with 10 µg/kg/day of hPTH(1-34) (PTH group), OVX with 20 ng/kg/day of eldecalcitol (eldecalcitol group) or OVX with 10 μg/kg/day of hPTH(1-34), and 20 ng/kg/day of eldecalcitol (combined group) for 4 or 8 weeks. As a consequence, the combined group showed a marked increase in bone volume/tissue volume (BV/TV), trabecular thickness (Tb.Th), and trabecular number (Tb.N) than OVX and had the highest bone mineral density (BMD) compared with other groups. OVX and PTH groups exhibited a high osteoblastic surface/bone surface (Ob.S/BS), mineral apposition rate (MAR), and bone formation rate/bone surface (BFR/BS) indices and many TRAP-reactive osteoclasts. Contrastingly, eldecalcitol and combined groups tended to attenuate the indices of osteoclastic surface/bone surface (Oc.S/BS) and Ob.S/BS than that the other groups. The combined group revealed histological profiles of minimodeling- and remodeling-based bone formation. Thus, the combined administration of eldecalcitol and hPTH(1-34) augments their anabolic effects by means of minimodeling and remodeling.
Introduction
Eldecalcitol, an analog of 1α, 25-dihydroxyvitamin D3, has been previously demonstrated to increase bone mass by suppressed bone turnover, consequently enhancing bone strength in rodents.1,2 We reported that eldecalcitol increased bone volume in a dose-dependent manner in ovariectomized rats by not only reducing osteoclastic bone resorption but also increasing bone formation. 3 Eldecalcitol diminished catabolic parameter indices of bone histomorphometry of femoral trabeculae, while it stimulated focal bone formation that started without prior osteoclastic bone resorption, which is a microscopic process known as minimodeling.4,5 Consistently, recent investigations on cynomolgus monkeys and human patients prescribed eldecalcitol have demonstrated an increase in the physiological cross-linkage of collagen fibrils (cynomolgus monkeys) and histological evidences of minimodeling (cynomolgus monkeys and human patients).6,7 During minimodeling, bone formation and bone resorption are not coupled, and bone deposition may take place on quiescent bone surfaces that had not been resorbed by osteoclasts.4,5 As a consequence of bone deposition onto a bone surface that has not been resorbed by osteoclasts, the boundary between the new bone and the old bone features smooth lines referred to as arrest lines, and a mildly convex shape of new bone is observed. A few preosteoblasts were overlying mature osteoblasts in the region of minimodeling-based bone formation, so eldecalcitol does not appear to stimulate preosteoblastic proliferation but accelerates differentiation into mature osteoblasts. 3 Thus, minimodeling, a type of anabolic effect induced by eldecalcitol, seems to be independent of cell coupling from bone-resorbing osteoclasts.
In contrast, human parathyroid hormone (1-34) (hPTH(1-34)) accelerates bone formation and resorption depending on dosing and administration intervals.8,9 Using rodent models, we reported that the intermittent administration of hPTH(1-34) promoted preosteoblastic proliferation and osteoblastic bone formation. 10 However, without osteoclasts, osteoblastic bone formation was not observed, indicating that the PTH-driven anabolic effect is mediated by cell coupling from osteoclasts.11–13 Bone remodeling, in which osteoclastic bone resorption triggers the differentiation and activation of osteoblasts, a process that is referred to as the “coupling phenomenon,”14–16 is one important aspect of PTH-driven anabolism. Previous reports have also documented that PTH-driven anabolic effects require the participation of osteoclasts to trigger preosteoblastic proliferation, differentiation, and ultimately osteoblastic bone formation.10,17,18 As PTH administration affects bone formation and bone resorption, and given that the activities of osteoclasts and osteoblasts are in synergy during normal bone remodeling, it seems plausible that PTH-driven bone anabolism is dependent on bone remodeling.
We have recently assumed the possibility that deferent frequency of hPTH(1-34) administration may affect the turnover of bone remodeling and resultant bone formation. To investigate this assumption, we injected hPTH(1-34) into mice at different administration intervals and histologically examined their bones. 19 As a consequence, a more frequent hPTH(1-34) administration led to the formation of thin trabeculae, thick preosteoblastic cell layers, many osteoclasts, and abundant scalloped-shaped cement lines, indicating accelerated bone remodeling. In contrast, in addition to bone remodeling-based bone formation, a less frequent hPTH(1-34) administration caused mildly convex new bone with arrest lines, that is, minimodeling.
Taken together, both eldecalcitol and hPTH(1-34) appear to have anabolic effects in bone. However, the cellular mechanism on anabolic effects seems to be different between eldecalcitol and hPTH(1-34). Eldecalcitol appears to basically suppress osteoclast formation and subsequent bone resorption and could induce new bone by means of minimodeling. By contrast, a high-frequency hPTH(1-34) administration appears to increase the preosteoblastic population and bone formation by bone remodeling. Therefore, we postulated the possibility that the combined administration of eldecalcitol and hPTH(1-34) could augment their anabolic effects by means of minimodeling and remodeling.
To provide some clues for clarifying this issue, in this study, we histologically examined the biological effects of the combined administration of eldecalcitol and hPTH(1-34) in the long bones of OVX rats.
Material and Methods
Animals and Tissue Preparation
Nine-week-old female Wistar rats either underwent an ovariectomy (OVX) or Sham operation. The rats were divided into the following groups with eight to nine animals each and treated from 4 weeks after operation (13 weeks old): Sham group (Sham operated), OVX group, PTH group (OVX administered 10 µg/kg/day of hPTH(1-34) five times per week; the dosage of the drug was decided according to the previous reports20–21) (Peptide Institute Inc., Osaka, Japan), eldecalcitol group (OVX administered 20 ng/kg/day of eldecalcitol five times per week, as reported by Takeda et al. 22 ), or a combined group (OVX administered 10 μg/kg/day of hPTH(1-34) and 20 ng/kg/day of eldecalcitol for 4 or 8 weeks). This study was performed according to the experimental protocol approved by the Institutional Animal Care and Use Committee at Chugai Pharmaceutical Co., Ltd.
Urinary deoxypyridinoline (DPD) was collected from each rat from all groups to estimate the index of DPD/Cre (n=8–9 for each group). All rats received the subcutaneous injection of tetracycline (20 mg/kg) and calcein (6 mg/kg) at 5 and 2 days before fixation, respectively. The rats were anesthetized with isoflurane, and blood was collected to determine the concentration of serum calcium (Ca) and serum osteocalcin (OCN) (n=8–9 for each group). Right tibiae were immersed in 4% paraformaldehyde diluted in a 0.1 M phosphate buffer (pH 7.4) for 18 hr at 4C, and right femora were immersed in a mixture containing 2% paraformaldehyde and 2.5% glutaraldehyde diluted in a 0.067 M cacodylate buffer (pH 7.4). Left femora were used to determine bone mineral density (BMD) using a bone densitometer (DCS-600EX; Aloka Co., Ltd., Tokyo, Japan). Left tibiae were immersed in 70% ethanol overnight before the Villanueva bone staining, according to Takeda et al. 22 The specimens were dehydrated with increasing concentrations of ethanol, infiltrated into acetone, and then embedded in methylmethacrylate without decalcification. After polymerization, undecalcified tibial sections with 10 µm thickness were prepared for observation under fluorescence microscope (Nikon E800; Nikon Instech Co., LTD) for bone histomorphometric analysis.
After fixation, some specimens were decalcified for 3 months with 10% EDTA for light microscopic observations or 5% EDTA for transmission electron microscopic (TEM) observations and then embedded in paraffin or epoxy resin. For paraffin embedding, decalcified tibiae were dehydrated in ascending ethanol solutions before paraffin embedding. For epoxy resin embedding, some specimens were decalcified, postfixed with 1% osmium tetraoxide in a 0.1 M cacodylate buffer for 4 hr at 4C, dehydrated in ascending acetone solutions, and embedded in epoxy resin (Epon 812; Taab, Berkshire, UK). The other specimens were undecalcified and postfixed with osmium tetraoxide for epoxy resin embedding. Ultrathin sections of the epoxy resin-embedded decalcified specimens were prepared with an ultramicrotome and then stained with uranyl acetate and lead citrate before TEM observations (Hitachi H-7100; Hitachi Co. Ltd, Tokyo, Japan) at 80 kV. 23
Staining for Tissue Nonspecific Alkaline Phosphatase (ALPase) and Tartrate-Resistant Acid Phosphatase (TRAPase)
Dewaxed paraffin sections were examined for tissue-nonspecific ALPase as reported elsewhere.24,25 In brief, after inhibition of endogenous peroxidases with methanol containing 0.3% hydrogen peroxidase for 30 min, dewaxed paraffin sections were pretreated with 1% bovine serum albumin (BSA; Serologicals Proteins Inc. Kankakee, IL) in PBS (1% BSA-PBS) for 30 min. Sections were then incubated for 2 to 3 hr at room temperature (RT) with rabbit polyclonal antisera against ALPase 26 diluted at 1:300 in 1% BSA-PBS. This was followed by incubation with horseradish (HRP)-conjugated antirabbit IgG (DakoCytomation, Glostrup, Denmark). Diaminobenzidine tetrahydrochloride (DAB) was used as a substrate to visualize all HRP-conjugated immunoreactions. Normal rabbit serum was used for negative control experiments. TRAPase activity was detected as previously described.27,28 In short, slides were rinsed with PBS and incubated in a mixture of 2.5 mg of naphthol AS-BI phosphate (Sigma-Aldrich), 18 mg of red violet LB (Sigma-Aldrich) salt, and 100 mM L (+) tartaric acid (0.76 g) diluted in 30 ml of a 0.1 M sodium acetate buffer (pH 5.0) for 15 min at 37C.
Double Staining of ALPase and Proliferating Cell Nuclear Antigen (PCNA)
For the double detection of ALPase/PCNA, which was employed to detect preosteoblasts, 19 PCNA staining was performed before ALPase detection. For PCNA immunohistochemistry, sections were pretreated with Tris-EDTA buffer (pH 9.0) at 70C for 2 hr and then preincubated with 1% BSA-PBS for 30 min at RT. Treated sections were incubated with a mouse anti-PCNA antibody (EMD Chemicals, Inc., San Diego, CA) at 1:50 for 1 hr. The secondary antibody used was an HRP-conjugated antimouse IgG (Bethyl Laboratories, Inc., Montgomery, TX). The immunoreactivity of PCNA was visualized with DAB. Normal mouse serum was used for negative control experiments. Then, ALPase immunoreactivity was detected with an ALP-conjugated antirabbit IgG (Jackson ImmunoResearch Laboratories, Inc., West Grove, PA) at 1:100 at RT. Sections were then incubated in a mixture of 2.5 mg of naphthol AS-BI phosphate (Sigma-Aldrich) and 18 mg of fast blue RR salt (Sigma-Aldrich) diluted in 30 ml of a 0.1 M Tris-HCl buffer (pH 8.5) for 15 min at 37C. Counterstaining with methyl green was performed on all sections before observation under a light microscope (Eclipse E800; Nikon Instruments Inc., Tokyo, Japan).
Von Kossa Staining
Histological sections of epoxy resin-embedded undecalcified specimens were soaked in an aqueous solution of silver nitrate until a dark brown/black staining of the bone tissue was discernible under a light microscope. 24
Bone Histomorphometry of BV/TV, Tb.N, Tb.Th, Tb.Sp, Ob.S/BS, Oc.S/BS, ES/BS, MAR, and BFR/BS
We measured bone volume/tissue volume (BV/TV), trabecular number (Tb.N), trabecular thickness (Tb.Th), trabecular separation (Tb.Sp), osteoblastic surface/bone surface (Ob.S/BS), osteoclastic surface/bone surface (Oc.S/BS), eroded surface/bone surface (ES/BS), mineral apposition rate (MAR), and bone formation rate/bone surface (BFR/BS) as recently reported. 19 A 600 μm × 600 μm region of interest (ROI) located 150 μm below the growth plate of the tibial metaphysis was employed for the assessment of the above-mentioned static parameters of bone histomorphometry. Whenever possible, abbreviations and calculations were conducted according to the recommendations of the American Society for Bone and Mineral Research (ASBMR) Histomorphometry Nomenclature Committee. 29
Statistical Analyses
All statistical analyses were assessed by a one-way ANOVA followed by a Tukey–Kramer multiple comparisons test. All values are presented as mean ± SD, and p<0.05 was considered statistically significant.
Results
The Indices of BMD, Serum Concentration of OCN and Ca, and Urinary Deoxypyridinoline
All the indices of BMD in the eldecalcitol, PTH, and combined groups were higher than those in the OVX group at 4 and 8 weeks (Fig. 1A and B). The highest index of BMD of femora was demonstrated in the combination group at both 4 and 8 weeks, and it reached a significant level compared with that in the eldecalcitol and PTH groups, except the eldecalcitol group at 4 weeks. Although there was no significant difference in BMD between the eldecalcitol and PTH groups at 4 weeks, the PTH group displayed a slightly higher index of BMD compared with the eldecalcitol group at 8 weeks. The index of serum OCN was higher in the PTH and combined groups compared with that in the OVX and eldecalcitol groups at 4 and 8 weeks (Fig. 1C and D). Conversely, DPD/Cre was significantly reduced in the eldecalcitol group at 4 weeks and also in the eldecalcitol and combined groups at 8 weeks than OVX group (Fig. 1E and F). There was no significant difference on the index of in the DPD/Cre between the eldecalcitol and combined groups. The serum concentration of Ca was higher in the eldecalcitol group at 4 weeks (Fig. 1G) and in the eldecalcitol and combined groups at 8 weeks compared with that in the other groups (Fig. 1H).
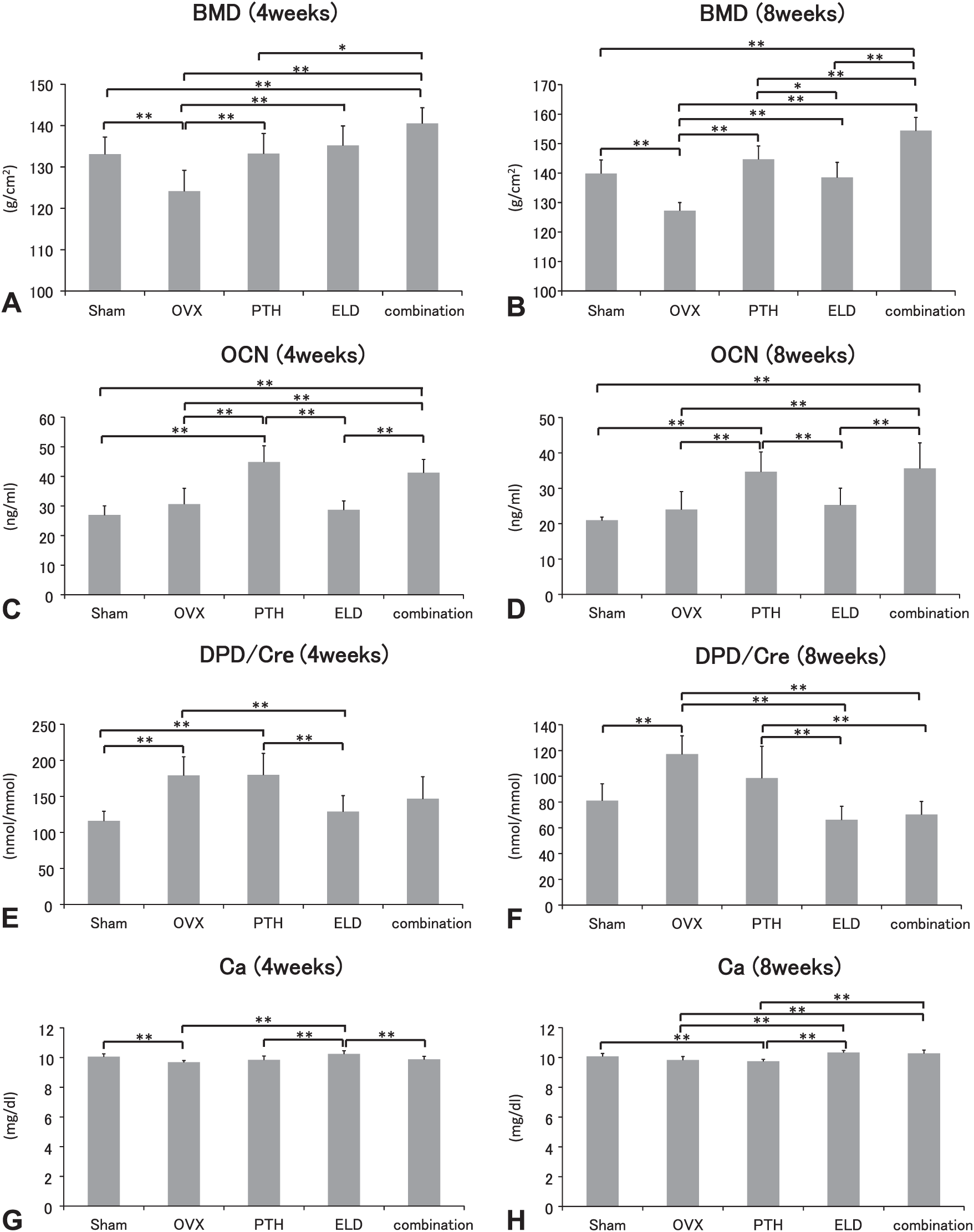
Tibial BMD, serum concentration of OCN, Ca, and Pi, and urinary DPD in OVX, PTH, eldecalcitol, and combined groups. All groups treated with PTH, eldecalcitol, and a combination of PTH and eldecalcitol show a higher index of BMD compared with the OVX group at both 4 and 8 weeks. Above all, the combined group reveals the highest value at both time points (A and B). Serum concentration of OCN is significantly higher in the PTH and combined group compared with that in the OVX and eldecalcitol groups at both 4 and 8 weeks (C and D). By contrast, the index of DPD/Cre is significantly reduced in the eldecalcitol group at 4 weeks and in the eldecalcitol and combined group at 8 weeks compared with that in the OVX group (E and F). The index of serum Ca is significantly elevated in the eldecalcitol group at 4 weeks and in the eldecalcitol and combined group at 8 weeks compared with that in the OVX group (G and H). Abbreviations: BMD, bone mineral density; OCN, osteocalcin; DPD, deoxypyridinoline; OVX, ovariectomy; PTH, parathyroid hormone.
Histological Observations and Static Parameters of BV/TV, Tb.Th, Tb.N, and Tb.Sp in OVX, PTH, Eldecalcitol, and Combined Groups
At 4 weeks, tibiae metaphyseal trabeculae in the OVX group was histologically markedly decreased, whereas those in the PTH, eldecalcitol, and combined groups demonstrated an increased volume of metaphyseal trabeculae (Fig. 2A–J). The indices of BV/TV in the PTH, eldecalcitol, and combined groups were significantly elevated than that in the OVX group at 4 weeks (Fig. 2K). Although Tb.Th was higher in the PTH, eldecalcitol, and combined groups compared with that in the OVX group, the index in the eldecalcitol group was significantly lower than that in the PTH and combined groups but tend to higher than OVX group (Fig. 2L). Tb.N was significantly increased in the PTH, eldecalcitol, and combined groups compared with that in the OVX group (Fig. 2M). Conversely, Tb.Sp in the OVX group was significantly higher than that in the other groups (Fig. 2N).
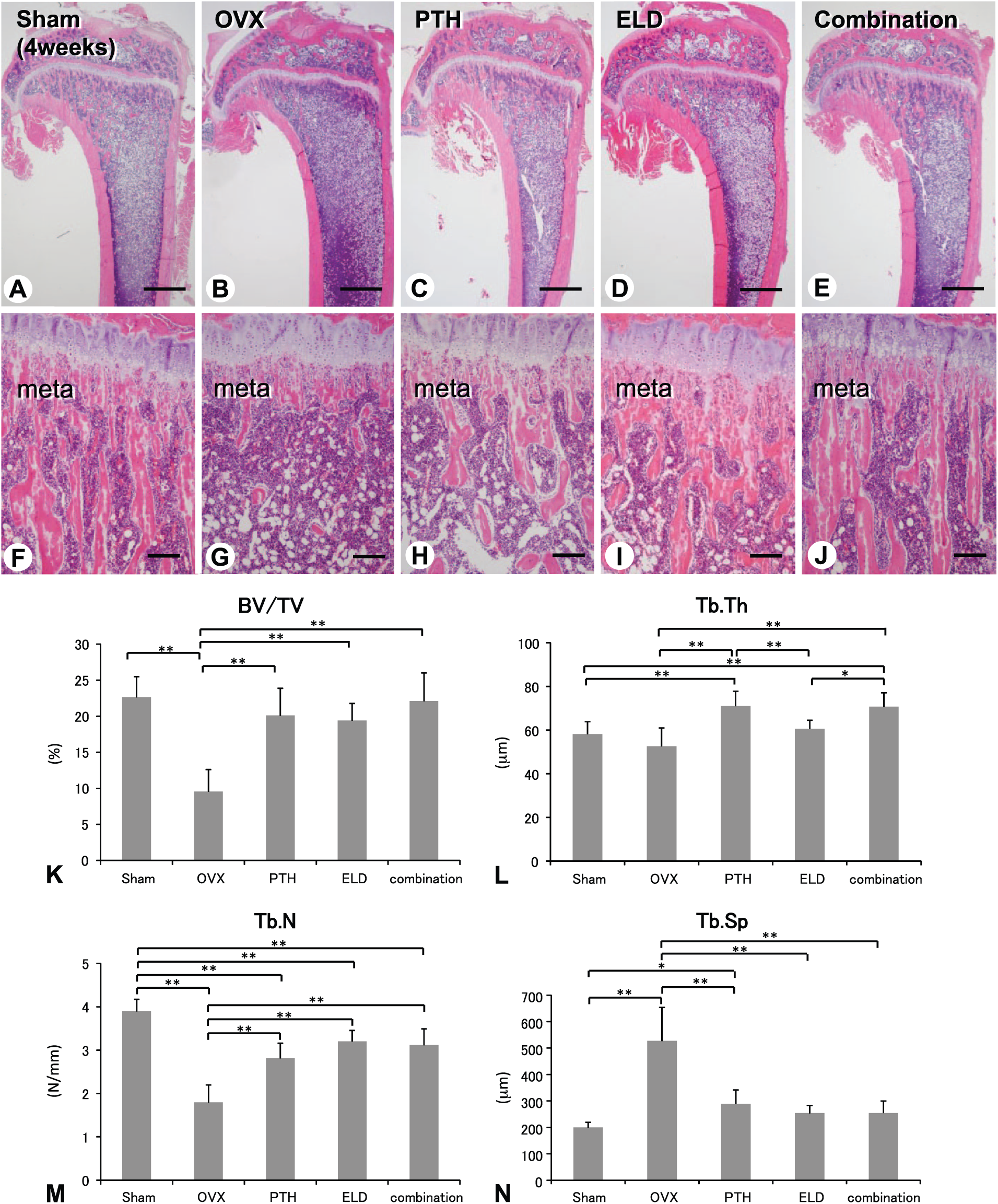
Bone histology and bone histomorphometry of tibiae in Sham, OVX, PTH, eldecalcitol, and combined groups (4 weeks). H&E staining demonstrates the markedly reduced metaphyseal trabeculae in the OVX group compared with that in the Sham group (A, B, F, and G). The PTH, eldecalcitol, and combined groups show the induced volume of metaphyseal trabeculae (C–E and H–J). The indices of BV/TV and Tb.N are significantly increased in the PTH, eldecalcitol, and combined groups compared with those in the OVX group (K and M). Tb.Th is higher in the PTH, eldecalcitol, and combined groups compared with that in the OVX group, whereas the index of Tb.Th is significantly lower in the eldecalcitol group compared with that in the PTH and combined groups (L). Tb.Sp is lower in the PTH, eldecalcitol, and combined groups compared with that in the OVX group (N). meta: metaphysis; Bars: A–E: 1 mm, F–J: 300 µm. Abbreviations: OVX, ovariectomy; PTH, parathyroid hormone; H&E, hematoxylin and eosin; BV/TV, bone volume/tissue volume; Tb.N, trabecular number; Tb.Th, trabecular thickness; Tb.Sp, trabecular separation.
Eight weeks after the administration, the tibial metaphyses of the combined group showed well-developed trabeculae compared with the other groups (Fig. 3A–J). Consistently, the indices of BV/TV and Tb.Th of the combined group were markedly elevated than OVX or eldecalcitol group although there was no significant difference compared with those in the PTH group (Fig. 3K and L). The PTH and combined groups revealed a higher Tb.N but a lower Tb.Sp. (Fig. 3M and N).
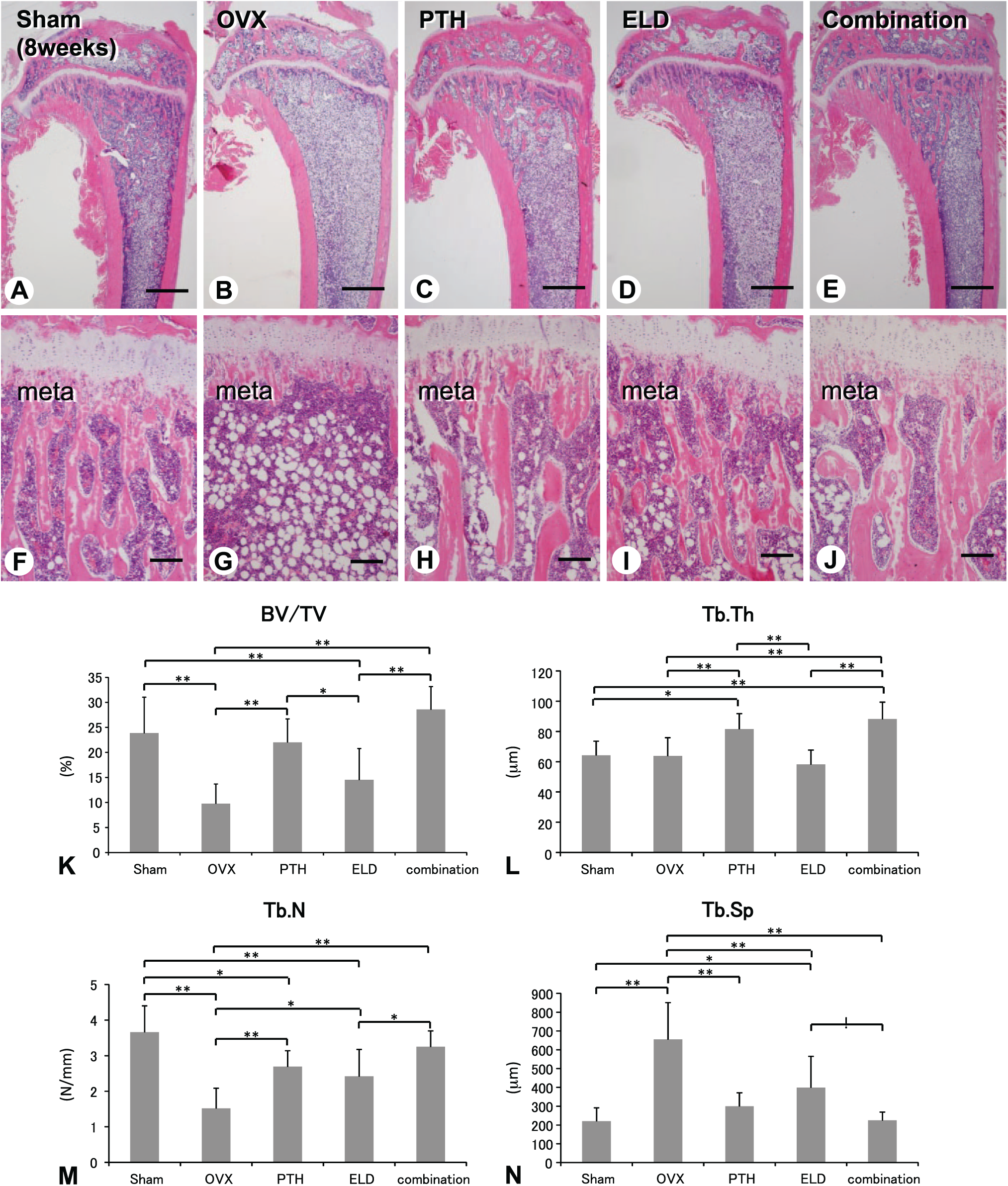
Bone histology and bone histomorphometry of tibiae in Sham, OVX, PTH, eldecalcitol, and combined groups (8 weeks). At 8 weeks, the combined group shows a thickened metaphyseal trabeculae (A–J) and the highest value of BV/TV and Tb.Th (K and L) compared with the other groups. The index of Tb.N in the PTH and combined groups is elevated (M), but Tb.Sp consistently decreased in those groups (N). meta: metaphysis; Bars: A–E: 1 mm, F–J: 300 µm. Abbreviations: OVX, ovariectomy; PTH, parathyroid hormone; BV/TV, bone volume/tissue volume; Tb.Th, trabecular thickness; Tb.N, trabecular number; Tb.Sp, trabecular separation.
Evaluation of Bone Formation and Bone Resorption in Tibiae of OVX, PTH, Eldecalcitol, and Combined Groups at 8 Weeks
At 8 weeks of the administration, the PTH and combined groups featured well-developed metaphyseal trabeculae and had the highest index of BV/TV. Therefore, we examined ALPase immunolocalization as well as anabolic parameters of Ob.S/BS, MAR, and BFR/BS in the tibial metaphyses of the specimens at 8 weeks. ALPase immunoreactivity was seen intensely in the PTH and combined groups but not in the eldecalcitol group (Fig. 4A–H). The index of Ob.S/BS was highest in the PTH group and second highest in the OVX and combined groups (Fig. 4I).
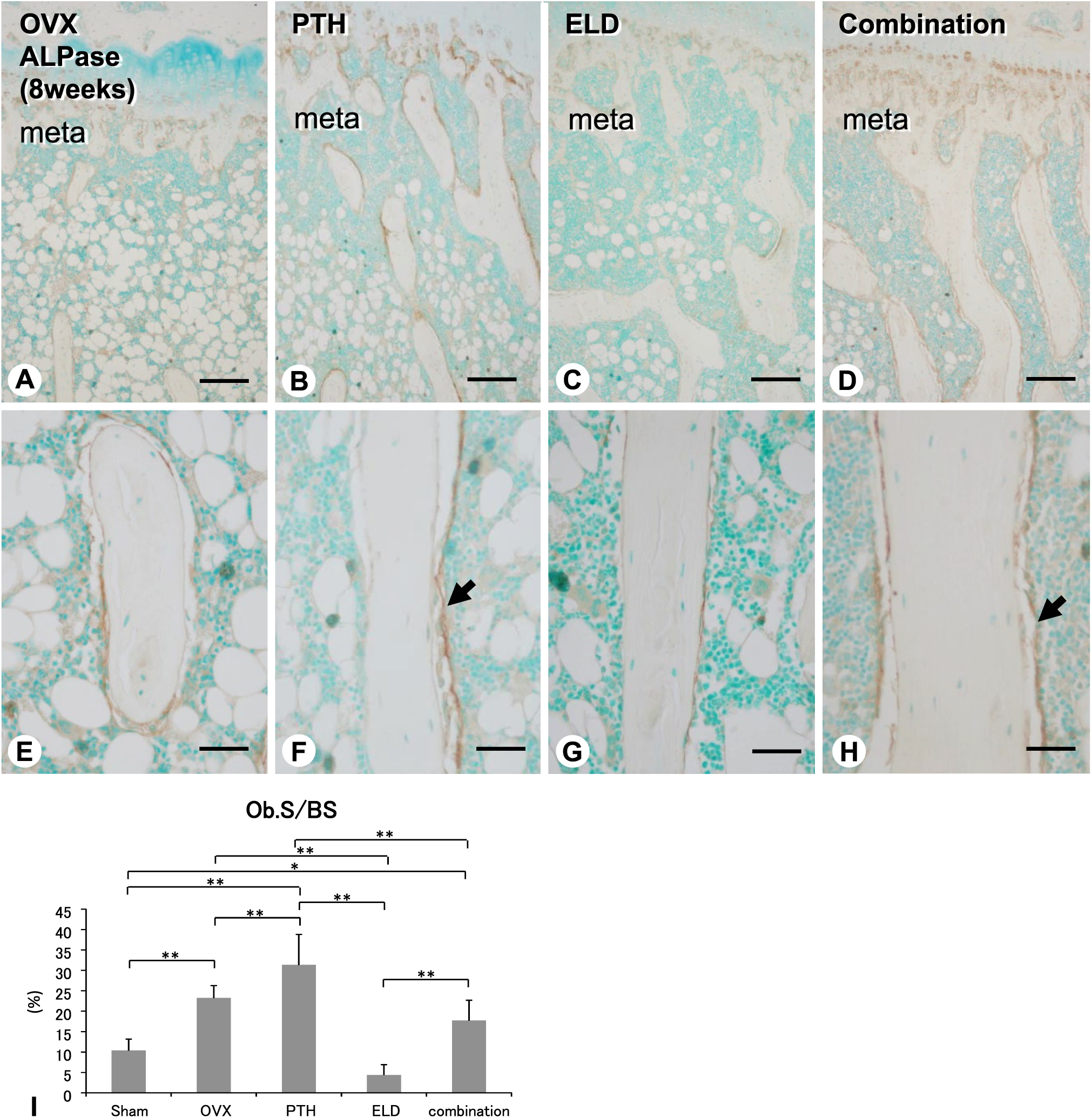
Distribution of ALPase-reactive cells and the index of Ob.S/BS in the tibiae of OVX, PTH, eldecalcitol, and combined groups (8 weeks). There is an intense immunoreactivity of ALPase on the bone surface in the tibial metaphysis of the PTH and combined groups (arrows, B, D, F, and H) but not in that of the Sham and eldecalcitol groups (A, C, E, and G) at 8 weeks after the administration. The indices of Ob.S in the PTH and combined groups are higher than in the Sham and eldecalcitol groups (I). meta: metaphysis. Bars: A–D: 250 µm, E–H: 25 µm. Abbreviations: ALPase, alkaline phosphatase; Ob.S/BS, osteoblastic surface/bone surface; OVX, ovariectomy; PTH, parathyroid hormone.
The double detection of calcein and tetracycline revealed a scalloped-shape labeling in the OVX and PTH groups while the eldecalcitol group demonstrated focal convex labeling (Fig. 5A–C and E–F). However, the combined group exhibited both focal convex labeling and scalloped labeling (see Fig. 5D and H). The OVX and PTH groups demonstrated the highest indices of MAR and BFR/BS, while the eldecalcitol and combined groups showed lower MAR, and BFR/BS of the eldecalcitol group was the lowest among all groups (Figs. 5I and J). Thus, histological and histomorphometrical parameters verified that the most abundant volume of metaphyseal trabeculae was in the PTH and combined groups at 8 weeks; however, the way in which the bone volume increased appeared to be different between the two groups.
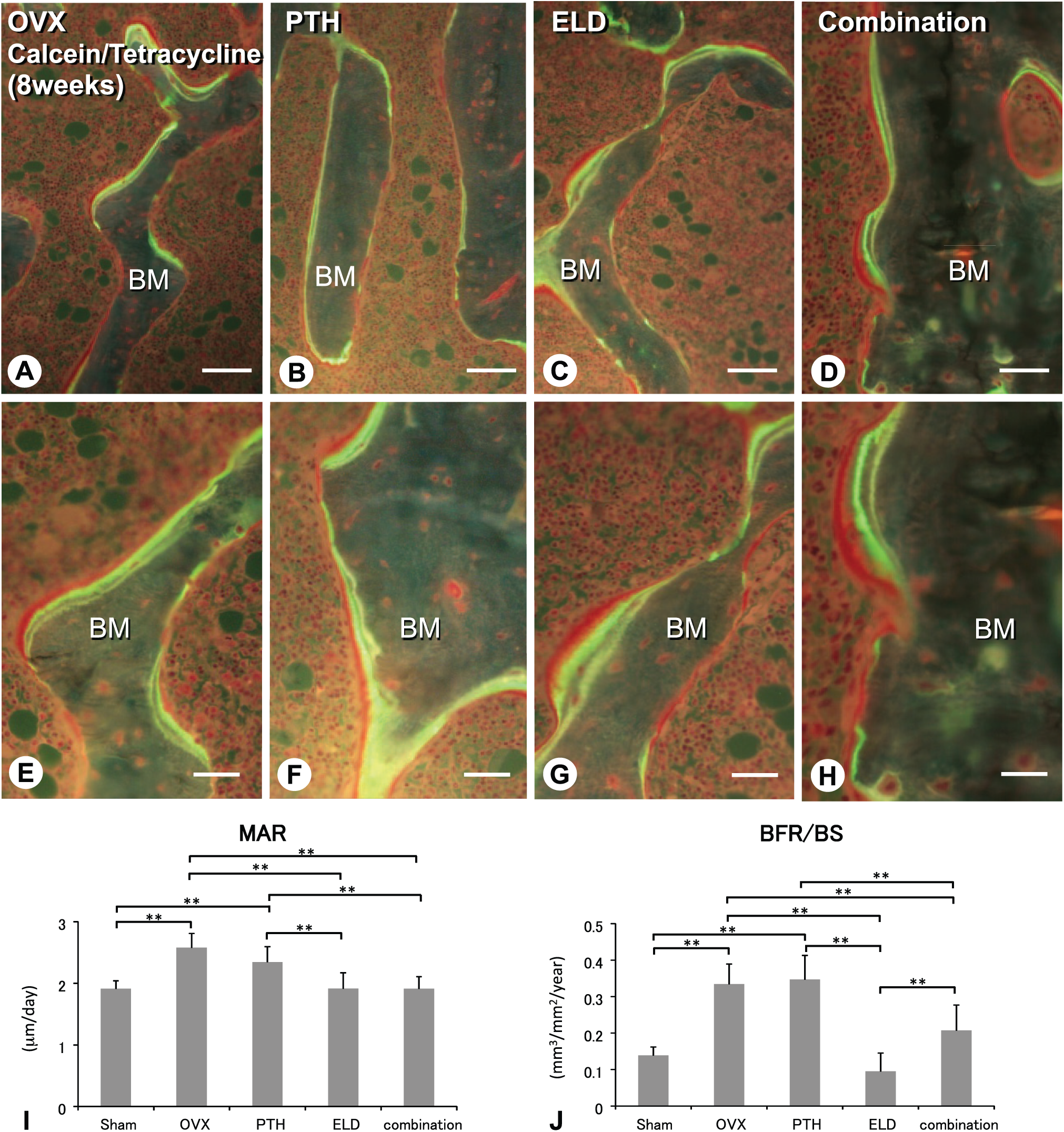
Histological aspects and static parameters of bone formation in the tibiae of OVX, PTH, eldecalcitol, and combined groups (8 weeks). The double labeling of calcein and tetracycline shows the scalloped shape, indicating remodeling-based bone formation in the OVX and PTH groups (A, B, E, and F), while the eldecalcitol group reveals focal convex labeling indicating minimodeling-based bone formation (C and G). The combined group demonstrates both focal convex and scalloped labeling in the metaphyseal trabeculae (D and H). The OVX and PTH groups show the highest value of MAR and BFR/BS, while the eldecalcitol and combined groups demonstrate lower MAR and BFR/BS values (I and J). Bars: A–H: 25 µm. Abbreviations: OVX, ovariectomy; PTH, parathyroid hormone; MAR, mineral apposition rate; BFR/BS, bone formation rate/bone surface; BM: bone matrix.
Therefore, we examined the catabolic effects, that is, osteoclastic bone resorption in these groups. As a consequence, the tibial metaphyses of the OVX group displayed more TRAPase-positive osteoclasts compared with the other groups (Fig. 6A–H). The indices of Oc.S/BS and ES/BS were consistently highest in the OVX group (Fig. 6I and J). By contrast, Oc.S/BS and ES/BS in the eldecalcitol and combined groups were attenuated (Fig. 6I and J).
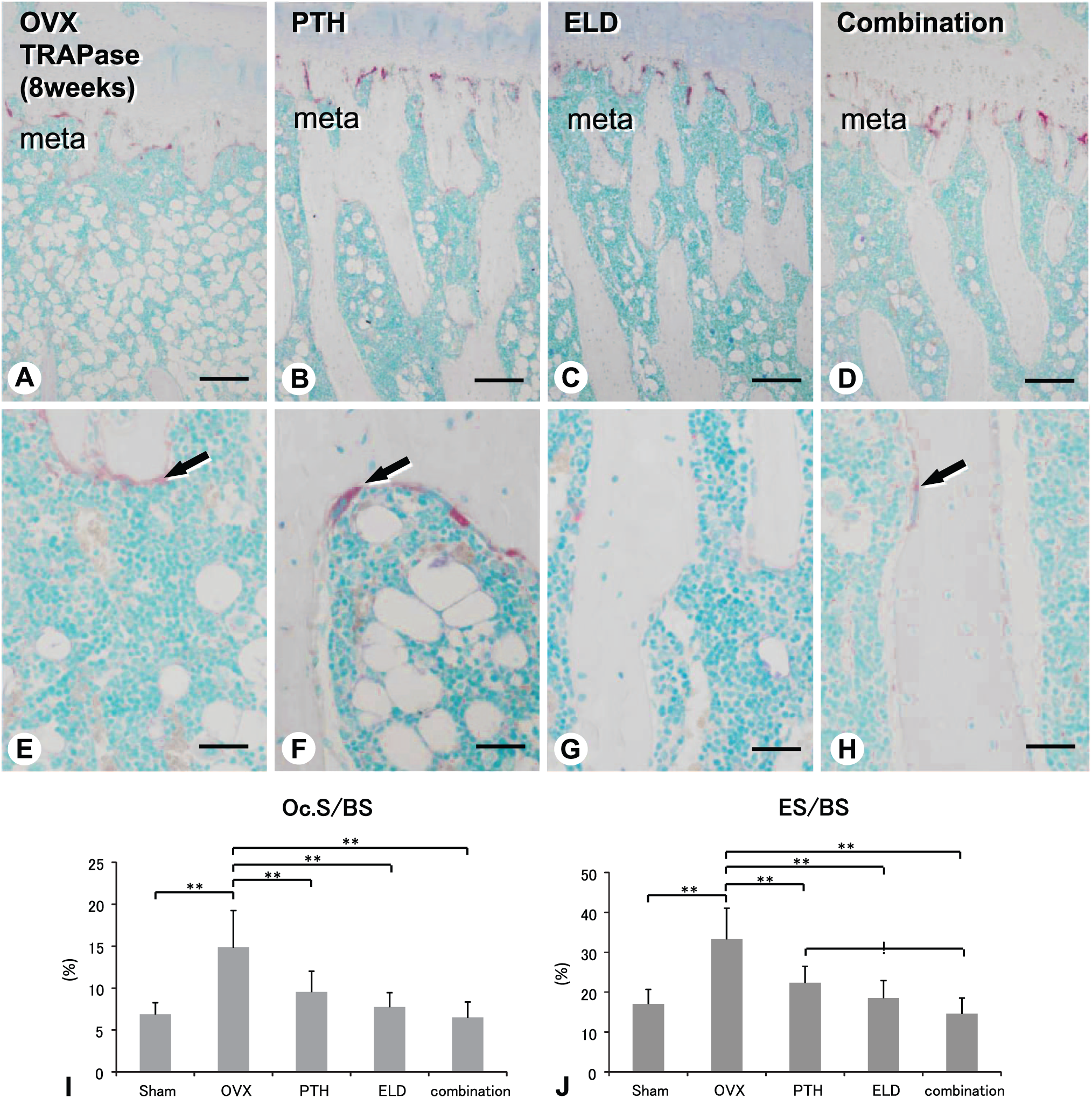
Distribution of TRAPase-positive osteoclasts and the index of Oc.S/BS and ES/BS in the tibiae of OVX, PTH, eldecalcitol, and combined groups (8 weeks). There are abundant TRAPase-positive osteoclasts in the tibial metaphysis of the OVX group compared with the other groups (B and F). The indices of Oc.S/BS and ES/BS are consistently highest in the OVX group (I and J). meta: metaphysis; Bars: A–D: 250 µm, E–H: 25 µm. Abbreviations: TRAPase, Tartrate-resistant acid phosphatase; Oc.S/BS, osteoclastic surface/bone surface; ES/BS, eroded surface/bone surface; OVX, ovariectomy; PTH, parathyroid hormone.
Histological Assessment of Osteoblasts and Osteoclasts in Tibial Metaphyseal Trabeculae Administered With PTH and Eldecalcitol
As calcein/tetracycline labeling demonstrated scalloped lines and smooth arrest lines after PTH and eldecalcitol administration, implying remodeling- and minimodeling-based bone formation, respectively, we examined von Kossa staining and double staining of ALPase/PCNA. Von Kossa staining verified the clear and smooth calcification front of the mineralized bone matrix in the eldecalcitol and combined groups, while it visualized a thicker osteoid and rough surfaces of calcification front in the PTH group (Fig. 7A–C). Many ALPase-reactive/PCNA-positive preosteoblasts were present in the PTH group but not in the eldecalcitol group (see Fig. 7D and E). In the eldecalcitol group, there was a thin layer of ALPase-positive flattened osteoblasts on the convex surface of trabecular bone (Fig. 7E). In the combined group, ALPase-reactive/PCNA-positive preosteoblasts were observed but not so many as were seen in the PTH group (Fig. 7F). Negative control experiments using normal rabbit serum or normal mouse serum did not show any nonspecific immunoreaction (data not shown).
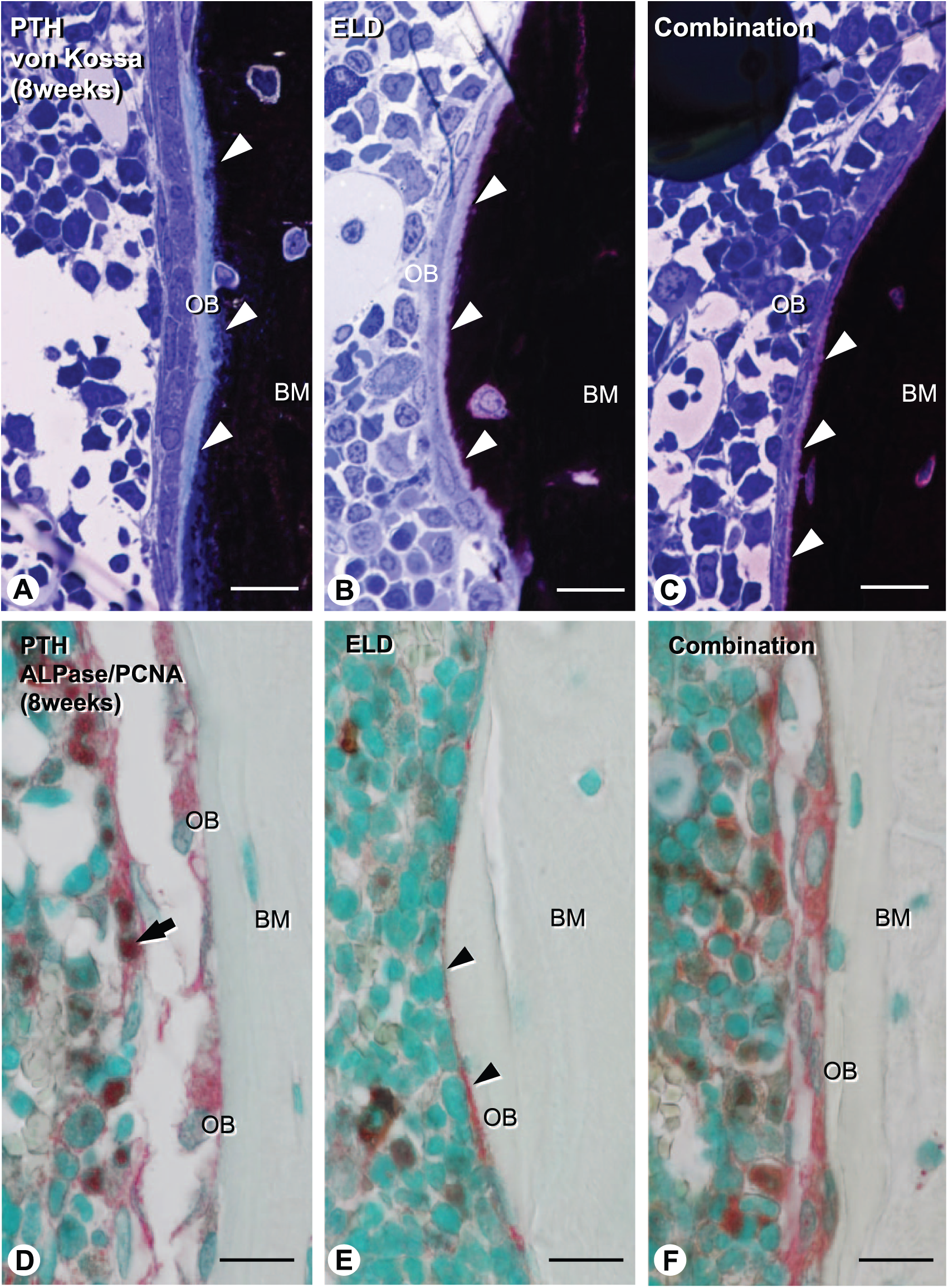
Histological analysis on osteoblastic cells and bone mineralization in the tibial metaphyseal trabeculae of the PTH, eldecalcitol, and combined groups (8 weeks). Von Kossa staining demonstrates the clear and smooth calcification front of the mineralized bone matrix in the eldecalcitol and combined groups (arrowheads, B and C), while it shows the rough surface of a mineralized bone matrix in the PTH group (arrowheads, A). ALPase and PCNA double-positive preosteoblasts are present in the both PTH and combined groups (arrow, D and F); however, not so many are seen in the combined group compared with the PTH group. In the eldecalcitol group, there is a thin layer of ALPase-positive osteoblasts on the bone surface (E). Bars: A–F: 10 µm. Abbreviations: PTH, parathyroid hormone; ALPase, alkaline phosphatase; PCNA, proliferating cell nuclear antigen; OB: osteoblast, BM: bone matrix.
Next, using a high resolution of light microscopy and TEM, we attempted to examine osteoblast and osteoclasts (Fig. 8). Consistent with ALPase/PCNA staining, the PTH group developed a thick layer of preosteoblasts overlying cuboidal mature osteoblasts (Fig. 8A and G), while the eldecalcitol group possessed a poorly developed layer of preosteoblasts and flattened osteoblasts (Fig. 8C and I). In contrast to osteoblastic cells, osteoclasts featured well-developed ruffled borders in the PTH group (Fig. 8B and H), while eldecalcitol-treated osteoclasts often lacked ruffled borders (Fig. 8D and J). The combined group showed a few preosteoblast layers (Fig. 8E and K), which were not as developed as that seen in the PTH group, as well as flattened osteoclasts (Fig. 8F and L).
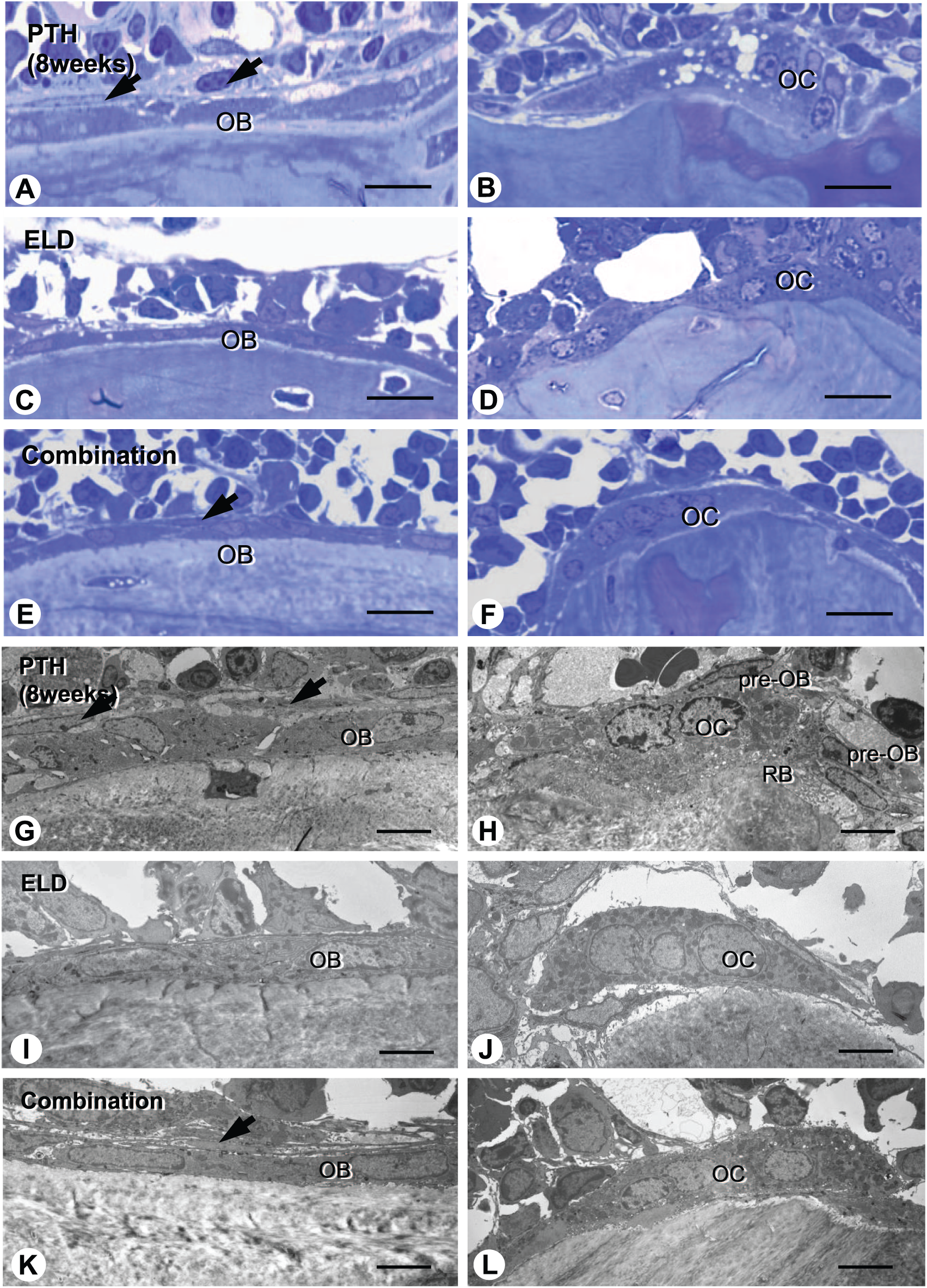
Histological assessment of osteoblast and osteoclast in the tibial metaphyseal trabeculae of the PTH, eldecalcitol, and combined groups (8 weeks). The PTH-group demonstrates cuboidal mature osteoblasts, a thick layer of preosteoblasts (arrows) and osteoclasts possessed well-developed ruffled borders on the bone surface (A, B, G, and H). By contrast, the eldecalcitol group shows flattened osteoblasts, a few preosteoblasts, and osteoclasts often lacking ruffled borders (C, F, I, and J). The combined group reveals mature osteoblasts, a few layers of preosteoblasts (arrow), and osteoclasts often lacking ruffled borders, such as in between the PTH and eldecalcitol groups (E, F, K, and L). Abbreviations: PTH, parathyroid hormone; OB, osteoblast; pre-OB, preosteoblast; OC, osteoclast; RB, ruffled border. Bars: A–F: 10 µm, G–I: 5 µm.
Discussion
We previously reported that the intermittent administration of hPTH(1-34) promoted preosteoblastic proliferation and osteoblastic bone formation mediated by cell coupling from osteoclasts.10,19 In contrast, we have also demonstrated that eldecalcitol induced minimodeling-based bone formation and suppressed bone resorption. 3 Thus, both reagents appear to have anabolic effects in bone. Therefore, in this study, we attempted to examine whether the combined administration of hPTH(1-34) and eldecalcitol may take advantage of additive anabolic effects on an osteoporotic animal model. Our main findings in this study can be summarized as follows:
The combined group showed markedly increased bone volume, trabecular thickness and number, and had the highest BMD of any of the groups.
OVX and PTH groups exhibited many ALPase-reactive osteoblasts, high histomorphometrical indices indicative of bone formation, and many TRAPase-positive osteoclasts, thereby, indicating a high bone turnover. By contrast, the eldecalcitol and combined groups tended to attenuate the parameters representing the number of osteoblasts and osteoclasts and eroded surfaces, implying a lower bone turnover.
The eldecalcitol-treated bone localized flattened osteoblasts, a few preosteoblasts, and osteoclasts that often lacked ruffled borders. By contrast, the hPTH(1-34)-administered group displayed many cuboidal osteoblasts and a thick layer of preosteoblasts, and many osteoclasts with well-developed ruffled borders.
Eldecalcitol administration induced focal, budding of bone formation, that is, minimodeling, while the PTH group demonstrated new bone with a scalloped shape of cement lines, indicating accelerated bone remodeling. The combined group revealed both histological profiles of minimodeling- and remodeling-based bone formation.
Thus, the PTH group increased bone resorption and bone formation, that is, accelerated bone turnover, but the PTH-driven anabolic activities appeared to be dominant to increase bone mass. By contrast, the combined group had relatively elevated anabolic indices of Ob.S/BS, MAR, and BFR/BS, but conversely, bone resorption activities, which might have been promoted by the catabolic action of hPTH(1-34), were much more attenuated. As a consequence, the combined group had the highest indices of BMD, BV.TV, and Tb.N, consistent with the histological finding of abundant metaphyseal trabeculae. It seems likely that the combined group would basically stimulate bone turnover, maintaining the anabolic effects derived from hPTH(1-34). Although accelerated bone turnover is supposed to lead to stimulated resorption activity, the eldecalcitol in the combined regimen may have diminished the resorption activity and also induced minimodeling-based bone formation.
It seems feasible that minimodeling-based bone formation does not disturb remodeling-based bone formation, because minimodeling-based bone formation is not involved in bone remodeling, that is, osteoclast-dependent cell coupling.4,5 Although there was no significant increase in BV/TV and Tb.Th in the combined group compared with the PTH group, it is of interest that the combined group revealed a significantly higher index of BMD rather than that in the PTH group at both 4 and 8 weeks (Figs. 1–3). We assume the possibility that the intermittent administration of hPTH(1-34) may accelerate bone turnover, somehow inducing an incomplete mineralization in bone 19 ; however, an inhibitory effect of eldecalcitol on bone resorption may at least slow down bone turnover, which could be seen in the indices of DPD/Cre, Oc.S/BS, ES/BS, MAR, and BFR/BS (Figs. 1, 4, and 5). If so, the combined regimen of eldecalcitol and hPTH(1-34) could form a well-mineralized bone matrix, as could be seen by von Kossa staining in this study (Fig. 7). Thus, we expect that the combined administration of hPTH(1-34) and eldecalcitol may not only induce more bone mass but also create a compact profile and good new bone quality, which could be resistant against mechanical loading and bone fracture.
Regarding the hPTH(1-34) regimen, daily and once-weekly administration of hPTH(1-34) or teriparatide are approved regimens for osteoporotic treatments in Japan. We have recently reported that a high-frequency of PTH administration seems to increase bone mass rapidly by forming abundant thin trabeculae. Alternatively, a low-frequency PTH administration leads to the formation of thicker trabeculae through bone remodeling and minimodeling. Our study can be confirmed by an investigation of biopsies taken from postmenopausal women treated with daily hPTH(1-34): Ma et al. 30 verified modeling-based bone formation, that is, minimodeling, even though it accounted for only 2.8% of the new bone formation in cancellous bone. Taking this into account, our regimen of hPTH(1-34) administration into OVX rats in this study appears to be closer to the high-frequency model of our previous study. Therefore, in this study, hPTH(1-34) administration may cause minimodeling-based bone formation, but it would be immediately replaced by an accelerated bone turnover.
In terms of eldecalcitol effects on osteoporotic treatments, clinically, a 3-year randomized, double-blind, active-comparator, clinical trial of eldecalcitol in postmenopausal women with osteoporosis demonstrated that eldecalcitol significantly decreased the incidence of vertebral fractures and wrist fractures, increased both lumbar spine and hip BMD, and suppressed bone turnover markers in comparison with alfacalcidol (1α-hydroxyvitamin D3).31,32 A post hoc analysis revealed that, compared with alfacalcidol, eldecalcitol reduced the incidence of vertebral fractures by approximately 50% in osteoporotic patients with prevalent fractures. 33 However, all of these studies were based on the inhibitory effects of eldecalcitol. More recently, minimodeling-based bone formation was found when microscopically examining human specimens administered eldecalcitol but not with bisphosphonates. 6 However, the measurement of anabolic serum markers may often fail to predict the occurrence of minimodeling, as shown in serum OCN in this study. It may be hard to detect an increment of anabolic serum markers induced by minimodeling, probably because minimodeling takes place in focal areas on the bone surface, and the population of preosteoblasts, which could produce anabolic markers, is limited. Therefore, the proper way to detect minimodeling may be by microscopic observations.
In conclusion, the combination of eldecalcitol and hPTH(1-34) augments their anabolic effects by means of minimodeling and remodeling. The inhibitory effects of eldecalcitol on bone resorption appear to reduce stimulated osteoclastic bone resorption that could be caused by PTH administration.
Footnotes
Acknowledgements
We thank Ms. Mai Haraguchi for her invaluable technical assistance in this study.
Competing Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Chugai Pharmaceutical Co., Ltd provided funding and supplied the test drugs for this study. S.S., S.T., and K.E. are employees of Chugai Pharmaceutical Co., LTD.
Author Contributions
TH and TY are the main researchers who contributed to this work, including preparation of paraffin sections and histochemical analyses. SS and ST performed animal experiments including fixation of animals and bone histomorphometry; TY conducted statistical analysis on body histomorphometry and serum concentration of OCN, Ca, Pi, and urinary DPD (deoxypyridinoline); YM, HH, ZQ, and MA assisted in preparation of paraffin sections; KO provided antitissue nonspecific alkaline phosphatase; PHLDF, ML, and NA participated in the discussion and preparation of the manuscript; and TH is a chief of this research project, organizing collaborators and providing a whole idea of this experiment. All the above authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by grants from Japanese Society for the Promotion of Science (JSPS, 15H05010, 15K20356), Promoting International Joint Research (Bilateral Collaborations) of JSPS and National Natural Science Foundation of China (NSFC) (Amizuka N, Li M) and the Founding from Chugai Pharmaceutical Co., LTD.
