Abstract
Summary
Viral infection of the insulin-producing cells in the pancreas has been proposed in the etiology of type 1 diabetes. Protein kinase R (PKR) is a cytoplasmic protein activated through phosphorylation in response to cellular stress and particularly viral infection. As PKR expression in pancreatic beta-cells has been interpreted as a viral footprint, this cross-sectional study aimed at characterizing the PKR expression in non-diabetic human pancreases. PKR expression was evaluated in pancreas tissue from 16 non-diabetic organ donors, using immunohistochemistry, qPCR, and western blot. Immunohistochemistry and western blot showed readily detectable PKR expression in the pancreatic parenchyma. The qPCR detected PKR mRNA in both endocrine and exocrine samples, with a slightly higher expression in the islets. In conclusion, PKR is constitutively expressed in both endocrine and exocrine parts of the pancreas and its expression should not be interpreted as a viral footprint in pancreatic beta cells.
Introduction
Viral infection of the insulin-producing cells remains implicated as a possible environmental trigger of type 1 diabetes (T1D).1–3 Protein kinase R (PKR), encoded by EIF2AK2, is a 68 kDa4,5 ribosome-associated serine/threonine kinase and a central mediator of the antiviral effects of interferons (IFN). 6 Upon binding dsRNA, PKR dimerizes and is activated by intermolecular autophosphorylation. 7 Activated PKR phosphorylates the α-subunit of eukaryotic initiation factor 2 (eIF2), which inhibits translation of most proteins. 8 PKR is also involved in apoptosis, primarily by inhibiting translation and possibly also by stimulating NF-κB signaling.9,10
Although EIF2AK2 is an IFN-stimulated gene, there is also a basal expression in various human tissues (GTEx portal V7. https://www.gtexportal.org. dbGaP Accession phs000424.v7.p2) and cell lines.11,12 The significance of the basal transcription is unknown. Likely, basally expressed PKR is in a latent state. Little is known about its function under physiological conditions. However, PKR has been found to modulate STAT1 through a mechanism independent of its kinase function 13 and has been identified as a mediator in signal transduction involving the growth factor PDGF (platelet-derived growth factor). 14 Elevated expression levels of EIF2AK2 and other IFN-stimulated genes have been found in laser-captured islets from subjects with recent onset T1D 15 and may suggest that type I IFNs play a role in the disease development.
A role for enteroviral infections in the etiology of T1D has previously been communicated based on immunohistochemical examinations of pancreatic biopsies from autopsy cases and organ donors.2,16 Dual immunofluorescent staining showed that islet cells positive for enterovirus capsid protein 1 (VP1) always coproduced PKR and that no PKR expression was detected in VP1-negative cells. 16 The selective overlapping expression of VP1 and PKR was interpreted as evidence of enteroviral infection.
The aim of this study was to investigate the basal expression of PKR in the human pancreas. Endocrine and exocrine pancreatic tissue from non-diabetic donors were studied through immunohistochemistry (IHC), western blot, and qPCR. Establishing the prevalence of PKR in normal tissue is an essential step in understanding the innate antiviral mechanisms in the pancreas. To further evaluate the interpretation of PKR expression as a viral footprint, islets infected with enterovirus were analyzed.
Materials and Methods
Human Pancreatic Samples
Pancreatic tissue samples from 16 multiorgan donors procured within the Nordic Network for Clinical Islet Transplantation were included in the study. Pancreatic biopsies were taken before islet isolation, formalin-fixed, and paraffin embedded (FFPE). The donors were non-obese and without known pancreatic disease. HbA1c and other known donor characteristics are described in ESM Table S1. Consent for organ donation (for clinical transplantation and for use in research) was obtained verbally from the deceased’s next of kin by the attending physician and documented in the medical records of the deceased in accordance with Swedish law and as approved by the Regional Ethics Committee (Dnr 2015/444). Human isolated islets, FFPE 3 days after infection with an enterovirus (Coxsackievirus B5) in a previous study 17 were included as a control for IHC.
RNA and Protein Extraction
Pancreatic tissue was harvested according to a previously described islet isolation and culture method. 18 Using a micropipette under a light microscope, samples of 100 islets or 100 exocrine cell clusters were handpicked from each donor. Islets were picked from fractions with high, and exocrine cell clusters from fractions with low, endocrine purity (as determined after the isolation process). RNA and protein were simultaneously extracted from harvested cells using an AllPrep DNA/RNA/Protein Mini kit (QIAGEN, Hilden, Germany), according to the manufacturer’s instructions with the following optional steps. The lysates were homogenized using QIAshredder spin columns (QIAGEN) in 2 mL collection tubes. After the final buffer RPE wash, the RNeasy spin column was transferred to a new 2 ml collection tube and centrifuged before the final step of adding RNase-free water.
cDNA-Synthesis and qPCR
The cDNA was synthesized from RNA using Invitrogen SuperScript VILO cDNA Synthesis Kit (Life Technologies, Carlsbad, CA) according to the manufacturer’s instructions. Real-time qPCRs were run with Quantifast SYBR PCR kit on a QuantStudio 7 Flex Real-Time PCR system. Predesigned gene-specific primer sets (QuantiTect Primer Assays, Qiagen) were used for detection of INS (Hs_INS_2_SG), GCG (Hs_GCG_1_SG), AMY2A (Hs_AMY2A_2_SG), EIF2AK2 (Hs_EIF2AK2_1_SG), GAPDH (Hs_GAPDH_2_SG), and 18S (Hs_RRN18S_1_SG) according to the manufacturer’s instructions. Each 20 μL reaction was run in duplicate and the veracity of the targets was confirmed by melt curve analysis. Relative expressions of mRNA were calculated by the 2−∆Ct method. The Ct average of reference genes GAPDH and 18S was used for normalization.
Validation of Sample Purity
For each donor, the successful handpicking of endocrine and exocrine cell clusters was validated based on the expression of endocrine (INS and GCG) and exocrine (AMY2A) genes. Donors were excluded from western blot and further qPCR analysis if the ratio of endocrine to exocrine genes was <1000 times higher in the islet samples than in the exocrine samples.
Western Blot
Extracted proteins were separated through SDS-PAGE (Mini-PROTEAN TGX gels, Bio-Rad, Hercules, CA) at 130V for 1 hr, using tris-glycine (TG)-buffer. Transferring to polyvinylidene difluoride (PVDF) membranes (Bio-Rad) was done at 100V for 1 hr using TG-SDS buffer. The membranes were cut at 50 kDa and incubated over night with the following antibodies: PKR, Abcam ab32506 diluted 1:5000 in 5% w/v milk-TBST; B-actin, 13E5 rabbit mAb diluted 1:2000. B-actin was used as a loading control. Secondary antibody (Goat antirabbit IgG, HRP-conjugated, Abcam Ab97051) was applied for 1 hr at room temperature. The antibodies were visualized using Clarity Western enhanced chemiluminescence (ECL) substrate (Bio-Rad). Images were acquired using Chemidoc MP Imaging system and ImageLab 4.1 software.
Immunohistochemistry
FFPE pancreatic biopsies and infected isolated islets were sectioned (6 µm) and placed on glass slides. The sections were blocked using Peroxidase-Blocking solution (Dako) and 5% goat serum.
Heat-induced epitope retrieval was performed in Target Retrieval Solution (Dako, Carpinteria, CA) at pH 9 (unphosphorylated PKR) or pH 6 (phospho-PKR, Vp1). Monoclonal rabbit anti-PKR antibody (Abcam, ab32506) diluted 1:1000 was applied for 1 hr in room temperature. Monoclonal rabbit anti-PKR phospho T446 antibody (Abcam, ab32036) diluted 1:50 was applied for 30 min in room temperature. Monoclonal mouse anti-Vp1 antibody (Dako, clone 5D-8.1) diluted 1:2000 was applied for 60 min in room temperature. Secondary antibody, EnVision+System-HRP labeled polymer antirabbit (Dako, K4002) or anti-mouse (Dako, K4001), was applied for 30 min. Liquid DAB+Substrate chromogen system (Dako) was applied for 4 min (PKR), 8 min (phospho-PKR), or 3 min (Vp1). The sections were counterstained with hematoxylin. Cover glasses were mounted using Pertex glue. Simultaneous isotype control stainings were used as controls for primary antibody specificity (Abcam, ab172730 and ab18415, respectively).
Statistical Analysis
Data from qPCR were analyzed through Wilcoxon matched-pair signed-rank test, using GraphPad Prism 6.0 software.
Results
PKR Is Constitutively Expressed on the Messenger Level in Non-infected Endocrine and Exocrine Pancreatic Tissue
A basal level of PKR mRNA (EIF2AK2) expression was found in all examined donors in both endocrine and exocrine pancreatic tissue. The relative expression varied between endocrine and exocrine samples from the same donor, with a slightly higher expression in endocrine samples (p=0.0313; Fig. 1).
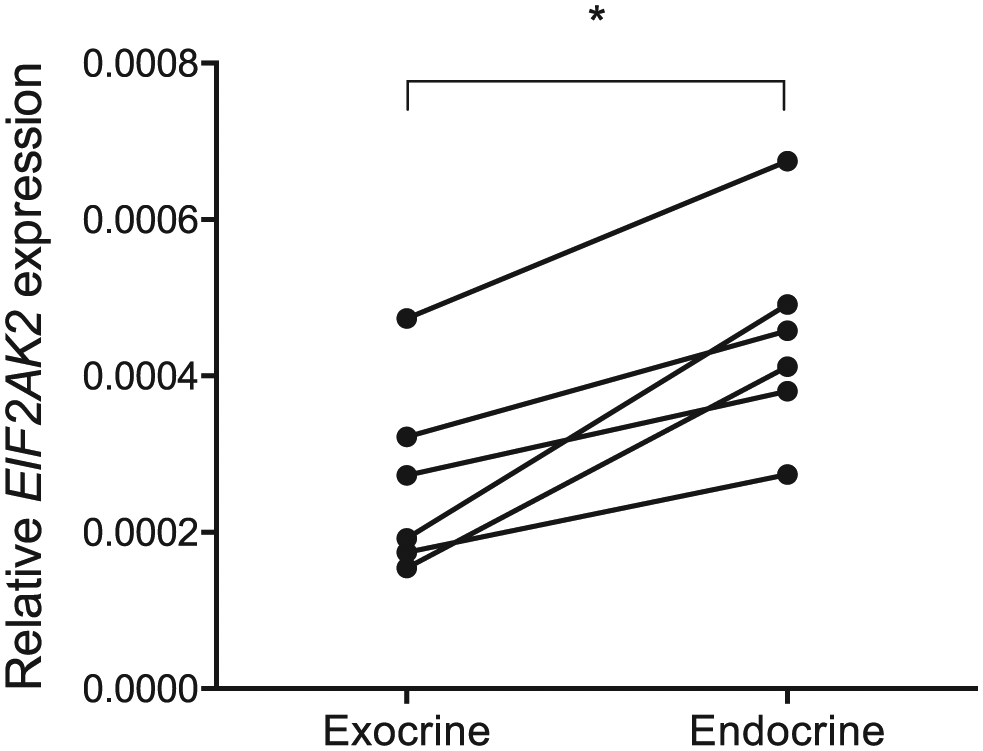
Expression of PKR mRNA (EIF2AK2) in isolated exocrine and endocrine pancreatic tissue from six different donors. The expression relative to the reference genes GAPDH and 18S is shown and samples from the same donor are connected with a line. Abbreviation: PKR, Protein kinase R.
PKR Is Constitutively Expressed on the Protein Level in Non-infected Endocrine and Exocrine Pancreatic Tissue
IHC showed moderate PKR staining of the cytosol in a majority of all cells in islets and in the exocrine parenchyma. PKR staining in islets was usually with increased intensity when compared with the exocrine pancreas (Fig. 2). PKR staining intensity varied among donors (Fig. 2A–D). Connective tissue, fibroblasts, and vessels were PKR negative. Occasional large, intensely PKR positive ganglion cells were found within nerve bundles (Fig. 2E). Staining using a commercial antibody developed to specifically label active phosphorylated PKR gave an almost identical staining pattern as did the antibody specific for PKR regardless of phosphorylation status. None of the organ donor sections stained positive for enterovirus protein (Vp1). All isotype controls were negative.
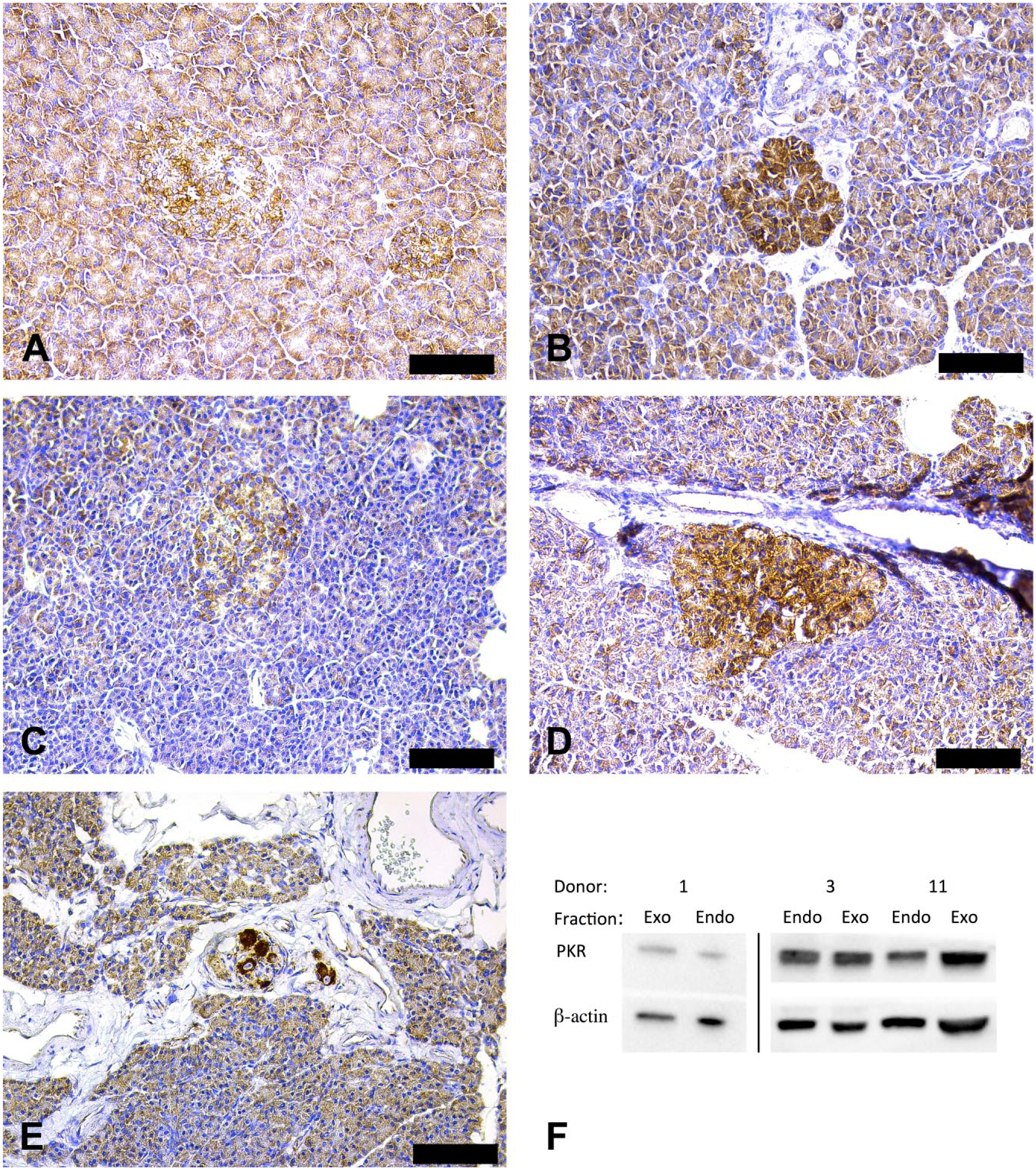
Expression and localization of PKR protein in exocrine and endocrine pancreatic tissue. Pancreatic tissue was stained for PKR (brown) and counterstained with hematoxylin (A–E). Representative images of PKR-stained islets and exocrine tissue from four different donors are shown (A; donor 2, B; donor 3, C; donor 5, D; donor 11). Ganglion cells, distinctly positive for PKR, could be seen (E; donor 8). Western blot of isolated tissue confirmed PKR expression in both islets (Endo) and exocrine cell clusters (Exo) from three different donors (1, 3, and 11) (F). Please note that the samples from donor 1 were run on a separate blot and that samples from donor 3 and 11 were run together. Scale bars 100 μm. Abbreviation: PKR, Protein kinase R.
Western blot analysis of PKR expression in isolated islets and exocrine cell clusters revealed distinct bands at ~68 kDa, consistent with the molecular weight of PKR. No apparent differences in intensity were seen between islets and exocrine tissue from the same donor (Fig. 2F).
PKR Is Constitutively Expressed in Infected and Non-infected Cells of Isolated Human Islets
Islets infected with enterovirus in vitro contained several cells that stained distinctly for enterovirus Vp1 (Fig. 3A). PKR staining of a consecutive section revealed positive staining of most cells regardless of VP1 positivity (Fig. 3B).
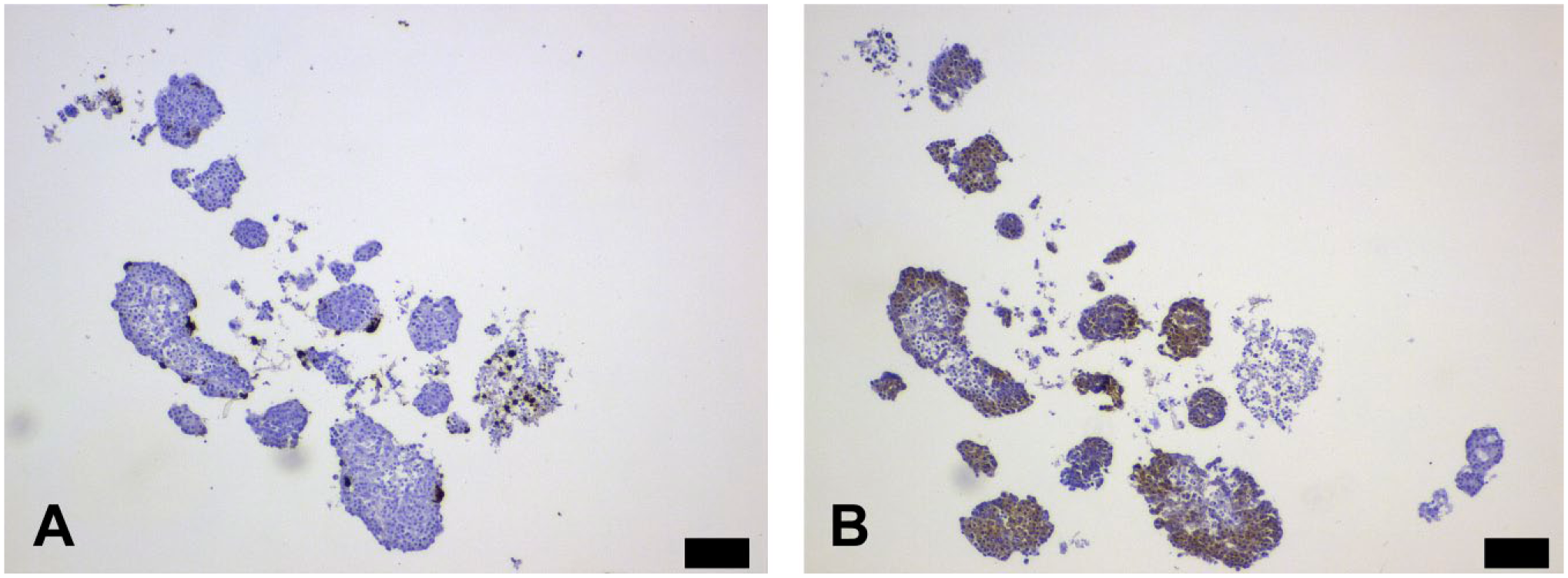
Infected islets stained for Vp1 (A) and PKR (B). Consecutive sections. Scale bars 100 μm. Abbreviation: PKR, Protein kinase R.
Discussion
In the present study, we demonstrate that PKR is constitutively expressed in high levels in the human pancreatic parenchyma. This is demonstrated on both the mRNA and the protein level. Expression of the messenger for PKR is slightly increased in islet cells when compared with the acinar cells. This finding is corroborated by the increased intensity of the IHC staining in islets, although this technique is not quantitative. In vitro infected islets display PKR staining in most cells, regardless of Vp1 positivity. The fact that no increased PKR staining intensity was seen in cells positive for virus protein argues against the merits of PKR as a suitable indicator of viral infection.
Pancreatic tissue from a total of 16 organ donors from the Nordic network for clinical islet transplantation was studied through qPCR, Western Blot, and IHC. Studying every pancreas through all three methods was unfortunately not possible (ESM Table S1).Unfortunately, pancreatic material from living human donors is scarce. Tissue intended for organ donation is handled with great care but still subject to abnormal conditions, such as those during the process of dying and tissue extraction, before being available for study. In addition, the isolated cells used for western blot and qPCR were cultured for 1 to 3 days before being available for research, which could arguably alter gene expression. From our study, we cannot with certainty conclude that PKR is constitutively expressed under physiological conditions in the human pancreas. However, the consistency of results within the same donor in different methods suggests that our findings reflect a common feature in the studied material. Furthermore, the above limitations apply to most studies on human pancreatic material, and certainly have implications for the use of PKR as a viral footprint.
The objective of this study was to investigate the prevalence of PKR in pancreases from non-diabetic donors. Three different analysis methods confirm constitutive expression of PKR in both the endocrine and exocrine pancreas. Thus, the detection of PKR should not be interpreted as a viral footprint. It is important to note that the methods used herein, as well as in previous studies of pancreatic tissue, do not distinguish between the inactive and the active, phosphorylated form of PKR. Successful and specific staining for phosphorylated PKR would be very informative and potentially useful as a marker of an activated antiviral response. In this study, we tried using a commercially available antibody against the phosphorylated variant of PKR. However, reliable phospho-specific IHC on tissue sections is notoriously difficult 19 and our results suggest that this antibody detected also the non-phosphorylated variant of PKR. Because IHC is not a quantitative method, the constitutive expression of PKR in non-infected pancreatic cells, demonstrated in this study, precludes the use of PKR expression as a marker for infection. An immunoblot approach could likely give information on the phosphorylation status of PKR when enough fresh or frozen tissue is available. Still, reliable methods to detect only active phosphorylated PKR in tissue sections need to be developed to determine the activation status of this viral sensor in various pancreatic cell types.
In conclusion, we demonstrate that PKR is constitutively expressed in the exocrine and endocrine pancreas. The physiological role of this latent expression is not known but the relatively higher PKR expression in islets compared with in the exocrine tissue is in line with a high antiviral defense mechanism in islet tissue.
Supplemental Material
DS_10.1369_0022155418802838 – Supplemental material for Protein Kinase R Is Constitutively Expressed in the Human Pancreas
Supplemental material, DS_10.1369_0022155418802838 for Protein Kinase R Is Constitutively Expressed in the Human Pancreas by Alexander Jonsson, Erik Yngve, Marie Karlsson, Sofie Ingvast, Oskar Skog and Olle Korsgren in Journal of Histochemistry & Cytochemistry
Footnotes
Acknowledgements
The authors acknowledge everyone in the Nordic Network for Clinical Islet Transplantation involved in the procurement of pancreases and islets.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AJ and EY executed the study with technical support from MK and SI and drafted the manuscript. AJ, EY, OS, and OK designed the study and performed the data analyses. All authors contributed to writing and/or approved the final version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Swedish Medical Research Council (65X-12219-15-6, K2015-54X-12219-19-4), the Novo Nordic Insulin Fund, the Åke Wiberg foundation, the Tore Nilsson Foundation, The Magnus Bergvall fund, the Ernfors Family Fund, Barndiabetesfonden, the Swedish Diabetes Association, the Diabetes Wellness Foundation Sweden (junior grant to O.S. 720-747 JDWG), and the Juvenile Diabetes Research Foundation International.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
