Abstract
Lipoxygenases (LOXs) are enzymes likely to be involved in corneocyte lipid envelope formation and skin barrier function. In humans, mutations in epidermis-type lipoxygenase 3 (eLOX-3) and 12R–lipoxygenase (12R-LOX) genes are associated with autosomal recessive congenital ichthyosis (ARCI), whereas deletion of these genes in mice causes epidermal defects. LOXs also represent a matter of interest in psoriasis as well as in cancer research. However, their expression as well as the exact role of these enzymes in normal human skin have not been fully described. Our goal was to characterize the expression of epidermal LOXs in both normal human skin and Tissue-Engineered Skin Substitutes (TESS) and to consider TESS as a potential model for LOX functional studies. Staining for epidermal differentiation markers and LOXs was performed, in parallel, on normal human skin and TESS. Our results showed similar expression profiles in TESS when compared with native skin for e-LOX3, 12R-LOX, 12S-lipoxygenase (12S-LOX), and 15-lipoxygenase 2 (15-LOX-2) but not for 15-lipoxygenase 1 (15-LOX-1). Because of their appropriate epidermal differentiation and LOX expression, TESS represent an alternative model for future studies on LOX function.
Keywords
Introduction
The lipoxygenase (LOX) family contains enzymes catalyzing dioxygenation or, for epidermis-type lipoxygenase 3 (eLOX-3), hydroperoxide isomerization of polyunsaturated fatty acids (for a review see Brash 1 ). Five LOX mRNAs are known to be expressed in human epidermal cells: eLOX-3, 12R–lipoxygenase (12R-LOX), 12S-lipoxygenase (12S-LOX), 15-lipoxygenase 1 (15-LOX-1), and 15-lipoxygenase 2 (15-LOX-2).2–6 In the last few years, a lot of attention has been given to 12R-LOX and eLOX-3 as mutations in their genes were found to be a frequent cause of nonsyndromic autosomal recessive congenital ichthyosis (ARCI)7–9: a rare human skin disease encompassing a broad spectrum of clinical manifestations including notably the “collodion baby.” 10 A role for these LOXs in skin barrier function was confirmed in various models. In mice as well as in human reconstructed skin, the depletion of eLOX-3 or 12R-LOX increased skin permeability.11–15 In 12R-LOX-deficient mice, the recruitment of covalently bound ceramides into the corneocyte lipid envelope (CLE), a crucial structure involved in skin barrier function, 16 was reduced. 17 From these observations came models proposing the successive processing of ceramids by 12R-LOX and eLOX-3 as a prerequisite step for the recruitment of these lipid molecules to the CLE, thus promoting their participation in skin barrier function.13,17,18
In contrast, and unlike eLOX-3 and 12R-LOX, the role of other epidermal LOXs in the skin still needs to be clarified. Analyses in 12S-LOX-deficient mice did show a slight increase in basal epidermal water loss with no major changes in skin lipid composition thus suggesting a more ambiguous role for 12S-LOX in CLE formation and skin barrier function. 19 Whereas the function of 15-LOX-2 in human skin is obscure, 15-LOX-1 is known to promote appropriate wound healing in this tissue. 20 Among epidermal LOXs, 12S-LOX, 15-LOX-1, and 15-LOX-2 were associated with the regulation of cell proliferation and carcinogenesis (for a review see Kuhn et al. 21 ). Changes in the expression or the activity of 12S-LOX, 15-LOX-1, and 15-LOX-2 were also noted in psoriasis.22–24 However, the involvement of these LOXs in such diseases as well as their functions in human epidermis still need to be studied more extensively.
Mice have frequently been used as models for the understanding of LOX function in the skin. 25 However, their use in such studies presents some drawbacks. Indeed, previous works have reported that human and mouse LOX orthologs do not share exactly the same substrates, products or expression pattern, as is the case for eLOX-3 and 15-LOX-2.26,27 Furthermore, the 15-LOX-2 ortholog in mice, namely 8S-LOX, is not constitutively expressed in murine epidermis but is rather induced following skin treatment with phorbol ester, 28 thus introducing an additional challenge to the study of this enzyme in mice and encouraging the development and use of human models.
Before proceeding to LOX functional studies in human skin, a more extensive characterization of their expression in this tissue was required. Whereas LOXs have been widely characterized in mice,25,28–30 their expression in human skin still needs to be clarified. Indeed, eLOX-3, 12R-LOX, 12S-LOX, and 15-LOX-2 expression patterns have not been the subject of a systematic examination but have rather been reported in distinct and independent studies with some discrepancies among the results.22,31–33 As for 15-LOX-1, to our knowledge, no immunohistochemical data have been presented for this enzyme in human epidermis.
In this context, the goal of this study was to characterize LOX expression in both normal human skin and in Tissue-Engineered Skin Substitutes (TESS) produced in vitro, and to assess the potential of TESS as models for future studies on human LOX function. The expression profiles of five LOXs, namely, eLOX-3, 12R-LOX, 12S-LOX, 15-LOX-1, and 15-LOX-2, were assessed in both TESS and normal human skin. Our results show that these LOXs are similarly expressed in both tissues, except for 15-LOX-1, which is not detected in normal human skin but is widely expressed in TESS. We suggest TESS as an interesting model for future studies on LOX function as epidermal differentiation and LOX expression in TESS are similar to what is observed in native epidermis.
Materials and Methods
Skin Biopsies
This study was approved by the Ethical Review Board at the Centre Hospitalier Universitaire de Québec and conducted in conformity with the Declaration of Helsinki principles. After informed consent, healthy human skin specimens were obtained following foreskin, face-lift, or breast resection surgeries.
Cell Culture
Fibroblasts derived from the breast skin biopsy of a 21-year-old adult woman were isolated as previously described.34,35 After isolation, the cells were cultured in Dulbecco’s modified Eagle’s medium (Gibco, Burlington, Canada #12800) supplemented with 10% fetal calf serum (HyClone, Logan, Utah), 100 IU/mL penicillin (Sigma-Aldrich, St-Louis, Missouri, #P3032-100MV), and 25 mg/mL gentamicin (Galenova, Saint-Hyacinthe, Canada, #GE152-100) and kept in an 8% CO2 atmosphere at 37C with 95% relative humidity. Culture medium was changed three times per week. Human keratinocytes were isolated from a 7-day-old human foreskin and cultured as previously described. 34 Briefly, cells were cultured on a feeder layer of irradiated 3T3 mouse fibroblasts in Dulbecco’s modified Eagle’s medium with Ham’s F-12 medium in a 3:1 proportion (Gibco, #21700) supplemented with 24.3 mg/mL adenine (Sigma-Aldrich, #A2786), 5% Fetalclone II serum (Hyclone, Logan, Utah), 5 mg/mL insulin (Sigma-Aldrich, #I5500-16), 0.4 µg/mL hydrocortisone (Calbiochem, La Jolla, California, #386898), 1 nM cholera toxin (Sigma-Aldrich, #C8052), 10 ng/mL epidermal growth factor (Austral Biologicals, San Ramon, California, #GF-0108), 100 IU/mL penicillin and 25 mg/mL gentamicin. Fibroblasts and keratinocytes were selected from a cell bank for their capacity to respectively produce thick dermal sheets and well-differentiated epidermis.
Human Skin Produced by Tissue Engineering
Human TESS were produced in vitro using the self-assembly approach.36,37 Fibroblasts (passage 3) were allowed to grow in T25 culture dishes in the previously described medium supplemented with 50 µg/mL of ascorbic acid (Sigma-Aldrich, #A7631) during 28 days, thus allowing the production of extracellular matrix and the formation of dermal sheets. Then, two dermal sheets were peeled from the dishes, superimposed and allowed to fuse for 7 days in culture to form dermal tissues. Human keratinocytes (passage 3) were then seeded on top of these dermal tissues and cultured in immersion for 1 week in the previously described keratinocyte culture medium, thus promoting keratinocyte proliferation and adhesion to the tissue. To induce keratinocyte differentiation, tissues were placed at the air–liquid interface for 14 days and cultured in keratinocyte culture medium deprived of Epidermal Growth Factor and supplemented with 50 µg/mL of ascorbic acid. During the entire process, culture media were refreshed three times a week. Samples were harvested after 14 days of air-liquid interface culture.
Histological and Immunofluorescence Staining
Skin and TESS biopsies were fixed with fresh 4% paraformaldehyde (pH 7.4) and embedded in paraffin. Microtome sections were stained with Masson’s trichrome and photographed using AxioVision software (Carl Zeiss, Toronto, Canada).
For immunofluorescence studies, TESS and normal human skin samples from breast surgeries were embedded in Tissue-Tek OCT compound (Sakura Finetek, Torrance, California, #4583), frozen at −80C and cryosectioned at a thickness of 5 µm. For LOX detection, tissues were fixed at 37C for 30 minutes in a solution containing 80 mM PIPES (Sigma-Aldrich, #P1851) pH 6.9, 75 mM NaCl (Fisher scientific, Ottawa, Canada, #S271-3), 3 mM MgCl2 (Sigma-Aldrich, #M9272), 1 mM CaCl2 (Sigma-Aldrich, #C7902), 0.5% Triton X-100 (Bio-Rad, Hercules, California, #161-0407), 3% paraformaldehyde (Electron Microscopy Services, Hatfield, Pennsylvania #15710), and 0.5% glutaraldehyde (Canemco & Marivac, Lakefield, Canada, #O121). After three washes in PBS, sections were incubated in 1 mg/mL of sodium borohydride (Sigma-Aldrich, #45882) for 15 minutes at room temperature and washed again three times in PBS before incubation with primary antibodies. For keratin 16 and transglutaminase 1 staining, samples were fixed in 100% acetone at −20C for 10 minutes. All immunostainings were performed by incubating primary antibodies for a 45-minute period followed by three washes in PBS, a 30-minute incubation in a combination of a fluorophore–conjugated secondary antibody and Hoechst 33258 (Sigma-Aldrich, #B2883) nuclear stain followed by three washes in distilled water. Negative controls, with omission of the antibody during the first antibody incubation, were routinely performed. Samples were visualized and pictures acquired using Carl Zeiss (Oberkochen, Germany) Axioimager microscopes coupled with AxioCam ICc1 and AxioCam Hrm cameras and operated by AxioVision 4.8.2 software. For the same staining, pictures were taken using the same exposition time and levels adjusted equally for all samples using Adobe Photoshop CS2. The antibodies used for immunostaining are presented in Table 1.
List of the Antibodies Used for This Study Including Host, Supplier and Catalog Number Information.
Abbreviation: LOXs, lipoxygenases.
Results
Antibody Selection
As many different LOXs are expressed in human skin and that these enzymes are highly homologous (between 34% and 65% identity),6,38 antibody selection was carefully performed to avoid cross-detection of different LOX subtypes. Antibodies for eLOX-3, 12R-LOX, 12S-LOX, 15-LOX-1, and 15-LOX-2 were selected in a way to maximize staining specificity. As shown in Fig. 1, the recognition sites of eLOX-3, 12R-LOX, and 15-LOX-2 antibodies are located in areas showing no sequence identity with other epidermal LOXs. Recognition site for 15-LOX-1 antibody is located in an area showing only 53% identity with 12R and 12S-LOX. To further assess the specificity of the anti-15-LOX-1 antibody, sequence alignment starting with antibody’s immunogen sequence was performed using blastp suite (https://blast.ncbi.nlm.nih.gov). For this antibody, as well as for anti-eLOX-3, anti-12R-LOX, and anti-15-LOX-2 antibodies, no other LOXs or known proteins were significantly detected in sequence alignment results, supporting the choice of these antibodies (data not shown).
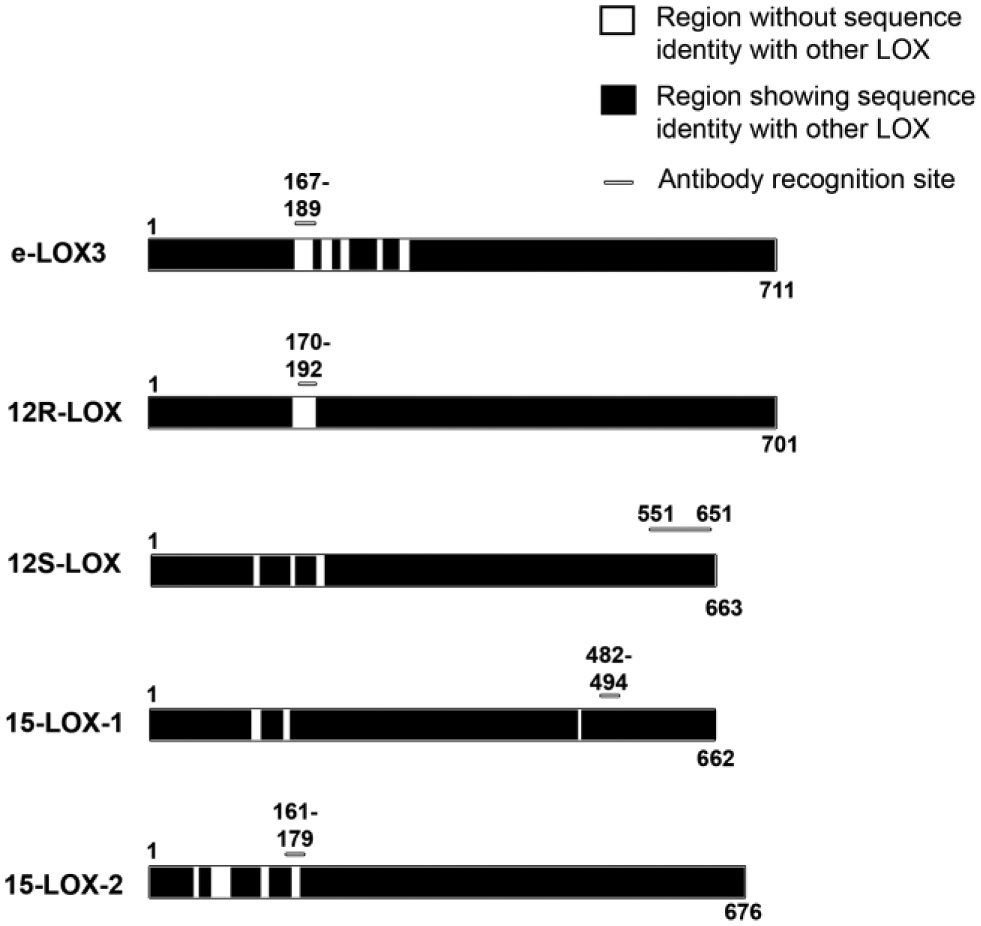
Antibody recognition sites and sequence identity between the different LOXs shown in this study. eLOX-3, 12R-LOX, 12S-LOX, 15-LOX-1, and 15-LOX-2 protein sequences are illustrated by long rectangles. Dark areas represent a protein section sharing sequence identity with another lipoxygenase of this study. Numbers indicate amino acid position (above) or protein length (below). Bars indicate the recognition site of the antibody used in this study. Abbreviation: LOXs, lipoxygenases.
As 12S-LOX is relatively similar to 15-LOX-1 (about 65% identity), 39 an antibody produced using a protein epitope signature tag (PrEST) was selected. PrEST s are protein fragments designed to have minimal sequence identity when compared with other human proteins and are thus, in this case, theoretically expected to uniquely detect 12S-LOX. 40 The antibodies selected for this study are described in Table 1.
Histology and Epidermal Differentiation of Normal Human Skin and Human Tissue-Engineered Skin Substitutes
Human TESS are models of epidermal differentiation expressing various epidermal markers (such as keratins 10 and 14) and cell adhesion proteins (such as laminin 5, β-catenin, desmoglein-1, and claudin 4), as previously characterized by our team.41,42 Masson’s trichrome staining of transversal sections of normal human skin and TESS shows proper epidermal cell cohesion and differentiation in both tissues. Normal human skin and TESS presented the four expected epidermal layers: the basal, spinous, granular, and cornified layers (Fig. 2A and B). The human TESS epidermis appears thicker, probably because of keratinocyte apparent increase in size in the reconstructed tissues. Larger cell size could be attributed to culture conditions (such as the addition of serum 43 ) or to tissue processing and preservation. To evaluate the epidermal differentiation process, the expression of transglutaminase 1, a well-known enzyme highly involved in cornified envelope formation and nonfunctional or mutated in some ARCI patients, 44 was evaluated in TESS and normal human skin. As shown in Fig. 2C and D, transglutaminase 1 was expressed in both tissues: starting from the granular layer in normal human skin and from the spinous layer in TESS. Earlier expression of transglutaminase 1 is commonly observed in various human skin models produced in vitro.14,45,46
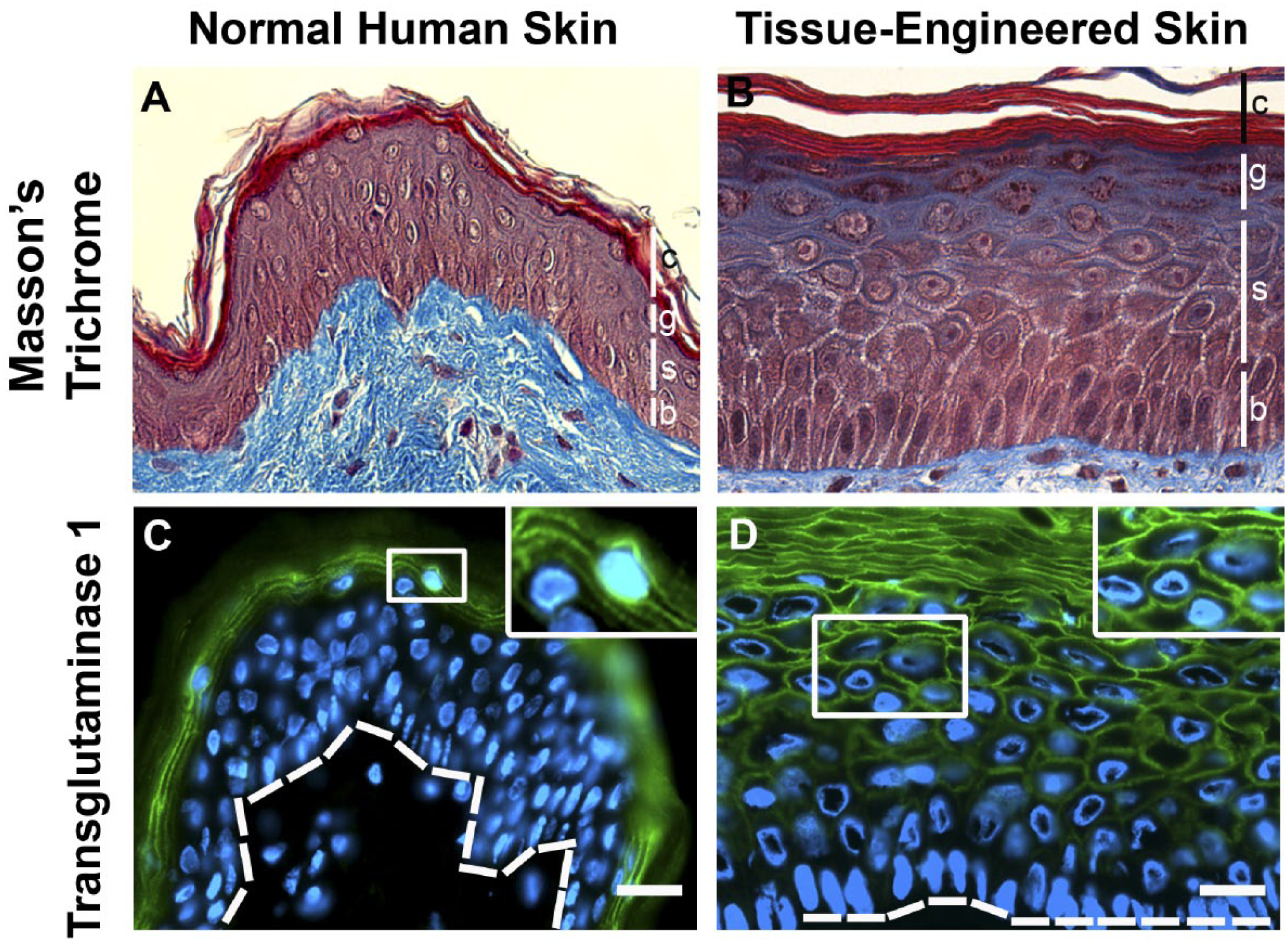
Epidermal cell differentiation in normal human skin and TESS. (A and B) Masson’s trichrome staining of normal human skin (A) and Tissue-Engineered Skin Substitutes (B). Epidermal layers are indicated by vertical bars. b: basal layer, s: spinous layer, g: granular layer and c: cornified layer. (C and D) Staining of normal human skin (C) and TESS (D) for transglutaminase 1 (green) and nuclei (blue). Magnification of the framed area is shown in the upper right corner of the corresponding picture. Dotted line underlines the basal layer. Scale bars: 25 µm. Abbreviation: TESS, Tissue-Engineered Skin Substitutes.
Similar Lipoxygenase Expression Profiles in Normal Human Skin and Human Tissue-Engineered Skin Substitutes
To characterize LOX expression in TESS, immunofluorescence staining was performed for eLOX-3 (Fig. 3A and B), 12R-LOX (Fig. 3C and D), 12S-LOX (Fig. 3E and F), and 15 LOX-2 (Fig. 3G and H) on both native and reconstructed tissues. As shown in Fig. 3, these LOXs were similarly expressed in TESS when compared with normal human skin. In both cases, these four LOXs were present at the periphery of the cells from the basal to the granular layers of the epidermis with, for all these LOXs, some intermittent staining observed near the nucleus.
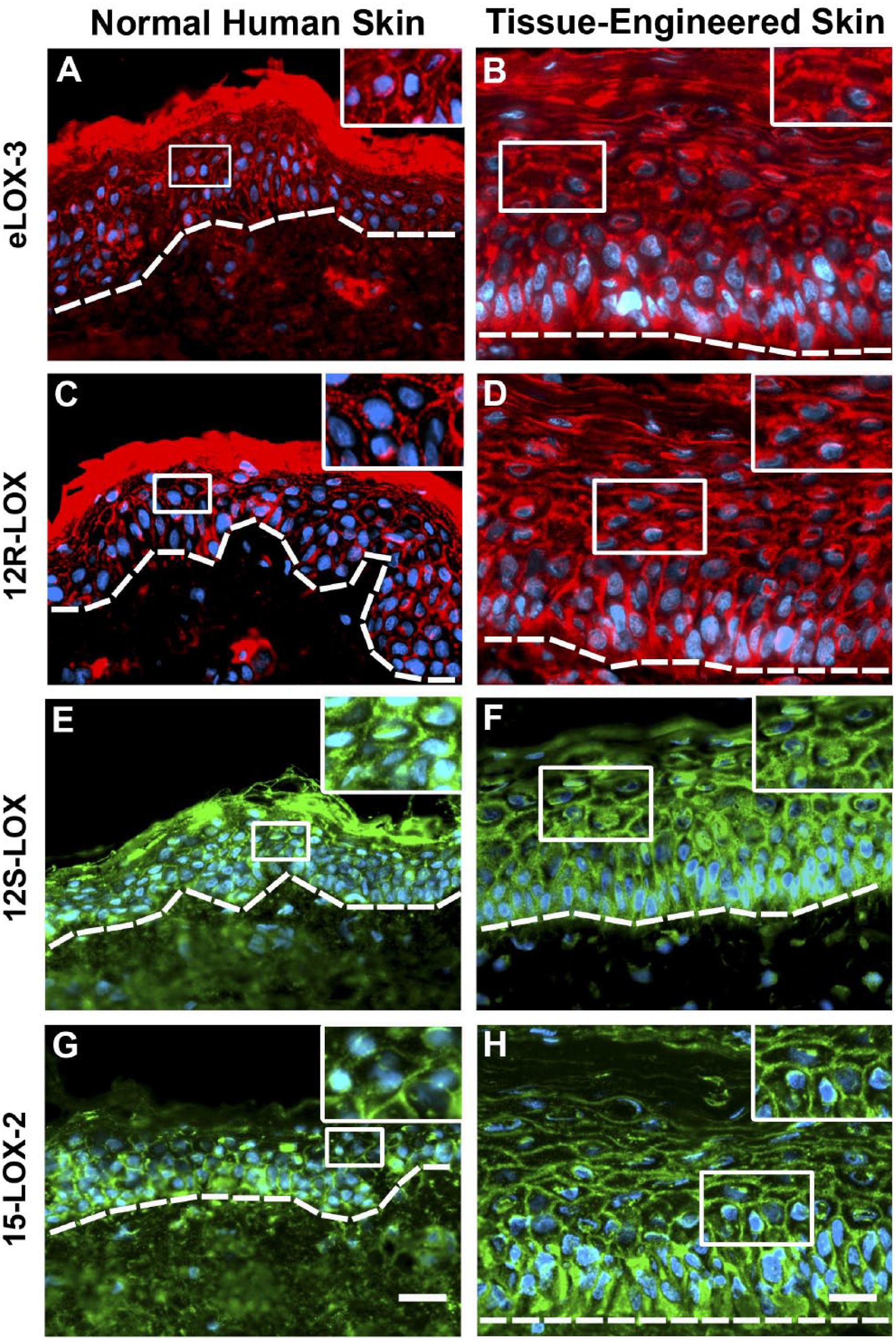
Immunofluorescence staining for eLOX-3 (red; A and B), 12R-LOX (red; C and D), 12S-LOX (green; E and F) and 15-LOX-2 (green; G and H) in normal human skin (A, C, E, and G) and Tissue-Engineered Skin Substitutes (B, D, F, and H). Magnification of the framed area is shown in the upper right corner of the corresponding picture. Nuclei are stained in blue. Dotted line underlines the basal layer. Scale bars: 25 µm. Abbreviation: LOXs, lipoxygenases.
15-LOX-1 Is Expressed in Tissue-Engineered Skin Substitutes But Not in Normal Human Skin
As 15-LOX-1 mRNA was previously detected in keratinocytes in culture, 5 the expression of this LOX was investigated in TESS and normal human skin using immunofluorescence techniques. As seen in Fig. 4A, 15-LOX-1 was not observed in normal human skin. However, 15-LOX-1 was strongly expressed at the periphery of TESS epidermal cells from the basal to the granular layer, thus revealing the induction of 15-LOX-1 expression in this model (Fig. 4B). Once again, perinuclear staining was observed within epidermal cells. As 15-LOX-1 was reported to be associated with wound healing in epithelial cells of the cornea, 47 we investigated the expression of a well-known marker of this process within the skin, namely keratin 16. 48 As expected, keratin 16 was absent in normal human skin but highly present in all suprabasal layers of the epidermis in TESS (Fig. 4C and D).
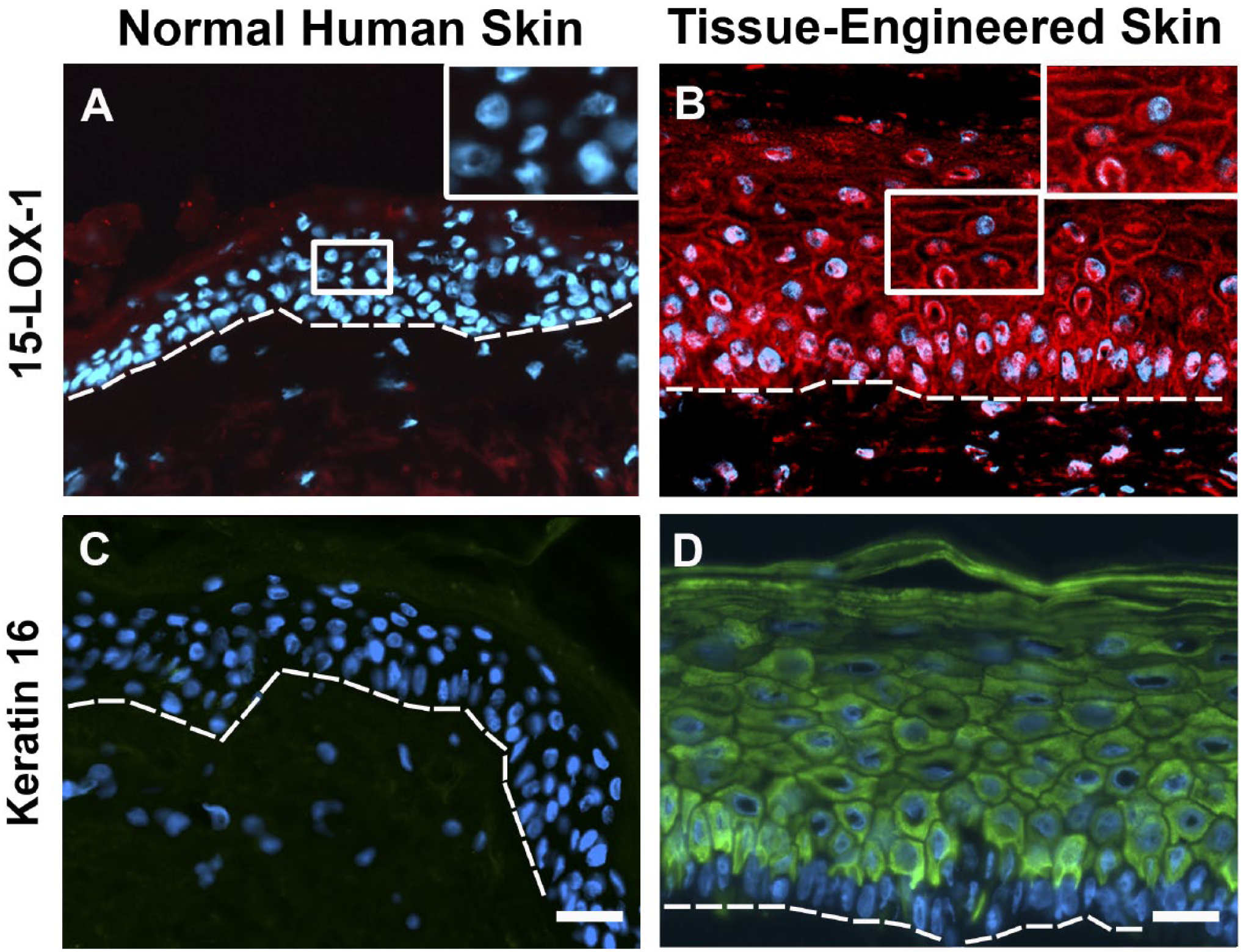
Immunofluorescence staining for 15-LOX-1 (red; A and B) and keratin 16 (red; C and D) in normal human skin (A and C) and Tissue-Engineered Skin Substitutes (TESS; B and D). Magnification of the framed area is shown in the upper right corner for A and B. Nuclei are stained in blue. Dotted line underlines the basal layer. Scale bars: 25 µm. Abbreviation: LOXs, lipoxygenases.
Discussion
Reports of mutations in LOX genes in patients suffering from ARCI as well as epidermal defects noted in 12R-LOX and eLOX-3-deficient mice and human reconstructed skin recently pointed out the importance of these LOXs in the establishment and maintenance of skin barrier function in mammals.7–9,11–14 However, the exact contribution of other epidermal LOXs to this process as well as to disorders such as cancer and psoriasis is relatively unknown. The aim of this study was to characterize LOX expression in a fully differentiated human epidermis model that could eventually be used for studies on LOX function.
This study brings more information about LOX expression in human skin models. The main finding of this report is that all detected LOXs were preferentially located at the cell periphery in epidermal cells of native and reconstructed human skin. Such a result is in agreement with previous reports showing a similar distribution for eLOX-314,31 and 12R-LOX 14 but not for 12S-LOX 22 and 15-LOX-2. 24 The distribution of LOXs at the periphery of epidermal cells is supported by various reports describing, in other cell types, the membrane association of some members of the LOX family. Indeed, early works found that 12-LOX enzymatic activity was mostly redistributed to the membrane fraction in platelets and erythroleukemia cells (HEL) upon cell activation.49,50 The translocation of 15-LOX-1 and 15-LOX-2 to membranes was also described in reticulocytes 51 and prostate epithelial cells. 52 Apparently, such a membrane association was not confirmed for 12R-LOX or eLOX-3. However, these proteins share, along with the other LOXs, an homologous ß-barrel-forming amino acid sequence, coined as the PLAT (for Polycystin-1, LOX, Alpha-Toxin) domain, 53 which is known to play a role in the membrane binding of mammalian 12S-LOX, 54 15-LOX-1, 55 and 15-LOX-2. 56 Interestingly, several lines of evidence suggest that the Ca2+ ion is involved in the regulation of membrane binding through the PLAT domain. Such a Ca2+-dependent membrane association was reported for 15-LOX-1 and 15-LOX-2 at concentrations as low as 10.4 µM.28,51,56,57 As epidermal cells from the basal, spinous, and granular layers are known to have an intracellular calcium content mostly ranging between 7 and 20 µM, 58 the cortical distribution of LOXs can be promoted, thus explaining their detection at the cell periphery in normal human skin and in TESS. In our samples, staining for all LOXs was also sometimes observed near the nucleus. Nuclear or perinuclear localization of LOXs is possible as some reports did describe interaction with intracellular or nuclear membranes for 12S-LOX, 59 15-LOX-1, 60 and 15-LOX-2. 61 Further analysis should therefore be performed to confirm such an interaction in differentiating epidermal cells.
The LOX expression reported herein, ranging from the basal to the granular layer, is consistent with what was previously observed in human native epidermis and/or reconstructed skin for eLOX-314,31 and 12R-LOX 14 but not in mice where these enzymes are restricted to the granular layer.11,13 Such a difference in eLOX-3 and 12R-LOX expression may be due to interspecies variations. It could seem surprising for eLOX-3 and 12R-LOX to be expressed early in keratinocyte differentiation when they play a role in CLE formation, a process considered as a late event and associated with the granular layer of the epidermis. However, appearance of some proteins involved in cornified envelope assembly, another late event in keratinocyte differentiation, also occurs before the formation of the granular layer of the epidermis even if the main part of this process is thought to take place within this layer. 62 Indeed, involucrin, also described as the scaffold protein responsible for ceramide attachment to the cornified envelope, 63 is expressed starting from the spinous layer of the epidermis. 64 In the context where eLOX-3 and 12R-LOX are considered responsible for ensuring esterified omega-hydroxyacyl-sphingosine ceramide oxidation, a prerequisite to ceramide recruitment to the CLE13,18 it is credible for these LOXs to be expressed before the proteins involved in later events of CLE formation.
As for the other LOXs, our results show some discrepancies with the previously reported distribution of 12S-LOX, 12R-LOX, and 15-LOX-2 in human skin. Indeed, these enzymes were reported not to be expressed in all the living epidermal layers and seldom in the plasma membrane zone.22,31,32 We believe that these discrepancies could be due to differences in fixation techniques used during tissue processing. Indeed, whereas other research teams used standard fixation agents such as acetone or formaldehyde, we rely on a mix of paraformaldehyde and glutaraldehyde known to minimize lipid extraction, thus maximizing the preservation of cell membrane integrity.65,66 As plasma membrane lipids act as an anchoring point for the binding of some PLAT domain-containing enzymes,28,67 dissolving these components with fixators such as acetone may lead to leaking of the antigen and thus to the loss of the signal from the original LOX localization.
Unlike other epidermal LOXs studied in this work, the 15-LOX-1 expression profile in TESS was different from the one observed in native tissue. Indeed, while 15-LOX-1 was absent in normal human skin, it was strongly expressed at the cell periphery throughout the TESS living epidermis. This evident difference between 15-LOX-1 and the staining of the remaining LOXs in native epidermis supports the anti-15-LOX-1 antibody specificity. As mentioned earlier, 15-LOX-1 activity and mRNA expression are also induced in cell monolayer culture but are not observed in native epidermis.5,68 15-LOX-1 is not the only protein showing different levels of expression in culture when compared with native tissues. Indeed, culture-induced expression is also observed for keratin 16 in every epithelial cell culture system in vitro such as classical monolayer culture,69,70 skin explants, 71 organotypic skin cultures,72,73 collagen-coated inserts (Leiden model), 74 and TESS. 75 Keratin 6, the binding partner of keratin 16 and also involved in the wound healing process, 76 is aberrantly expressed as well in commercially available reconstructed skin models such as Episkin, SkinEthic, and EpiDerm. 45 Keratin 16 is also expressed in TESS produced in serum-free culture media and produced with cells originating from the same donor thus pointing out the fact that keratin 16 expression in culture is not related with to the exposition to serum or to the mixing of cell populations. 75 Interestingly, keratin 16 and 15-LOX-1 are two proteins whose expression is known to be induced during wound healing.47,77,78 Expression of these proteins in vitro can be explained by the presence of required factors in the culture media that induce proliferation or wound healing processes. Once again, culture conditions, as well as tissue processing and preservation, could be factors explaining the apparent larger cell and nucleus size observed in TESS when compared with the native tissue. Despite the differences in cell size and keratin 16 expression, TESS present a permeability very similar to the one measured in normal human skin, 79 thus suggesting that the keratinocyte differentiation process in this model ultimately leads to the production of a skin barrier function similar to the one observed in vivo. As LOXs are known to be involved in skin barrier function, we believe that TESS represent an accurate model for the study of their function. However, such a difference in 15-LOX-1 and keratin 16 expression in TESS should be kept in mind in future studies on LOX function.
In summary, this study enabled the characterization of epidermal LOX expression in normal human skin as well as in TESS. The expression profiles of eLOX-3, 12R-LOX, 12S-LOX, and 15-LOX-2 were similar in both tissues whereas 15-LOX-1 expression was only observed in TESS. Despite this difference between native and reconstructed skin, our data and previous results from other teams 14 suggest TESS as an interesting model for the study of human LOXs. Indeed, TESS recapitulate LOX expression and epidermal cell differentiation, a process much more difficult to study using classic monolayer cell culture techniques and in which keratin 16 and 15-LOX-1 are also expressed. TESS would notably be relevant for the functional study of 15-LOX-2; a LOX that is not constitutively expressed in mouse epidermis. Expression of 15-LOX-1 in TESS should, however, be kept in mind. The existence of TESS models for psoriasis, 80 wound healing, 81 and cancer 82 also offers an opportunity to further analyze the function of these enzymes within these pathophysiological processes.
Footnotes
Acknowledgements
The authors thank Dr. Peter Krieg for kindly providing epidermis-type lipoxygenase 3 (eLOX-3) and 12R-lipoxygenase (12R-LOX) rabbit antibodies and Dr. Cindy J. Hayward for revising the manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
CS-B prepared the manuscript, designed and performed experiments. LAP designed and performed experiments and participated in manuscript revision. VJM and BFDL contributed to experiment design and manuscript revision.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Fondation du CHU de Québec and by the Quebec Cell, Tissue and Gene Therapy Network–ThéCell (a thematic network supported by the Fonds de recherche du Québec–Santé).
