Abstract
Ulcerative colitis (UC) is a chronic inflammatory disease histologically characterized by diffuse mononuclear cell infiltrates in colonic mucosa. These inflammatory cells are considered to be recruited via high endothelial venule (HEV)-like vessels displaying mucosal addressin cell adhesion molecule 1 (MAdCAM-1), the ligand for α4β7 integrin, and/or peripheral lymph node addressin (PNAd), an L-selectin ligand. 6-O-sulfation of N-acetylglucosamine in the carbohydrate moiety of PNAd is catalyzed exclusively by N-acetylglucosamine-6-O-sulfotransferase 1 (GlcNAc6ST-1) and GlcNAc6ST-2. To determine the role of 6-O-sulfation of N-acetylglucosamine on HEV-like vessels in UC, we used a chronic dextran sulfate sodium–induced colitis model using mice deficient in both GlcNAc6ST-1 and GlcNAc6ST-2. We found that more inflammatory cells, with expression of tumor necrosis factor α, were infiltrated in double knockout mouse colitis compared with that in wild-type mice. Moreover, the number of MAdCAM-1-positive vessels was increased in double knockout mouse colitis, and these vessels were bound by E-selectin•IgM chimeras that bind to unsulfated sialyl Lewis X (sLeX). These findings suggest that interactions between MAdCAM-1 and α4β7 integrin and/or unsulfated sLeX and L-selectin may become a dominant mechanism for inflammatory cell recruitment in the absence of 6-sulfo sLeX and contribute to more severe colitis phenotypes seen in double knockout mice.
Introduction
Ulcerative colitis (UC) is a chronic, relapsing inflammatory disorder affecting colonic mucosa that, together with Crohn’s disease, constitutes inflammatory bowel disease (IBD). Although its etiopathogenesis is not fully understood, UC is currently considered an abnormal inflammatory response to intestinal microbial flora with or without components of autoimmunity.1,2
In UC, diffuse mononuclear inflammatory cell infiltrates composed mainly of lymphocytes and plasma cells are almost universally present in the colonic lamina propria.3,4 It is widely accepted that these inflammatory cells use the same extravasation mechanisms operating in normal conditions, namely, lymphocyte homing in “physiological inflammation” seen in gut-associated lymphoid tissue (GALT), but in an exaggerated and uncontrolled manner. 5
Lymphocyte recruitment to a chronic inflammation site is mediated by a cascade of adhesive interactions between circulating lymphocytes and endothelial cells that comprise venules resembling high endothelial venules (HEVs),6,7 which are called HEV-like vessels. 8 At least one of two molecular interactions is required for the “rolling” step of lymphocyte recruitment: one via L-selectin and peripheral lymph node addressin (PNAd), and the other via α4β7 integrin and mucosal addressin cell adhesion molecule 1 (MAdCAM-1).9,10
PNAd describes a set of glycoproteins decorated with 6-sulfo sialyl Lewis X (sLeX)-capped O-glycans that are detected by the monoclonal antibody MECA-79.8,11 6-O-sulfation of N-acetylglucosamine (GlcNAc) in the sLeX structure is required for optimal recognition of PNAd by L-selectin. 12 Thus far, five GlcNAc-6-O-sulfotransferase (GlcNAc6ST) family members have been identified in humans and four orthologues in mice. 13 Among them, GlcNAc6ST-114,15 and GlcNAc6ST-216,17 are expressed not only in HEVs but also in HEV-like vessels13,18 and play a pivotal role in biosynthesis of PNAd carbohydrates on these vessels.
Gene expression of GlcNAc6ST-1 and GlcNAc6ST-219,20 as well as MAdCAM-1 21 22 in endothelial cells that constitute HEV-like vessels in GALT is reportedly upregulated by proinflammatory cytokines such as tumor necrosis factor α (TNF-α) and lymphotoxin α/β. Besides MAdCAM-1, other immunoglobulin superfamily cell adhesion molecules such as platelet endothelial cell adhesion molecule 1 (PECAM-1), which is commonly used as a marker of vascular endothelial cells, 23 are also expressed on HEV-like vessels. PECAM-1 is expressed on endothelial cells as well as platelets and leukocytes, and plays a pivotal role in leukocyte migration through vessels.24,25
Two authors of this article independently demonstrated that mice deficient in both GlcNAc6ST-1 26 and GlcNAc6ST-227,28 show a complete lack of MECA-79+ HEVs in secondary lymphoid organs, and that lymphocyte homing to peripheral lymph nodes and mesenteric lymph nodes is reduced to approximately 25% and 50%, respectively, relative to that seen in wild-type mice, while lymphocyte homing to Peyer’s patches is maintained.29,30 However, whether elimination of both enzymes would alter lymphocyte recruitment in gastrointestinal mucosal inflammation had not been determined.
One author of this article and his colleagues previously reported that GlcNAc6ST-2 catalyzes GlcNAc-6-O-sulfation of not only L-selectin ligands on HEVs but also mucins secreted from colonic epithelial cells. 31 The same group also demonstrated that after administration of dextran sulfate sodium (DSS), the number of leukocytes infiltrating the colonic lamina propria of GlcNAc6ST-2 single knockout mice was significantly greater than that seen in wild-type mice, suggesting that colonic mucins with GlcNAc6ST-2-mediated sulfated glycans have a protective function. 32 However, whether loss of GlcNAc6ST-1 and GlcNAc6ST-2 perturbs leukocyte recruitment in gastrointestinal mucosal inflammation remains to be investigated. Such an investigation might require analysis in a chronic model, which is more applicable to human UC, than the acute model in which mice die within a week.
Here, using a chronic DSS-induced animal colitis model33,34 established in GlcNAc6ST-1 and GlcNAc6ST-2 double knockout mice, we evaluated the consequences of deficiency in optimal L-selectin ligands, specifically, 6-sulfo sLeX-capped O-glycans. To this end, we undertook quantitative immunohistochemical analysis to assess colonic mucosal inflammation focusing particularly on MAdCAM-1+ HEV-like vessels.
Materials and Methods
Animals
Eight-week-old male mice deficient in both GlcNAc6ST-1 and GlcNAc6ST-229,30 (n=12) and their wild-type littermates (n=12) on a C57BL/6 background were used in this study. Mice were housed in specific pathogen-free (SPF) conditions (namely, negative for all the following microorganisms: Clostridium piliforme, Corynebacterium kutscheri, Mycoplasma pulmonis, Salmonella spp., ectromelia virus, lymphocytic choriomeningitis virus, mouse hepatitis virus, Sendai virus, ectoparasites, intestinal protozoa, and pinworm), fed an autoclaved chow diet (Oriental Yeast, Tokyo, Japan), and given ultraviolet-irradiated, filtered non-acidified tap water ad libitum at the Animal Facility of Hoshi University, in accordance with institutional guidelines.
Induction of Experimental Colitis
Experimental colitis resembling human UC was induced in mice doubly deficient in GlcNAc6ST-1 and GlcNAc6ST-2 (n=6, randomly assigned) as well as in control wild-type mice (n=6, randomly assigned) by including 2% DSS (MP Biomedicals, Santa Ana, CA) in drinking water for a week followed by drinking water without DSS for another week. This cycle was repeated three times (6 weeks in total) as described previously. 33 As controls, both groups of mice (n=6 each, randomly assigned) received drinking water without DSS throughout the 6-week experimental period. The experimental protocol was approved by the Animal Research Committee of Hoshi University (reference number 26-138, approved on June 5, 2015).
Histological Evaluation of Experimental Colitis
At the end of the 6-week experimental period, mice were sacrificed by cervical dislocation and the entire colon was collected and divided evenly into three parts: proximal (cecal side), middle, and distal (anal side) parts. Samples were fixed in 20% buffered formalin, embedded in paraffin, and sections of 3-μm thickness were stained with hematoxylin and eosin (HE). Serial tissue sections were used for immunohistochemistry with a battery of antibodies to be hereinafter described. Based on the grading system proposed by Wirtz et al., 35 we evaluated four properties of colitis, including (1) ulceration, (2) polymorphonuclear cell infiltration of the lamina propria, (3) cryptitis, and (4) crypt abscess, semiquantitatively: Factors 1 and 2 were graded from 0 to 3, and factors 3 and 4 from 0 to 2 (Table 1). This assessment was done in a blinded fashion. The resultant colitis score, which is the sum of the four subscores, ranged from 0 to 10.
The Colitis Scoring System Used in This Study.
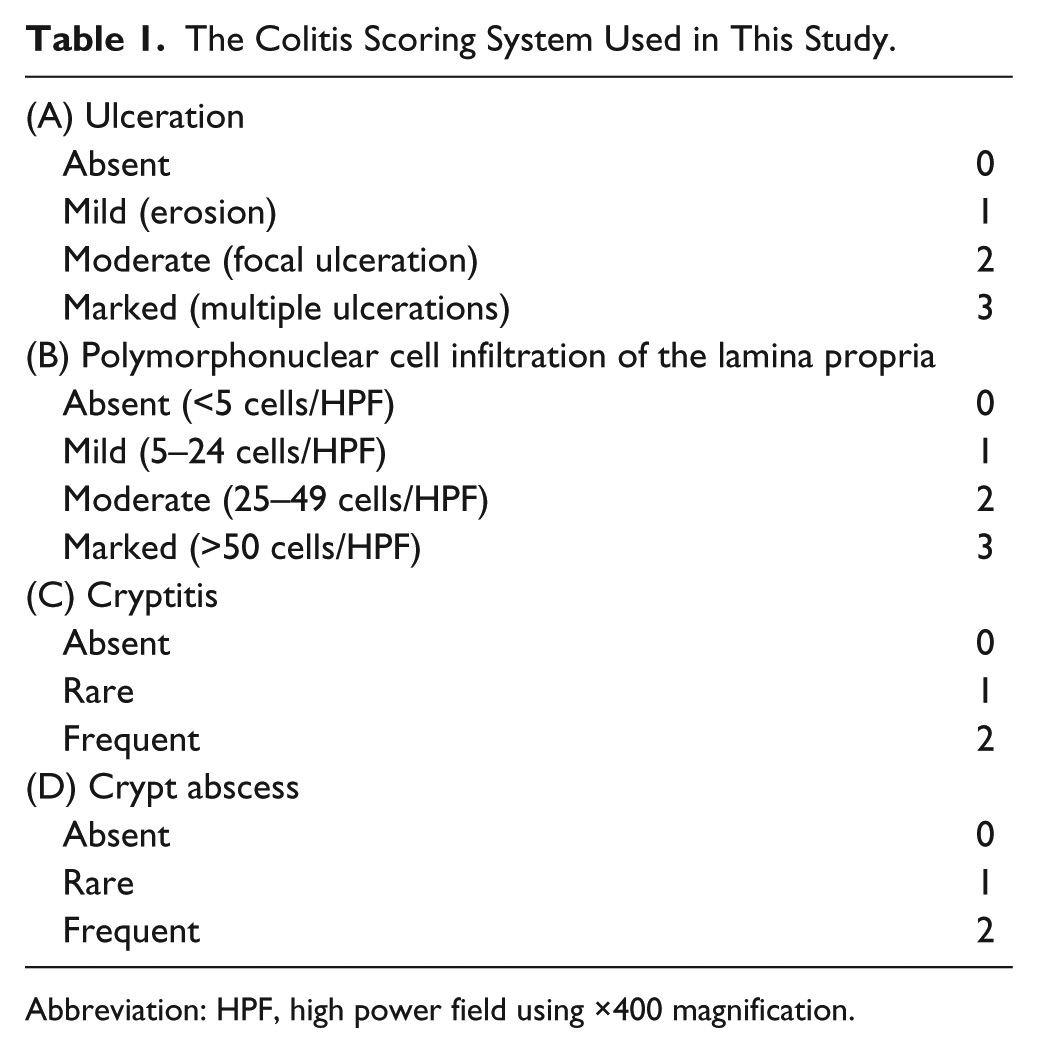
Abbreviation: HPF, high power field using ×400 magnification.
Immunohistochemistry
The following antibodies served as primary antibodies: MECA-79 (rat IgM; BD Pharmingen, San Diego, CA) recognizing 6-sulfo N-acetyllactosamine attached to extended core 1 O-glycans, which overlaps with 6-sulfo sLeX, a recognition determinant for L-selectin11,13,36; MECA-367 (rat IgG; BD Pharmingen) recognizing mouse MAdCAM-1; SZ31 (rat IgG; Dianova GmbH, Hamburg, Germany) recognizing mouse PECAM-1 (also known as CD31), a vascular endothelial cell marker; and rabbit anti-TNF-α polyclonal antibody (Abcam, Cambridge, MA). In addition, #15-8G-61 (mouse IgG) recognizing human MAdCAM-1 37 and JC70A (mouse IgG; Leica Biosystems, Newcastle Upon Tyne, UK) recognizing human PECAM-1 (CD31) were used for human samples. In mice, immunohistochemistry was conducted using the labeled streptavidin–biotin (LSAB) method, as described 38 ; human samples were prepared as described. 39 Tissue sections of mesenteric lymph nodes and/or GALT, which harbor HEVs that express PECAM-1 as well as MAdCAM-1 and PNAd, served as positive controls. Negative controls were obtained by replacing primary antibodies with isotype-matched immunoglobulins. Use of human UC tissues was approved by the Ethics Committee of the Faculty of Medical Sciences, University of Fukui (reference number 20160082, approved on November 2, 2016).
Quantification of HEV-Like Vessels
To quantitate MECA-79+ and MAdCAM-1+ HEV-like vessels, we determined MECA-79+/PECAM-1+ and MAdCAM-1+/PECAM-1+ vessel ratios, as PECAM-1 (CD31) proteins are universally expressed on vascular endothelial cells, including those that constitute HEV-like vessels.23,40 To calculate ratios, the number of MECA-79+, MAdCAM-1+, or PECAM-1+ vessels in the entire colonic mucosa of each animal specimen was determined under a microscope at ×200 magnification without knowledge of colitis status, and then the number of MECA-79+ or MAdCAM-1+ vessels was divided by the number of PECAM-1+ vessels, yielding the MECA-79+/PECAM-1+ or MAdCAM-1+/PECAM-1+ vessel ratios, respectively.
E-Selectin•IgM Chimera Binding Assay
E-selectin•IgM chimeric proteins were obtained from the culture medium of COS-1 cells transiently transfected with pcDNA1.1-E-selectin•IgM. The E-selectin•IgM chimera in situ binding assay was conducted as described. 23
Statistical Analysis
Differences between groups were statistically analyzed using Student’s t-test with GraphPad Prism 5 software (GraphPad Software, La Jolla, CA). Values of p<0.05 were considered significant.
Results
Successful Establishment of Experimental Colitis
As shown in Fig. 1, without DSS administration, both wild-type mice and mice doubly deficient in GlcNAc6ST-1 and GlcNAc6ST-2 were healthy and did not show histological findings indicative of colonic mucosal injury or inflammation. However, following DSS administration, both mutant and wild-type mice demonstrated signs of active colitis, including reduced mobility, a hunched back, diarrhea, and bloody stool. On histological examination, colonic walls of DSS-administered mice of both genotypes were substantially thickened due to hyperplasia of crypt-forming epithelial cells and thickening of proper muscle layers. In addition to cryptitis and crypt abscess, ulcerative lesions (Fig. 1, low magnification view, right panels, arrowheads) were found chiefly from the distal to middle part of the colon, and to a much lesser extent in the proximal part. It is noteworthy that a proportion of the above-mentioned hyperplastic epithelial cells were highly dysplastic (Fig. 1, high magnification view, right lower panel), a change that likely emerges from continuous exposure to inflammatory stimuli and one observed in long-standing human UC, from which colon cancers develop. Taken together, these findings indicate that our DSS administration protocol successfully established mouse experimental colitis resembling human UC.
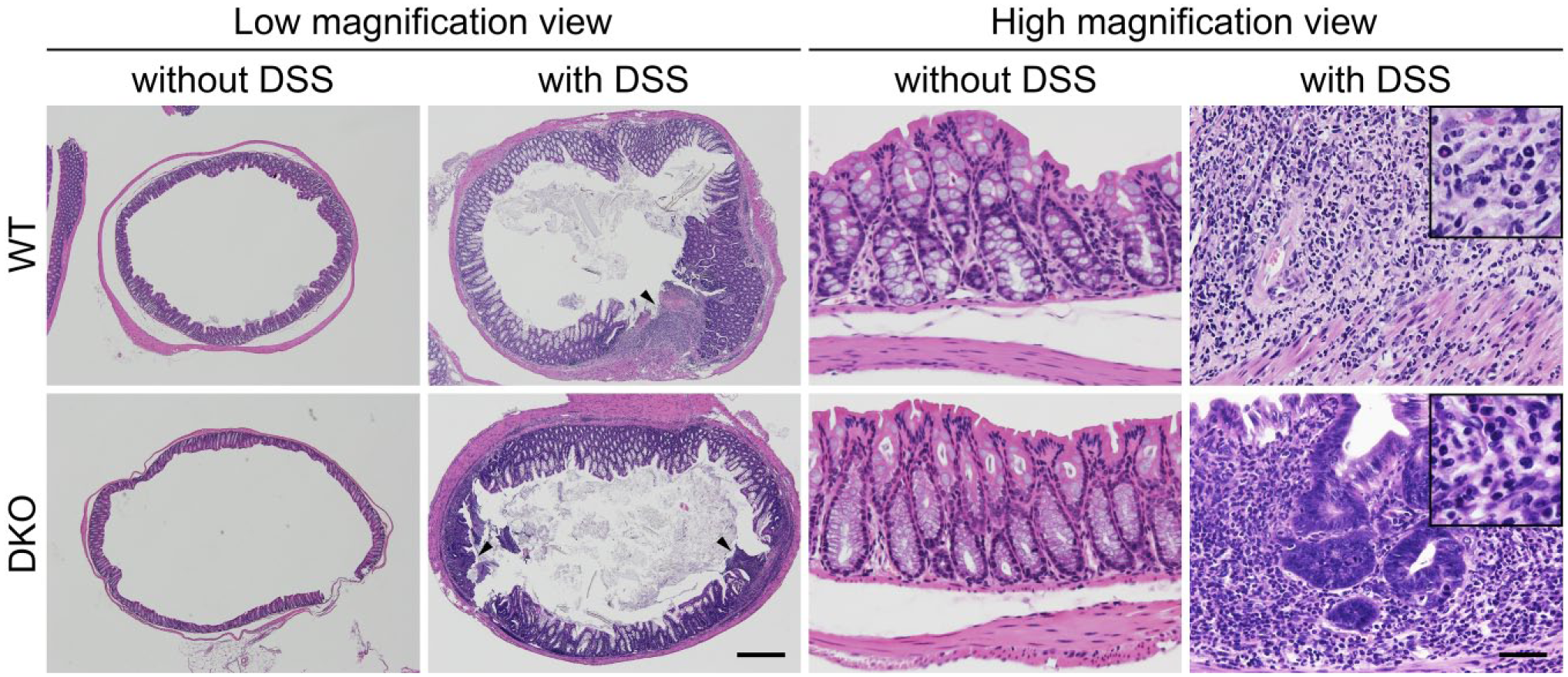
Representative histology of DSS-induced chronic mouse colitis. Without DSS administration, colonic mucosae are normal in both WT mice and GlcNAc6ST-1 and GlcNAc6ST-2 DKO mice. Following DSS administration, colonic walls are thickened due to hyperplasia of crypts and thickening of proper muscle layers. Arrowheads show ulcers formed after administration. In and around ulcerative lesions, inflammatory cell infiltrates composed of polymorphonuclear and mononuclear cells are observed (see insets in high magnification view). Note that a proportion of crypt-forming epithelial cells show severe dysplasia (right lower panel). Hematoxylin and eosin staining. Bar = 1000 μm for low magnification view, 50 μm for high magnification view, and 20 μm for insets in high magnification view. Abbreviations: DSS, dextran sulfate sodium; WT, wild type; GlcNAc6ST, N-acetylglucosamine-6-O-sulfotransferase; DKO, double knockout.
Mice Deficient in GlcNAc6ST-1 and GlcNAc6ST-2 Develop More Severe Colitis
We then evaluated colonic inflammatory changes histologically and determined a colitis score for double knockout and wild-type mice. Although ulcerative lesions were observed following DSS administration in both genotypes, multiple ulcerative lesions were more frequently observed in double knockout compared with wild-type mice (Fig. 1, low magnification view, right panels, arrowheads). Moreover, the degree of polymorphonuclear cell infiltration of the colonic lamina propria of double knockout mice was greater than that seen in wild-type mice (Fig. 1, high magnification view, right panels and Fig. 2, left panel). Consequently, the total colitis score was greater in DSS-administered double knockout (7.33 ± 1.37) compared with wild-type (4.0 ± 1.27) mice with high statistical significance (Fig. 2, right panel). We note, however, that the degree of mononuclear cell infiltration of the colonic lamina propria was comparable between genotypes. Besides inflammatory cell infiltrates, we also examined tissue distribution of TNF-α proteins in the two groups of mouse colitis. As shown in Fig. 3, in both double knockout and wild-type colitis mice, signals for TNF-α proteins were detected around inflammatory cell aggregates formed in the colonic lamina propria. In addition, in double knockout mice, signals for TNF-α proteins were also detected beneath colonic epithelial cell layers (Fig. 3, right panels).
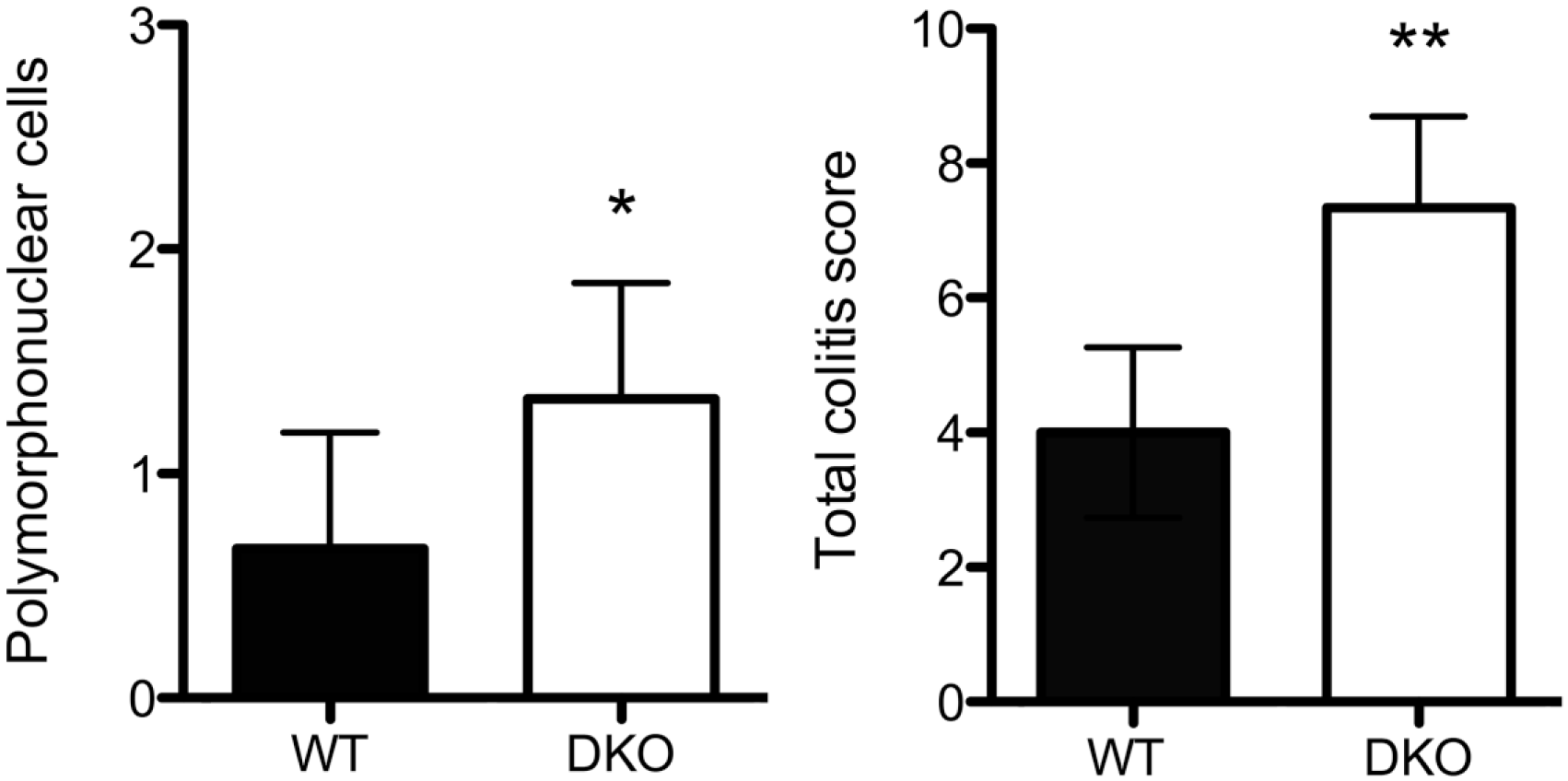
The degree of polymorphonuclear cell infiltration (left) and total colitis score (right) in chronic DSS-induced colitis in WT mice and GlcNAc6ST-1 and GlcNAc6ST-2 DKO mice. Data are presented as means with standard deviation. *p<0.05, **p<0.01. Abbreviations: DSS, dextran sulfate sodium; WT, wild type; GlcNAc6ST, N-acetylglucosamine-6-O-sulfotransferase; DKO, double knockout.
Tissue distribution of TNF-α proteins in DSS-induced colitis in WT and GlcNAc6ST-1 and GlcNAc6ST-2 DKO mice. Images in lower panels are enlarged from boxes in upper panels. Inset in lower right panel shows accumulation of TNF-α proteins beneath colonic epithelial cell layer. Signals were visualized with 3,3′-diaminobenzidine (brown), and tissues were counterstained with hematoxylin. Bar = 100 μm for upper panels and 50 μm for lower panels (including inset). Abbreviations: TNF-α, tumor necrosis factor α; DSS, dextran sulfate sodium; WT, wild type; GlcNAc6ST, N-acetylglucosamine-6-O-sulfotransferase; DKO, double knockout.
Mice Deficient in GlcNAc6ST-1 and GlcNAc6ST-2 Show Increased Numbers of MAdCAM-1+ Vessels
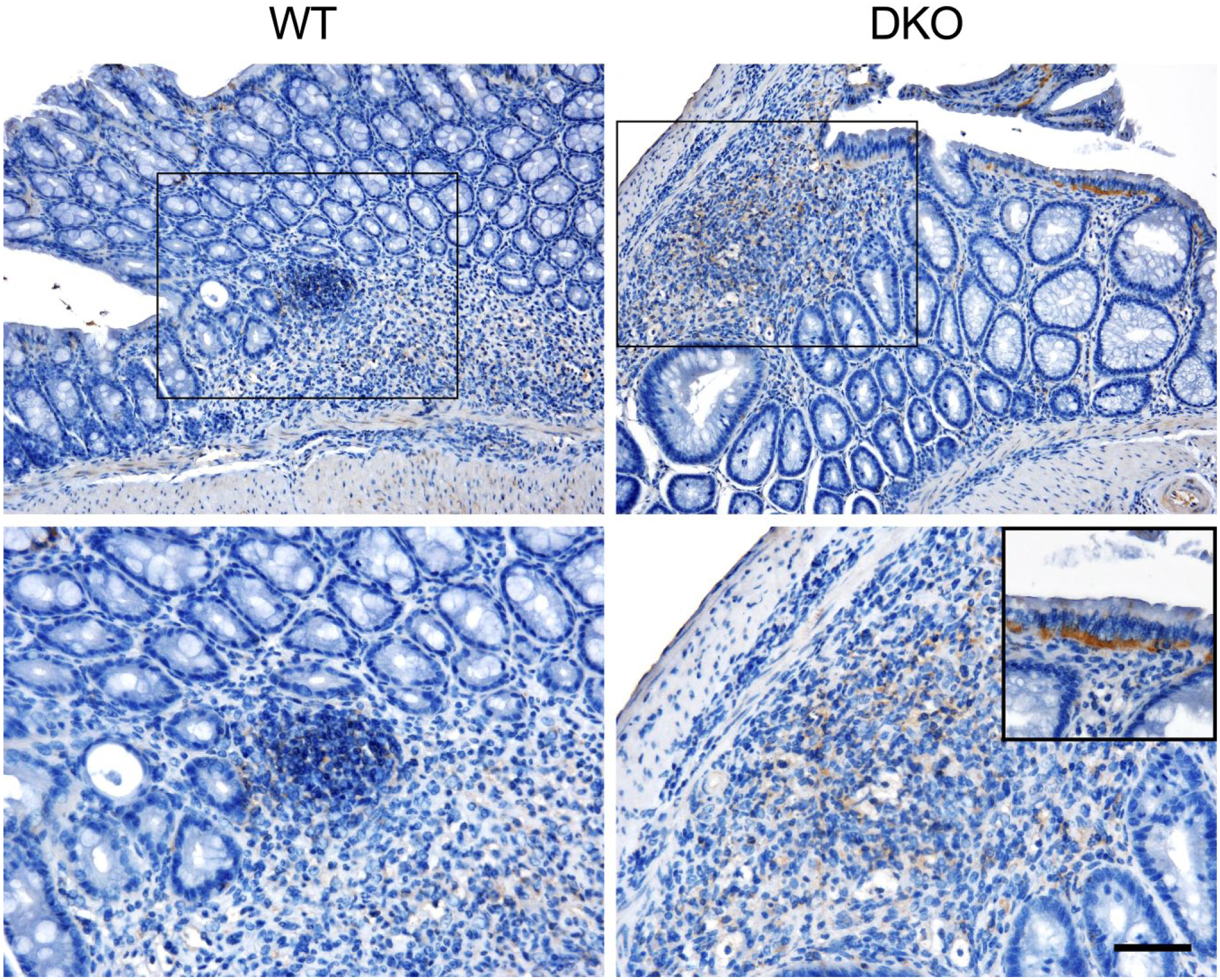
To understand mechanisms underlying mononuclear cell recruitment to inflamed colonic mucosa in the absence of 6-sulfo sLeX-capped O-glycans, we performed quantitative immunohistochemical analysis for MECA-79+ and MAdCAM-1+ HEV-like vessels in DSS-treated mice of both genotypes. As shown in Fig. 4, MECA-79+ HEV-like vessels were found in and around ulcerative lesions in wild-type mice, as is seen in human samples of active UC, while, as expected, MECA-79+ vessels were completely lacking in mice doubly deficient in GlcNAc6ST-1 and GlcNAc6ST-2. On the contrary, MAdCAM-1+ vessels were observed in both groups of colitis mice as well as in human samples of active UC, although the number of MAdCAM-1+ vessels in double knockout mice was significantly greater than that seen in wild-type mice (Fig. 5).
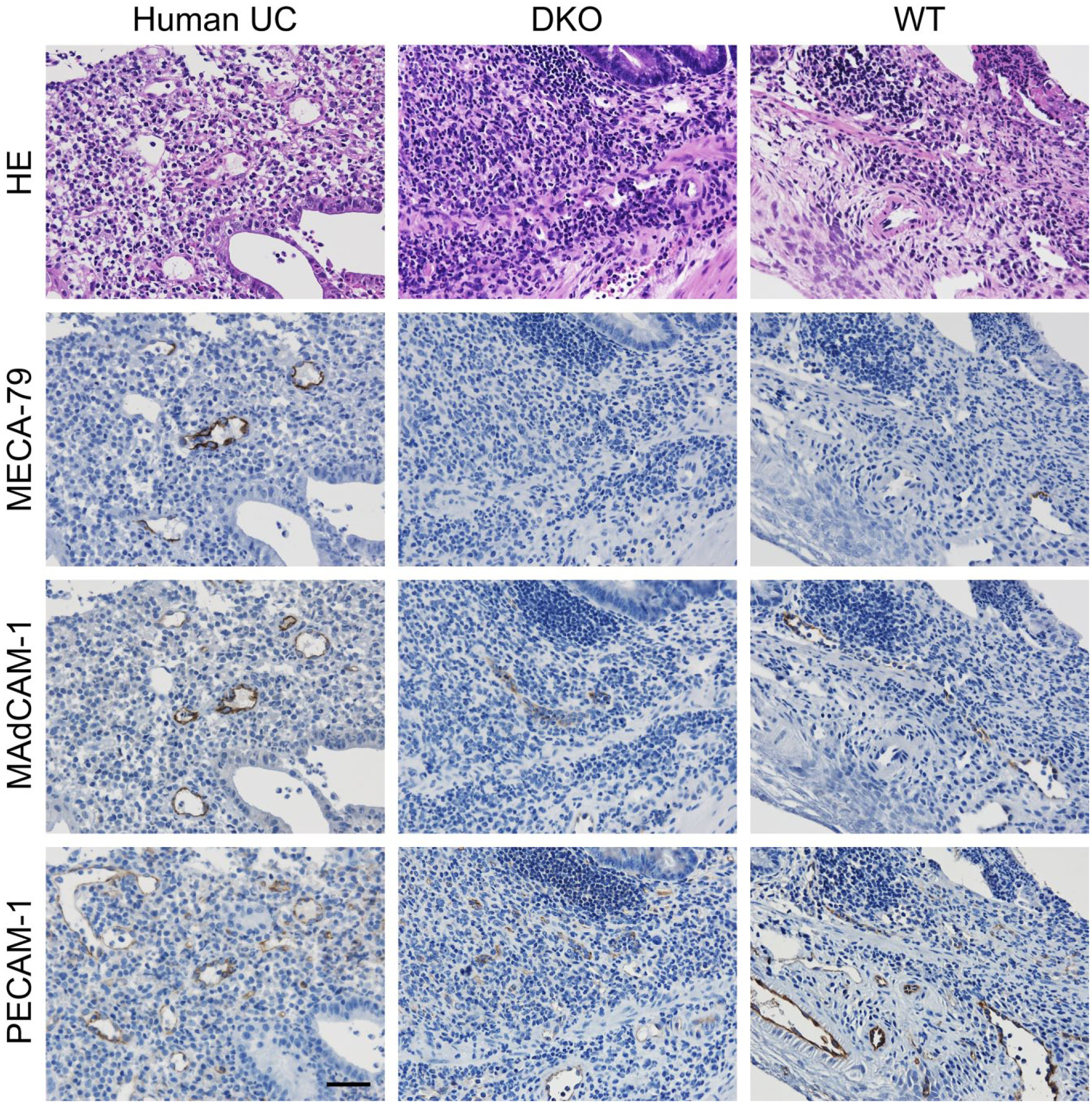
Immunohistochemical profiles of HEV-like vessels seen in chronic, DSS-induced experimental colitis in WT mice and GlcNAc6ST-1 and GlcNAc6ST-2 DKO mice. HEV-like vessels in human active UC are also shown (Human UC). Serial tissue sections were stained with HE and immunostained for MECA-79, MAdCAM-1, or PECAM-1. Signals were visualized with 3,3′-diaminobenzidine (brown), and tissues were counterstained with hematoxylin. Note that HEV-like vessels seen in double knockout mice are MECA-79-negative. Bar = 50 μm. Abbreviations: HEV, high endothelial venule; DSS, dextran sulfate sodium; WT, wild type; GlcNAc6ST, N-acetylglucosamine-6-O-sulfotransferase; DKO, double knockout; UC, ulcerative colitis; HE, hematoxylin and eosin; MAdCAM-1, mucosal addressin cell adhesion molecule 1; PECAM-1, platelet endothelial cell adhesion molecule 1.
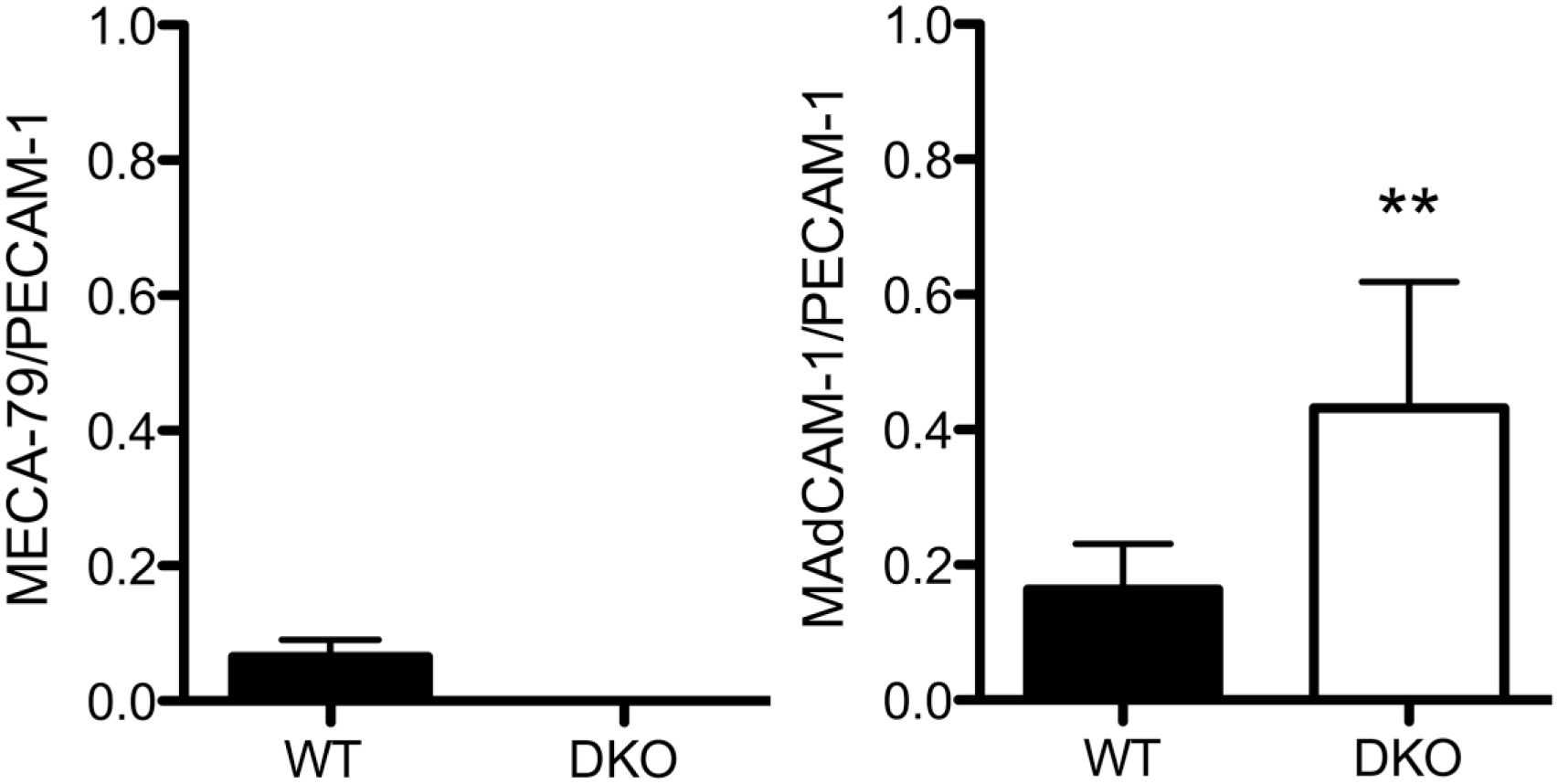
The percentage of MECA-79+ (left) or MAdCAM-1+ (right) vessels among PECAM-1+ vessels in chronic colitis mucosa of WT mice or GlcNAc6ST-1/GlcNAc6ST-2 DKO mice. Data are presented as means with standard deviation. **p<0.01. Abbreviations: MAdCAM-1, mucosal addressin cell adhesion molecule 1; PECAM-1, platelet endothelial cell adhesion molecule 1; WT, wild type; GlcNAc6ST, N-acetylglucosamine-6-O-sulfotransferase; DKO, double knockout.
MAdCAM-1 Serves as a Scaffold Protein for Unsulfated sLeX
It has been reported that mice doubly deficient in GlcNAc6ST-1 and GlcNAc6ST-2 lack 6-sulfo sLeX-capped O-glycans on HEVs and show increased amount of unsulfated sLeX.29,30 To determine whether MAdCAM-1 proteins can serve as scaffold proteins for unsulfated sLeX in GlcNAc6ST-1/GlcNAc6ST-2 double knockout mice, we conducted an E-selectin•IgM chimera in situ binding assay, as E-selectin•IgM can be used as a functional probe for sLeX. 30 As shown in Fig. 6, MAdCAM-1+ HEVs in GALT formed in colitis mucosa, which are negative for MECA-79, were decorated with E-selectin•IgM in a calcium-dependent manner. This finding strongly suggests that in GlcNAc6ST-1/GlcNAc6ST-2 double knockout mice, MAdCAM-1 proteins can function as a scaffold for unsulfated sLeX.
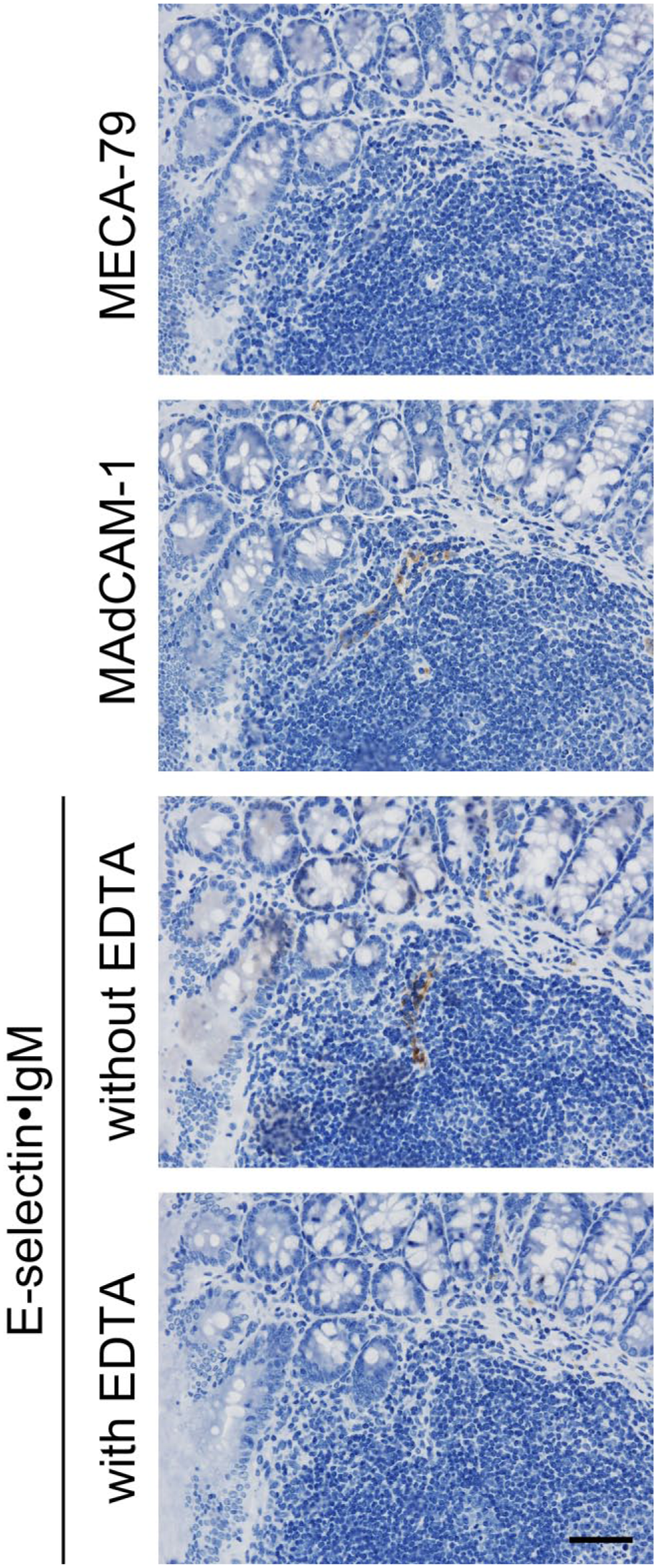
MAdCAM-1+ HEVs in GALT formed in colitis mucosa of GlcNAc6ST-1/GlcNAc6ST-2 double knockout mice are bound by E-selectin•IgM chimeras, a functional probe for sLeX. Serial tissue sections were immunostained for MECA-79 or MAdCAM-1 and subjected to an E-selectin•IgM chimera in situ binding assay. Chimera binding to vessels in the absence of EDTA is completely abrogated in calcium-free (with EDTA) conditions. Signals were visualized with DAB (brown) and tissues counterstained with hematoxylin. Note that MAdCAM-1+ HEVs in double knockout mice are MECA-79-negative. Bar = 50 μm. Abbreviations: MAdCAM-1, mucosal addressin cell adhesion molecule 1; HEVs, high endothelial venules; GALT, gut-associated lymphoid tissue; GlcNAc6ST, N-acetylglucosamine-6-O-sulfotransferase; sLeX, sialyl Lewis X; DAB, 3,3′-diaminobenzidine.
Discussion
Here, we show that mice doubly deficient in GlcNAc6ST-1 and GlcNAc6ST-2 develop more severe DSS-induced colitis than do wild-type mice. Although we observed histological findings similar to human UC, including cryptitis, crypt abscess, and subsequent multiple ulcerative lesions, in both wild-type and double knockout DSS-treated mice, polymorphonuclear cell infiltration of the colonic lamina propria was more prominent in double knockout mice. This finding indicates that elimination of 6-sulfo sLeX-capped O-glycans does not alter polymorphonuclear cell recruitment but rather enhances colonic mucosal inflammation in this experimental context. It is reasonable to assume that the marked polymorphonuclear cell infiltration seen in colitis in double knockout mice is likely caused by a breakdown in the protective function of colonic mucins displaying GlcNAc6ST-2-dependent sulfated glycans. 32
In the DSS-induced colitis model, tight junctions between intestinal epithelial cells are interrupted due to increased permeability of the colonic mucosa.34,41 Both duration and dosage of DSS administration vary from experimenter to experimenter, ranging from 5 days to 10 weeks of duration and a dosage in drinking water from 2% to 5%. 34 As UC is a chronic relapsing disease, use of short-term, high-dose DSS administration to induce an acute form of the disease may not fully represent human UC. In the present study, we chose a low-dosage, long-duration protocol of DSS administration to generate human UC-like acute on chronic inflammation showing histological features of human UC, including cryptitis, crypt abscess, and, most importantly, chronic inflammatory cell infiltrates.
Lymphocyte homing to Peyer’s patches is not affected in mice doubly deficient in GlcNAc6ST-1 and GlcNAc6ST-2.29,30 One author of the current study previously demonstrated that MAdCAM-1 is the primary lymphocyte homing counterreceptor on Peyer’s patch HEVs in double knockout mice by infusing the anti-MAdCAM-1 function-blocking antibody MECA-367 into recipient mice before injecting fluorescence-labeled donor lymphocytes for a short-term in vivo homing assay. 30 MECA-367 infusion did not alter lymphocyte homing to peripheral lymph nodes, but homing to mesenteric lymph nodes and Peyer’s patches in mutant mice was substantially reduced to 5.4% and 1.1%, respectively, of wild-type levels. This finding is in line with our present finding that mononuclear cell recruitment to the DSS-induced colitis mucosa is comparable in wild-type and GlcNAc6ST-1/GlcNAc6ST-2-deficient mice; however, in double knockout mice, whether MAdCAM-1 (most likely decorated with non-sulfated sLeX) plays a principal role in lymphocyte recruitment in colitis mucosa awaits further studies. In these studies, functional analysis such as in vivo lymphocyte homing assay would be required.
Here, we also found that the number of MAdCAM-1+ vessels in the double knockout mouse colitis mucosa exceeded that seen in wild-type mice. This increase suggests a potential compensatory mechanism in which MAdCAM-1 expression is induced, most likely by proinflammatory cytokines including TNF-α,21,22 to recruit lymphocytes when the optimal L-selectin ligand, 6-sulfo sLeX-capped O-glycans, is absent. We previously reported that the number of MAdCAM-1+ HEV-like vessels in the colonic lamina propria of UC patients increases relative to healthy subjects, regardless of clinical stage. 2 Furthermore, Berg et al. reported that through its immunoglobulin-like domain, MAdCAM-1 could serve as a vascular addressin for α4β7 integrin-expressing lymphocytes to trigger L-selectin-independent lymphocyte rolling. 42 Taken together, these facts suggest that L-selectin-independent lymphocyte rolling may become dominant in colonic inflammation induced in GlcNAc6ST-1/GlcNAc6ST-2 double knockout mice.
In the present study, we found that TNF-α proteins were detected around inflammatory cell aggregates in both groups of mouse colitis. In addition, in the case of double knockout mice colitis, TNF-α proteins were found to be accumulated beneath colonic epithelial cell layers. It has been reported that TNF-α is produced by intestinal epithelial cells. 43 Moreover, genetically overproduced TNF-α results in Crohn’s disease-like spontaneous colitis in mice. 44 These facts combined suggest that TNF-α produced either by inflammatory cells or by intestinal epithelial cells can potentially contribute to more severe colitis seen in double knockout mice.
Here, we also show that in a binding assay, MAdCAM-1+ HEVs in GALT formed in colitis mucosa of the double knockout mice become decorated with E-selectin•IgM chimeras calcium dependently. Given that 6-sulfo sLeX-capped O-glycans on these vessels are completely eliminated in GlcNAc6ST-1/GlcNAc6ST-2 double knockout mice, this finding indicates that E-selectin•IgM chimeras likely bind to unsulfated sLeX-capped O-glycans displayed on MAdCAM-1 proteins. It might be helpful to see whether the preincubation of tissue sections with MECA-367 antibody interferes with the binding of E-selectin•IgM chimeras to GALT HEVs. Although unsulfated sLeX-capped O-glycans are not the optimal L-selectin ligand, this carbohydrate structure may contribute in part to L-selectin-dependent lymphocyte rolling.
As noted, we observed marked polymorphonuclear cell infiltration in and around ulcerative lesions particularly in double knockout mice. Several reports demonstrate that neutrophils express α4 integrin, which interacts not only with vascular cell adhesion molecule-1 (VCAM-1) but also with MAdCAM-1, both of which are expressed on vascular endothelial cells.9,45–47 Here, the number of MAdCAM-1+ vessels increased in double knockout mice, suggesting a potential role for α4 integrin/MAdCAM-1 interaction in the substantial polymorphonuclear cell infiltration and more severe colitis seen in double knockout mice. Support for this hypothesis, however, awaits additional studies.
Footnotes
Acknowledgements
The authors thank Messrs. Hisataka Kato, Shinichi Fujii, and Moriyasu Hattori and Ms. Noriko Maruta for technical assistance, and Dr. Elise Lamar for critical reading of the manuscript.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SL designed and performed the research, analyzed the data, and wrote the manuscript; JH and HH performed the research; KU and HK designed the research; MK conceived of and designed the research, analyzed the data, and wrote the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant-in-Aid for Scientific Research 15K08343 (to M.K.) from the Japan Society for the Promotion of Science.
