Abstract
Toll-like receptor 4 (TLR-4), a bacterial lipopolysaccharide sensor, is an innate immunity essential modulator. It is expressed on both immune and non-immune cells and may contribute to the cutaneous and renal manifestations during lupus erythematosus (LE). Our purpose is to evaluate TLR-4 expression and analyzing its role in lupus nephritis (LN) and chronic cutaneous lupus erythematosus (CLE) pathogenesis. TLR-4 immunohistochemical staining was performed on 30 LN renal biopsies compared with 11 healthy renal tissues and 30 skin biopsies from CLE patients compared with 15 normal individuals. CLE patients’ biopsies showed a strong and diffuse TLR-4 expression throughout the epidermis and labeled inflammatory infiltrate and glands in the dermis whereas controls’ skin expressed weakly TLR-4 only in the epidermis basal layer. LN glomeruli and tubules showed an increased and more intense TLR-4 expression compared with normal controls where TLR-4 expression was weak and rarely detected in glomeruli, diffuse and weak in tubules. A significant difference in TLR-4 expression between LN classes, both in glomeruli and tubules, was observed. These data confirm an up-regulation of TLR-4 expression in the affected tissues of CLE and LN patients and highlight the critical role of TLR-4 in the pathogenesis of cutaneous and renal disorders in LE.
Keywords
Introduction
Lupus erythematosus (LE) is a complex autoimmune disease with a broad spectrum of clinical manifestations reaching from localized skin lesions to systemic disease. Classically, in pure cutaneous subtypes of LE, the disease is limited to the skin. While in systemic LE (SLE), it involves internal organs such as kidneys in lupus nephritis (LN). 1 LN is associated with significant patient morbidity and mortality. 2 Overlaps and transitions from cutaneous LE into SLE are observed in 10% to 40% of patients. 1
LE has multifactorial etiopathology and is characterized by the production of a wide variety of auto-antibodies(-Abs) and immune complex deposition resulting in inflammation and eventually, permanent organ damage.
This inflammation is driven by interferon (IFN)-regulated pro-inflammatory cytokines that orchestrate the B- and T-cell–mediated lesional inflammation. During the last years, therapeutic strategies have focused on these players: Biologics targeting type I IFNs and their receptors as well as anti–B-cell drugs have been investigated in clinical trials with variable success. 3 Currently, there are diverse agents under development for lupus treatment by targeting Toll-like receptors (TLRs). 4 TLR activation is associated with runaway inflammation. Thus, controlling the expression of TLRs or the downstream signaling of TLRs is a common mechanism to down-modulate inflammation.5,6
TLR-4 was the first human TLR to be identified in 1997 by Medzhitov et al. 7 It was then found to contribute to the detection and recognition of infectious agents as well as numerous endogenous ligands released by cells undergoing apoptosis, necrosis, or stress.7–9
Several researches have proved that TLR-4 promotes mesangial cell injury and renal fibrosis in chronic kidney disease.10–12 In fact, in pristane-induced experimental lupus, TLR-4−/− mice have decreased pro-inflammatory cytokines, anti-dsDNA, and anti-ribonucleoprotein (RNP) Abs and attenuated renal injury. 13 In addition, TLR-4 up-regulation at the protein or gene level is a potent trigger to induce lupus-like autoimmune disease. 14
Few studies have examined the role of TLR-4 in the development of LN, but no study has investigated its implication for chronic cutaneous lupus erythematosus (CLE). In this work, we aim to evaluate the involvement of TLR-4 in the physiopathogenesis of LE by the assessment of its renal and cutaneous expression and distribution in LN and CLE patients, respectively.
Materials and Methods
Biopsies
For TLR-4 immunohistochemical staining, all biopsies were collected from the Anatomopathology Department of the Habib Bourguiba Hospital of Sfax, Tunisia.
Skin Biopsies
Cutaneous expression of TLR-4 was evaluated on 30 skin biopsies of newly diagnosed CLE patients (21 women and 9 men) with a mean age of 44 years (17–74), coming from rural regions and attending the Dermatology Department at the Hedi Chaker Hospital of Sfax. The diagnosis of CLE was established according to the standard clinical, histological, and immunological criteria of the disease. The laboratory parameters, performed for clinical purpose, were collected for each patient, including indirect immunofluorescence tests for IgG, IgA, IgM, and C3 deposit.
Fifteen skin specimens were obtained from normal individuals who underwent plastic surgery. No one of them suffered from autoimmune or inflammatory disease.
Renal Biopsies
Thirty renal biopsy specimens of patients with LN, followed up in the Nephrology Department of the Hedi Chaker Hospital of Sfax (age, sex and duration of the disease are mentioned in supplementary data) and classified according to the International Society of Nephrology/Renal Pathology Society classification of LN (2003ISN/RPS), 15 three with class II, five with class III, seven class IV, three with class V, eight with class IV+V and III+V, and four with class VI, were included in this study. All the patients had received neither corticoids (>10 mg) nor immunosuppressive drugs during the last 3 months. The immunofluorescence tests data for IgG, IgM, IgA, C1q, C3, anti-kappa light chain Abs, anti-lambda light chain Abs and fibrinogen deposits were collected for each biopsy. The activity and sclerosis index were used to assess disease activity and cumulative damage. Five renal tissues obtained from the normal part of nephrectomized kidneys (because of renal carcinoma) and five cadaver kidneys (autopsy) from subjects without renal disease served as normal controls.
Laboratory Evaluation
For all patients, results of immunological tests performed in the clinical laboratory of immunology, Habib Bourguiba Hospital of Sfax [antinuclear Abs (ANA), anti-DNA Abs, anti-RNP were collected from patients’ sera simultaneously with the skin/kidney biopsy], were used for correlation tests.
All samples were collected after obtaining a written informed consent that was approved by the ethical committee of the Habib Bourguiba Hospital, University of Sfax, Tunisia (protocol number of ethical committee, 4/12).
TLR-4 Immunohistochemistry
Paraffin-embedded tissue sections, 3- to 4-µm-thick, were mounted on positively charged slides, heated at 60C for 30 min and then deparaffinized and rehydrated through a series of alcohol baths before staining. After antigen retrieval with micro-wave treatment in a citrate buffer, pH 6.0, endogenous peroxidase was blocked with 3% hydrogen peroxide for 5 min. Sections were washed three times with the cold 0.01 M phosphate buffered saline after blocking with the 0.4% buffered casein solution. Slides were then incubated for 2 hr with anti–TLR-4 (H-80) Abs [rabbit polyclonal raised against amino acids 242–321 of TLR-4 of human origin (Santa Cruz Biotechnology Inc; Santa Cruz, CA); dilution 1:75 for renal staining and 1:100 for cutaneous staining]. After a secondary antibody incubation, a prepared 3,3-diaminobenzidine substrate chromogenic solution was applied and incubated for about 10 min until color intensity was reached. Finally, sections were lightly counterstained with hematoxylin. For negative control preparation, the primary Abs were replaced with irrelevant isotype-matched control immunoglobulin (data not shown). Photographic images of representative results were captured using the Olympus BX 40 microscope equipped with an Olympus DP 70 camera (Olympus; Tokyo, Japan).
Scoring of Immunostaining
Stained biopsy sections were evaluated by anatomopathologist with a semi-quantitative method. For every sample, TLR-4 expression score was assigned by giving a score ranging from 0 to 12 resulting of the product: intensity score X distribution score.
The Intensity Score
The score was 0 for negative, 1 for weakly positive, 2 for moderately positive, and 3 for strongly positive.
The Distribution Score
The score was 0 for 0%, 1 for 0% to 25%, 2 for 25% to 50%, 3 for 50% to 75%, and 4 for more than 75%.
For renal biopsies, the number of glomeruli and tubules per sample ranged from 3 to 9 and 12 to 35, respectively.
Statistical Analysis
The results were analyzed using SPSS version 20; the Mann–Whitney U-test was used to analyze the difference in TLR-4 expression in renal and cutaneous tissues between healthy controls and patients. Statistical differences between the different classes were analyzed using one-way analysis of variance followed by post hoc Tukey’s multiple comparison tests.
For correlation studies, the Spearman rank correlation was used, and for the association studies between the expression score and the absence or presence of antibodies deposit, we used the Mann–Whitney U-test. A p-value less than 0.05 was considered as statistically significant.
Results
Cutaneous Immunostaining
The TLR-4 immunostaining was principally intra-cytoplasmic for almost all biopsies.
In epidermis, we reported a significant increase in TLR-4 expression in CLE patients’ biopsies compared with controls (p<0.0001; Fig. 1E).
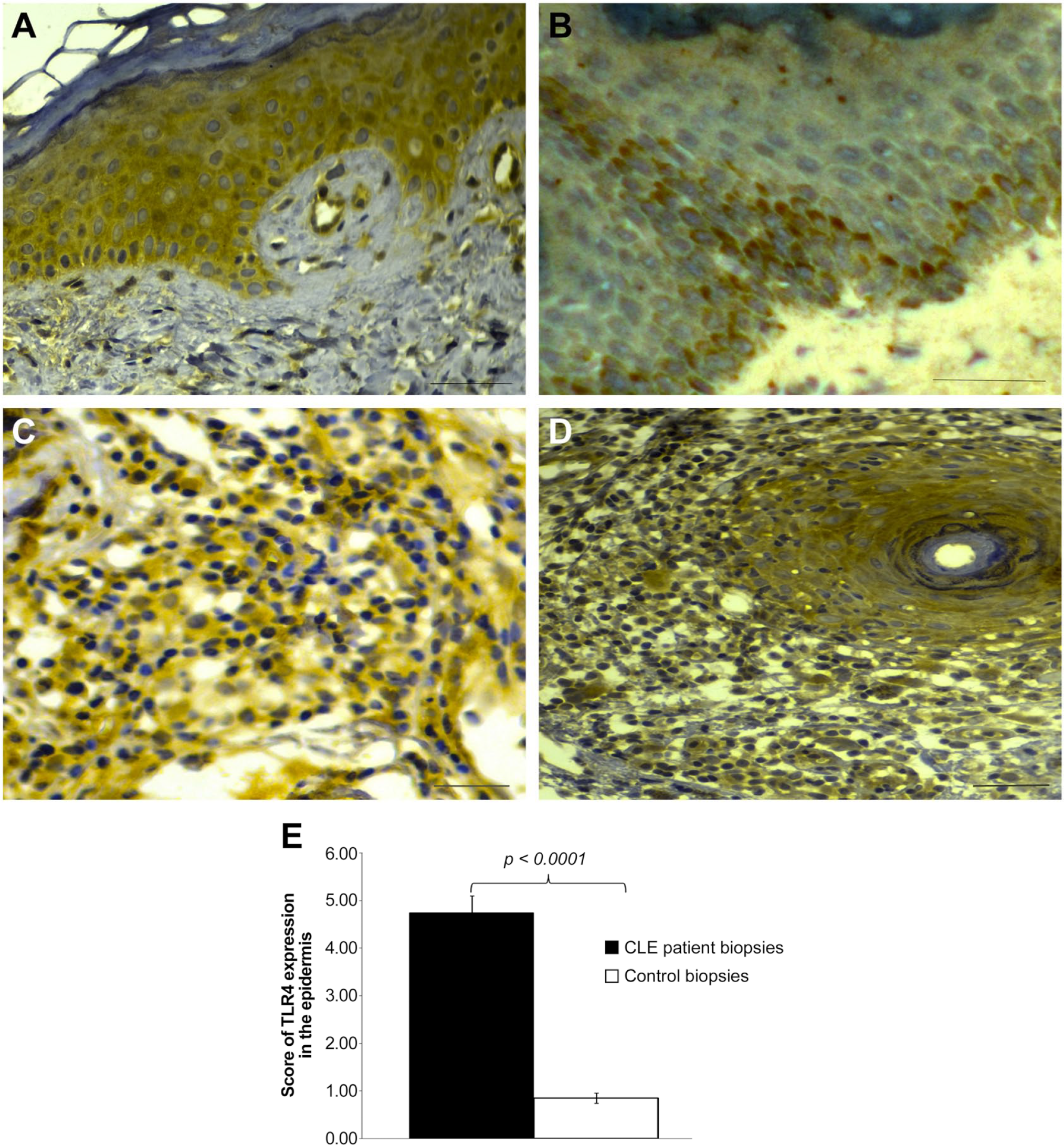
Immunohistochemical staining of Toll-like receptor 4 (TLR-4): (A) in affected epidermis from a patient with cutaneous lupus erythematosus (CLE), (B) in the epidermis of a healthy control, (C) in affected dermis of CLE biopsy showing a labeled infiltrate, (D) in affected dermis of CLE biopsy showing glands expression of TLR-4, and (E) TLR-4 expression score in the epidermis. Scale bar = 50 µm.
Indeed, the epidermis of the control group was weakly labeled and TLR-4 staining was limited to the basal and suprabasal layers. More precisely, TLR-4 expression was barely detected in 8 biopsies, focally enhanced in 7 others, and well-marked in only 1 out of the 20 specimens analyzed (Fig. 1B).
A variation in the expression level of TLR-4 was observed in the 30 biopsies of CLE patients. Thus, 8 patients’ biopsies exhibited a strong and diffuse expression of TLR-4 throughout all layers of keratinocytes (KCs; Fig. 1A) whereas 16 showed a weak and diffuse staining focally enhanced in the basal layer. For the last six biopsies, TLR-4 was not expressed in the upper epidermis whereas a slight and diffuse immunoreactivity was detected in the two-third lower epidermis layers.
In the dermis, we observed an intense labeling of both the glands and the cell infiltrate in CLE patients’ biopsies. However, within the control biopsies, dermis did not exhibit a labeled cell infiltrate and glands were not stained (Fig. 1C and D).
When correlation tests were done, no significant results were revealed between the TLR-4 cutaneous expression score and any of laboratory parameters.
Renal Immunostaining
In all kidney biopsies, we observed that TLR-4 expression was predominantly pronounced in tubular epithelial cells and low expressed in the glomeruli.
In glomeruli, we observed a significant increase in intensity and distribution, from focal to diffuse, in glomeruli of patients with LN compared with normal controls (Fig. 2C). More precisely, although this expression was low in LN glomeruli, it was detected in all of them in contrast to normal biopsies (Fig. 2A). In fact, in control renal biopsies, TLR-4 expression was slightly detected on glomeruli (Fig. 2B).
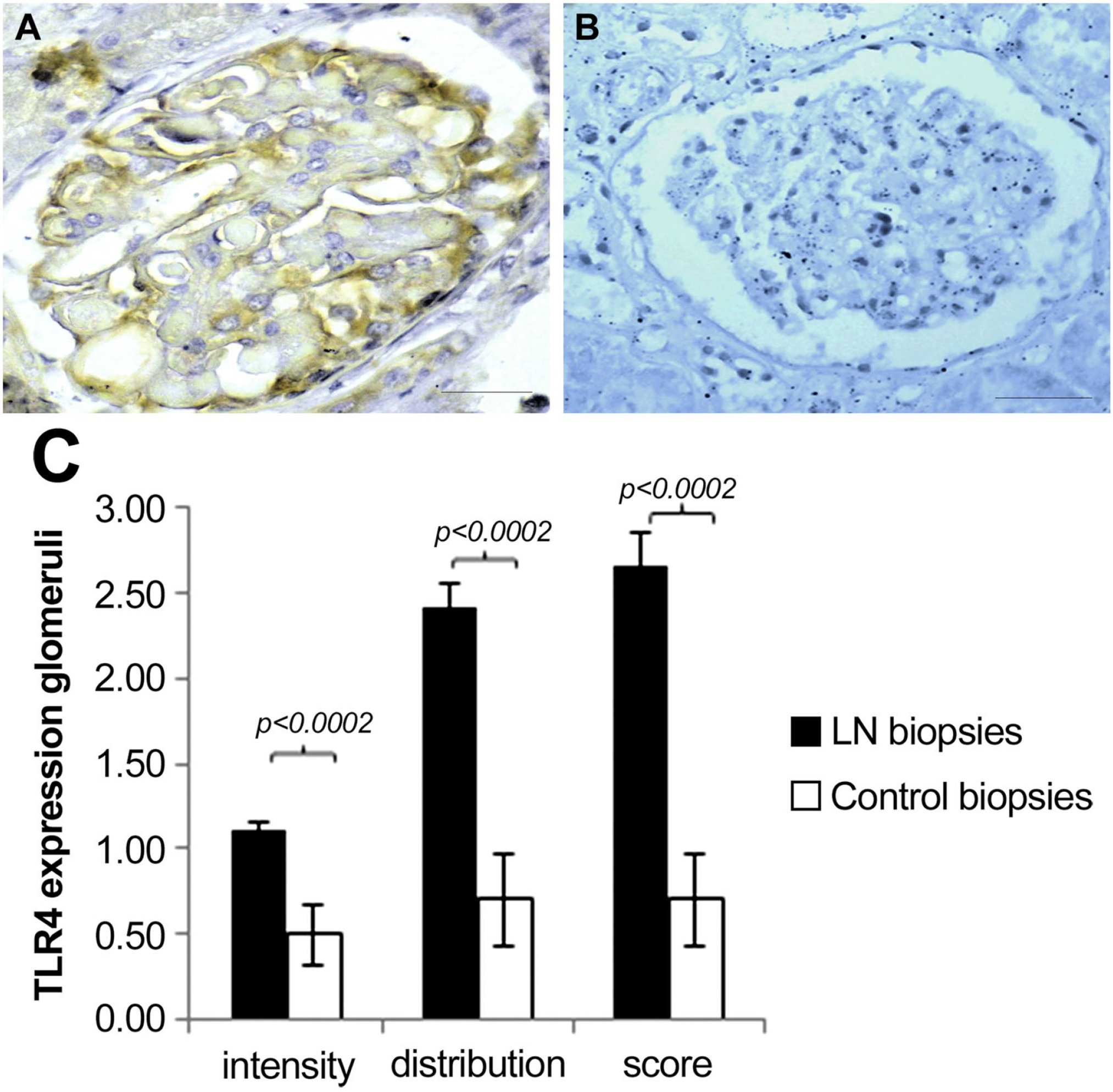
TLR-4 expression in glomeruli: (A) in LN patient glomeruli, (B) in healthy control glomeruli, (C) histogram according to intensity and distribution. Scale bar = 50µm. Abbreviations: TLR-4, Toll-like receptor 4; LN, lupus nephritis; CLE, cutaneous lupus erythematosus.
In tubules, TLR-4 immunostaining intensity was significantly higher in LN biopsies (p=0.014) than in control specimens (Fig. 3C). However, the distribution score was similar between them. Indeed, there was no difference in the TLR-4 expression score when comparing the two groups (Fig. 3A–C).
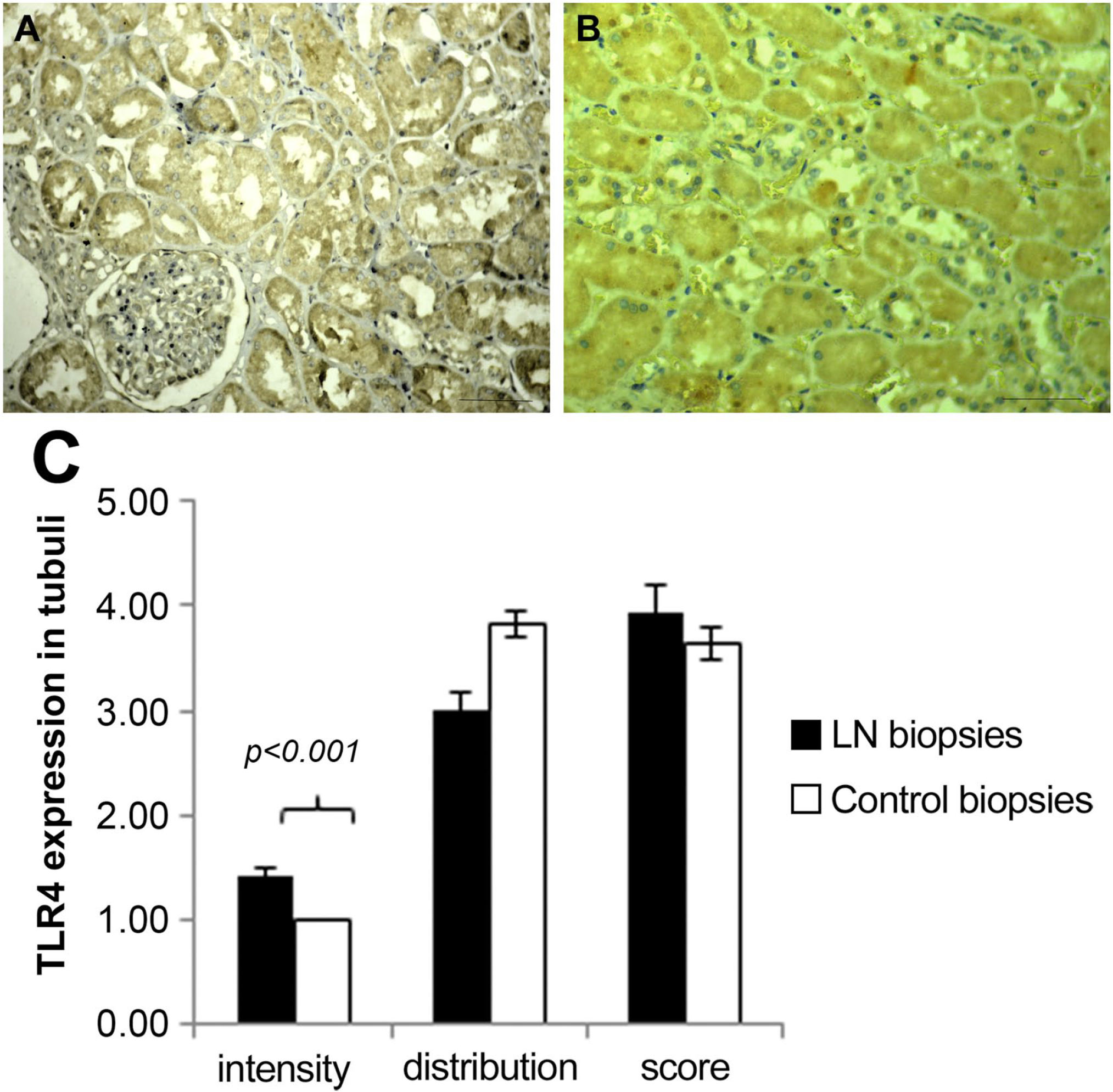
TLR-4 expression in tubules: (A) in LN patient tubules, (B) in a normal biopsy, (C) histogram according to intensity and distribution. Scale bar = 50µm. Abbreviations: TLR-4, Toll-like receptor 4; LN, lupus nephritis.
When comparing TLR-4 expression score between tubules and glomeruli in LN patient biopsies, a positive correlation appeared (p=0.012; r = 0.453).
On inter-subset analysis, the TLR-4 expression score in glomeruli was significantly different between class II, class V+VI, proliferative classes (class III, class IV, and class III+V; class IV+V), and the control group (p<0.001). Regarding LN classes, we noted that the class II expression score was high and that class V+VI has the lowest score (Fig. 4)
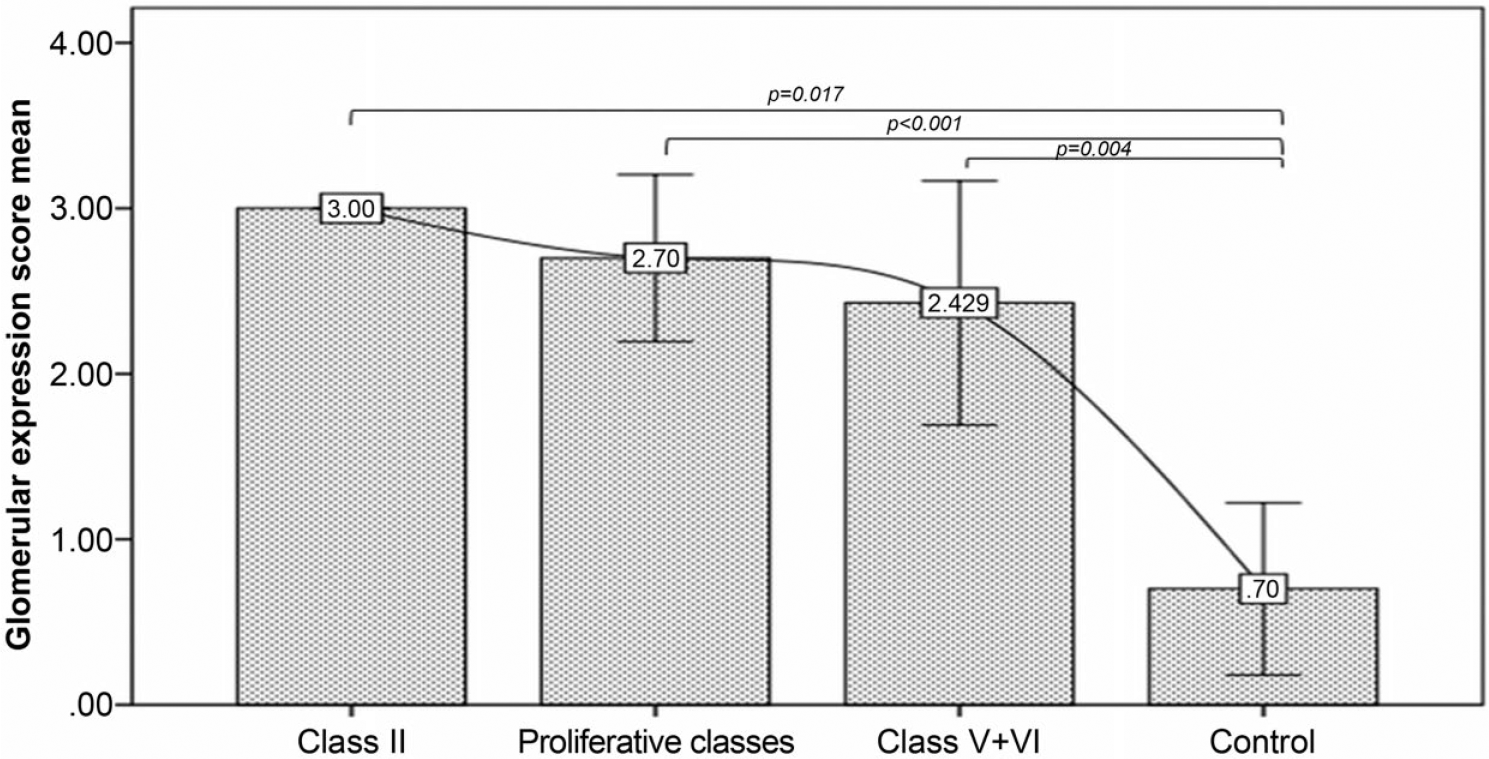
Representation of the Toll-like receptor 4 (TLR-4) expression score in glomeruli.
In tubules, there was no significant difference in the TLR-4 expression score between the classes mentioned above. Taken together, these results show that in class II of the disease, TLR-4 expression in both glomeruli and tubules was the highest.
However, we investigated the correlation between the expression of TLR-4 in LN patients’ samples, immunofluorescence test results, and clinical findings (proteinuria, hematuria, and glomerular filtration rate); significant results are mentioned in Table 1. The IgA presence was associated with the TLR-4 expression intensity in tubules, whereas in glomeruli, TLR-4 expression correlated negatively with the sclerosis index and positively with proteinuria.
Association Between TLR Expression in Renal LN Biopsies and Immunofluorescence Test Results.

Abbreviations: TLR-4, Toll-like receptor 4; LN, lupus nephritis; NS, not significant.
Spearman rank correlation.
Mann–Whitney U-test.
Discussion
During a long period, researchers have been interested in the implication of intracellular TLRs, such as TLR-3, TLR-7, and TLR-9, in the pathogenesis of lupus. However, recent studies have revealed that TLR-4, a cell surface TLR, plays an essential role in autoimmune disorder and may represent a promising target for new immune-therapeutic approaches. Indeed, this receptor in addition to lipopolysaccharide, can also bind different exogenous ligands, like Taxol, viral glycoproteins, rSV fusion protein, mouse mammary tumor virus envelope protein, as well as endogenous ligands, such as necrotic cells, heat-shock proteins (HSPs), fibronectin, fatty acid, minimally modified low-density lipoprotein, and fibrinogen.9,16 This is what prompts us to examine cutaneous and renal expression of TLR-4 in lupus (CLE and LN) patients and controls.
Analyzing TLR-4 cutaneous expression, we noticed that normal controls’ skin expresses weakly TLR-4 in the basal layer of epidermis whereas in CLE patients’ skin, this expression is more diffused and even stronger and extended to all epidermal layers.
Even though weak and limited to the basal layer, the expression of TLR-4 in healthy skin epidermis seems surprising; especially, in a preceding work, measuring its mRNA expression, TLR-4 was not detected either in human epidermis or in primary KCs derived from normal controls’ skin. 17 However, it may reflect the great exposition of our healthy controls to various infectious agents widespread in the rural areas where they come from.
Findings on TLR-4 expression in healthy tissues are controversial. In fact, our observation is not surprising as it was already reported in two other previous studies that confirmed that normal skin specimens express TLR-4. The discrepancy between these two reports is the TLR-4 localization in the epidermis.18,19 In the study by Abida et al., TLR-4 immunohistochemical staining was weak and focally limited in basal layer KCs 19 whereas in the study by Panzer et al., TLR-4 was found to be expressed in the entire nucleated epidermis, more concentrated in the upper tip of the basal layer. 18
Another common point between these two previous studies and our results is the prevalently intracellular expression pattern of TLR-4. This intra-cytoplasmic expression was also reported by Begon et al. 20 and may be due to a localization of TLR-4 in cell organelles, such as endoplasmic reticulum or Golgi apparatus. 21
The cutaneous TLR-4 expression profile was examined in some autoimmune diseases such as pemphigus 19 and psoriasis 22 but not in CLE. In this work, we report an intense and diffuse TLR-4 expression throughout the epidermis combined to a labeled inflammatory infiltrate and an intense TLR-4 expression in glands in CLE biopsies.
This strong epidermal and dermal expression of TLR-4 may prove its potential function in this chronic inflammation state in the skin. In fact, the role of TLR-4 in the production of inflammatory cytokines and the recruitment of neutrophils has been largely documented.23–25 According to these data, it seems that the cutaneous barrier engages actively TLR-4 in the development and progression of CLE.
If the cutaneous manifestations are the most frequent in LE patients after the general signs, the kidney injury represents the most common serious damage in LE.
To the best of our knowledge, this is the first study to assess renal expression of TLR-4 in LN patients in different stages of the disease compared with a control group; albeit, a study in 2014 has enrolled six renal patients’ biopsies as a disease control. 26
We showed an increased and more intense TLR-4 expression, respectively, in LN glomeruli and tubules compared with normal controls where TLR-4 expression was weak and rarely detected in glomeruli, and diffuse and constantly weak in tubules. Our results are in concordance with those of Wang et al. who reported that patients with LN exhibited weak expression of TLR-4 in glomeruli and diffuse expression in renal tubules and interstitium. 26
In a murine model study, TLR-2 and TLR-4 mRNA expression in the kidney was predominantly located in the renal tubular epithelium in healthy kidneys and this expression was up-regulated during renal sterile inflammation induced by unilateral renal pedicle clamping. Moreover, the authors confirmed that TLR-4 mRNA renal synthesis was enhanced by the action of IFN-γ. 27 In fact, the IFN-γ responsive element is found in the promoter region of the gene encoding for TLR-4. 28 As SLE is characterized by high levels of IFN-γ, particularly at the severe stages of the disease, 29 we speculate that this renal up-regulation of TLR-4 expression may be due to the presence of a high amount of IFN-γ in LN patients’ sera.
It is noteworthy that, in our results, the positive stain in glomeruli appears almost restricted to podocytes. This result is sustained by an in vitro study on the murine model that demonstrated that podocytes express TLR-4 constantly and that its expression is up-regulated in glomerulonephritis of immune complex origin. 30 In fact, podocytes may have immunomodulatory activities that contribute to renal injury. 31
In another murine model study, Bei Liu et al. reported that TLR-4 up-regulation at the protein or gene level alone without any exogenous insult induces a similar lupus-like autoimmune glomerulonephritis. 14 These results consolidate our findings and support our belief that TLR-4 up-regulation in LN kidneys may reflect the potential function of this receptor in the physiopathology of LN.
Furthermore, TLRs are described to be involved in non-infectious inflammatory disease, in which they are activated by endogenous danger–associated molecular patterns that are released from injured tissue. 32
Thus, we speculate that TLR-4 up-regulation revealed in both CLE and LN patients’ biopsies can be tightly linked to the presence and/or high level of endogenous ligands, such as HSPs, which characterize the inflammatory state observed in lupus. Indeed, HSP intracellular synthesis is known to increase remarkably under several pathological conditions associated with lupus such as inflammation, viral and bacterial infections, and autoimmunity. 33 As TLR-4 activation is known to induce several pro-inflammatory events, triggering of TLR-4 by these endogenous ligands will aggravate this chronic inflammation in a positive feed-back loop. Interestingly, Minota et al. showed that serum from patients with SLE frequently contained IgM and IgG auto-Abs to the constitutively expressed 73-kDa/pl 5.5 member of the hsp70 family of HSPs. 34 Furthermore, it has been demonstrated that hsp70 induces pro-inflammatory cytokine production that is mediated via the MyD88/NFκB signal transduction pathway and that hsp70 utilizes both TLR-2 (receptor for Gram-positive bacteria) and TLR-4 to transduce its pro-inflammatory signal in a CD14-dependent fashion. 35
In other similar studies, hsp60 was identified as an endogenous molecule that can be released by tubular epithelial cells on injury and leads to glomerular necrosis. 36 The pro-inflammatory signaling of human hsp60 was found to be dependent on a functional TLR-4. 37
These data support the role of TLR-4 endogenous ligands in the development and exacerbation of the disease. In consequence, it seems that the assessment of HSP in both CLE and LN patients is worth investigating.
However, and on subset analysis, we observed a significant difference between classes in TLR-4 expression glomeruli. In fact, high scores were recorded in class II and in proliferative forms of the disease, whereas in non-proliferative forms, TLR-4 expression was dramatically decreased. A negative correlation between the sclerosis index and TLR-4 glomerular expression score was revealed that highly supports the result above. Controversially, some markers of LN activity, such as index activity, and clinical markers (complement and anti-DNA Abs deposit) were not associated with TLR-4 expression neither in glomeruli nor in tubules. As lupus disease undergoes active and remission phases, we suppose that TLR-4 renal expression does not follow the disease activity. Moreover, we noticed that TLR-4 glomerular expression was differently over-expressed between patients with proliferative and non-proliferative classes of the disease. This supports that TLR-4 is highly implicated in the trigger of the immune-mediated glomerular injury mechanisms and that the persistence of the chronic inflammatory conditions may favor the over-expression of TLR-4 receptor.
An interesting significant association between the intensity of TLR-4 tubule expression and the IgA deposit on biopsies was noted. In a previous work, Lepenies et al. demonstrated that TLR-4 expression was significantly up-regulated in patients with severe proteinuria, and in patients with chronic ischemic renal damage and IgA nephropathy. 10 Besides, a mouse model study showed that TLR-4 signaling is involved in IgA-stimulated mesangial cell activation. 11
Taken together, these results indicate that LN patients have altered TLR-4 renal expression. However, what remains is to determine functionally how this dysregulation affects development and/or progression of the disease.
In conclusion, we confirm that TLR-4 is up-regulated in both epidermal KCs of CLE and in tubular epithelial and glomerular cells of LN patients. The renal TLR-4 expression is expressed differently between LN classes and correlates negatively with the sclerosis index. These findings suggest that TLR-4 has an important pathogenic role in CLE inflammation and LN damage. Thus, TLR-4 may represent a novel target in the treatment of LE: Modulating TLR-4 expression and/or blocking its inappropriate activation with antagonists could be a novel way in the treatment of this chronic and disabling disease.
Footnotes
Acknowledgements
This study was supported by the Ministry of High Education and Scientific Research of Tunisia.
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
NE is the main researcher in this work; she performed experiments, data acquisition, and drafted the work. RF contributed in the design and the interpretation of the work and did the statistical analysis. LA and KK contributed in the analysis and interpretation of data. KS, OA, MBJ, and TS contributed in the collection of samples. HM is the chef of the project; he revised the work and gave the final approval of the version to be published. Each author has read and approved the manuscript before submission.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
