Abstract
Corneal epithelial cells exhibit continuous centripetal movements at a rate of about 30 µm per day, but neither the driving force nor the mechanism that determines the direction of movements is known. To facilitate the investigation of homeostatic cell movement, we examined if the intracellular position of a centriole can be used as a directional marker of epithelial cell movements in the mouse cornea. A direction of cell movements was estimated in fixed specimens from a pattern of underlying subepithelial nerve fibers. Intracellular position of centrioles was determined by gamma-tubulin immunohistology and plotted in a narrow strip along the entire diameter of a cornea from limbus to limbus. When we determined the position of centrioles in the peripheral cornea where cell movements proceed generally along a radial path, about 55% of basal epithelial cells contained a centriole in the front half of a cell. However, in the central cornea where cells exhibit a spiral pattern of movements, centrioles were distributed randomly. These results suggest that centrioles tend to be positioned toward the direction of movement in corneal basal epithelial cells when they are moving centripetally at a steady rate.
Introduction
Corneal epithelium is a dynamic tissue in which cells are constantly renewed and lost, and yet the total mass is kept steady by a mechanism yet to be elucidated.1–4 Continual maintenance of epithelium is essential for its physiological functions, including corneal transparency, and our understanding of it at a molecular and cellular level would be crucial in coping with various pathologic conditions such as wound healing. Three major factors that govern epithelial homeostasis are cell division, cell movements, and cell loss; this study is concerned about the cell movements.
A strong body of evidence suggests that corneal epithelial cells exhibit continuous and steady movement from the limbus toward the center of the cornea.5–11 In the mouse cornea, our study showed that an average rate of movement was about 26 µm per day. 11 However, the driving force of this centripetal movement is not known. The precise direction of cell movements—mostly centripetal—can be experimentally determined by a time-lapse recording with multiple time points, but currently it cannot be determined with histological materials or live tissues with a single time point. It would be useful to have a histological marker that can be used to examine the dynamic aspect of cell movements.
For this purpose, we focused on the intracellular position of a centriole as a potential directional marker of epithelial cell movements based on several lines of evidence. When endothelial cell movements were studied in a wound healing assay, a clear correlation was found between the position of microtubule-organizing center (MTOC), where the centriole is located, and the direction of endothelial cell migration; that is, most cells along the leading edge of a wound contained MTOC located between the wound edge and the nucleus.12,13 Based on this observation, it was suggested that MTOC or associated cellular components may function as a guiding cue for determining the direction of cell movements. The data also suggested that the MTOC position in reference to a nucleus could serve as a marker for the direction of cell movements. An idea has been advanced that the centrosome assists positioning of the nucleus at the cell center and that the relative positioning of the nucleus and centrosomes during migration indicates where the cytoplasmic mass is moving. 14 However, cells migrating on micro-patterned lines of fibronectin in vitro were found to contain the Golgi complex and the centrosome behind the nucleus, 15 suggesting that the centriole position may be dependent on the cell type and the extracellular environment. In Caenorhabditis elegans, centrosomes appear to play an active role in polarization of intestinal cells by reassigning MTOC functions 16 while it has been suggested that centrosomes could release diffusible signal molecules to induce polarity in C. elegans zygotes. 17
We report our observations here that centrioles tend to be positioned in the front half of a cell in corneal basal epithelial cells when they are moving centripetally at a steady rate. The results raise a possibility that aggregate data about centriole positions from a group of cells may be used to predict the direction of cell movements in the corneal epithelium.
Materials and Methods
Animals
All methods involving live animals were approved by the institutional animal care and use committee, and adhered to the ARVO Statement for the Use of Animals in Ophthalmic and Visual Research. C57BL/6 mice were purchased from Jackson Laboratories (Bar Harbor, ME) and used at ages between 12 and 20 weeks. The data reported here were obtained from 13 eyes from 9 mice (7 males and 2 females).
Histology
The mouse eyes were obtained after the animal was sacrificed with a lethal injection of pentobarbital (200 mg/kg body weight), and they were immediately fixed in cold methanol at −10C for 15 min, followed by a rinse and equilibration in PBS at 4C. The eyes were dissected to isolate the cornea and processed for staining.
Immunostaining was carried out by first incubating the whole-mount cornea with PBS containing 10% donkey serum (catalog 017-000-121; Jackson ImmunoResearch Laboratories, West Grove, PA) and 0.2% saponin (Sigma-Aldrich, St Louis, MO) for 3 hr at room temperature. The cornea was then incubated with 100 µl of primary antibody solution in PBS containing 4% donkey serum and 0.1% saponin in a well of a polyvinyl chloride flat-bottom 96-well plate (BD-Falcon catalog 353912, Corning Inc., Corning, NY) on an oscillating platform shaker at room temperature overnight. After a rinse with PBS (four times, 20 min each), the cornea was incubated with fluorescently labeled secondary antibody solution in the same manner as the primary antibody except that the incubation time was 6 hr. The cornea was rinsed with PBS (four times, 10 min each), and the epithelial cell nuclei were stained with nuclear fluorescence dyes, 4′,6-diamidino-2-phenylindole (DAPI) or Hoechst 33258 (both from Sigma-Aldrich, used at 1 µg/ml in PBS), by incubating the cornea in the dye solution for 10 min at room temperature, followed by a rinse with PBS (four times, 10 min each). The cornea was mounted in 50% glycerol/PBS, containing 0.1% n-propyl gallate (Sigma-Aldrich) before microscopy. Rabbit anti-gamma-tubulin antibody (catalog T5192, 1:200 dilution; Sigma-Aldrich) was used to stain the centrioles, and rat anti-CD90.2 antibody (catalog 550543, 1:20 dilution; BD Pharmingen, San Diego, CA) or rabbit anti-beta-tubulin antibody (catalog T2200, 1:100 dilution; Sigma-Aldrich) was used to stain subepithelial nerves. Secondary antibodies, purchased from Jackson ImmunoResearch Laboratories, were donkey anti-rat IgG(Fab′)2 conjugated with Cy3 (catalog 712-166-150, 1:400 dilution) and donkey anti-rabbit IgG(Fab′)2 conjugated with Cy2 (catalog 711-226-152, 1:400 dilution). Specificity of these antibodies is documented in manufacturer’s respective technical brochures, and we determined that it is suitable for the purpose of this study in all instances.
Whole-mount corneas were placed under an upright fluorescence microscope (Zeiss Axioskop 2, Carl Zeiss Inc, Thornwood, NY, with a water immersion Axioplan 40X objective, Carl Zeiss Inc, Thornwood, NY, which gives an effective depth of focus of a few micrometers) with focus on the basal epithelial cell nuclei. All images were acquired digitally (MetaMorph, Molecular Devices, LLC., Sunnyvale, CA) at a resolution of 0.167 µm/pixel (Hamamatsu ORCA camera, Hamamatsu Photonics K.K., Shizuoka, Japan), and assembled to generate a single panorama image of a corneal strip from limbus to limbus, using Photoshop (Adobe, San Jose, CA). All three parts (nuclei, centriole, and nerves) were registered and overlaid for analysis on the computer. To outline the area to be measured, a box of five equal-sized grids with a width of 100 µm was overlaid on the composite image between the cornea–limbus border and the central cornea. Another box of five grids was placed on the other side of the corneal strip. The cornea–limbus boarder was determined by the difference in the nuclear size and shape between corneal and limbal cells as described previously. 18 The nominal center of the cornea was defined from the nerve fiber pattern that formed a spiral that converged to the center; this was usually slightly off the geometric center. The total area of measurements of 10 grids was approximately 0.35 mm2, containing about 5000 basal epithelial cells, with an average density of about 14,000 cells/mm2.
Nuclei and centrioles were manually plotted in the entire strip of the cornea from limbus to limbus and tallied within each of the 10 grids. All centrioles were plotted with a “front” or “back” label, referring to a front half or a back half of a cell in reference to the direction of cell movement, which is assumed to be along the length of the subepithelial nerve fibers. Although appearance of immunologically detectable centrioles depends on the cell cycle position of each cell, we did not determine cell cycle positions in this study. Since dividing cells have centrioles at two ends of mitotic poles, centriole positions are not useful for estimating cell movement direction. It is also likely that dividing cells are stationary and not taking part in homeostatic centripetal cell movements. For this reason, any mitotic cells that could be identified from a nuclear staining pattern were excluded from measurements. Among non-mitotic cells, two centrioles could be identified in some cells while others exhibited only one detectable centriole. And yet some cells contained no detectable centrioles. In the end, we were able to score for the centriole position in about a half of total basal epithelial cells.
The plots were then analyzed using ImageJ. 19 Statistical analyses were carried out with XLSTAT (Addinsoft, New York, NY).
Results
Nerve Fiber Patterns in the Cornea
Corneal epithelial cells move centripetally from the limbus to the central cornea in general. However, the exact path of cell movements is not always along a straight radial line; rather the direction of cell movements often deviates from the radial line and sometimes they move along a line that is nearly parallel to the limbus. To precisely determine the direction of epithelial cell movements in the normal cornea, we took advantage of our observation (Fig. 1) that the direction of homeostatic centripetal cell movements coincides nearly perfectly with the pattern of subepithelial nerve fiber organization. Note that green fluorescent protein (GFP) stripe patterns in Fig. 1A and C represent paths of epithelial cell movements in the cornea as we have shown previously. 11 Thus, we determined the nerve fiber orientation by histology and made an assumption that this is the path that epithelial cells took to crawl toward the central cornea.
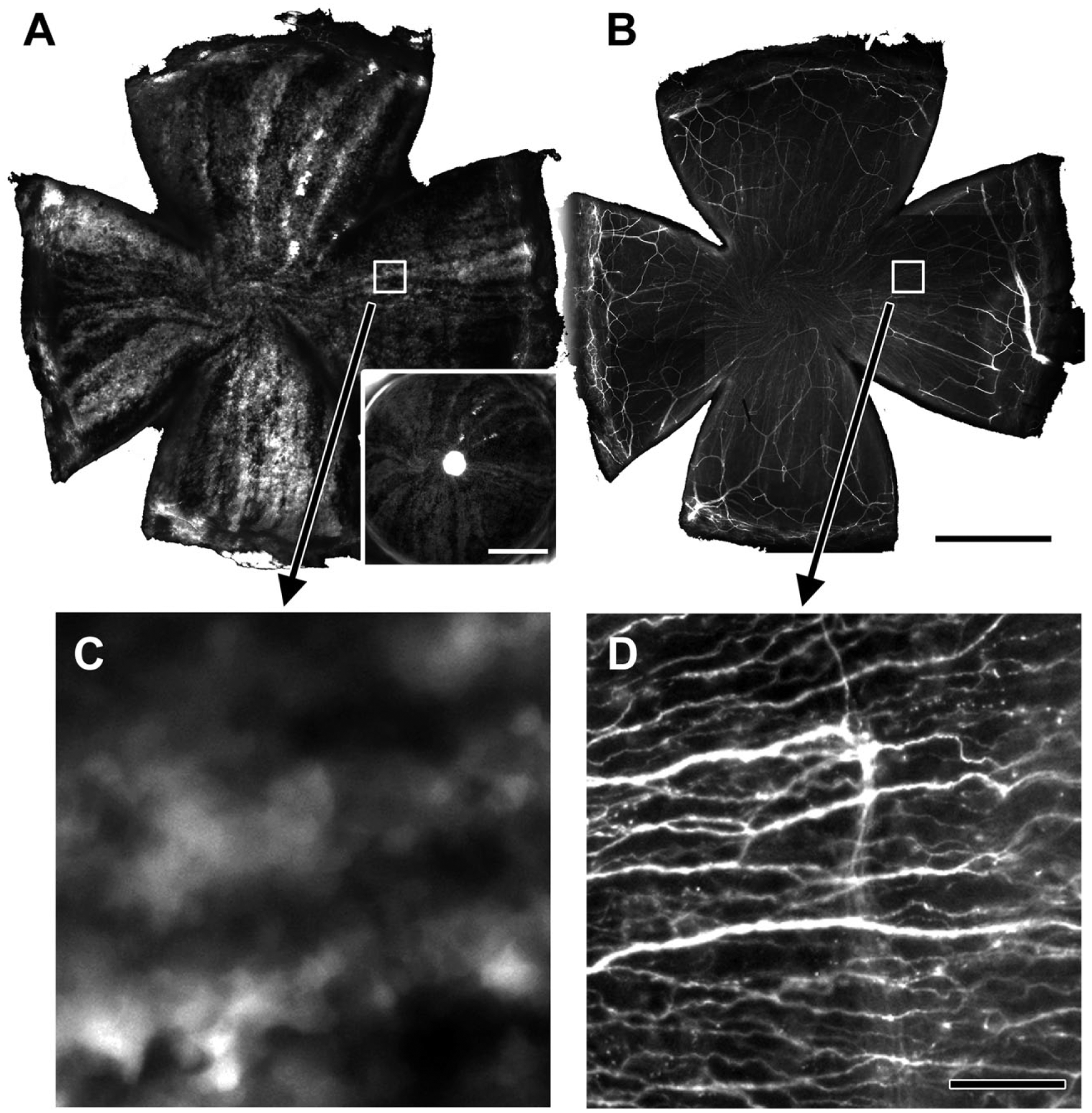
An epithelial green fluorescent protein (GFP) stripe pattern matches the distribution of subepithelial nerve fibers. Nerve fibers were visualized by immunostaining with beta-tubulin antibody. GFP stripe patterns, by virtue of some lineages of epithelial cells being bright and others dim, identify a migration track of epithelial cell clusters. 11 Panel A, a low-power image of corneal GFP patterns. An inset at lower right shows an epithelial GFP pattern of the same cornea in a live mouse immediately before sacrificing the mouse. Panel B, the same cornea as in panel A that was stained for nerves with beta-tubulin antibody. Note the fine fibers in the radial pattern that matches GFP patters in panel A. Panel C, a magnified image of an area indicated by a white box in panel A. Panel D, a magnified image of an area indicated by a white box in panel B. Panels C and D show the same area, demonstrating that the GFP stripe patterns (surrogate of an epithelial movement path) coincide with nerve fiber patterns. Bars A and B = 1 mm; bar D = 50 µm.
Centriole Positions in the Moving Epithelial Cells in the Basal Epithelium of the Cornea
We determined the intracellular position of centrioles in the basal epithelium of the mouse cornea in histological specimens, as depicted in Fig. 2. Typical examples are shown in Figs. 3 and 4. For brevity, the position was categorized as either a front half or a back half of a cell, using the direction of cell movements as the reference to determine front and back. Measurements were carried out in cells along the entire length of a diameter of the cornea from limbus to limbus. A preliminary analysis indicated that there was no significant difference between a corneal strip of superior-inferior orientation and that of nasal-temporal orientation, and therefore they were combined for the subsequent data analysis.
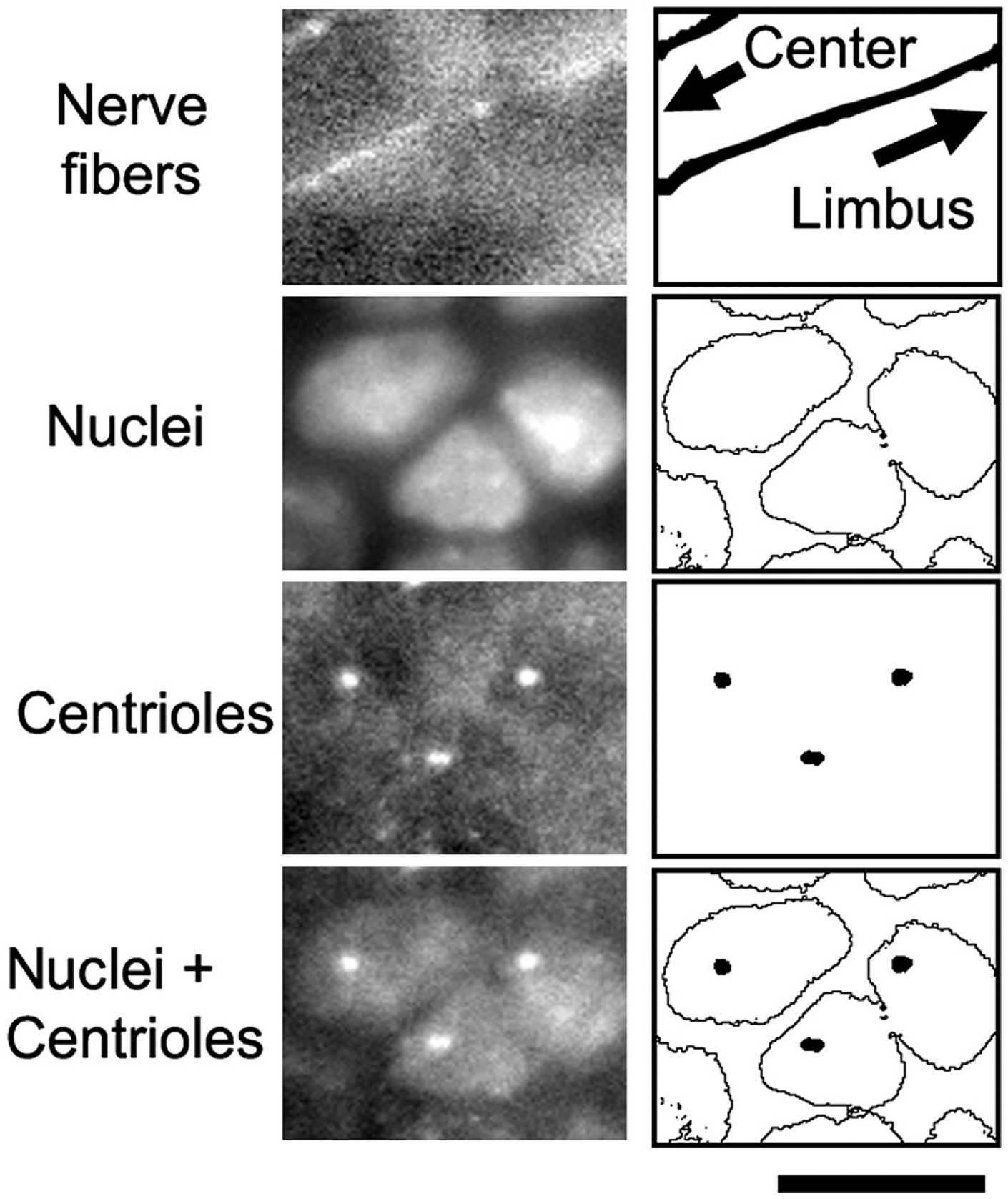
A schematic of scoring intracellular centriole positions. A process of scoring is depicted with binary images (panels at right) for a demonstration purpose. Actual scoring is done with gray-scale images (panels at left) displayed on a computer monitor. A nucleus occupies most of the horizontal area of a basal epithelial cell, and the cytoplasm is approximated by the area of the nucleus. In the three cells in this figure, centrioles were scored to be in the front half of a cell because they were found at an intracellular location nearer to the central cornea than to the limbus. Bar = 10 µm.

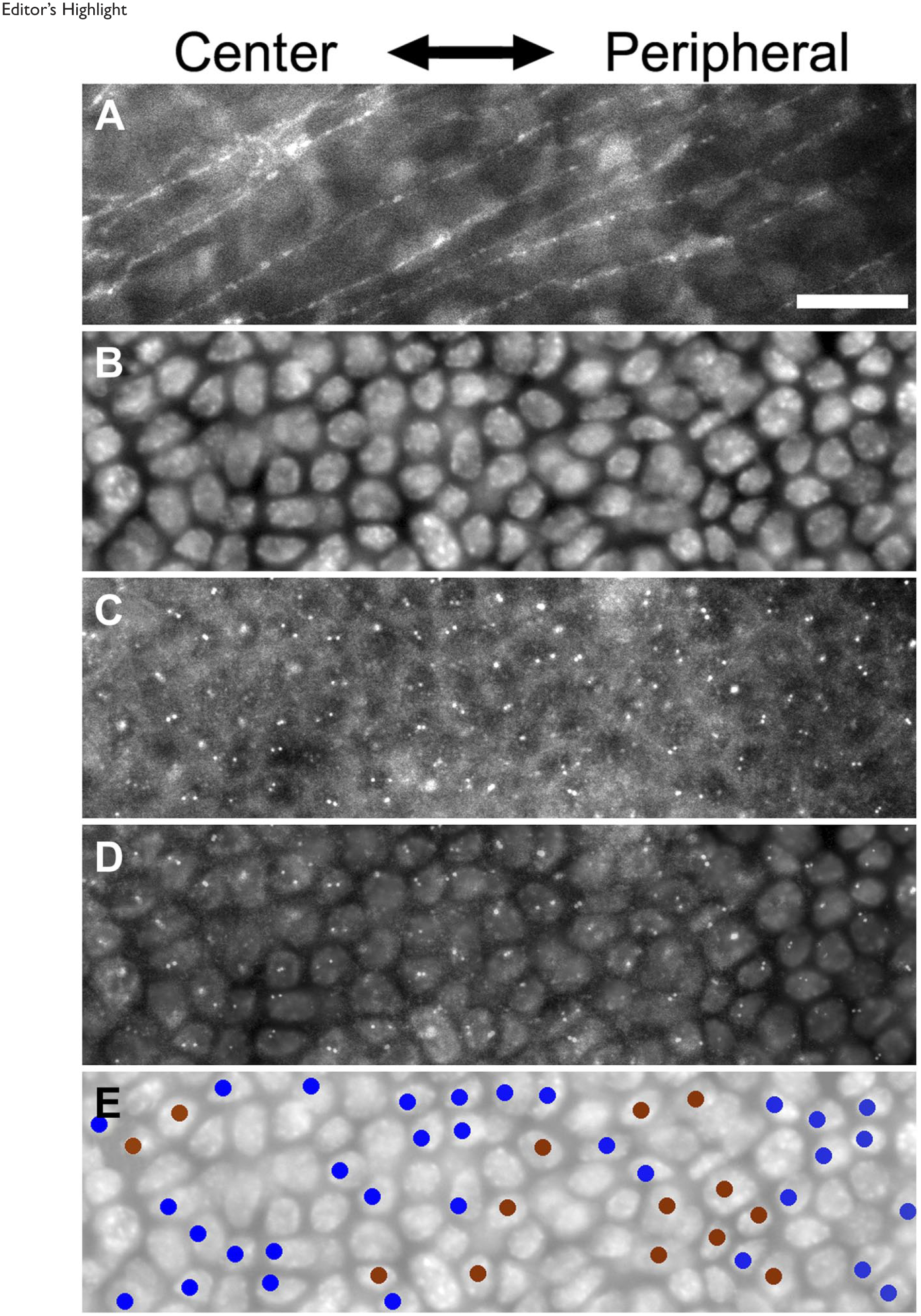
A part of a typical whole-mount cornea strip that was used to determine the position of intracellular position of centrioles. Panel A, nuclei of basal epithelial cells stained with 4′,6-diamidino-2-phenylindole. Panel B, centrioles stained with gamma-tubulin antibody in the same area. Panel C, subepithelial nerves as determined by beta-tubulin staining in the same area. Panel D, a plot of cells with a centriole in the front half of a cell. Panel E, a plot of cells with a centriole in the back half of a cell. Subepithelial nerve fibers were used to determine cell movement directions. Roughly 50% of basal epithelial cells were scored for centriole position because they contained unambiguous centriole staining. Bar = 50 µm.
Editor’s Highlight
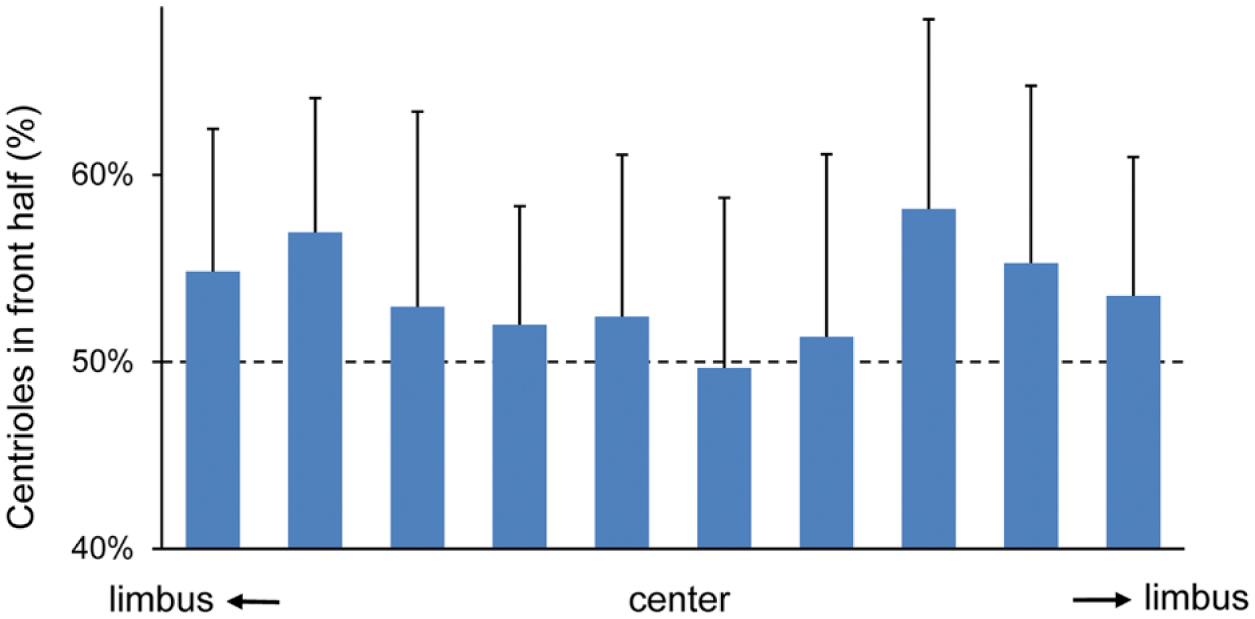
Percentage of centrioles in the front half of a cell as a function of corneal position along the entire diameter from limbus to limbus. The front half is determined from the direction of cell movements, which in turn was determined by subbasal nerve fiber patterns. A limbus-to-limbus strip was divided into 10 boxes, and the percentage of centrioles in the front half in each box was calculated from measurements of 25 limbus-to-limbus strips from 13 eyes. Average and standard deviation are shown in the figure. Dotted lines indicate 50%, which is the expected value for random distribution of centrioles.
We were able to determine the position of centrioles in about 50% of all basal epithelial cells because their intracellular position was unambiguous. The summary data of 10 grids in the limbus-to-limbus strip show the effect of corneal positions along the corneal radius (Fig. 5). The percentage of cells with centriole position in the front half of a cell was calculated within each boxed area, and the average of the percentage values was obtained for each box (Fig. 5). The data were grouped into two categories: those in the peripheral cornea (60% of the diameter) and those in the central cornea (40% of the diameter). A statistical analysis indicated that the percentage values of centriole position in peripheral cells were statistically distinct from 50% that would be expected in a random distribution, while those of central cells were not significantly different from 50% (Table 1). In other words, peripheral cells tended to contain centrioles in front of a cell while central cells showed no such preference. Thus, the correlation of centriole positions versus cell movement direction was lost at the central cornea where epithelial cells move in a spiral pattern at a decreasing rate (to zero at the center, data not shown).
Intracellular Position of Centrioles in Basal Epithelial Cells of the Mouse Cornea.
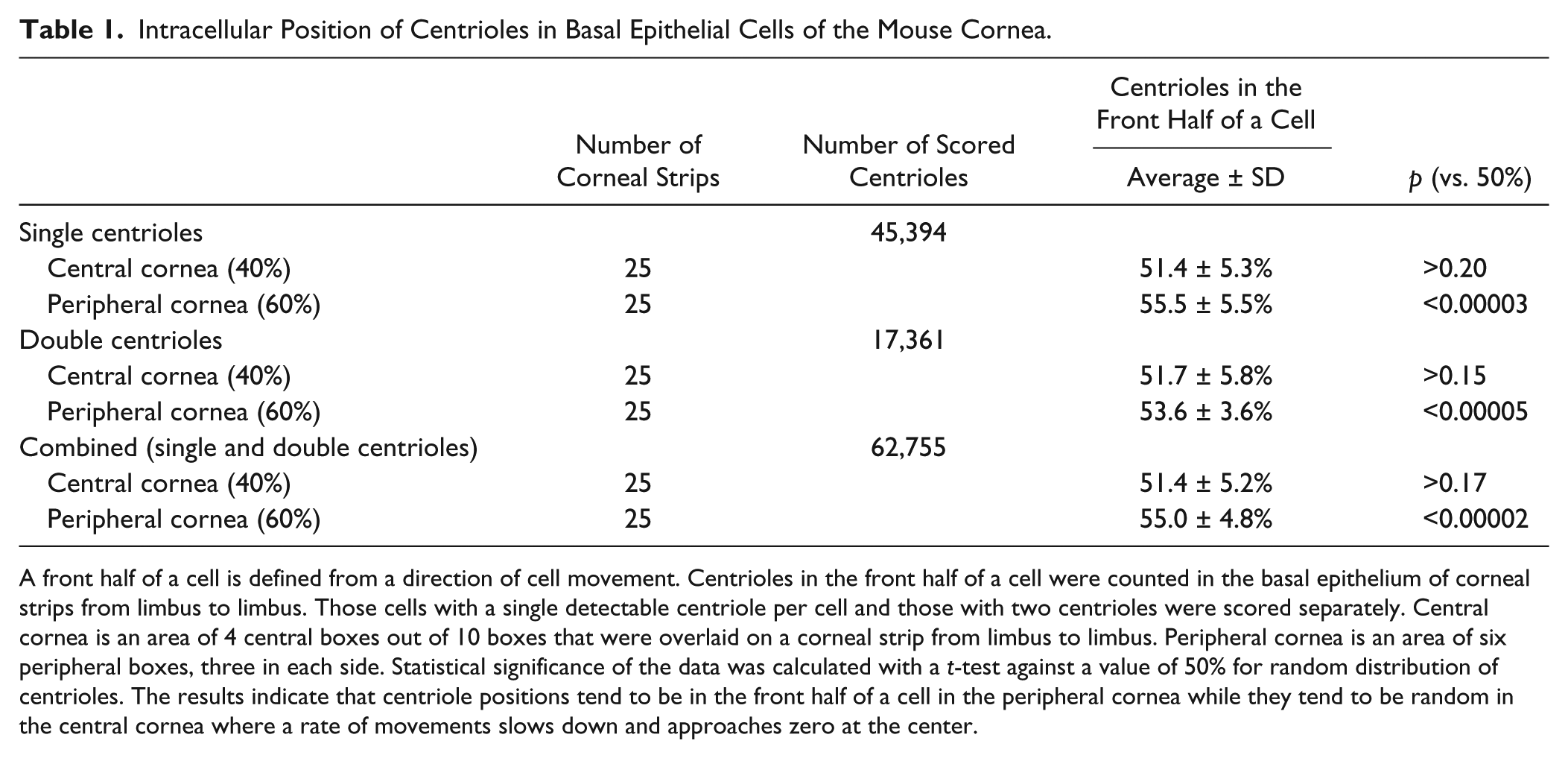
A front half of a cell is defined from a direction of cell movement. Centrioles in the front half of a cell were counted in the basal epithelium of corneal strips from limbus to limbus. Those cells with a single detectable centriole per cell and those with two centrioles were scored separately. Central cornea is an area of 4 central boxes out of 10 boxes that were overlaid on a corneal strip from limbus to limbus. Peripheral cornea is an area of six peripheral boxes, three in each side. Statistical significance of the data was calculated with a t-test against a value of 50% for random distribution of centrioles. The results indicate that centriole positions tend to be in the front half of a cell in the peripheral cornea while they tend to be random in the central cornea where a rate of movements slows down and approaches zero at the center.
Discussion
One of the problems of studying epithelial homeostasis is lack of practical techniques to examine dynamic events such as cell movements. In vivo time-lapse experiments are ideal for this, but examination of individual cell movements is technically demanding and time-consuming. With this background, we set out to find a histological marker that reflects parameters of cell movements—a dynamic event. Our data suggest that a general direction of cell movements is likely to be toward an average position of centrioles within a cell that is determined from a group of moving cells.
Our results showed that the percentage of centrioles that are in the front half of a cell was consistently more than 50%, indicating that centrioles are not distributed randomly within a cell. The direction of movements of individual cells cannot be determined from the centriole position because the correlation is not 100%. However, the data allow for a statistical prediction of a group of cells because epithelial cells move together as a group in the cornea. 11 A power analysis (assuming a normal distribution with alpha=0.05 and beta=0.2) based on the combined centriole data (Table 1) suggests that the average centriole position of a cluster of eight or more neighboring cells in the peripheral cornea is likely to indicate the direction of cell movements.
Cells in the central cornea, however, showed no statistical correlation of centriole position versus cell movement direction. There are two features in the central cornea that are worth consideration: (1) cell movements are not linear but they are curved, frequently generating a track of a logarithmic spiral; and (2) rates of cell movement slow down and approach zero at the center. Perhaps the slow rates of movement may be related to the random centriole positioning, but only future investigation can clarify such relationship.
Several studies have demonstrated that intracellular centriole position could be a marker of cell movement direction,12–15 and our results with homeostatic corneal epithelial cell movements are in line with these reports. A molecular mechanism that leads to the correlation is not known, and thus it is not clear whether a centriole position dictates the cell movement direction or whether a centriole is positioned as a consequent of cell movement. In this regard, it is curious to note that an active role of centrioles has been indicated in cellular polarization,16,17 suggesting that centriole positioning could potentially influence the cell movement direction.
Finally, we would like to discuss the value of the technique presented here. We envision that the technique can be used (1) in vivo with a live mouse or (2) with a histological specimen after fixation. The first use is possible with a transgenic mouse with fluorescently tagged centriole and nuclear molecules; for example, transgenic mice with GFP-labeled centrin 2 20 and GFP-labeled histone H2B 21 have been available (GFP can be substituted with cyan fluorescent protein (CFP), red fluorescent protein (RFP), mCherry, etc.), and we have used CAG::H2B-EGFP mice (Jax stock 006069) for live imaging of corneal basal epithelial cells previously. 22 The second histological use is a direct application of techniques presented in this study. The most valuable feature is that histological specimens can be used to infer dynamic events (i.e., cell movements) albeit in a limited scope; a direction can be inferred but rates of movements remain unknown.
Clearly, more studies are needed to verify what we reported here vigorously with normal and pathological corneas. It is possible that corneas with abnormal epithelial homeostasis, such as those of Pax6+/− mice 23 and Dstncorn1 mice 24 that exhibit abnormal cell movements, are different from normal corneas in intracellular centriole positions. We intend to test this hypothesis with these pathological corneas using the technique described in this study.
The present study excluded mitotic cells and also those cells whose centrioles could not be determined unambiguously by immunostaining, resulting in about 50% of cells being scored. With the understanding of the shortcomings, our results represent an attempt to establish histological markers to study cell movements. We are hopeful that this line of investigation will eventually lead to a robust method of estimating the direction of cell movement (dynamic events) from histological specimens (static observations), assisting studies of corneal epithelial homeostasis.
Footnotes
Competing Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
ES and JZ performed immunohistochemistry, ES performed image analysis, JZ performed corneal preparation, ES, JCM, and TN designed the study, carried out data analysis, and wrote the manuscript. All authors have read and approved the final manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by grants from the Research to Prevent Blindness, Eye Surgery Fund, and NIH grant R01-EY015835.
