Abstract
We determined the expression of ORAI1 protein in rodent and non-rodent tissues using a monoclonal antibody directed against an extracellular loop of the protein. Previous reports using antibodies directed at the C-terminus of ORAI1 have not detected central nervous system (CNS) expression. Our results demonstrate broad tissue expression that includes the CNS using a unique monoclonal antibody specific to an extracellular loop of ORAI1. In addition, we present in situ hybridization (ISH) results using a probe within the middle of the mouse coding region showing CNS expression of Orai1 RNA. We contrast the patterns of rodent and human tissue expression and conclude that rodents have similar expression of ORAI1 in most tissue types when compared to primates, with an important exception being the male reproductive system, where human-specific expression is observed.
Introduction
Calcium is an important second messenger used in the regulation of cell differentiation, gene transcription, and cell-type specific function of many cells. One of the best studied cell types where calcium signaling is critical for cell function is lymphocytes (Feske 2007; Putney 1986). The primary pathway that induces an increase in intracellular Ca2+ signaling in lymphocytes is mediated through store-operated calcium (SOC) influx, and much of SOC is mediated by calcium release-activated calcium (CRAC) channels. Although the mechanism of SOC is not exclusive to CRAC channels, they are perhaps the best described channels that mediate the phenomenon. CRAC channels have been documented to have functions in multiple cell types where SOC is observed outside the immune system (Lewis 2001; Oh-hora 2009; Oh-hora and Rao 2008; Oh-hora et al. 2008; Parekh and Putney 2005).
The CRAC channel is made up of two essential components: stromal interaction molecule 1 (STIM1) and ORAI1. STIM1 is localized in the endoplasmic reticulum (ER) and senses a decrease in calcium levels in the ER through an EF hand region located in the ER lumen (Liou et al. 2005; Roos et al. 2005; Zhang et al. 2005). ORAI1 forms the plasma membrane component of the channel and, when coupled with STIM1, opens and allows for calcium entry from the extracellular space, thus initiating signaling events (Prakriya et al. 2006; Vig et al. 2006; Yeromin et al. 2006).
The main intracellular signaling events that induce an increase in intracellular Ca2+ through SOC are shared by all cell types, although the initiation of the signal from the plasma membrane varies depending on the specific cell type in question. Given an initial signal, phospholipase Cγ1 becomes activated and hydrolyzes phosphatidylinositol-3,4-bisphosphate (PIP2) into diacylglycerol (DAG) and inositol-1,4,5-triphosphate (IP3). IP3 subsequently binds to IP3 receptors (IP3R) located in the ER membrane. The binding of IP3 results in IP3R opening, and subsequent depletion of Ca2+ from the ER store. This ER store depletion triggers store-operated channels, resulting in sustained cytoplasmic Ca2+ influx. This sustained Ca2+ influx is required for persistent calcineurin phosphatase activation, which causes transcriptional activation of downstream genes through dephosphorylation and subsequent nuclear translocation of the transcription factor nuclear factor of activated T cells (NFAT) (Feske 2007; Feske et al. 2003).
The CRAC channel, given its key role in activation of T cells and downstream cytokine-mediated events, is of great interest for the development of immunosuppressive drugs. The importance of CRAC function is perhaps best demonstrated in a subset of severe combined immunodeficiency (SCID) patients who have mutations that render either ORAI1 or STIM1 non-functional or cause the absence of the proteins (Feske et al. 2001; Feske et al. 2006; Feske et al. 2010; Feske et al. 2005; McCarl et al. 2009). These SCID patients have a similar clinical presentation of immunodeficiency, congenital myopathy, and anhydrotic ectodermal dysplasia at birth, and autoimmunity in later life (Feske et al. 2010; McCarl et al. 2009). Although the limited clinical presentation of Orai1-deficient individuals is in sharp contrast to its broad tissue expression, the clinical phenotype of loss-of-function of ORAI1, as well as the biological importance of SOC function in cell types outside the immune system, garners legitimate concern about target-related toxicity of any agent that inhibits CRAC function. Given our own interest in the pharmacological modulation of the CRAC channel (Lin et al. 2013), we wanted to explore what nonclinical species would be relevant to investigate toxicity associated with CRAC channel inhibition. In this paper, we describe immunohistochemistry results using a mouse monoclonal antibody, mAb 266.1, which binds to an extracellular loop of ORAI1 and detects the rodent, cynomolgus monkey, and human proteins. We present a focused look at expression in the central nervous system (CNS) across nonclinical species. Previous reports using antibodies directed at the C-terminus of ORAI1 have not detected CNS expression by immunohistochemistry (IHC) in mouse or human tissues (Gwack et al. 2008; McCarl et al. 2009), although there has been one published report of CNS expression using western blotting of mouse brain extracts (Linde et al. 2013). We also present in situ hybridization (ISH) results using a probe directed to the middle of the RNA transcript for mouse Orai1 where CNS expression is also detected. Comparing the expression patterns across species, we conclude that rodents and primates have similar expression patterns of ORAI1 in most tissue types, with an important exception being in the male reproductive system.
Materials & Methods
Antibody Generation
Crude cell membrane fractions from transient transfected hOrai1-expressing 293 cells were prepared and used as the antigen for conventional immunization of B6/129 mice (The Jackson Laboratory, Bar Harbor, ME). After several rounds of immunization, lymphocytes were released from the spleen and were fused with mouse myeloma cells, Sp2/0-Ag14 (ATCC, CRL-1581), at a ratio of 2.5:1 by electrofusion. Fused cells were seeded in 96-well plates at 2×104 cells/well in 100 µl of BD media supplemented with 10% FBS, 5% Origen Cloning Factor (BioVerisTM; Gaithersburg, MD; Cat# 210001), 1× Penicillin-Streptomycin-Glutamine (Life Technologies; Grand Island, NY; Cat# 10378-016), and 1×OPI (oxaloacetate, pyruvate, and insulin; Sigma-Aldrich; St. Louis, MO; Cat# O-5003). After 24 hr in culture, 100 µl of 2×HAT (0.1 mM hypoxanthine, 0.16 mM thymidine, 4 mM aminopterin; Sigma-Aldrich; Cat# H-0262) was added to each well. Medium was changed 7 days and 10 days post-fusion, and the conditioned media was collected after two days of incubation for primary screening. Positive clones were expanded, single-cell cloned, and confirmed by multiple assays.
Hybridoma supernatants were analyzed via the cell-based FMAT (fluorometric microvolume assay technology) with human Orai1- expressing CHO cells (CHO-hOrai1) in parallel with parental CHO cells. The hybridoma clones containing ORAI1-specific antibodies were selected based on their specific binding to CHO-hOrai1 but not to CHO parental cells. Monoclonal antibodies were partially purified from the expanded hybridoma cultures, and specific binding was confirmed through FMAT and FACS (fluorescence-activated cell sorting), where cross-species reactivity was also evaluated using methods previously described (Lin et al. 2013), using goat
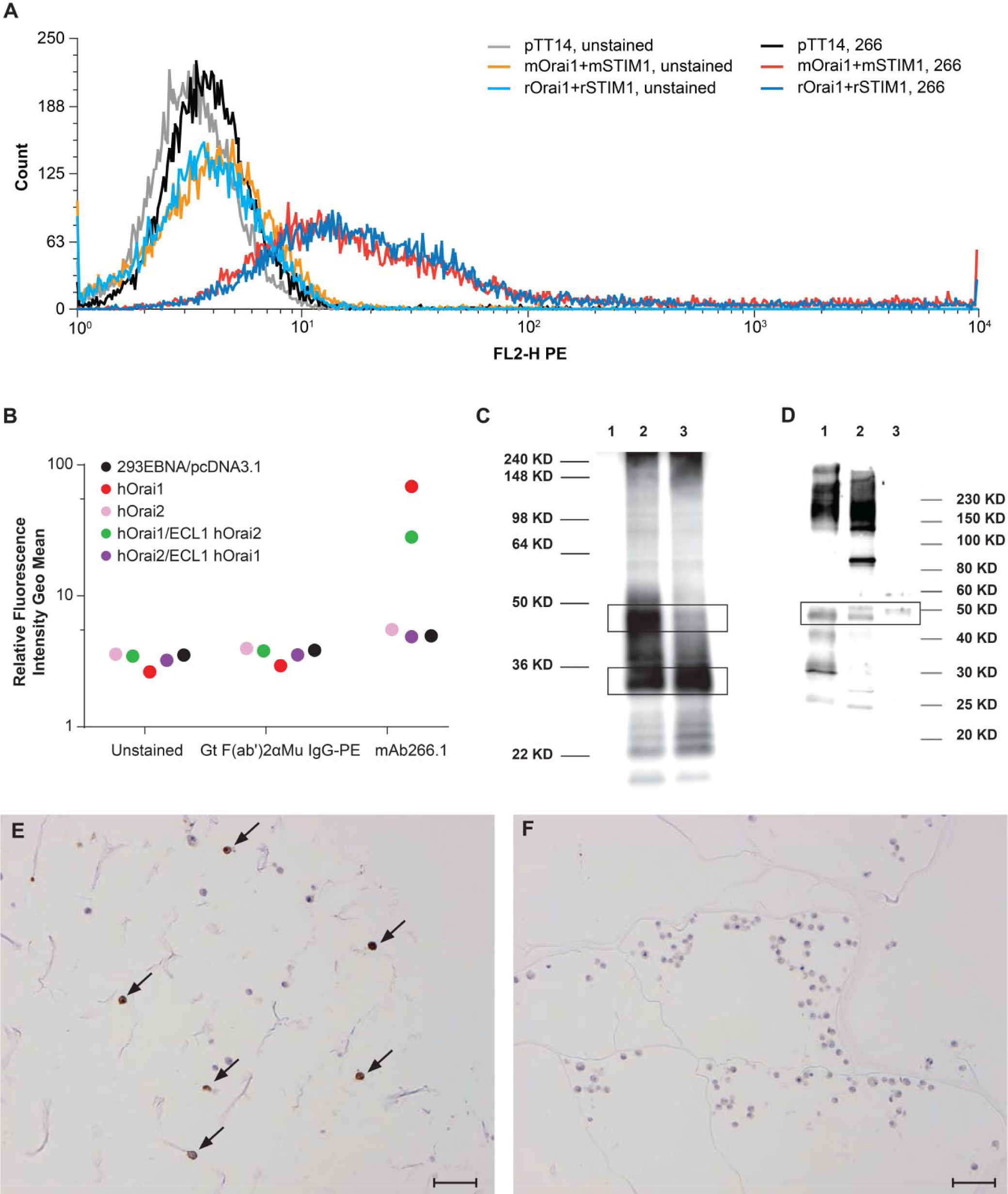
Monoclonal antibody, mAb266.1, is specific for Orai1 and cross-reacts with human, mouse, and rat Orai1. (A) mAb266.1 detects mouse and rat ORAI1 expressing cells by FACS. Note that controls are included for 293 EBNA cells expressing the control vector or mouse or rat constructs for Orai1 and Stim1 in both the presence and absence of mAb266.1. (B) Selectivity of mAb266.1 for binding to ORAI1 but not ORAI2. The geo mean was plotted from FACS experiments with mAb266.1 binding to 293 EBNA cells expressing either a control vector, ORAI1, ORAI2 or a limited set of chimeric proteins. Results are shown along with unstained and secondary antibody controls. mAb266.1 bound specifically to ORAI1 but not ORAI2. A chimera containing the first extracellular loop of ORAI2 showed a minimal decreased in binding of mAb266.1 to ORAI1, whereas a chimera with the first extracellular loop of ORAI1 within ORAI2 did not show increased binding above background. (C) Western blot of mAb266.1 demonstrating cross-reactivity of the antibody to 293 EBNA cells expressing mouse (Lane 2) or rat (Lane 3) Orai1. Lane 1 represents un-transfected 293 EBNA cells. In this cell line, the black boxes highlight that both the glycosylated (~45 kD) and un-glycosylated (~30 kD) forms of the protein are produced in different amounts depending on the construct transfected. (D) Western blot of endogenous ORAI1 protein in human ovary (adult ovary-Lane 1; fetal ovary-Lane 2) and adult spleen (Lane 3) lysates. Both the glycosylated and un-glycosylated forms of ORAI1 are detected in tissue lysates. (E–F) IHC staining of collaplugs of 293 EBNA cells expressing human ORAI1. Positive-expressing cells are marked with black arrows (E) vs. non-expressing control cells (F). Scale bars E and F = 100µm.
Western Blotting
Cell lines expressing mouse and rat Orai1 were generated and lysates prepared as previously described (Lin et al. 2013). Tissue lysates were purchased from Prosci, Inc (Poway, CA; Cat #1316-ovary and #XBL-10422-fetal spleen). Western blots were processed as previously described (Lin et al. 2013).
Animals
Sprague-Dawley rats (Rattus norvegicus) and C57BL6 mice (Mus musculus) of both sexes were used between 6–8 weeks of age and were cared for in accordance to the Guide for the Care and Use of Laboratory Animals, 8th Edition (National Research Council 2011). Rats were pair-housed; female mice were group housed and male mice were single housed at an AAALAC, international-accredited facility in non-sterile ventilated micro-isolator housing on corn cob bedding. All research protocols were approved by the Institutional Animal Care and Use Committee.
Animals had ad libitum access to pelleted feed (2020X, Harlan Laboratories; Madison, WI) and water (reverse osmosis-purified with the addition of 0.20-0.50 ppm chlorine) via an automatic watering system. Animals were maintained on a 12:12 hour light:dark cycle in rooms held at 20–26C with 20-70% relative humidity. Animals had access to enrichment opportunities. All rodents were determined specific pathogen-free for standard Level 1 pathogens, including viruses, bacteria, enteric protozoa, arthropod ectoparasites, and helminth endoparasites.
Immunohistochemistry
Immunohistochemistry was performed on formalin-fixed, 5-µm sections. Deparaffinized tissue sections were heat-treated with Diva Decloaker (Biocare Medical; Concord, CA; cat. # DV2004). Tissue sections were blocked with CAS BLOCK (Life Technologies; Grand Island, NY; cat. # 00-8120) and incubated with antibody at a concentration of 0.25 μg/ml. Tissue sections were quenched with 3% peroxidase solution and detected with Envision Labelled Polymer HRP Mouse (DAKO; Carpinteria, CA; cat. # K4001). Reaction sites were visualized with DAB+ Substrate-Chromagen System (DAKO; cat. #K4007) and counterstained with hematoxylin. mAb266.1 was generated by Amgen, as described above. The polyclonal rabbit anti-ORAI1 antibody was purchased from Prosci Inc. (catalog #4281).
In Situ Hybridization (ISH) Probes
A cDNA template containing a 312-bp fragment of the mouse ORAI1 gene was generated by polymerase chain reaction and cloned into the pCR4-TOPO vector (Invitrogen; Carlsbad, CA). The cDNA vector template was verified by sequencing and corresponds to mid-coding region nucleotides 481-792 (Genbank NM_175423) of the mouse ORAI1 gene. An antisense 33P-labeled RNA probe was synthesized with T7 RNA polymerase after linearization with SpeI restriction enzyme. A control sense 33P-labeled RNA probe was synthesized from the same template with T3 RNA polymerase after linearization with NotI restriction enzyme.
Tissues
Tissues used for immunohistochemistry were derived from a normal mouse necropsy or normal human tissue set, with the exception of two human tonsil samples that showed non-specific inflammation. Human tissue microarrays were procured from US Biomax (Rockville, MD). Cynomolgus monkey tissue microarrays were purchased from Asterand US (Detroit, MI). Tissues for in situ hybridization consisted of immersion formalin-fixed, paraffin-embedded material from a normal mouse from which 5-µm sections were cut under RNase-free conditions.
ISH Method
Isotopic in situ hybridization was performed as described (Oliner et al. 2012). Briefly, sections were deparaffinized, rehydrated, and pretreated by microwave heating to 100C for 10 min in a citric acid buffer solution (Citra, Biogenex; Freemont, CA). Tissue sections were then subjected to deproteination, proteinase K treatment, acetylation, and pre-incubation in hybridization buffer for 2 hr in a humidified chamber at 56C. After pre-hybridization, the sections were hybridized with 1.0×106 cpm of 33P-labeled probe at 56C for 16 hr. After overnight hybridization, all slides were treated to RNase digestion, followed by a series of SSC washes with highest stringency of 0.1× SSC at 55C for 30 min. The slides were coated with Kodak NTB emulsion (Eastman Kodak, Rochester, NY) and exposed for 3 weeks in the dark at 4C, developed, and then counterstained with hematoxylin and eosin.
Results
Generation and Characterization of mAb266.1
mAb266.1 was isolated during a screen for monoclonal antibodies in C57BL6 mice using membrane preparations from cells expressing human ORAI1. After an initial screen to ascertain binding to cells expressing human ORAI1, candidate antibodies were further screened by FACS staining using mouse and rat Orai1-expressing cells. mAb266 was selected for subcloning to generate mAb266.1.
mAb266.1 detects both glycosylated and un-glycosylated forms of mouse and rat ORAI1 by FACS and western blotting in cell lines (Fig. 1A, 1C, 1D). mAb266.1 is specific for ORAI1 and, using a limited chimera set between ORAI1 and ORAI2, we concluded that the antibody likely binds to the second external loop of ORAI1 (Fig. 1B). In addition, the antibody can detect endogenous human ORAI1 protein in tissue lysates from ovary and spleen (Fig. 1D). As a final test of the suitability of this antibody for use in immunohistochemistry, we evaluated the ability of mAb266.1 to stain cell pellets expressing human ORAI1 (Fig. 1E, 1F). mAb266.1 demonstrated good sensitivity and specificity based on this characterization; therefore, we proceeded with our IHC experiment using tissues from rat and human, in addition to a focused set of CNS tissues from cynomolgus monkey.
Tissue Microarray (TMA) IHC
The results of the IHC assessment of a range of rat and human tissues are presented in Table 1. In addition, tissues from the nervous system of a non-human primate (cynomolgus macaque) were included in the evaluation and these results are also summarized in Table 1. Overall, the distribution of staining was fairly widespread in rat and human tissues. Significant staining was observed in the nervous system, muscle, surface epithelium, testes, endocrine pancreas, kidney, and lung. Widespread staining was also evident in the nervous system of the cynomolgus macaque.
IHC Expression of ORAI1 in Normal Rodent and Primate Tissues using Monoclonal Antibody, mAb266.1.
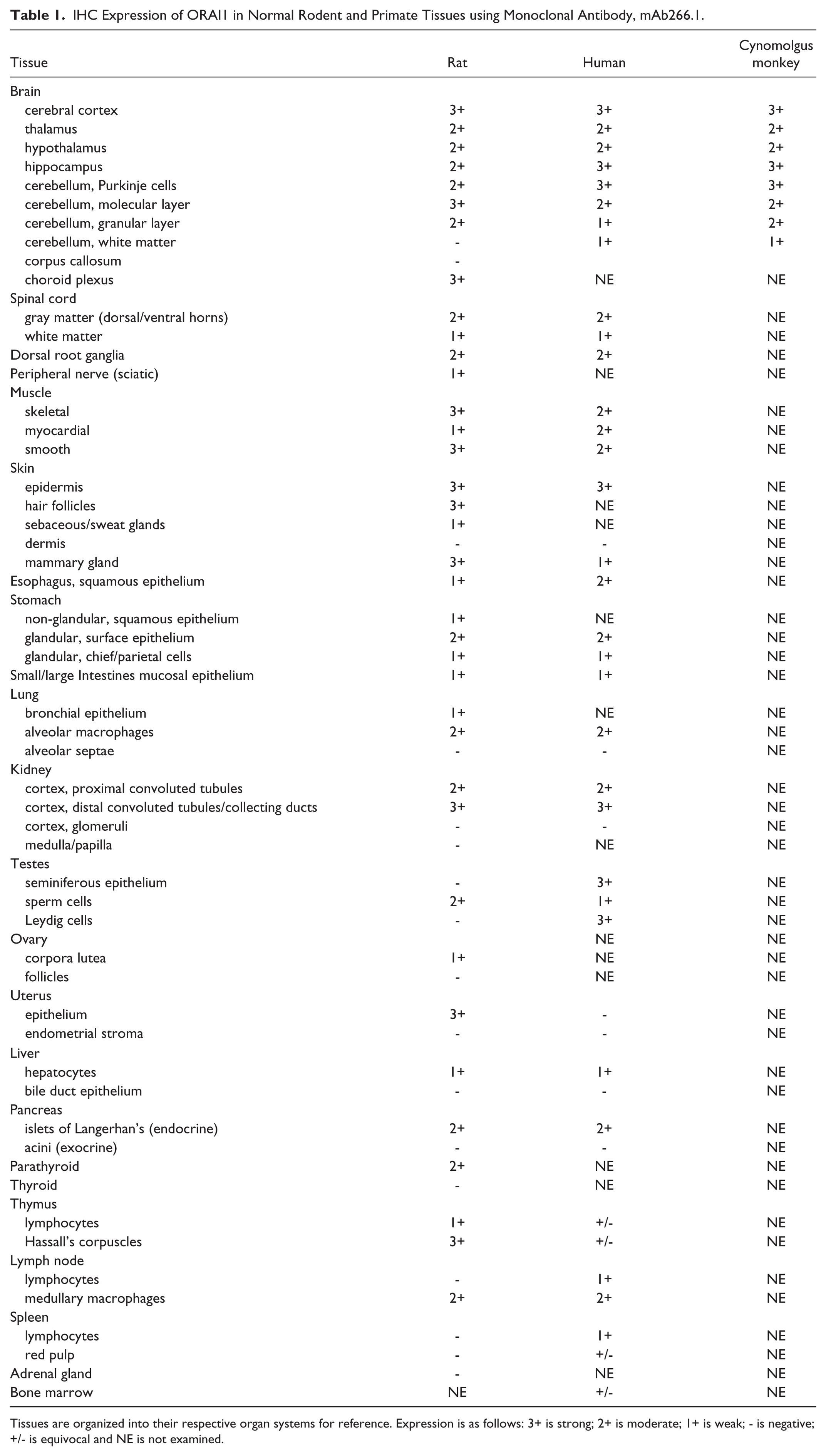
Tissues are organized into their respective organ systems for reference. Expression is as follows: 3+ is strong; 2+ is moderate; 1+ is weak; - is negative; +/- is equivocal and NE is not examined.
Staining in the nervous system had a similar pattern of distribution in rat, human, and cynomolgus monkey. Staining was observed in the brain, spinal cord, and root ganglia. In brain, moderate to strong staining was evident in multiple anatomic locations but most notably in the cerebral cortex, hippocampus, and cerebellum. In rat brain (Fig. 2A), staining in the cerebral cortex appeared to be diffusely localized to the gray matter neuropil, with weak to moderate staining of neurons and glial cells. The underlying white matter tracts were negative. Within the lateral ventricles, cells of the choroid plexus were strongly stained and showed a membranous distribution (Fig. 2A). A similar pattern of staining was observed in the hippocampus, where glial cells and neurons were negative or exhibited a weak, membranous staining, whereas the surrounding neuropil and associated dendritic processes had strong staining (Fig. 2B–2C). In the cerebellum (Fig. 2D–2F), there was moderate to strong staining of the molecular layer, Purkinje cells, and granular layer.
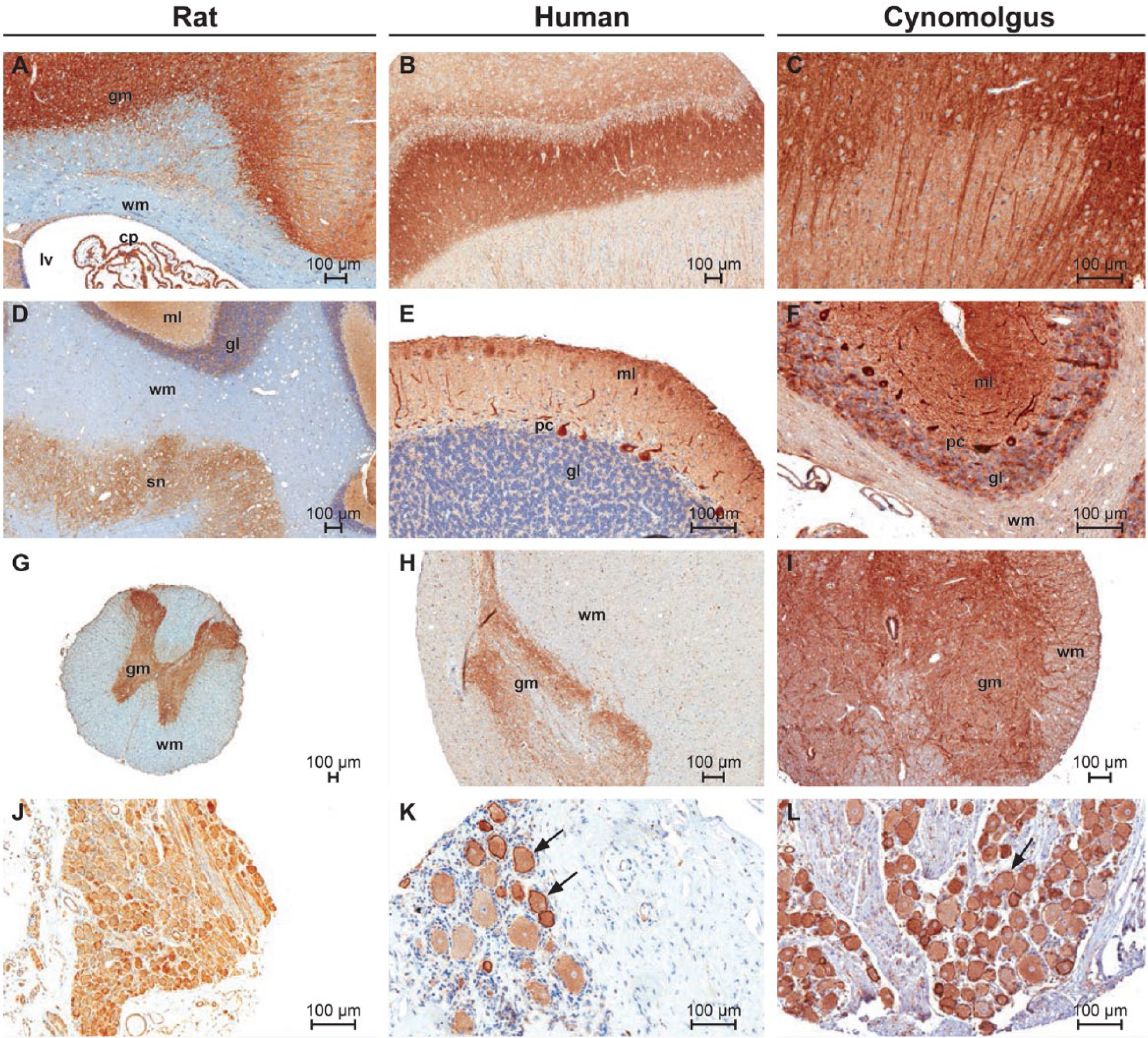
Immunohistochemical staining of normal central nervous system tissues from rat, human and cynomolgus macaque with ORAI1 monoclonal antibody 266.1. (A) Rat cerebral cortex; gray matter (gm), white matter (wm), choroid plexus (cp). (B) Human hippocampus. (C) Cynomolgus hippocampus. (D) Rat cerebellum; molecular layer (ml), granular layer (gl) and brain stem nuclei (sn). (E–F) Human and cynomolgus cerebellum. (G-I) Rat, human and cynomolgus spinal cord. (J–L) Dorsal root ganglia. (K) Note membranous pattern of staining in some neuronal cell bodies (arrows). Scale bars A–L = 100µm.
In the rat (Fig. 2), the underlying cerebellar and brain stem white matter tracts were negative, whereas, in the human and cynomolgus monkey tissue, white matter was generally weakly positive. In the rat, the neuropil associated with brainstem nuclei also had strong, localized staining, with some groups of neuronal cell bodies exhibiting weak cytoplasmic or membranous staining. In the spinal cord (Fig. 2G–2I), there was moderate to strong staining of the gray matter, with weak to moderate staining of individual nerve fibers in the white matter. The pattern of staining in the spinal cord gray matter was similar to that observed in gray matter elsewhere, in that staining was most prominent in the neuropil whereas the neuronal cell bodies and glial cells had weak membranous staining or were negative. In the dorsal root ganglia (Fig. 2J–2L), neuronal cell bodies had moderate to strong staining. Some scattered neurons had a pronounced membranous distribution, a pattern that was consistent in all species. The surrounding bundles of nerve fibers were weakly to moderately positive. Nerve fibers from peripheral nerves (sciatic) showed weak staining in individual fibers.
Staining was observed in all muscle types of both rat and human. Staining of similar intensity was generally observed in human and rat smooth muscle from multiple anatomic locations, including gastrointestinal, vascular, and reproductive systems. In rat uterus (Fig. 3A), the smooth muscle layer was positive whereas the endometrial stroma was negative. Smooth muscle from human prostate (Fig. 3B) had positive staining of a similar intensity and subcellular distribution. Human skeletal and myocardial muscle showed moderate, diffuse staining (Fig. 4A–4B), which was cytoplasmic, had a fine, granular pattern, and was evenly distributed within individual myofibers. Weak staining occurred in rat myocardial muscle and was distributed in a diffuse, cytoplasmic pattern in individual myofibers.
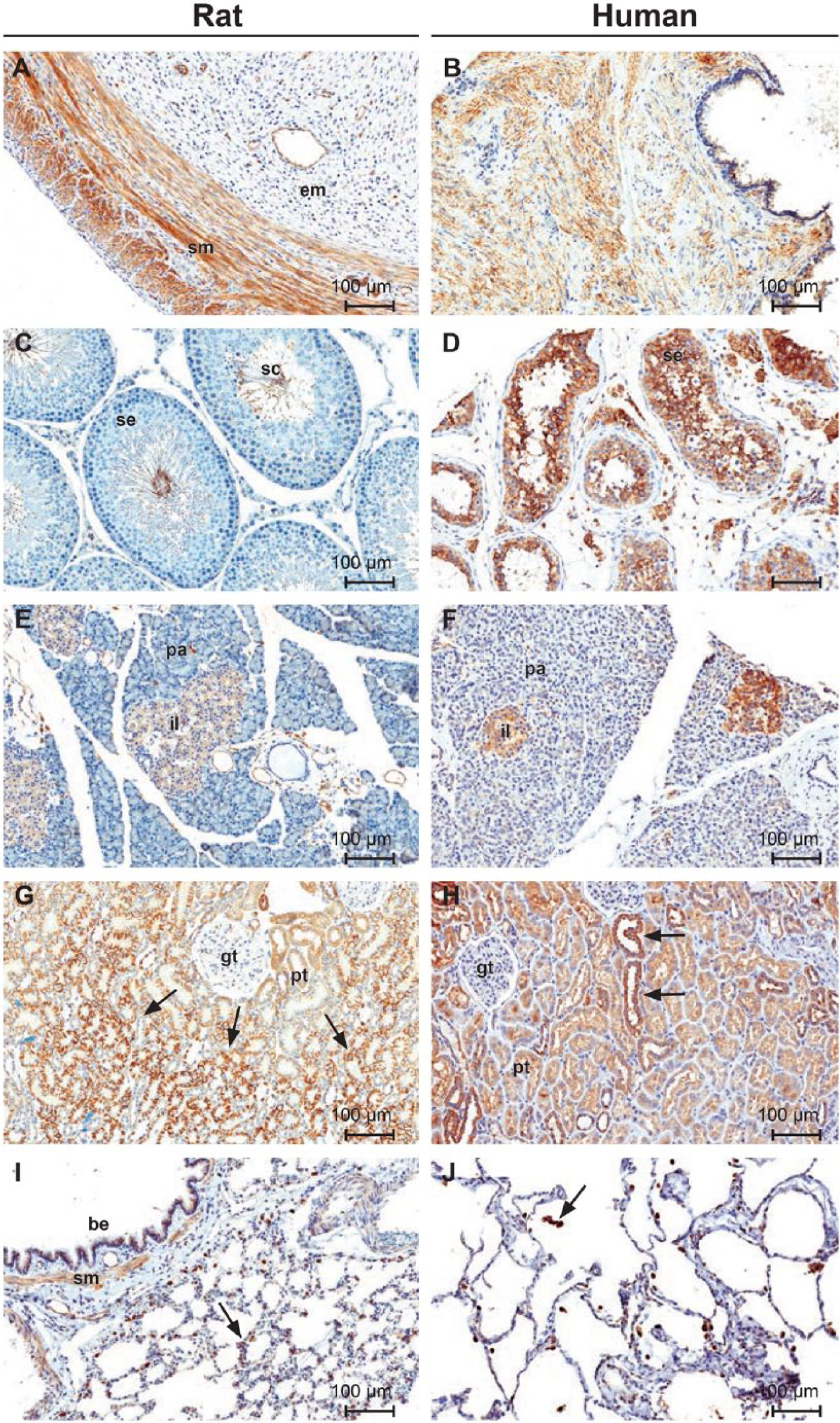
Immunohistochemical staining of normal rat and human tissues with ORAI1 monoclonal antibody mAb266.1. (A) Uterus; smooth muscle (sm), endometrium (sm). (B) Prostate gland. (C–D) Testes; spermatogenic epithelium (se), sperm cells (sc), interstitial cells (ic). (E–F) Pancreas; islets of Langerhans (il), pancreatic acini (pa). (G) Kidney; glomerular tuft (gt), proximal convoluted tubules (pt). Note the increased intensity of membranous staining in tubules at the corticomedullary junction (arrows). (H) Kidney; glomerular tuft (gt), proximal convoluted tubules (pt). Note the increased staining of the distal convoluted tubules and collecting ducts (arrows). (I–J) Lung; bronchiolar epithelium (be). Note staining of alveolar macrophages (arrows). Scale bars A–J = 100µm.
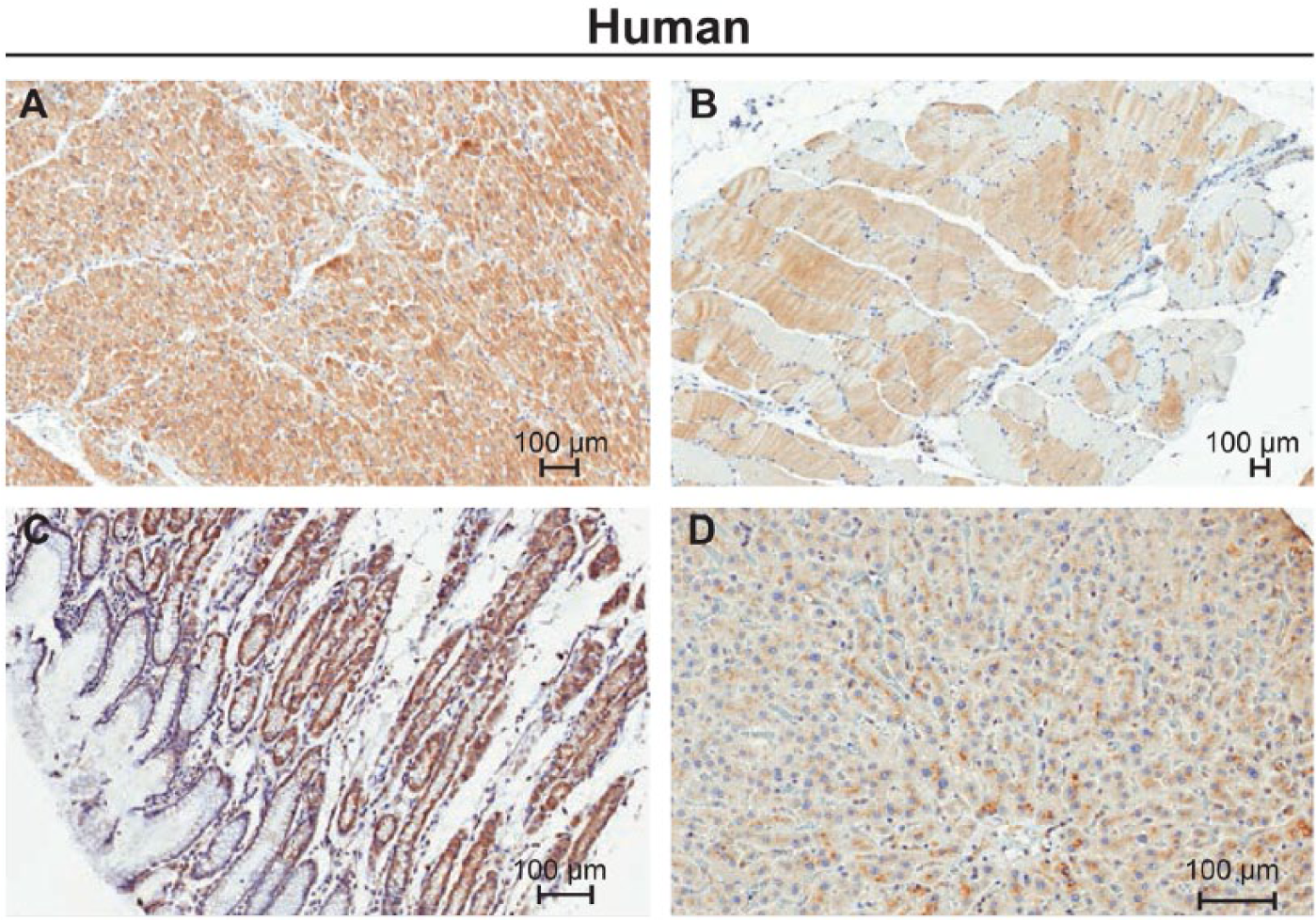
Immunohistochemical staining of normal human tissues with ORAI1 monoclonal antibody mAb266.1. (A) Heart, (B) Skeletal muscle, (C) Stomach, and (D) Liver. Scale bars = 100µm.
Staining was observed in multiple epithelial surfaces of rat and human, most notably in skin. Staining of a lesser intensity was also observed in gastrointestinal and glandular epithelium. In skin, staining was present in the epidermis and hair follicles, with minimal expression in sebaceous glands. Staining in the epidermis was uniform across all cell layers and exhibited a distinct distribution along cellular membranes and at intercellular junctions. In the esophagus and non-glandular stomach (rat), weak staining was present mostly along the basal cell layer of the stratified squamous epithelium. In the glandular stomach of rat and human, weak staining was present in glandular epithelial cells (chief and parietal cells), with moderate staining observed in the surface epithelial cells in both species. Human staining is shown in Figure 4C. In the small and large intestines, weak staining was present in the villous and crypt epithelial cells. Weak to strong staining was also observed in prostate, uterine, and mammary gland epithelium.
Significant species differences in staining were observed in the male reproductive system. In rat testes (Fig. 3C), luminal sperm cells were moderately positive whereas seminiferous epithelium and interstitial cells were negative. In contrast, strong staining was present in the seminiferous tubules and interstitial cells of human testes (Fig. 3D); a prominent membranous pattern was evident in both anatomic structures. In rat epididymis, there was staining of the tubular epithelial cells, smooth muscle, and luminal sperm cells. Human epididymal tissue was not available for examination.
In rat ovary, weak staining was present in most corpora lutea, whereas developing follicles were negative. Human ovary was not available for examination.
Rat (Fig. 3E) and human (Fig. 3F) pancreas showed weak to moderate staining of the islets of Langerhan’s. Islets exhibited a uniform cytoplasmic staining in all cell types. Pancreatic acini were negative in both species. Of the remaining rat endocrine tissues, only the parathyroid gland had moderate staining with a clear membranous pattern.
In rat kidney (Fig. 3G), staining had a distinct regional distribution, which was most prominent along the corticomedullary junction. Moderate staining was present in the cortex, whereas the renal medulla and papilla were negative. In the cortex, there was diffuse staining of proximal convoluted tubules, but the glomeruli were negative. Cells of the proximal convoluted tubules had moderate diffuse cytoplasmic staining. Cells of the distal convoluted tubules and collecting ducts had strong staining that was more prominent in the apical domain of the cells. Strong staining was present in tubules along the corticomedullary junction, an area that correspond mostly with the straight segment of the proximal convoluted tubules. Individual cells had strong staining with a distinct membranous pattern that involved the apical, basal, and basolateral aspects of the cell. The pattern of staining in human kidney was similar; however, only the renal cortex was available for examination.
Alveolar macrophages in rat and human lung were positive whereas cells of the alveolar lining were negative (Fig. 3I, 3J). Rat bronchiolar epithelium and surrounding smooth muscle were also positive. Equivalent structures in human lung were not available for examination.
In rat liver, individual hepatocytes had a membranous distribution of staining that was more prevalent in the sinusoidal domain of the cells. Staining was distributed across all regions of the hepatic lobule with less intensity in periportal areas. In human liver, weak staining was observed in centrilobular hepatocytes. Unlike the rat, individual human hepatocytes had a coarsely granular pattern of staining distributed in the basolateral (canalicular) domain of the cells (Fig. 4D).
CNS expression by IHC has not been reported previously and, in fact, was clearly excluded in previous reports for both mouse and human tissues using polyclonal antibodies directed against the C-terminus of the protein. To confirm this difference for ourselves, we performed a second human tissue microarray experiment contrasting mAb266.1 and a commercially available polyclonal antibody similar to the one previously reported (McCarl et al. 2009) (Supplemental Table 1). The expression patterns outside the CNS were grossly the same for these two antibodies with the only clear difference in the CNS where mAb266.1 was able to detect ORAI1 protein. The Allen Brain Atlas shows no brain expression using an in situ hybridization (ISH) probe that spans the C-terminal coding region of the protein, and overlaps with the polyclonal antibody binding epitope (Sunkin et al. 2013) (Fig. 5).
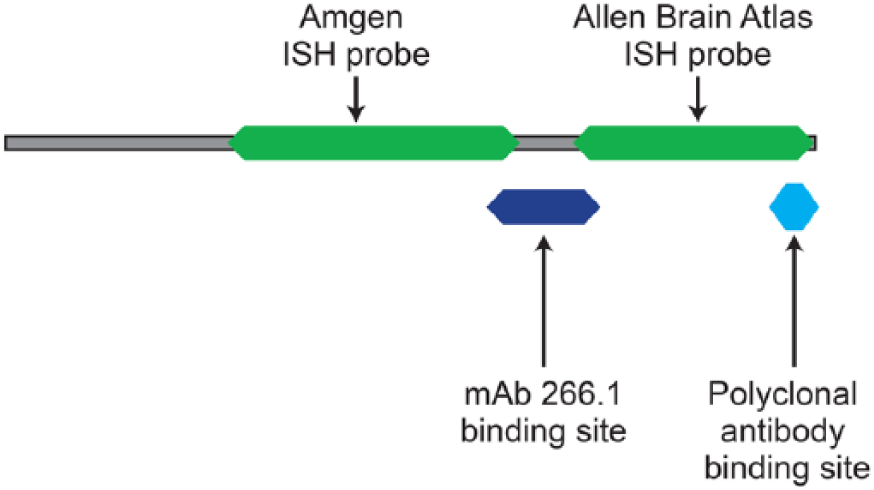
Location of reagent binding sites mapped to the mouse ORAI1 protein sequence. The monoclonal antibody used for IHC in this study, mAb266.1, binds to the second extracellular loop of the protein, whereas the polyclonal antibody binds to a region in the C-terminal tail of the protein. The ISH probe for this study has been mapped to its respective protein sequence. The Allen Brain Atlas probe is shown for comparison (Sunkin et al. 2013).
Mouse ISH
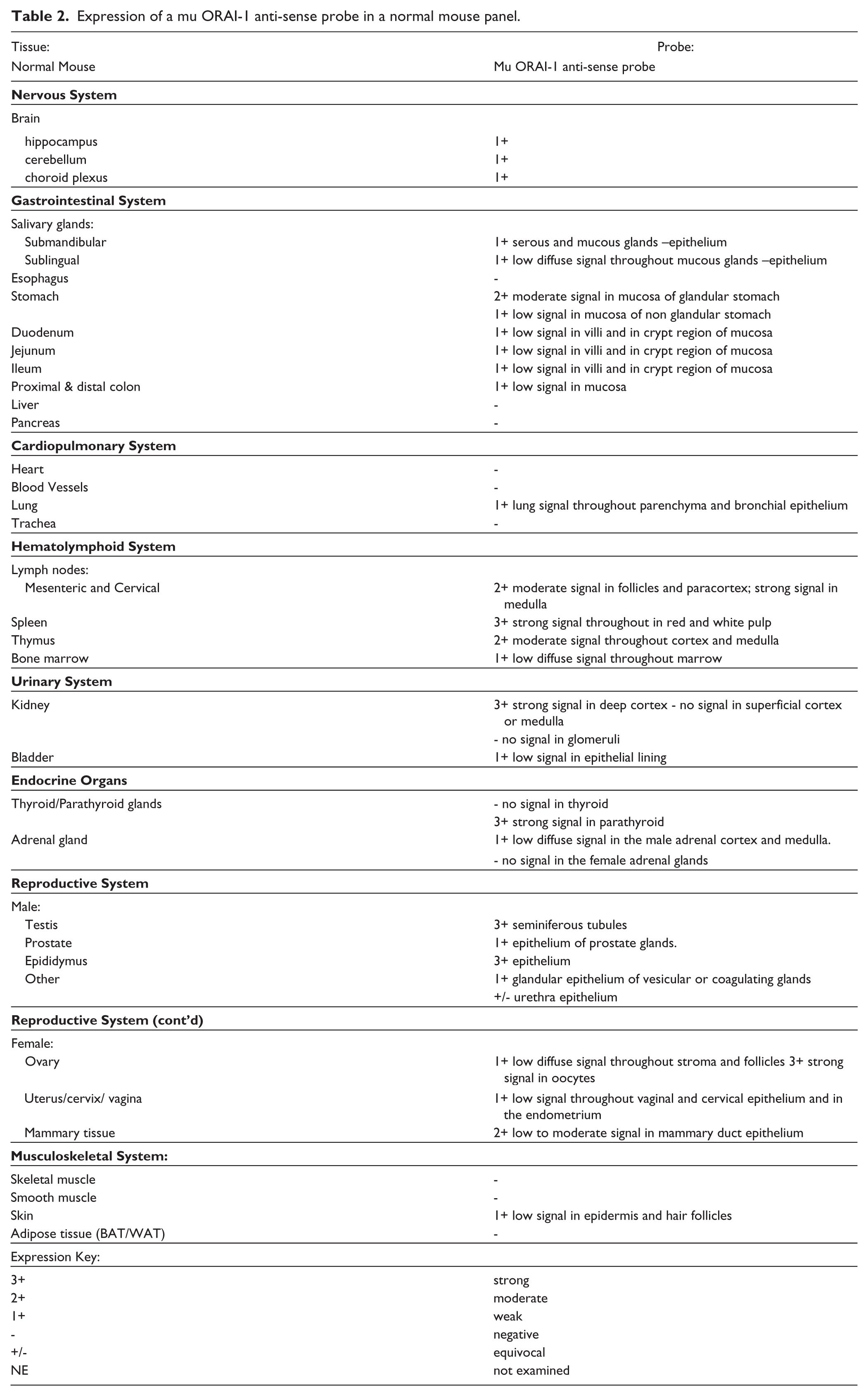
We performed ISH on a set of normal mouse tissues using a probe directed against the middle of the Orai1 message in a similar location to the coding region of the binding site for mAb266.1 (Fig. 5). Table 2 details the results of that experiment and a selected image set is shown in Figure 6. The negative control sense probe was negative in most tissues, with the exception of low background staining observed in densely cellular regions of the lymph node, spleen, thymus, marrow, testis, and mammary duct epithelium. Positive Orai1 expression was determined when the signal from the antisense probe exceeded this background. Using this probe, there is clear CNS expression and broad expression across other tissue types, mimicking our IHC results. There is clear CNS expression detected using this probe in addition to its broad expression across other tissue types, which also mimics our IHC results. For example, strong Orai1 mRNA expression was observed in the mouse parathyroid (Fig. 6B) similar to the protein expression seen by IHC. However, we noted some interesting exceptions. For instance, ISH was not able to detect Orai1 in any muscle type, even though protein expression has been well documented in our analysis as well as in literature reports (Gwack et al. 2008; McCarl et al. 2009). Also of note is the lack of detectable message in the liver or pancreas using ISH when compared to our IHC results. RNA expression of Orai1 is reported for human tissues between 1-10 copies per cell, and this is based on the Broad GTex RNA-seq analysis (GTex Consortium 2013). Given this low level of RNA expression for Orai1, we are very close to the sensitivity limit of ISH; therefore, results across tissues may be inconsistent using this method. We consider the IHC results to be much more robust given the relatively higher protein levels when compared to the RNA expression of ORAI1.
Expression of a mu ORAI-1 anti-sense probe in a normal mouse panel.
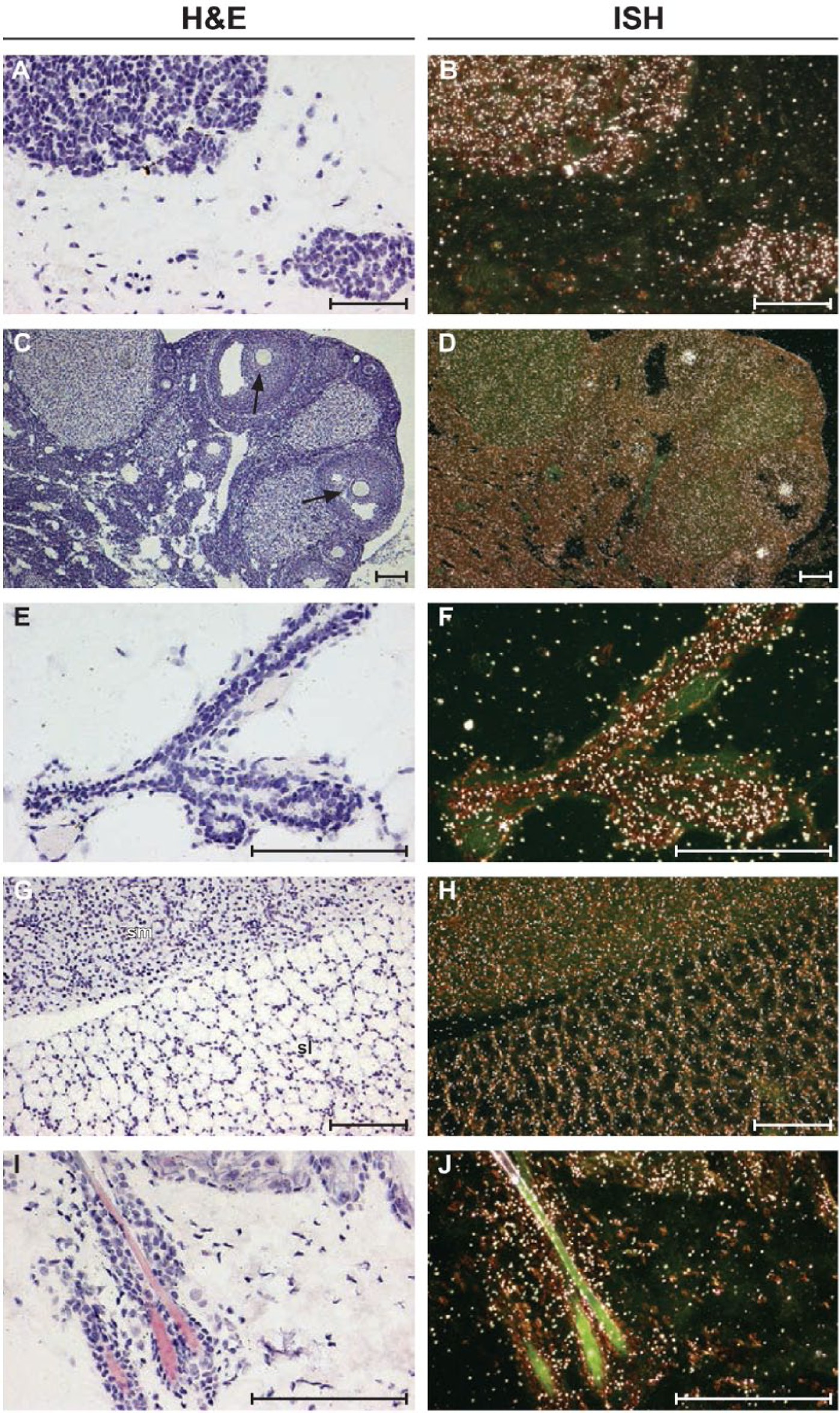
In situ hybridization images of normal mouse tissues for Orai1. Antisense ISH dark-field images are shown on the right; H&E-stained bright-field images are shown on the left. (A–F) Strong Orai1 mRNA expression is detected in the parathyroid gland (A–B), oocytes of ovary tissue (arrows; C–D), and mammary duct (E–F). Low Orai1 mRNA expression is detected in the submandibular (sm) and sublingual (sl) salivary glands (G–H) and in the epidermal cell layer and hair follicle of the skin (I–J). Scale bars A–J = 100µm.
The set of mouse tissues used in the ISH study provided an expanded tissue list from our rat tissue set. Of particular importance was the female reproductive system, because we were able to obtain ovaries from young mice, which allowed us to see clear expression in the follicles, consistent with previous observations of the expression of protein in ovum from multiple species (Fig. 6D) (reviewed in (Martín-Romero et al. 2012)). In addition, we were able to detect expression in the mouse salivary gland, which had been previously reported (Fig. 6H) (Cheng et al. 2008), as well as in the skin and hair follicles (Fig. 6J) (Gwack et al. 2008).
Discussion
Given our interest in the pharmacological modulation of the CRAC channel and ORAI1 in particular (Lin et al. 2013), we examined the tissue expression of ORAI1 across rodent and non-rodent species with the goal of identifying a relevant toxicology test species. Expression of a protein does not always correlate with function in any particular tissue and, in fact, ORAI1-deficient human patients exhibit a limited spectrum of defects as compared with the broad tissue expression of this ion channel (McCarl et al. 2009). However, if there is no expression of a protein in a test species, this may exclude it from consideration as a relevant toxicology species. In the case of ORAI1, there is only one major difference between the primate and rodent, and this is its expression within the male reproductive tract.
We wanted a reagent that could specifically recognize both rodent and non-rodent ORAI1 to characterize the expression of the protein across species, and mAb266.1 was well suited to our needs. The antibody is capable of detecting cell-surface ORAI1 using FACS and binds to an external loop in the middle of the protein, in contrast to the majority of polyclonal antibody reagents against ORAI1, which are specific for a peptide located in the C-terminus of the protein.
We detected significant ORAI1 CNS expression in rodent and non-rodent species using mAb266.1, which could not be detected using the polyclonal C-terminal antibody. We confirmed this difference at the message level using ISH in the mouse using an ISH probe that is just 5’ to the binding region for mAb266.1 in the protein. Northern blot of Orai1 mRNA using a probe similar to our ISH probe has also previously detected mouse brain expression (Gross et al. 2007) in addition to another report using qPCR techniques (Czeredys et al. 2013). Consistent with the hypothesis that there is a CNS-specific truncated form of ORAI1, ISH was performed using a probe that binds 3’ to the one described in this study, as part of the Allen Brain Atlas project; no CNS expression was apparent using that probe (Sunkin et al. 2013). Although there is only one predicted full-length transcript for the mouse, it is interesting to note that there is a transcript in the rat that is predicted to truncate the protein at amino acid 208, as determined using Ensembl, as well as an even shorter transcript in humans (Flicek et al. 2014). What function a truncated form of ORAI1 would have within the CNS and the consequence of this truncation in terms of its inhibition are unknown, although there are reports of store-operated calcium flux in cerebellar granular neurons regulating neuronal excitability (Lalonde et al. 2014) and in mouse neuronal progenitor cells (Somasundaram et al. 2014). Given that human patients with an activating mutation in ORAI1 exhibit miosis, there may also be a role for CRAC in the parasympathetic system (Nesin et al. 2014).
We demonstrated strong Orai1 parathyroid expression by ISH in mouse and by IHC in the rat. The human parathyroid was not available for examination in our TMAs, but expression has been previously reported using a C-terminal-directed polyclonal antibody to ORAI1 (McCarl et al. 2009). The kidney cortex-specific expression of ORAI1 is suggestive of a role for this protein in the collecting tubules, although there have been no reports in the literature of such a phenotype in ORAI1-deficient mice or humans. Given the specific expression of ORAI1 in both the parathyroid and kidney collecting tubules, it is tempting to speculate that ORAI1 may play a role in the regulation of serum calcium levels, which would be consistent with the association of genetic variants in ORAI1 with nephrolithiasis in the Taiwanese population (Chou et al. 2011).
We have also been able to confirm expression of ORAI1 in primary tissues that was previously only reported in cell lines, including the lung epithelium. There have been multiple reports of ORAI1 expression in the lung epithelium of human and mouse, and some functional studies have been performed in rodents (Abcejo et al. 2012; Balghi et al. 2011; Gross et al. 2007; Lin et al. 2010; McCarl et al. 2009; Suganuma et al. 2012) . It is of note that surviving human patients deficient in ORAI1 protein and/or function suffer from recurrent lung infections even after bone marrow reconstitution (S. Feske, personal communication). Whether this reflects a lung-specific role for ORAI1 remains to be determined.
In general, the rest of our observations were consistent with those previously reported in the literature. One tissue where the literature reports of expression are mixed is mouse heart muscle. Our results show no expression of Orai1 in mouse heart, but both mAb266.1 and a polyclonal antibody to ORAI1 detected expression in rat and human heart. Consistent with the mouse ISH result, negative protein expression has been reported in mouse heart using a custom-made polyclonal C-terminal antibody to ORAI1 (Gwack et al. 2008). However, using northern blotting to detect mouse Orai1, a separate group has reported positive expression in the heart (Gross et al. 2007). The reason for such disparate results is not clear.
We were not able to profile ovary expression thoroughly in our study due to a lack of appropriate tissues, although we observed strong mRNA expression by ISH in mouse ovum, and protein expression by IHC in rat corpus luteum. There have been previous reports of ORAI1 expression in mouse, pig, and human ova, and these observations could be confirmed in further-focused studies using appropriate female reproductive tissues (reviewed in (Martín-Romero et al. 2012)).
There is a literature report of mouse salivary gland protein expression (Cheng et al. 2008), although the antibody used in that publication has been shown to be non-specific in western blots, so those results should be interpreted with caution (Lin et al. 2013). We observed strong salivary gland expression using mouse ISH; however, salivary gland tissue was not represented on our tissue micro-arrays (TMA), and so we are unable to confirm this protein expression. SOC flux is important for saliva secretion (reviewed in (Parekh and Putney 2005)) and, therefore, further characterization of salivary gland ORAI1 expression and the potential impact of ORAI1 inhibition in this tissue may be warranted.
The only significant species-specific difference in expression occurred within the male reproductive system. Although sperm cells were positive for both rat and human, human tissue showed additional expression within both the Leydig cells and seminiferous epithelium; therefore, caution should be taken in judging the risk of male reproductive liability from ORAI1 inhibition using rodents as the toxicology species.
In conclusion, mAb266.1 is a useful reagent for evaluating ORAI1 expression across rodent and non-rodent species, particularly within the nervous system.
Supplementary Material
Supplementary material
Footnotes
Acknowledgements
Thanks to Diane Duryea for pathology support. Thanks to David Smith and Mehrdad Ameri for helpful discussions.
Declaration of Conflicting Interests
All authors were employed at Amgen Inc., Thousand Oaks, California at the time of this study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
