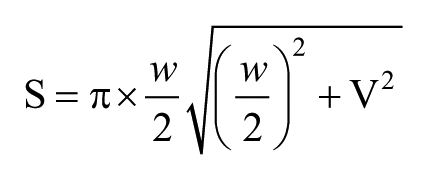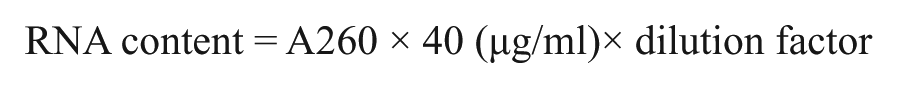Abstract
Intrauterine growth restriction (IUGR) is a very common problem in both piglet and human neonate populations. We hypothesized that IUGR neonates have impaired intestinal mucosal immunity from birth. Using neonatal piglets as IUGR models, immune organ weights, the weight and length of the small intestine (SI), intestinal morphology, intraepithelial immune cell numbers, levels of cytokines and immunoglobulins, and the relative gene expression of cytokines in the SI were investigated. IUGR neonatal piglets were observed to have lower absolute immune organ weight and SI length, decreased relative weights of the thymus, spleen, mesenteric lymph node, and thinner but longer SIs. Damaged and jagged villi, shorter microvilli, presence of autophagosomes, swelled mitochondria, and decreased villus surface areas were also found in the SIs of IUGR neonatal piglets. We also found a smaller number of epithelial goblet cells and lymphocytes in the SIs of IUGR neonates. In addition, we detected reduced levels of the cytokines TNF-α and IFN-γ and decreased gene expression of cytokines in IUGR neonates. In conclusion, IUGR was shown to impair the mucosal immunity of the SI in neonatal piglets, and the ileum was the major site of impairment.
Introduction
Intrauterine growth restriction (IUGR) refers to the impaired growth and development of a mammalian embryo/fetus or its organs during pregnancy, which is a prevalent problem in both human medicine and animal production (McMillen and Robinson 2005; Wu et al. 2006). Approximately 5-10% of human neonates suffer from IUGR caused by multiple factors, including inadequate nutrient intake, environmental stress, and dysfunction of the placenta or uterus (McMillen and Robinson 2005). Pigs, as a domestic animal with multifetal pregnancies, exhibit the most severe naturally occurring IUGR due to placental insufficiency (Anderson and Parker 1976). IUGR not only can cause high mortality and morbidity in neonates but also has a permanent effect on postnatal growth and impairs long-term health (Widdowson 1971; Wu et al. 2006).
Previous studies have found that IUGR affects the thymus size and histopathology in both IUGR animal models and in humans (Lansdown 1977; Contreras et al. 2011). IUGR also alters T lymphocyte cell numbers and impairs cellular immunity in neonates (Contreras et al. 2011; Zhong et al. 2012). Previous studies have demonstrated that IUGR impairs the development of the small intestine (SI), damages the SI structure, and changes the bacterial colonization of the intestine (Wang et al. 2005; D’Inca et al. 2010a). The SI is one of the most important routes for both nutrition absorption and antigen entry (Gaskins 1996), and different sections of the SI have different biological characteristics. Numerous cells and bio-activators compose the barriers in the SI, including mechanical, mucus, immunological, and microbiota barriers (Pitman and Blumberg 2000; Clavel and Haller 2007), to prevent antigen invasion. However, studies of the effects of IUGR on mucosal immunity in the SI in neonates are relatively rare, and few studies have been published on the mucosal immunity of different parts of the SI. We hypothesized that IUGR may impair barriers in the SI in neonatal piglets, and we sought to determine the key site of impairment in the SI.
Materials & Methods
Animals and Experimental Design
Twelve newborn piglets (Duroc × (Landrace × Yorkshire)) from six sows were obtained from the Jiangsu Kangle Agricultural and Pastoral Co., Ltd. (Jiangsu, China). The sows were of similar birth order (3rd or 4th) and fed with the same gestating diet that met NRC (1998) nutrient requirements. Piglets with birth weights of 1.56 ± 0.07 kg and 0.73 ± 0.08 kg (mean ± SD) were selected as normal birth weight (NBW) and IUGR piglets, respectively (Wang et al. 2005). A total of 12 neonatal piglets (6 IUGR and 6 NBW) were stunned by electric shock and killed by jugular bloodletting within 2-4 hours after birth without suckling. All procedures were approved by the Institutional Animal Care and Use Committee of Nanjing Agricultural University, PR China.
Sample Collection
All 12 piglets were weighed at birth. The small intestine from the pylorus to the ileocecal valve was removed from the abdominal cavity immediately after the animal died and was divided into three segments: the duodenum, jejunum, and ileum. The weight and length of the emptied intestinal segments were measured. A sample of approximately 1 cm in length was collected from each section and fixed in 4% buffered formaldehyde for histological studies using a light microscope. Another two samples from the midpoint of the SI were fixed in 2.5% buffered glutaraldehyde for scanning and transmission electron microscopy studies. The whole intestine of the jejunum and ileum were also collected and stored in a −20C freezer and liquid nitrogen for double-antibody enzyme-linked immunosorbent assay (ELISA) and gene expression analysis, respectively. The thymus, spleen, mesenteric lymph nodes (MLN) (mesentery dissected MLN along the whole SI) and the small intestine were collected and weighed; the ratio of the organ weight versus body weight was also calculated.
Histological Study
Small sections of the SI were used for scanning and transmission electron microscopy studies (Desaki et al. 1984). Samples were fixed in 2.5% buffered glutaraldehyde, washed with PBS and then post-fixed in 1% osmium tetroxide for 2 hr at 4C. The samples were then dehydrated with an increasing gradient of acetone for 2 hr at room temperature (approximately 18C), and then critical-point dried in 3-methylbutyl (isoamyl) acetate. The dried samples were coated with gold-palladium prior to observation using a digital scanning electron microscope (S-3000N, Hitachi High-Technologies Corp.; Tokyo, Japan).
Another piece of SI, fixed and post-fixed as described above, was dehydrated with an increasing gradient of ethanol for 10 min at room temperature and embedded in EPOK 812 (Oukenn; Tokyo, Japan). The embedded block was cut using an ultramicrotome (MT-500) with a diamond knife (producing 70-nm thin sections) and stained with 2% uranyl acetate and Sato’s lead staining solution (containing lead citrate, lead nitrate, and lead acetate). The stained thin sections were then analyzed using a transmission electron microscope (H-7650; Hitachi High-Technologies Corp.; Japan).
Other SI samples were fixed in 4% buffered formaldehyde and then dehydrated using a graded series of ethanol and xylene and embedded in paraffin. Tissue sections (5 microns in size) were then deparaffinized using xylene and rehydrated with graded dilutions of ethanol. Slides were stained with hematoxylin and eosin (HE) and analyzed using a light microscope for morphological observations and lymphocyte counting. Five to ten slides for each tissue were prepared, and images were collected using an optical binocular microscope (Olympus BX5; Olympus Optical Co. Ltd, Tokyo, Japan) coupled with a digital camera (Nikon H550L; Nikon, Tokyo, Japan). Morphological measurements of the villus length (v), crypt depth, and villus width (w) were measured using the Image-Pro Plus (IPP) software, and the villus area (S) was calculated using the following formula:
Polysaccharide periodic acid-Schiff (PAS) staining was conducted for goblet cells (GCs) using a commercial kit (Nanjing JianCheng Bioengineering Institute, Jiangsu, China). Ten fields of view were selected using an Olympus light microscope (400× magnification), and images were collected using a digital camera (Nikon H550L). The GCs were counted among 100 enterocytes in 15 different intestinal villi.
Cytokine and Immunoglobulin Level Analysis by ELISA
Frozen intestines from each sample (200 mg) were homogenized in 1 ml of cold phosphate buffer solution (pH=7.4, 0.01 M). The homogenate was centrifuged at 1,200 × g for 15 min at 4C. The levels of cytokines (IL1β, IL2, IL10, TNF-α, and IFN-γ) and immunoglobulin (sIgA) in the jejunum and ileum were determined by ELISA (pig-specific) according to the manufacturer’s instructions (Nanjing JianCheng Bioengineering Institute, Jiangsu, China). Samples were assayed in triplicate. Protein contents of mucosal tissues were measured using the BCA protein assay (Beyotime Institute of Biotechnology, Jiangsu, China). ELISA results were detected using a microplate ELISA reader at 450 nm and were expressed as nanograms per gram protein (for cytokines) and milligrams per gram protein (for sIgA).
Cytokine Gene Expression Analysis
Total RNA was isolated from samples of jejunum and ileum using TRIzol (Invitrogen; Shanghai, China). RNA was quantified based on the absorption of light at 260 nm (A260) and 280 nm. The RNA content in each sample was calculated using the following formula:
The RNA quality was assessed by agarose gel electrophoresis. From each sample, 1 μg of RNA was used to synthesize cDNA in a 20 μl reaction mixture using the Primer-ScriptTM reagent kit (TakaRa Biotechnology Co. Ltd.; Dalian, China) according to the manufacturer’s instructions.
Gene-specific primers are listed in Table 1 and were synthesized by Invitrogen Biotech Co. Ltd. (Shanghai, China). β-Actin was chosen as a housekeeping gene (cyclophilin was also used as a control gene to normalize the expression of target genes). Reverse transcription-polymerase chain reaction (RT-PCR) assays were conducted using an ABI 7300 RT-PCR system (Applied Biosystems; Foster City, CA) with a SYBR Premix Ex TaqTM Kit (TakaRa Biotechnology Co. Ltd; Dalian, China). The following thermal profile was used for RT-PCR: 95C for 30 sec, followed by 40 cycles of 95C for 5 sec and 60C for 31 sec. The relative gene expression was determined using ABI software (Applied Biosystems) and calculated by the 2-ΔΔCT method (Livak and Schmittgen 2001).
Primer Sequences Used in Quantitative Real Time PCR Assays.

Statistical Analysis
Organ weights and the relative gene expression of cytokines were analyzed using independent t-tests (SPSS 17.0; Chicago, IL). Cytokine and immunoglobulin levels, histological data from the SI, and the numbers of lymphocytes and GCs were analyzed as a mixed model with group (G), intestine (I) and G × I as fixed effects and piglets as a random effect using the MIXED procedure. Independent t -tests were conducted between the IUGR and NBW groups for further analysis. Statistical significance was set at p<0.05.
Results
Development of Immune Organs
In this study, the body weight (BW) of IUGR neonatal piglets was 53.2% less (p<0.01) than that of NBW piglets. Compared with NBW piglets, IUGR neonatal piglets had decreased (p<0.01) absolute weights of the thymus, spleen, MLN, and SI and a shorter (p<0.05) absolute length of the SI (Fig. 1). Furthermore, the relative weights of the thymus (p<0.01), spleen (p<0.05), and MLN (p<0.05) as well as the SI weight per unit of length (p<0.01) were also lower in IUGR piglets than in NBW piglets. However, the relative SI length of IUGR piglets was higher (p<0.01) than that of NBW piglets (Table 2).
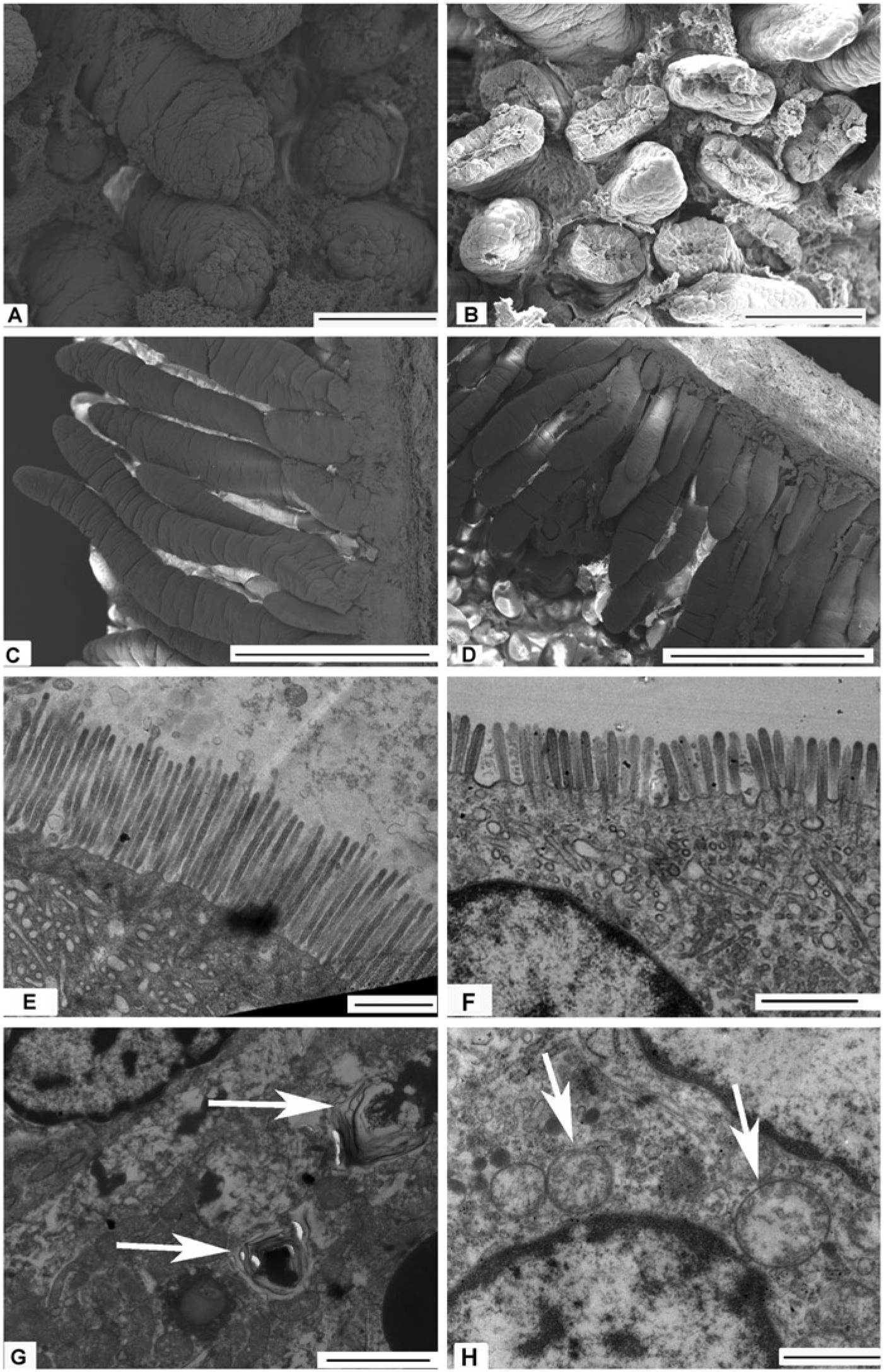
The effects of intrauterine growth restriction (IUGR) on the morphology and interior structure of the midpoint of the small intestine (SI) in neonatal piglets. Observation of villus integrity and uniformity in the SI was determined by scanning electron microscopy (S-3000N; Hitachi High-Technologies Corp; Japan) in piglets (normal birth weight (NBW): A, C; IUGR: B, D). Damaged (B), shorter, and jagged villi (D) in the SI were observed in IUGR neonatal piglets. Microvilli of the SI and its interior structure (NBW: E; IUGR: F, G, H) were observed by transmission electron microscopy (H-7650; Hitachi High-Technologies Corp; Japan). IUGR piglets had shorter and fewer microvilli compared with NBW piglets (E, F). Furthermore, autophagosomes (indicated by arrows in G) and swelled mitochondria (indicated by arrows in H) were also observed in the SI of IUGR neonatal piglets but not in NBW piglets. Scale bars: 100 micrometers in A and B; 500 micrometers in E and F; 1 micrometer in C, D, and H; and 2 micrometers in G.
Effects of Intrauterine Growth Restriction on the Development of the Small Intestine and Immune Organs in Neonatal Piglets a .
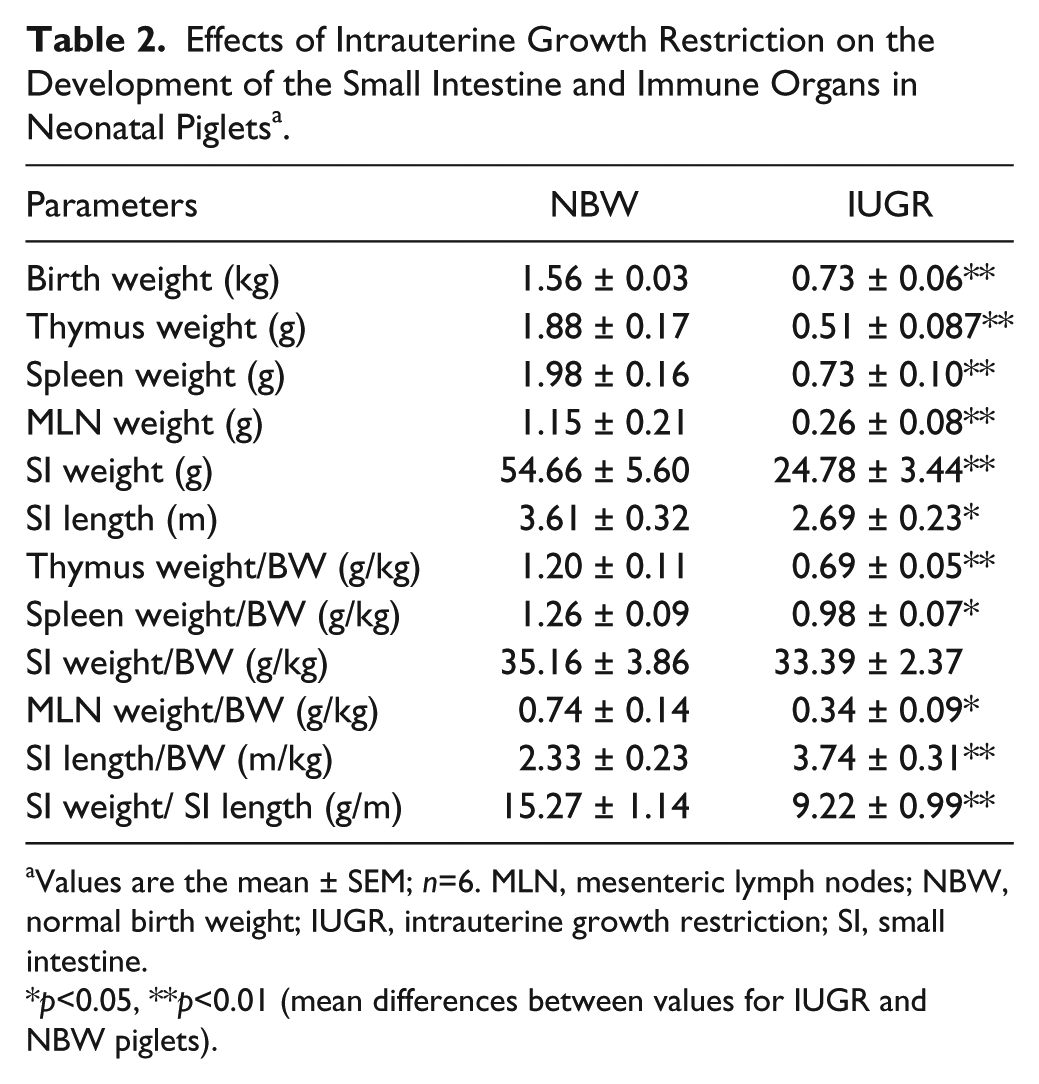
Values are the mean ± SEM; n=6. MLN, mesenteric lymph nodes; NBW, normal birth weight; IUGR, intrauterine growth restriction; SI, small intestine.
p<0.05, **p<0.01 (mean differences between values for IUGR and NBW piglets).
Histological Observations of the Small Intestine
IUGR neonatal piglets showed damaged, shorter, and jagged intestinal villi in comparison with NBW piglets (Fig. 1A-1D). IUGR neonatal piglets also had shorter and fewer microvilli (Fig. 1E & 1F). In addition, autophagosomes and swelled mitochondria were also observed in the SIs of some IUGR piglets, whereas none were found in NBW piglets (Fig. 1G, 1H). The villus length and calculated villi area were also reduced in IUGR neonatal piglets, especially in the jejunum and ileum (Table 3).
Effects of Intrauterine Growth Restriction on the Morphology of the Small Intestine in Neonatal Piglets a .
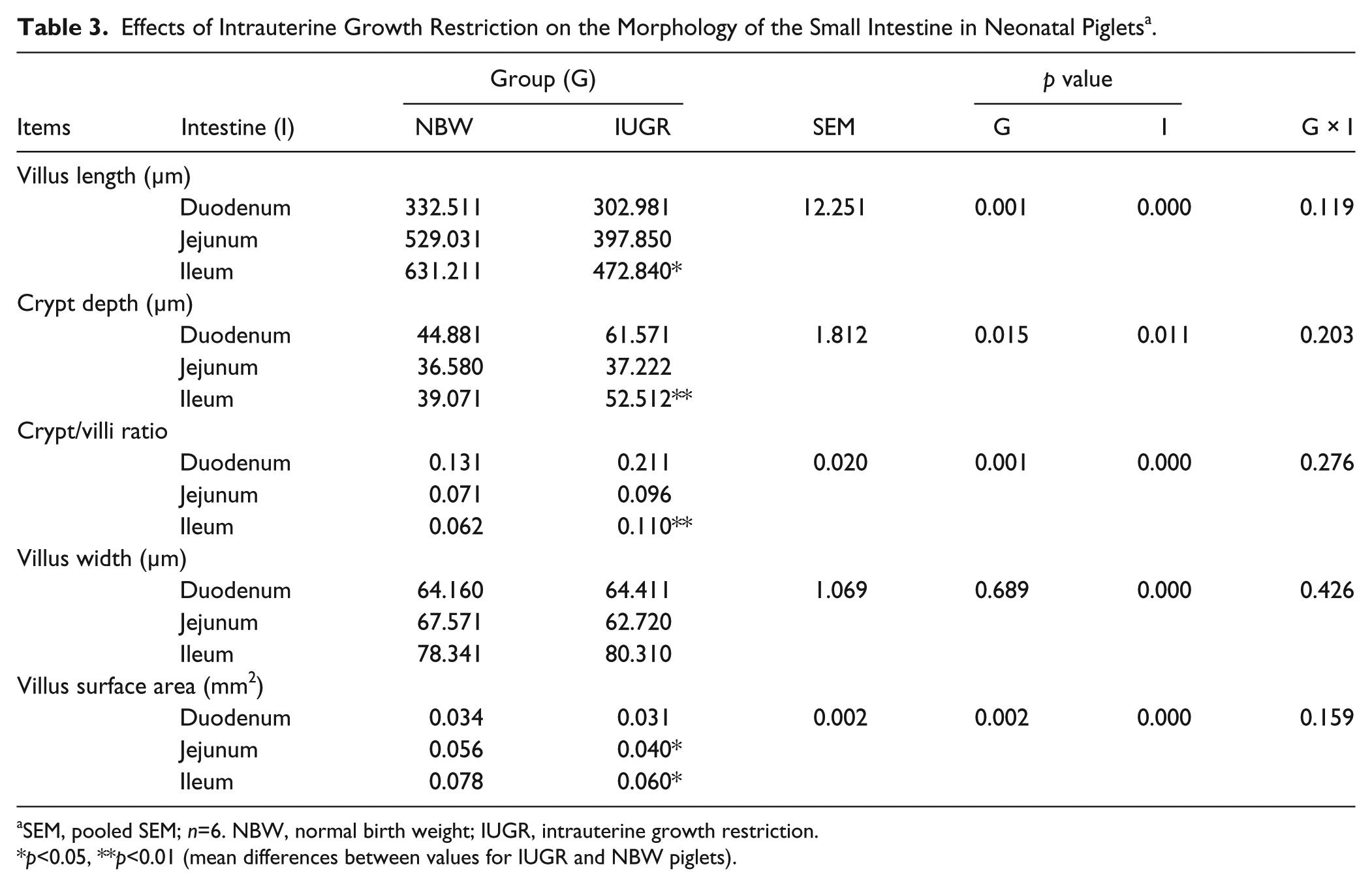
SEM, pooled SEM; n=6. NBW, normal birth weight; IUGR, intrauterine growth restriction.
p<0.05, **p<0.01 (mean differences between values for IUGR and NBW piglets).
We calculated GC and lymphocyte numbers in the epithelium of the SI. The results are shown in Table 4. Compared with NBW neonatal piglets, IUGR piglets had lower (p<0.01) numbers of GCs in the SI, especially in the ileum (p<0.01) and jejunum (p<0.05). Lymphocytes with large and round nuclei were observed in the epithelium of the SI by HE staining (data not shown). The effects of IUGR on the lymphocyte numbers in the SIs were segment dependent. IUGR neonatal piglets were also observed to have fewer (p<0.05) lymphocytes in the ileum than NBW piglets.
Effects of Intrauterine Growth Restriction on the Numbers of Goblet Cells and Intraepithelial Lymphocytes in the Small Intestine of Neonatal Piglets a .
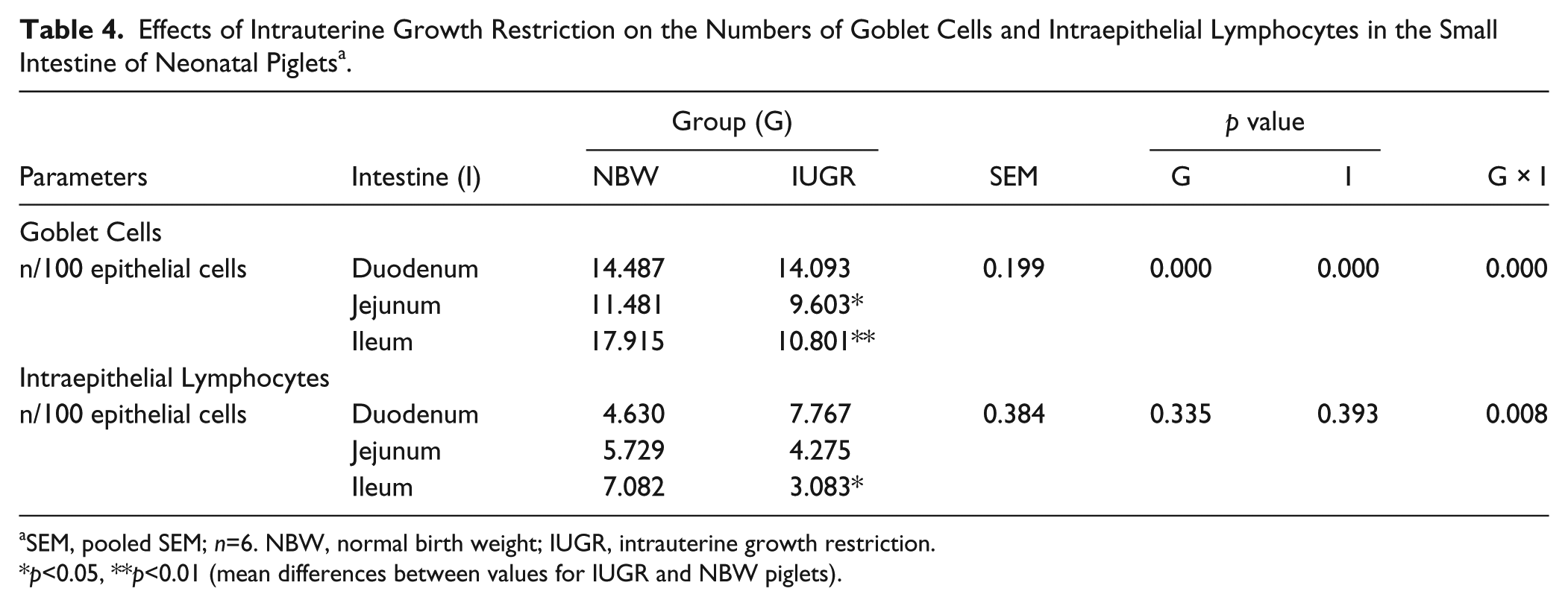
SEM, pooled SEM; n=6. NBW, normal birth weight; IUGR, intrauterine growth restriction.
p<0.05, **p<0.01 (mean differences between values for IUGR and NBW piglets).
Levels of Cytokines and Immunoglobulin
The concentrations of IL1β, IL2, IL10, TNF-α, IFN-γ, and sIgA in the jejunums and ileums of IUGR and NBW neonatal piglets are shown in Table 5. The IL1β, IL2, IL10, and sIgA contents were similar between the two groups and between the two segments of the SI (p>0.05). However, TNF-α (p<0.01) and IFN-γ (p<0.05) contents were lower in the SIs of IUGR neonatal piglets than in NBW piglets. Furthermore, the levels of TNF-α were dissimilar in different segments of the SIs in IUGR neonatal piglets. The concentration of TNF-α in the ileum of IUGR neonatal piglets was significantly lower (p<0.01) relative to that of NBW piglets.
Effects of Intrauterine Growth Restriction on the Levels of Cytokines and Immunoglobulin in the Small Intestines of Neonatal Piglets a .
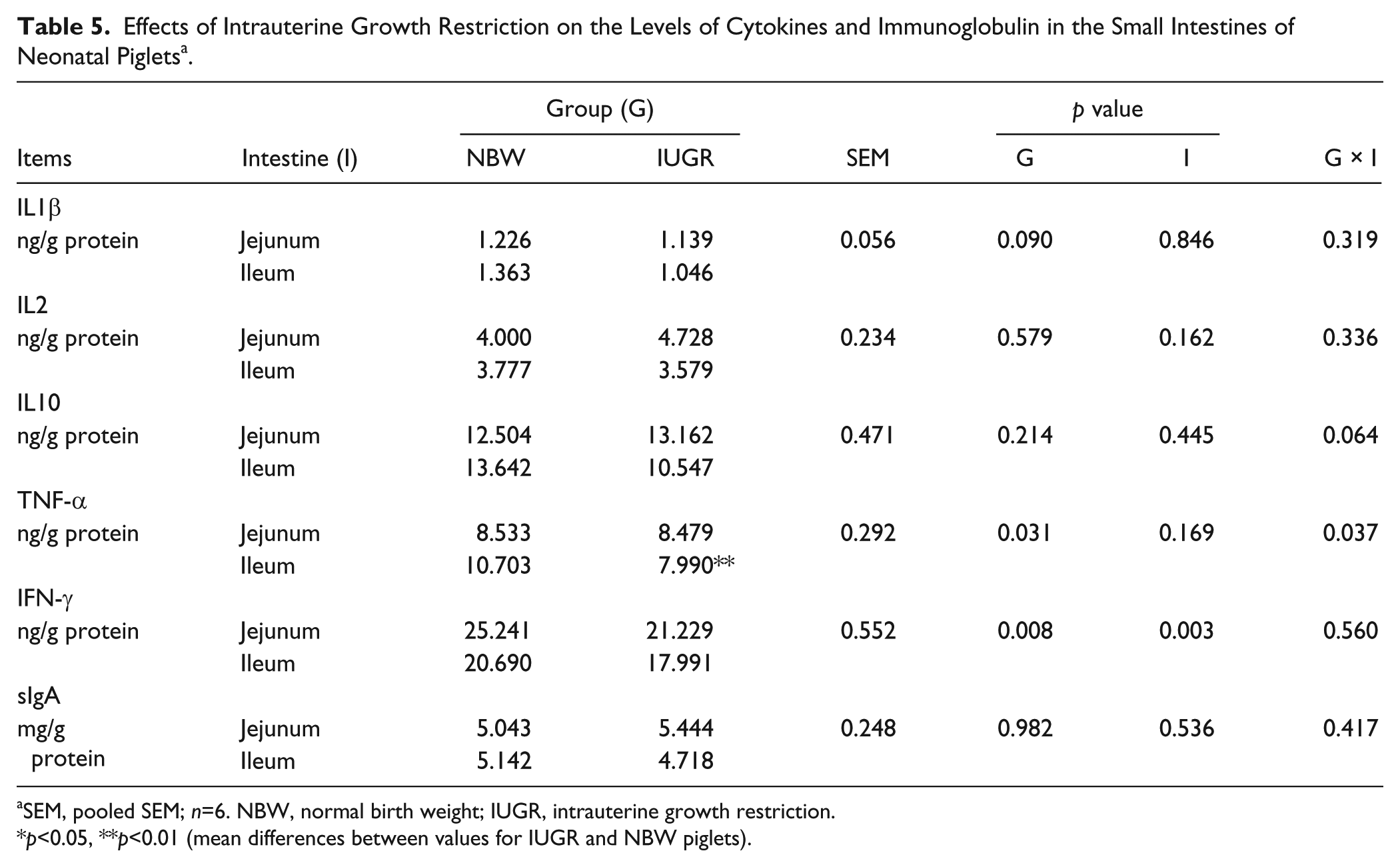
SEM, pooled SEM; n=6. NBW, normal birth weight; IUGR, intrauterine growth restriction.
p<0.05, **p<0.01 (mean differences between values for IUGR and NBW piglets).
Cytokine gene expression
Table 6 shows the relative gene expression of IL1β, IL2, IL10, TNF-α, and IFN-γ in the SIs of neonatal piglets with IUGR or NBW. In the jejunum, the relative mRNA expression of IFN-γ was reduced (p<0.05) in IUGR neonatal piglets in comparison with NBW piglets, whereas the relative mRNA contents of the other measured cytokines were similar (p>0.05) between the two groups. However, the relative mRNA expression of cytokines, including IL1β (p<0.05), IL2 (p<0.01), IL10 (p<0.01), and TNF-α (p<0.01), were all decreased in the ileums of IUGR piglets compared with the NBW piglets. (Similar results were obtained using cyclophilin as a control gene; data not shown.)
Effects of Intrauterine Growth Restriction on the Relative mRNA Expression of Cytokines in the Small Intestines of Neonatal Piglets a .
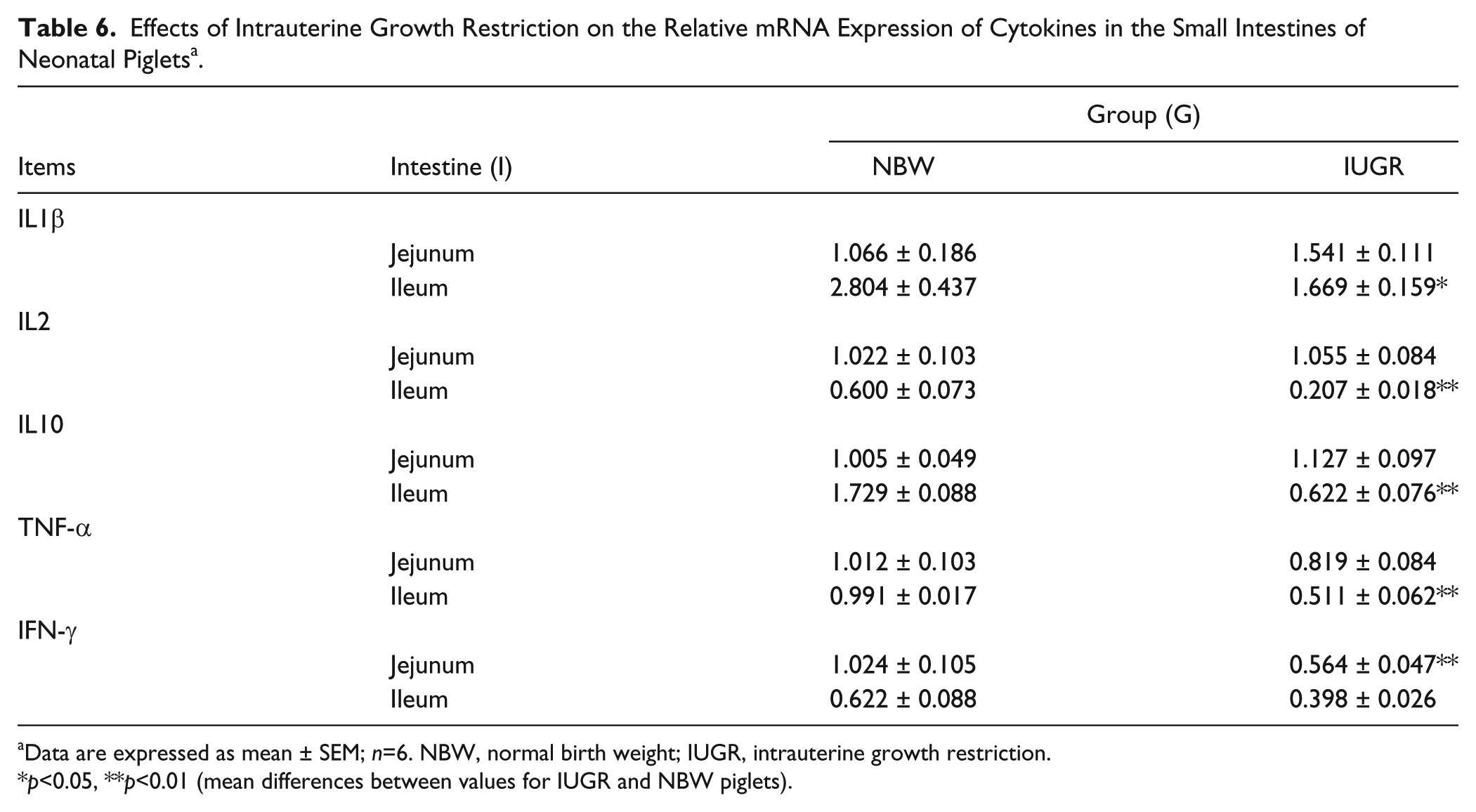
Data are expressed as mean ± SEM; n=6. NBW, normal birth weight; IUGR, intrauterine growth restriction.
p<0.05, **p<0.01 (mean differences between values for IUGR and NBW piglets).
Discussion
IUGR neonates are more susceptible to infection, and the SI is a major site for antigen invasion. Therefore, we hypothesized that IUGR may impair the mucosal immunity of the SI in neonatal piglets. In our study, maldeveloped immune organs and SIs, damaged SI morphology, the presence of autophagosomes and swelled mitochondria, lower levels of cytokines, and the down-regulated gene expression of cytokines in the SI were observed in IUGR neonatal piglets; these observations were indicative of impaired mucosal immunity in the SIs of IUGR neonatal piglets.
In the present study, the maldevelopment of the major immune organs, including the thymus, spleen and MLN, suggested that IUGR neonatal piglets suffer from poor immunity. Previous studies also observed changes in the size and histopathology of the thymus in animal models of IUGR and stillborn infants (Lansdown 1977; D’Inca et al. 2010c). Our findings agree with previous observations that also suggested that IUGR was associated with poorly developed SIs, which were shorter and thinner than those of normal piglets, although the relative length of the SI in IUGR was longer (Wang et al. 2005; D’Inca et al. 2010c; Shanklin and Cooke 1993). Michiels et al. (2013) also reported maldevelopment of the digestive tract of the IUGR piglets in the post-weaning period. This could be a consequence of lower circulating insulin-like growth factor-1 (IGF-1) levels. Though few reports concerning the levels of IGF-1 in the neonatal IUGR piglet could be found, multiple studies have demonstrated that human IUGR infants have lower fetal or cord IGF-1 concentrations as compared with appropriately sized infants (Woods et al.1996; Lassarre et al. 1991). Shorter villus length and smaller villus surface area in the SIs of newborn IUGR piglets were also observed in this study, which could be due to a high rate of apoptosis in the intestinal epithelia (Wang et al. 2005; Shirkey et al. 2006; D’Inca et al. 2010b). In addition, damaged and jagged villi and shorter microvilli of the SI were observed in IUGR newborn piglets, all of which indicate a vulnerable mechanical barrier in the SI of IUGR neonates.
Autophagosomes and swelled mitochondria were also observed in the SIs of IUGR neonates. Autophagosomes are involved in innate immunity in the process of autophagy (Deretic 2006). Nutrient starvation is the cause of autophagy (Mizushima 2007). Coincidentally, nutrient deficiency in the uterus is a major cause of IUGR (Wu et al. 2004), which may partly explain why autophagosomes were found in the SIs of IUGR piglets. Mitochondrial swelling is associated with oxidation and phosphorylation processes within the mitochondria (Mizushima 2007). The occurrence of swelled mitochondria suggested a weakened antioxidant capacity, which may be partly responsible for damage to the SI in IUGR piglets. However, this antioxidant impairment can most likely be reversed after adequate nutrients are supplied postnatally because mitochondrial swelling is reversible (Minamikawa et al. 1999).
Mucosal surfaces of the SIs are lined by epithelial cells, which establish a barrier between hostile external environments and the internal milieu (Turner 2009). Goblet cells (GCs) in the epithelium of the SI can synthesize and secrete glycoproteins, thus protecting the intestine from pathogens (Deplancke and Gaskins 2001). Lower epithelial GC numbers in the SIs of IUGR piglets suggested a possible weaker mucin barrier in the SIs of IUGR neonates. Intraepithelial lymphocytes (IELs) in the SI are the first immune barrier cells to encounter pathogens that have invaded an epithelial surface (Beagley and Husband 1998). Decreased numbers of IELs in the ileum of IUGR neonatal piglets suggests a potentially weaker response to antigen stimulation after birth. The antibody sIgA also acts as an inhibitor of bacterial/viral adherence and penetration of the underlying epithelium (Cunningham-Rundles 2001; Macpherson et al. 2001). However, sIgA levels in the SIs of IUGR piglets were not significantly lower than those in NBW piglets; this finding did not support our hypothesis, and further research is needed.
An immature immune system in neonates contributes to a weak and Th2-polarized immunity; thus, the immune system of a newborn is subjected to an educational process to obtain a Th1/Th2 balance (Morein et al. 2002). Although the ratio of Th1 to Th2 cytokines was similar between the two groups (data not shown), the TNF-α and IFN-γ contents (both of which are Th1 cytokines and pro-inflammatory cytokines) were lower in the SIs of IUGR neonatal piglets than in the NBW piglets. Lower levels of Th1 cytokines are indicative of a partially defective innate immune system (Morein et al. 2002). Lower cytokine expression per gram tissue could be explained by the presence of a lower total number of immune cells per tissue section. Research has reported that IUGR could reduce the total number of CD4+ and CD8+ T lymphocytes in the thymus (Contreras et al. 2011). Similar findings were also observed in our study: Both the gene expression of CD4 and CD8 were reduced in the jejunum and ileum of IUGR neonatal piglets (unpublished data). TNF-α and IFN-γ mRNA transcription varied with their corresponding protein levels, but IL2 and IL10 showed down-regulation only at the gene level. The gene expression of IL1β in the intestine of term IUGR piglets at birth was not changed in either our study or a previous study (D’Inca et al. 2010a). The decreased relative mRNA expression of these cytokines may contribute to complicated cytokine interactions (Braun et al. 2004). Additionally, decreases in IL2 and IFN-γ mRNA expression may represent a regional inhibition of the immune status that is associated with MHC-II expression (Zeitz et al. 1988; Bravery et al. 1995), which is consistent with the lower MHC-II expression detected in the present study (unpublished data). Although the rearing environment affects the development of the immune system in neonates (Inman et al. 2010), the process takes time. Piglets in this study were newborn and euthanized shortly after birth without suckling. Hence, we assumed that the piglets in this experiment attained a normal status without immune stimulation and that the lowered mRNA expression of both pro-inflammatory cytokines and anti-inflammatory cytokines in the SIs of IUGR newborn piglets indicated poor cytokine secretion.
Interestingly, we also found that the villus height and area, number of GCs and IEL, cytokine levels, and gene expression were affected by IUGR among the various intestinal segments. The ileum is the key site of mucosal immunity defects in the SIs of IUGR neonatal piglets. At present, this phenomenon could not be explained; however, the finding provided a direction for our next study (ongoing research).
It can be concluded that the mucosal immunity of the SI in IUGR neonatal piglets was impaired, with maldeveloped MLNs and SIs, a vulnerable mechanical SI barrier, fewer epithelial immune cells (GCs and IELs) and weaker cytokine secretion abilities. The ileum was found to be the main site of impairment.
Footnotes
Acknowledgements
We are sincerely grateful to Professor Bing Hu and Professor Ziyi He of the College of Life Science of Nanjing Agricultural University for their assistance with the scanning and transmission electron microscopy.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by grants from the