Abstract
Passive immunity of the nervous system has traditionally been thought to be predominantly due to the blood-brain barrier. This concept must now be revisited based on the existence of neuron-derived IgG. The conventional concept is that IgG is produced solely by mature B lymphocytes, but it has now been found to be synthesized by murine and human neurons. However, the function of this endogenous IgG is poorly understood. In this study, we confirm IgG production by rat cortical neurons at the protein and mRNA levels, with 69.0 ± 5.8% of cortical neurons IgG-positive. Injury to primary-culture neurons was induced by complement leading to increases in IgG production. Blockage of neuron-derived IgG resulted in more neuronal death and early apoptosis in the presence of complement. In addition, FcγRI was found in microglia and astrocytes. Expression of FcγR I in microglia was increased by exposure to neuron-derived IgG. Release of NO from microglia triggered by complement was attenuated by neuron-derived IgG, and this attenuation could be reversed by IgG neutralization. These data demonstrate that neuron-derived IgG is protective of neurons against injury induced by complement and microglial activation. IgG appears to play an important role in maintaining the stability of the nervous system.
Introduction
The nervous system is an immune-privileged site; however, the overall concept of the nature of this immunity is under reconsideration based on the neuro-immune crosstalk which results from intra-activity and mimicry of the nervous and immune systems (Carson et al. 2006). In particular, molecules traditionally regarded as being expressed exclusively by immune cells have also been found in nervous cells and vice versa (Carson et al. 2006; Kerschensteiner et al. 2009; Mix et al. 2007).
Immunoglobulin G (IgG), which is the most important antibody in mammalian serum, has traditionally been thought to be synthesized only by mature B lymphocytes. IgG has been detected in cerebral spinal fluid (CSF) and brain tissues of a few animal species (Bouloukos et al. 1980; Fabian 1988; Huang et al. 2008; Reiber et al. 1998; Thompson et al. 1983; Upender et al. 1997), and it was assumed that IgG reaches the CSF by passive transfer across the blood brain barrier (BBB) or through uptake by axon terminals of motor neurons projecting outside the BBB with retrograde transportation into the CNS (Fabian 1988; Mohamed et al. 2002).
Recently, several observations have pointed to another source of IgG in the CNS. Huang et al. found IgG can be produced by mouse CNS neurons, and our laboratory also confirmed IgG is produced extensively by neurons and microglia in the human CNS and peripheral nervous system (PNS) (Huang et al. 2008; Niu et al. 2011a). The eye, which is a specialized organ of the nervous system, is also an immune-privileged site, and IgG is synthesized there by intraocular cells (Niu et al. 2011b). Although the function of neuron-derived IgG is poorly understood, evidence suggests that neuron-derived IgG is involved in active immunity and in maintaining the stability of the nervous system.
In this study, we first confirmed the expression of IgG in rat cortex at the protein and mRNA levels. Distribution of FcγRI (IgG receptor I, CD64) was also evaluated. Neuronal injury and microglial activation were then induced with complement, and blockage of neuron-derived IgG was employed to investigate the function of endogenous IgG in the presence of complement.
Materials and Methods
Animals and Tissue Preparation
Twenty male Sprague Dawley (SD) rats (300-350 g), were purchased at the age of 6-7 weeks from the Experimental Animal Center of Peking University Health Science Center (Beijing, China). The use of rats for this study was approved by the Animal Care and Use Committee of Weifang Medical University.
Under anesthesia with 2% Nembutal (45 mg/kg), rats were perfused with heparinized saline, and the brains were removed and snap frozen with liquid nitrogen, and sectioned as 6-μm-thick coronal sections. Alternatively, after perfusion with saline, rats were perfused with 4% paraformaldehyde/PBS sequentially, and brains were removed and fixed in 4% paraformaldehyde and embedded in paraffin.
Immunohistochemistry
Immunohistochemistry (IHC) was performed on consecutive paraffin-embedded tissue sections (4-µm thickness), as described previously (Niu et al. 2009). Primary rabbit anti-rat antibodies to IgG (H+L, 1:100, Abcam, Cambridge, UK), and goat anti-rat CD64 (1:100; Santa Cruz Biotechnology, Santa Cruz, CA) were used to identify IgG and CD64 in the cortex. Primary mouse anti-rat antibodies to neurofilament (NF200, 1:500, Boster, Wuhan, China), glial fibrillary acidic protein (GFAP, 1:500, Sigma-Aldrich, St. Louis, MO), and CD11b (1:500, Millipore, Bilerica, MA) were used to identify neurons, astrocytes and microglia, respectively (Hulse et al. 2008; Niu et al. 2011a). Horseradish peroxidase (HRP)-conjugated anti-mouse/rabbit IgG (PV9000 Immunohistochemistry Kit, Zymed Laboratories, South San Francisco, CA) or HRP-conjugated anti-goat IgG (PV9003 Immunohistochemistry Kit, Zymed Laboratories) was used as the secondary antibody. 3-Amino-9-ethyl-carbazole (AEC, Zymed Laboratories) was used for visualization of the signal. Spleen tissue was used as a positive control. Normal serum of homologous origin with the secondary antibody was substituted for the primary antibody was employed as a negative control.
Cell Culture
To further investigate the synthesis and function of IgG, primary neural cell culture was performed as previously described (Caggiano and Kraig 1999), with minor changes. Media specific for neurons and for microglia were employed to assure purity of these cells. Neuron cultures were prepared from the cortex of 18-day-old fetal SD rats in 10 cases with serum-free media consisting of Neurobasal Medium, 2% B27, 0.5 mM GlutaMAX™-I supplement and Penicillin-Streptomycin (100 U/ml, 100 µg/ml) (all from GIBCO, Carlsbad, CA). Glial cells were prepared from whole brains of 10 one-day-old neonate SD rats, as described previously (Gao et al. 2002). Microglia were cultured with complete Microglia Medium (ScienCell, Carlsbad, CA) containing 5% FBS, 1% microglia supplement and Penicillin-Streptomycin. Astrocytes were cultured with complete medium for astrocytes (ScienCell).
Immunofluorescence
To confirm the identity of IgG- or CD64-positive cells and observe the morphological changes in neurons, double- or single-label immunofluorescence was performed on paraffin-embedded sections of 4-µm thickness or on frozen sections of 6-µm thickness, or on primary neural cells, as described previously (Niu et al. 2009). Primary antibodies to IgG, CD64, NF200, GFAP and CD11b for these experiments were the same as those used for IHC. MAP-2 antibody (Abcam) was used to visualize neuronal dendrites. Fluorescein (FITC)- or rhodamine (TRITC)-conjugated secondary antibodies (1:100, Jackson Immunoresearch Laboratories, West Grove, PA) was applied for double staining. DAPI (1:1000, Sigma-Aldrich) was used to stain the nucleus. Blank controls were employed, including replacement of primary or secondary antibodies or both with PBS. After mounting with glycerol-PBS (9:1), slides were evaluated with a BX51 microscopic (Olympus, Tokyo, Japan).
RT-PCR
Total RNA was isolated from primary neurons using TRIzol reagent (Invitrogen, Carlsbad, CA). RQ1 RNase-free DNase (Promega, Madison, WI) was used to treat RNA samples to exclude contamination by genomic DNA. One μg of total RNA was extracted and reverse transcribed with oligo (dT)18 primer (Fermentas, Burlington, Ontario, Canada) using SuperScript III Reverse Transcriptase (Invitrogen), according to the manufacturer’s protocol.
To identify IgG mRNA, PCR was carried out with LA Taq polymerase (TaKaRa, Dalian, PR China). Thirty-five cycles were performed as follows: denaturing at 95C for 30 sec; annealing at 52–60C (according to the melting temperature of the primer) for 40 sec and polymerization at 72C for 1 min, followed by complete extension at 72C for 10 min. CD19 was employed to evaluate for B lymphocyte contamination and GAPDH was amplified as an endogenous reference. The conditions for PCR amplification reactions for each transcript are listed in Table S1, including the sequence of specific primers, denaturation temperatures, and the length of each product. Spleen was employed as a positive control. Water was substituted for template for a negative control. Identification of the PCR product was confirmed by DNA sequencing and blasting.
cRNA Probe Preparation and In Situ Hybridization
The cRNA antisense probe against the constant regions of rat IgG1 was prepared as described previously (Niu et al. 2011a). The tissue sections used for in situ hybridization (ISH) were consecutive with sections used for IHC. ISH was performed as previously described (Niu et al. 2011a). Application of IgG1 cRNA sense probe instead of IgG1 cRNA antisense probe was used as a negative control. Normal rat spleen tissue was used as a positive control.
Complement-dependent Cytotoxicity
To investigate the effect of complement on primary neurons, the complement-dependent cytotoxicity (CDC) assay was performed according to the standard protocol, as described previously (Griffioen et al. 2009; Zhao et al. 2010). Briefly, 1×105 cultured mixed neural cells per well were plated in 96-well plates and cultured in the serum-free medium described above. Freshly prepared rabbit baby complement (1:10000, 1:1000, 1:100, 1:10, 1:1; Cedarlane Labs, Burlington, NC) was added to the medium and incubated for 2 hr on the sixth day when neurons showed sufficient extension. After exposure to complement, neurons were further cultured with fresh medium for 48 hr and neuron survival was evaluated using the MTS cell proliferation assay (CellTiter 96 AQ One Solution Cell Proliferation Assay Kit, Promega), as previously described (Maloney et al. 2003). Results were analysed using an OD 490 nm. As a negative control, 0.01 M PBS was used instead of complement.
Western Blot
To observe the effect of complement on the expression levels of neuron-derived IgG, western blotting was performed as previously described (Huang et al. 2008). Total protein (40 µg) was extracted from primary neurons treated with complement at different concentrations as described above. IgG protein (0.02 µg) from normal rat serum was used as a positive control. Rabbit anti-rat IgG antibody (Bethyl, Montgomery, TX) and HRP-labeled goat anti-rabbit (Jackson Immunoresearch Laboratories) were used as primary and secondary antibodies.
Cell Survival and Analysis of Apoptosis with IgG Neutralization
To determine whether neuron-derived IgG is protective against CDC (induced by complement), IgG was blocked, and MTS and apoptotic assays were performed. Briefly, mixed neural cells were plated in 6-well plates (2×106/well, for apoptosis analysis) and 96-well plates (1×105/well, for MTS analysis) and cultured in serum-free medium. Freshly prepared rabbit baby complement (1:100) was added and incubated for 2 hr on day 6. Goat anti rat IgG antibody (R&D Systems, Minneapolis, MN) was administered for 48 hr at doses of 0.2, 2.0, 20, 200 and 2000 µg/ml before exposure to complement. Normal goat IgG at these same concentrations was used as a homologous control for the IgG antibody. Neuron survival was then evaluated with the MTS cell proliferation assay (Promega) as previously described (Maloney et al. 2003).
For analysis of apoptosis, primary neurons were analyzed with the Annexin V-PI kit (Peking University Human Disease Genomics Research Center, Beij,ing, China) as described previously (Qiu et al. 2003). According to the standard method for analysis suggested by the Annexin V-PI kit manufacturer, cells (Annexin V +, PI-) distributed in the right lower quadrant of the flow cytometry plot are in early apoptosis, while those (Annexin V +, PI+) in the right upper quadrant are in late apoptosis or are dead. The gates for assessing cells and apoptosis ratios were set and analyzed against blank and isotype controls.
Caspase-3 staining was performed on primary cortical neurons to further confirm induction of apoptosis by CDC. Rabbit anti-rat active caspase-3 antibody (Cell Signaling, Danvers, MA) and FITC-labeled goat anti-rabbit antibody (Jackson Immunoresearch Laboratories) were applied as primary and secondary antibodies. DAPI was used to visualize the nucleus.
Extraction and Purification of Neuron-derived IgG
To further study its function and mechanism, neuron-derived IgG was purified from primary cortical neurons with the Montage Antibody Purification Kit (Millipore) according to the manufacturer’s protocol and assessed with western blotting, as described above.
Microglial Activation and Nitric Oxide Assessment
To investigate the effect of neuron-derived IgG on microglia induced by complement (1:100, 2 hr exposure time), microglia were evaluated with CD11b (a marker for microglia) by immunofluorescence, as described above. Using a Total Nitric Oxide Assay Kit (Beyotime Institute of Biotechnology, Shanghai, China), total nitric oxide (NO) production in microglial culture medium was determined by measuring the concentration of nitrate and nitrite, which are stable metabolites of NO, by modified a Griess reaction method, according to the manufacturer’s protocol.
Real Time RT-PCR
To confirm whether neuron-derived IgG affects the expression of CD64 in microglia, purified neuron-derived IgG or IgG antibody was added into the primary microglia medium, which was then exposed to complement (1:100) for 2 hr. Total microglial RNA was then extracted and RT was performed, as described above. Real time PCR was carried out in triplicate with primers described previously (Li et al. 2008) and SYBR Premix Ex Taq (TaKaRa Bio Inc., Shiga, Japan) using the ABI PRISM 7500 Real-Time PCR system (Applied Biosystems, Foster City, CA), according to the standard protocol.
Statistical Analysis
Representative sections of cortex from 20 SD rats were selected for statistical analysis after carrying out IHC. Five random fields of consecutive sections stained for NF200 and IgG were photographed with a light microscope at ×400 magnification (BX51; Olympus). Total numbers of NF- and IgG-positive neurons were counted and IgG-positive ratios (number of IgG-positive cells/number of NF-positive cells) were calculated. Differences in IgG-positive ratios in SD rats, and differences in apoptosis in test and control groups with primary-cultured neurons were evaluated using the χ2 test with SPSS 12.0 software (SPSS Inc., Chicago, IL). Two-sided p values with p<0.5 were considered statistically significant.
Results
IgG Protein and CD64 in Rat Cortex
Cell type was established with neural markers, including NF200 for neurons, GFAP for astrocytes and CD11b for microglia. In the positive control, IgG immunoreactivity was visualized in plasma cells or plasmablasts in SD rat spleen tissues, demonstrating the antibody to IgG used in this experiment was specific (Fig. S1A). In the cerebral cortex, IgG immunoreactivity was found in most neurons (marked with NF), with an total average positive ratio of 69.0 ± 5.8%, but was more prevalent in pyramidal cells, whose cell bodies are identifiable by their prominent triangular tapered shape, with an average positive ratio of 81.1 ± 6.3% (Fig. 1A). For tissue sections and primary neural cultures, IgG immunoreactivity co-localized with NF-positive zones, and these positive signals were distributed in the cytoplasm of the neuron body and in the cytoplasm of dendrites and axons near the cell body (Fig. 1A and 1B).
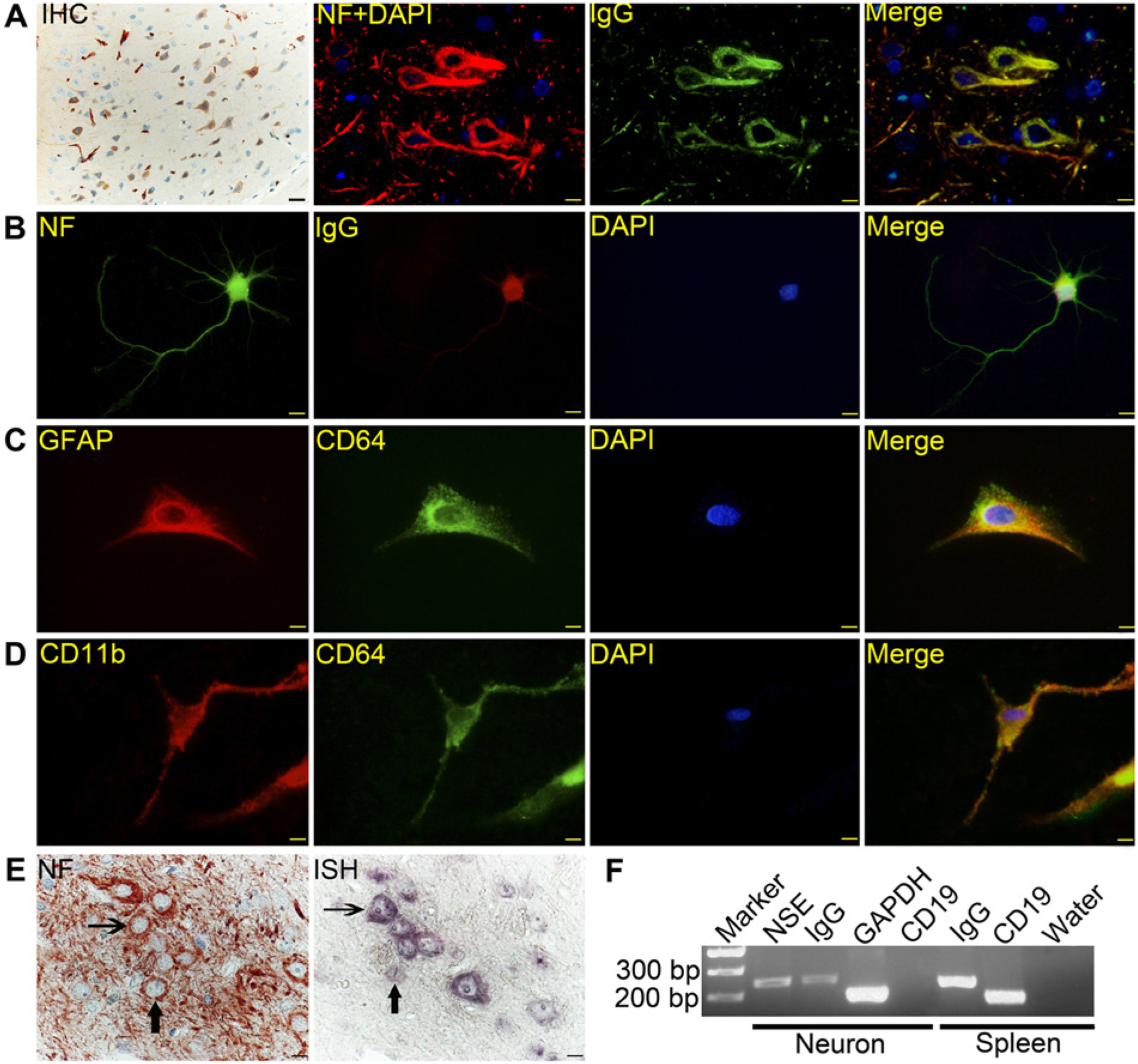
Expression and distribution of IgG and CD64 in rat cortex. (A) Most cortical neurons are positive for IgG (left panel) by immunohistochemistry, as visualized by AEC (red). Neurofilament (NF) (red, labeled with TRITC) and co-localizing IgG (green, labeled with FITC) in pyramidal cells. (B) IgG (red), distributed in the cytoplasm and cell processes of primary-cultured neurons (green). (C, D) CD64 (green) is detected in the membrane and cytoplasm of astrocytes (red, identified by GFAP) and microglia (red, identified with CD11b). (E) Consecutive sections of rat cortex showing co-expression of NF (left panel) and IgG mRNA (right panel) in the cytoplasm of several neurons using NF antibody staining and in situ hybridization. Arrows of the same shape point to the same single neuron in consecutive sections. (F) Agarose gel electrophoresis of RT-PCR amplification products following mRNA extraction from primary cultured neurons. Bands of IgG (282 bp) and NSE (255 bp) were detected in neurons, but no CD19 was found. GAPDH: endogenous reference. Spleen: positive control. Water (instead of RNA): negative control. Bars: 20 µm.
CD64 is the receptor with the highest affinity for IgG. It was found on the membrane and in the cytoplasm of astrocytes and microglia (Fig. 1C and 1D), which suggested that neuron-derived IgG may engage in cross-talk with glial cells by binding with CD64.
Expression of IgG mRNA in Rat Cortical Neurons
To confirm IgG mRNA expression in neurons, ISH was performed on sections consecutive with those used for IHC. Special probes for the constant region of rat IgG heavy chain were used. In splenic tissue, significant positive signals were found in plasma cells (Fig. S1B) and no signal was seen when the sense probe was used as the control (Fig. S1C). In the cortex, positive IgG mRNA signals were found in the cytoplasm of neuronal bodies. As shown in Fig. 1E, NF protein (left panel), determined by IHC, and positive IgG mRNA signals (right panel), determined by ISH, co-localized in the cytoplasm of a single neuron, which was large enough to be present in two consecutive sections. No signal was found in microglia or astrocytes.
IgG transcripts were further amplified by RT-PCR from total RNA extracted from primary–cultured neurons. Splenic tissue was used as a positive control. The products were analyzed with agarose gel electrophoresis (Fig. 1F) and confirmed by sequencing. IgG heavy chain and NSE were found in primary neurons, and the intensity of positive bands was weaker than those in splenice tissues. There was no identifiable amplification of CD19 transcript, excluding the possibility of lymphocyte contamination in the samples under investigation. Sequencing showed that the amplified products of IgG heavy chain were 100% identical with HQ640951.1.
CDC Effects on Neurons, and Complement-induced Increase in IgG Levels
It is known that there are several complement receptors on the neuron surface (Woodruff et al. 2010) and fresh complement has been used to induce CDC (Lee et al. 2012). In the present study, freshly-prepared rabbit complement was applied to evoke CDC, and significant injury was observed. Compared with the control (Fig. 2A, left panel), the morphology of primary–cultured neurons was altered in CDC, with irregular cell outlines and a decreased numbers of dendrites (Fig. 2A, middle panel). Neutralization of the neuron-derived IgG with IgG antibody was able to decrease these complement-induced morphological changes in neurons (Fig. 2A, right panel). When the neuronal dendrites were visualized with MAP-2 staining (Fig. 2B), the morphological neuronal changes in the test and control groups were clearer. Compared with the blank control (Fig. 2B, left panel), neuronal dendrites exposed to complement (1:100) were fewer and thinner (Fig. 2B, middle panel), and IgG neutralization affected the loss of neuronal dendrites (Fig. 2B, right panel). We further investigated the effect of step-wise concentrations of complement on IgG expression levels. It is worth noting that, after 2 hr exposure to complement, followed by incubation for 48 hr with fresh serum-free medium, an increase in IgG expression was observed (Fig. 2 C). IgG expression increased at concentrations of 1:10,000, 1:1000, and 1:100, but decreased at 1:10 and 1:1. This may have resulted from excessive death of neurons induced by high concentrations of complement. For the following experiments, we therefore chose 1:100 as the most effective complement concentration. Moreover, as compared with the control, IgG expression by neurons triggered by complement (1:100) was increased by 5.8 ± 1.1-fold (Fig. 2D, p=0.021).
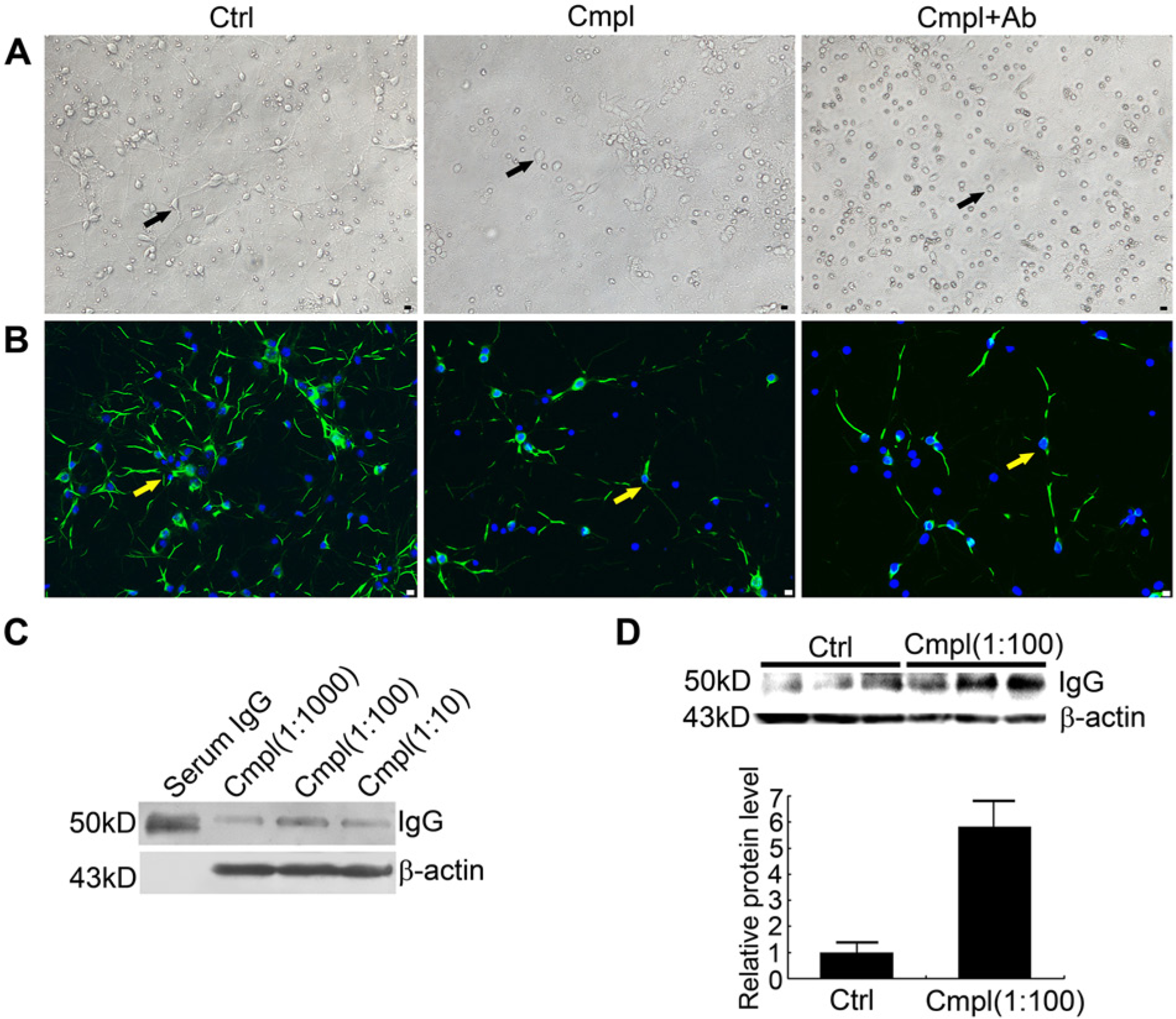
Morphological neuronal changes and changes in IgG expression level induced by complement-dependent cytotoxicity (CDC). (A-B) Morphological changes in primary neurons (indicated with arrows) caused by exposure to PBS (left panel, Ctrl (control)), complement (1:100) (middle panel, Cmpl), or complement (1:100) and IgG antibody (20 µg/ml) (right panel, Cmpl+Ab). (A) Visualization by phase contrast microscopy. (B) Photographs of primary neurons stained using MAP-2 (green). (C) Expression levels of IgG protein in primary neurons were affected by the concentration of complement, as detected using western blotting. (D) Levels of IgG protein in test neurons exposed to complement (1:100) and the control, as detected by western blotting (upper panel) with relative expression analysis (lower panel). Bars: 20 µm.
Exposure to step-wise increases in complement resulted in decreasing numbers of viable cells in the survival assay (Fig. 3A), demonstrating that more neurons died as the dose of complement increased. A significant point of decrease in this curve was found at a concentration of 100 µl per 1 ml medium (1:10).
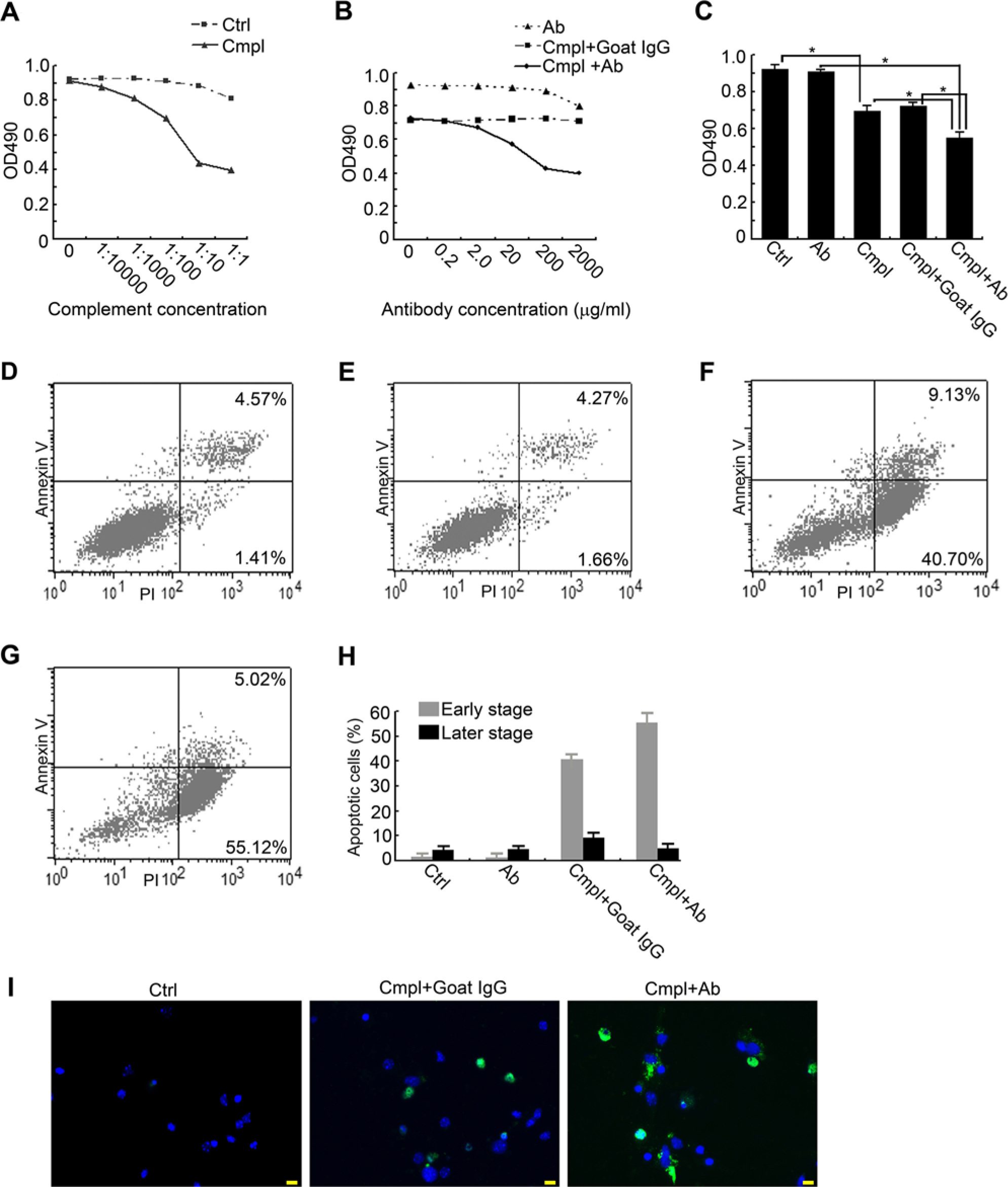
Effect of IgG neutralization on complement-dependent cytotoxicity (CDC) of cortex neurons induced by complement. (A) MTS analysis of primary neuron exposure to gradient concentrations of complement. (B) In the presence of complement (1:100), MTS analysis showed that IgG neutralization with gradient doses of goat anti-rat IgG antibody caused a reduction in OD 490 values. (C) OD 490 value comparison of different treatments. Significant differences are indicated with an asterisk. (D-G) Apoptosis analysis of primary neuron exposure to (D) antibody (20 µg/ml), (E) PBS (control), (F) complement (1:100) and goat IgG (employed as the homologous control for IgG antibody), and (G) complement (1:100) and anti-IgG antibody (20 µg/ml). Right lower quadrant: early apoptosis; right upper quadrant: late apoptosis or cell death. (H) Comparison of apoptotic ratio of different stages resulting from different treatments. (I) Caspase-3 staining (green) of neurons in the subgroups of the blank control (left panel, 2.1 ± 1.8%), complement and goat IgG (middle panel, 35.8 ± 8.3%), complement and IgG antibody (56.2 ± 9.1%). Cmpl: complement. Bars: 20µm.
Neutralization of IgG Aggravates CDC in Neurons Exposed to Complement
Antibody neutralization has been used extensively in functional studies of protein (Qiu et al. 2003), and IgG antibody has been employed to neutralize secreted IgG (Lee et al. 2012). To investigate the function of neuron-derived IgG, gradient concentrations of goat anti-rat IgG antibody were applied before and during exposure of primary neurons to complement at a concentration of 1:100.
The survival assay showed that IgG antibody aggravated injury induced by complement at a concentration of 1:100. Goat anti-rat IgG antibody alone was not toxic to primary neurons at a concentration of 0.2 to 200 µg/ml but showed minor toxicity at 2000 µg/ml (Fig. 3B). This may have resulted from the high osmotic pressure secondary to the high density of molecules in the medium. Normal goat IgG, used as a homologous control for IgG antibody, had no effect on neurons and showed a curve similar to that of PBS (Fig. 3B), indicating that it could be employed as another negative control for IgG antibody in the following functional experiments in addition to PBS. Compared with goat IgG, anti-rat IgG antibody led to a decrease in the OD 490 nm readings at concentrations of 0.2 and 2.0 µg/ml, and led to a significant decrease at 20 and 200 µg/ml (Fig. 3B). Compared with complement alone, neutralization of IgG resulted in a significantly lower neuron survival rate (Fig. 3C). In subsequent experiments, 20 µg/ml was therefore selected as the most suitable concentration for IgG antibody.
Apoptotic analyses showed that IgG antibody alone did not significantly increase the apoptotic rate (2.98 ± 3.12 %, Fig. 3D) as compared with the blank control (3.26 ± 2.79%, Fig. 3E; p>0.05). In the presence of complement, compared with treatment with normal goat IgG (37.13 ± 5.62 %, Fig. 3F), neutralization with IgG antibody resulted in a higher rate of neuronal apoptosis (52.46 ± 8.02 %, Fig. 3G, p=0.036). Notably, IgG neutralization led to significant increases in apoptosis in the early stages of the experiments (54.85 ± 4.37 vs 39.24 ± 3.65%, Fig. 3H, p=0.029), and no significant decreases in later stages (8.73 ± 2.16 vs 5.02 ± 2.91%, Fig. 3 H). This showed that neuron-derived IgG may protect neurons from complement-induced apoptosis in the early stages. Moreover, the role of neuron-derived IgG in neuronal apoptosis induced by complement was confirmed by activated caspase-3 staining. When cells undergo apoptosis, activated caspase-3 signals are found in the cytoplasm or nucleus. As shown in Fig. 3I, compared with the blank control (2.1 ± 1.8%) and the negative and homologous controls (complement and goat IgG, 35.8 ± 8.3%) for IgG antibody, blocking of neuron-derived IgG with IgG antibody resulted in a significant increase in the percentage of cells (56.2 ± 9.1%, p=0.021) positive for activated caspase-3, while the percentage of cells in the negative control was 35.8 ± 8.3%.
Neuron-derived IgG Attenuates the Microglial Activation Induced by Complement
Inflammation caused by microglial activation is involved in many pathological conditions, such as Parkinson’s disease (Panaro and Cianciulli 2012). Considering the proposed protective function of neuron-derived IgG, we further investigated its effect on microglia with purified IgG derived from neurons by observing the expression of CD11b, measuring the release of neuron-toxic NO, and the evaluating levels of CD64.
The purity of extracted neuron-derived IgG was confirmed by western blotting and the result showed a band of 150 kD identical to standard IgG from normal rat serum (Fig. 4A). Compared with the normal microglia (Ctrl, Fig. 4B), exposure to complement triggered stronger CD11b signals (a parameter of microglial activation) and caused alterations to microglial morphology. Activated microglia became larger in size, with more abundant cytoplasm as compared with resting microglia, which were smaller and thinner (Cmpl, Fig. 4B). Administration of neuron-derived IgG into microglial medium before stimulation by complement attenuated this microglial activation, which was manifested by reduced CD11b signal intensity, and more slender microglial morphology (Cmpl+IgG, Fig. 4B). These effects could be diminished with IgG antibody (Cmpl+IgG+Ab, Fig. 4B).
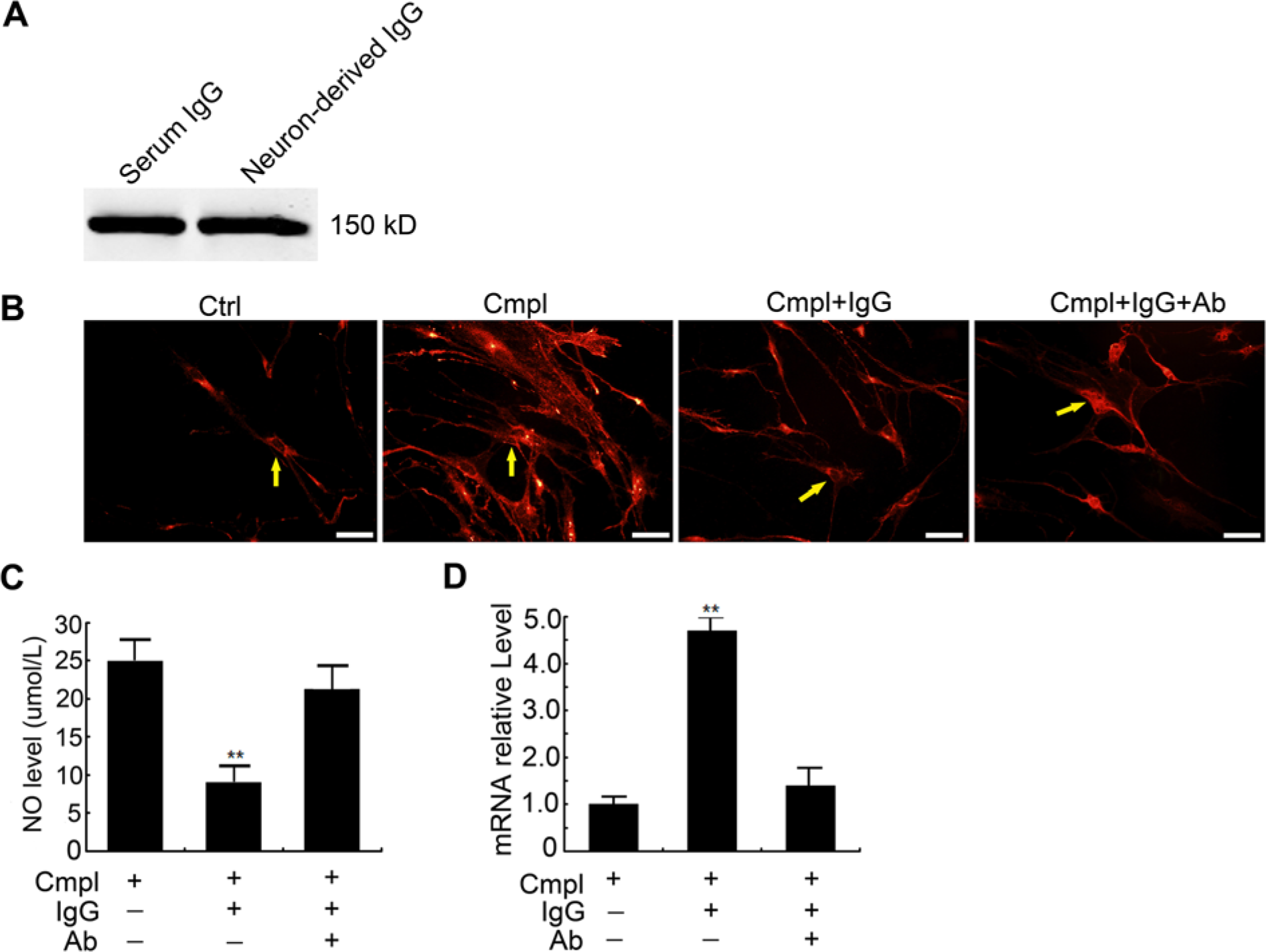
Effect of neuron-derived IgG on microglia. (A) Confirmation of neuron-derived IgG purified from primary-cultured neurons by western blotting. (B) Activation of primary-cultured microglia marked with CD11b (red) triggered by PBS (the control), complement (1:100), complement (1:100) and neuron-derived IgG (20 µg/ml), complement (1:100) and neuron-derived IgG (20 µg/ml) and IgG antibody (20 µg/ml). Examples of typical microglial cells are indicated with yellow arrows. Bars, 20 µm. (C) Nitric oxide (NO) levels in the medium of microglia triggered by complement (1:100, for 2 hr) after treatment with neuron-derived IgG (20 µg/ml, for 24 hr) or neuron-derived IgG and IgG antibody (20 µg/ml, for 24 hr). (D) Relative level of CD64 expressed by microglia treated with complement and neuron-derived IgG (20 µg/ml) with or without IgG neutralization.
NO is produced by activated microglia and is a factor that causes injury in neurons. NO has roles, therefore, in several pathological processes, including Parkinson’s disease (Wang et al. 2007). To study the effect of neuron-derived IgG on microglia, we evaluated total NO levels in microglial media where complement was applied as a stimulus. Compared with the control (25 ± 3.1 µmol/L) in which only complement was exposed, production of NO was reduced to by 64 ± 6.3% to 9.1 ± 2.5 µmol/L (p=0.016) with pre-exposure to neuron-derived IgG (20 µg/ml, Fig.4C), whereas there was only a 15% decrease when IgG and IgG antibody were added together (21.2 ± 3.4 µmol/L). This suggested that neuron-derived IgG can protect neurons from the injury caused by excessive NO released from complement-induced microglia.
Microglial CD64 has the highest affinity for monomeric IgG and mediates phagocytosis and production of several inflammatory molecules, such as IL-10 (Gerber and Mosser 2001). Compared with the control, the relative level of CD64 in microglia pretreated with neuron-derived IgG was elevated significantly by 4.8 ± 0.4-fold (p=0.018), whereas the CD64 level was elevated only 1.4 ± 0.4-fold (p>0.05) by IgG neutralization with IgG antibody (Fig. 4D). These data suggest that neuron-derived IgG has the capacity to affect the function of microglia.
Discussion
Here we demonstrated that IgG is endogenously produced by cortical neurons in the rat, and in our previous paper we demonstrated the presence of neuron-derived IgG in the human nervous system. In tissue sections of rat cortex, IgG protein and mRNA were detected in most neurons (69%) via IHC and ISH. To rule out the possibility of contamination by serum IgG, primary neurons were cultured with neuron-specific serum-free medium. IgG protein and mRNA were further demonstrated by double immunofluorescence staining, together with RT-PCR with DNA sequencing. These data clearly show that cortical rat neurons can produce IgG. Under physiologic conditions, there is 16 ± 2 µg/ml IgG in the CSF (Hulse et al. 2008) and this may be derived from neurons.
The complement system is a vital component of the innate immune system, which protects the host from infection and injury (Horstman et al. 2011). Complement receptors are expressed in all cell types within the CNS, and activation of complement is thought to play a significant role in the progression of CNS diseases, such as infection, ischemia, Alzheimer’s disease and multiple sclerosis (Horstman et al. 2011; Woodruff et al. 2010). In particular, C5a may induce neuronal apoptotic cell death through inhibition of mitochondrial respiration, dehydrogenase and cytochrome c oxidase activity (Woodruff et al. 2010). In the present study, exposure to complement provoked apoptotic death of primary cultured neurons and resulted in a significant increase in IgG levels, indicating that IgG expression may occur in reaction to complement stimulation. Neutralization of IgG with IgG antibody led to increased apoptosis and cell death at an early stage of neuron culture, suggesting that neuron-derived IgG has the ability to protect neurons from early apoptosis and cell death induced by complement. It is known that the exogenous IgG can bind to and scavenge complement, and relieve complement-mediated injury (Basta et al. 2003). In a stroke model, intravenously administered IgG inhibited neuron loss caused by experimental ischemia and reperfusion injury through neutralization of complement components (Arumugam et al. 2007). There is a strong likelihood that neuron-derived IgG protects neurons from inflammatory injury by neutralizing complement.
Microglial cells have traditionally been thought to be the only cell type exerting certain immune functions vital to immune regulation of the CNS. Reactive oxygen species, including NO, and inflammatory cytokines, such as IL-6 and TNF-α produced by excessive or uncontrolled activation of microglia in a burst, are toxic to neurons and contribute to many neuropathies, such as Parkinson’s disease and Alzheimer’s disease (Floden et al. 2005; Gao et al. 2002; Hulse et al. 2008). In the present study, neuron-derived IgG was found to attenuate microglia activation and NO release induced by complement.
CD64 (FcγRI), the high-affinity subtype of FcγR (including CD64, CD32, CD16 and FcRn), was found to be expressed in microglia and in astrocytes of rat cortex in the present study. Upon binding with IgG, activation of CD64 results in a series of biological functions in neural cells. In the neurons of the dorsal root ganglia (DRG), this results in increased concentration of Ca2+ and release of substance P, and elicits scratching of the skin in mice (Andoh and Kuraishi 2004). In motor neurons of the spinal cord, this leads to increased intracellular concentration of Ca2+, acetylcholine release and IgG uptake (Mohamed et al. 2002). Our present study verified neuron-derived IgG can elevate the expression of CD64 in microglia. Together, these data demonstrate that neuron-derived IgG may function with surrounding neural cells, and especially with microglia, via binding with CD64 and is involved in the immune regulation of CNS under physiological and pathological conditions.
To our knowledge, this is the first study demonstrating a neuron-protective function for neuron-derived IgG. At physiological levels, exogenous monomeric IgG is protective of neurons via enhancement of endocytotic recycling, and release of microglial TNF-α following binding to FcγRs (Hulse et al. 2008). IgG also takes part in neural development by inducing the differentiation of oligodendrocyte precursor cells (OPC) into myelinating oligodendrocytes through the activation of Fyn tyrosine kinase by binding with FcγRs (expressed on OPCs) (Nakahara et al. 2003). Under pathological conditions, invading pathogens may be opsonized by locally produced IgG and then phagocytosed by microglial cells upon binding of IgG to FcγRs expressed on their surface (Aloisi 2001). Neuron-derived endogenous IgG may be involved in these pathways.
Multiple studies have shown that neuron-derived IgG is likely to be a vital component of the active immunity in the nervous system, and likely plays an important role in maintaining the stability of the microenvironment of the neuron. This area of inquiry calls for further study.
Footnotes
Author Contributions
Study concept and design: NN. Acquisition of data: JZ, NN. Statistical analysis and interpretation of data:
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (No. 81100885) and Young and Middle-Aged Scientists Research Awards Fundation of Shandong Province (No. BS2011SW051) to Na Niu.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
