Abstract
Lymph vessels play an important role in tumor progression. Pulmonary adenocarcinomas, accounting for half of non-small-cell lung carcinomas, compose a spectrum of histological types, exclusively or without a lepidic growth pattern (LGP) along preserved interalveolar septa. In that context, this study was designed to investigate the lymphatic vascular pattern associated with LGP and the concomitant invasive component of pulmonary adenocarcinomas. Using the D2-40 monoclonal antibody as a marker of lymphatic endothelial cells, the lymphatic vessel density (LVD) and vessel-area fraction (LVAF) were morphometrically analyzed in four adenocarcinomas in situ (AIS) and the LGP of eight invasive adenocarcinomas (LPIA), and compared with their invasive pattern (IPIA). LVD in AIS (2.1 ± 0.7 mm−2) and LPIA (2.4 ± 1 mm−2) were significantly lower than that in IPIA (14.9 ± 13.6 mm−2) (p=0.001). Moreover, the lymphatic vascular pattern in LGP was similar to that of normal lung, with isolated small lymphatic vessels within the interalveolar septa. Our results showing the scarcity of lymphatics in LGP suggest an absence of septal lymphangiogenesis associated with the LGP pattern in lung adenocarcinomas, which could explain, at least partially, the better prognosis observed in tumors with exclusive or predominant lepidic spread compared with other subtypes.
Non-small-cell lung carcinoma (NSCLC) accounts for more than 80% of all lung cancers and is a leading cause of cancer deaths (International Agency for Research on Cancer, 2008). Adenocarcinomas, representing approximately half of NSCLC, compose a spectrum of various histopathological types, ranging from the non-invasive, so-called adenocarcinoma in situ, to solid invasive adenocarcinoma. According to the new International Association for the Study of Lung Cancer/American Thoracic Society and European Respiratory Society (IASLC/ATS/ERS) classification (Travis et al. 2011), based on histomorphological subtypes, the designation bronchioloalveolar carcinoma has been deleted and the term “adenocarcinoma in situ” was introduced for solitary adenocarcinomas ≤3 cm, with pure lepidic growth.
Lepidic growth refers to a characteristic growth pattern along preexisting alveolar structures, without invasion, commonly associated with mild septal thickening and fibrosis (Travis et al. 2004). Other lung adenocarcinoma subtypes can also exhibit, at least in part, a lepidic growth pattern, like minimally invasive adenocarcinoma when lepidic growth is predominant with an invasive pattern ≤5 mm, or lepidic-predominant adenocarcinoma, for the invasive adenocarcinomas that were previously considered to have a predominant bronchioloalveolar carcinoma pattern (Travis et al. 2011). That classification has demonstrated prognostic value. Indeed, in a retrospective series of 514 patients with stage I adenocarcinomas of the lung (7th edition of the UICC/AJCC staging system) (Sobin et al. 2009; Edge et al. 2010; Yoshizawa et al. 2011), adenocarcinomas in situ and minimally invasive adenocarcinomas had 100% 5-year disease-free survival, whereas lepidic-predominant adenocarcinomas had an intermediate clinical behavior compared with other solid invasive adenocarcinomas, with 90% 5-year disease-free survival.
Lymph vessels within and around malignant tumors have been shown to play an important role in the progression of several tumor types (Van der Auwera et al. 2006). In NSCLC, an association between poor prognosis and lymphatic vessel density (LVD) was clearly demonstrated (Renyi-Vamos et al. 2005; Kadota et al. 2008; Sun et al. 2009).
In that context, taking advantage of the identification of intralobular lymphatic vessels in normal lung (Kambouchner and Bernaudin 2009; Sozio et al. 2012) and using the D2-40 monoclonal antibody as a reliable marker of lymphatic endothelial cells (Evangelou et al. 2005; Van der Auwera et al. 2006), we evaluated the lymphatic vascular pattern associated with lepidic growth or the invasive component, to obtain insight into lymphangiogenesis in pulmonary adenocarcinomas.
Materials and Methods
Materials
Tumor-tissue samples investigated herein were retrospectively obtained from the archives of two departments of pathology (Hôpital Bichat, Paris and Hôpital Avicenne, Bobigny). Formalin-fixed, paraffin-embedded tissues were obtained from 12 patients (four women and eight men; median age: 68 [range, 20–85] years), who underwent surgical pulmonary resection for lung adenocarcinomas (lobectomy n=10, pneumonectomy n=1, or large wedge excision n=1). To obtain a panel of adenocarcinomas with exclusive or associated lepidic growth pattern, we specifically chose a set of samples from tumors identified according to the IASLC/ATS/ERS classification of lung adenocarcinomas: four adenocarcinomas in situ (two mucinous and two non-mucinous) and eight invasive adenocarcinomas exhibiting a lepidic growth pattern (six non-mucinous: one minimally invasive, three lepidic-predominant, one micropapillary-predominant, one acinar-predominant, and two mucinous adenocarcinomas). Their clinicopathological features are summarized in Table 1. For each of the samples examined, it was verified on hematoxylin-and-eosin–stained sections that they contained a lepidic growth pattern.
Clinical and Histopathological Features.
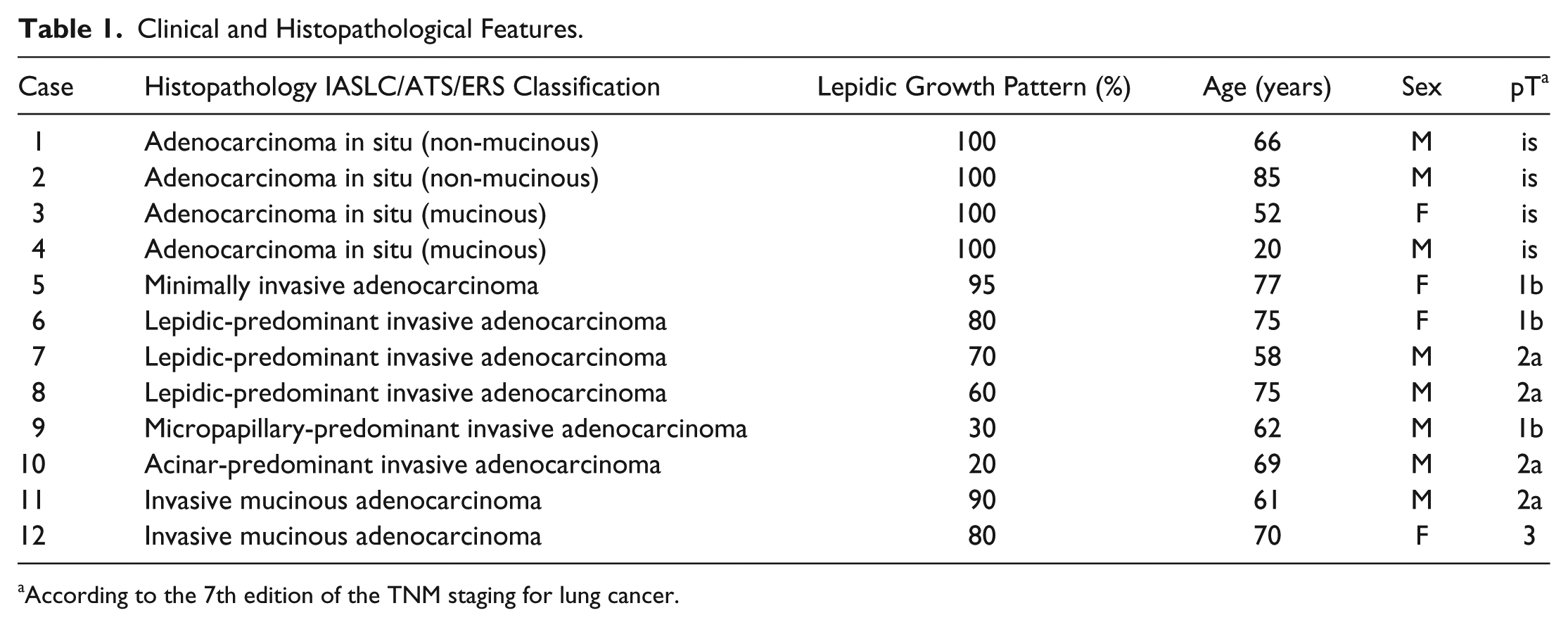
According to the 7th edition of the TNM staging for lung cancer.
Moreover, for comparison, normal lung tissue was obtained from surgical specimens of two patients who underwent pulmonary resection for lung cancer; the samples were taken far from the tumor.
This retrospective study was conducted in accordance with French legislation on medical research, exempting this type of study from obtaining informed consent.
Immunohistochemistry
Sections (5-µm thick) were deparaffinized in xylene and rehydrated in graded ethanol concentrations. Thereafter, sections were immunolabeled using an automated procedure (Ventana Medical Systems, Tucson, AZ). Antigens were retrieved by boiling at 95–100C. The D2-40 mouse IgG1 monoclonal antibody (Dako, Glostrup, Denmark; clone 07 3611) recognizes podoplanin, a 38-kDa transmembrane glycoprotein expressed on lymphatic vascular endothelium (Van der Auwera et al. 2006). The anti-human smooth muscle actin (SMA) monoclonal antibody (Dako, clone 1A4) recognizes smooth muscle cells and myofibroblasts and was used here to identify small arteries or veins.
Sections were incubated for 30 min with D2-40 monoclonal antibody (diluted 1:100) or anti-human SMA monoclonal antibody (diluted 1:100); then the biotinylated universal anti-mouse immunoglobulin secondary antibody (Ventana) for 16 min at 37°C; followed by the avidin–biotin–horseradish peroxidase complex, using the Ventana Medical Systems Basic diaminobenzidine detection kit for 8 min at 37C, with the peroxidase-diaminobenzidine reaction forming the visualized chromogen.
As negative controls, sections from each paraffin block were incubated with a normal mouse IgG1 (Dako; X0931 culture supernatant) instead of the primary antibody.
Microscopy and Morphometry Analysis
Two different image acquisitions were obtained. Images of the D2-40–immunolabeled slides were acquired with a Leica DM2500 microscope equipped with a Leica DFC300 FX digital color camera connected to a computer loaded with image scan software (Tribvn, Châtillon, France), whereas SMA-immunolabeled slides were digitized using a ScanScope digital slide scanner (Aperio, Vista, CA).
Each D2-40–immunolabeled slide was examined with a ×10 objective, as for Fig. 1. Then, for each pattern (i.e., adenocarcinoma in situ, lepidic pattern of invasive adenocarcinoma, and invasive pattern of invasive adenocarcinoma), two sets of 10 × 10-objective fields were digitized for each pattern: 10 consecutive fields in one part of the pattern and 10 other consecutive fields, far from the first 10 fields, randomly selected in another part of the section showing the same pattern. Using a calibrated micrometer grid, it was possible to calculate that each ×10 field equaled 553,500 µm2; therefore, 20 consecutive ×10 fields cover a total area of 11.07 mm2.
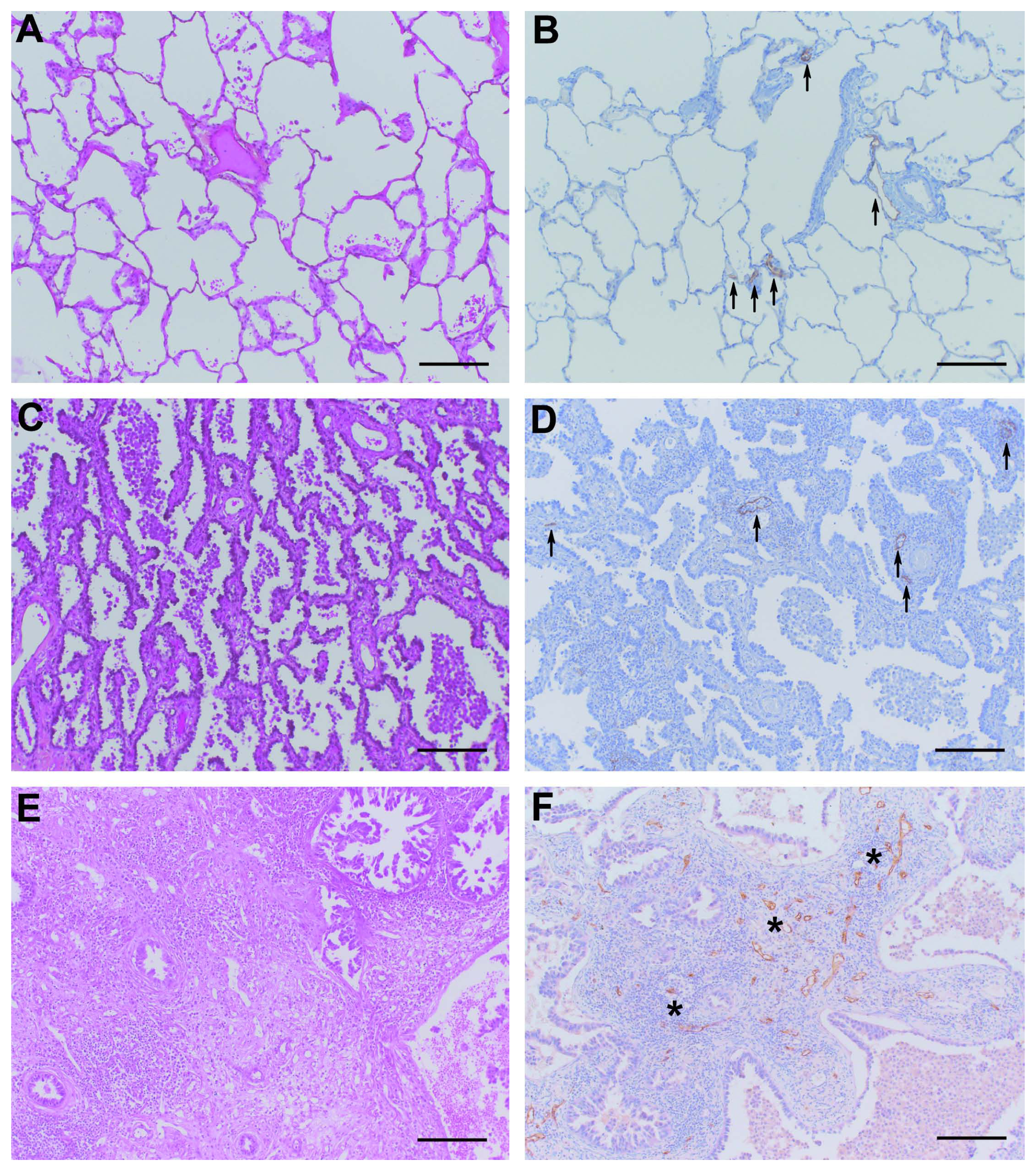
Hematoxylin–eosin-stained sections (A, C, E) and D2-40 immunolabeling (B, D, F) of normal lung (A, B), non-mucinous adenocarcinoma in situ (AIS; C, D), and invasive pattern of invasive adenocarcinoma (E, F) (×10 magnification). Arrows point to the few D2-40–labeled lymphatic vessels in normal lung (B) and adenocarcinoma in situ (D). However, in the invasive adenocarcinoma (F), lymphatic vessels (labeled brown, stars) are too numerous to be individually indicated. Bars = 200 µm.
For morphometry analysis, the digitized fields were analyzed by the same observer (AS) using Photoshop software (Adobe Photoshop CS5 extended, http://www.adobe.com/products/photoshop/photoshopextended/).
D2-40–immunolabeled lymphatics with a clear lumen were manually colored red. In normal lung and the lepidic patterns, the lymphatic vessels were classified according to their location, either in an interlobular septum or interalveolar within the pulmonary lobule. As previously reported for normal lung (Kambouchner and Bernaudin 2009; Sozio et al. 2012), these intralobular lymphatic vessels were either associated with interalveolar arterioles or venules, or isolated intralobular septal lymphatic vessels at a distance from interalveolar arterioles or venules.
Next, each picture was converted with Photoshop software into a grayscale image with gray for lymphatic vessels and white (0 grayscale) for the rest of the picture. These grayscale images were then saved as TIF files, measuring 1024 × 768 pixels.
For morphometry analyses, the images were thresholded, and lymphatic vessel numbers and areas were determined with Image J software (http://rsbweb.nih.gov/ij/). Then, in the lepidic pattern of each tumor (adenocarcinoma in situ and invasive adenocarcinoma), the area occupied by intra-alveolar air was manually colored red with Photoshop software, converted to a grayscale image and evaluated by Image J software. Knowing the intra-alveolar air area enabled calculation of the tissue area and comparisons of the LVD (i.e., calculated as the number of lymphatic vessel sections per mm2) and lymphatic vessel-area fraction (LVAF; i.e., calculated as the fraction of area occupied by lymphatic vessels in the tissue after exclusion of the intra-alveolar air area) within the lepidic pattern, with those observed in the invasive fraction devoid of intra-alveolar air. Moreover, measurements after excluding intra-alveolar air can be considered to overcome the artifacts of inflated conditions at the time of lung-sample fixation. Data were finally transferred to an Excel chart (Microsoft Office Excel 2007).
Digitalized anti-human SMA-labeled slides were used to determine whether D2-40–immunolabeled intralobular lymphatics were or not in the vicinity of intralobular arterioles or venules.
Statistical Analysis
Data were compared using Student’s t-test or the Mann–Whitney U test; p<0.05 was considered significant.
Results
Morphology
All the studied samples contained a lepidic growth pattern, constituting either 100% of the four adenocarcinomas in situ or 20–95% of the other analyzed tumors (Table 1). Mucinous adenocarcinomas were identified by a foamy cytoplasm in the presence of numerous microvacuoles or a single large mucin-filled vacuole. Lymphatic vessels identified by the D2-40–labeled endothelium surrounding a distinct lumen were clearly observed in all the studied samples. D2-40 antibody did not bind to tumor cells, regardless of the adenocarcinoma pattern. Within the lepidic fraction of tumors, only lymphatics were immunolabeled. However, in the invasive areas, some other labeling, which might correspond to collapsed small lymphatics or tumor-associated fibroblasts, was observed in addition to lymphatics. SMA labeling clearly identified the muscle layer of intralobular arterioles or venules. No immunolabeling was seen on the sections after incubation with the normal mouse IgG1 instead of the primary antibody.
LVD
No significant differences were observed between the mean ± SD LVD (the number of lymphatics per mm2) measured in the four adenocarcinomas in situ (2.1 ± 0.7 mm−2) and the eight lepidic patterns of invasive adenocarcinomas (2.4 ± 1 mm−2) (p=0.73). In contrast, after pooling the twelve adenocarcinoma in situ and lepidic pattern of invasive adenocarcinoma values, their mean LVD of 2.3 ± 0.8 mm−2 differed significantly from that of the eight invasive patterns of invasive adenocarcinoma (14.9 ± 13.6 mm−2) (p=0.001) (Fig. 2A). When LVD was calculated after exclusion of the space occupied by air in adenocarcinomas in situ and lepidic patterns of invasive adenocarcinomas, the difference persisted: the pooled mean of adenocarcinoma in situ (4.4 ± 2.5 mm−2) and lepidic pattern of invasive adenocarcinoma (4.1 ± 1.8 mm−2) LVD of 4.2 ± 1.9 mm−2 versus that of invasive patterns of invasive adenocarcinomas (14.9 ± 13.6 mm−2) (p=0.02), as illustrated in Fig. 3.
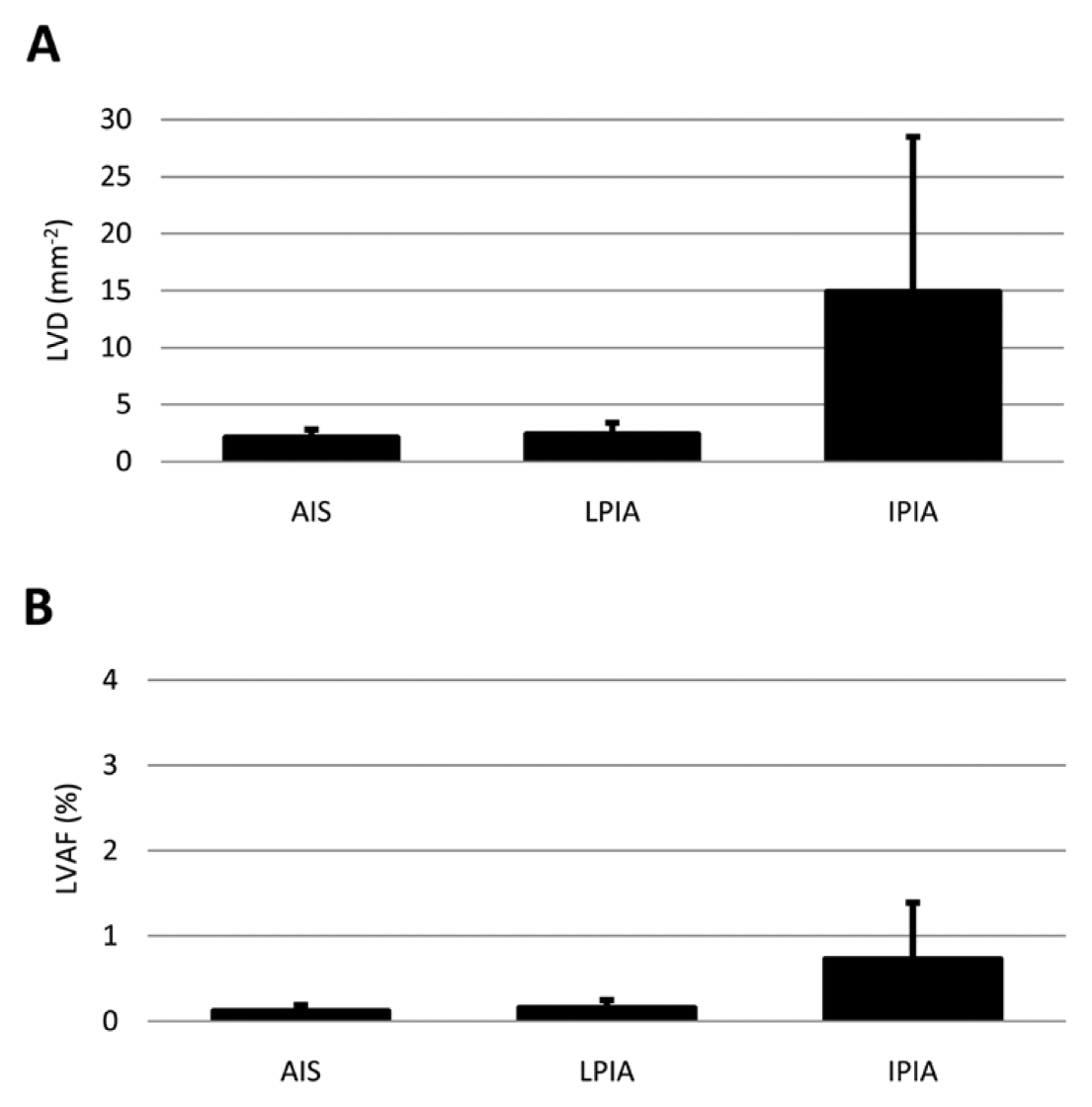
A, Lymphatic vessel density (LVD), calculated as the number of sections of lymphatic vessels per mm2 in adenocarcinoma in situ (AIS), lepidic pattern of invasive adenocarcinoma (LPIA), and invasive pattern of invasive adenocarcinoma (IPIA). B, Lymphatic vessel-area fraction (LVAF), calculated as the fraction of area occupied by the lymphatic vessels in the tissue (i.e., intra-alveolar air area excluded), in AIS, LPIA, and IPIA (mean ± SD). LVD did not differ between AIS and LPIA (p=0.73). AIS and LPIA LVD and LVAF differed significantly from IPIA (p=0.001 and p=0.002, respectively) (Mann–Whitney U test).
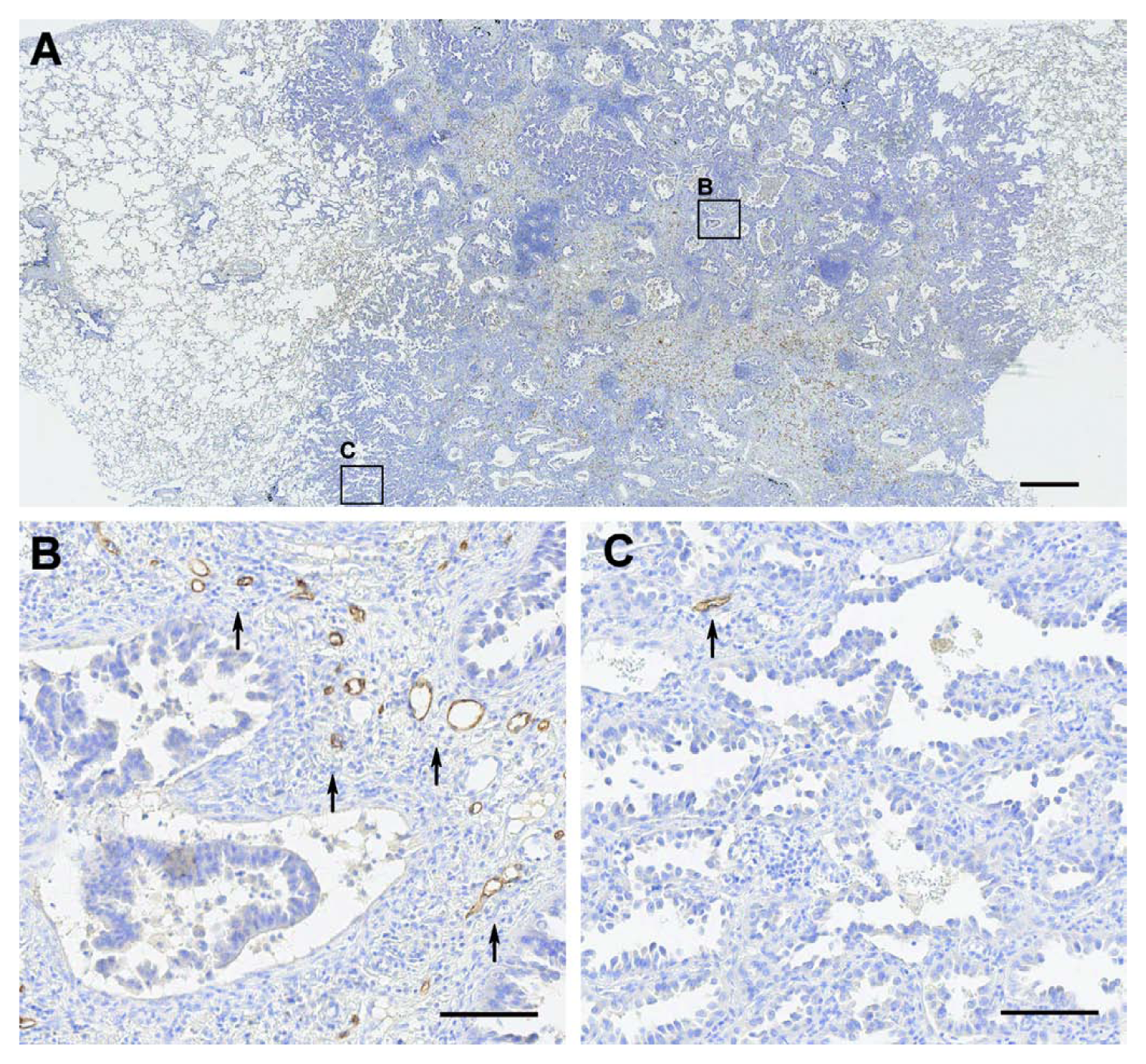
D2-40 monoclonal antibody-immunolabeled section of a micropapillary-predominant invasive adenocarcinoma with a peripheral lepidic pattern, at ×2.5 (A) and ×20 magnification (B, C). High magnification of the invasive pattern showing high D2-40–labeled lymphatic vessel density (arrows) (B). Lepidic pattern with a rather poorly developed lymphatic network (arrow) (C). Bars (A) = 500 µm; (B, C) = 100 µm.
LVAF
Mean ± SD LVAF (the fraction of tissular area occupied by lymphatics) was 0.12% ± 0.07% in adenocarcinomas in situ, 0.16% ± 0.09% in lepidic patterns of invasive adenocarcinomas and 0.73% ± 0.66% in invasive patterns of invasive adenocarcinomas (Fig. 2B). Comparing adenocarcinoma in situ and the lepidic pattern of invasive adenocarcinoma versus invasive pattern of invasive adenocarcinoma, a significant difference (p=0.002) was observed.
Area of Lymphatic Vessel Cross Sections
The mean areas occupied by lymphatic vessel cross sections in adenocarcinomas in situ or lepidic and invasive patterns of invasive adenocarcinomas were comparable (p=0.24) (Table 2). However, lymphatics observed in interlobular septa were larger than those located in the interalveolar septa, either perivascular or isolated, in adenocarcinomas in situ or lepidic patterns of invasive adenocarcinomas (p=0.0002). Furthermore, intralobular lymphatic vessels located in the interalveolar septa but close to an arteriole or venule were larger than those at a distance from those blood vessels (p=0.0002), as detailed in Table 2. Although lymphatics in interlobular septa were narrower in adenocarcinomas in situ and lepidic patterns of invasive adenocarcinomas than in normal lung, they were still larger, as in the normal lung, than those located within the interalveolar septa.
Mean ± SD Areas of Lymphatic Vessel Sections (µm2).
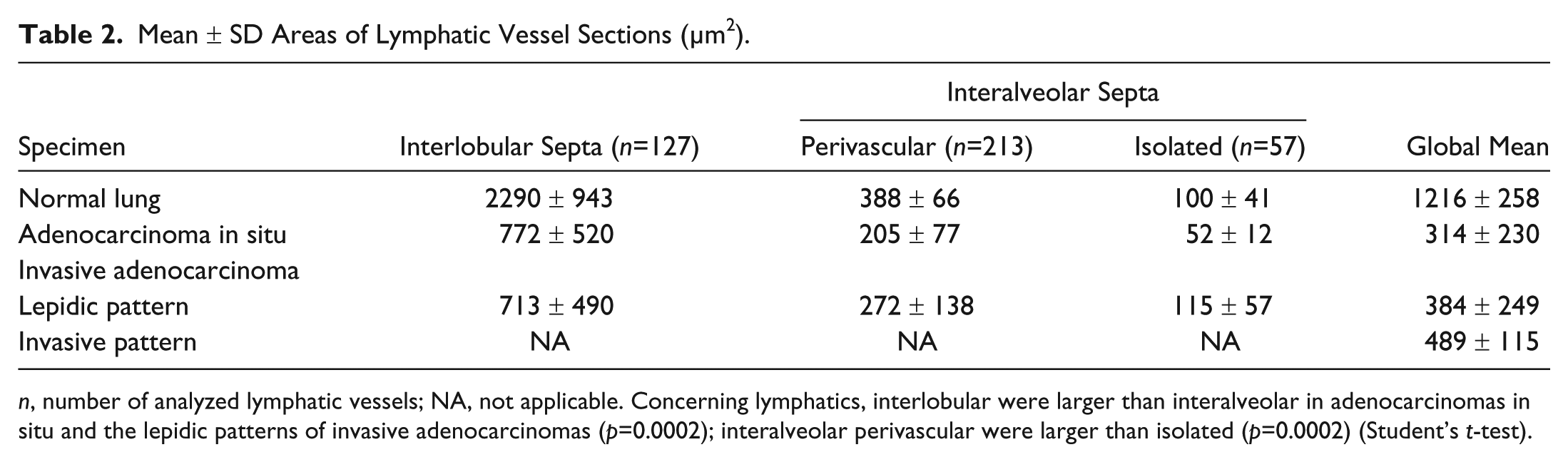
n, number of analyzed lymphatic vessels; NA, not applicable. Concerning lymphatics, interlobular were larger than interalveolar in adenocarcinomas in situ and the lepidic patterns of invasive adenocarcinomas (p=0.0002); interalveolar perivascular were larger than isolated (p=0.0002) (Student’s t-test).
Pulmonary Lymphatic Network Topography
Considering adenocarcinomas in situ or lepidic pattern of invasive adenocarcinomas, respectively, 23.7% and 29.9% of lymphatic vessels were located within interlobular septa, and 76.3% and 70.1% within interalveolar septa. Among the latter, respectively, 78.9% and 80% of lymphatic vessels were close to an arteriole or a venule, as identified by their open lumen on serial sections and by anti-SMA labeling. Therefore, respectively, 21.1% and 20% of lymphatic vessels were located in interalveolar septa far from those blood vessels. In normal lung, 45.6% of the lymphatic vessels were located in the interlobular septa and 54.4% in interalveolar septa: 75.5% of them close to an intralobular arteriole or venule and 24.5% far from those blood vessels.
Discussion
To identify lymphatic vessels, antivascular endothelial growth factor receptor-3 (VEGFR-3) or antilymphatic vessel endothelial hyaluronan receptor-1 (LYVE-1) antibodies were formerly used but we opted for monoclonal antibody D2-40 because of its higher sensitivity and specificity for labeling lymphatic endothelial cells (Evangelou et al. 2005; Van der Auwera et al. 2006; Kambouchner and Bernaudin 2009; Sozio et al. 2012). Anti-VEGFR-3 antibodies were shown to lack specificity and do not clearly discriminate between vascular and lymphatic endothelial cells, due to VEGFR-3 upregulation during tumor angiogenesis (Niki et al. 2001). Moreover, some studies found a lack of LYVE-1 sensitivity because it is not always expressed in lymphatic vessels associated with tumors (Van der Auwera et al. 2006). Normal or tumor cells other than endothelial cells from lymphatics, such as cancer-associated myofibroblasts (Kawase et al. 2008; Ito et al. 2012) or squamous carcinoma cells (Iwakiri et al. 2009), have been shown to express podoplanin, recognized by the D2-40 antibody. However, when considering vascular systems, podoplanin is exclusively expressed by the lymphatic endothelium, thereby explaining why D2-40 is widely considered specific to lymphatic identification. Moreover, using D2-40 as a marker of lymphatic endothelium enabled reappraisal of the organization of the intralobular lymphatic network in normal lung (Kambouchner and Bernaudin, 2009; Sozio et al. 2012) and pulmonary interstitial diseases (Kambouchner et al. 2011). In this study, only D2-40–immunolabeled lymphatics clearly exhibiting an open lumen were counted, to avoid taking into account cancer-associated myofibroblasts (Kawase et al. 2008; Ito et al. 2012) that might be recognized by D2-40 antibody, as previously described.
In most studies devoted to lymphangiogenesis in carcinomas, only lymphatic vessel “hot spots” were considered (Koukourakis et al. 2005; Takanami 2006; Faoro et al. 2008; Iwakiri et al. 2009; Sun et al. 2009) and their density was often demonstrated to be a prognostic factor (Takanami 2006; Iwakiri et al. 2009; Sun et al. 2009). These so-called hot spots are tumor zones giving the impression, at low magnification, of containing numerous lymphatic vessels but their identification is observer-dependent (Van der Auwera et al. 2006). The interobserver variability inherent in this method was underlined by the discordant results of several studies evaluating LVD. Therefore, Shields et al. (2004) suggested that absolute lymphatic vessel-density determination would be more appropriate and facilitate data comparisons. Thus, our aim was to establish a map of lymphatic vessels associated with the lepidic pattern of lung adenocarcinomas, as previous reports on lymphangiogenesis in NSCLC only dealt with invasive carcinomas (Koukourakis et al. 2005; Renyi-Vamos et al. 2005; Kadota et al. 2008; Iwakiri et al. 2009; Sun et al. 2009). Niki et al. (2001) studied a series of pure bronchioloalveolar carcinomas (i.e., adenocarcinoma in situ according to the new classification) but with anti-VEGFR-3 antibody, which does not clearly discriminate between vascular and lymphatic endothelial cells.
To do so, we counted lymphatic vessels and determined their density per mm2 (LVD) or the fraction of area occupied by lymphatics (LVAF) in 20 fields randomly selected for each case and found that the lymphatic microvasculature observed in the lepidic pattern of tumor growth in adenocarcinomas in situ or invasive adenocarcinomas was similar. Moreover, it must be underscored that the lymphatic network topography in the lepidic growth fraction is comparable to that in the intralobular distribution in normal lung (Kambouchner and Bernaudin 2009; Sozio et al. 2012). In contrast, a higher LVD was observed in the invasive fractions of tumors, suggesting that a different network pattern is associated with invasion.
Because lymphatics may collapse during fixation and tissue processing, some bias might have been introduced into the quantitative data, particularly in terms of lymphatic vessel size. However, the interalveolar matrix in the lepidic fractions of growth is thicker and, therefore, more rigid than in normal lung, which could explain why interalveolar lymphatics almost always had clear lumens. Hence, the main bias could have been to underestimate the data for normal lung or the invasive patterns of the tumors, which would reinforce the message on the low number of lymphatics associated with lepidic growth.
The results of this study suggest that lymphangiogenesis is not associated with the lepidic growth of pulmonary adenocarcinomas, which is a pattern of intraepithelial tumor growth and/or spread. To date, only a few studies have evaluated the LVD in intraepithelial neoplasias in other organs and dissimilar results have been reported. For example, Jach et al. (2011) found the highest LVD in vulvar intraepithelial neoplasia compared with vulvar invasive cancer, whereas Heindl et al. (2010) showed that the LVD of proliferating lymphatic vessels was higher in conjunctival squamous cell carcinomas than in conjunctival intraepithelial neoplasias, with a statistically significant difference between the high-grade and low-grade latter lesions.
We also confirmed the presence of isolated, small-caliber lymphatic vessels within interalveolar septa, far from bronchioles, arterioles or venules, in adenocarcinomas in situ (21.1% of intralobular lymphatic vessels) and lepidic patterns of invasive adenocarcinomas (20%), as in normal lung (24.5%). These findings are in agreement with other observations showing normal lung lymphatic vessels extending into the interalveolar septa (Kambouchner and Bernaudin 2009; Sozio et al. 2012). Although Koukourakis et al. (2005) did not observe any intratumoral lymphatic network within a series of lung carcinomas with LYVE-1 antibody, we found lymphatic vessel hot spots within the invasive patterns of lung adenocarcinomas. Our observation is in agreement with Renyi-Vamos et al. (2005), who observed D2-40–positive lymphatic vessels in the central areas of tumors, but none of those lymphatic vessels was recognized by anti-LYVE-1, despite LYVE-1 and podoplanin having similar labeling patterns in normal lung and at the tumor periphery. Thus, the discrepancy of our results with those of Koukourakis et al. (2005) could, in part, be explained by the better sensitivity of anti-D2-40 monoclonal antibody than anti-LYVE-1 to recognize lymphatic vessels entrapped in the invasive tumor.
LVD has been associated with poor prognosis of various tumors, notably NSCLC (Renyi-Vamos et al. 2005; Kadota et al. 2008; Sun et al. 2009). Thus, the good prognosis of adenocarcinomas in situ, minimally invasive adenocarcinomas and lepidic-predominant adenocarcinomas (Russell et al. 2011; Yoshizawa et al. 2011) could be explained, at least in part, by the maintenance of a rather poorly developed lymphatic network, as shown herein. In contrast, LVAF was 5–6-fold lower in the lepidic pattern of invasive adenocarcinomas and adenocarcinomas in situ than the invasive patterns of invasive adenocarcinomas.
Lymphangiogenesis is considered to be mainly related to tumor-cell secretion of VEGF-A, VEGF-C and VEGF-D, which interact with VEGFR-3 expressed on lymphatic endothelial cells (Van der Auwera et al. 2006). Low VEGF levels were reported in adenocarcinoma in situ, that is, formerly bronchioloalveolar carcinoma, compared with “conventional adenocarcinoma” (Saad et al. 2004). Enatsu et al. (2006) observed an association between hypoxia-inducible factor-1α (HIF-1α) and VEGF-A expression in pulmonary adenocarcinomas, and little or no reactivity of HIF-1α was observed in the bronchioloalveolar components, in contrast to the solid components of adenocarcinomas with mixed subtypes. Therefore, the absence of lymphangiogenesis in the areas of lepidic growth might reflect the absence of VEGF secretion by tumor cells, in contrast to the VEGF-C secretion in invasive adenocarcinomas reported by Saintigny et al. (2007).
In conclusion, the results of our study suggest the absence of septal lymphangiogenesis associated with the lepidic tumor-growth pattern in lung adenocarcinomas. This characteristic could explain, at least partially, the better prognosis of tumors with an exclusive lepidic growth pattern in which an absence of nodal metastatic spread is observed.
Footnotes
Acknowledgements
The authors thank Mrs. Véronique Albano (Department of Pathology, Hôpital Bichat, AP–HP, Paris, France) for her technical assistance, Pr. Dominique Henin for her constant support, and Ms. Janet Jacobson for editorial assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
