Abstract
Birth dating neurons with bromodeoxyuridine (BrdU) labeling is an established method widely employed by neurobiologists to study cell proliferation in embryonic, postnatal, and adult brain. Birth dating studies in the chick dorsal telencephalon and the mammalian striatum have suggested that these structures develop in a strikingly similar manner, in which neurons with the same birth date aggregate to form “isochronic clusters.” Here we show that isochronic cluster formation in the chick dorsal telencephalon is an artifact. In embryos given standardly employed doses of BrdU, we observed isochronic clusters but found that clusters were absent with BrdU doses close to the limits of detection. In addition, in situ hybridization experiments established that neurons in the clusters display errors in cell type specification: BrdU cell clusters in nidopallium adopted a mesopallial neuronal fate, mesopallial clusters were misspecified as nidopallial cells, and in some instances, the BrdU clusters failed to express neuronal differentiation markers characteristic of the dorsal telencephalon. These results demonstrate that the chick dorsal telencephalon does not develop by isochronic cluster formation and highlight the need to test the integrity of BrdU-treated tissue with gene expression markers of regional and cell type identity.
Keywords
Birth dating studies with tritiated thymidine ([3H]dT) and bromodeoxyuridine (BrdU) have generated critical insights into the cellular mechanisms of brain development. Classic studies with [3H]dT established, for example, that cortical structures such as the mammalian neocortex develop in an “inside-out” pattern, where early born neurons populate deep layers and later born neurons occupy more superficial layers (Angevine and Sidman 1961). By contrast, many nuclear structures such as the mammalian dorsal thalamus are generated with the inverse “outside-in” pattern (Angevine 1970). In the late 1980s, BrdU birth dating with immunohistochemistry essentially replaced the more demanding [3H]dT autoradiography, with one key advantage being that BrdU-labeled cells can be directly studied with molecular markers of specific neuronal cell types (Miller and Nowakowski 1988; Nowakowski et al. 1989). This technique has been widely employed to investigate cell type–specific birth dating during embryonic, postnatal, and adult neurogenesis in a variety of vertebrate species, including both birds and mammals.
Experimental birth dating in avian and mammalian systems involves different technical challenges. Because mammals develop in utero, it is possible to deliver a pulse of label that is rapidly cleared from the embryo within minutes or hours (Nowakowski and Rakic 1974; Hayes and Nowakowski 2000; reviewed in Taupin 2007). The cells with the heaviest labeling are identified as those born during the window of the pulse. By contrast, the chick egg is a closed system, and the DNA synthesis labels, once administered, are retained. Consequently, labeling with [3H]dT or BrdU in the chick is cumulative, and cell birth dating is most securely based on the time of detection of unlabeled cells (Sidman 1970; Lavail and Cowan 1971; Tsai et al. 1981a, 1981b).
In 2000, Striedter and Keefer addressed this limitation in the temporal resolution of birth dating in birds. They developed a technique where a relatively low dose of BrdU was injected directly into the vitelline veins of a developing chicken embryo and tested for pulse labeling with [3H]dT delivery a day later. They found that cells were strongly labeled with BrdU only if they were born within 24 hr of BrdU delivery. With this technical innovation, they observed that in the dorsal telencephalon, some neurons with the same birth date aggregate into patches, or “isochronic clusters.” These results generated considerable excitement because isochronic cluster formation is strikingly similar to the cellular development of the striosome system of the mammalian striatum, a nuclear structure in the ventral telencephalon (Angevine and McConnell 1974; Brand and Rakic 1979; Graybiel and Hickey 1982). Although the mammalian striatum is not homologous to any nucleus in the bird dorsal telencephalon (Reiner et al. 2004), these findings raised the intriguing possibility that isochronic cluster production is a general mechanism by which the vertebrate forebrain makes large, complex nuclear structures (Redies et al. 2002).
We revisited BrdU birth dating of the chick dorsal telencephalon to relate isochronic cluster generation to general processes of dorsal telencephalon nucleogenesis. We readily confirmed isochronic cluster formation, but when we studied the BrdU-labeled tissue with in situ hybridization (ISH) for developmental control genes, we saw expression patterns that were never present normally. We found that if we greatly reduced the dose of BrdU, the gene expression patterns were normal and no isochronic clusters were evident. Our findings indicate isochronic clusters are a BrdU-induced artifact and that “routine” doses of BrdU can produce covert, but extensive, errors in regional cell type specification.
Materials and Methods
BrdU Injections
Fertilized White Leghorn chicken (Gallus gallus) eggs were obtained from Phil’s Fresh Eggs (Forreston, IL) and incubated on their side at 37C with 90% relative humidity in a forced-draft incubator. Between embryonic day 2 (E2) and E4, approximately 3 ml of albumen was removed from each egg using an 18-gauge needle attached to a syringe. Eggs were returned to the incubator. BrdU (Sigma-Aldrich, St. Louis, MO) solutions were prepared at 0, 0.15, 0.5, 1.5, 7.5, or 15 mg/ml BrdU in phosphate-buffered saline (PBS; pH 7.4) with 0.01% Fast Green dye added (Sigma-Aldrich). On E5 to E9, a small window was cut in the eggshell and 1 to 3 µl BrdU solution was injected into one of the embryo’s vitelline veins through a glass micropipette (tip diameters: 20–40 microns) connected to a picospritzer. The 15-mg/ml dose is a “routine” dose similar to that employed in other studies of chick forebrain development (Striedter and Keefer 2000; Redies et al. 2002; Heyers et al. 2003). In scout experiments, we found that with the 0.15-mg/ml dose, BrdU was not detected over background (n=8 where all failed detection), whereas with the 0.5-mg/ml dose, a BrdU signal could be regularly, but not invariably, detected (n=18 where 2 failed detection) and is referred to in this study as the threshold dose. Eggs were resealed with Scotch transparent tape and returned to the incubator until tissue collection, which ranged from 24 hr postinjection to embryonic day 20 (1 day before hatching).
Tissue Processing
For chick embryos harvested at E7 through E14, the dissected brains were immersion-fixed overnight in 4% paraformaldehyde in PBS (PFA) at 4C. Embryos collected at E19 or E20 were anesthetized on ice, given a lethal dose of sodium pentobarbital, and perfused through the heart with 20 ml PFA. All animal procedures were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of the University of Chicago. Fixed tissue was cryogenically protected by equilibration in 30% sucrose/PFA at 4C for at least 24 hr. Coronal sections were cut at 28 to 42 microns on a sledge microtome. Sections were mounted and dried for at least 2 hr on Superfrost Plus slides (Fisherbrand, Pittsburgh, PA). Slides were incubated for 15 min in PFA, washed three times in PBS for 5 min, and incubated in proteinase K solution (Roche Diagnostics, Indianapolis, IN; 1 µg proteinase K per milliliter of 100 mM Tris-HCl [pH 8.0], 50 mM EDTA [pH 8.0]) for 30 min at 37C. Sections were postfixed for 15 min in PFA, washed three times for 5 min in PBS, and stored in hybridization solution (200 µg/ml heparin, 12.5 mg/ml yeast RNA, 5× SSC, 1% SDS, 50% formamide) at −20C until BrdU detection or ISH processing.
In Situ Hybridization
The nidopallium marker DACH2 was generously provided by C. J. Tabin (Heanue et al. 1999). The excitatory neuron marker VGLUT2/SLC17A6, the inhibitory neuron marker GAD67/GAD1, and the regional mesopallial marker EMX1 were generated by PCR on chick cDNA. The primers employed were VGLUT2 forward, 5′-GATAATGAACTG TGGAGGCTTTGG-3′; VGLUT2 reverse, 5′-ACCTGGGTT GTAGCACCATACGAC-3′; GAD67 forward, 5′-GTCGTG GACATACTCCTCAACTACG-3′; GAD67 reverse, 5′-GGACCAGAATGGCAGAACACTG-3′; EMX1 forward, 5′-TTCCCCCCTCCTGACACTGCTGTTC-3′; and EMX1 reverse, 5′- ACGTCGATGTCTTCTCCGCTC-3′. The PCR fragments were subcloned in the pCRII-TOPO vector (Invitrogen, Carlsbad, CA). Digoxigenin (DIG)–labeled riboprobes were synthesized from cDNA plasmids by in vitro transcription. Single-color ISH was carried out as previously described (Rowell et al. 2010). Tissue was prehybridized for 1 hr at 72C in hybridization solution. Then, 0.5 to 2 mg antisense RNA probe was added to the hybridization solution, and the incubation proceeded overnight at 72C. The next day, slides were washed three times for 45 min at 72C in Solution X (2× SSC, 1% SDS, 50% formamide). The sections were rinsed three times for 15 min in Tris-buffered saline with Tween-20 (TBST) ((25mM Tris HCl [pH 7.5], 0.136M NaCl, 2.68mM KCl, 1% Tween-20) and blocked for 1 hr in 10% lamb serum/TBST at 4C. Riboprobe-mRNA duplexes were detected with anti-DIG Fab fragments coupled to alkaline phosphatase (Roche Diagnostics). The antibody-phosphatase conjugate was preadsorbed with chick embryo powder/1% lamb serum and added to the tissue at a 1:5000 dilution in TBST/1% lamb serum. The antibody reaction proceeded overnight at 4C. The tissue was washed three times for 15 min in TBST and then once for 10 min in NTMT (100 mM Tris-HCl [pH 9.5]; 100 mM NaCl, 50 mM MgCl2, 1% Tween-20). Signal was detected by phosphatase histochemistry at room temperature employing 5-bromo-4-chloro-3-indolyl phosphate (BCIP) and the formazan nitro blue tetrazolium (NBT) (Denville Scientific, South Plainfield, NJ). BCIP/NBT incubation time was 1 to 5 days.
BrdU Detection
Processed tissue was incubated in hybridization solution for 12 to 18 hr at 72C. Slides were rinsed with preheated 72C Solution X, washed twice in Solution X for 30 min at 72C, washed twice in TBST for 5 min, incubated in 2 N HCl for 30 min, washed five times in 250 mM Tris-HCl (pH 8.5) for 5 min, rinsed three times in TBST for 5 min, and blocked in 10% lamb serum in TBST for 1 hr at 4C. BrdU was detected with a monoclonal mouse anti-BrdU fluorescein antibody (Becton Dickinson, Franklin Lakes, NJ; Leuner et al. 2009), which was added to the tissue at a 1:50 dilution in 2% lamb serum/TBST and incubated for 90 min. Tissue was washed three times in TBST and blocked for 1 hr in 2% lamb serum/TBST at 4C, and the primary antibody was detected with antifluorescein Fab fragments coupled to alkaline phosphatase (Roche Diagnostics). The secondary antibody was preadsorbed with chick embryo powder/1% lamb serum and added to the tissue at 1:5000 in TBST/1% lamb serum for an overnight incubation at 4C. The tissue was washed three times for 15 min in TBST and then once for 10 min in NTMT. The BrdU was detected by phosphatase histochemistry employing BCIP and NBT. Incubation times varied greatly depending on the BrdU dose injected. For the 0-mg/ml dose, no signal and very little background were detected even if slides were incubated for 5 hr in color solution (Suppl. Fig. S1). For the 0.15-mg/ml dose, after 5 hr in color solution, very little signal was detected, and the background was very high. For the threshold 0.5-mg/ml dose, signal was detected after approximately 3 hr, and for the routine 15-mg/ml dose, the tissue needed incubation times of only 10 to 30 min. Slides were rinsed, postfixed, and coverslipped with Eukitt mounting medium (Sigma-Aldrich).
Multicolor Fluorescence ISH and BrdU Detection
Fluorescence ISH was performed as previously described (Rowell et al. 2010). DIG- or 2,4-dinitrophenyl (DNP)–labeled riboprobes were synthesized. After hybridization and high-stringency washes, the tissue was blocked in TBST/B (20% DIG blocking solution from Roche Diagnostics in TBST). Riboprobe-mRNA duplexes were incubated for 2 hr with anti-DIG (Roche Diagnostics) or anti-DNP (PerkinElmer, San Jose, CA) horseradish peroxidase–coupled antibodies at a final concentration of 1:200 in TBST/B. After antibody reaction, slides were washed three times for 15 min in TBST and rinsed twice for 5 min in TNT (0.1M Tris-HCl [pH 7.5], 0.15 M NaCl, 0.05% Tween-20). Signal was demonstrated by incubation with CY3 or CY5 tyramide dyes (PerkinElmer; 1:50 in 1 Plus Amplification Diluent) for 1 hr. Slides were rinsed three times for 15 min in TNT, washed twice for 15 min in detergent mix (1% IGEPAL, 1% SDS, 0.5% deoxycholate, 50 mM Tris-HCl [pH 7.5], 1 mM EDTA [pH 8.0], 150 mM NaCl) at 72C, placed for 30 min in Solution X at 72C, and washed three times for 15 min in TBST. BrdU detection was performed after fluorescence ISH, beginning with the 2 N HCl incubation. We found that the 2 N HCl incubation did not disrupt the fluorescence gene expression signal (Suppl. Fig. S2). The fluorescence BrdU protocol diverges from the tetrazolium BrdU detection method after the anti-BrdU antibody incubation and subsequent TBST washes: Slides were blocked with TBST/B, incubated for 2 hr in preadsorbed antifluorescein horseradish peroxide antibody (PerkinElmer; 1:50 in TBST/B), washed three times for 15 min in TBST, rinsed twice for 5 min in TNT, and incubated with FITC tyramide dye (PerkinElmer; 1:50 in 1 Plus Amplification Diluent) for 1 hr. After fluorescence processing, the slides were rinsed in PBS and coverslipped in ProLong Gold Antifade Reagent (Invitrogen).
Microscopy
Single-color tetrazolium section images were captured on an Axioskop 40 microscope (Zeiss, Oberkochen, Germany) or a MZ FLIII fluorescence dissecting microscope (Leica, Wetzlar, Germany). Fluorescence signals were studied on a TIRF Live Cell microscope (Olympus, Tokyo, Japan) or a DSU spinning disk confocal microscope (Olympus). Images were processed with Photoshop CS3 (Adobe Systems, San Jose, CA) and ImageJ (National Institutes of Health, Bethesda, MD) with hue, saturation, brightness, and contrast altered as necessary.
Results
The avian dorsal telencephalon is divided into 4 broad regions, the hyperpallium, mesopallium, nidopallium, and arcopallium, each of which contains multiple nuclei (Reiner et al. 2004). It is in the central regions, the mesopallium and nidopallium, that isochronic cluster formation has been described (Fig. 1A, F).
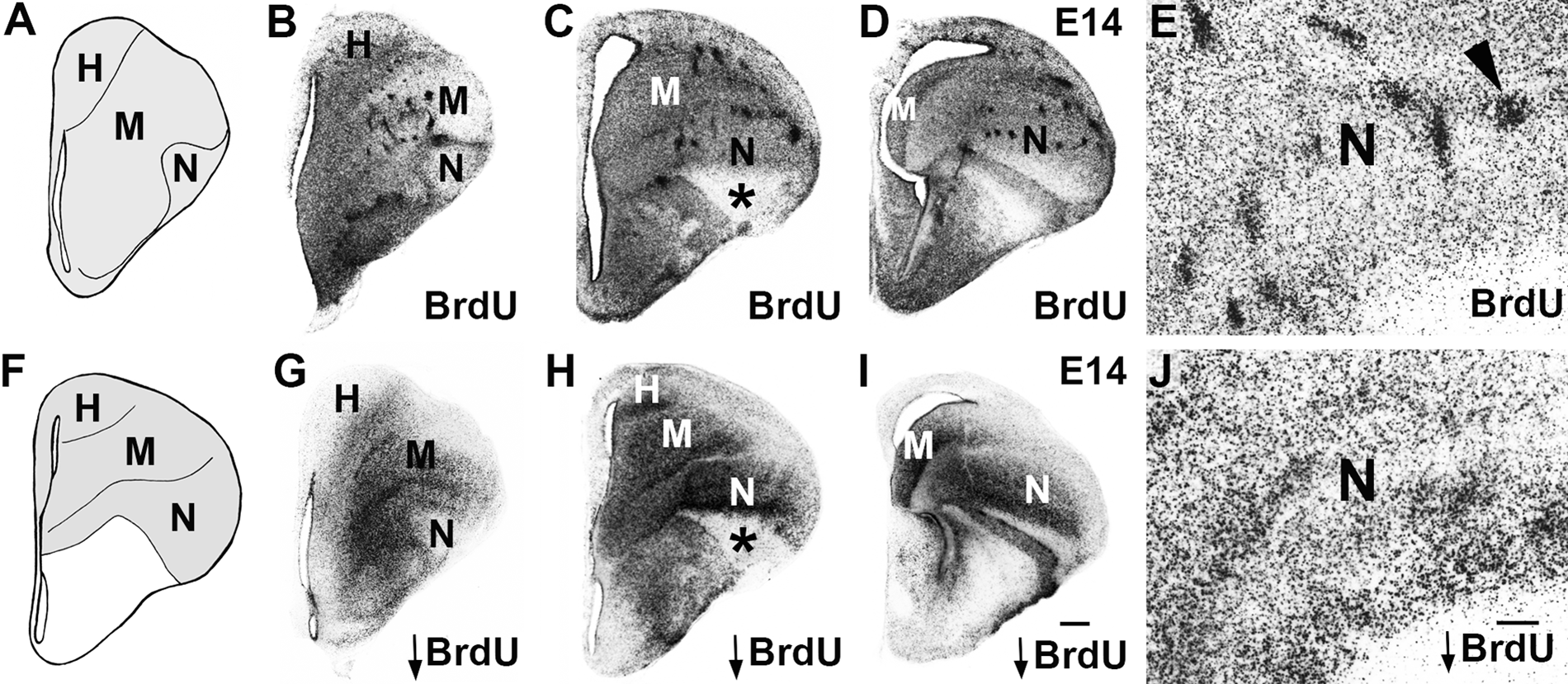
Isochronic clusters are experimental artifacts. (A, F) Coronal section schematics of chick E14 telencephalon at anterior (A, corresponding to panels B, G) and intermediate (F, for panels C, H) levels. The chick dorsal telencephalon (gray) has 4 subdivisions: hyperpallium (H), mesopallium (M), nidopallium (N), and arcopallium, which is located more posteriorly. (B–E, G–J) E14 chick brain processed for BrdU immunohistochemistry following BrdU delivery at E6. (B–E) Isochronic clusters (E, arrowhead) are seen in the mesopallium and nidopallium after a routine BrdU dose (15 µg). (G–J) No isochronic clusters are observed with a threshold dose (0.5 µg, indicated by downward arrow). (B–D, G–I) Series of anterior to intermediate levels. (E, J) High-power photomicrographs of the nidopallium. The lateral or “outside” part of the nidopallium is clear of BrdU signal with both doses (* in C, H). Scale bar in I (for B–D, G–I) is 500 microns and in J (for E and J) is 200 microns. Gamma was adjusted in B–E, G–J.
We used two doses of BrdU to study dorsal telencephalon development, the “routine” dose (15 microgram/microliter) and the “threshold” dose (0.5 microgram/microliter), with the latter representing the lowest dose with which we were able to detect a BrdU signal consistently (see Materials and Methods). These doses resulted in dramatically different patterns of cellular labeling. The routine dose, injected at embryonic day 6 (E6), generated large BrdU clusters in the mesopallium and nidopallium of E14 brains (Fig. 1B–E), as expected (Striedter and Keefer 2000). With the threshold dose, BrdU clusters were not observed, and labeled cells were distributed more uniformly (Fig. 1G–J). We found in a dose-response assessment that increasing the BrdU dose resulted in progressively larger and more prominent BrdU clusters (Suppl. Fig. S3). With all doses, we saw evidence for the “outside-in” pattern of chick dorsal telencephalon generation described by Tsai and colleagues (1981a, 1981b) in [3H]dT birth dating experiments. For example, with E6 delivery, the ventrolateral-most part of the nidopallium was relatively clear of BrdU-labeled cells, demonstrating that this “outside” region of the dorsal telencephalon is generated prior to E6 (cf. Fig. 1C, H). Because BrdU isochronic clusters appear only with the higher dose and not with the threshold dose, we conclude these clusters are an experimental artifact and do not reflect the native pattern of chick dorsal telencephalon neurogenesis.
We studied the molecular character of the BrdU isochronic clusters with gene expression for the regional transcription factors EMX1 and DACH2, as well as the glutamate and GABA neurotransmitter markers VGLUT2 and GAD67. EMX1 and DACH2 are expressed in mutually exclusive domains in chick dorsal telencephalon, with EMX1 identifying the mesopallium and DACH2 marking out the nidopallium (Fernandez et al. 1998; Szele et al. 2002) (Suppl. Figs. 1 and 4). This expression pattern, with a sharp and smooth expression boundary between the EMX1 and DACH2 territories, was also observed following threshold doses of BrdU (Fig. 2A and Suppl. Fig. S4). Gene expression in brains injected with the routine BrdU dose, however, was abnormal. Fluorescence in situ hybridization (FISH) demonstrated that the boundary between EMX1 and DACH2 gene expression territories was severely disrupted, forming corrugations, and within the EMX1 and DACH2 expression domains, there were gaps (Fig. 2B). Strikingly, these disruptions of the native gene expression patterns often coincided with the BrdU clusters, indicating that neurons in many BrdU clusters were misspecified (Fig. 2B–D and Suppl. Fig. S5). We observed a variety of cell fate errors. In the most dramatic examples, cells took on a regionally inappropriate fate where BrdU clusters in the nidopallium had an expression profile indicative of the mesopallium (EMX1+/DACH2−) (Fig. 2E–G and Fig. 3A, B), and BrdU clusters in the mesopallium molecularly resembled the nidopallium (EMX1–/DACH2+) (Fig. 3C, D, F–H). In other examples, the BrdU clusters expressed DACH2 and EMX1 either weakly or not at all (Suppl. Fig. S5). In addition, although many BrdU clusters were enriched with excitatory neurons (Fig. 4A–C), as indicated by expression of vesicular glutamate transporter VGLUT2, other clusters had very little or no VGLUT2 expression (Fig. 3E, F and Fig. 4D–J) and did not show enhanced expression of the inhibitory neuron marker GAD67 (Fig. 4H, J). These VGLUT2–/GAD67– clusters were often located at the mesopallium-nidopallium border (Fig. 4D–F), suggesting that they were sequestered at this boundary. Finally, some clusters contained cells with a mixture of different fates (Fig. 4K–N). These findings demonstrate that BrdU can produce multiple cell fate errors, with some cells adopting the wrong regional gene expression profile and others failing to complete a neuronal differentiation program to acquire glutamatergic or GABAergic phenotypes.
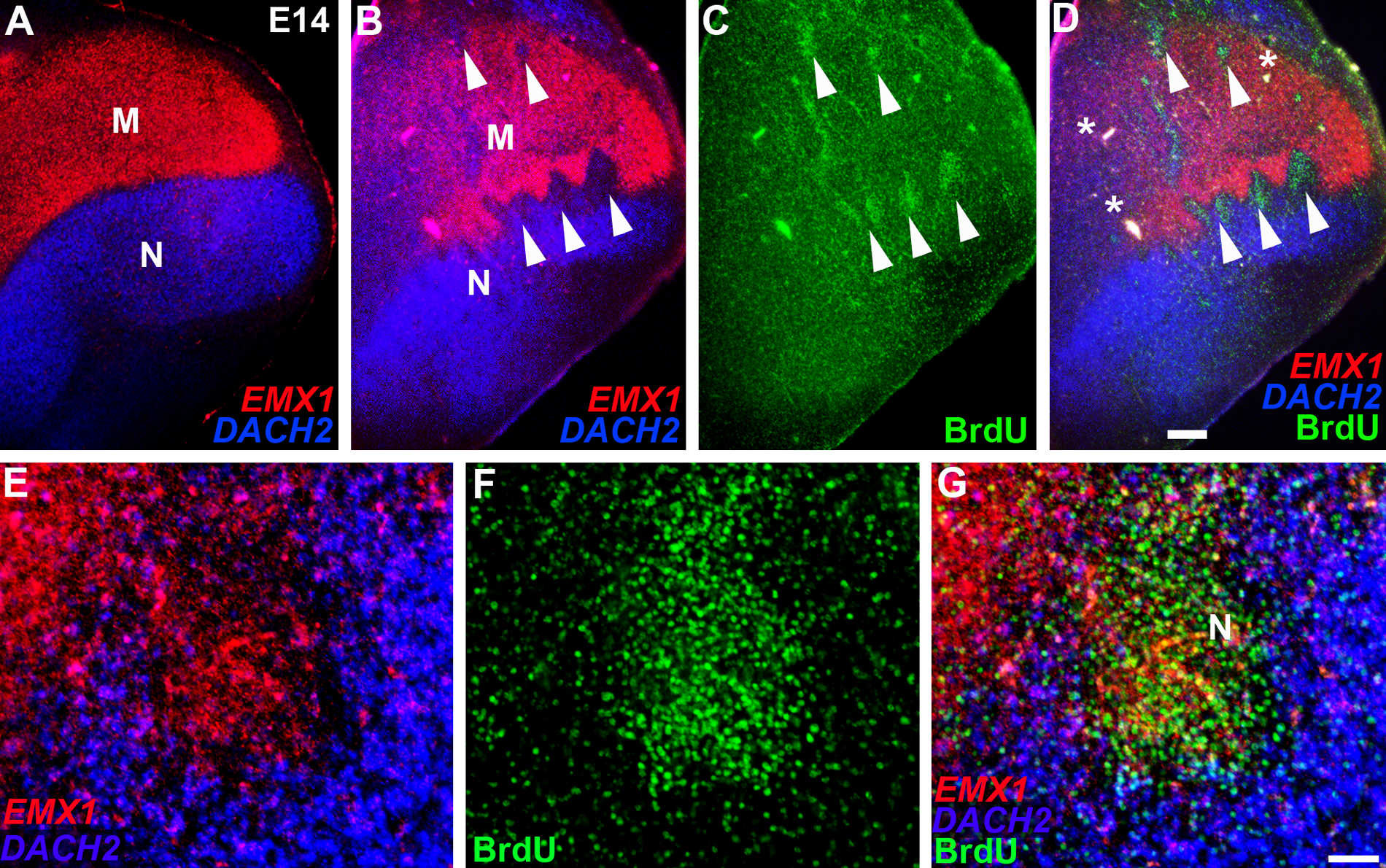
BrdU can produce cell fate specification errors in the chick dorsal telencephalon. (A–G) E14 chick telencephalon following BrdU delivery at E6. (A) Regional gene expression markers EMX1 (red, in the mesopallium) and DACH2 (blue, in the nidopallium) retain their native patterns after a threshold dose of BrdU. (B–D) The boundary between the mesopallium (M) and nidopallium (N) is severely disrupted by a routine BrdU dose. Holes in EMX1 (red) and DACH2 (blue) expression coincide with BrdU (green) isochronic clusters (white arrowheads). Panel D is an overlay of B and C. The asterisks in panel D identify vasculature. (E–G) Higher power photomicrographs of the nidopallium/mesopallium boundary showing a routine dose-induced BrdU cluster (green) is EMX1+ (red) and DACH2– (blue). Panel G is an overlay of E and F. Scale bar in D (for A–D) is 500 microns and in G (for E–G) is 100 microns. Gamma was adjusted in all panels.
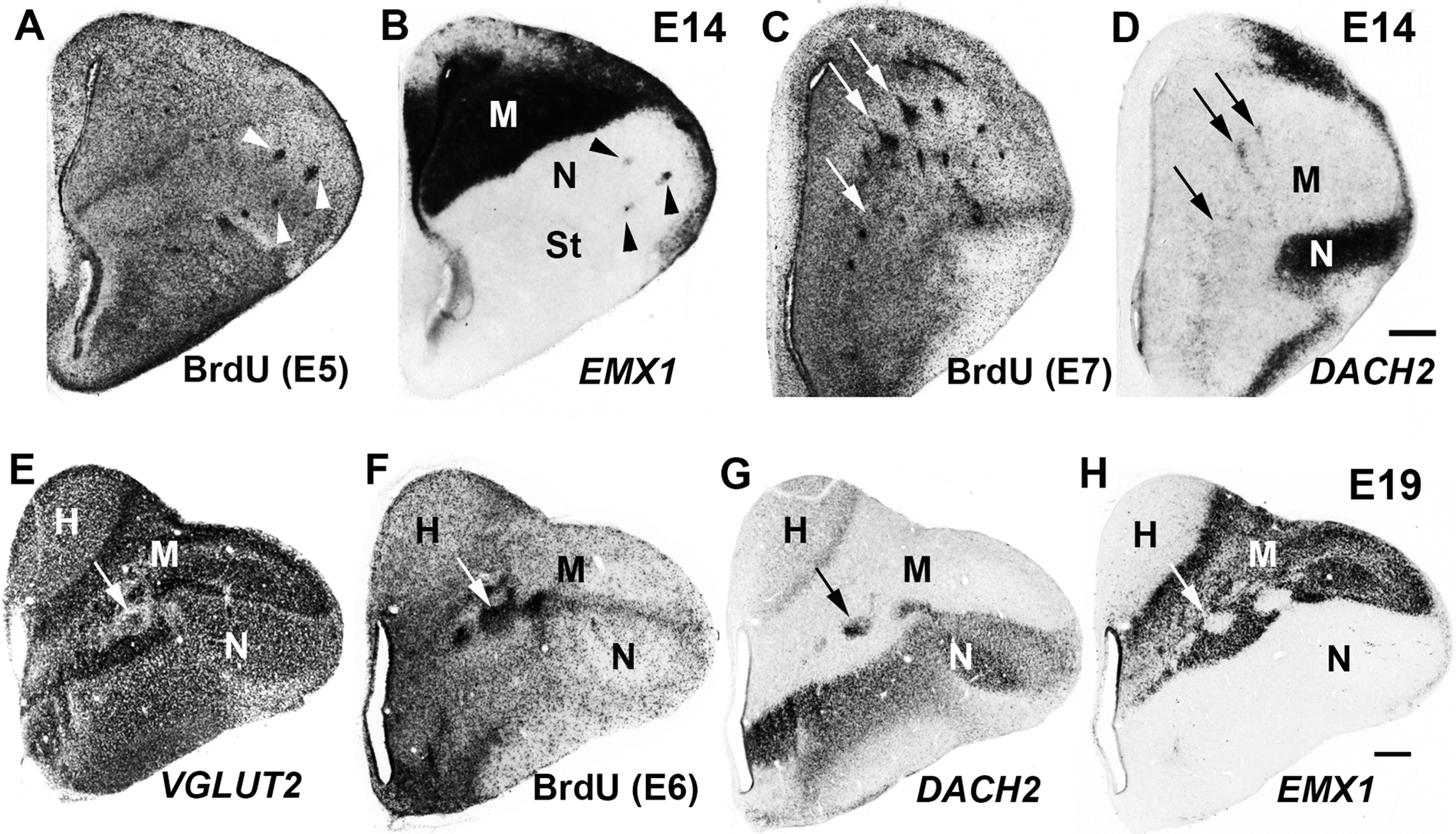
Isochronic clusters with inappropriate gene expression are produced during peak neurogenesis and survive throughout embryogenesis. (A–H) Coronal sections of E14 (A–D) and E19 (E–H) chick telencephalon processed for BrdU detection (A, C, F) or gene expression (B, D, E, G, H) after routine doses of BrdU delivered on E5 (A, B), E7 (C, D), or E6 (E–H). A and B, C and D, and E to H illustrate serially adjoining sections. (A, B) BrdU-positive clusters in the nidopallium (N) misexpress the mesopallial marker EMX1 (arrowheads). (C, D) BrdU-positive clusters in the mesopallium (M) misexpress DACH2, a marker of nidopallium (arrows). (E–H) BrdU-positive clusters in the mesopallium (F) show a depletion of the glutamatergic marker VGLUT2 (E), misexpress the nidopallial marker DACH2 (G), and fail to express the mesopallial marker EMX1 (H). Arrows indicate corresponding clusters in E to H. H, hyperpallium; St, striatum. Scale bar in D (for A–D) and in H (for E–H) is 500 microns. Gamma was adjusted in all panels.
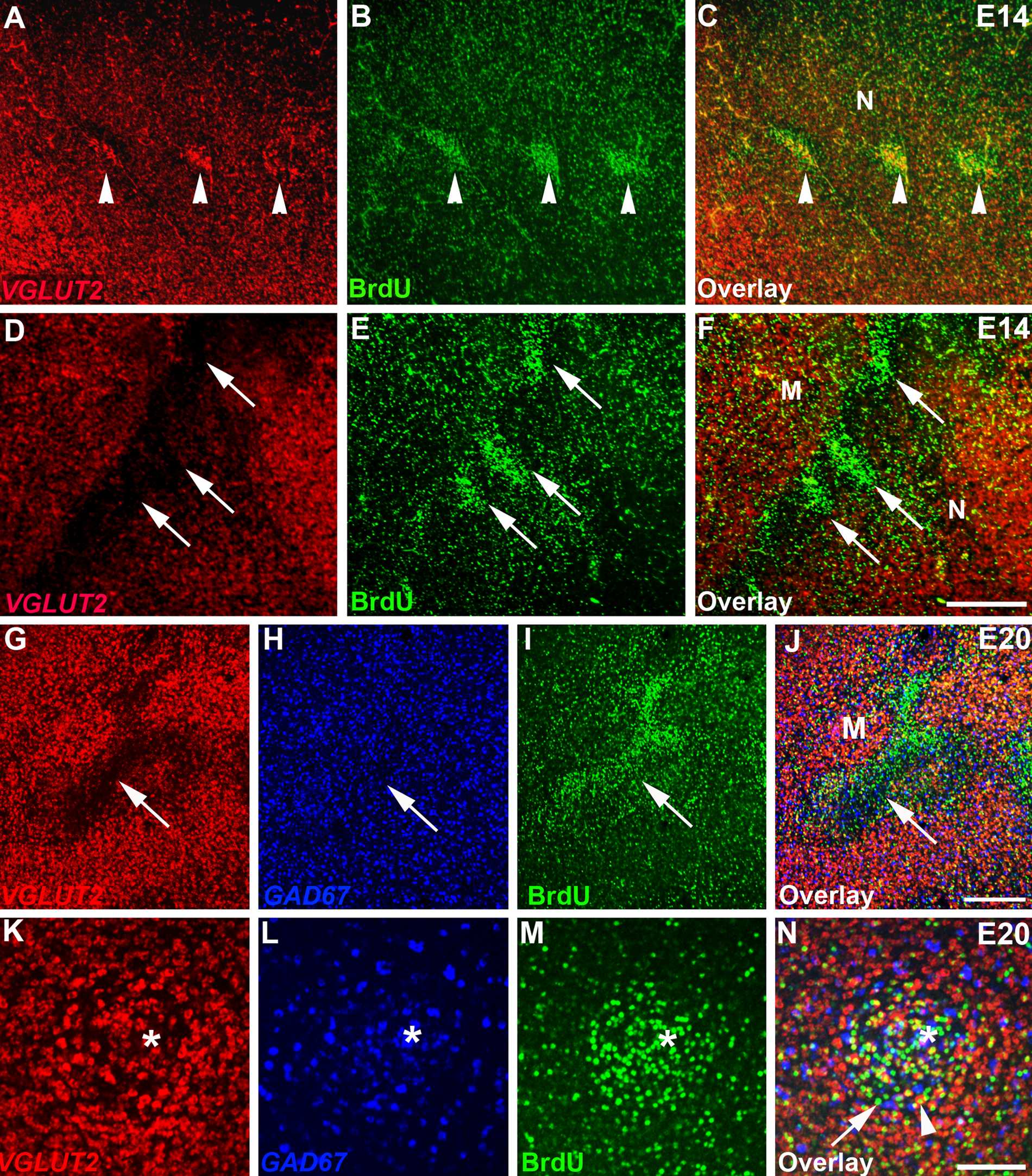
BrdU label incorporation can elicit a range of cell fate errors. (A–N) High-power photomicrographs of E14 (A–F) and E20 (G–N) chick telencephalon after delivery of a routine BrdU dose at E6. (A–F) Some BrdU-positive clusters (green in B, C) are enriched in VGLUT2+ neurons (red in A, C; white arrowheads), whereas others (green in E, F) lack VGLUT2 expression (red in D, F; white arrows). (G–J) Cells in a large BrdU-positive cluster in the mesopallium (green in I, J) are impoverished in the excitatory neuron marker VGLUT2 (red in G, J) and are not enriched in the inhibitory neuron marker GAD67 (blue in H, J). (K–N) Fluorescence detection of VGLUT2 (red in K, N) and GAD67 (blue in L, N) gene expression and BrdU labeling (green in M, N) captured on a confocal microscope. BrdU-positive clusters can contain a range of molecularly identified cells, including some that are VGLUT2−/GAD67− (arrow in N) and others that are VGLUT2+/GAD67− (arrowhead in N). Interspersed within the cluster are BrdU-/GAD67+ cells. Endothelial cell indicated by * in K to N. M, mesopallium; N, nidopallium. Scale bar in F (for A–F) is 500 microns; in J (for G–J), 500 microns; in N (for K–N), 50 microns. Gamma was adjusted in all panels.
Neurogenesis in the chick dorsal telencephalon occurs between E4 and E9 (Tsai et al. 1981a, 1981b), and isochronic cluster formation has been observed with BrdU delivery during peak neurogenesis (E5–E7) (Heyers et al. 2003). By injecting E5 to E9 brains with the routine BrdU dose and collecting brains at E14 for BrdU detection and in situ hybridization, we established that BrdU clusters can be produced from E5 to E8 and are consistently associated with inappropriate EMX1 and DACH2 expression (Fig. 3A–D). We confirmed the observation of Striedter and Keefer (2000) that BrdU isochronic clusters persisted into the third week of embryogenesis and found that the EMX1 and DACH2 misexpression associated with BrdU clusters was evident even in late-stage embryonic brains (E19) that had been injected with the routine dose nearly 2 weeks earlier (on E6; Fig. 3E–H).
Striedter and Keefer (2000) reported that isochronic clusters generated from E6 BrdU delivery were first detected 2 to 3 days later. To determine whether the onsets of cluster formation and gene misexpression coincided, we injected E6 embryos with either routine or threshold doses of BrdU and analyzed the tissue 1 to 4 days postinjection, at E7 to E10. At 1 day postinjection, there was no detectable difference in BrdU labeling between the two doses and, in each case, labeled cells were present in the ventricular zone and in the adjoining mantle layer (Fig. 5A, B). Scattered labeled cells were also detected in the lateral tissue, likely representing vascular endothelial cells. By 3 days postinjection, small aggregates of cells were detected at the migration front in brains injected with the routine dose but not with the threshold dose (Fig. 5C, D). Gaps in gene expression were first evident 4 days postinjection, when the BrdU clusters were more prominent (Fig. 5G, H). Neither the BrdU clusters nor gene misexpression was observed with the threshold dose at 4 days postinjection, as anticipated (Fig. 5E, F). Aside from the isochronic clusters, the overall pattern of the BrdU-labeled wave front was similar for both threshold and routine doses, indicating that the routine BrdU staining pattern was not wholly unreliable and that clusters of later-born cells did not “overshoot” into lateral territories occupied by earlier born neurons (Figs. 1 and 5).
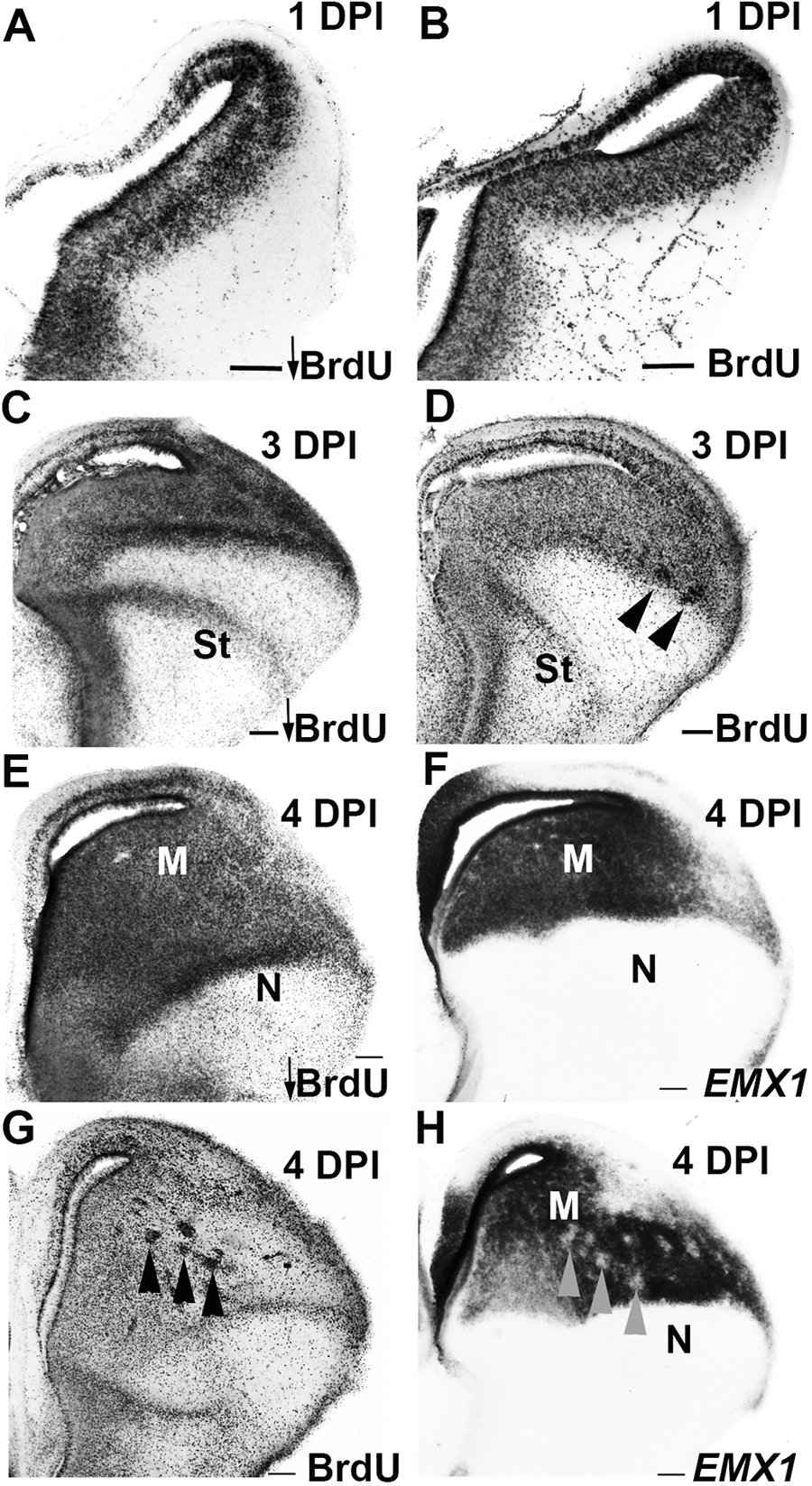
Delayed appearance of isochronic clusters and inappropriate gene expression following delivery of routine BrdU doses on E6. (A, B) BrdU labeling patterns are similar between embryos injected with threshold (A) or routine (B) BrdU doses and collected 1 day after injection (E7). (C, D) Small BrdU-labeled clusters are evident with the routine dose (D, arrowheads) collected at 3 days postinjection (DPI, E9). (E–H) BrdU clusters (G, arrowheads) and gaps in EMX1 gene expression (H, corresponding arrowheads) are observed 4 DPI (E10) after routine (G, H) but not threshold (E, F) BrdU doses. M, mesopallium; N, nidopallium; St, striatum. Scale bars represent 200 microns. Gamma was adjusted in all panels.
Discussion
It has been known for almost 50 years that BrdU is toxic (DiPaolo 1964; Taupin 2007 for review). In rodent embryos, BrdU can cause exencephaly, cleft palate, limb deformities, and behavioral abnormalities (Ruffolo and Ferm 1965; Skalko et al. 1971; Bannigan 1985; Bannigan et al. 1990; Kuwagata and Nagao 1998; Kolb et al. 1999; Sekerkova et al. 2004; Breunig et al. 2007). In chicken embryos, BrdU injection can result in developmental delay, growth retardation, increased mortality, and the appearance of ventral body wall defects (Lee et al. 1974; Bannigan et al. 1981; Gould et al. 1999). Incorporation of BrdU, a halopyrimidine, into DNA can produce base pairing of the bromouracil with guanine instead of adenine, so it is perhaps not surprising that frank genetic problems, including mutations and breaks in double-stranded DNA, can be caused by BrdU (Hsu and Somers 1961; Roy-Burman 1970; Bannigan and Langman 1979; Saffhill and Ockey 1985; Morris 1991; Morris et al. 1992; Duque and Rakic 2011).
Researchers have made substantial efforts to mitigate the toxicity of BrdU. Gould and colleagues (1999) addressed BrdU toxicity in chicken embryos by incrementally decreasing a cytotoxic dose of BrdU until the injected embryos survived, no gross morphological defects were observed, and neuronal numbers in the studied motor neuron pools were normal. Striedter and Keefer’s (2000) strategy was to reduce the BrdU dose to the point where BrdU incorporation was limited to a 24-hr window, thereby establishing a BrdU “pulse” labeling protocol for avian brains. Heyers and colleagues (2003) specifically addressed whether the chick dorsal telencephalon isochronic clusters were a product of BrdU toxicity, and they concluded this was not the case because the isochronic clusters lined up with heterogeneities in cadherin expression reminiscent of those seen in uninjected embryos. We confirmed many features of isochronic cluster formation in chick dorsal telencephalon previously reported, including that the BrdU-induced clusters survive at least until late in embryogenesis. Indeed, the reproducibility of isochronic cluster formation in the chick dorsal telencephalon appeared to establish it as an authentic developmental process.
The data presented in this report demonstrate conclusively that the isochronic clusters are a BrdU-induced malformation. Clusters were not produced with threshold doses (Fig. 1), and the clusters elicited with routine doses were associated with misexpression of mesopallial marker EMX1 and nidopallial marker DACH2 (Figs. 2–5), indicating that BrdU, even at the relatively low routine doses, was capable of altering cell fates. This finding demonstrates that it is insufficient to look only at the survival of BrdU-labeled cells to determine whether BrdU is exerting a toxic effect on brain development. It is critical to ensure that gene expression patterns in BrdU-treated tissue are normal. Because the toxic effects of BrdU are dose dependent, we recommend as a precaution employing as low a BrdU dose as can be detected, as was done with the threshold dose in this study.
Adverse effects of BrdU can be challenging to detect. Studies in the rodent central nervous system addressing this point directly have suggested that results obtained with [3H]dT and BrdU labeling are similar (Miller and Nowakowski 1988; del Rio and Soriano 1989). Recently, however, Duque and Rakic (2011) revisited this issue in the macaque monkey neocortex, a large structure with a long generation time. They concluded that BrdU labeling leads to more cell death than does [3H]dT labeling and that migration patterns differed between the two labeling methods. To reach these conclusions, however, the authors needed extensive quantitative analyses.
The manner in which the central regions of the chick dorsal telencephalon develop may facilitate detection of the BrdU-induced cell fate errors. Isochronic clusters were observed in the mesopallium and nidopallium but not the hyperpallium (Figs. 1 and 3; Striedter and Keefer, 2000). The hyperpallium develops in a gradual “throughout” pattern across a relatively long time window (E4–E8). By contrast, cell populations in the mesopallium and nidopallium are produced in a tight “outside-in” pattern over a short period of time, with some nuclei produced in a single day (Tsai et al. 1981a, 1981b). It is possible that this pattern of neurogenesis, where large numbers of neurons are produced and migrate out en masse (Fig. 5), promotes the formation of isochronic clusters. The BrdU-labeled hyperpallial cells may also change fate, but because these misspecified cells do not cluster, quantitative analyses such as those performed by Duque and Rakic (2011) would likely be necessary to detect hyperpallial BrdU-induced cell fate errors.
We did not observe BrdU cluster formation until 3 days postinjection, well after cell cycle exit and migration from the ventricular zone (Fig. 5; Striedter and Keefer 2000), suggesting that isochronic cluster production was the result of cell fate misspecification followed by cell migration defects. Cell fate errors producing alterations in cell affinities provide a potential mechanism through which, in this system, misspecified neurons aggregate together to form the isochronic clusters. In support of this model, Heyers et al. (2003) found that the BrdU isochronic clusters express different levels of cadherins from the surrounding tissue. Differences in cadherin expression levels are known to be sufficient to make populations of cells immiscible with each other (Steinberg and Takeichi 1994).
Given the toxicity of BrdU and the cell fate errors it can produce, it is critical to develop less toxic techniques for studying developmental timing. The approach we describe here is to reduce the BrdU dose to “just detectable” levels. It is also possible that alternative thymidine analogues may be less toxic than BrdU and could be delivered at high doses. Other halopyrimidines such as IdU and CldU have been employed to study neuronal birth dating (Bauer and Patterson 2005; Burns and Kuan 2005; Thomas et al. 2007), but these molecules have similar chemical structures to BrdU and are likely to prove as deleterious. EdU (5-ethynyl-2′-deoxyuridine), a thymidine analogue with an alkynyl substitution that can be detected with “click” chemistry (Salic and Mitchison 2008), is a possible alternative to halopyrimidines. It is unknown, however, whether EdU also exerts toxic effects on developing cells. The development of the chick dorsal telencephalon, where BrdU-induced errors can be readily identified, appears a particularly attractive system to test the labeling toxicities of future candidate birth dating analogues.
Footnotes
Acknowledgements
We thank Wujun Ke for technical assistance and Elizabeth Grove for comments on the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by a grant from the National Eye Institute/National Institutes of Health.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
