Abstract
Two soft tissue masses from different locations in 2 dogs were submitted for histopathologic examination. Each was well demarcated and consisted of interweaving streams of bland spindle cells among which numerous plasma cells and lymphocytes were scattered. All the spindle cells reacted strongly to antibodies against vimentin and calponin, whereas a subset of the spindle cells expressed smooth muscle actin and desmin. Immunohistochemistry results were consistent with a myofibroblastic derivation for the spindle-cell population and the diagnosis of inflammatory myofibroblastic tumor (IMT) was made. This is the second report of IMT in the veterinary literature.
Inflammatory myofibroblastic tumors (IMT) are discrete masses composed of a mixture of bland fusiform myofibroblastic cells and an inflammatory infiltrate composed of varying proportions of lymphocytes, plasma cells, and histiocytes. They are well described in humans and occur most commonly in the lungs of children and young adults, although there are numerous examples of extrapulmonary IMT. 3, 4, 8 IMTs are regarded as pseudoneoplasms and are generally accepted to represent an aberrant or exaggerated inflammatory response to some unidentified tissue injury. 3 Despite their appearance, IMT typically have a benign clinical course in humans although, as these tumors are more commonly recognized, there are more reports of aggressive or malignant behavior. 4, 8 A confusing array of synonyms for this tumor exists in the human literature, including plasma cell granuloma, inflammatory pseudotumor, inflammatory fibrosarcoma, and myofibrohistiocytic proliferation. 5 In this report, we describe histologic and immunohistochemical characteristics of 2 inflammatory myofibroblastic tumors removed from different locations (Table 1) in 2 dogs.
Signalment, tumor location, immunohistochemistry results for proliferating spindle cells, and clinical outcome for 2 dogs with inflammatory myofibroblastic tumors.
In each dog, the excised masses were fixed in 10% neutral buffered formalin and routinely prepared for histologic examination. Sections for immunohistochemistry were prepared according to a routine protocol. Using an autostainer, slides were incubated with antibody against vimentin (1 : 80 dilution; mouse monoclonal), calponin (1 : 100; rabbit polyclonal), smooth muscle actin (SMA) (1 : 30; mouse monoclonal), desmin (1 : 20; mouse monoclonal), pan-cytokeratin (AE1/AE3; 1 : 200; mouse monoclonal), glial fibrillary acidic protein (GFAP) (1 : 3,000; rabbit polyclonal), S-100 protein (1 : 200; rabbit polyclonal), Melan A (1 : 50; mouse monoclonal), Mac 387 (1 : 300, mouse monoclonal), BLA.36 (1 : 100, mouse monoclonal), and CD18 (1 : 50, mouse monoclonal). All antibodies were previously titrated for optimum reactivity. A streptavidin/immunoperoxidase staining procedure was used for immunolabeling. The immunoreaction was visualized with a 3,3′-diaminobenzadine substrate. Sections were counterstained with Mayer's hematoxylin. Negative controls were performed by substituting the primary antibody with an isotype-matched irrelevant mouse monoclonal antibody or normal (nonimmune) rabbit serum. All reagents except antibody to CD18 were made by Dakocytomation (Carpinteria, CA, USA); CD18 antibody was obtained from PF Moore (University of California, Davis, CA, USA).
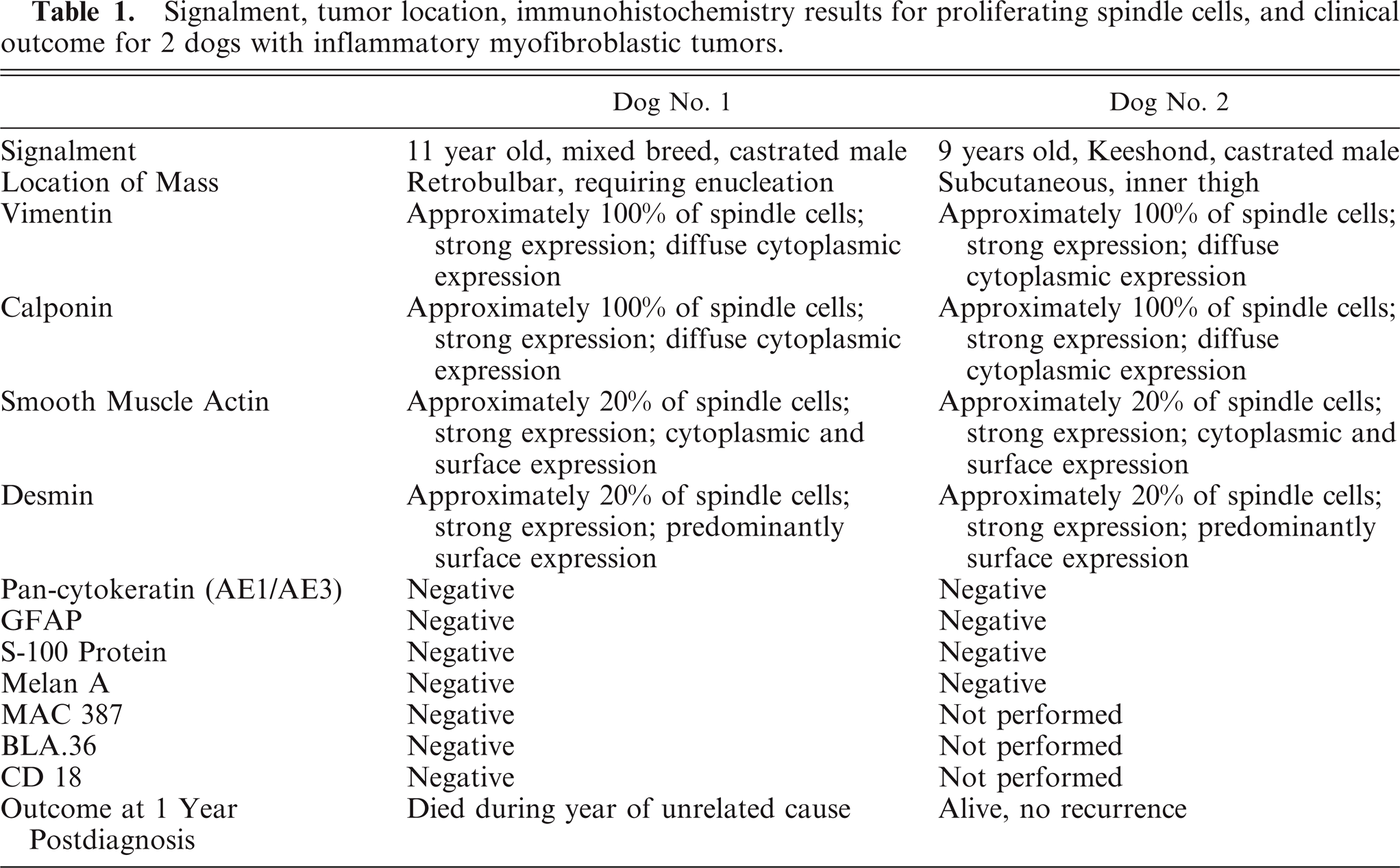
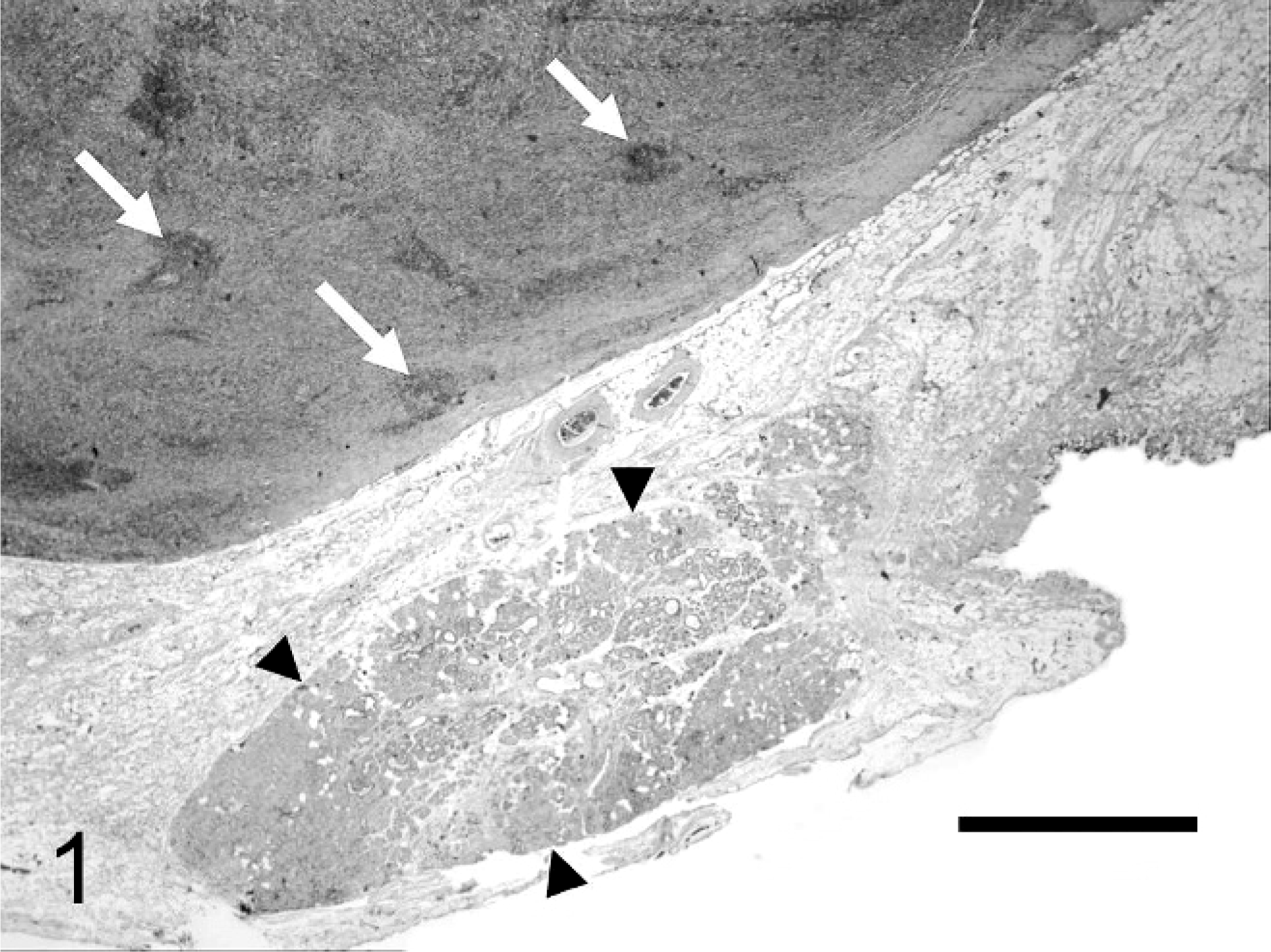
The mass in dog No. 1, an 11-year-old mixed-breed castrated male, was retrobulbar, visible on ultrasound and computed tomography, and caudoventral to the globe, requiring enucleation for removal. An incidental anterior uveal melanocytoma was present in this same eye, but all other ocular structures, including the optic nerve, were normal. The retrobulbar mass measured approximately 20 × 20 × 10 mm, was sharply demarcated but unencapsulated, expansile, and surrounded by a thin rim of compressed adipose tissue, lacrimal glandular tissue, and skeletal muscle (Fig. 1). Histologically the mass consisted of elongated spindle cells arranged in dense, haphazardly whorling or interwoven streams, among which were scattered numerous plasma cells and numerous lymphoid follicles, many with germinal centers (Fig. 2). Tumor cells had moderate to large amount of very faintly fibrillar, eosinophilic cytoplasm. Nuclei were large with mild anisokaryosis and ovoid to round with finely stippled to granular chromatin and 1 to 4 inconspicuous nucleoli. Mitotic figures were not observed. All spindle cells reacted strongly to antibodies against vimentin and calponin; approximately 20% of the spindle cells also strongly expressed SMA and desmin. Cells did not react with antibodies against pan-cytokeratin (AE1/AE3), GFAP, S-100 protein, or Melan A (Table 1). Small numbers of MAC 387-positive cells were randomly dispersed among the spindle cells but were excluded from the nodular aggregates of lymphocytes, compatible with tumor-infiltrating macrophages and granulocytes. Small numbers of CD18-positive cells with prominent cytoplasmic extensions were clustered primarily around vessels. A similar pattern was detected with BLA.36, which also labeled B-cells. Although double-label immunohistochemistry was not performed, the cellular morphology and similar distribution of CD18 and BLA.36 positive cells supported the conclusion that the mass was infiltrated by small numbers of dendritic cells that preferentially localized in perivascular spaces.
Inflammatory myofibroblastic tumor; 11-year-old male dog; dog No. 1. Well-demarcated, expansile mass that contains lymphocytic foci (arrows) and compresses adjacent structures, including lacrimal glandular tissue (arrowheads). HE. Bar = 1.5 mm.
Inflammatory myofibroblastic tumor; 11-year-old male dog; dog No. 1. Numerous plasma cells and lymphoid aggregates are scattered among streams of bland spindle cells. HE. Bar = 50 µm.
The mass in dog No. 2, a 9-year-old castrated male Keeshond, was in the subcutis of the medial aspect of the thigh, near the inguinal region. This mass measured 20 × 20 × 20 mm and, histologically, was virtually identical to the mass described in dog No. 1. Immunohistochemistry results (Table 1) were also identical to those in dog No. 1. (Immunohistochemistry for CD18, MAC 387, and BLA.36 was not performed in this case.)
Based on gross and histologic appearance, immunohistochemistry results, and similarity to previously reported cases, both masses were diagnosed as inflammatory myofibroblastic tumors. In each case the mass was removed without recurrence. The outcome for each dog is provided in Table 1.
Reports of inflammatory myofibroblastic tumors in the veterinary literature are extremely rare, and, to the best of our knowledge, only a single case has been described and given this designation (in a brush-tailed porcupine). 10 Several reports exist, however, of animal neoplasms with similar histologic features but alternate names. A splenic mass with marked histologic similarity to the 2 masses described in this article was reported in a dog; this mass was termed inflammatory pseudotumor by the authors. 6 Two reports exist of peritoneal masses in horses in which tumor cells were confirmed to be of myofibroblastic origin, but neither mass had a significant inflammatory component, and each was designated simply as a myofibroblastic tumor. 2, 14 Immunohistochemistry was performed on all but 1 of these masses (1 equine tumor was not examined), and each shared similar immunohistochemical characteristics with the 2 masses described in this article, namely 1) strong vimentin expression by all spindle cells; 2) strong expression of SMA in some spindle cells; 3) variable expression of desmin; and 4) no expression of cytokeratin or S-100 protein. Other antigens, not tested for in each case, included calponin, NSE, GFAP, CD3, CD31, CD34, and factor VIII–related antigen. There was no expression by the spindle-cell population of any of these antigens except for calponin, which was intensely expressed by the spindle-cell population of the 2 canine masses described in this report.
Other masses designated as pseudotumors have also been reported in several species. In cats they are most often reported as sclerosing orbital pseudotumors that consist of inflammatory foci (predominantly lymphoplasmacytic) within a desmoplastic stroma. 1, 12, 16 These are generally progressive and infiltrate between extraocular structures, encapsulating muscles, and orbital fat and cause scleral thickening. Involvement of the contralateral eye is common. In the largest study to date on sclerosing orbital pseudotumors of cats, immunohistochemistry was not performed, but trichrome staining confirmed the collagenous nature of the infiltrative tissue, and the authors proposed that a primary fibrosing disorder be considered as the cause. 1 In a separate report, a mass in the paw of a cat was designated as an inflammatory pseudotumor. 11 In this cat, an ulcerated and fistulous spindle-cell proliferation in the hind foot contained neutrophils and histiocytes and was associated with intralesional acid-fast bacteria (cutaneous mycobacteriosis). A mass designated as an orbital pseudotumor was described in a lesser bushbaby. 9 This mass developed within the bulbar conjunctiva at the limbus, was unencapsulated but easily separated from adjacent tissue, and consisted of a mixture of fibroblasts, plasma cells, lymphocytes, and scattered neutrophils. A similar mass, designated as a lacrimal pseudotumor, was reported in a young Bull Terrier. 19 The mass arose from the lower conjunctival sac and was composed of granulation tissue with abundant collagen deposition and a mixed perivascular infiltrate of lymphocytes, plasma cells, mast cells, neutrophils, and eosinophils. Of these pseudotumors, none was consistent histologically with the IMT described in this report, and immunohistochemistry was performed in only 1 case (the hind foot of the cat with cutaneous mycobacteriosis). We believe that these reports do not describe IMT and that the term pseudotumor in these cases is used in a more generic way to designate a reactive spindle-cell mass bearing resemblance to a neoplasm rather than as a synonym for IMT.
The term equine conjunctival pseudotumor is commonly used for smooth, nonulcerated conjunctival masses resembling neoplasms clinically but which are found to be inflammatory nodules on histologic examination. 13 These are suspected to have an immune-mediated pathogenesis and share some similarities with so-called nodular granulomatous episcleritis (NGE) of dogs. 17 NGE is a nonneoplastic disease primarily affecting the canine limbus and sclera. Its various names (including ocular nodular fasciitis, fibrous histiocytoma, and proliferative keratoconjunctivitis) reflect its variable clinical presentation, but it generally behaves as would a locally infiltrative neoplasm and consists of a proliferative, nonencapsulated mixture of histiocytic cells, spindle cells, and mononuclear leukocytes. 18 We believe that the location, lack of infiltrative behavior, and paucity of histiocytic cells (as demonstrated by immunohistochemistry) within the masses described in this report allow a diagnosis of NGE/ocular nodular fasciitis to be ruled out.
With the scarcity of reports of this tumor in the veterinary literature, uncertainty in nomenclature, and definitive diagnosis requiring immunohistochemistry, it is possible that this disease is underdiagnosed or reported under different names. At this point, the biologic behavior in animals cannot be predicted, particularly given the range of species affected and the different presentation in each species. Masses in both dogs in this report were discrete, focal, relatively small, and removed without recurrence, whereas the peritoneal IMT in the brush-tailed porcupine was disseminated within the peritoneal cavity.
In summary, we believe that masses corresponding to human inflammatory myofibroblastic tumors exist in animals and will be diagnosed with increasing frequency as more unclassified spindle-cell masses are examined immunohistochemically. This diagnosis should be considered for any spindle-cell mass with the particular set of immunohistochemical characteristics described above and a prominent inflammatory infiltrate.
The status of IMT in human medicine is still controversial and under investigation. Originally accepted as a benign pulmonary pseudoneoplasm, the frequent diagnosis of IMT in extrapulmonary sites and occasional reports of aggressive behavior, including local infiltration, recurrence after excision, multifocality, vascular invasion, and malignant transformation, have led to the belief that these may be true neoplasms. 4, 5 This possibility is supported by studies showing that these tumors have clonal characteristics. 7, 15 Whether these findings will also apply to IMT in animals requires recognition of more of these tumors, followed by immunohistochemical confirmation of diagnosis, clonality studies, and long-term monitoring of patients.
