Abstract
A 13-year-old, spayed, female Australian Cattle Dog had at least a 10-year history of numerous subcutaneous nodules for which fine-needle aspiration and cytologic evaluation were nondiagnostic. Abdominal ultrasound 3.5 months before necropsy detected a small left kidney but no cysts or neoplasms. At gross necropsy, innumerable, firm, round to oval, white, 0.25 to 2 cm masses were detected throughout the subcutaneous tissues of the axial and appendicular skeleton, epimysium of numerous muscles, and parietal peritoneum of the lateral abdominal body wall. The left kidney was approximately half the size of the right, and there was severe bilateral renal medullary (papillary) necrosis. Histologically, the subcutaneous nodules were well-demarcated masses of mature, hypocellular collagen that were consistent with previous reports of nodular dermatofibrosis and renal cystadenomas or cystadenocarcinomas. In addition to diffuse acute medullary necrosis, both kidneys were affected by severe chronic lymphoplasmacytic interstitial nephritis. This is the first known report of nodular dermatofibrosis in a dog without renal cysts, cystadenoma, or cystadenocarcinoma.
A 6-year-old spayed female Australian Cattle Dog was presented to the Colorado State University Veterinary Teaching Hospital (CSU VTH) with a 3-year history of generalized nonpruritic and nonalopecic subcutaneous nodules mainly located on the head and legs. Cytologic evaluation of fine-needle aspirates of numerous nodules was nondiagnostic. Seven years later (January 2007) the dog was presented again to the CSU VTH with a chief complaint of urinary incontinence. At that time the number of subcutaneous nodules had significantly increased. Cytologic evaluation of fine-needle aspirates of 14 nodules was again nondiagnostic. Abdominal ultrasound revealed that the left kidney was slightly smaller than the right, but no renal cysts or neoplasms were detected. The only abnormal urinalysis result was isosthenuria (specific gravity, 1.011); urine culture yielded no organisms. Serum creatinine concentration was mildly elevated; blood urea nitrogen (BUN) was within the reference range. Three months later, the dog was returned to the CSU VTH for persistent incontinence. The dog was azotemic, had a serum BUN concentration of 65 mg/dl (reference range, 7–32 mg/dl) and a creatinine concentration of 3.5 mg/dl (0.4–1.5 mg/dl), and remained isosthenuric (specific gravity, 1.011). An additional abnormality detected at this visit was systemic hypertension with a mean arterial pressure of 180 mm Hg. The dog was euthanatized 2 months later when it became anorexic with frequent vomiting and elevated concentrations of creatinine (8.1 mg/dl) and BUN (63 mg/dl).
At necropsy, innumerable, firm, spherical to oval, white, 0.25 to 2 cm (diameter) masses were found to be scattered throughout the subcutis of the axial and appendicular skeleton, epimysium of numerous muscles, and peritoneum of the lateral abdominal body wall (Figs. 1, 2). The left kidney was approximately half the size of the right, and there was severe bilateral renal medullary necrosis (Fig. 3). Other lesions included mild left and right atrioventricular myxomatous valvular degeneration (endocardiosis) and mild to moderate bilateral thyroid gland atrophy. Histologically, the nodules were well-demarcated collections of mature, hypocellular collagen (Figs. 4–6) that were consistent with previous reports of nodular dermatofibrosis and renal cystadenomas or cystadenocarcinomas (NDRC). 1, 12, 19, 25 Renal medullary necrosis was acute but accompanied by severe chronic lymphoplasmacytic interstitial nephritis with fibrosis. A few dilated tubules (without formation of macroscopic cysts) were present, but there were no dysplastic or neoplastic changes within the tubular epithelium. This is the first known report of generalized nodular dermatofibrosis without concomitant renal cysts, cystadenomas, or cystadenocarcinomas.
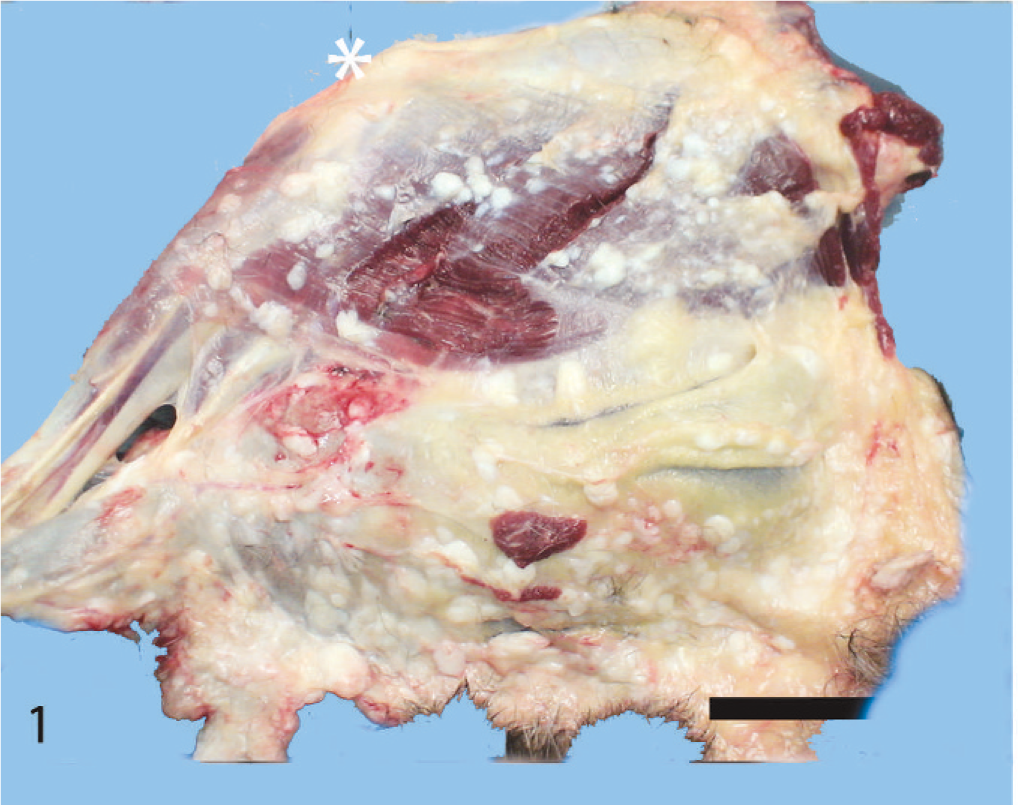
Left hind leg, lateral view with the skin reflected; dog. ∗ = stifle joint. Note the numerous variably sized white nodules within the subcutis and associated with the epimysium of the muscles. Bar = 4 cm.
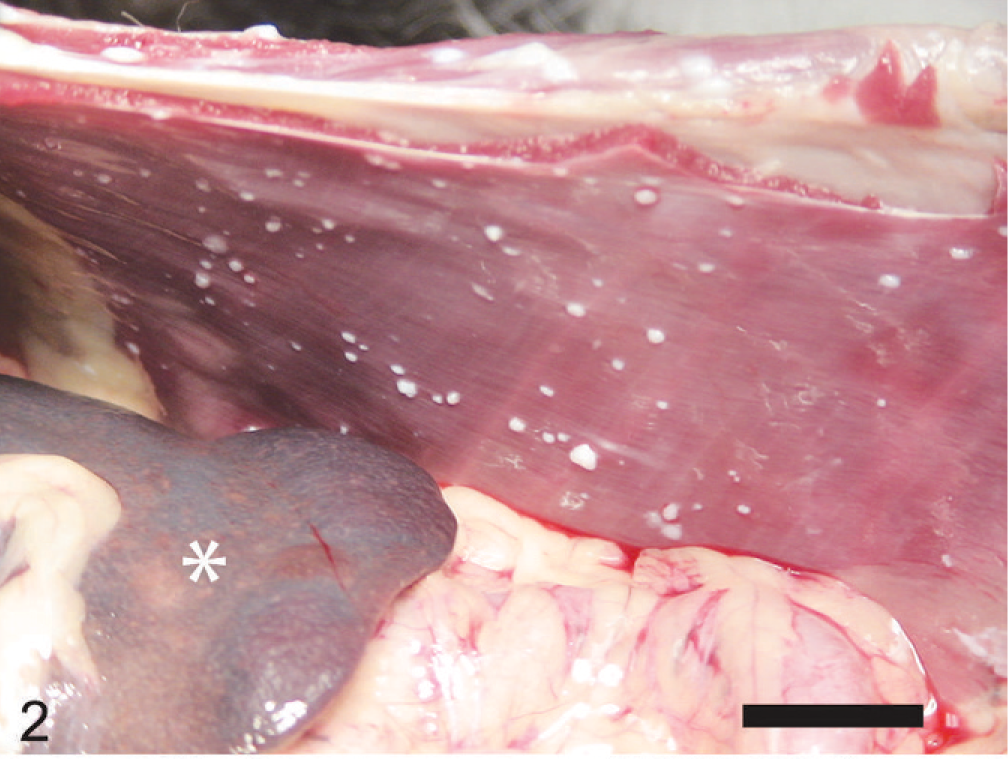
Peritoneal cavity and right body wall; dog. ∗ = body of the spleen. Note the numerous variably sized white nodules on the parietal peritoneum. Bar = 2.5 cm.
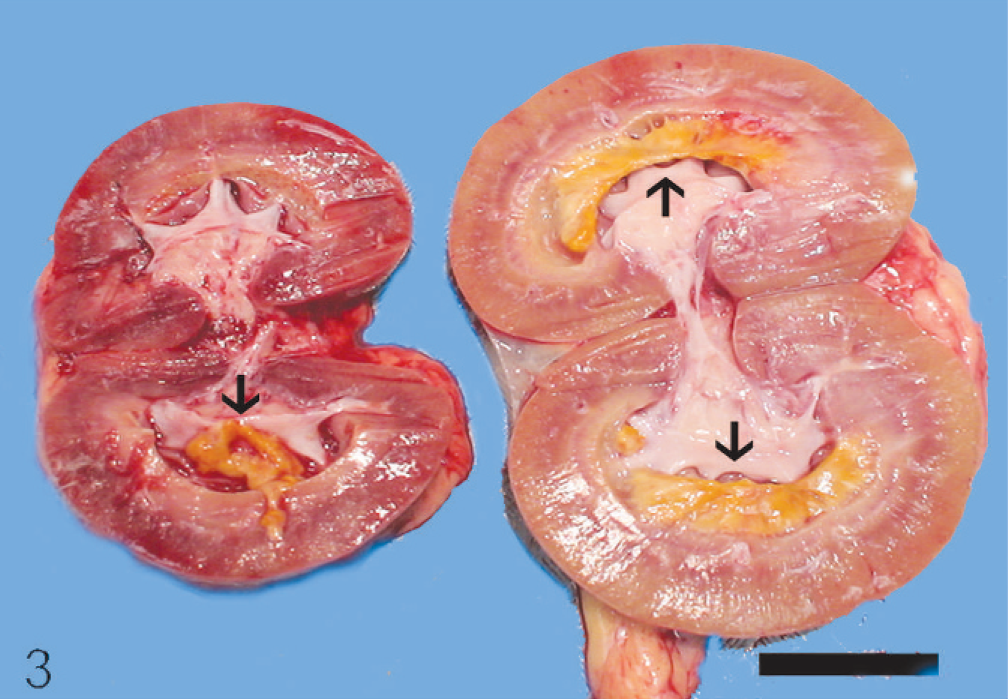
Left and right kidneys sagittally sectioned; dog. Necrotic medullary tissue (black arrows) is intact in the right kidney, but sloughed into the pelvis of the left kidney. Note lack of cysts or neoplastic nodules. Bar = 2.5 cm.
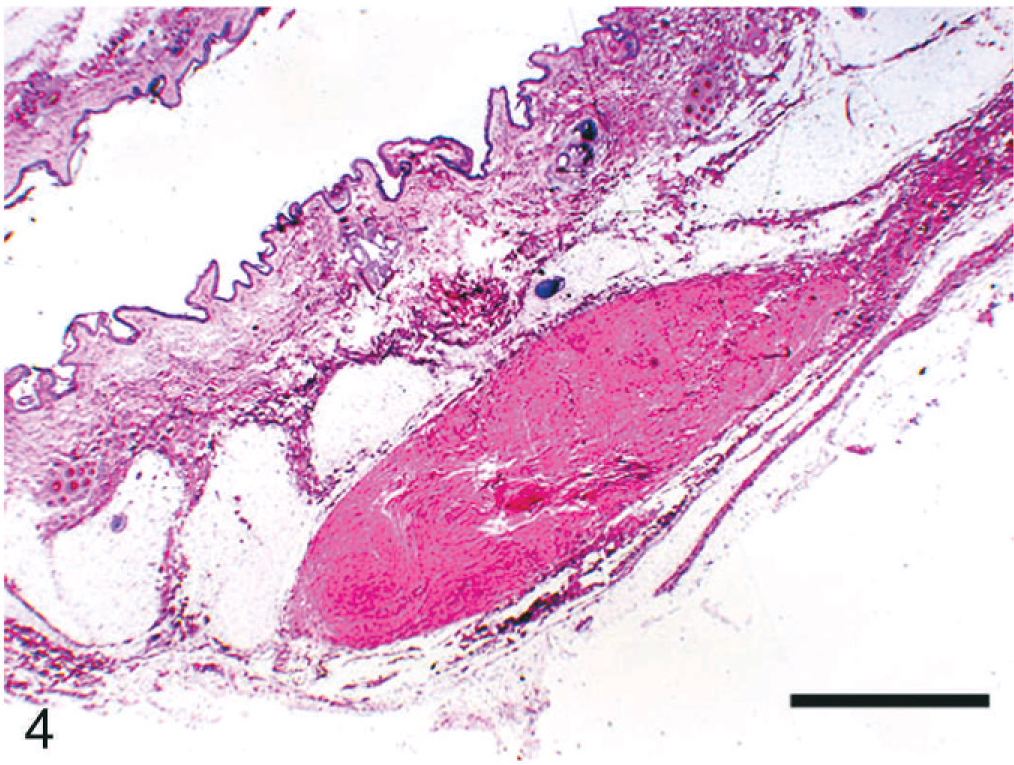
Skin; dog. Note the well-demarcated, hypocellular collection of mature collagen in the subcutaneous adipose tissue. HE. Bar = 2 mm.
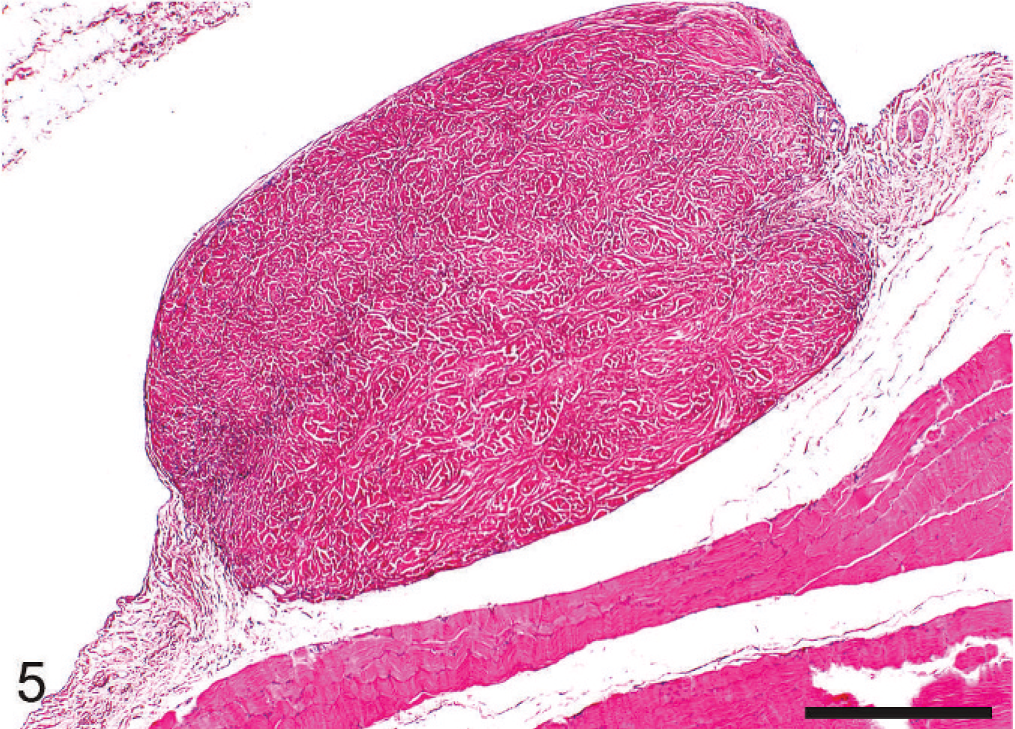
Cutaneous trunci muscle; dog. A mass similar to that in Fig. 4 arises from the epimysium. HE. Bar = 500 µm.
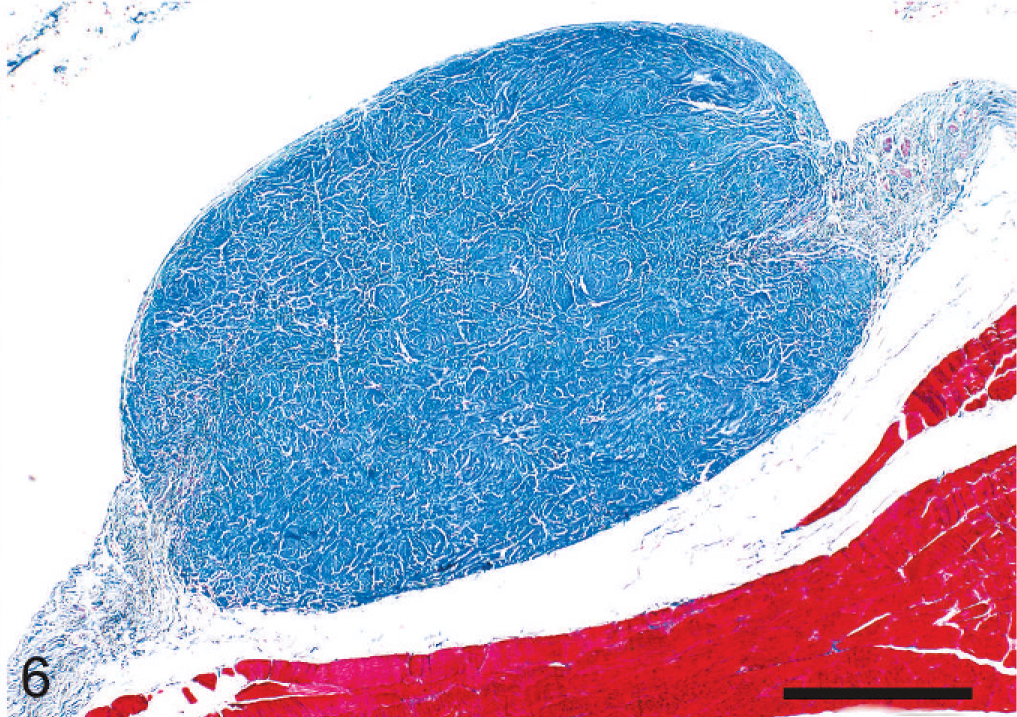
Cutaneous trunci muscle; dog. Same nodule as in Fig. 5. Note differential staining of collagen (blue) and skeletal muscle (red). Masson's trichrome. Bar = 500 µm.
Generalized nodular dermatofibrosis is typically seen in German Shepherd Dogs in association with renal cysts, cystadenomas, or cystadenocarcinomas. Diagnoses of surgical biopsy samples from the dermal and subcutaneous masses include collagenous hamartoma, focal dermal fibrosis, focal dermal scarring, reactive fibrosis, and fibroma. 1, 4, 6, 13, 17, 22 Affected dogs typically are presented first for the presence of numerous cutaneous and subcutaneous masses that are often concentrated on the extremities or head. Masses vary in size and shape but typically are nonpruritic and nonpainful on palpation; they may be associated with ulceration, hyperpigmentation, a pitted or dimpled surface (peau d'orange), and alopecia. 1, 6, 12, 13, 15, 17, 19, 22 Nodular dermatofibrosis has been described histologically in the dermis, subcutis, and fascial planes of various muscles. 1, 4, 6, 12, 13, 17, 19, 22 Polyps within the small intestine, fibrotic nodules in the tongue, and tinctorial changes to the adventitia of the bladder have also been described in cases of NDRC. 3, 4, 15 The connective tissue masses can be ill defined or well demarcated. 1, 4, 6, 12, 13, 19 In this case the distribution of dermatofibrosis was overwhelmingly subcutaneous and most masses were well demarcated.
Upon presentation for the cutaneous masses most dogs with NDRC have renal clinical pathologic, radiographic, or ultrasonographic abnormalities. A few dogs have no evidence of renal neoplasia at presentation for the cutaneous masses but invariably develop renal neoplasia within months to years. 15, 25 NDRC is a disease almost exclusive to German Shepherd Dogs, although there have been reports of the disease in a Golden Retriever, a Boxer, a German Shorthaired Pointer, and 3 mixed-breed dogs. 7, 13, 25 This is the first known report of a similar cutaneous syndrome in an Australian Cattle Dog.
Results of a study of 43 German Shepherd Dogs with NDRC suggested that renal lesions occur as a continuum from hyperplasia to adenoma to adenocarcinoma. 12 Renal lesions thought to be preneoplastic have been detected in two 1-year-old dogs from an NDRC-affected dam. 14 The fact that the dog in this report had had severe dermatofibrosis for at least 10 years without developing renal neoplasia suggests that more than one syndrome of generalized dermatofibrosis may exist in dogs or that dermatofibrosis in NDRC can persist for many years without development of renal neoplasia or cysts. A number of human diseases include connective tissue masses of the skin in addition to lesions in other organ systems—Shagreen patches of tuberous sclerosis complex, dermatofibrosis lenticularis disseminata (Buschke-Ollendorff syndrome), Cowden syndrome (PTEN hamartoma tumor syndrome), and familial cutaneous collagenoma. 5, 8, 10, 18 None of these human diseases perfectly matches the disease detected in this dog, and dermatofibrosis in canids of the extent seen in this case has only been reported in NDRC.
NDRC in the German Shepherd Dog is probably inherited in an autosomal dominant manner. 10, 12 The putative genetic defect has been located on exon 7 of a gene on chromosome 5. 9, 11 The mutation is thought to have a homozygous lethal effect. 11 No information regarding parents or siblings of this dog was available. The disease-associated canine gene has a human orthologue, the Burt-Hogg-Dubé (BHD) gene. 11 Germline mutations in the BHD gene are associated with the Burt-Hogg-Dubé syndrome (BHDS) in humans. 16 The human disease is phenotypically similar to NDRC in dogs and is characterized by numerous skin tumors (fibrofolliculomas) of the head, renal neoplasms, lung cysts, and pneumothorax. 20 The gene product is a protein termed “folliculin” (the gene is also referred to as the folliculin gene or FCLN), which is of unknown function but a tumor suppressor role is suspected. 23 Supportive of a tumor suppressor role, somatic second-hit mutations or loss of heterozygosity (LOH) were detected in 70% of the wild-type BHD alleles in 77 human renal tumors from 12 patients with BHDS. 23 In humans, BHD mRNA is expressed in a variety of tissues, including fibrofolliculomas, but not in renal tumors from patients with BHDS. 24 The development of fibrofolliculomas without detectable LOH therein is thought to be due to haploinsufficiency at the BHD locus. 21 Mutational analysis of NDRC-associated canine renal tumors revealed somatic second-hit mutations of predicted functional significance in 12 of 17 (71%) tumors from 2 NDRC-affected dogs, but no mutations were identified in skin nodules from the same dogs. 2 Similar to the disease in humans, the cutaneous nodules in dogs with NDRC may be due to haploinsufficiency. 2
In accordance with these findings, the nodules in the case described herein could be due to haploinsufficiency at the BHD locus. The lack of renal neoplasia may reflect the absence of a functionally significant second-hit mutation in the BHD gene within the kidneys. Genetic and molecular studies would have been helpful to explore these possibilities but were not done in this case.
Causes for the renal medullary necrosis remain undetermined. In the absence of known nonsteroidal anti-inflammatory administration, which would be more likely to affect both kidneys, either fibrosis leading to reduced perfusion or hypertensive vasculopathy was considered. However, no microscopic lesions were identified consistent with hypertensive vasculopathy. This case indicates that generalized nodular dermatofibrosis can occur in dogs in the absence of renal neoplasia, or at least it may exist for years without development of renal neoplasia, or more than one syndrome of generalized nodular dermatofibrosis may exist in the dog.
