Abstract
Two cases of patent ductus arteriosus are described in 2 Murrah buffalos, a 7.5-month-old heifer calf and a 5-month-old bull calf. The main clinical signs consisted of exercise intolerance, dyspnea, and tachycardia. At necropsy, lesions were consistent with congestive heart failure as indicated by dependent subcutaneous edema, body cavity effusions, and nutmeg liver. The lungs were extensively atelectatic. The heart was enlarged and had a globous shape. The ductus arteriosus, measuring 0.8 cm in length and 0.4 cm in caliber, was patent. The proximal pulmonary artery had an approximately 50-mm-long aneurysm. Eccentric cardiac hypertrophy was evident primarily in the right but to a lesser degree also in the left ventricle. Additionally, there was diffuse subendocardial fibrosis in the left and right ventricle. To the best of the authors' knowledge, patent ductus arteriosus was not previously described in buffalos. Since both calves were sired by the same bull, it is possible that this cardiac anomaly has a hereditary basis.
Keywords
The ductus arteriosus is a normal fetal structure that allows pulmonary arterial blood to bypass the unexpanded lungs and enter the descending aorta for oxygenation in the placenta. Shunting of blood from the venous to the arterial side is a consequence of the high vascular resistance of the fetal pulmonary bed. At birth, the increase in oxygen tension leads to inhibition of local prostaglandins and causes functional closure of the duct, followed by its anatomic closure during the first weeks of life. If the duct fails to close, blood shunts from the descending aorta to the pulmonary artery. 1, 9 This report describes two cases of patent ductus arteriosus (PDA) in Murrah buffalos (Bubalus bubalis bubalis).
The 2 affected buffalos belonged to a herd that was kept in pasture and received supplemental feed as necessary. Most females were artificially inseminated. The first case (buffalo No. 1) was a 7.5-month-old heifer calf, with a 20% lower weight gain than the average within the herd. As the calf grew older it got remarkably tired when exercised. During the last couple of weeks of its life, an increase in respiratory rate, tachycardia, and heart murmur could be observed whenever the animal moved around. At physical examination, the abdomen was distended. The animal was found dead a few days later. The second case (buffalo No. 2) was a bull calf from a different dam but the same sire as the first one. It was born 12.5 months after buffalo No. 1. Buffalo No. 2 died at 5 months of age after developing clinical signs similar to the ones described for buffalo No. 1. A complete necropsy of both calves was performed. Specimens of various tissues were collected and fixed in neutral buffered formaldehyde (10% formalin), embedded in paraffin, sectioned, and stained with hematoxylin and eosin (HE).
The gross findings were similar in both animals. The carcasses were in good nutritional condition. There was diffuse subcutaneous edema, particularly of the lower parts of the body. The thoracic cavity contained about 5 liters of yellow fluid (marked hydrothorax). The trachea and bronchi were filled by moderate amount of foamy fluid. The ventral portions of the apical lobes and most of the caudal lobes of the lungs were atelectatic. The pericardial sac was dilated and contained 60 ml of clear fluid. The heart was enlarged and had a globous shape (Fig. 1). The right atrium was dilated. The right ventricle was eccentrically hypertrophied as indicated by dilation of the chamber and mild thinning of the wall, despite overall increase of the heart mass (Fig. 2). The left ventricle was similarly eccentrically hypertrophied, although to a milder degree than the contralateral ventricle. The endocardium of the left and right ventricle was diffusely thickened. A patent duct with a length of 0.8 cm and with a caliber of 0.4 cm (Fig. 3) connected the aorta to the pulmonary artery. The vascular lumen of the pulmonary artery proximal to ductus arteriosus was dilated over an extension of approximately 50 mm, forming a thin-walled sacculated structure (aneurysm). The left ventricle and the aorta contained large cruoric clots. Ten liters of clear fluid filled the abdominal cavity (marked hydroperitoneum). The liver was intensely enlarged, was bluish subserosally, and had rounded margins. Abundant dark red blood oozed from its cut surface. The hepatic parenchyma was moderately firm, with whitish lines alternating with depressed dark red areas (“nutmeg liver”) (Fig. 4). The abomasal submucosa and mesentery were markedly expanded by edema. Kidneys were severely and diffusely congested.
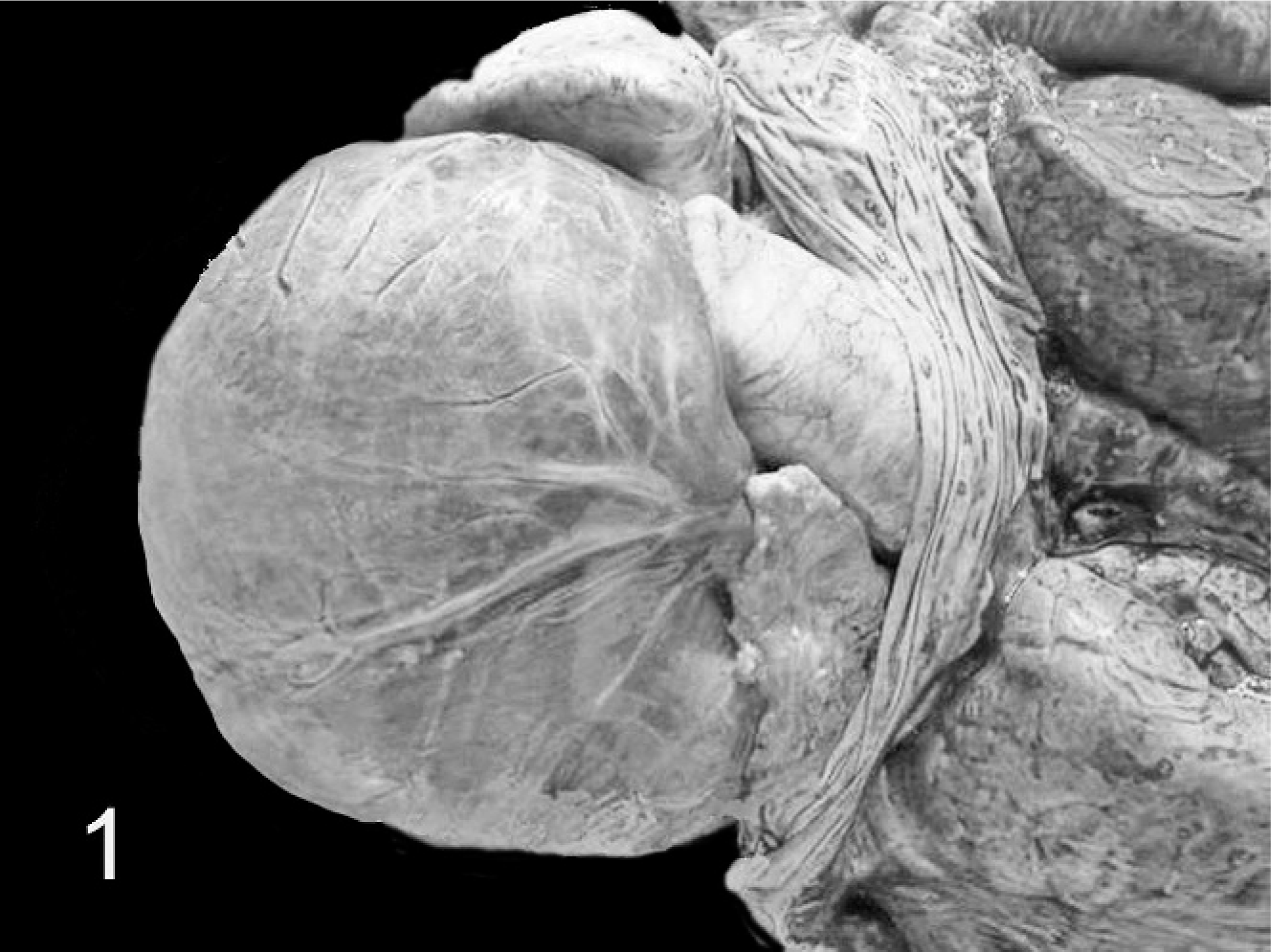
Heart; buffalo No. 2. The heart is too round with a markedly dilated right atrium.
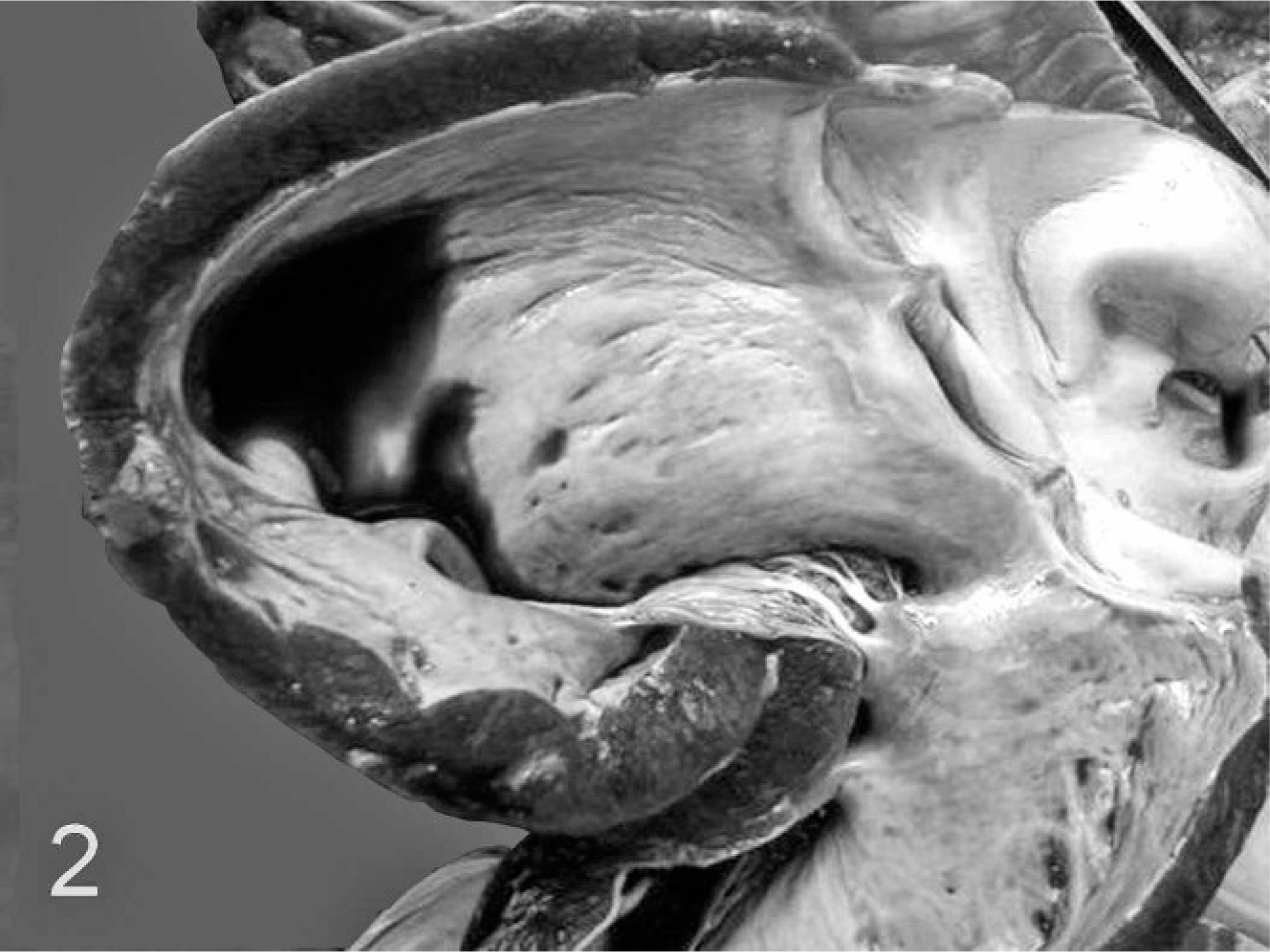
Heart; buffalo No. 2. The right ventricle is dilated and the wall is mildly thinner than normal (eccentric hypertrophy). There is subendocardial fibrosis.
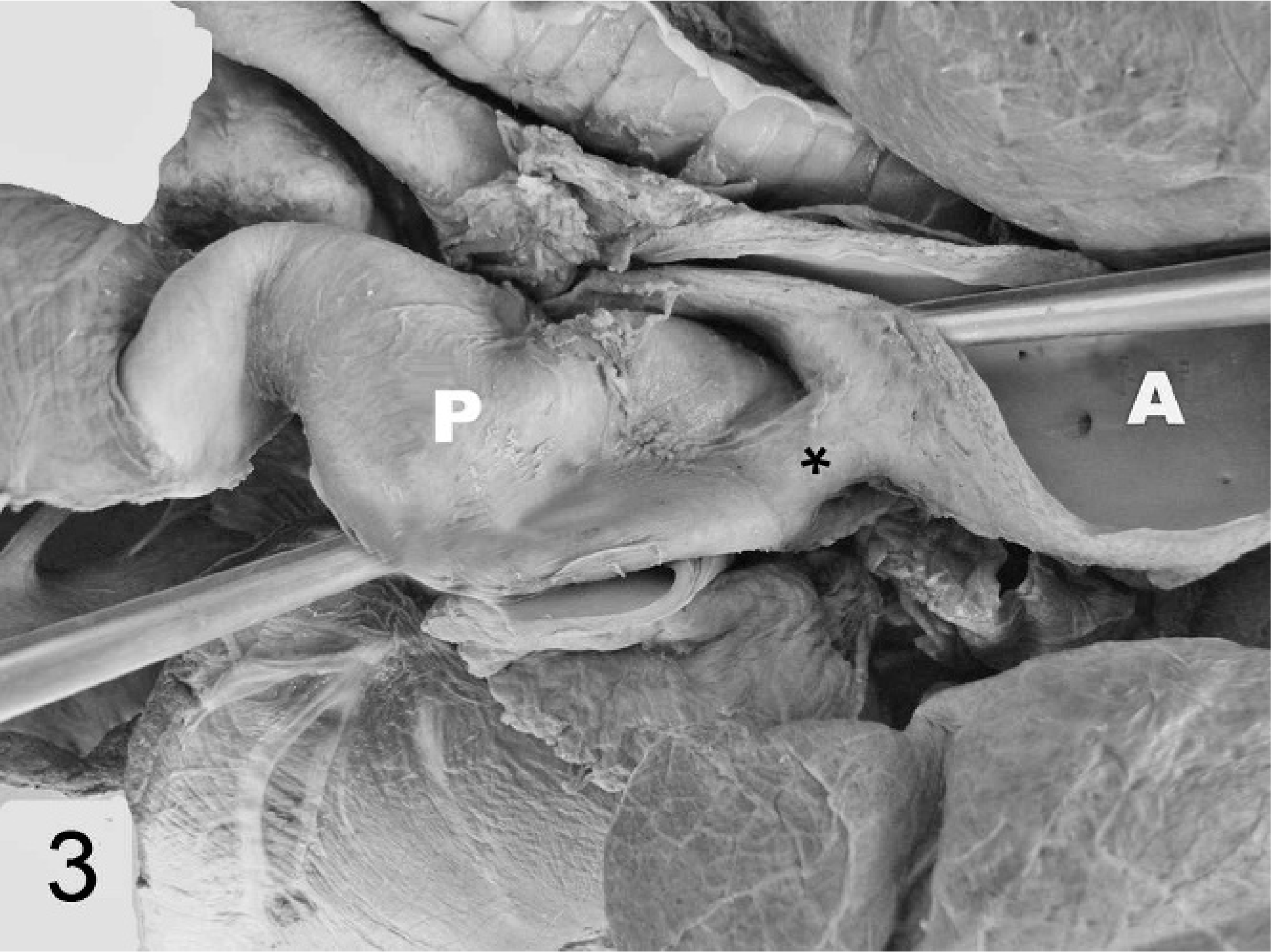
Heart; buffalo No. 2. A patent ductus arteriosus (∗) connects the pulmonary artery (P) to the aorta (A).

Liver; buffalo No. 1. The hepatic parenchyma has whitish lines alternating with dark red areas (nutmeg liver).
The histopathologic changes were also similar in both animals. The pulmonary parenchyma was markedly collapsed (compressive atelectasis). Abundant fibrous tissue surrounded and separated the Purkinje fibers in the subendocardial region of the left and right ventricle (Fig. 5). The liver had irregular sinusoidal distension in the midzonal region. Centrilobular hepatocytes were lost and replaced by fibroblasts and collagen (Fig. 6). The fibrosis bridged centrilobular areas and also linked centrilobular zones to portal triads (cardiac fibrosis). In some hepatic lobules, only a few blood-distended sinusoids remained, surrounded by hepatocytes, some of which were degenerate or necrotic. Additional findings included multifocal bile duct proliferation.
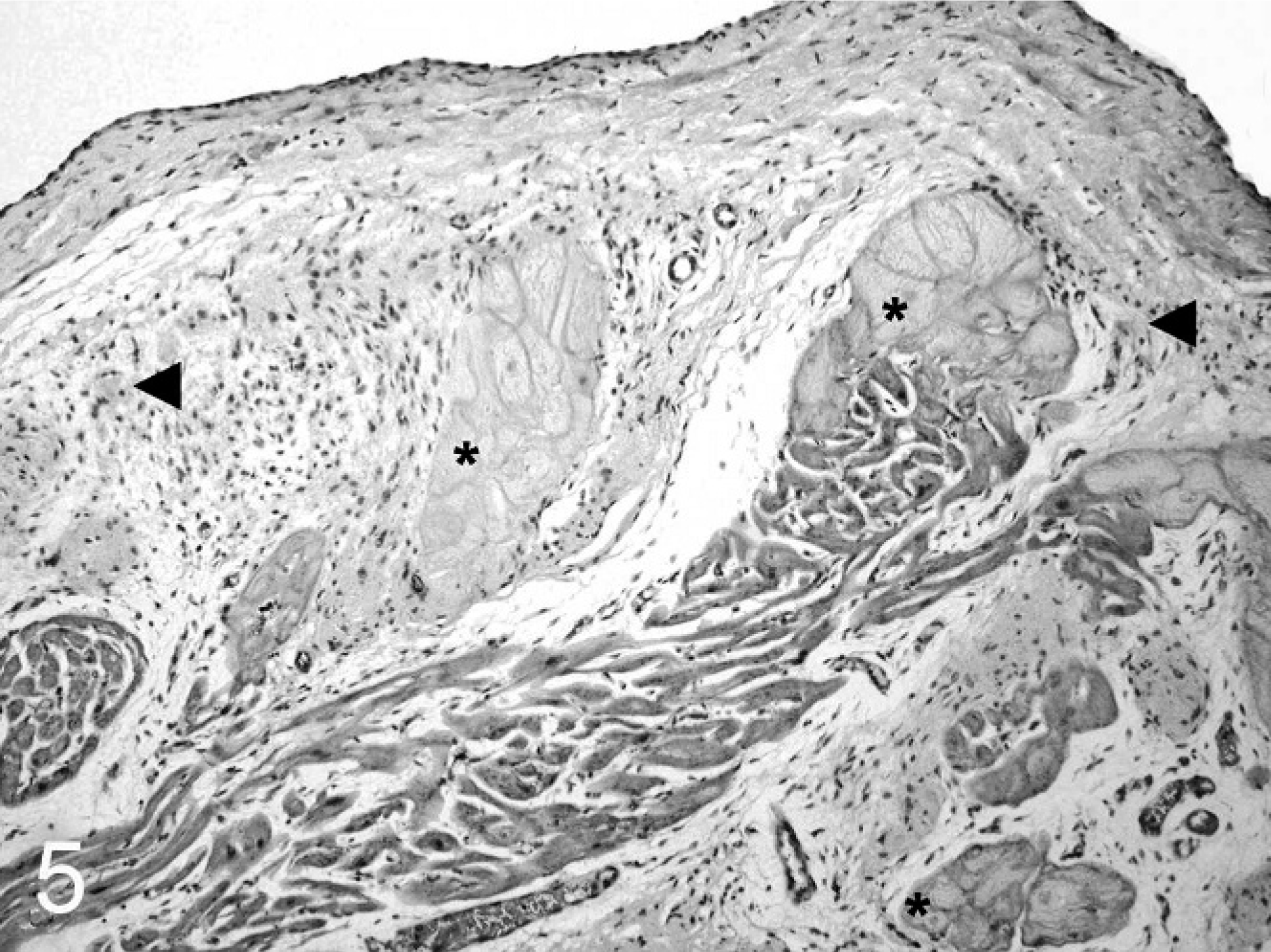
Heart; buffalo No. 2. Abundant fibrous tissue (◂) surrounds Purkinje fibers (∗) in the right ventricle and extends into the underlying myocardium (subendocardial fibrosis). HE.
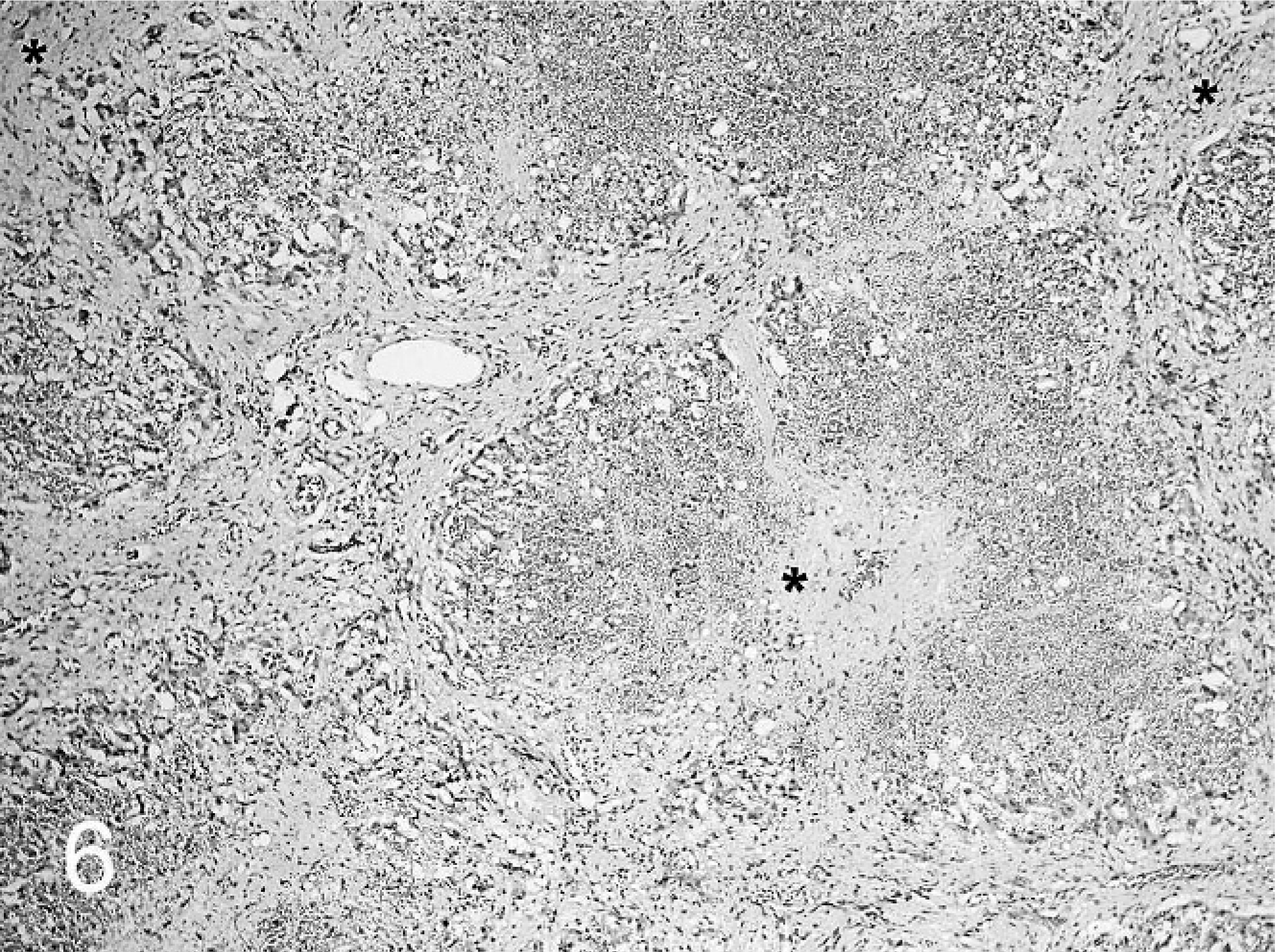
Liver; buffalo No. 1. Hepatocytes in the centrilobular area are completely replaced by fibrous tissue (∗). HE.
The main findings in both buffalos were PDA accompanied by biventricular compensatory myocardial hypertrophy and right-sided heart failure. Chronic fatigue and respiratory signs exhibited by both animals during exercise were probably a result of hypoxemia caused by low blood perfusion of the lungs and extensive compressive atelectasis as a result of the hydrothorax. Patent ductus arteriosus is a common anomaly in dogs, but it is rare in other domestic animals. 5 The most common cardiac abnormalities in bovine are defective ventricular and atrial septa and transposition of the great vessels. 6 In buffalos there are no reports of cardiac anomalies. Most cases reported in bovine describe early onset of clinical signs, often soon after birth, followed by death between the ages of 5 days and 11 months. 4, 8 There is one report of an adult cow that developed thromboarteritis of a PDA at 4 years of age. 6 Animals with PDA have a good body condition at birth; they may, however, become apathetic soon after and die within a week. 10 Others survive for months with a history of poor growth, signs of right heart insufficiency and tachypnea before dying suddenly. 8
Clinical signs generally depend on the size of the PDA, on the direction of the blood flow, on the concurrent presence of other congenital lesions, and on the accompanying complications. 9, 10 Fatigue, dyspnea, cyanosis, and exercise intolerance are the most common signs of a left-to-right shunting PDA. 3, 10 Reversed (right-to-left) shunting may be associated either with a large PDA, which causes progressively increasing pulmonary vascular resistance, or with primary pulmonary hypertension. 2, 3 Pulmonary hypertension in such a reversed PDA is generally the result of persistently underdeveloped, essentially fetal pulmonary circulation, or a combination of both mechanisms. Pulmonary vascular lesions causing right-to-left blood shunting frequently are advanced and irreversible, even when diagnosed early. 2 In cases in which the arterial duct has a small internal diameter, the blood flow from the aorta to the pulmonary artery (left-to-right shunt) is minimal during both systole and diastole, without any precocious clinical signs. 9 In the 2 cases reported here, the right eccentric ventricular hypertrophy, the pulmonary arterial aneurysm, and the alterations indicative of right-sided heart failure likely resulted from blood flow from the aorta to the pulmonary artery (left-to-right shunt). Nonetheless, the eccentric hypertrophy of the left ventricle indicates that reversed blood flow is likely to have also occurred. This eccentric hypertrophy probably resulted from a combination of an increased diastolic volume and pressure load. Dilatation of the aorta near the PDA is one of the recognized abnormalities that accompany this condition. Aneurysmal dilatation results from and contributes to abnormal blood flow dynamics and increased tension of the vascular wall, leading to luminal expansion of the vessel. 7 Subendocardial fibrosis is regarded as a reaction of the endocardium to abnormal jets of blood or to turbulence following congenital or acquired valvular disorders. 9 The pulmonary arterial aneurysm and subendocardial fibrosis in the 2 buffalo calves probably resulted from a similar combination of turbulent blood flow and blood pressure overload.
Cardiac anomalies may be associated with inherited genetic defects, with teratogen exposure, or with nutritional deficiencies. 4 Hereditary factors have been suggested for some types of cardiac defects in cattle, dogs, sheep, and humans. In this case, both calves had different dams, but the same sire, suggesting an underlying hereditary factor although no other developmental anomalies were thus far seen in this herd.
