Abstract
Pallido-nigral spheroids associated with iron deposition have been observed in some aged clinically normal nonhuman primates. In humans, similar findings are observed in neurodegeneration with brain iron accumulation diseases, which, in some cases, show associated mutations in pantothenate kinase 2 gene (PANK2). Here we present an aged gorilla, 40 years old, suffering during the last 2 years of life from progressive tetraparesis, nystagmus, and dyskinesia of the arms, hands, and neck, with accompanying abnormal behavior. The postmortem neuropathologic examination revealed, in addition to aging-associated changes in the brain, numerous corpora amylacea in some brain areas, especially the substantia nigra, and large numbers of axonal spheroids associated with iron accumulation in the internal globus pallidus. Sequencing of the gorilla PANK2 gene failed to detect any mutation. The clinical, neuropathologic, and genetic findings in this gorilla point to an age-related pallido-nigral degeneration that presented PKAN-like neurologic deficits.
Keywords
Age-related neurologic lesions, such as neuronal loss, lipofuscin and neuromelanin accumulation, gliosis, corpora amylacea (CA), amyloid deposits, and vascular changes, have been described, not only in human beings, but also in dogs 4 and nonhuman primates. 14, 27, 28 Moreover, in nonhuman primates, pallido-nigral spheroids associated with iron deposition have been observed in clinically normal rhesus monkeys, baboons, and macaque monkeys as a normal aging phenomenon. 5, 25, 27, 29 To our knowledge, in aged gorillas, pallido-nigral degeneration has not previously been reported; the only age-associated lesion described in the brain was the accumulation of senile plaques. 14 Here, we report the clinical, neuropathologic, and genetic findings in an aged male albino gorilla with neurologic deficits. Among normal aging lesions, abundant iron deposition and numerous spheroids in the globus pallidus (GP) and corpora amylacea in the substantia nigra (SN) are described.
A 38-year-old albino male Gorilla gorilla gorilla kept at the Barcelona Zoo developed squamous-cell carcinoma that started in the right nipple and extended to the armpit; it eventually led to its euthanasia 2 years later. The tumor was surgically removed, unsuccessfully, several times, and the gorilla never received chemotherapy. During the last year of life, the gorilla presented with a progressive neurologic disorder characterized by nystagmus, tetraparesis, and decreased muscle tone in the forelimbs, with difficulties in quadruped locomotion that forced him to support his body weight with the posterior extremities. Moderate chorea and dyskinesia that mainly involved the arms and hands, and the neck to a lesser extent, appeared later. At 2 months before his death, the severity of nystagmus and dyskinesia in the neck hampered the gorilla's ability to hold his gaze. One month before death, the tetraparesis was so evident that the gorilla was unable to climb the platforms in his pen. This was accompanied by increasing resting behavior and social withdrawal. Because of his oculocutaneous albinism, the gorilla presented photophobia and reduced visual acuity throughout his life. Two years after the presentation of the tumor, the generalized metastasis, which affected the lymph nodes and liver, that had brought on increased pain led to euthanasia after general anesthesia.
At postmortem, cervical spinal cord and coronal sections of the brain were fixed by immersion in 10% neutral buffered formalin and embedded in paraffin for the neuropathologic study. Other organs were also included in the postmortem study. Four-micrometer-thick sections were obtained with a sliding microtome and were stained with several histochemical and immunohistochemical stains. 3 Histochemical staining studies were performed with Perls’, von Kossa, Bielschowsky, Masson's trichrome, and Congo red stains. Immunohistochemical studies were performed for ubiquitin (1 : 100, Dakocytomation), tau (1 : 1000, Dakocytomation), α-B-crystallin (1 : 500, Novocasta), β-amyloid (βA4) (1 : 500, Dakocytomation), neuron-specific enolase (NSE) (1 : 400, Dakocytomation) and glial fibrillary acidic protein (GFAP) (1 : 500, Dakocytomation), 200 KDa phosphorylated neurofilaments (1 : 50, clone RT97, Boehringer), 200 KDa phosphorylated and nonphosphorylated neurofilaments (1 : 100, Sigma) and α-synuclein (1 : 500, Chemicon). Positive immunoreactivity was visualized with diaminobenzidine-tetrahydrochloride (DAB) (Sigma). Morphometric analyses were performed by using the software VideoTest Analysis System (VideoTest Company, St. Petersburg, Russia). On macroscopic examination, only a light brown discolouration of the GP and a few hemorrhagic foci in the striatum were observed.
The microscopic examination revealed axonal spheroids (15 per 20× magnification field) and abundant iron deposition in the internal GP (Fig. 1A), together with moderate neuronal loss and gliosis. Spheroids were round or oblong eosinophilic structures of variable size, between 15 µm and 30 µm in diameter, strongly stained with Bielschowsky stain. Immunohistochemical studies showed ubiquitin, α-B-crystallin, tau, and 200 KDa neurofilament accumulation within the spheroids (Table 1). Perls’ method clearly revealed iron deposition in the neuropil and within some spheroids (Fig. 1B). Increased neuromelanin pigment was also observed in this area.
Immunoreactivity of corpora amylacea and spheroids in different brain areas.

ND = not determined.
Halo: peripheral staining.
Other: brain stem, spinal cord, cortex, and cerebellum.
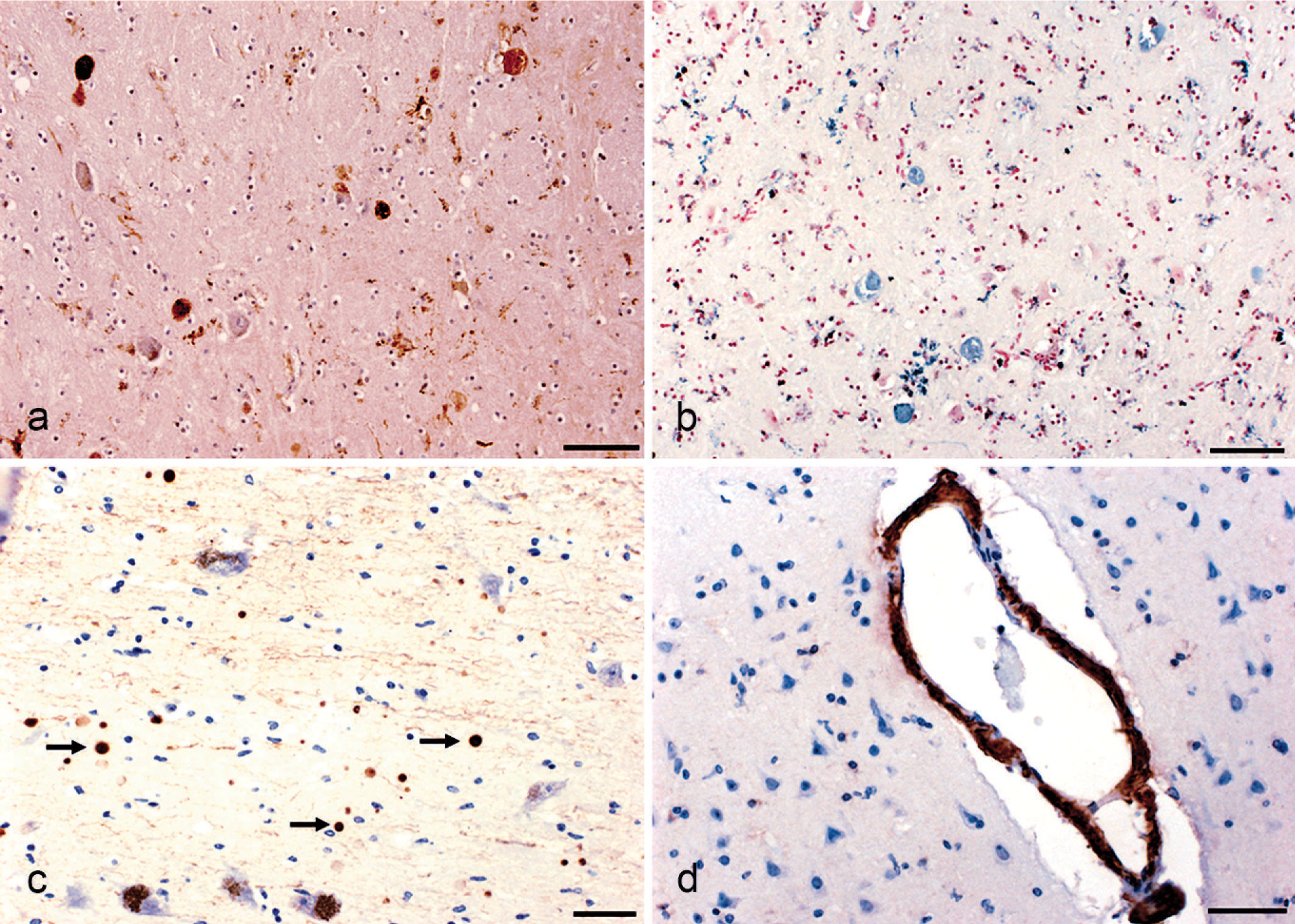
Brain parenchyma; Gorilla gorilla gorilla.
Spheroid ultrastructure was examined by using transmission electron microscopy (H-7000 at 100 kV, Hitachi). To this end, the internal GP was excised from the paraffin block, dewaxed, rehydrated, postfixed with 1% osmium tetroxide, and embedded in SPURR resin. On ultrastructure examination, these spheroids had a dystrophic nature (Fig. 2A and C), evidenced by the accumulation of abnormal aggregates of electron-dense vesicles that measured from 0.1 to 1.0 µm, together with granular and filamentous material, and lipofuscin instead of the normal cellular organelles (Fig. 2B and D). Electron-dense material consistent with iron had also accumulated in some spheroids. Surrounding microglia cells had accumulation of a similar material in their perikaryon (Fig. 2E and F).
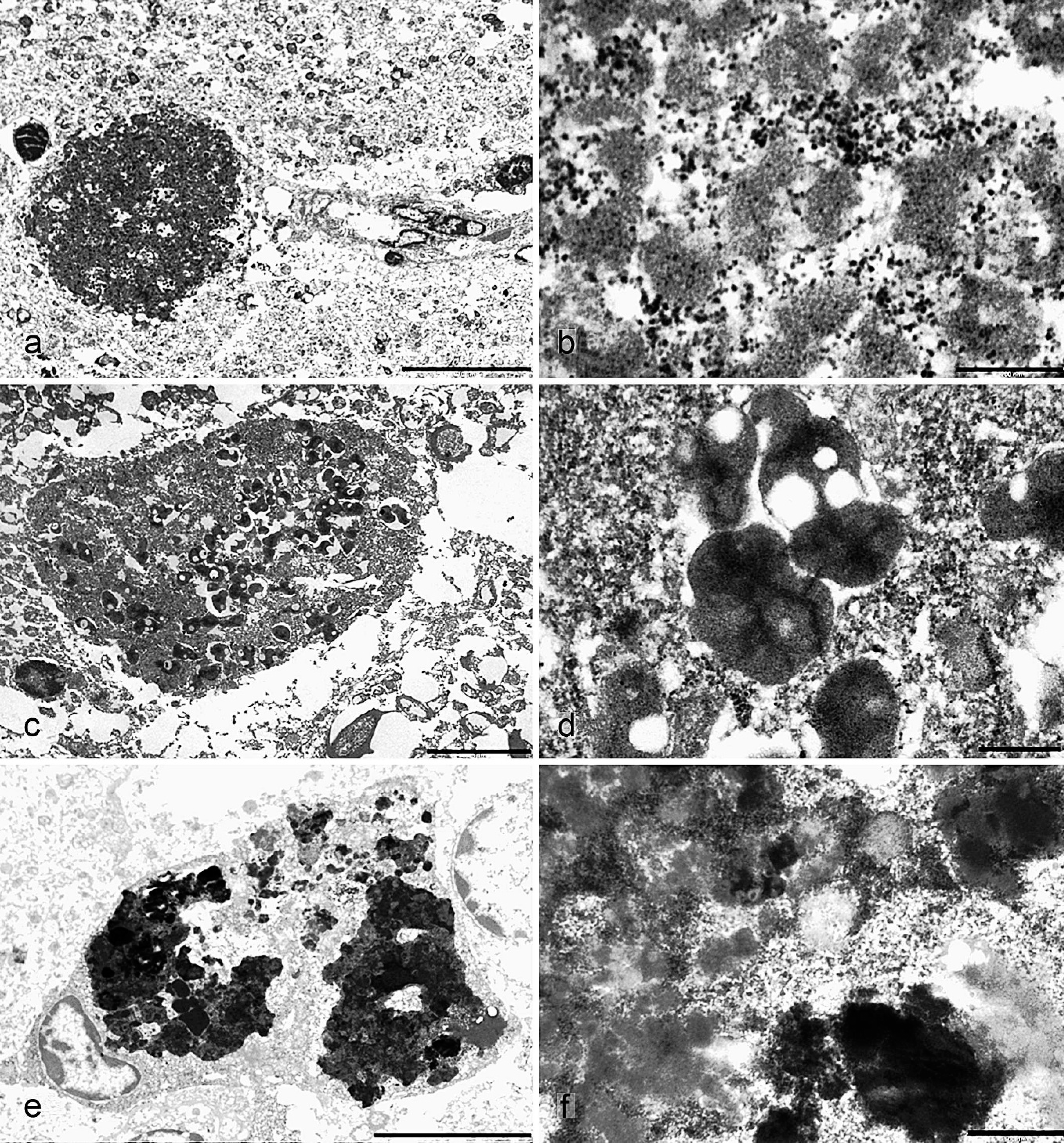
Electron micrographs. Brain parenchyma; Gorilla gorilla gorilla.
CA were observed throughout the brain but with a topographic predilection for the periventricular white matter, hippocampus, medial temporal cortex, medulla oblongata, and, more abundantly, the pars reticulata of the SN (Fig. 1C). CA from the SN and hippocampus were smaller than those found in the rest of the brain. Immunohistochemical studies showed area-dependent immunoreactivity of CA (Table 1).
Vascular changes were also observed and were characterized by thickening, fibrosis (Masson's trichrome staining) and hyalinosis of small arteries, calcification of the tunica externa of blood vessels in the striatum and choroid plexus (von Kossa staining) and β-amyloid deposition (Fig. 1D) in some small blood vessels of the cerebral cortex (Congo red staining and βA4 immunostaining). Diffuse microspongiosis, which was more accentuated in perivascular spaces, sporadic irregular glial nodule formation and widespread accumulation of lipofuscin in neurons and glial cells, were observed throughout the nervous parenchyma.
Vascular changes, microspongiosis, lipofuscin accumulation, and gliosis are common features in the aged brain of nonhuman primates and are not usually accompanied by neurologic signs. 14, 22, 27, 28 CA accumulation in the brain is also a normal aging phenomenon. 6 Most of the CA found in the gorilla had accumulations of ubiquitin and microtubule-associated proteins similar to CA accumulated in human aging. 6 However, CA located principally in the hippocampus and SN in the gorilla were almost all negative to both these proteins and mostly had accumulations of phosphorylated neurofilaments and α-synuclein. These findings suggest that CA found in the hippocampus and principally in the SN of this gorilla might have a different origin than CA associated with aging.
Even though the gorilla was albino, he presented neuromelanin deposits in the GP and inside the SN neurons. The albinism was from deficiencies in the tyrosinase enzyme, responsible for eumelanin synthesis. The neuromelanin synthesis process that takes place in the brain is obscure. However, several studies proved that tyrosinase is not necessary for the synthesis of neuromelanin; other enzymes, such as tyrosine hydroxylase and peroxidase, might be involved. 31
Spheroids and iron deposition in the GP and SN are described in nonhuman aged primates 5, 25, 27, 29 and aged humans. 2 The ultrastructural and immunohistochemical studies suggest that these spheroids originate from degenerating axons 5, 8 and/or astroglia. 2, 27, 29 In our gorilla, the Bielschowsky staining, the positive tau and NF200KDa immunostaining, and the negative immunostaining of GFAP suggest a neuronal and not an astroglial origin for the spheroids. The accumulation of neurofilaments indicates that at least part of the accumulated material inside the spheroids comes from degraded neuronal cytoskeleton. Tau accumulation in spheroids would indicate an attempt to repair the axonal damage. 18 The α-β-crystallin overexpression in the gorilla spheroids would protect axons from the aggregation of intermediate filaments and proteins triggered by iron-mediated oxidative stress. 8, 26 The ubiquitin immunostaining of the spheroids would indicate an activation of the proteolytic nonlysosomal system for the degradation of abnormal filamentous cytoskeletal proteins. 18
No data on iron and spheroids in the brain of aged gorillas has previously been reported. Spheroids in our gorilla could correspond to a normal aging phenomenon. However, all the reported nonhuman aged primates who presented with these pallido-nigral lesions were clinically normal; none of the reported cases manifested neurologic signs. In contrast, this gorilla presented with a progressive neurologic deterioration, including gait ataxia, involuntary movements, and tetraparesis. In the postmortem study, the squamous carcinoma was observed to have penetrated to the fifth intercostal muscle and the regional lymph nodes; no other tissues, including peripheral nerves, were affected. We did not find evidence of neoplastic involvement of the nervous system, peripheral nerve (brachial plexus and sciatic nerve) or skeletal muscle (triceps) lesions, or other nervous lesions that might be causative of these disorders. No lesions were observed in the cervical spinal cord; unfortunately, the whole spinal cord was not available to examine the causes of tetraparesis. To elucidate the cause of our findings, a different diagnosis was undertaken that included human disorders that present with extrapyramidal signs associated with iron and spheroids in the brain.
Brain iron accumulation and axonal spheroids associated with progressive extrapyramidal disorders are observed in human neurodegeneration with brain iron accumulation (NBIA) disease, formerly Hallervorden-Spatz disease. 9 The subgroups of NBIA include, among others, neuroferritinopathy, aceruloplasminemia, and pantothenate kinase-associated neurodegeneration (PKAN).
Neuroferritinopathy is a disease caused by mutations in the ferritin gene. Spheroids are observed in GP, putamen, and white matter tracts throughout the central nervous system, together with an increased amount of iron in several brain areas, including the GP. Cavitations of the basal ganglia are also observed. In some neuroferritinopathy forms, these findings are also accompanied by intranuclear and intracytoplasmic inclusions in glial and neuronal cells. 16 In addition, intranuclear and intracytoplasmic iron deposits are also observed inside cells of some parenchymal organs, such as hepatocytes, or in epithelial cells of renal tubes, fibroblasts in the skin, and endothelial cells of muscle capillaries and retina.
Aceruloplasminemia is a ceruloplasmin deficiency characterized by iron accumulation in the visceral organs and several areas of the brain, including the GP. In addition, deformed astrocytes with irregular nucleus and globular structures of astrocytic origin are some of the more characteristic histopathologic findings. On clinical examination, extrapyramidal neurologic signs are accompanied by diabetes mellitus. 15
In PKAN, usually caused by mutations in pantothenate kinase 2 gene (PANK2), 13 iron accumulation is specific to the GP and SN. Spheroids may be limited to both these areas, although they can also be observed in other brain areas. 9 Adult and senile onset of the disease is characterized by presenting a great heterogeneity of clinical signs, such as chorea, head movements, hand tremors, orofacial dyskinesia, an inability to walk, generalized paratonia and akinesia, muscular rigidity, epileptic seizures, and dystonia. 10, 11, 23, 30 Corroborative features may include retinal degeneration and acanthocytosis. 9
All 3 NBIA diseases are characterized by presenting clinical signs and pathologic findings similar to those observed in our gorilla. However, except for the clinical signs and iron and spheroids in the GP, none of the additional features of neuroferritinopathy and aceruloplasminemia were observed in the gorilla. Cavitations in basal ganglia, intraneuronal and intracytoplasmic inclusions, deformed astrocytes, or diabetes mellitus were not found in our gorilla. In addition to the GP, the gorilla presented high iron deposition in the liver and spleen, moderate deposition in the kidney, and low iron accumulation in the triceps muscle. Increased iron deposition in these organs may correspond to aging 1, 7 and the feed given to captive gorillas. 17 Dietary iron overload seems not to significantly increase the brain iron deposition. 21 Eyes, heart, pectoral lymph nodes, skin, lung, stomach, adrenal, and sciatic peripheral nerve in the gorilla did not show significant iron deposition.
None of the corroborative features included in PKAN were observed in the gorilla. However, these features are not usually observed in late-onset PKAN. 9 In our gorilla, spheroids and iron were only observed in the GP; however, in some late-onset human NBIA cases, this is the only brain area inlvolved. 11, 18 The presence of numerous CA in the gorilla SN is not surprising, because, in some NBIA cases, principally, those with late onset, additional findings are observed, such as neurofibrillary tangles, Lewy bodies, neuritic plaques, and Lafora bodies. 11, 23, 24, 30
Immunohistochemical studies of human NBIA spheroids show, as in our gorilla and some aged nonhuman primates, a neuronal origin of the spheroids with expression of stress proteins. 18, 24, 30 On ultrastructural examination, spheroids from human NBIA 18 are also similar to those observed in nonhuman aged primates 5, 27 and our gorilla. Consequently, neither immunohistochemistry nor ultrastructure were useful tools to differentiate between spheroids associated with aging and with NBIA. For this reason, to determine whether PANK2 mutations might be causative of the clinical signs observed in our gorilla, sequencing of this gene was undertaken.
To this end, PANK2 exons were sequenced from the gorilla, 1 human, and 2 nongenetically related gorilla controls from the Barcelona Zoological Garden and the European Collection of Cell Cultures. Primers were designed in accord with Hayflick et al., 13 whose sequence and polymerase chain reaction conditions were kindly provided by Shawn K. Westaway (Oregon Health and Science University). Sequencing was performed on an ABI 3700 (AB) DNA sequencer. Both strands were sequenced in all samples. Sequence alignment was carried out by using BioEdit v.7.0.5.3 and then were manually checked. Successful complete sequences were obtained for PANK2 exons 2, 3, 5, 6, and 7 from the present case and controls. Up to now, the gorilla PANK2 gene sequence had been unknown. GenBank accession numbers for gorilla PANK2 sequenced exons are in progress.
The sequence for the 5 exons revealed few differences between the gorilla and human PANK2 gene. The exon sequence obtained was identical in the 6 gorilla chromosomes analyzed. Thus, among the sequenced exons from our gorilla, no PANK2 missense mutations were observed. However, mutations in the unviable sequencing exons 1 and 4 cannot be ruled out as possible causes of the pathologic findings. In any event, many human NBIA cases that present a PKAN phenotype do not show PANK2 mutations, 12, 13 which suggests the presence of sporadic cases or the implication of a second gene in this syndrome, 12, 19 such as the PLA2G6 gene. 20
In summary, because of the clinical, neuropathologic, and genetic findings, together with the absence of other evident lesions that could be causative of the signs of the gorilla, we considered the possibility that the gorilla had a non-PKAN NBIA-like syndrome. However, with the exception of the clinical signs and the neuropathologic findings in the GP, we have no evidence of other NBIA corroborative features, such as PANK2 mutations, retinal degeneration, or acanthocytosis. 9 Consequently, we can only conclude that the findings in the gorilla point to age-related pallido-nigral degeneration, similar to that found in other nonhuman primates but present with neurologic deficits. It is worth noting that the gorilla was monitored and recorded 24 hours a day in his last year and a half of life by the zoo veterinarians. It may be the case that careful continuous monitoring and recording of the behavior of other aged nonhuman primates may reveal similar clinical signs. Because there is a great similarity in the pallido-nigral degeneration between aged nonhuman primates and human PKAN, it would not be surprising to observe similar neurologic deficits. Additional neuropathologic studies of other aged gorillas would be needed to determine whether our findings are really because of normal aging that occur in gorillas.
Footnotes
Acknowledgements
We thank B. Perez, S. Roca, M. Carmona, and R. Blanco of the Institut de Neuropatologia for their technical assistance; Dr. C. Maté and Dr. J. Fernández-Morán of the Barcelona Zoological Garden for management support, and T. Yohannan for editorial assistance. The authors also wish to acknowledge the care and expert dedication with which the primate curator (M. Teresa Abelló) and primate keepers of the Barcelona Zoo tended the gorilla, particularly during his final years. This work was supported by the Animal Tissue Bank of Catalunya (BTAC), the MCT project SAF2001–4772, PTA-2003–01–00568 and the UAB project PRP2005–09. The genetic project has been funded by grant BOS2003–08070 from the Ministerio de Educación y Ciencia, and by the Direcció General de Recerca, Generalitat de Catalunya (JB). Ultrastructural studies were carried out at the Electron Microscopy Service of the Universitat Autònoma de Barcelona.
