Abstract
As part of a high-throughput mutagenesis and phenotyping process designed to discover novel drug targets, we generated and characterized mice with a targeted mutation in Slc24a5, a gene encoding a putative cation exchanger. Upon macroscopic examination, Slc24a5—/— mice were viable, fertile, and indistinguishable by coat color from their heterozygous and wild-type litter mates. Ophthalmoscopic examination revealed diffuse retinal hypopigmentation, and a histologic examination of the eye confirmed the presence of moderate-to-marked hypopigmentation of the retinal pigmented epithelium (RPE), ciliary body, and iris pigment epithelium (IPE). Hypopigmentation was most severe in the anterior layer cells of the IPE, where melanosomes were smaller, paler, and more indistinct than those of the anterior stroma and posterior IPE. The pigment granules of the posterior IPE appeared to be nearly as dark as those in stromal melanocytes; however, both cell layers were thinner and paler than corresponding layers in wild-type mice. Ultrastructural analysis of the RPE, IPE, and ciliary body pigmented cells confirmed that mutation of Slc24a5 results in marked hypopigmentation of melanosomes in optic cup-derived pigmented neuroepithelium in the eyes. Milder reductions in melanosome size and pigmentation were noted in neural crest-derived melanocytes. The severe hypopigmentation of neuroepithelium-derived cells in the eyes resulted in a novel form of ocular albinism in Slc24a5—/— mice. Our findings suggest that SLC24A5 may be a candidate gene for some forms of ocular albinism and for the BEY1/EYCL2 locus previously associated with central brown eye color in humans.
Introduction
High-throughput mutagenesis and phenotyping approaches permit rapid characterization of protein function and identification of novel drug targets. 45 These approaches also identify genes that, when mutated in mice, result in pathologic phenotypes that can clarify the pathogenesis of specific diseases and that may serve as models for human diseases. Molecular cloning and analysis of many of the approximately 130 different genes involved in mouse coat color mutations helped elucidate the biochemical pathways and pathogenesis of pigmentation disorders. 37 Genes responsible for mouse coat color mutations are involved in several processes required for normal pigmentation, including the viability, migration, and proliferation of melanocytes, and the signaling pathways that control the production of melanosomes and melanin, as well as melanin biosynthesis itself. Because mutations within orthologous genes often produce similar pigmentation phenotypes or genetic diseases in both humans and mice, 3 several mouse coat color mutations proved valuable in explaining aspects of the pigmentation process and related diseases in humans. 31 For example, mutant mouse models helped in elucidating several genes and processes involved in melanin production or accumulation, the absence of which results in albinism. The 2 general forms of albinism include 1) oculocutaneous albinism (OCA), which affects pigmentation of the eye, skin, and hair; and 2) ocular albinism (OA), in which pigmentation defects only involve the eye. The common albino mouse is a spontaneous mutant with a defective tyrosinase gene Tyrc , which results in the inability to produce melanin. Tyrosinase is also nonfunctional in human oculocutaneous albinism type 1 (OCA1), which results in the absence of pigmentation in the eye and skin. 20 Other genes, such as oculocutaneous 2 (OCA 2), tyrosinase-like protein (TYRP1), or the melanocortin receptor (MC1R), are also involved in the tyrosinase pathway and influence iris pigmentation (see review by Sturm et al. 40 ) However, normal tyrosinase activity is present in several other forms of albinism, the most common being oculocutaneous albinism type 2 (OCA2). Mutations in the p gene result in the mouse pink-eyed dilution (p) phenotype, the equivalent of OCA2. 32
Recent investigations on zebrafish homozygous for the golden mutation showed that the hypopigmentation of the skin and retinal pigmented epithelium in mutants was because of melanin granules that were smaller, less dense, and more irregular in shape than melanin granules of wild-type fish. 21 The zebrafish golden gene encodes a putative cation exchanger slc24a5 (nckx5) localized to intracellular membranes (most likely on melanosomes or their precursors). Human SLC24A5 has also been localized to melanosomes. 5 Slc24a5 activity is believed to increase uptake and accumulation of calcium in melanosomes, and the paler melanin phenotype in golden zebrafish is thought to result from abnormally low calcium levels in the melanosomes. 21 Later studies associated polymorphisms in the human ortholog of golden SLC24A5 with lighter skin pigmentation in humans. A single nucleotide polymorphism in SLC24A5 changes an alanine at position 111 (A111) to threonine (T111); the T111 variant is almost ubiquitous in European populations, whereas, the ancestral A111 genotype is present in 93% to 100% of African, Native American, and East Asian populations. 21
Interestingly, no mouse coat color mutations were reported in the chromosome 2 region that contained the orthologous mouse Slc24a5 gene. 37 To investigate the biological function of Slc24a5, we generated mice in which the Slc24a5 gene was disrupted through targeted mutation. The studies confirm the conserved function of Slc24a5 in controlling pigmentation in mammals.
Materials and Methods
Generation of mutant Slc24a5 mice
The Slc24a5 targeting vector was derived by using the Lambda KOS system. 42 The Lambda KOS phage library was screened by polymerase chain reaction (PCR) by using exon 3-specific primers 24a5-1 [5′-CTGCAACATCCTGCGACAGTCC-3′] and 24a5-2 [5′-CGTTGCAGCTATAGAAACCCTATG-3′]. The PCR-positive phage superpools were plated and screened by filter hybridization by using the 428 base pair (bp) amplicon derived from primers 24a5-1 and 24a5-2 as a probe. Two pKOS genomic clones, pKOS-39 and pKOS-57, were isolated from the library screen and confirmed by sequence and restriction analysis. Gene-specific arms (5′-GTAATCACTTCCTTCATCTTCAGCCCTTGGACTG-3′) and (5′-CAGCTGAGACTGCAATAGCTGTTGAAGTGAAGG-3′) were appended by PCR to a yeast selection cassette that contained the URA3 marker. The yeast selection cassette and pKOS-39 were cotransformed into yeast, and clones that had undergone homologous recombination to replace a 784-bp region that contained coding exons 3–5 with the yeast selection cassette were isolated. The yeast cassette was subsequently replaced with a BetaGeo (BetaGalactosidase-Neomycin resistance fusion)/Puro selection cassette to complete the Slc24a5 targeting vector. The NotI linearized targeting vector was electroporated into 129S5/SvEvBrd (Lex-2) ES cells. G418/FIAU resistant ES cell clones were isolated, and correctly targeted clones were identified and confirmed by Southern analysis by using a 399-bp 5′ external probe (11/12), generated by PCR by using primers 24a5-11 [5′-CTATAGATGCTGTCTGTAGCCA-3′] and 24a5-12 [5′-CACTATAACTGCAGTGTGGACC-3′], and a 413-bp 3′ external probe (15/16), amplified by PCR by using primers 24a5-15 [5′-CAGTGATGGGCCTTACTTTGTTG-3′] and 24a5-16 [5′-CTGTTATTGTTAGAGCAAGAGTC-3′]. Southern analysis by using probe 11/12 detected a 15.8-Kb wild-type band and a 21.5-Kb mutant band in BglII digested genomic deoxyribonucleic acid (DNA), whereas, probe 15/16 detected a 16.1-Kb wild-type band and a 18.5-Kb mutant band in HpaI-digested genomic DNA. Six targeted ES cell clones were identified, and one was microinjected into C57BL/6-TyrBrd (albino) blastocysts to generate chimeric animals that were bred to C57BL/6-TyrBrd (albino) females, and the resulting heterozygous offspring were interbred to produce homozygous Slc24a5 deficient mice. Knockout F2 mice used in phenotyping studies were produced by intercrossing the F1 heterozygous knockout (−/+) offspring of chimeric founder parents; wild-type, heterozygous, and homozygous F2 litter mates were produced in an approximately a 1 : 2 : 1 Mendelian ratio. Use of the albino variant of C57BL/6 permits simple visual recognition of chimeric offspring, 47 because they have dark eyes and patches of dark hair that derive from stem cells from the agouti 129S5/SvEvBrd. In addition, the 129S5/SvEvBrd mouse was used as a source of embryonic stem cells because of its demonstrated reliability in production of tractable embryonic stem cells. 35 Determination of the genotype of mice at the Slc24a5 locus was performed by screening DNA from tail biopsy samples by using quantitative PCR for the Neo cassette. This strategy enabled discrimination of zero, one, or two gene disruptions per genome that represented Slc24a5+/+ (wild type), Slc24a5+/− (heterozygous), and Slc24a5−/− (homozygous) mice, respectively.
Mouse husbandry
Mice were housed in a barrier facility at 24°C on a fixed 12-hour light and 12-hour dark cycle and were fed rodent chow no. 5001 (Purina, St. Louis, MO) ad libitum. Procedures that involved animals were conducted in conformity with the Institutional Animal Care and Use Committee guidelines that are in compliance with the state and federal laws and the standards outlined in the Guide for the Care and Use of Laboratory Animals (National Research Council, 1996). 13
Phenotypic screen
F2-generation wild-type and homozygous (null) mice were subjected to a comprehensive battery of phenotyping screens to include behavioral tests (such as circadian rhythm, open field, inverted screen, prepulse inhibition of the acoustic startle response, tail suspension, marble burying, and context trace conditioning), funduscopy, and angiography examinations, blood pressure measurements, serum chemistries, complete blood cell count, urinalysis, quantitative magnetic resonance, computed axial tomography (CAT) scans, and Micro-CAT scans as previously described. 44
Histopathology
Eyes from F2-generation wild-type and null mice (in groups of five) were collected at 4, 6, 8, and 12 weeks of age. Eyes from more than 40 Slc24a5 null mice at ages up to 1 year of age were also examined. Immediately after euthanasia, Slc24a5−/− mice and age-matched normal control mice were fixed by cardiac perfusion with 10% neutral buffered formalin. Tissues were collected and immersed in 10% neutral buffered formalin for an additional 48 hours, except the eyes, which were removed and fixed by immersion in Davidson's fixative (Poly Scientific, Bay Shore, NY) overnight at room temperature. All tissues were embedded in paraffin, sectioned at 4 μm, and mounted on positively charged glass slides (Superfrost Plus, Fisher Scientific, Pittsburgh, PA) and stained with hematoxylin and eosin (HE) for histopathologic examination.
Electron microscopy
For the ultrastructural studies, deep anesthesia was induced by administering avertin (1.25% solution 2,2,2, tribromoethanol in 2-methyl-2-butanol in phosphate buffered saline solution) at 0.2 ml per 10 g body weight given intraperitoneally. Anesthetized mice were perfused with 3.5% glutaraldehyde in 0.1 M phosphate buffer. Eyes were removed and held in the same fixative overnight before dissection. Selected sections of ocular tissues were then incubated in 2% osmium tetroxide, stained with 2% uranyl acetate, dehydrated in graded ethanols, and embedded in Spurr's resin. Semithin sections were stained with 1% toluidine blue. Ultrathin sections were obtained and collected on 200-mesh grids, poststained with 4% methanolic uranyl acetate and Venable's lead citrate, and examined with a transmission electron microscope.
β-Galactosidase staining and LacZ histochemistry
Mice were anesthetized as described above, and the anesthetized mouse was then perfused sequentially via the left ventricle with 8 ml of saline solution; then 10 ml of β-Gal fixative (0.2% glutaraldehyde, 1.5% paraformaldehyde, 2 mM MgCl2, 5 mM ethylene glycol, 100 mM sodium phosphate [pH 7.3]); followed by 2 ml of β-Gal rinse (0.2% Nonidet-P40 [NP-40], 0.1% sodium deoxycholate, 2 mM MgCl2, 100 mM sodium phosphate); and, finally, 10 ml of β-Gal stain (5 mM K3Fe(CN)6 [potassium ferricyanide], 5 mM K4Fe(CN)6 [potassium ferrocyanide], 1 mg of 5-bromo-4-chloro-3-indolyl-D galactopyranoside [β-Gal] [dissolved in dimethylformamide] per ml, 0.2% NP-40, 0.1% sodium deoxycholate, 2 mM MgCl2, 100 mM sodium phosphate [pH 7.3]). Tissues were then dissected and postfixed in a β-Gal fix for 20 minutes, rinsed 3 times for 10 minutes in the β-Gal rinse, and then incubated in the β-Gal stain solution for 48 hours. After three additional 10-minute washes in β-Gal rinse, the tissues were placed in Bouin's fixative for 24 hours before dehydration and embedding in paraffin. Sections were cut at 4 μm and counterstained with nuclear fast red.
Results
Gross and histologic findings
Slc24a5−/− mice were behaviorally and anatomically normal and had normal survival, growth rates, and fertility. Routine serum chemistry and hematology workups were unremarkable. On macroscopic examination, the coat colors (either albino, agouti, or black) and eye color of Slc24a5−/− mice were indistinguishable from wild-type litter mates. However, only one Slc24a5−/− mice with black coat color was produced in the F2 generation under a randomized breeding scheme of F1 heterozygous mice (Table 1). Similarly, a higher than normal Mendelian ratio of agouti to black (nonagouti) coat color was noted in mice heterozygous for the Slc24a5 targeted mutation. This increased agouti :black ratio was expected, because the agouti coat color and Slc24a5 genes are located close enough on mouse chromosome 2 to be linked. The appearance of small numbers of black mice heterozygous or homozygous for the Slc24a5 mutation showed that some chromosomal crossover events had occurred.
Genotype and coat color of F2–generation mice

Under microscopic examination, longitudinal sections of hairs taken from agouti wild-type and Slc24a5−/− mice could not be differentiated at lower magnifications, with hairs from both genotypes having similar diameters and pigmentation. However, subtle differences were detected at high magnification; medullary hair cells in agouti Slc24a−/− mice contained pigment granules that were smaller and paler than those in agouti wild-type litter mates (Figs. 1, 2). A similar total volume of melanin granules appeared to be packed into each medullary cell within the hair shaft, which resulted in a comparable gross appearance.

Hair; agouti Slc24a5+/+ mouse. Melanin pigment granules fill medullary hair cells. Inset: Large globular melanin granules (arrowheads). HE.

Hair; agouti Slc24a5−/− mouse. Melanin pigment granules also fill medullary hair cells in null mice. Inset: Individual melanin granules are smaller and more variable in size in null mice (arrowheads). HE.
Although haircoat coloration of Slc24a5 null mice was grossly normal, a careful examination of skin surfaces of the tail, footpads, and ears of agouti Slc24a5−/− mice revealed that the skin covering the ear pinna was paler than normal. In mice, pigmented cells in the skin are generally restricted to the hair follicles except on the ears, around the nose and perineal areas, footpads, and tail. In these areas, melanocytes are most common in the superficial dermis, although melanin pigment is also present in the epidermis in some foci. A comparison of microscopic sections of skin of agouti wild-type and Slc24a5−/− mice, showed notably reduced amounts of melanin in dermal and epidermal locations in the ear pinna. Again, by light microscopy, the melanin pigment granules in the epidermis of wild-type mice (Fig. 3) were noticeably larger and darker than those observed in Slc24a5−/− mice (Fig. 4). In the epidermis of wild-type and Slc24a5 null mice, melanin granules tend to cluster in caps that cover the more superficial surface of the cell nuclei (Figs. 3, 4) but in wild-type mice, granules were also scattered more widely in the cytoplasm.
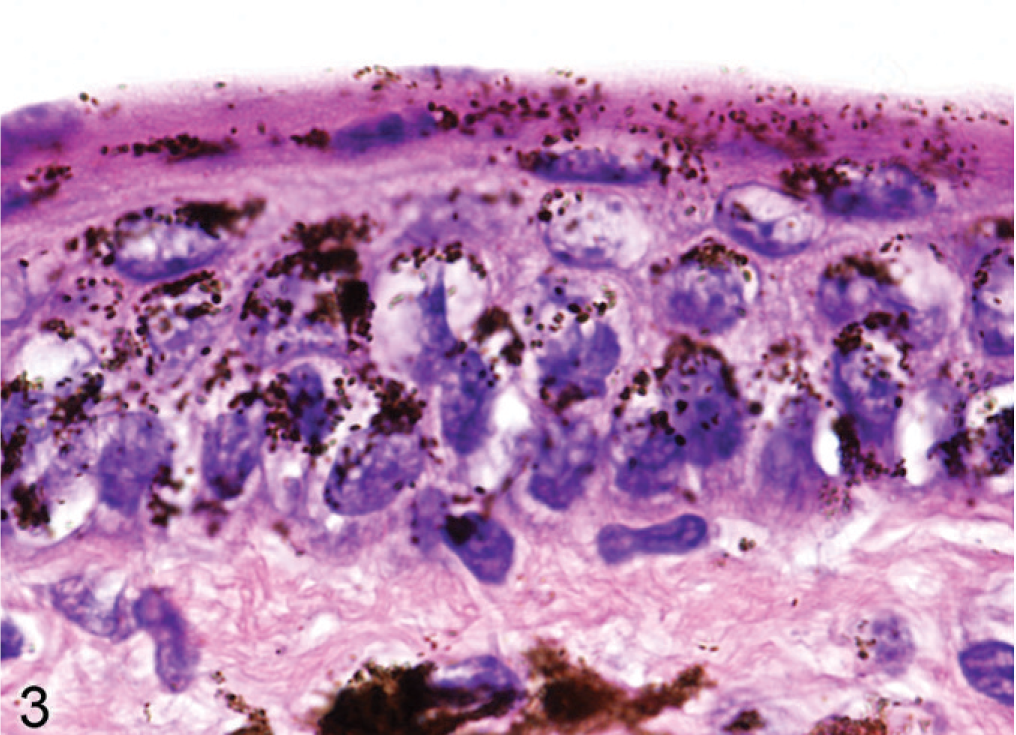
Pinna epidermis; agouti Slc24a5+/+ mouse. Large melanin granules are widely dispersed in the epithelial cells and dermal melanocytes. HE.
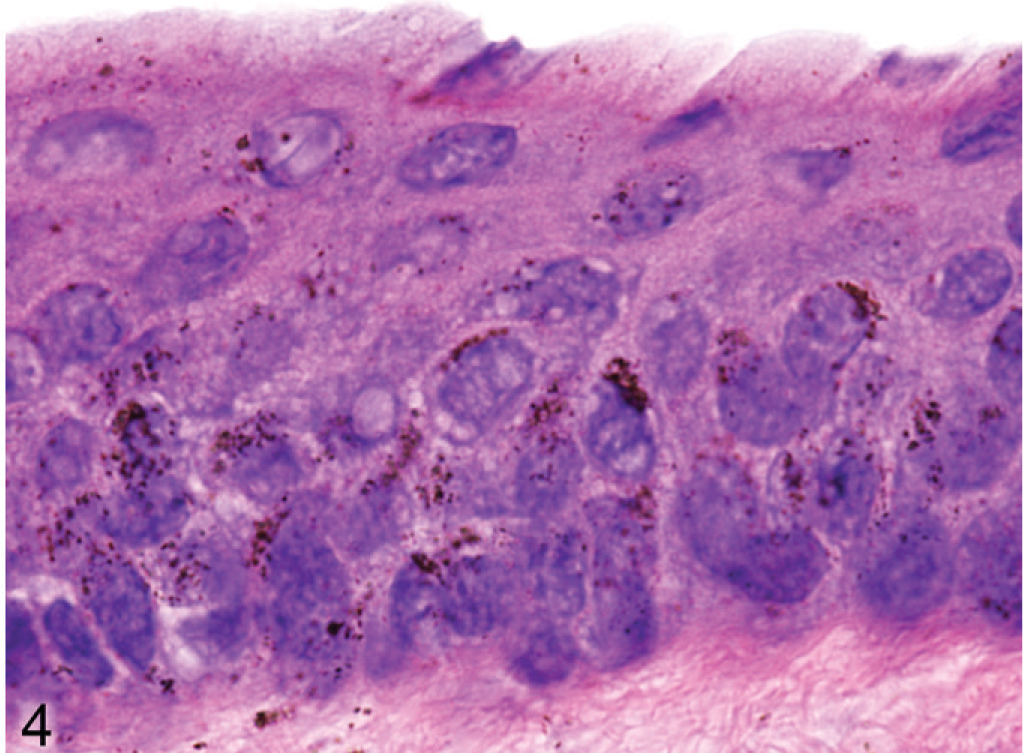
Pinna epidermis; agouti Slc24a5−/− mouse. Melanin granules appear to be reduced in number, and are smaller and paler than those in wild-type litter mates. HE.
Although the hypopigmentation phenotypes that affect the integument were relatively subtle, being evident only with microscopy, dramatic differences in ocular pigmentation between wild-type and Slc24a5−/− mice were identified by gross examination of both the retina and iris. The marked hypopigmentation of the fundus was detected easily by ophthalmoscopy, and histologic evaluation showed that the most severe hypopigmentation involved the neuroepithelium-derived pigmented cells of the eye, which resulted in a novel form of ocular albinism. Although irides appeared grossly normal, the decreased pigment density in Slc25a5−/− mice could be detected by transillumination of dissected irides. In wild-type mice, dense iridal pigmentation completely blocked the passage of light (Fig. 5), whereas, the markedly reduced volume of melanosomal pigmentation in irides null mice made the irregular tubular cytoplasmic extensions of stromal melanocytes visible by transillumination (Fig. 6). Ophthalmoscopic examination of agouti Slc24a5−/− mice also revealed diffuse retinal hypopigmentation in comparison with agouti wild-type litter mates. In both wild-type and Slc24a5 null agouti mice, the superficial retinal blood vessels were clearly visible radiating from the optic cup; however, in eyes of wild-type mice, the underlying choroidal vessels were mostly obscured by the dark retinal pigmented epithelium (Fig. 5 inset), whereas, in Slc24a5 null mice, the markedly reduced retinal pigmentation resulted in an albino-like fundus, with choroidal vessels that appeared as ill-defined pale pink blotches and streaks that covered the entire fundus field (Fig. 6 inset).
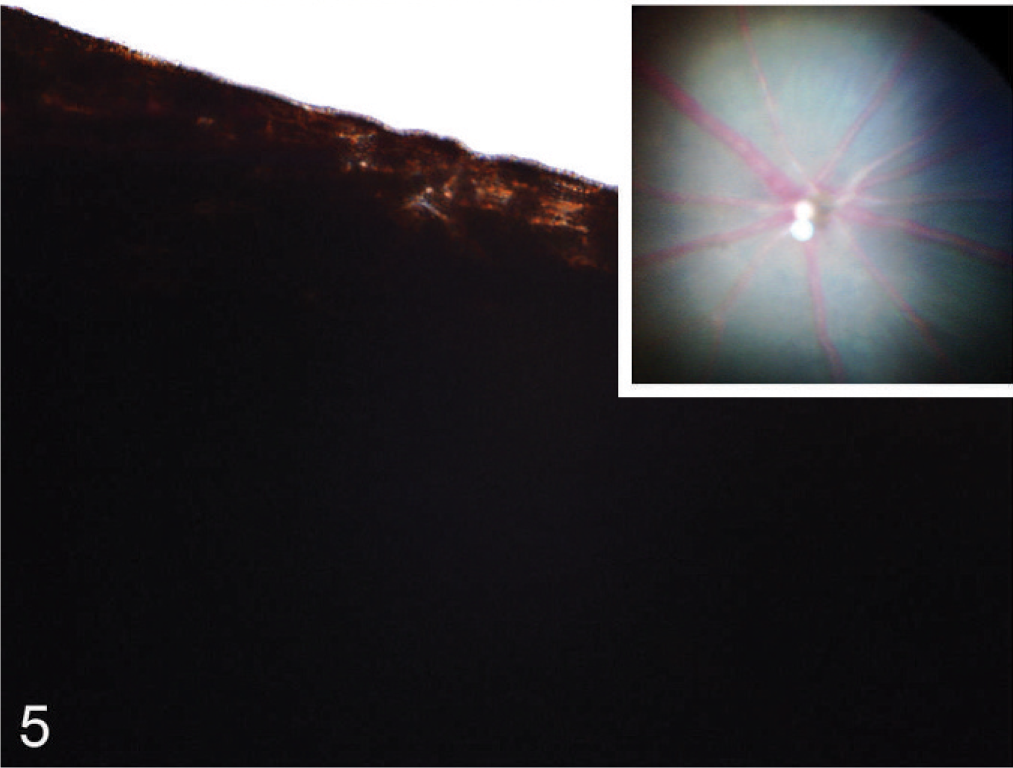
Iris; agouti Slc24a5+/+ mouse. Dense iridal pigmentation blocks the passage of light in a transilluminated iris. Inset: Radiating retinal vessels on a blue background of pigmented RPE, but only the largest choroidal vessels are visible as pink blotches. Funduscopic image.
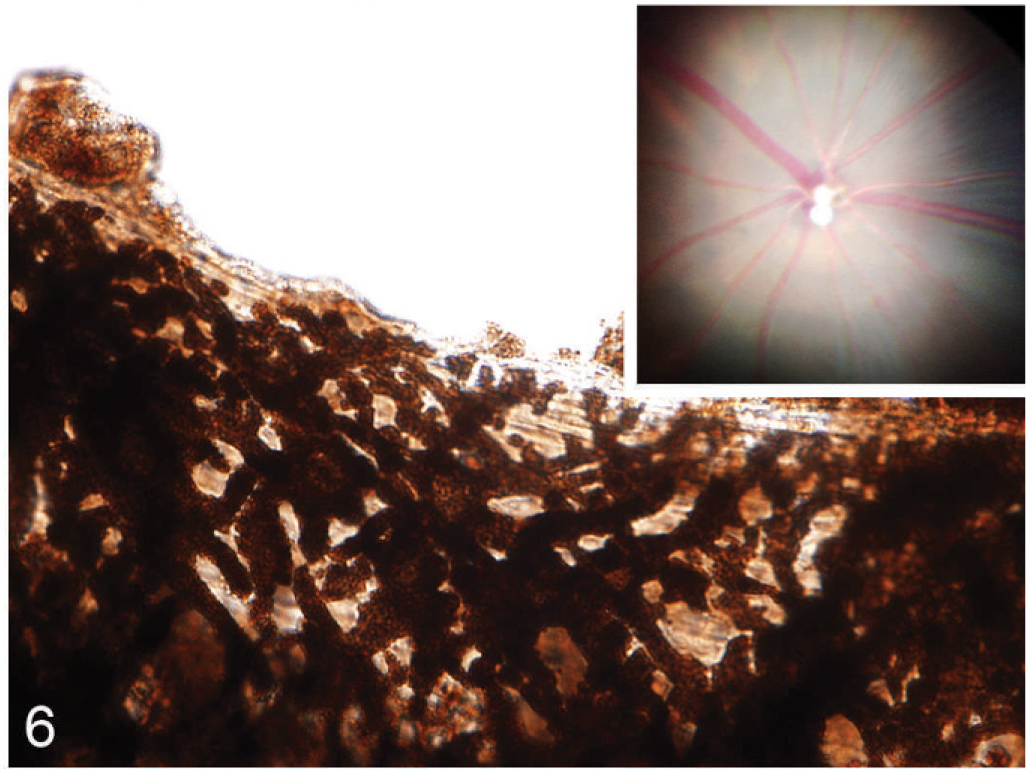
Iris; agouti Slc24a5−/− mouse. A markedly reduced amount of melanin pigmentation makes the irregular tubular cytoplasmic extensions of stromal melanocytes visible in a transilluminated iris. Inset: The reduced RPE pigmentation results in an albino-like fundus, with overall pink coloration and choroidal vessels are easily seen. Funduscopic image.
Microscopic examination of the eyes and skin showed that hypopigmentation was most prominent in the neuroepithelial pigmented cells, which included the retinal pigmented epithelium (RPE), the anterior and posterior layers of the iris pigment epithelium (IPE), and the ciliary body epithelium. Pigmented cells are present in all 3 layers of the iris, with neural crest-derived melanocytes that fill the loose fibrovascular stroma that makes up the anterior iris stroma, whereas, optic cup-derived neuroepithelial cells formed 2 cell layers of tightly fused, cuboidal cells that comprised the densely pigmented IPE. In normal agouti or black mice, all 3 layers of the iris were densely pigmented and difficult to delineate because of the numerous large melanin pigment granules filling the cytoplasm of each cell (Fig. 7). In contrast, the 3 iridal layers of the Slc24a5−/− iris were histologically distinct because of the markedly reduced volume, size, and darkness of melanosomes in all iridal layers (Fig. 8). The characteristic large round melanosomes of the posterior IPE were visible by light microscopy in wild-type mice (Fig. 7) but were almost entirely absent in Slc24a5−/− mice (Fig. 8). Notably, although melanin was reduced throughout the iris of null mice, hypopigmentation was most severe in the cells of the anterior layer of the IPE, which is sandwiched between the anterior iris stroma and thin posterior IPE cells (Fig. 8). By light microscopy, the melanosomes of the anterior IPE melanosomes appeared to be smaller, paler, and more indistinct than those of the iris stroma or posterior IPE. The marked hypopigmentation of neuroepithelium-derived cells was also evident in the ciliary body. In wild-type mice, the pigmented cells of the anterior layer of ciliary body epithelium blended imperceptibly with the adjacent pigmented choroidal melanocytes because of the similar coloration and high density of melanosomes in both cell types (Fig. 9). In marked contrast, in Slc24a5 null mice, the pigmented ciliary body epithelial cells were easily distinguished from choroidal melanocytes because of the lighter pigmentation of this cell layer in knockout mice (Fig. 10). Similarly, in the retina, there was diffuse hypopigmentation of the RPE. In agouti and black wild-type mice, round-to-oval melanosomes filled the basilar cytoplasm, whereas melanosomes at the apical interface with photoreceptors generally appeared as long thin granules that palisade perpendicular to the RPE cell surface (Fig. 11). In contrast, apical melanin granules appeared to be markedly reduced in size and number, and basal melanosomes were scarce in Slc24a5 null mice (Figs. 12, 13). Although the overall severity of ocular hypopigmentation in null mice varied between individuals, reduced melanization was evident in the neuroepithelium-derived pigmented cells of agouti and black Slc24a5−/− mice. The reduced pigmentation of choroidal melanocytes was more subtle and was most evident when tissue sections were directly compared side by side. No gross or histologic differences were detected between black and agouti wild-type mice, and the ocular hypopigmentation in the single black knockout mouse was indistinguishable from that noted in the agouti knockout mice.
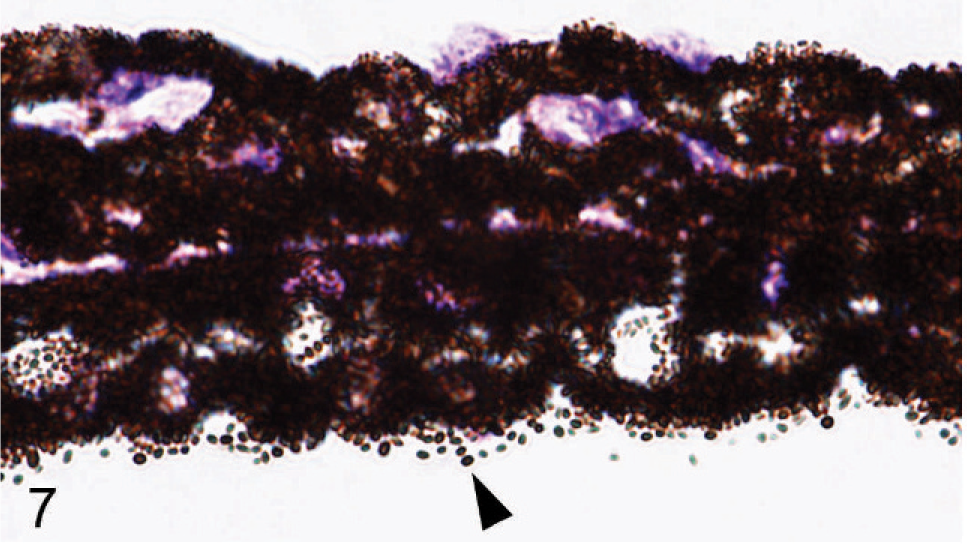
Iris; agouti Slc24a5+/+ mouse. All 3 layers of the iris are densely pigmented and difficult to delineate because of the numerous large pigment granules. Note the characteristic large round melanin pigment granules of the posterior iridal pigmented epithelium (arrowhead). HE.
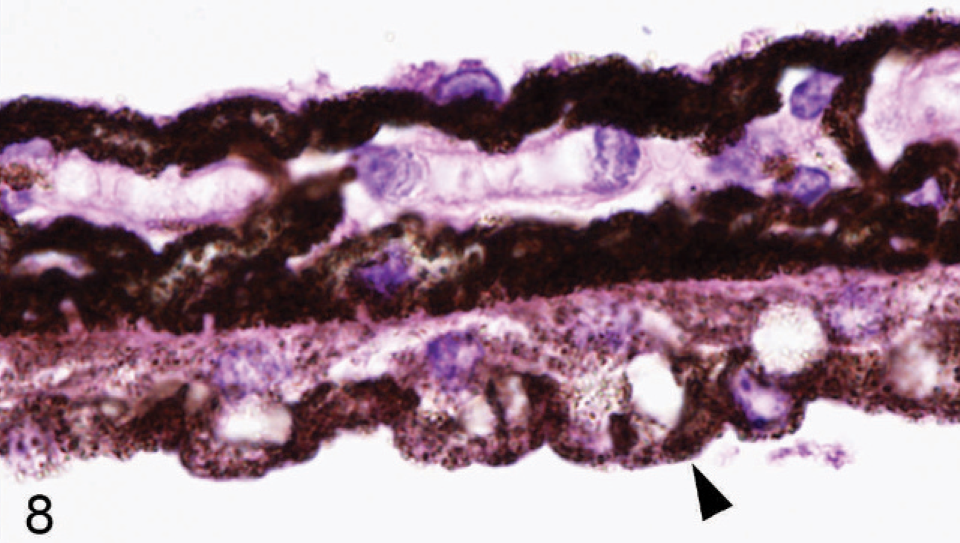
Iris; agouti Slc24a5−/− mouse. The 3 iridal layers of iris are easily identifiable because of the markedly reduced size and melanin content of pigment granules. Note the absence of large round melanin pigment granules in the posterior iridal pigmented epithelium (arrowhead) and that hypopigmentation is most severe in the anterior IPE layer. HE.
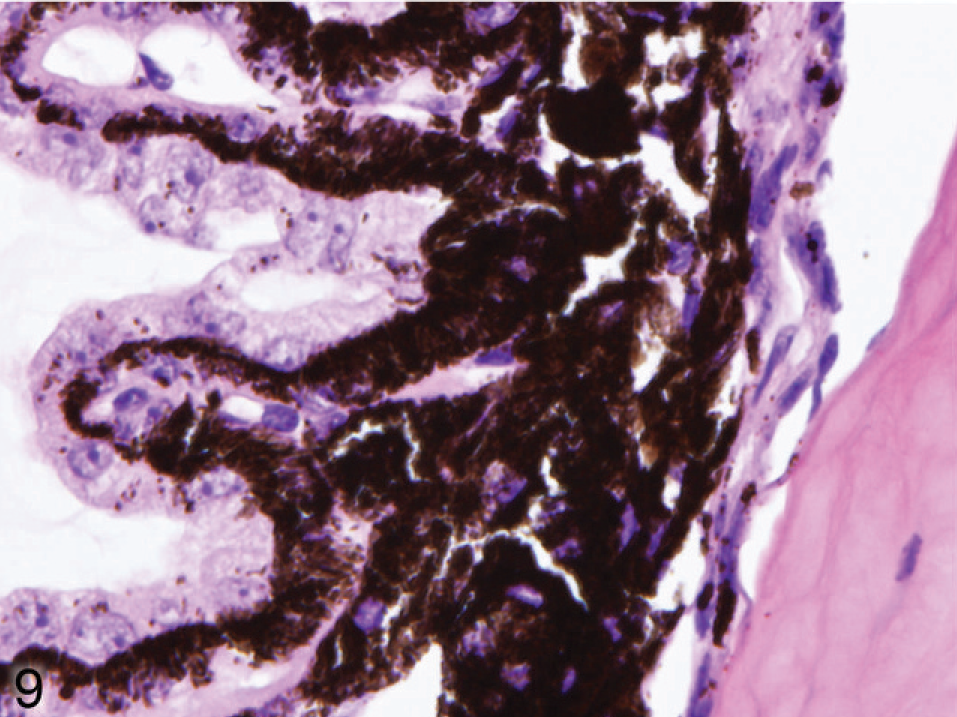
Ciliary body; agouti Slc24a5+/+ mouse. The cells of the anterior layer of ciliary body epithelium often blend imperceptibly with the adjacent pigmented choroidal melanocytes because of the similar size and pigmentation of melanin granules. HE.
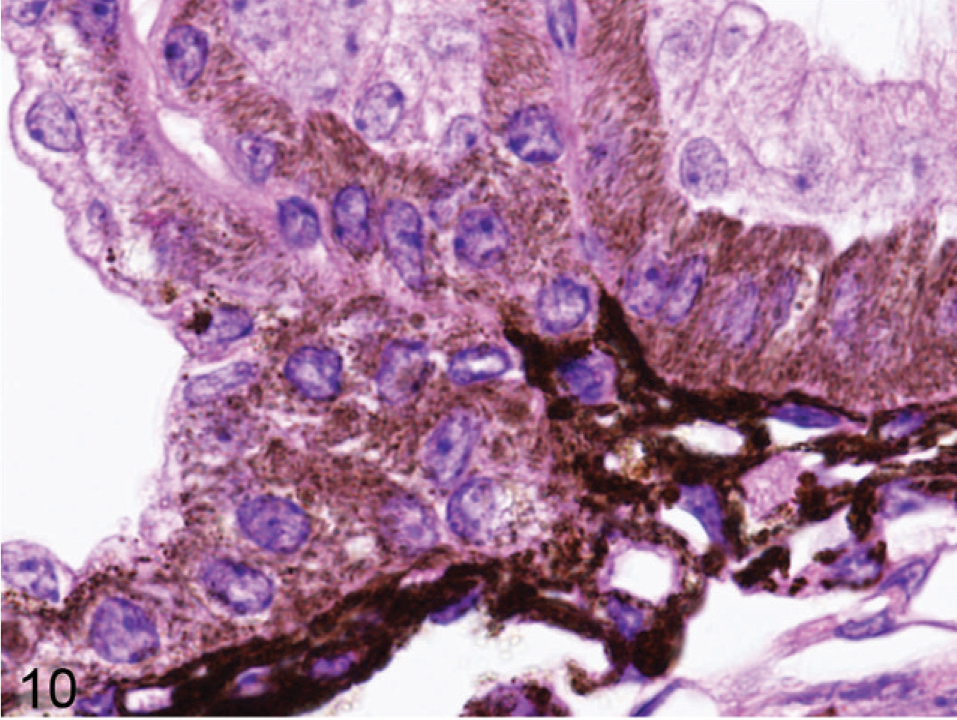
Ciliary body; agouti Slc24a5−/− mouse. The cells of the anterior layer ciliary body epithelial cells are easily distinguished from choroidal melanocytes because of their smaller, more lightly pigmented cytoplasm. HE.
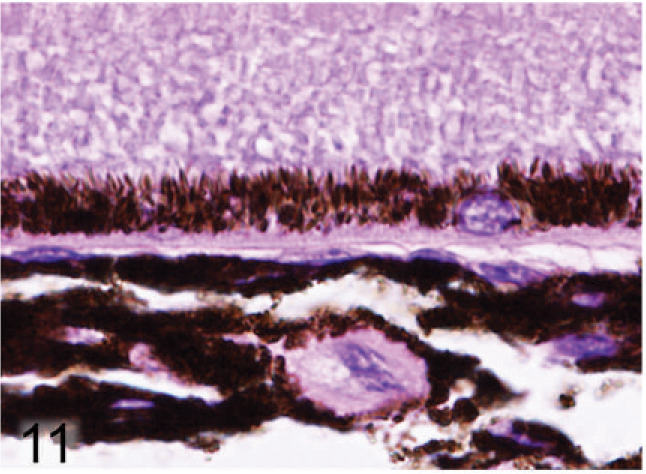
Retinal pigmented epithelium and choroidal melanocytes; agouti Slc24a5+/+ mouse. Round-to-oval melanin granules fill the basilar cytoplasm, while at the apical interface with photoreceptors, elongated melanin granules generally line up in rows perpendicular to the RPE cell surface. HE.
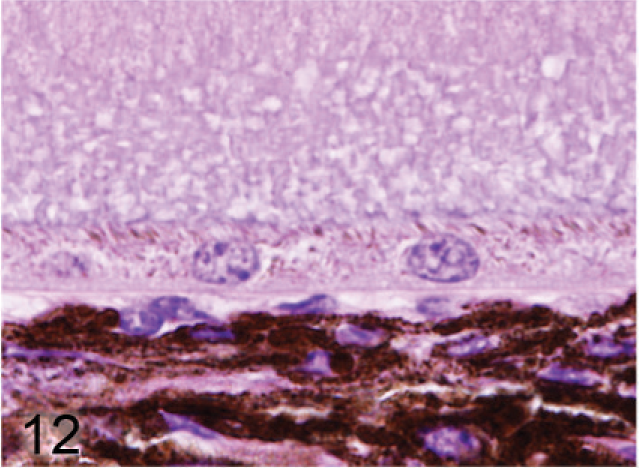
Retinal pigmented epithelium and choroidal melanocytes; agouti Slc24a5−/− mouse. Pigment granules are barely visible in the RPE and the size and pigmentation of melanin granules in the choroid is noticeably reduced. HE.
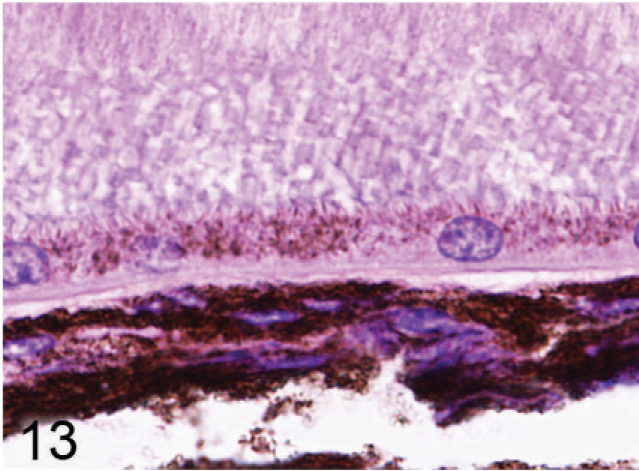
Retinal pigmented epithelium and choroidal melanocytes; agouti Slc24a5−/− mouse. Hypopigmentation is less severe in this null mouse, but the size and melanization of pigment granules are reduced in comparison to wild-type control tissues. HE.
Tissue distribution of β-Gal
A lacZ reporter gene is controlled by the endogenous Slc24a5 promoter in our Slc25a5−/− mice. Eyes from Slc24a5−/− mice were prepared and stained by using a
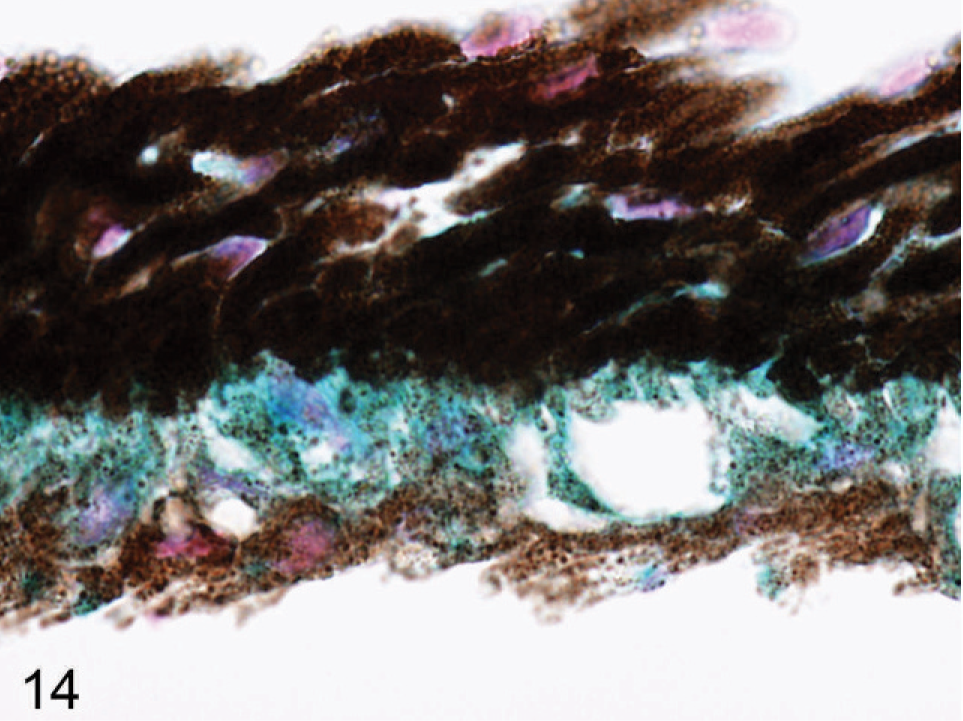
Iris; agouti Slc24a5−/− mouse. Positive signal that represent endogenous promoter activity was detected in all pigmented cells in the eye, but highest levels are in the anterior IPE (blue). X-gal stain with nuclear fast red counterstain.
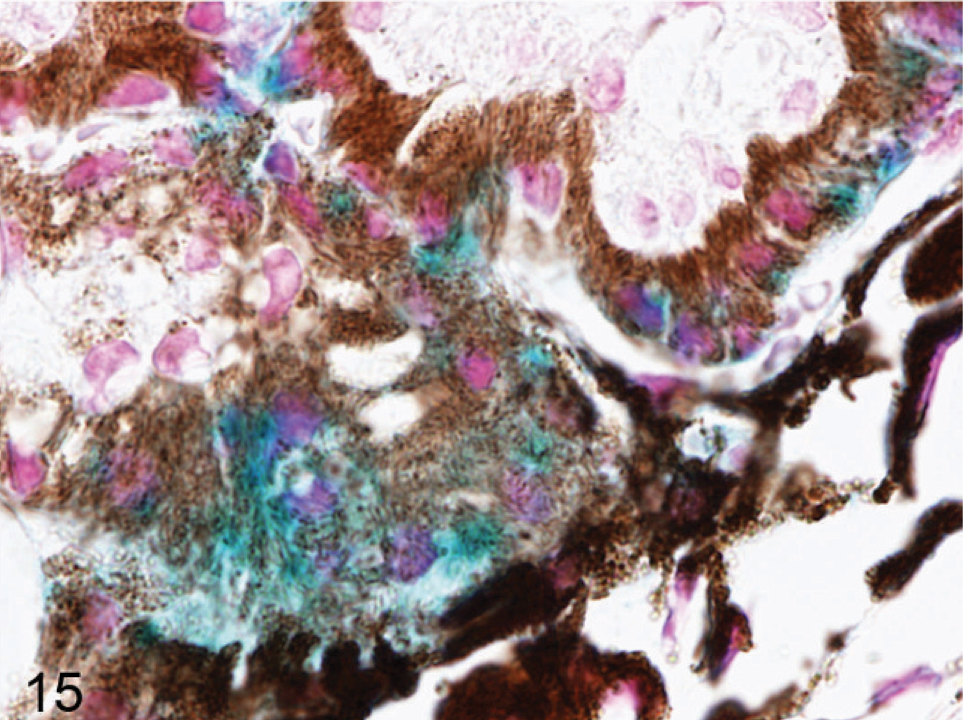
Ciliary body; agouti Slc24a5−/− mouse. Strong positive signal representing endogenous promoter activity was detected in the anterior pigmented cell layer of the ciliary body (blue). X-gal stain with nuclear fast red counterstain.
Electron microscopy findings
Ultrastructural analysis of the pigmented cells of the RPE, IPE, iris stroma, and ciliary body supported and extended the light microscopy findings. Melanosomes in Slc24a5−/− mice were reduced in size and electron density in all pigmented layers of the eye compared with the wild type. In the RPE of Slc24a5−/− mice, there was a clear reduction in the volume of melanosomes, which tended to be smaller and less electron dense than that of wild-type mice (Figs. 16, 17). The differences in melanization were also striking in the irides, where electron microscopy documented a marked reduction in overall thickness because of decreased melanization of all 3 layers of the iris of Slc24a5−/− mice in comparison with wild-type mice (Figs. 18, 19). The reduced thickness of the irides in Slc24a5−/− mice appeared to result more from reduced cell volume because of the smaller, less electron-dense melanosomes than from a reduction in melanosome abundance. The ultrastructural features of melanosomes in the irides of Slc24a5−/− mice differed significantly from those of wild-type mice in all 3 layers of the irides, but the reduction in size and pigmentation of melanosomes was most prominent in the anterior layer of the IPE of knockout mice (Figs. 18, 19). The reduced size and electron density of iridal melanosomes appeared to be at least partly because of deficient melanization. Melanosome development has been divided into 4 stages based on the degree of organelle maturation and melanization. The rarely identified stage I melanosomes represent the early stages of matrix organization. In stage II eumelanosomes, the matrix is well organized but not yet melanized, whereas, in stage III eumelanosomes, various amounts of melanin are deposited on internal structures. Mature stage IV melanosomes are fully melanized, and the matrix is no longer visible.
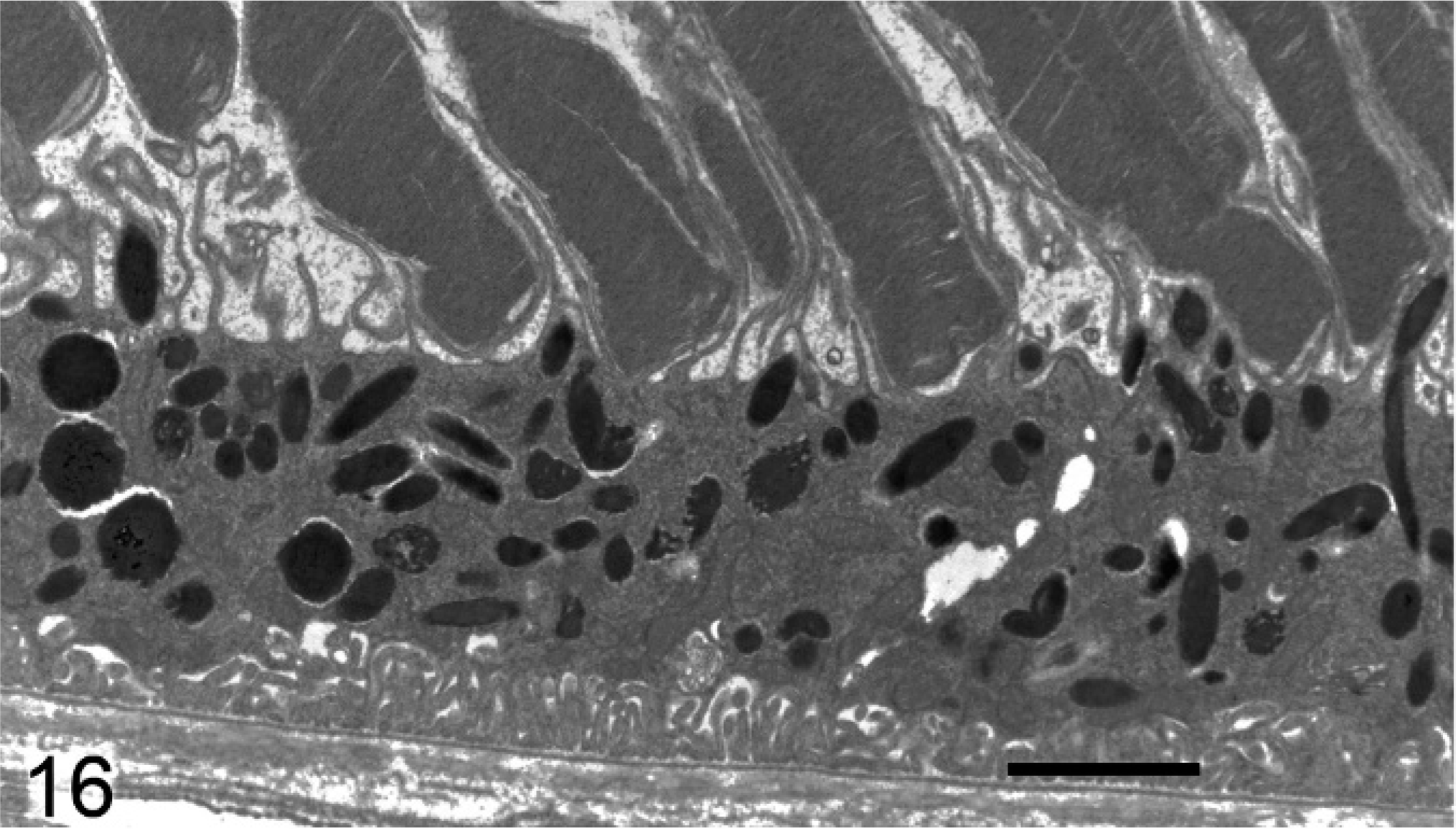
Retinal pigmented epithelium; agouti Slc24a5+/+ mouse. Abundant large electron dense melanosomes are present in the RPE. Transmission electron microscopy. Bar = 2 μm.
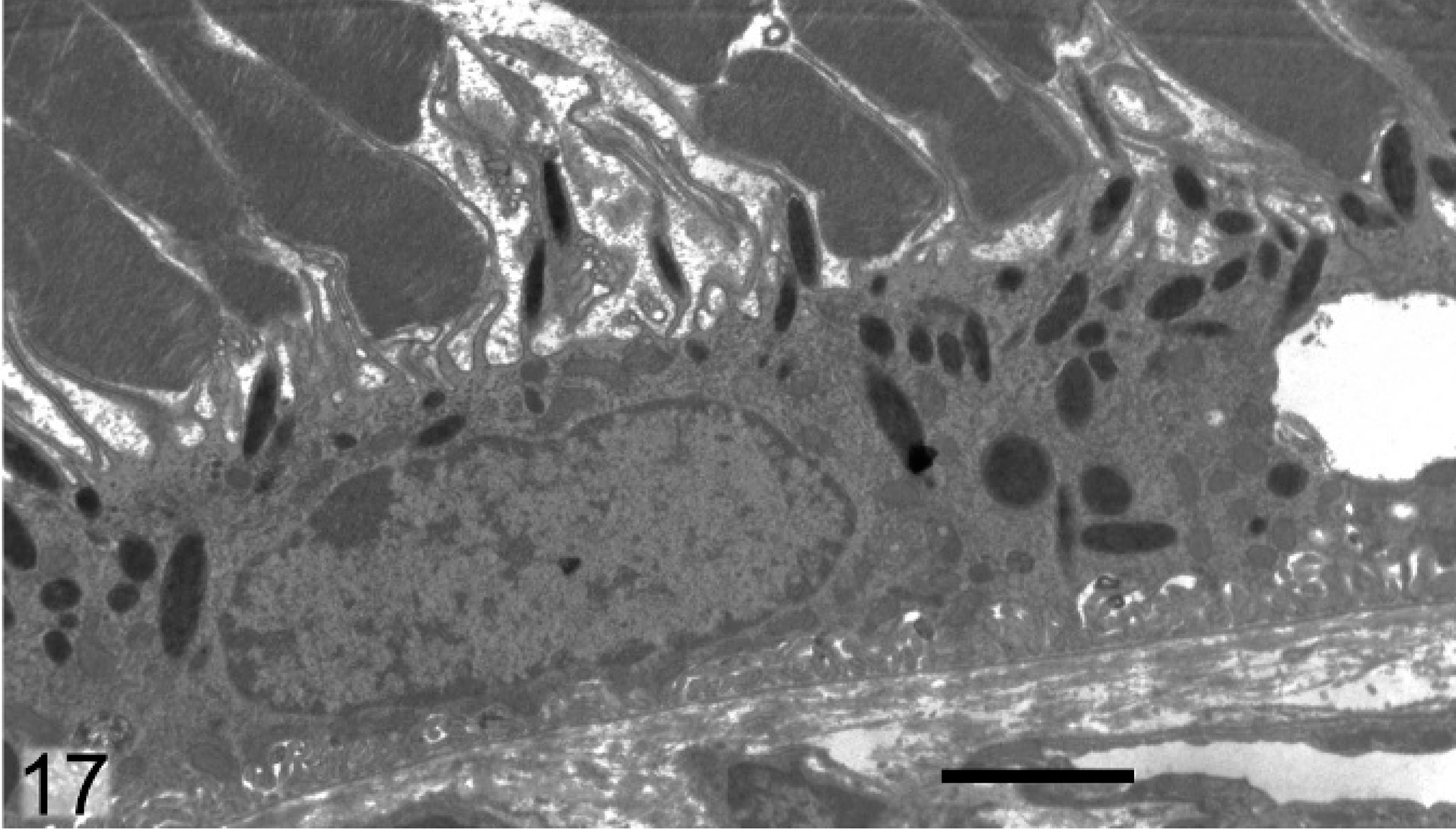
Retinal pigmented epithelium; agouti Slc24a5−/− mouse. Melanosomes are generally smaller and many are less electron dense. Transmission electron microscopy. Bar = 2 μm.
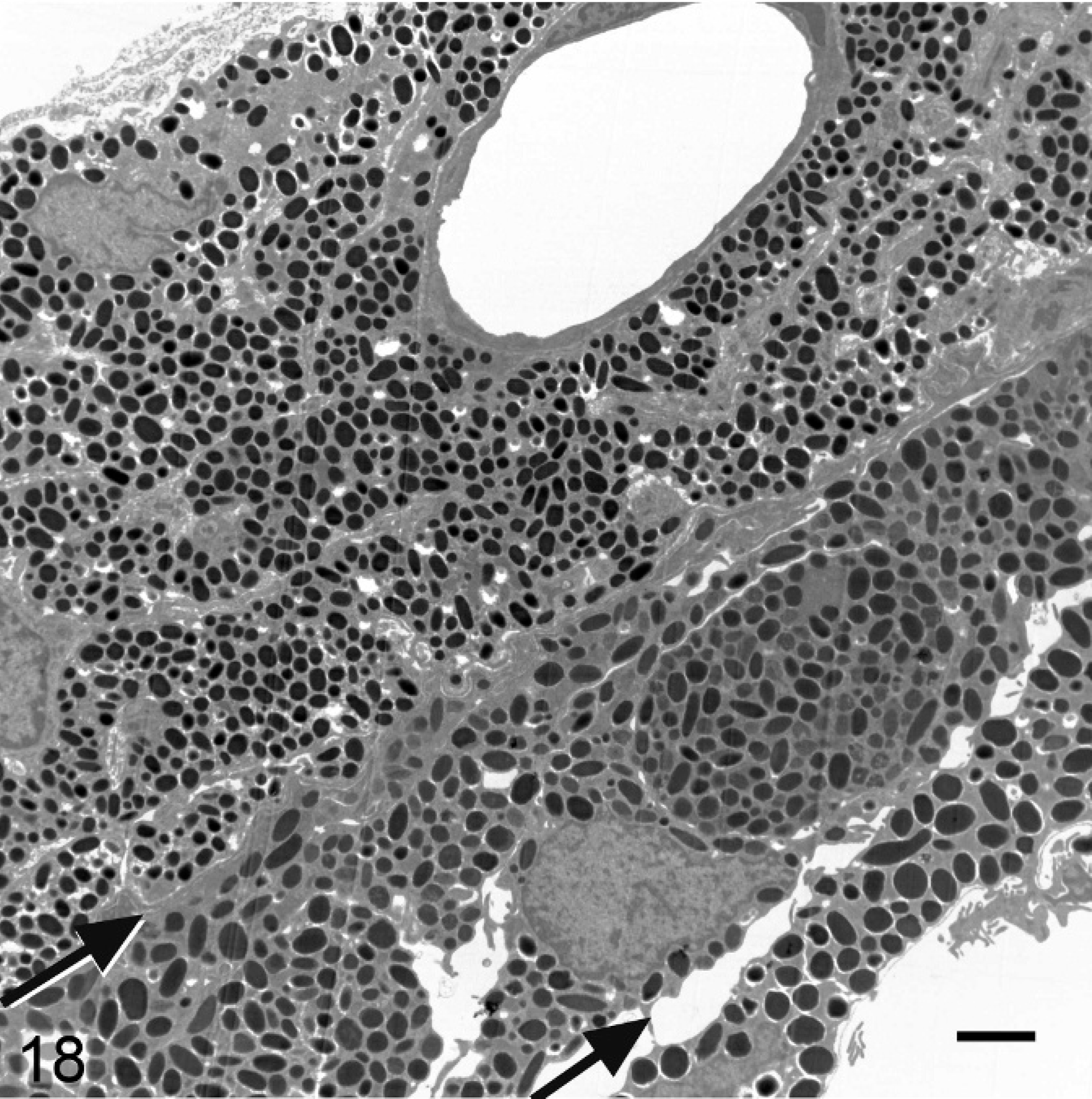
Iris; agouti Slc24a5+/+ mouse. The 3 layers of the iris, with margins indicated by arrows from upper left to lower right, comprise the iridal stroma, anterior IPE, and posterior IPE. Transmission electron microscopy. Bar = 2 μm.
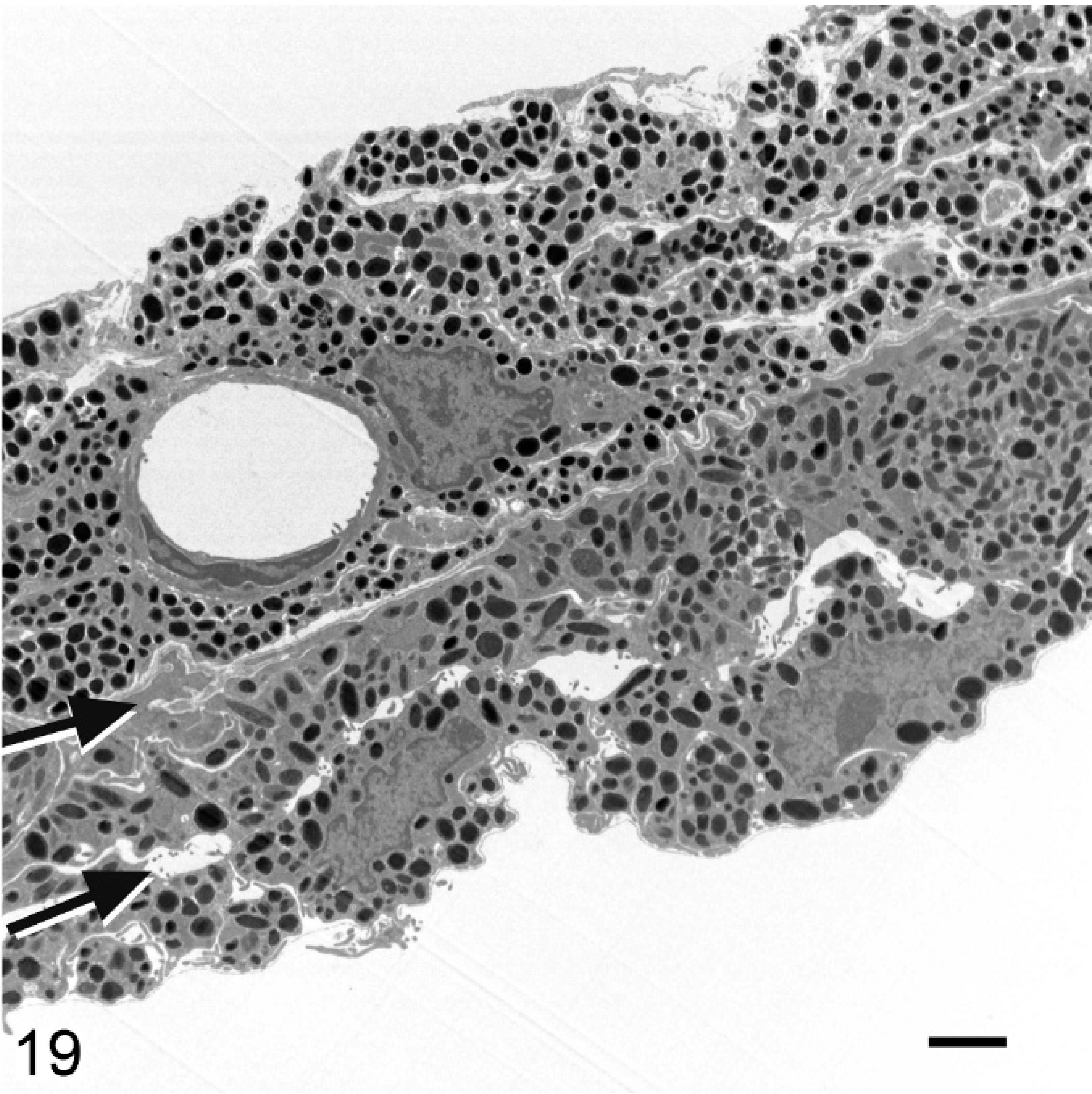
Iris; agouti Slc24a5−/− mouse. The reduced thickness of the iris of Slc24a5−/− mice is because of the reduced size and pigmentation of melanosomes. Hypopigmentation is most severe in the anterior layer of the IPE (layer between arrows). Transmission electron microscopy. Bar = 2 μm.
In wild-type mice, the stromal melanocytes in the anterior region of the iris were filled with densely packed, round-to-oval stage IV melanosomes (Fig. 20). In contrast, in stromal melanocytes of Slc24a5−/− mice, the melanosomes were typically smaller, less electron dense, and often incompletely melanized (Fig. 21). Many melanosomes in stromal melanocytes of Slc24a5−/− mice had densely melanized cores and comparatively pale peripheral zones, giving them a targetoid appearance (Fig. 21). A few incompletely melanized stage III melanosomes were also present in stromal melanocytes in the knockout mice.
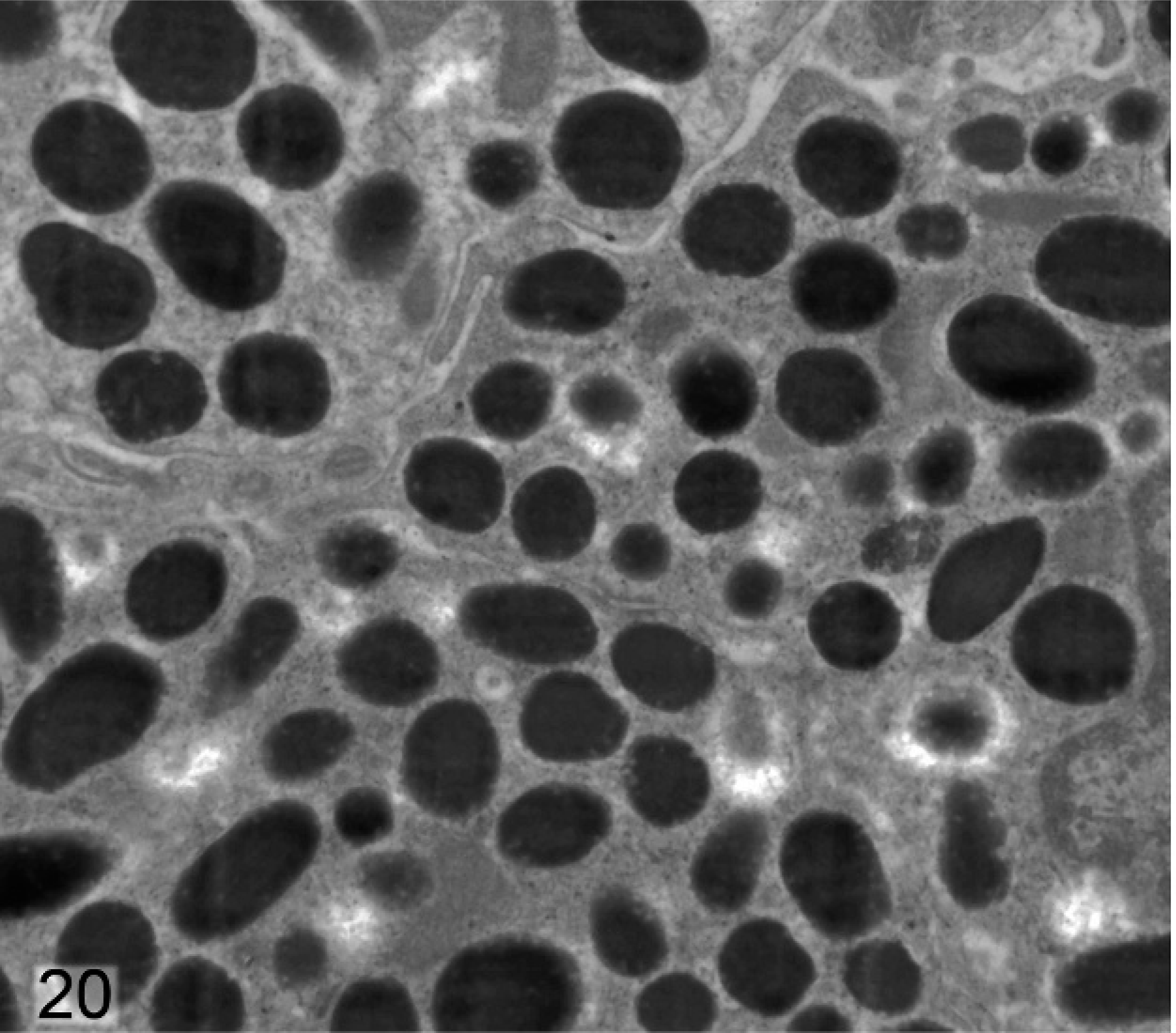
Iris stromal melanocytes; agouti Slc24a5+/+ mouse. Wild-type stromal melanocytes contain densely packed, round-to-oval stage IV melanosomes.
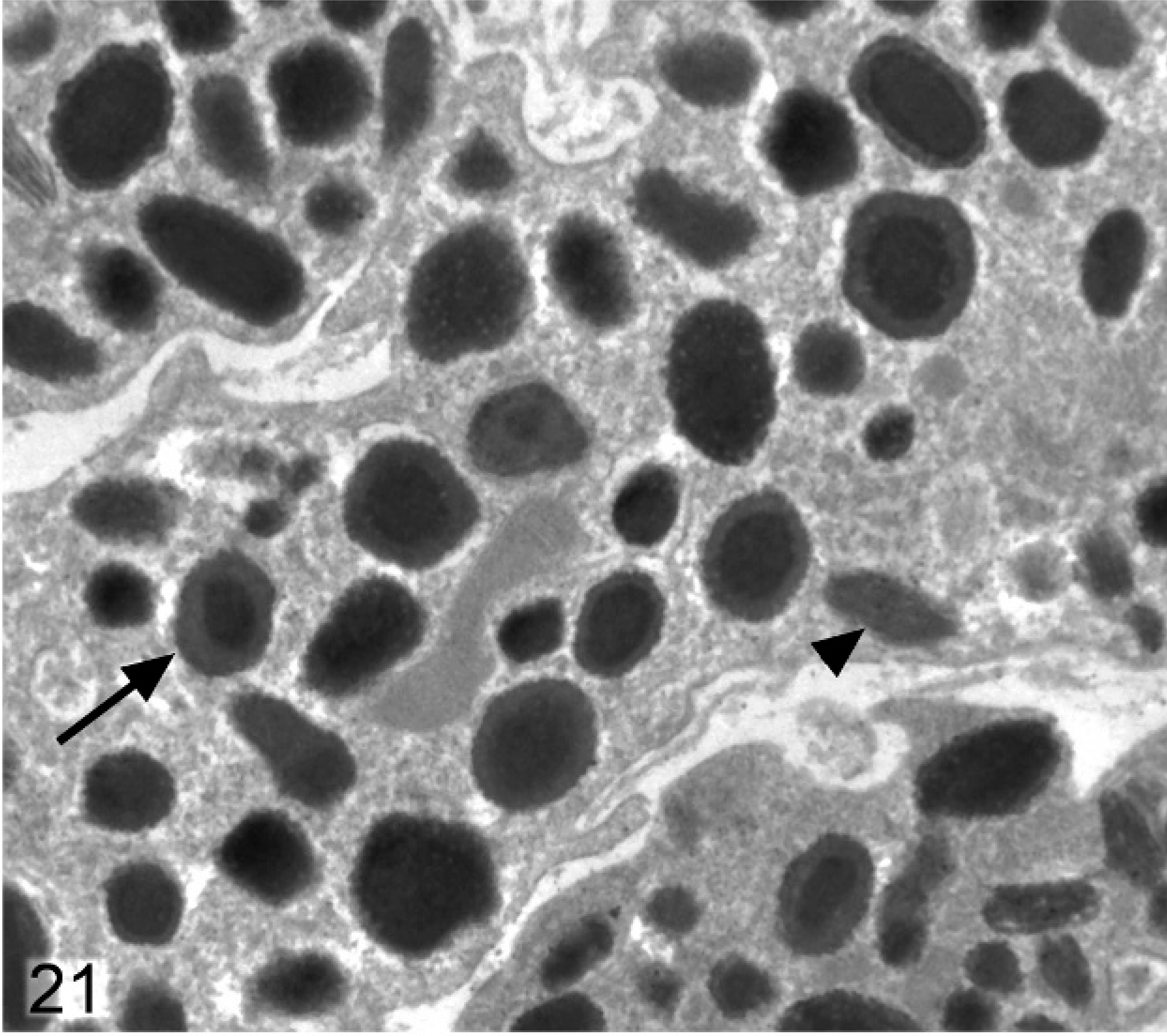
Iris stromal melanocytes; agouti Slc24a5−/− mouse. Stromal melanocytes contain smaller and irregular, less electron dense, and often incompletely melanized stage IV melanosomes. Some melanosomes have densely melanized cores and relatively pale peripheral zones, giving them a targetoid appearance (arrow), and a few incompletely melanized stage III melanosomes (arrowhead) are also present.
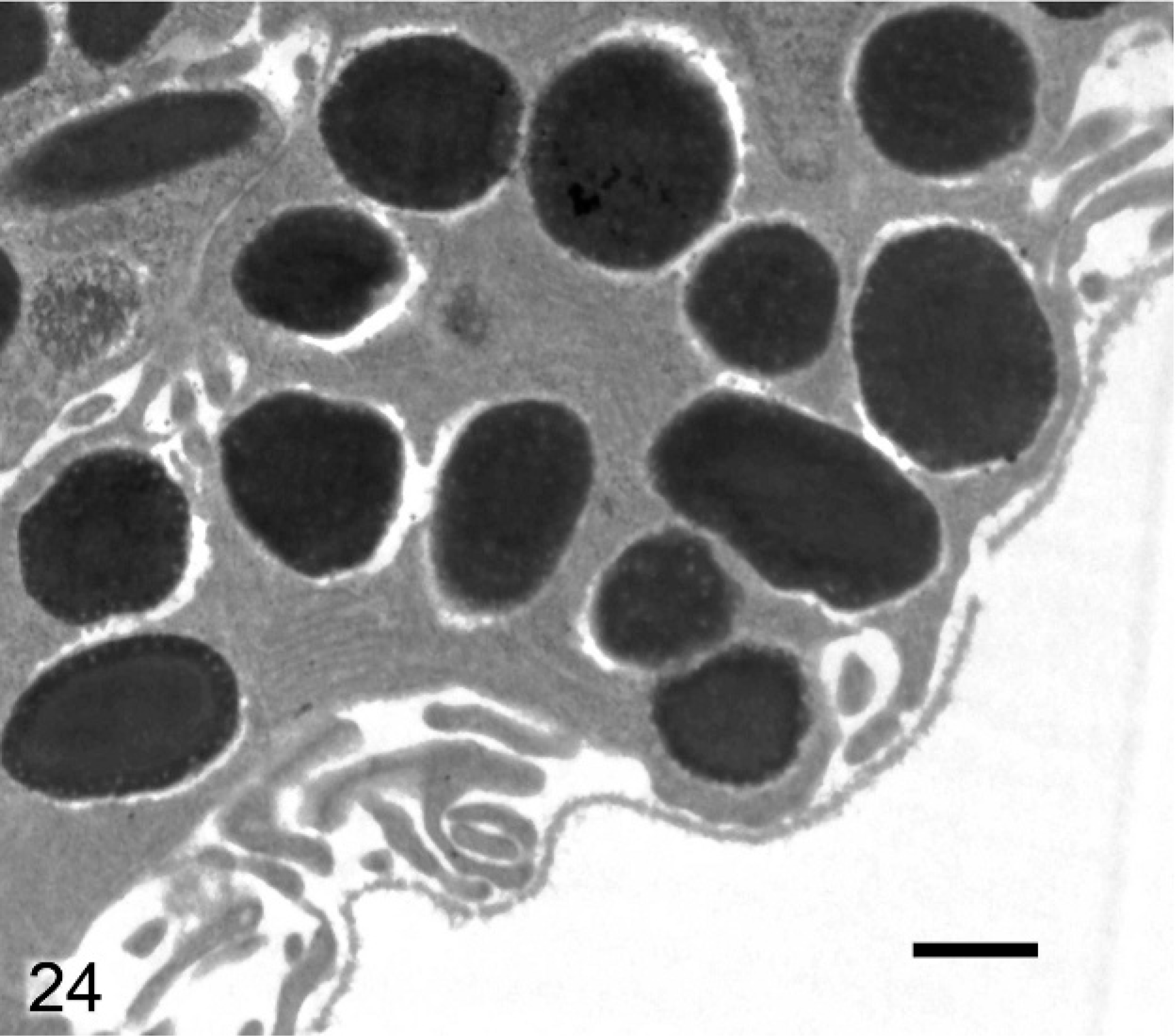
Differences between melanosomes of wild-type and knockout mice were even more striking in the anterior IPE (Figs. 22, 23). Anterior IPE cells of wild-type mice had a mixture of round and ovoid fully melanized stage IV melanosomes (Fig. 22), which were generally larger than those found in the stromal melanocytes. In marked contrast, the numerous melanosomes in the anterior IPE of Slc24a5−/− mice were generally smaller and incompletely melanized (Fig. 23). Numerous stage III and even some stage II (nonmelanized) melanosomes were present in the anterior IPE of Slc24a5−/− mice (Fig. 23). In wild-type mice, the third layer of the iris (the posterior IPE) contained numerous, uniformly dense, black, and generally round melanosomes (Fig. 24); even the ovoid melanosomes of the posterior IPE tended to be larger and wider than those found in the other 2 layers of the iris. Melanosomes in the posterior IPE of the Slc24a5−/− mice also tended to be smaller and electron lucent than in wild-type counterparts; numerous incompletely melanized small fragments and targetoid melanosomes were present (Fig. 25) in this layer. Overall, these results suggested that loss of gene function resulted in impaired maturation and melanization of melanosomes and that the defect was more prominent in the pigmented cells of the anterior IPE than in the posterior IPE and was least prominent in the neural–crest derived iridal stromal melanocytes.
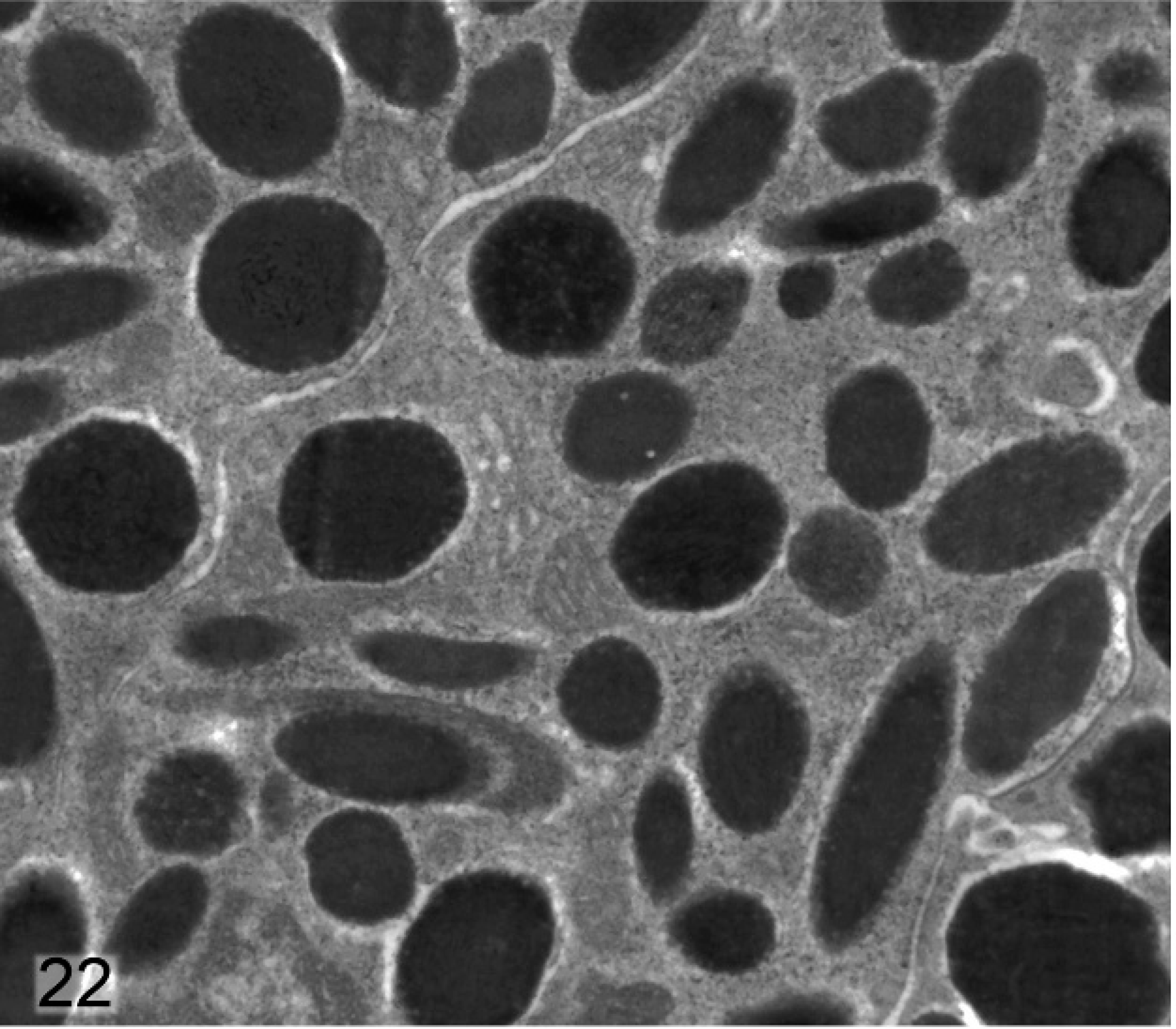
Anterior iris pigmented epithelium; agouti Slc24a5+/+ mouse. The round-to-ovoid fully melanized stage IV melanosomes in this layer of the iris are generally larger than those within stromal melanocytes.
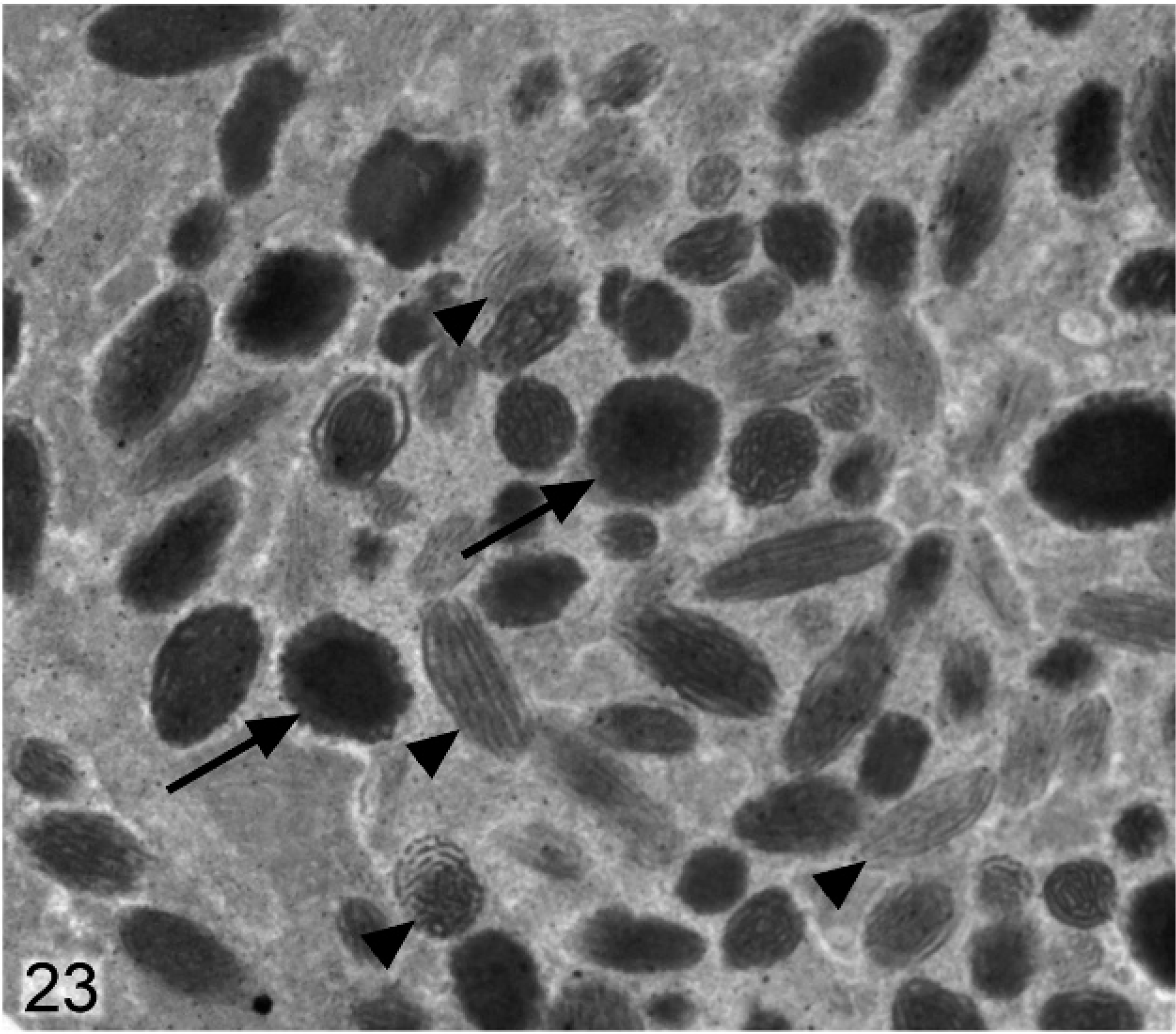
Anterior iris pigmented epithelium; agouti Slc24a5−/− mouse. In general, melanosomes in this layer are smaller and more irregular, with some incompletely melanized targetoid forms (arrow) and numerous incompletely melanized stage III and nonmelanized stage II melanosomes (arrowheads) present.
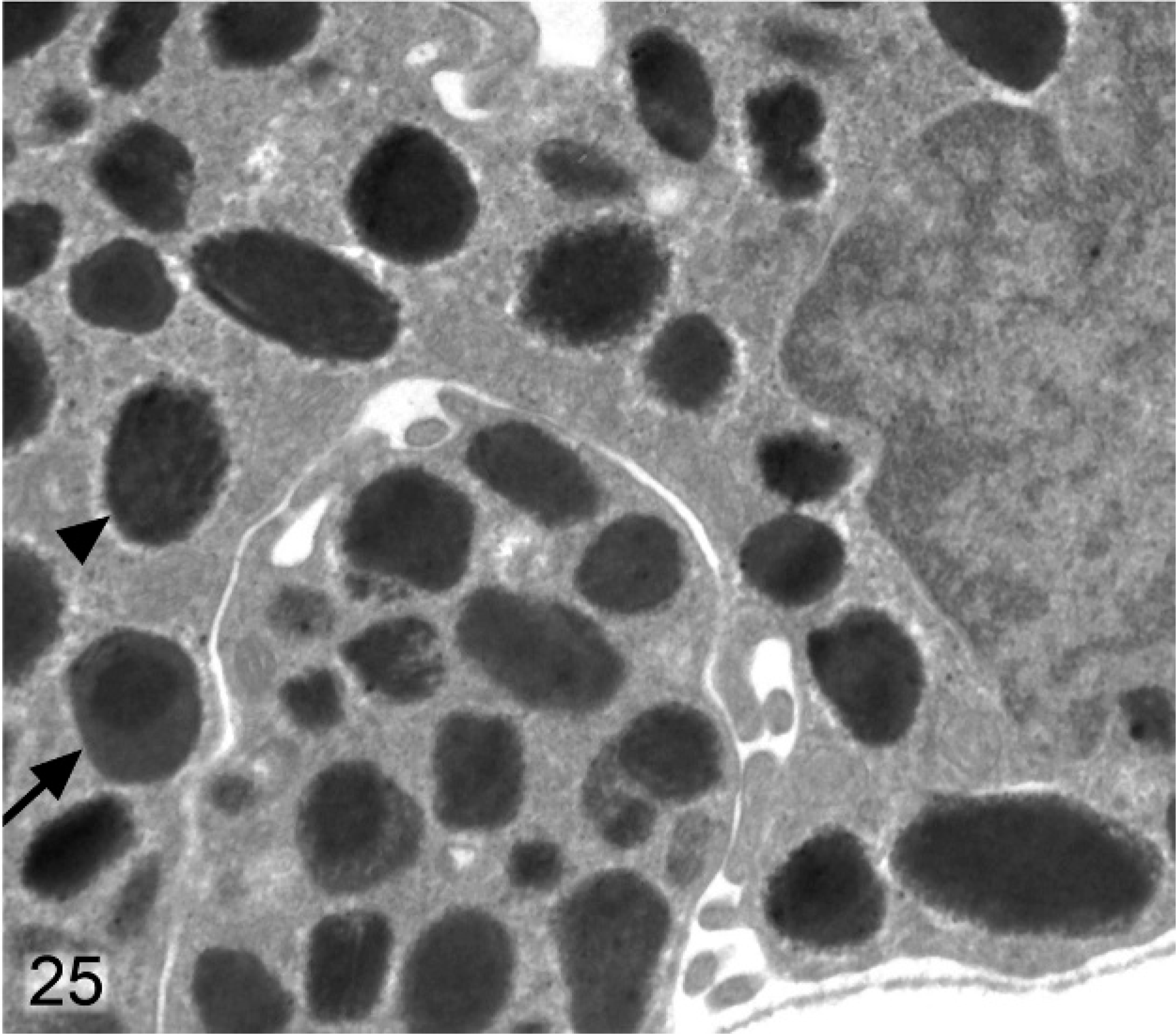
Posterior iris pigmented epithelium; agouti Slc24a5−/− mouse. Melanosomes tend to be smaller than normal (arrowhead), and irregular and incompletely melanized targetoid melanosomes (arrow) are also present.
Discussion
This study was designed to determine the phenotypic effects induced by disrupting the Slc24a5 gene in mice. Our findings indicate that Slc24a5 is involved in melanin biosynthesis in all pigment-producing cells, and we noted changes in melanosome number, size, and type within the integument and the eyes of Slc24a5−/− mice. However, the more severe hypopigmentation of the RPE, IPE, and pigmented epithelial cells of the ciliary body indicated that normal Slc24a5 gene function was especially important in the melanization process within the neuroepithelium-derived pigmented cells of the eye. As well, the most intense reporter gene activity was found in the anterior IPE, which correlated well with the area of most pronounced hypopigmentation and melanosomal abnormalities. We found that Slc24a5−/− mice developed hypopigmentation, which most severely affected the optic cup neuroepithelium-derived cells of the eye.
A role for Slc24a5 in pigmentation of mice is not unexpected given the demonstrated importance of this gene in melanization processes in both zebrafish and humans. 21 The human ortholog of zebrafish golden Slc24a5 is located on chromosome 15q21.1 and encodes a 500 amino acid protein (SLC24A5 or NCKX5), which is a member of the potassium-dependent sodium-calcium exchanger family. High levels of calcium were reported in pigmented tissues of the vertebrate eye, such as RPE and melanin granules that contained high calcium concentrations. 33 Because SLC24A5 has been localized to melanosomes 5 and appeared to be involved in calcium uptake into these organelles, 21 it seemed likely that the loss of SLC24A5 function would preferentially affect melanization in ocular structures.
Although mutations in two other genes also located within band 15q21 (RAB27A and MYO5A) result in hypopigmentation disorders in humans, they are invariably associated with the silver hair and immunologic deficits of Griscelli disease 2 that were not present in our Slc24a5−/− mice. 27 The SLC24A5 protein was localized to melanosomes 5 and appeared to be involved in calcium uptake into these organelles. 21 Mutations of slc24a5 in golden zebrafish resulted in hypopigmentation of skin melanophores and RPE. 21 The melanophores of golden zebrafish are thinner and contain fewer melanosomes than normal. Moreover, the melanosomes in golden zebrafish are smaller, less electron dense, and more irregularly shaped than those of wild-type zebrafish. 21 The melanosome was also the critical organelle responsible for pigmentation differences in humans. Interestingly, most of the variation in human eye and skin color is not because of differences in the numbers and density of melanocytes 36 but is instead determined by differences in the number, size, distribution, and type of melanosomes carried by melanocytes. 19, 21
The more complete melanization of neural crest–derived melanocytes in the skin and parts of the eye, along with the normal appearance of the coat color in our mice, suggested that Slc24a5-dependent impairment of melanization was not global. Although coat color variations in mice are generally easy to identify, it is more difficult to assess differences in skin pigmentation because pigmented cells in mouse skin are usually restricted to the hair follicles and in scattered foci on the ears, nose, perineum, footpads, and tail. Nevertheless, the outer ears of Slc24a5−/− mice were visibly paler than those of wild-type litter mates, and we confirmed, by light microscopy, that reduced amounts of melanin pigment were in the dermis and epidermis of the ears and nose in Slc24a5−/− mice. The smaller size, reduced number, and hypomelanization of pigment granules in the skin and hair of Slc24a5−/− mice were consistent with findings in golden zebrafish and some groups of humans with paler skin. Interestingly, the coat colors of non-albino Slc25a5−/− mice were virtually indistinguishable from those of wild-type litter mates. As in other pigmented cell types, the individual pigment granules in the medullary hair cells in Slc24a5−/− mice tended to be smaller, paler, and more irregular in shape than those of wild-type litter mates. However, the decreased size and pigmentation of melanosomes in hairs coincided with increased numbers of melanin granules being packed into each individual hair cell, resulting in a normal overall appearance and color of hairs.
Although changes in skin pigmentation were detected, the most obvious and severe phenotypic differences between wild-type and Slc24a5−/− mice were present in the eye and presented as OA. Hypopigmentation of the fundus was evident on ophthalmoscopic examination of Slc24a5−/− mice and transillumination of dissected irides clearly demonstrated the decreased pigment density in knockout mice. The 2 major classes of albinism that affect pigmentation in the eye have been defined as OCA and OA. The major difference between these types of albinism is that, in OCA, pigmented cells in both the integument and eyes are severely affected, whereas, in OA, the pigment of the hair and skin is normal or only slightly diluted. In the 4 major types of OCA identified to date, skin pigmentation phenotypes are accompanied by a variety of lesions that affect the pigmented cells of the eye. OCA1 is associated with mutations of the TYR gene, which encodes tyrosinase (the rate-limiting enzyme in the production of melanin pigment). OCA1 accounts for approximately 40% of OCA worldwide. The tyrosine-positive forms of OCA prove that TYR is not the only gene involved in iris pigmentation. OCA2 is the most common form of OCA and is associated with mutations of the mouse p gene. OCA3 is a rare form of OCA associated with mutations in TYRP1 (encoding tyrosinase-related protein 1). The mutated gene responsible for OCA4 encodes the membrane-associated transporter protein (MATP) and was first identified in the underwhite (uw) mouse. 25 Defective MATP causes abnormal processing and intracellular trafficking of the TYR protein to the melanosomes. 6 The phenotype of our Slc24a5−/− mouse fits better into the OA category given that a diffuse marked hypomelanization of neuroepithelium-derived cells in Slc24a5−/− eyes is coexistent with a relatively mild reduction in pigmentation of neural crest–derived melanocytes in the hair, skin, iris, and choroid. The recognized types of human OA include the X-linked recessive form (Nettleship-Falls type), which is called ocular albinism type 1 (OA1), and the less well-defined forms of autosomal recessive ocular albinism (AROA), which include OA2 and OA3. However, genetic analyses showed that many of the patients diagnosed with AROA actually represent mild cases of either OCA1 or OCA2, with 14% having abnormalities of the tyrosinase gene (making them OCA1), and 36% having abnormalities of the P gene (making them OCA2). 12 Although a genetic locus for OA in the vicinity of SLC24A5 in humans has not yet been identified, the demonstrated role of this gene in ocular pigmentation in zebrafish and now in mice suggests that it is involved in some forms of human ocular hypopigmentation or albinism.
Our findings also suggest that the overall reduced pigmentation was mainly because of defective melanization and, to a lesser extent, to reduced numbers of melanosomes. In all pigmented cells, the reduced number of large stage IV melanosomes was not associated with autophagosomes, which indicate that increased degradation of melanosomes was not responsible for the hypopigmentation. The different degrees of hypopigmentation we observed in the various pigmented cells of the eye are most likely linked to the differing embryologic origins of the affected cell types. The melanocytes in the iris stroma, ciliary stroma, and choroid develop from melanoblasts that migrate from the neural crest to the uveal tract and develop into melanocytes. In contrast, the RPE, IPE, and the outer pigmented and inner nonpigmented ciliary epithelium are all derived from the optic cup neuroepithelium. The IPE is formed by a 2-cell thick layer of fused, cuboidal, densely pigmented cells termed the posterior and anterior IPE. In normal agouti or black mice, both IPE layers are black because of the numerous large granular melanosomes that fill the cytoplasm of each cell. At the periphery of the iris, the IPE is continuous with the double-layered epithelium of the ciliary body, of which only the anterior layer is pigmented. The pigmented anterior layer of the ciliary epithelium is, in turn, continuous with the retinal pigmented epithelium.
In normal agouti or black mice, all 3 layers of the iris are densely black and difficult to delineate because of the numerous large granular melanosomes that fill the cytoplasm of each cell. In contrast, the relatively thin iridal layers of Slc24a5−/− mice appeared to contain reduced numbers of small pigment granules in the stromal melanocytes and IPE, especially in the anterior layer cells of the IPE. Electron microscopy confirmed that neuroepithelium-derived pigment cells of the eye in Slc24a5−/− mice contained large numbers of melanosomes with irregular borders that were smaller and less electron dense than normal. Incomplete melanization was most evident in the anterior IPE cells, where the reduced number and size of stage IV melanosomes was accompanied by a marked increase in stage-II and stage-III melanosomes. The more severe hypomelanization of neuroepithelium-derived cells of the RPE/IPE compared with the neural crest–derived melanocytes of the iris stroma and choroid correlated with the most intense reporter gene activity we detected with X-gal staining. The more severe hypomelanization of optic cup neuroepithelium-derived pigmented cells is likely to result from differences in the regulatory networks that control melanin pigment production in these 2 classes of pigmented cells. 22– 24 Interestingly, the only neuroepithelial-cell layer that maintained near-normal pigmentation density in the Slc24a5 knockout mice was the posterior IPE, which suggested that these cells used some of the same alternative pathways for melanosomal Ca++ transport and melanization as neural crest–derived melanocytes.
The hypopigmentation that we observed histologically in the iris might be expected to influence eye color. Eye color is determined by the amount and wavelengths of light that are absorbed, scattered, and reflected in the iris. The number of melanocytes in the iris is relatively constant, regardless of eye color, 1, 43 which instead depends primarily on variations in the amount, type, and distribution of melanin pigment in the iridal melanocytes. Of these factors, the absolute amount of melanin is of primary importance in determining eye color. 14, 29 Given its clear role in the melanization process, SLC24A5 would presumably have its greatest effect on eye color by influencing the amount and extent of stromal melanization. In blue eyes, melanin pigment is virtually absent in melanocytes within the anterior border layer and stroma of the iris. The eyes appear blue because shorter wavelengths of light (blue) are scattered and reflected (via the Tyndall effect) by the nonpigmented tissues and molecules of the iris stroma. At the same time, the longer wavelengths of light (red to yellow) readily penetrate the nonpigmented stroma of blue eyes but are absorbed by the densely pigmented IPE, which acts as a light sink. In contrast, in brown or black irides the melanocytes in anterior border layer and stroma are densely packed with melanin granules that absorb most of the incident and backscattered light. Other eye colors contain intermediate amounts of melanin pigment within anterior border and stromal melanocytes. 14
Eye color is also influenced by the type of the melanin pigment produced by stromal melanocytes. Mammalian melanins exist in 2 chemically distinct forms: 1) eumelanin, which is a brown-black pigment packaged in ellipsoid eumelanosomes that contain fibrillar matrix; and 2) pheomelanin, which is a red-yellow pigment packaged in round pheomelanosomes in a vesiculoglobular matrix. 15, 16, 26 Eumelanin is the predominant pigment in blue and green-blue mixed-color irides, whereas, pheomelanin is a major pigment type in green irides. Brown and green-brown mixed-color irides contain a mixture of eumelanin and pheomelanin. 29 Although melanin granules were generally smaller and irregular in Slc24a5−/− mice, most were ultrastructurally identifiable as eumelanosomes, and we did not detect any shift from eumelanin to pheomelanin that might influence eye color. The small size and thin irides of the mouse eye result in a severely limited range of possible eye colors in mice, an interesting biological phenomenon that contrasts with the many different coat color variations. Mouse eye colors, which range from pink to ruby to brown-black, correlate with increasing amounts of melanin pigment in ocular structures. In albino mice, eyes lack melanin pigment in all layers and are pink. Similarly, agouti mice with mutations at the mouse pink-eyed dilution locus (pgen, the ortholog of human OCA2) 4 have minimal amounts of melanin pigment in ocular structures and consequently have pink eyes. Ruby-eyed mice, which have mutations in genes orthologous to those that cause Hermansky-Pudlak syndrome in humans, have intermediate levels of ocular pigmentation. 46 All agouti Slc24a5−/− mice had dark brown to black eyes that were indistinguishable from those of wild-type litter mates. Although the decreased iridal pigmentation was detectable by transillumination of dissected irides, apparently there was sufficient melanin pigment present in the eyes of Slc24a5−/− to produce essentially normal eye color under normal lighting conditions.
The severe hypopigmentation and high levels of Slc24a5 promoter activity that we noted in the anterior IPE suggested that Slc24a5 played a more important role in melanization in this cell layer than in the posterior IPE. In vertebrates, the IPE consists of 2 densely pigmented monolayers of neuroepithelial cells that are apposed apex to apex, and tightly fused by intercellular connections. Normally, in both humans and mice, the 2 layers of the IPE are maximally pigmented and act as a light sink at the back of the iris and thus are not believed to affect eye color. 8 We hypothesize that SLC24A5-dependent hypopigmentation of the anterior IPE in humans might prevent this layer from acting as an efficient light sink and that the resulting backscatter of incident light from the more lightly pigmented anterior IPE would influence iris color. Hence, it is possible that SLC24A5-dependent differences in pigmentation density between the anterior and posterior layers of the IPE may contribute to some eye color variation in humans.
Indeed, genetic studies support a possible role for SLC24A5 in influencing human eye color. The genetic basis for eye color in humans is now known to be quite complex; rather than a simple dominance of one allele over another, eye color is the result of a developmental process that involves many genetic interactions. At least 3 genes have been inferred from genetic linkage analysis to explain the many variations in human eye color. 9 The first of these, the EYCL1/GEY gene is located on chromosome 19 and is associated with green or blue eye color. A second important eye color gene, EYCL3/BEY2, was originally mapped to chromosome 15q11–15q21. 9 More recently, EYCL3/BEY2 proved to be the same gene as OCA2, which encodes the P melanosomal protein, 48 and which is homologous to the mouse pink-eye dilution gene. Polymorphisms in OCA2 are associated with color variations in eyes and hair, 9 and later studies confirmed that the OCA2 gene is the major determinant of brown and/or blue eye color in humans. 11, 39 Two OCA2 coding region variant alleles, Arg305 to Trp and Arg419 to Gln, are associated with brown and green/hazel eye colors, respectively. 18, 30 Blue eye color is also linked to the OCA2 locus. 11, 28 Three different single nucleotide polymorphisms in intron 1 of the OCA2 gene are associated with blue eye color, light brown hair, and fair and medium skin types. 7 A dominance order model that uses the BEY2 and GEY genes is used to explain the inheritance of blue, green, and brown eyes. In this model, the brown allele of BEY2 gene on chromosome 15 is always dominant over its corresponding blue allele. Similarly, the green allele of the GEY gene on chromosome 19 is also dominant over blue. In this model, the brown allele of BEY2 is dominant to the green allele of GEY, and blue eyes occur only if all 4 alleles on chromosomes 15 and 19 are for blue eyes. However, this model does not explain how 2 blue-eyed parents can produce a brown-eyed child, so additional genes are clearly involved. Although 74% of the variance in eye color is estimated to be because of the QTL that contain OCA2, other loci that affect eye color are believed to exist telomeric of OCA2 on chromosome 15. 48 It is worth noting that SLC24A5 is located within band 15q21.1, a site that is telomeric to OCA2.
Our discussion focused on the effects of iris pigmentation differences on eye color, however, SLC24A5-related hypopigmentation of the RPE may have implications for human eye diseases beyond rare cases of autosomal recessive ocular albinism. It is possible that severe hypomelanization of neuroepithelium-derived cells in the eye might contribute to the pathogenesis and progression of age-related macular degeneration (ARMD). 38 Studies have linked a deficit of melanin pigment in the RPE and long-term exposure to bright light to an increased risk of developing ARMD, a leading cause of blindness. 34 Similarly, there is a significant association between light iris color/reduced fundus pigmentation and an increased incidence of ARMD. 10 It is posited that melanin in the RPE might have an antioxidant role in protecting the retina from oxidative damage that accumulates over time and eventually progresses to ARMD. 41 Thus, it seems reasonable to propose that hypopigmentation of the RPE because of aberrant SLC24A5 function might increase susceptibility to development of ARMD. Interestingly, evidence for a ARMD-associated locus has been reported on the same band of chromosome 15 (15q21) 17 that contains SLC24A5, which supports a possible link to the disease.
In conclusion, our findings demonstrated that Slc24a5 is important for normal melanization processes in all pigmented cells but that the protein is especially critical for normal melanization of neuroepithelium-derived pigmented cells in the mouse eye. We hypothesize that SLC24A5 may be involved in the development of some forms of ocular albinism and possibly macular degeneration in humans. We also propose SLC24A5 as a candidate gene for EYCL2/BEY1, a poorly characterized locus linked to central brown eye color in humans.
Footnotes
Acknowledgements
We thank Claire Gelfman and Kim Paes for providing funduscopic images and helpful discussion, Karen Harris and Mary Thiel for histology support, and Joe Shaw for his invaluable assistance in preparing the manuscript.
