Abstract
We describe a disseminated biphasic neoplasm in a young Madagascar tree boa utilizing transmission electron and light microscopy. Discrete neoplastic cells identified within pulmonary capillaries and hepatic sinusoids represented the leukemic phase. Spindloid cells represented the sarcomatous phase, which comprised hepatic and fat body nodules. A zone of transition of the neoplastic cells, from discrete to spindloid, was noted along the periphery of the hepatic and fat body nodules. Ultrastructural examination elucidated similar nuclear features in the discrete and spindloid neoplastic cells and revealed collagen fibers within the spindloid neoplastic cells. These ultrastructural findings indicate that the discrete and spindloid cells represent a single neoplastic process with a subpopulation of cells exhibiting mesenchymal differentiation.
Neoplastic disease in captive reptiles is growing in prevalence, in part, as a result of increased life expectancy caused by improved husbandry and management. 10 Commonly reported ophidian neoplasms include mesenchymal, 5 epithelial, 7, 10 and lymphoid/hematopoietic. 1 Among ophidian species, the most reported neoplasms are in aged colubrids, followed by crotalids, vipers, and boids. 6 Occurrence of oral fibrosarcoma, 6 multicentric lymphoma, pancreatic adenoma, leiomyosarcoma, adrenal adenocarcinoma, 1 and granulocytic sarcoma 3 have been reported in boids. However, few reports provide detailed documentation of neoplasm in both captive and wild ophidian species. Herein, we describe, in a Madagascar tree boa, histologic and ultrastructural features of a biphasic metastatic neoplasm exhibiting an intravascular discrete cell phase and an extravascular sarcomatous phase.
A 5-year-old female Madagascar tree boa was presented to the referring veterinarian with a history of having a 60-minute seizure a day earlier, sluggishness, and a decrease in mental awareness. Upon physical examination, body condition appeared normal, but the snake exhibited dysecdysis. Analysis of peripheral blood revealed a monocyte count of 4,070/μl (reference range for Madagascar tree boa: 290 to 8,800/μl). 14 There was no evidence of hemoparasites. Six days later, the snake was found dead.
Upon necropsy, the snake was found in good body condition. The proximal portion of the right fat body contained an approximately 1-cm-diameter, firm, raised, pale, mottled hemorrhagic nodule. Petechial hemorrhages were noted throughout the fat bodies. A 1-cm-diameter, pale–tan, poorly demarcated firm mass was also noted in the liver (Fig. 1). Multiple foci of red to tan pinpoint nodules covered both the lung and the liver. Multiple, pale–tan streaks were noted on the capsular surface and within the parenchyma of the kidneys. Aerobic culture of liver tissue did not reveal any significant bacterial pathogens.
Tissue samples from, brain, heart, liver, lung, kidney, and fat body were collected, fixed in 10% neutral-buffered formalin, and were routinely processed for histopathology by light and transmission electron microscopy.
The solid phase of the neoplasm was manifested in the hepatic and fat body nodules. Neoplastic cells were spindloid with indistinct borders and were embedded in a pale fibrillar eosinophilic stroma. These cells contained pleomorphic oval to elongate nuclei with sparsely stippled chromatin, which contained 1 to 2 prominent nucleoli. Multinucleated and karyomegalic cells were frequently observed (Figs. 2, 3). The neoplasm contained hemorrhagic foci and heterophils.
The discrete cell phase (leukemic phase) was characterized by conspicuous infiltration of hepatic sinusoids and pulmonary capillaries by these cells, resulting in their distension (Figs. 4, 5). Individual cells were round to polygonal, had distinct cell borders, and contained mild to moderate amounts of pale eosinophilic cytoplasm. Nuclei were round, with a coarsely stippled chromatin pattern, and contained 1 to 2 prominent nucleoli and thus demonstrated nuclear morphological features similar to the spindloid cells comprising the discrete nodules within the liver and fat body. Along the peripheral margin of the liver and fat body nodules, neoplastic cells exhibited a unique hybrid feature characterized by elongating discrete cells, which gradually and imperceptibly merged into the spindloid population (Fig. 6). These cells with intermediate morphological features suggest that the spindloid cells and discrete cells represent 2 morphologic manifestations of the same neoplasm.
The endocardium was covered focally by small clusters of neoplastic cells that exhibited variable morphological features, ranging from discrete cells to intermediate cells that appeared to be flattening against the endocardium (Fig. 7). The cortical surface and parenchyma of the kidney was also infiltrated by similar neoplastic cells. Within the brain, there was a focal area comprised of mononuclear inflammatory cells, which was circumferentially surrounded by mild fibrous connective tissue. The neuronal elements were histologically unremarkable. No neoplastic cells were noted in the brain. Given the lack of obvious neoplastic infiltration in the brain, the neurological signs noted in this snake were likely caused by energy deprivation and/or hyperviscosity syndrome. It is also possible that an affected area of the brain was not included in the histologic sections examined.
In an attempt to determine the histogenesis of the neoplastic cells, immunohistochemistry was performed using antibodies against vimentin, lysozyme, and CD18 antibodies. The results were inconclusive (results not shown), possibly a result of poor cross reactivity of the primary antibodies generated in laboratory mammals with the ophidian tissue or the lack of differentiation of the neoplastic cells, resulting in an indistinct phenotype.
The liver, neoplastic nodule in the fat body, and lungs were examined ultrastructurally by transmission electron microscopy. Ultrastructural examination of the solid hepatic neoplasm exhibited elongated neoplastic cells embedded in a stroma containing sparse irregular collagen fibrils with faint and irregular cross-striations and banding patterns. Occasionally, these fibrils were noted within the cytoplasm of the spindloid neoplastic cells (Fig. 8). The cytoplasm also contained a few membrane-bound vesicles, some of which could be identified as swollen mitochondria. Intercellular junctions and desmosomes were not encountered. Lack of desmosomes and intercellular junctions suggested that the neoplasm was not of epithelial origin. Nuclei were elongated and the nuclear envelope was frequently characterized by multiple folds and invaginations. The nucleoli were large with characteristic reticulated dense fibrillar and granular areas (Fig. 8). Ultrastructurally, the discrete neoplastic cells in the hepatic sinusoids were large, round, and exhibited distinct cell borders. A few membrane-bound vesicles were noted within the cytoplasm. Nuclei were large and round, but otherwise exhibited ultrastructural features similar to those of the spindloid cells. In contrast to the spindloid cells, collagen fibrils were not found in the discrete cells, which also were not associated with a sparse fibrillar matrix (Fig. 9). Although virus-associated neoplasms have been reported in boids, no viral particles were identified ultrastructurally or histologically. 8
Thus, this disseminated neoplasm was biphasic with an intravascular discrete cell phase and an extravascular sarcomatous phase. It is possible that these 2 phases represent 2 distinct neoplasms; however, both cell types exhibit similar nuclear morphology and a convincing zone of transition at the periphery of the fat body and hepatic neoplastic nodules. Additionally, along the endocardial surface of the heart, small clusters of neoplastic cells contained discrete cells, elongated discrete cells, as well as spindloid cells. Ultrastructurally, both discrete and spindloid cells exhibit similar nuclear and cytoplasmic morphology. These morphological features point to a common histogenesis of the neoplasm. The presence of collagen in the spindloid population suggests fibroblastic differentiation of a subpopulation of neoplastic cells.
Current reports of mammalian mesenchymal tumors, 12 brain tumors, 13 and mammary cancer 4 support the concept of “neoplastic stem cells.” These cells are described as primitive cells with capacity to regenerate phenotypically diverse populations of cells, which express differentiated cell products. 2 As an example, in a report of canine mammary tumors it was experimentally shown that a single stem cell formed multiple neoplasms, including mammary osteosarcoma, mammary spindle-cell tumors, fibromas, and rhabdomyoid tumors. In this particular case of a biphasic tumor, the circulating discrete cell phase may represent a circulating neoplastic stem cell, which transforms into a differentiated, collagen-producing sarcomatous phase as seen in the discrete nodules in the liver and fat body. In scientific literature, there are reports of facultative fibroblasts in human beings, which are histiocytes that could appear and function as fibroblasts and can give rise to neoplasms, including histiocytomas, fibrous histiocytomas, or pleomorphic fibrosarcomas. 11 Experimental data show that these fibroblasts express monocytic cell surface markers, suggesting that these cells are of true histiocytic origin. 9 Hence, it is possible that the leukemic component of this neoplasm may represent a facultative fibroblast with potential for transformation into a fibroblastic phenotype with ability to produce collagen.
Finally, this case demonstrates that evaluation of neoplasms by light microscopy alone may not lead to sufficient characterization. In such instances, additional diagnostic techniques such as immunohistochemistry, in situ hybridization, and transmission electron microscopy are helpful in determining histogenesis. Electron microscopy is especially useful in diagnosing neoplasms in exotic animal species as routine molecular reagents are often not available for these species. In this particular case of a disseminated neoplasm in a young Madagascar tree boa, we were able to provide evidence for mesenchymal differentiation of a subpopulation of neoplastic cells by demonstrating collagen fibrils ultrastructurally when molecular diagnostic techniques failed to be of any value.
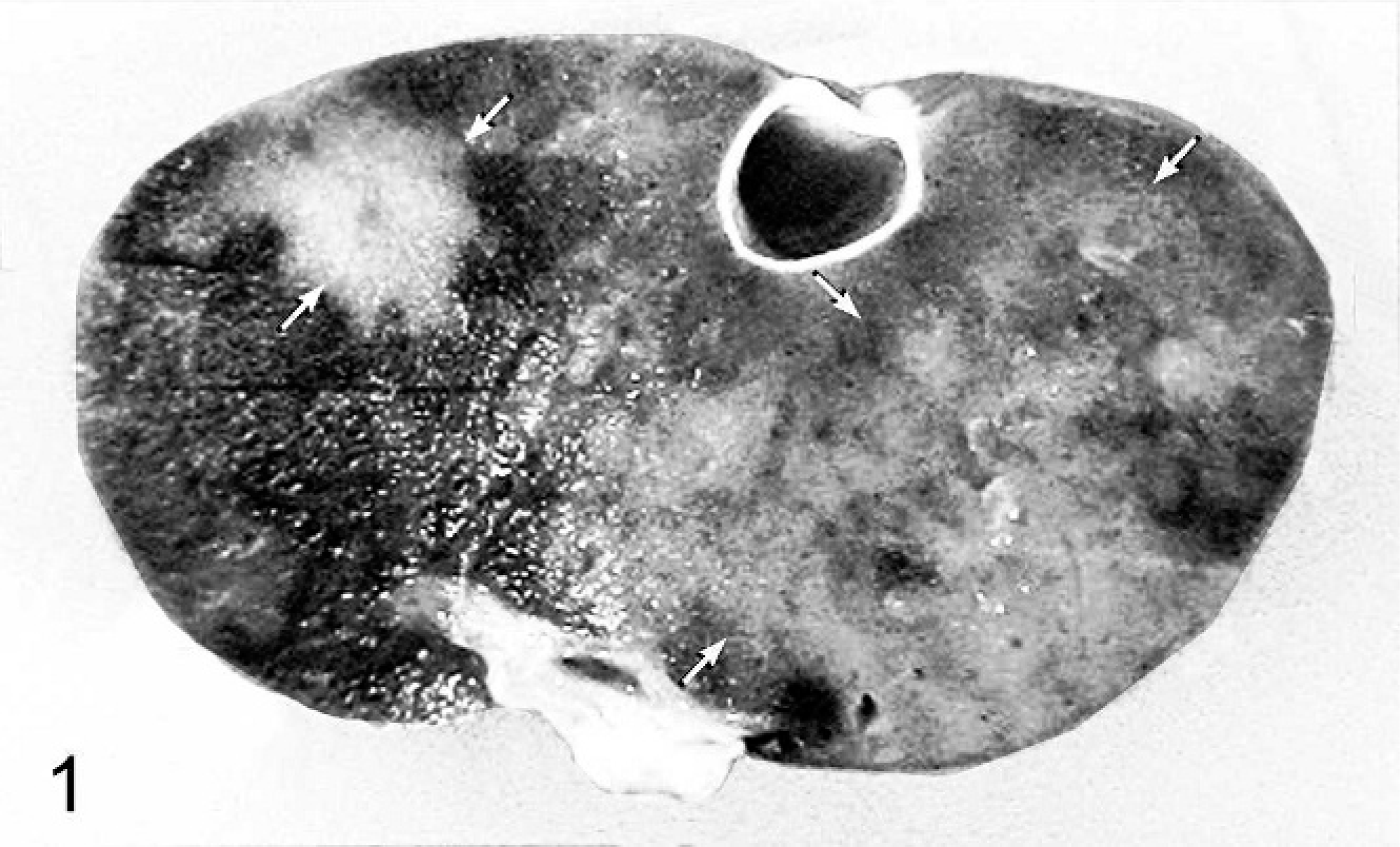
Liver; boa constrictor. Poorly demarcated firm–tan nodule (white arrows) within the liver (formalin-fixed sample).
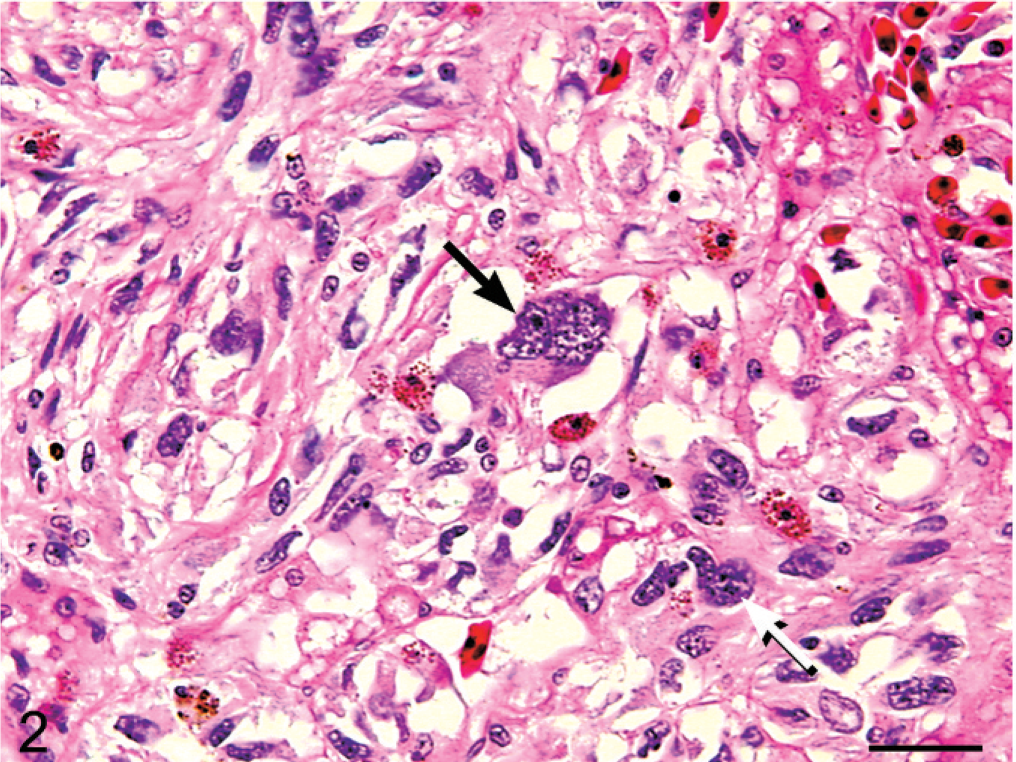
Liver; boa constrictor. Image demonstrating moderate to marked anisocytosis and anisokaryosis characterized by multinucleated (black arrow) and karyomegalic (white arrow) forms of the neoplastic cell population. HE. Bar = 30 μm.
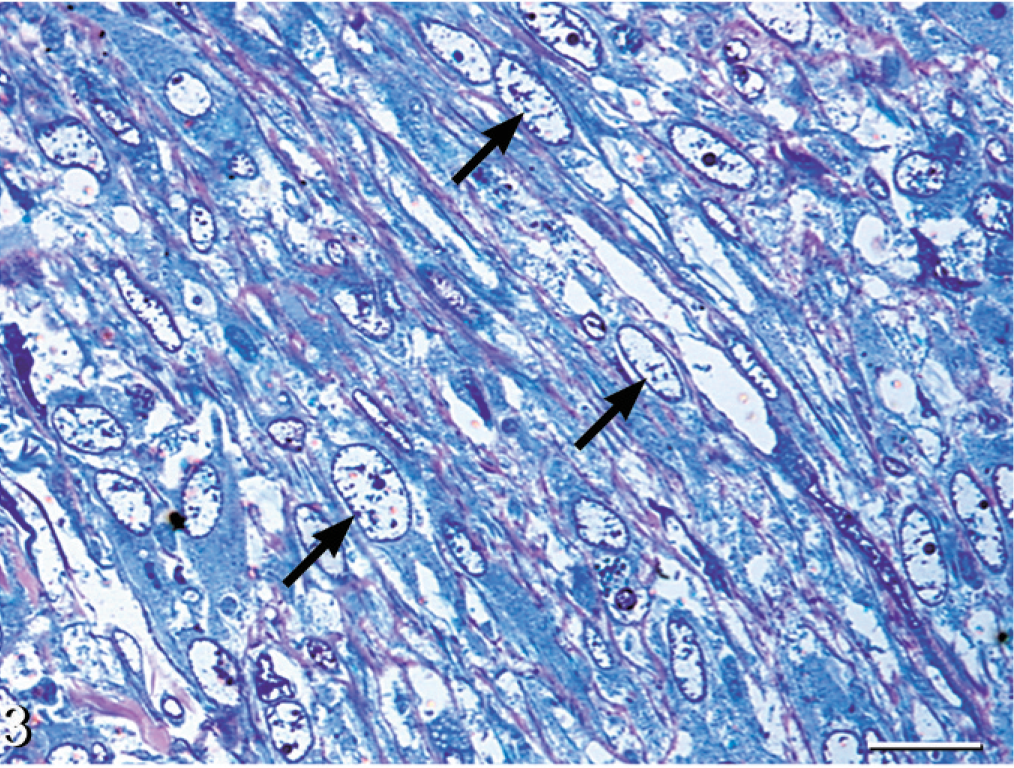
Liver, nodule; boa constrictor. Sheets of elongated neoplastic cells with coarsely stippled nuclear chromatin (black arrows) embedded in a scant fibrillar stroma. Methylene blue and safranin-O. Bar = 20 μm.
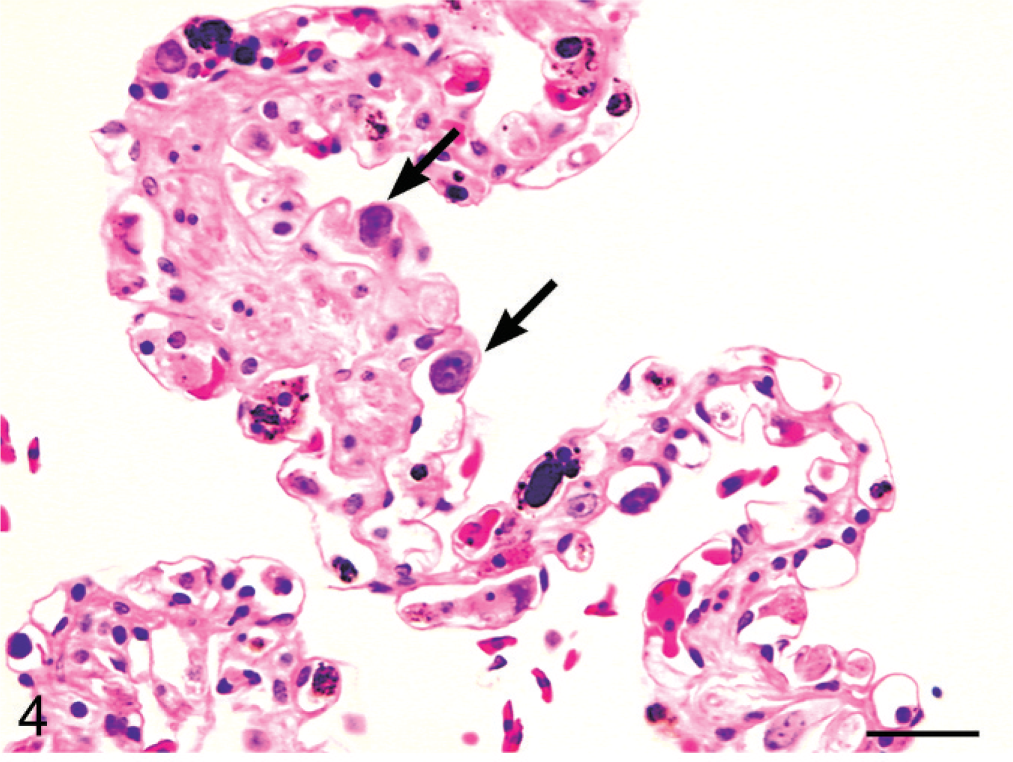
Lung; boa constrictor. Distended alveolar capillaries filled with discrete cells (black arrows) represent the discrete cell phase of the biphasic neoplasm (black arrow). HE. Bar = 30 μm.
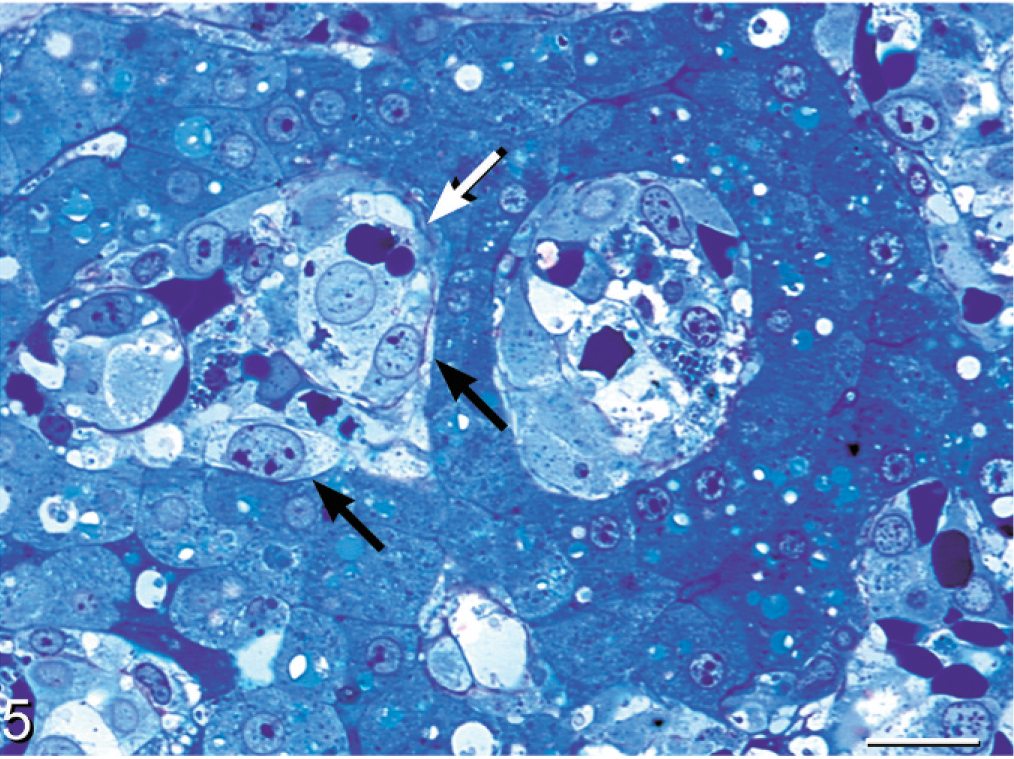
Liver; boa constrictor. The discrete cell phase (black arrow) is evident within hepatic sinusoids admixed with nucleated red blood cells (white arrow). Toluidine blue. Bar = 20 μm.
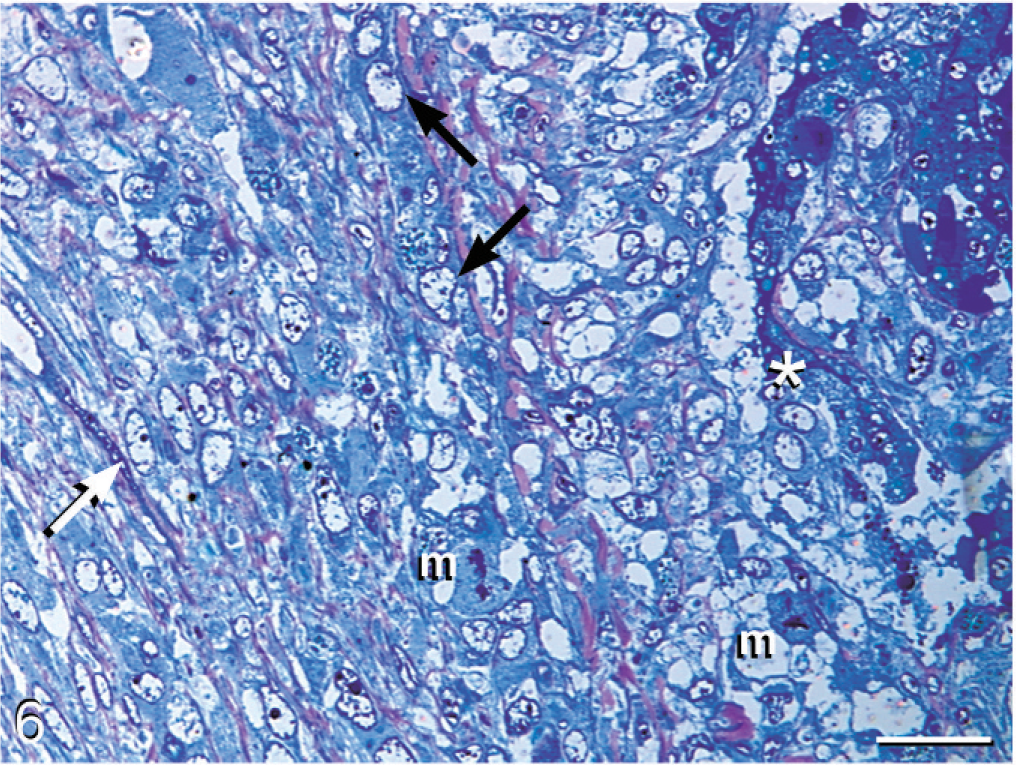
Liver; boa constrictor. Discrete cells (asterisk) within hepatic sinusoids adjacent to hepatocytes transition into an intermediate phase (black arrow) and then into an elongated cell phase (white arrow), which is embedded in a fibrillar stroma. Mitotic figures (m) are present along the zone of transition. Methylene blue and safranin-O. Bar = 40 μm.
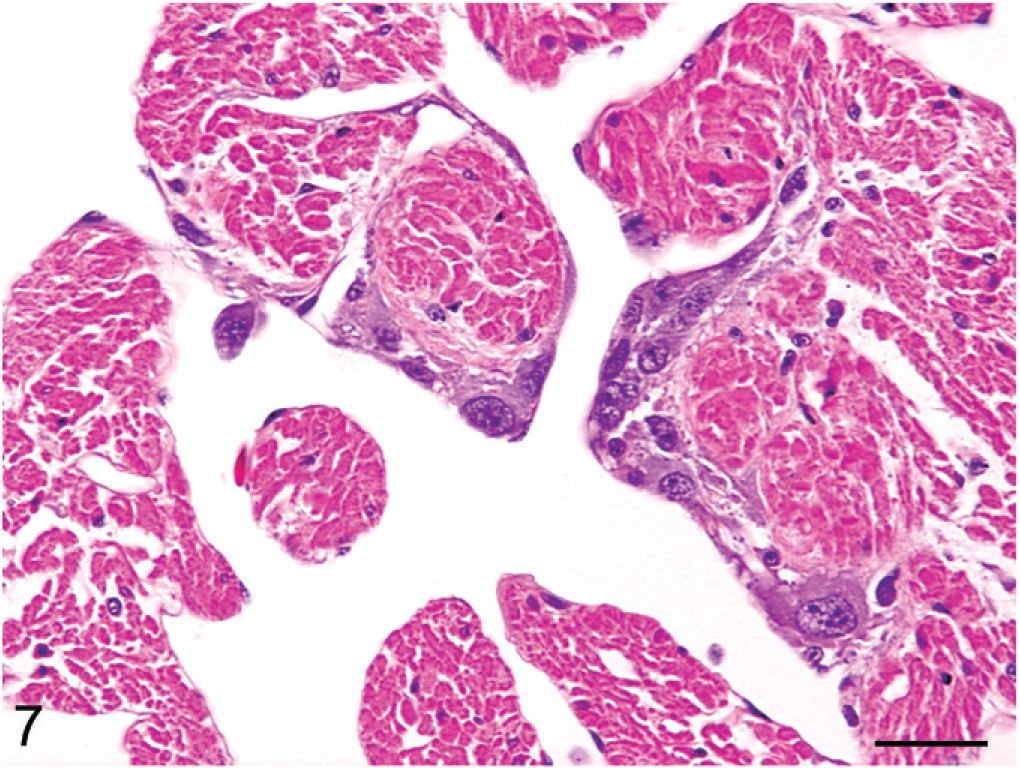
Heart; boa constrictor. Metastatic neoplastic cells adhering to the endocardial surface exhibit variable morphologic features ranging from polygonal to spindloid. HE. Bar = 30 μm.
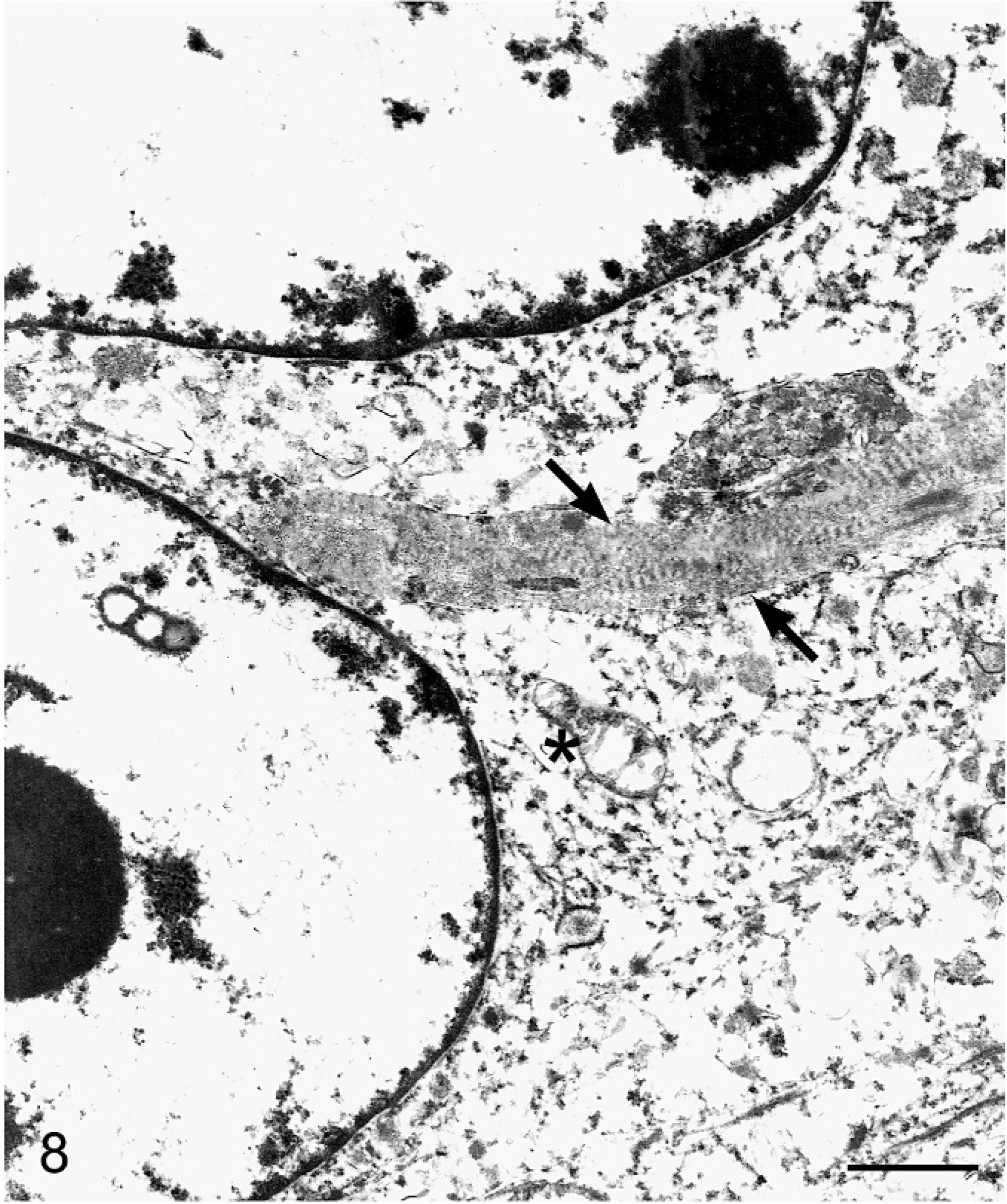
Liver; boa constrictor. Ultrastructural morphology of the solid neoplastic nodule demonstrating partial profiles of two spindle-shaped neoplastic cells with euchromatic nuclei and intracytoplasmic collagen with cross-banding in close proximity to the nucleus (black arrows). Cytoplasmic features include loose fibrillar material, membrane-bound vesicles, and swollen mitochondria (asterisk). Uranyl acetate and lead citrate. Bar = 2 μm.
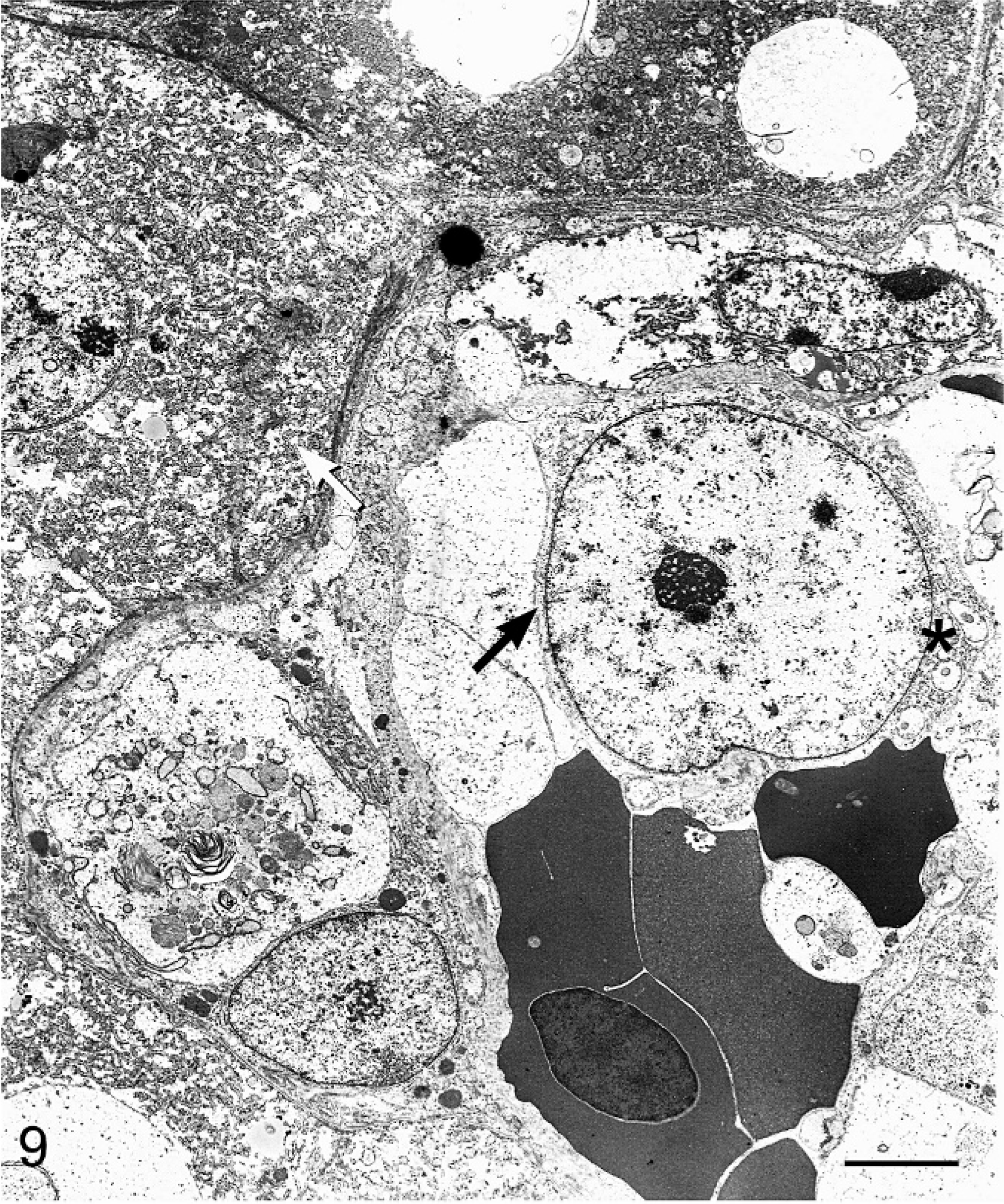
Liver; boa constrictor. Within hepatic sinusoids admixed with nucleated red blood cells is a discrete cell with a large nucleus (black arrow). Intracytoplasmic features include membrane-bound vesicles (asterisk). Hepatocytes (white arrow) are located along the peripheral margin of the sinusoid. Uranyl acetate and lead citrate. Bar = 5 μm.
Footnotes
Acknowledgements
We thank Carol Moeller and Jennifer Palmer for technical assistance with electron microscopy.
