Abstract
A case of cerebral angiomatosis in a cat was associated with neurologic signs characterized by clusters of severe generalized seizures. Bilaterally in the gray matter, most prominent in the cingulate gyrus, there was focal accumulation of garlandlike arrangements of blood vessels. Vessels exhibited activated, hypertrophic endothelial cells and thickening and progressive dystrophic mineralization of the basement membrane, with complete luminal obstruction of some affected vessels. Thickening of the basement membrane was due to accumulation of endothelium-derived proteins such as laminin and von Willebrand factor. Furthermore, moderate diffuse astrogliosis was observed. Findings indicate an idiopathic angiomatosis, with clinical signs possibly due to ischemia resulting from narrowing or complete obliteration of vessel lumina. Changes represent a unique endothelial cell-derived lesion within the brain not previously described in humans or domestic animals.
Keywords
In humans, vascular lesions such as atherosclerosis are very common and are a major cause of disease. 4,14 In contrast, cats rarely exhibit noninflammatory vascular alterations. If present, they are mainly represented by arteriolosclerotic lesions due to hypertension, predominantly developing in the course of chronic renal failure and hyperthyroidism. 5 Further reports on vascular alterations in cats include cases of arteriosclerosis and angioendotheliomatosis. 9,13,16 Here, we describe a case of bilateral cerebral angiomatosis that was associated with generalized seizures in a cat.
A female neutered 2-year-old stray domestic shorthair cat was taken into an animal shelter 5 months prior to death. While at the shelter, it had a seizure attack. After transfer to a new owner, the cat developed seizure attacks with increasing frequency, starting 5 weeks prior to death. The cat was euthanatized after having at least five generalized tonic-clonic seizures per day. Attacks were characterized by spasms, falling over, jumping up, bouncing, rowing, and unpredictable movements, accompanied by autonomic signs such as urination.
At necropsy, the cat did not show any macroscopic lesions. The complete brain and spinal cord and specimens from most visceral organs were fixed in 10% nonbuffered formalin for 18 hours and routinely embedded in paraffin. Sections (5 µm thick) were stained with hematoxylin and eosin (HE). Brain sections were also stained with Congo red, periodic acid–Schiff (PAS), and the von Kossa stain. For immunohistology, the following cross-reacting antibodies were applied: mouse anti-swine desmin (Dako Diagnostika GmbH, Hamburg, Germany), mouse anti-swine vimentin (Dako), mouse anti-human HLA-DR (α-chain, major histocompatibility complex [MHC] class II; Dako), mouse anti-human collagen IV (Dako), mouse anti-human laminin (Sigma-Aldrich, Deisenhofen, Germany), rabbit anti–von Willebrand factor (Dako), and rabbit anti–glial fibrillar acidic protein (GFAP; Dako). For the demonstration of laminin and von Willebrand factor, slides were pretreated with 0.5% protease (type XXIV: bacterial; Sigma-Aldrich). For the demonstration of MHC class II antigen and collagen IV, slides were incubated in citrate buffer (10 mM, pH 6.0) at 96 C for 30 minutes. Swine anti-rabbit IgG (Dako) and rabbit peroxidase antiperoxidase (PAP) complex (Dako) were used as the detection system for polyclonal antibodies, and rat anti-mouse IgG (Dianova GmbH, Hamburg, Germany) and mouse PAP complex (Dianova) served as the detection system for mouse monoclonal antibodies. Visualization was achieved by incubation with 0.05% 3,3′-diaminobenzidine tetrahydrochloride in 0.1 M imidazole/HCl buffer (pH 7.1), and counterstaining was performed with Papanicolaou's hematoxylin. For transmission electron microscopy, the detachment technique was performed on uncovered HE-stained sections embedded in Epon® (Fluka Ag, Buchs, Switzerland). One 0.4-µm-thick semithin section and 50-nm thin sections were prepared. 1 The thin sections were stained with uranyl acetate and lead citrate.
Microscopically, the frontal third of the cingulate gyrus, part of the gyrus proreus, and the head of the caudate nucleus exhibited focal bilateral blood vessel alterations in the gray matter. Alterations extended throughout all cortical layers but were most prominent in the molecular layer. Affected cortical areas exhibited an increased number of blood vessels, accumulating in a garlandlike pattern (Fig. 1). Vessels showed hypertrophic endothelial cells (Fig. 2) and were often surrounded by a variable thickness subendothelial layer of homogeneous eosinophilic material (Fig. 3), which was negative with the Congo red stain but stained faintly red with the PAS reaction. Ultrastructurally, this material appeared amorphous, finely granular, and variably electron dense (Fig. 4) and was similar to the basal laminae of unaffected blood vessels. Within this material, spotlike focal mineralization, identified by the von Kossa method, was observed. Some vessels were completely encircled by a layer of mineralization (Fig. 5), and others were obliterated and exhibited degenerated endothelial cells. In the choroid plexus, single blood vessels exhibited similar alterations, including mineralization. In the left hippocampus, some vessels showed thickening of the walls.
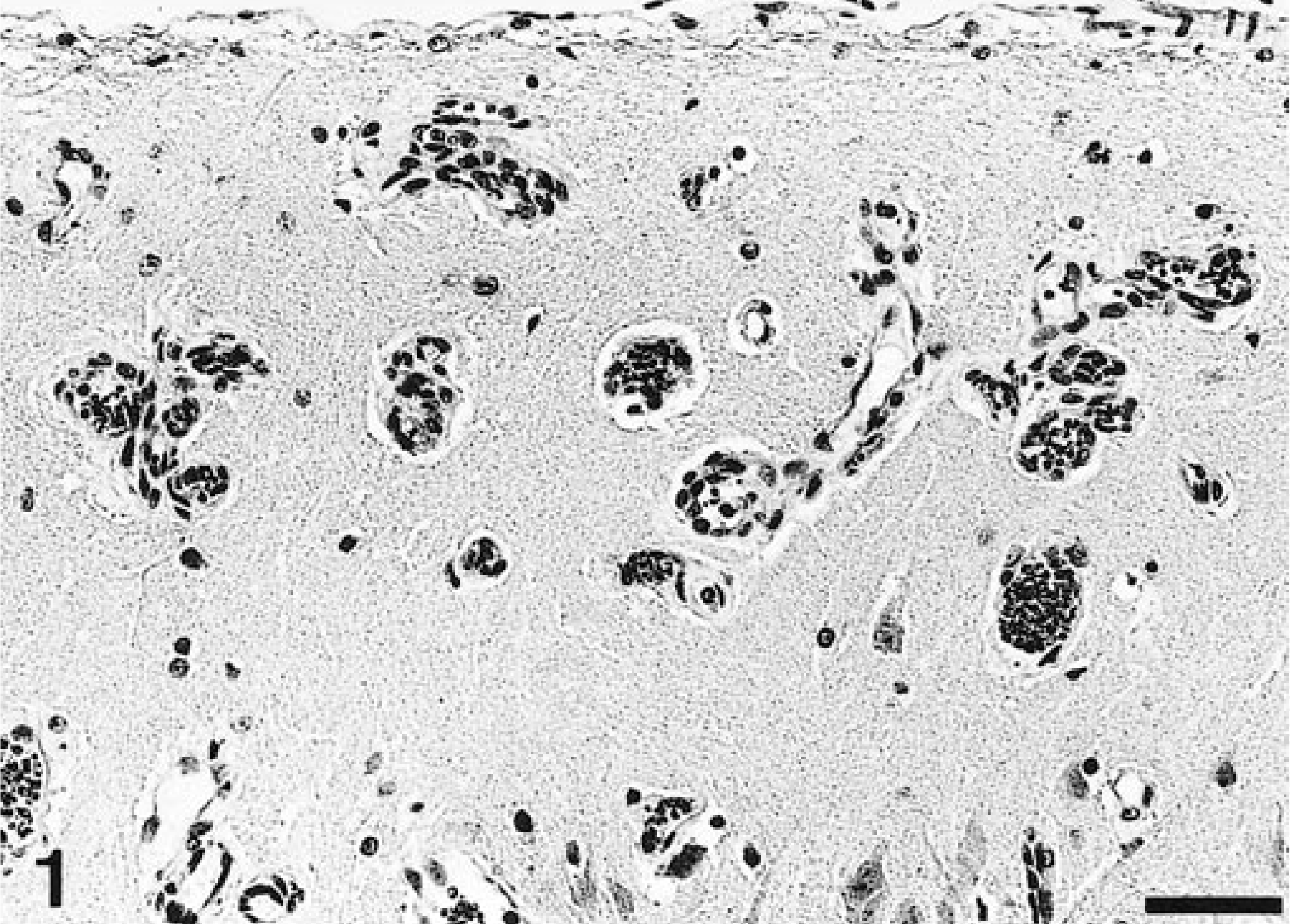
Cingulate gyrus; cat. Garlandlike arrangement of cortical blood vessels. HE. Bar = 50 µm.
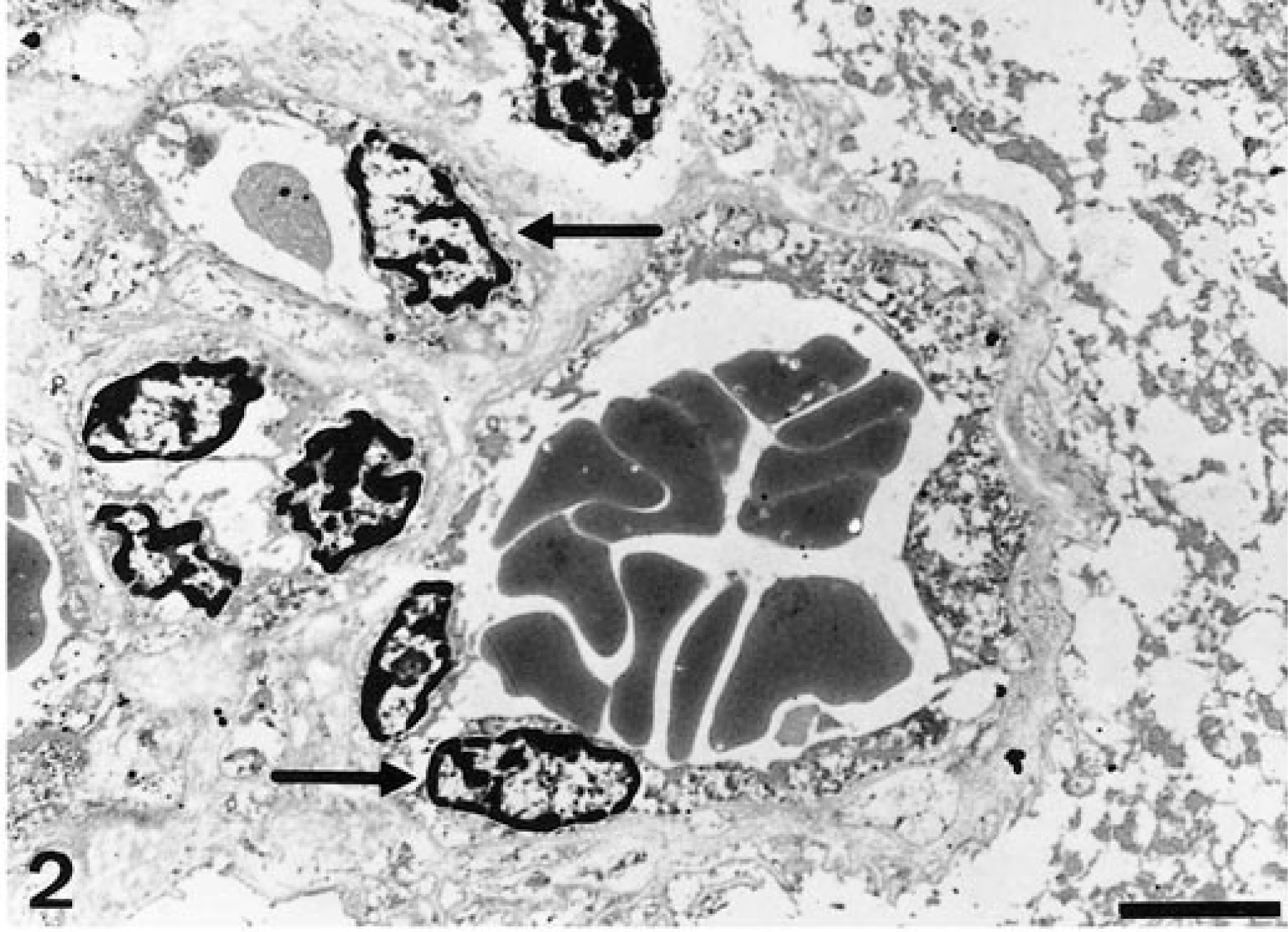
Transmission electron micrograph. Cingulate gyrus; cat. Blood vessels exhibit distinct hypertrophy of endothelial cells (arrows). Bar = 2.5 µm.
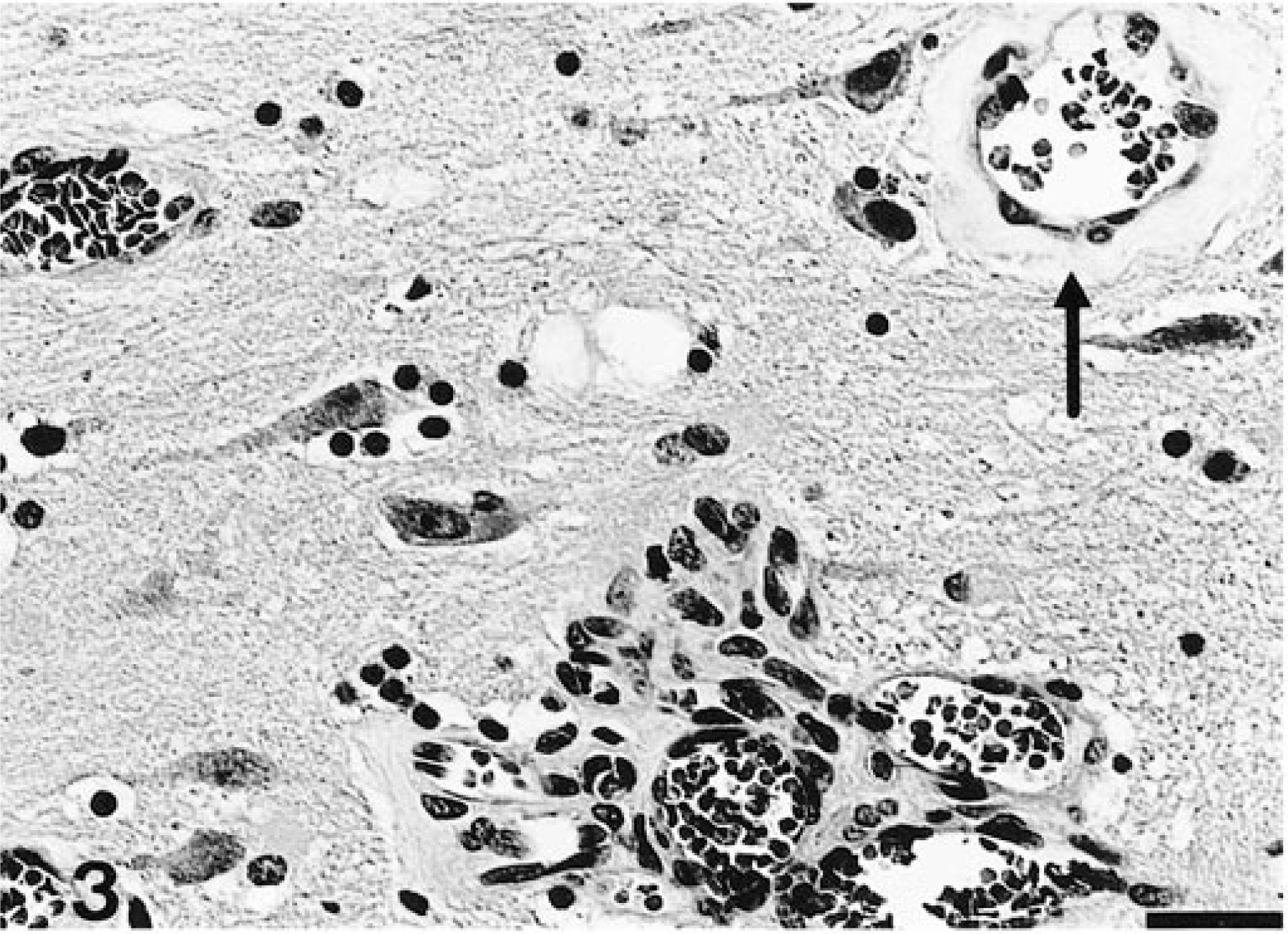
Cingulate gyrus; cat. Blood vessel with thickened basement membrane due to subendothelial deposition of homogeneous material (arrow). HE. Bar = 25 µm.
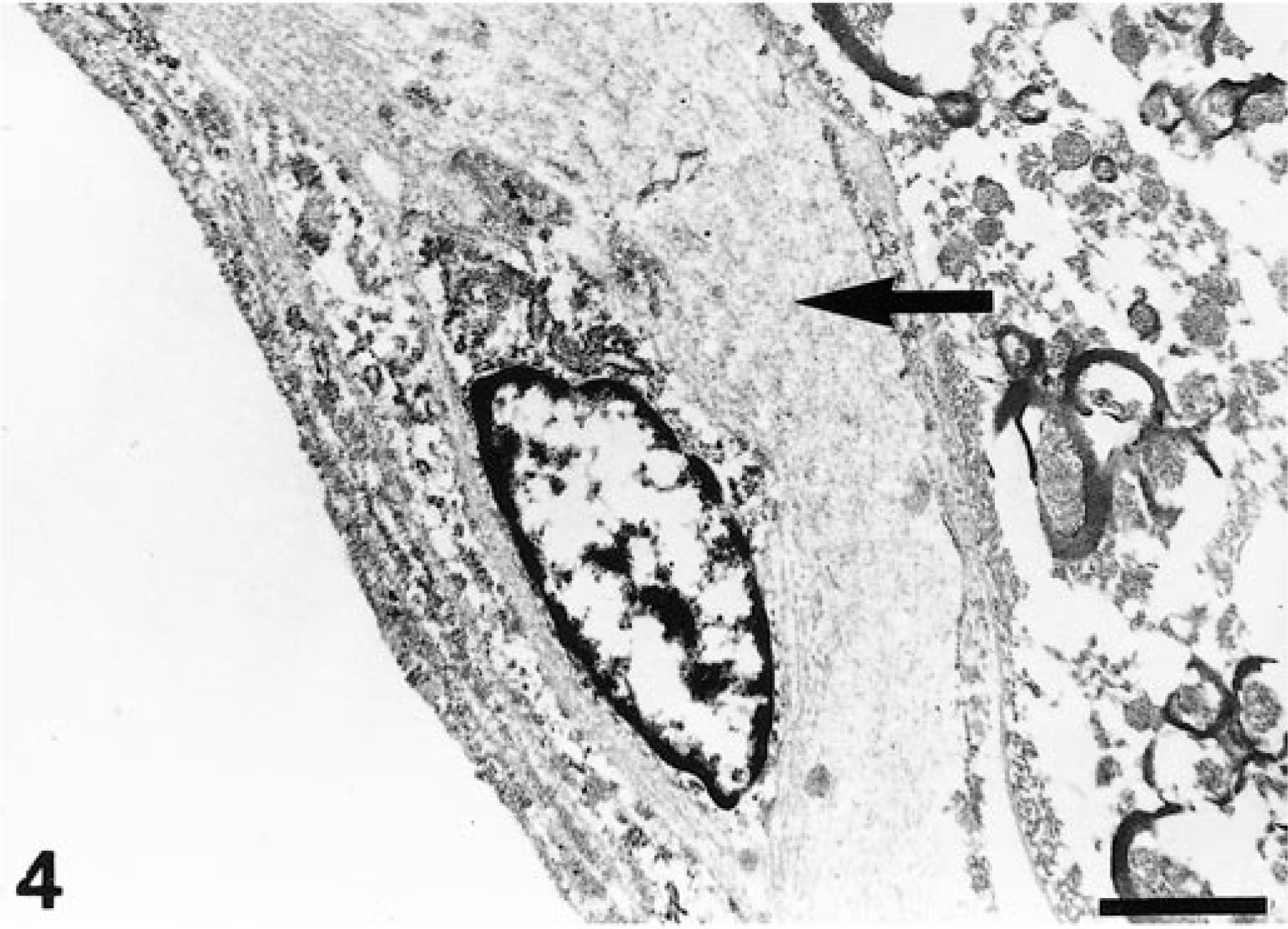
Transmission electron micrograph. Cingulate gyrus; cat. Blood vessel with deposited subendothelial material that is amorphous, finely granular, and variably electron dense (arrow). Bar = 1 µm.
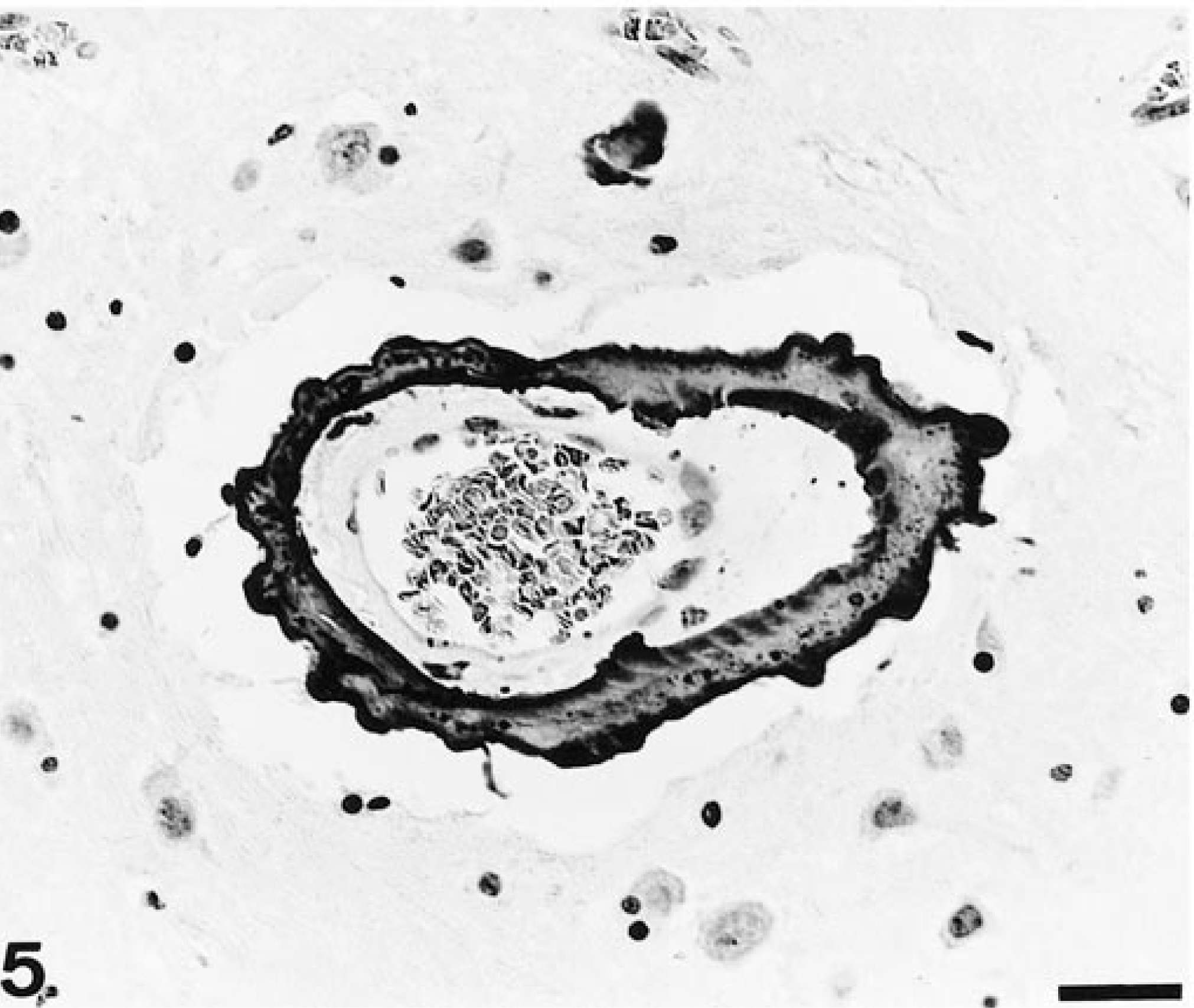
Cingulate gyrus; cat. Blood vessel with thickened basement membrane, encircled by a thick mineral layer. HE. Bar = 25 µm.
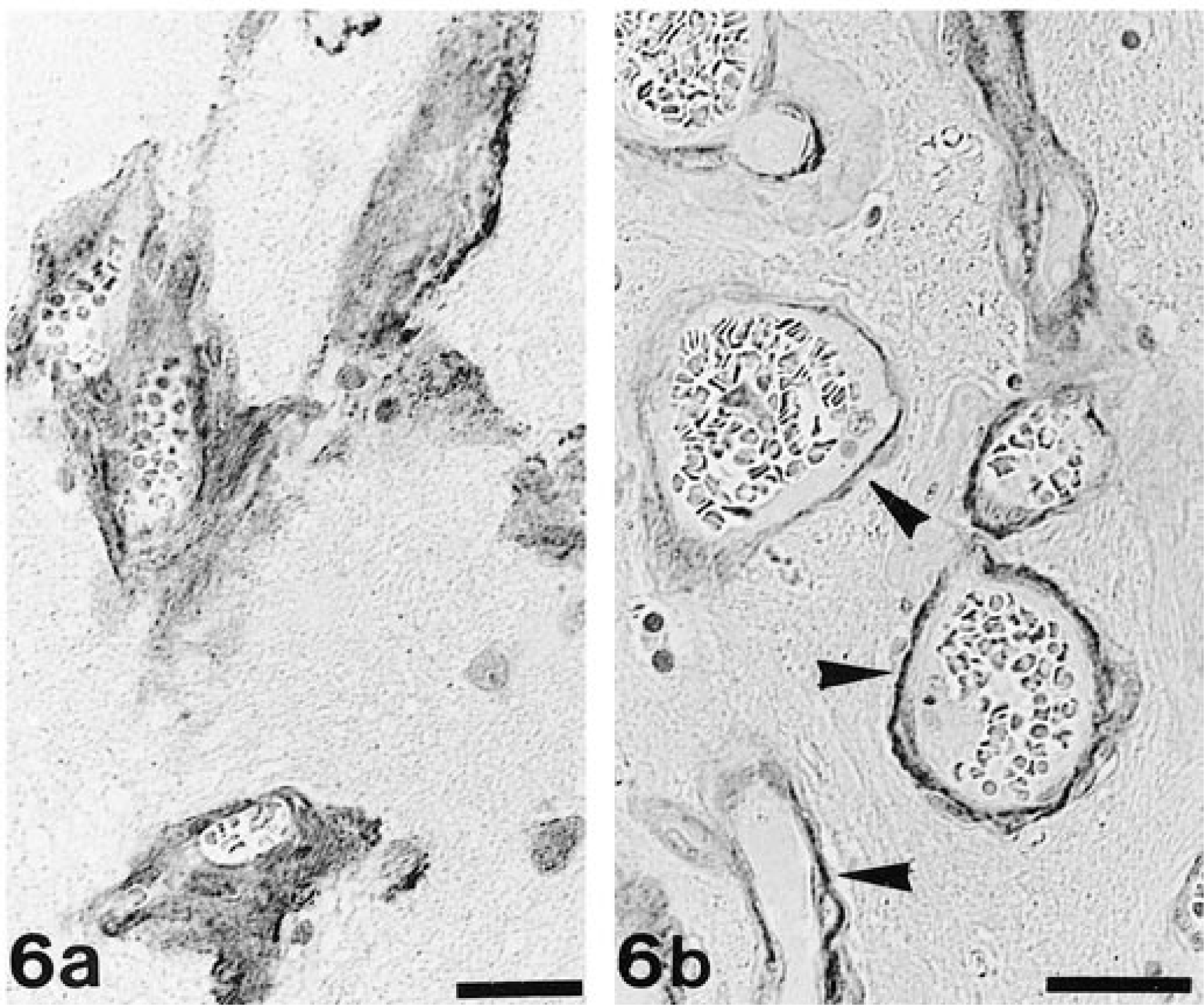
Immunohistologically, the homogeneous material stained strongly positive for laminin (Fig 6a) and von Willebrand factor. Staining for collagen IV was represented by a fine, mainly subendothelial line in unaltered vessels; in altered blood vessels, it was visible as a variable thickness band located subendothelially or outside the homogeneous material (Fig 6b). The cytoplasmic staining of vascular smooth muscle cells/pericytes for desmin depicted these cells as abluminal from endothelial cells and within or abluminal from the deposited material. The homogeneous material did not stain for desmin, vimentin, or GFAP. Staining for GFAP revealed moderate diffuse astrogliosis, represented by numerous hypertrophic astrocytes within altered cortical areas. Plump cytoplasmic astrocyte processes often surrounded both altered blood vessels and deposited material and were demonstrable in the distinct thickening of the adjacent glia limitans. MHC class II antigen was mildly expressed by perivascular microglial cells in both altered and unaltered brain areas and in hypertrophic endothelial cells of altered blood vessels.
All other organs examined (spinal cord, heart, lungs, liver, spleen, kidneys, jejunum, mesenteric lymph nodes, thymus, and bone marrow) did not show any alterations.
The focal bilateral cerebral angiomatosis in this cat was associated with severe clusters of seizures. Seizures in cats are mostly caused by morphologic changes in the brain and are mainly of inflammatory, traumatic, ischemic, or neoplastic origin. 12 Vascular lesions have not been previously identified as a possible cause of seizures in cats.
The increased number of blood vessels in a garlandlike arrangement in affected regions indicates vessel proliferation. In affected vessels, endothelial cells were hypertrophic; although their number seemed to be increased, there was no evidence of cell proliferation. Morphologic changes included subendothelial deposition of endothelial cell–derived proteins undergoing dystrophic mineralization. 7 The presence of excessive laminin and von Willebrand factor abluminal from the endothelial cell layer in affected vessels indicates that hypertrophic, activated endothelial cells produced and secreted these basement membrane proteins. 11,14 There was no evidence of smooth muscle cell proliferation and/or synthesis of extracellular matrix proteins such as collagen, ruling out a potential intimal thickening in response to vascular injury. 14 Astrogliosis develops after brain injury as a consequence of local ischemia due to total occlusion and/or loss of single blood vessels. 10
Vessel lesions in this cat were similar to those of hypertensive microangiopathy (small artery disease) in humans. 4 In cats, systemic hypertension is rarely seen and is usually related to chronic renal disease, hyperthyroidism, or a high-salt diet. 5,8,17 Vessel wall damage due to systemic hypertension is most prominent in heart, kidneys, and retina, and the deposited material is composed of various plasma components such as amyloid or fibrin because of increased vascular permeability. 4,5,17 In the present case, however, vessel lesions were clearly restricted to the brain. In humans, diabetic microangiopathy is also characterized by hyaline thickening of basement membranes mainly due to deposition of type IV collagen. 14 However, in this cat, neither massive deposition of collagen IV nor indication of diabetes were present. Furthermore, vascular lesions clearly differ from atherosclerosis in that there was no evidence of lipid deposition within vessel walls. 14 Therefore, the pathogenesis and cause of the vascular lesions in this cat remain unclear, and a possible congenital condition can be assumed.
Brain vessel alterations in the cat have been reported twice in association with multisystemic vascular lesions. 13,16 Because of the proliferation of factor VIII–related antigen-expressing endothelial cells in small arteries and veins, alterations were regarded as equivalent to human Masson's “intravascular pseudoangiosarcoma.” 6,16 However, in the present case, there was no indication of systemic vascular proliferation or of endothelial cell proliferation as the cause of luminal obstruction.
Lesions in this cat were similar to those observed in other syndromes. Encephalofacial angiomatosis (Sturge-Weber syndrome) has been reported in humans and in a horse, and proliferative vasculopathy and hydrancephaly-hydrocephaly (Fowler's syndrome) has been observed in humans. 3,8 In both syndromes, tortuous and calcified blood vessels can be found; however, these vessels bear features of neoplasia. Abnormal calcified blood vessels are also seen in meningioangiomatosis, which has been described in humans and in a dog. 2,15 The lack of the characteristic discrete meningeal and cortical nodules formed by proliferated blood vessels and the spindle-shaped and concentrically arranged meningothelial cells, however, exclude this diagnosis. 2,15 In cerebral autosomal dominant arteriopathy of humans, a nonarteriosclerotic and nonamyloid arteriopathy can be observed mainly in the white matter. 4,18 Because both media and adventitia are thickened by a collagen-dominated material and degeneration of medial smooth muscle cells is stated, this lesion is not similar to those in this cat. 18
The observed bilateral cerebral angiomatosis predominantly in the cingulate gyrus represents a unique lesion associated with nervous signs in a cat.
Footnotes
Acknowledgements
We thank Dr. G. Preuss for providing both cat and clinical information, Mrs. K. Franke for preparation of the detachment samples, and Mrs. U. Zeller for photographic assistance.
