Abstract
Vascular endothelial growth factor (VEGF) is an important regulator of tumor angiogenesis and vascular permeability, and has been implicated both in progression of central nervous system (CNS) tumors and development of vasogenic peritumoral edema. A retrospective study was done to characterize the levels of expression of the 3 major canine VEGF isoforms (VEGF120, VEGF164, VEGF188) in a variety of spontaneous canine CNS tumors using quantitative TaqMan reverse transcription real-time polymerase chain reaction. Presence and degree of peritumoral edema also were determined in sampled tumors using magnetic resonance imaging (MRI). Increased expression of VEGF relative to normal cerebral cortex tissue was seen predominantly in high grade astrocytic (grade IV) and oligodendroglial (grade III) tumors, with lower expression in low grade astrocytomas (grade II) and meningiomas (grade I). All 3 major VEGF isoforms were present; VEGF164 was the predominant isoform, particularly in the tumors with the highest VEGF expression. Peritumoral edema was present in all tumor types; however, a significant association between the extent of peritumoral edema and the level of VEGF expression was not apparent.
Keywords
Introduction
Vascular endothelial growth factor (VEGF), also termed VEGFA and vascular permeability factor, is a member of a family of angiogenesis-related growth factors. It is a potent endothelial cell mitogen and survival factor, and a strong inducer of vascular permeability, a thousand times more potent than histamine. 17, 22 It is an essential factor for angiogenesis and vasculogenesis during normal development and is proposed to play a major role in tumor neovascularization and associated edema. 11, 28 The VEGF gene consists of 8 exons, and in humans at least 9 VEGF isoforms are produced by alternative splicing: VEGF121, VEGF145, VEGF148, VEGF162, VEGF165, VEGF165b, VEGF183, VEGF189, VEGF206. 2, 10, 11, 48 VEGF121, VEGF165, and VEGF189 are the major isoforms secreted by most cell types, with VEGF165 being the most abundant isoform in both normal and transformed cells. 10, 44 The major VEGF isoforms bind cell-surface heparan sulphate proteoglycans to varying degrees, with VEGF121 being freely diffusible, approximately half of secreted VEGF165 being bound, and almost all VEGF189 remaining sequestered in the extracellular matrix.
VEGF is overexpressed at both the mRNA and protein level in a variety of human brain tumors, including astrocytomas, 13, 14, 29, 33, 38, 39, 45, 49, 54 oligodendrogliomas, 6, 7, 14, 19, 45, 52 and meningiomas. 13, 14, 21, 36, 45, 49 VEGF expression has been associated with a variety of factors, including necrosis and hypoxia, growth factors such as transforming growth factor (TGF) and fibroblast growth factor (FGF), cytokines including IL-1α and IL-6, and oncogenes or tumor suppressor genes such as Ras and TP53. 11, 28 Most studies have linked the degree of overexpression of VEGF with increasing tumor grade in astrocytomas, oligodendrogliomas, and meningiomas; 5, 6, 13, 14, 19, 21, 29, 36, 38, 50, 52, 54 however, this has not been universally reported. 35, 45, 47
Vasogenic peritumoral edema is a well-documented complication contributing to morbidity and death in patients with central nervous system (CNS) tumors. The exact mechanisms responsible for increased vascular permeability in specific tumors have not been elucidated fully and are likely to be multifactorial; however, VEGF has been implicated as a contributory agent in tumor-related edema in humans. Peritumoral edema, increased vascular permeability, and presence of intratumoral cysts have been correlated with increased VEGF expression in human gliomas 20, 26, 47, 51 and meningiomas. 12, 16, 34, 35, 37, 41 In addition, alleviation of peritumoral edema in high grade gliomas following administration of VEGF-receptor-specific tyrosine kinase inhibitors indirectly suggests an important role for VEGF in edema formation. 1
Five VEGF isoforms have been reported in dogs: VEGF120, VEGF144, VEGF164, VEGF182, and VEGF188. These isoforms have approximately 95% homology to the human amino acid sequence and, similar to most other mammals, lack a single amino acid residue in the N-terminal region compared with human VEGF. 15, 46 VEGF expression has been demonstrated by means of immunohistochemistry and polymerase chain reaction (PCR) and in a variety of canine neoplasms, 30, 42, 43, 46 including 2 canine grade IV astrocytomas 23 and a series of grade 1 meningiomas. 40 Few data exist describing the relative expression of VEGF isoforms in canine tissues, although all available data suggest that VEGF120, VEGF164, and VEGF188 are the most abundant isoforms, as in other mammalian species; VEGF120, VEGF164, and VEGF188 were shown by RT-PCR and sequencing to be the major isoforms in canine lung carcinoma, 46 and similar data were reported in canine heart using RT-PCR, ribonuclease protection assays, and Western blotting. 15 The objectives of this study were twofold: 1) to evaluate the mRNA expression of the major VEGF isoforms in a variety of primary canine CNS tumors using quantitative real-time TaqMan polymerase chain reaction and to correlate any mRNA expression with tumor type and grade, and 2) to determine whether there was an association between the level of VEGF mRNA expression in canine CNS tumors and the presence or severity of peritumoral edema evaluated by magnetic resonance imaging (MRI).
Materials and Methods
Sample collection
All tumor tissue was obtained from surgical biopsy/resection specimens, necropsy, or from archival paraffin-embedded material from clinical cases presented to the Veterinary Medical Teaching Hospital, University of California, Davis. Samples of tumor from necropsy were collected within 20 minutes of death and snap frozen in liquid nitrogen. Surgical samples were similarly stored following collection. Samples of adjacent tumor tissue were processed for routine paraffin embedding and histology whenever fresh tissue was collected in liquid nitrogen. Normal brain samples were collected from both necropsy and archival paraffin-embedded material. All tumors were graded by a board certified pathologist (RJH, AWB) essentially according to the international World Health Organization classification of human tumors of the nervous system. 18, 24, 25 Meningiomas were graded as either grade I (benign), grade II (atypical), or grade III (malignant); astrocytomas were graded as either grade II (diffuse), grade III (anaplastic), or grade IV (glioblastoma multiforme); oligodendroglial tumors were graded as either grade II (oligodendroglioma) or grade III (anaplastic oligodendroglioma). Clinical records were reviewed for evidence of corticosteroid administration prior to either MRI or acquisition of tumor samples.
Magnetic resonance imaging
MRI was done using either a 0.4T scanner (Resonex 5000, Resonex Inc., Sunnyvale, CA) or a 1.5T scanner (General Electric, Sigma, Milwaukee, WI). The imaging sequences were obtained from transverse 5-mm slices with an interslice gap of 1 mm generated with T1-weighted (T1W), T2-weighted (T2W), and proton-density weighted (PDW) spin echo pulse sequences. T1-weighted images were also acquired after the intravenous administration of gadolinium-diethylenetriaminepenta-acetic acid (Gd-DTPA; Magnevist, Berlex Laboratories, Wayne, NJ) at 0.2 mmol/kg. Peritumoral edema was determined to be present or absent based on the presence of hyperintensity on T2-weighted images beyond the tumor boundary. Tumor boundaries were determined based on T1W, T1W postcontrast, T2W, and PDW images. Edema was graded either as absent, focal if edema was present on 1 or 2 consecutive T2W images beyond the tumor margin, or diffuse if present on 3 or more consecutive images beyond the tumor margin.
Canine VEGF sequence
Canine VEGF cDNA sequence data and clones for VEGF isoforms VEGF120, VEGF164, and VEGF188 were kindly provided by Dr. Rolf Jaussi (Institute of Medical Radiobiology of the University of Zurich and the Paul Scherrer Institute, Zurich, Switzerland).
TaqMan PCR
Total RNA extraction, cDNA preparation, and real-time TaqMan PCR were done as previously described. 9 VEGF PCR primers for canine isoforms VEGF120, VEGF144, and VEGF188 were designed based on canine sequence data described above using Primer Express software (Applied Biosystems, Foster City, CA) and are listed and illustrated in Table 1 and Fig. 1. TaqMan PCR primers for canine housekeeping genes glyceraldehydes-3-phosphate dehydrogenase (GAPDH), ribosomal protein L13A, glycosyltransferase (HPRT1), and glucuronidase beta (GUSB) (Table 1) were designed and validated as previously described. 9 PCR products were designed to be less than 150 base pairs in length, with either one of the primer pairs or internal probe placed over an exon-exon junction to allow discrimination between cDNA and gDNA. PCR products were initially validated by agarose gel electrophoresis. Subsequently, products from triplicate PCR reactions, generated using control canine pooled tissue cDNA and each primer pair, were combined and purified (QIAquick PCR purification kit, QIAGEN Inc., Valencia, CA). Specificity of the PCR product was then confirmed by direct sequencing using an automated big dye termination sequencing procedure (Davis Sequencing, Davis, CA). Transcript quantitation was done using the comparative CT method and reported as relative transcription, or the n-fold difference relative to the mean value for individual normal cerebral cortex samples (n = 15). Tumor samples that had GAPDH CT values weaker than 3 times the average GAPDH CT value were considered low-quality cDNA samples and were discarded.
TaqMan PCR primers.

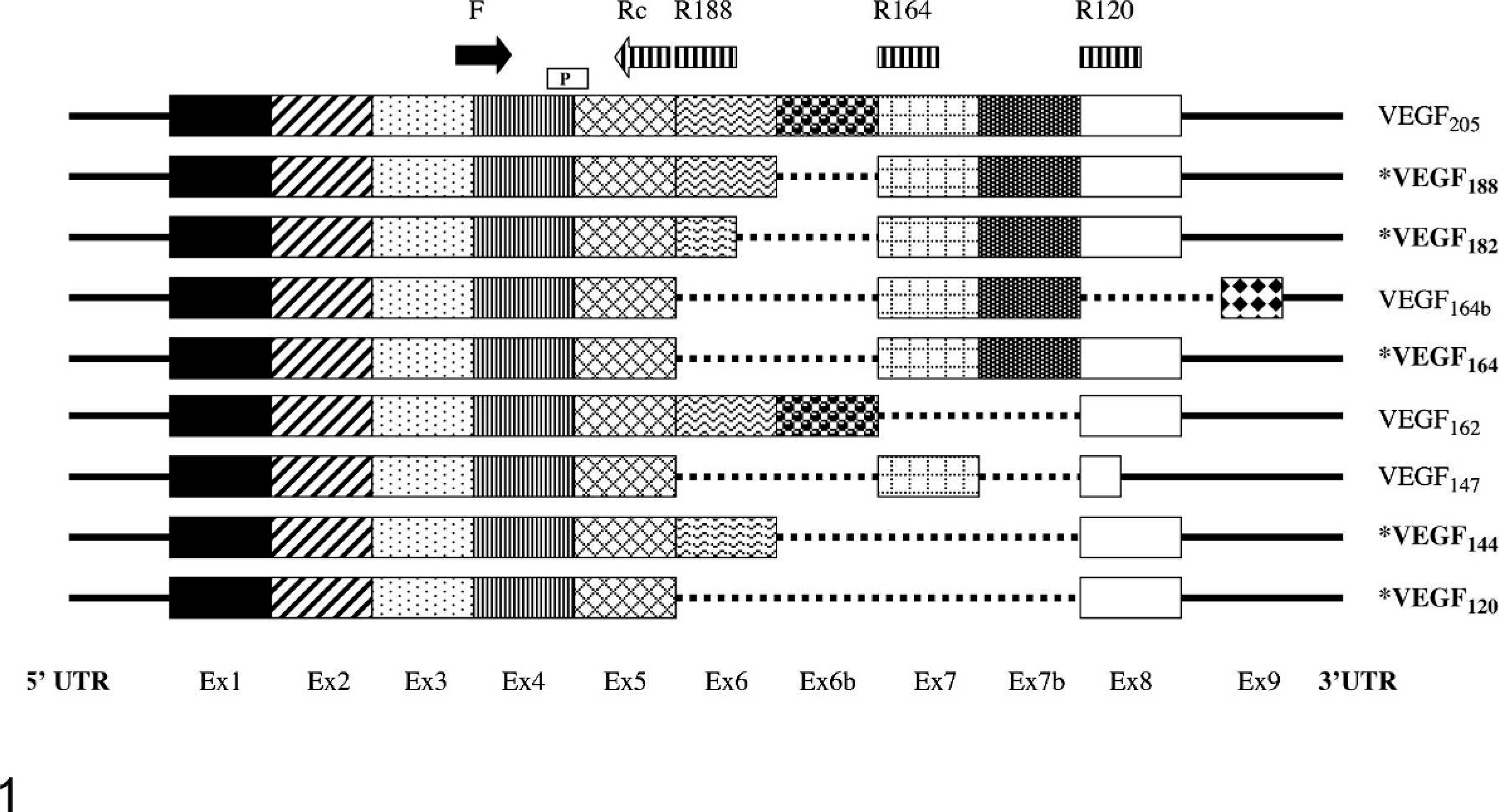
Schematic representation of the exon structure and splice variants of canine and human VEGF. Known canine splice variants are in bold marked with an asterisk (∗). Isoform lengths are numbered as for canine. The canine VEGF primers and probes were designed to amplify canine VEGF isoforms 120, 164, and 188 using a common forward primer (F), a common internal probe (P), and reverse primers containing a common terminus (Rc) located at the end of exon 5 linked to isoform-specific sequence in exon 8 (VEGF120), exon 7 (VEGF164), and exon 6 (VEGF188). It is unknown whether additional VEGF isoforms reported in humans are present in dogs. It is possible that the VEGF164 primers might also amplify VEGF164b and VEGF147, and the VEGF188 primers might amplify VEGF182, VEGF162, VEGF145, and VEGF205. Ex = exon, UTR = untranslated region. Sequence specificity was confirmed by direct sequencing.
Statistical analysis
Data were divided into nominal categories of tumor type, and for astrocytomas, they were further divided into ordinal categories of grade. Kruskal-Wallis one-way analysis of variance (ANOVA) was used to compare the variation in expression of VEGF mRNA among tumor types. When significant differences were evident, Mann-Whitney tests were used for pairwise comparisons. A sequentially rejective modification of Bonferroni's multiple comparison adjustment was used to confirm significant results. Variation in expression of specific VEGF isoforms within tumor groups was assessed using Friedman two-way ANOVA and Wilcoxon signed rank test for paired data. Peritumoral edema data were divided into ordinal categories of severity: 0 = no edema, 1 = focal edema, 2 = diffuse edema. Trends in expression of VEGF isoforms with severity of peritumoral edema were evaluated using the Jonckheere-Terpstra test. Evaluation of VEGF expression with presence or absence of edema was done using Mann-Whitney tests. The effect of presence or absence of corticosteroid administration on total VEGF expression and presence or absence of edema was assessed using Mann-Whitney tests. Statistical significance was defined as P ≤ .05.
Results
VEGF expression
PCR products from the 3 VEGF primer sets were of the appropriate size based on agarose gel electrophoresis, and direct sequencing of gel-extracted product resulted in single primer specific sequences corresponding to the 3 major isoforms, VEGF120, VEGF164, and VEGF188. VEGF PCR product was amplified from 56 separate samples from 56 dogs; 15 samples of normal cerebral cortex, 9 grade I meningiomas, 1 grade II (atypical) meningioma, 6 grade II astrocytomas, 5 grade IV astrocytomas (glioblastoma multiforme [GBM]), 2 grade II oligodendrogliomas, and 18 grade III (anaplastic) oligodendrogliomas (Fig. 2). Three grade I meningiomas and 1 grade III oligodendroglioma were of spinal cord origin. Averages of normalized values and the standard deviations for both frozen and paraffin-embedded samples were not significantly different.
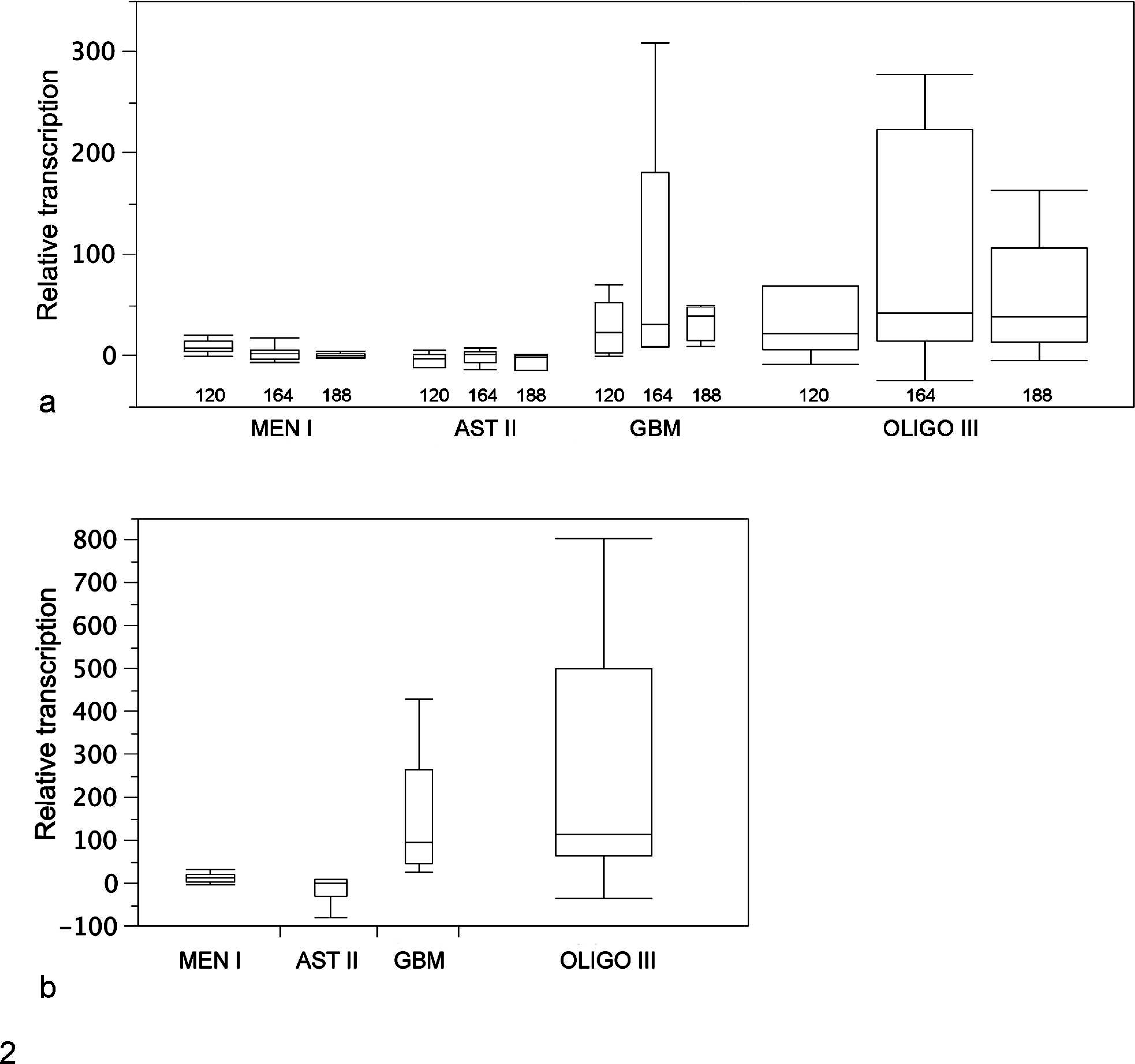
Expression of the major canine VEGF mRNA isoforms
Increased expression of VEGF transcripts was seen most often with high grade gliomas, including GBMs and oligodendrogliomas. Combined expression of all isoforms was significantly greater in GBMs than grade II astrocytomas (P = .026) and significantly greater in oligodendrogliomas than meningiomas (P = .009) or grade II astrocytomas (P = .001). There was no significant difference between total VEGF expression in GBMs and oligodendrogliomas (P = .25) (Fig. 2b). Expression of VEGF isoforms in the single atypical meningioma was similar to that in the grade I meningiomas. Expression of VEGF isoforms in the 2 grade II oligodendrogliomas were ranked the second and fifth lowest of all oligodendroglial samples as a single group. All 3 major VEGF isoforms were present in tumor samples, and VEGF164 was the predominant isoform in grade III oligodendrogliomas (P = .016) (Fig. 2a).
Information relating to administration of corticosteroids was available for 38/41 tumors. Corticosteroids of varying dose and duration were administered in 23/38 dogs prior to collection of tumor samples. There was no statistical difference in VEGF transcript levels in treated and untreated tumor samples when looking at individual tumor groups. Total VEGF expression with administration of corticosteroids approached significance when looking at all tumor groups combined (P = .055).
Peritumoral edema
MRI was available for 37 dogs from which tumor samples were analyzed. This included 9 meningiomas, 4 grade II astrocytomas, 5 GBMs, 2 grade II oligodendrogliomas, and 17 grade III oligodendrogliomas. Edema was absent in 2/9 meningiomas, 1/4 grade II astrocytoma, 2/2 grade II oligodendrogliomas, and 5/17 grade III oligodendrogliomas; focal in 4/9 meningiomas, 2/4 grade II astrocytomas, 5/5 GBMs, and 11/17 grade III oligodendrogliomas; diffuse in 3/9 meningiomas, 2/4 grade II astrocytomas, and 1/17 grade III oligodendrogliomas. No significant association was seen between edema scores or presence/absence of edema and increased expression of VEGF for any tumor group individually, or for all tumor groups combined.
Information relating to administration of corticosteroids was available for 35/37 MRI studies. Corticosteroids were administered in 12/37 dogs prior to MRI. There was no statistical association between the presence or absence of edema and corticosteroid administration.
Discussion
VEGF and its receptors are essential regulators of angiogenesis and vascular permeability and are postulated to play a major role in human tumor angiogenesis, carcinogenesis, invasion, and metastasis. 48 In the current study, we have demonstrated the presence of all 3 major VEGF mRNA isoforms in canine meningiomas, astrocytomas, and oligodendrogliomas and shown that VEGF mRNA expression is highest in high grade gliomas, specifically GBMs and grade III oligodendrogliomas.
Although 9 alternatively spliced human VEGF isoforms have been described (Fig. 1), 3 isoforms (VEGF121, VEGF165, and VEGF189) predominate in the majority of human tissues and tumors. In particular, VEGF165 and to a lesser extent VEGF121 have been shown to be the predominant isoforms expressed in several human tumors, including astrocytomas, oligodendrogliomas, and meningiomas. 3, 14, 32, 36 Individual isoforms have been shown to have distinct roles in vascular patterning and venous and arterial development; however, the roles of specific isoforms in neoplastic tissues remain unclear. The findings of the current study suggest that VEGF164 (canine VEGF isoforms are 1 aa residue shorter than their human counterparts) is the predominant mRNA isoform in canine CNS tumors, similar to their human tumor counterparts, at least when significant overexpression is present. This predominance also parallels a previous study where VEGF164 was found to be the major isoform in the canine heart. 15 The TaqMan PCR primers designed to amplify VEGF120, VEGF164, and VEGF188 for this study could potentially have amplified other additional isoforms based on human data (Fig. 1). Specifically, the VEGF164 primers might also amplify VEGF147 and VEGF164b, and the VEGF188 primers might amplify VEGF145, VEGF162, VEGF182, and VEGF205. The VEGF120 primers should not amplify any other published human isoforms. Of these isoforms, only VEGF182 has been described in dogs; 15 however, it is likely that other isoforms are also present and remain to be documented. Isoforms other than VEGF121, VEGF165, and VEGF189 have only been described in relatively small amounts in humans. Agarose gel analysis and sequencing of canine products generated with the current primer sets revealed single bands with product sequences consistent with the expected major VEGF isoforms, making the presence of significant amounts of most of the potential additional isoforms unlikely.
Increased expression of VEGF mRNA in canine high grade gliomas is not surprising since the presence of microvascular proliferation, presumably driven in part by VEGF, is a histologic criterion for their classification. Similar findings have been reported in human studies with high grade gliomas having greater expression than meningiomas, 13, 14, 49 and GBMs and high grade (III) oligodendrogliomas having greater expression than low grade astrocytomas and low grade (II) oligodendrogliomas. 5 Some studies, however, reported no marked differences in VEGF expression between different tumor types and grades. 45 Only 2/20 oligodendrogliomas in this study were low grade, and statistical analysis was not possible; however, VEGF mRNA expression for these samples were the second and fifth lowest values for the oligodendroglial tumors. Care should be taken when attempting to interpret individual samples since transcription levels were assessed for small samples of the whole tumor, and VEGF mRNA expression may vary markedly in different regions of a tumor depending on intratumoral factors such as necrosis and hypoxia. As such, significant variation between individual tumors is to be expected, as seen in this study, and overall conclusions should be restricted to describing broad trends in tumor types and grades as a whole. Although the current study demonstrates increased VEGF mRNA expression in high grade canine tumors, no specific comments can be made relating to VEGF protein expression. However, increased levels of both VEGF mRNA and protein have been documented independently in a variety of human CNS tumors, 6, 7, 13, 14, 19, 21, 29, 33, 39, 45, 49, 52, 54 and some studies have specifically demonstrated elevated VEGF protein associated with elevated VEGF mRNA in the same tumor samples. 36, 38 It is therefore likely that the general patterns seen in VEGF mRNA expression in canine CNS tumors will be reflected in similar studies quantifying protein expression.
The effect of corticosteroid administration on VEGF expression is poorly defined. Corticosteroids have been shown to decrease VEGF expression, predominantly at the transcriptional level in vitro; 8 however, in vivo, human tumors with peritumoral edema may express VEGF whether or not pretreated with corticosteroids. 27, 47, 51 No significant difference was seen within individual tumor groups when comparing levels of VEGF mRNA expression in corticosteroid treated to untreated samples in the current study. However, decreased total VEGF expression with corticosteroid administration approached significance (P = .055) when looking at all tumors combined. Dosage and duration of corticosteroid administration varied markedly (from less than 24 hours to several weeks) between individual dogs, and it is possible that VEGF mRNA expression could have been decreased following treatment in certain tumors. However it is unlikely that this had a significant effect on the broad conclusions made regarding VEGF mRNA expression in the different canine tumors.
VEGF is known to be a potent factor resulting in increased vascular permeability and has been associated with the presence of peritumoral vasogenic edema in human CNS tumors. No significant association between the level of VEGF mRNA expression and the degree or presence of peritumoral edema was seen in this study for any group of tumors, although MRI data were only available for a subset of the tumor samples and overall sample numbers were low. Several human studies have reported a correlation between VEGF expression and peritumoral edema 47 or vascular permeability 26 in human gliomas, and the decreased edema following treatment with specific inhibitors of VEGFR tyrosine kinase receptors in human clinical trials is strong circumstantial evidence for a major role of VEGF in edemagenesis. 1 However, a lack of correlation also has been reported. 51 A more consistent correlation between VEGF expression and edema has been reported for human meningiomas, 12, 16, 34, 35, 37, 41 although several other factors have been implicated, including tumor size and location, presence of a cerebral pial blood supply, 4, 53 other cytokines, vascular obstruction, and intrinsic permeability differences in tumor blood vessels. None of these additional factors were assessed in the current study. As discussed above, the exact role of corticosteroid administration on VEGF expression and its subsequent effects on peritumoral edema in vivo are unclear. Although the limited data in the current report must be interpreted with caution, no significant differences were seen between corticosteroid treated and untreated tumors and the presence or degree of edema, in either canine meningiomas or gliomas, and it is likely that the presence of peritumoral edema, similar to human tumors, is dependent on many factors in addition to VEGF expression.
Both presence 31, 33, 54 and absence 19, 50 of a correlation between VEGF expression and clinical outcome has been reported for individual human patients with specific types and grades of CNS tumors, and high VEGF expression has been associated with poor outcome in canine grade I meningiomas. 40 No attempt was made to correlate prognosis with VEGF mRNA expression in the current study; however, intuitively, the higher level of expression in high grade gliomas as a group is likely to be associated with a poorer prognosis relative to the lower expression in lower grade gliomas and meningiomas.
VEGF, with its multiple isoforms, each potentially having multiple effects, is one factor in a family of proteins involved in the highly complex processes of angiogenesis, carcinogenesis, and control of vascular permeability in CNS tumors. We have previously shown that mRNAs for the 2 major tyrosine kinase receptors for VEGF (VEGFR-1, VEGFR-2) are overexpressed in many canine CNS tumors, particularly high grade gliomas. 9 The current study further supports a major role for VEGF in the biology of these tumors and reinforces the striking similarities between canine CNS tumors and their human counterparts. Although the validity and efficacy of VEGF targeted therapy in neuro-oncology has still to be proven, these data support the further investigation of VEGF expression as a prognostic marker and therapeutic target in canine primary CNS tumors.
Footnotes
Acknowledgements
This research was supported by the Paul C. and Borghild T. Petersen Foundation and the Center for Companion Animal Health, University of California, Davis, CA. We are grateful to John Doval for his assistance with the illustrations.
