Abstract
Mycobacterium bovis, the causative agent of bovine tuberculosis, persists within granulomas. Formation of granulomas involves a complex array of immune activation and cellular migration. To examine temporal changes in granuloma development, we inoculated 32 cattle with M. bovis of deer origin. Tissues from 4 calves each were examined at 15, 28, 42, 60, 90, 180, 270, and 370 days after inoculation. Granulomas in the medial retropharyngeal lymph node were staged (I–IV) on the basis of cellular composition and the presence or absence of necrosis and peripheral fibrosis. Immunohistochemistry for inducible nitric oxide synthase (iNOS), CD68, CD4, CD8, and γ/δ T cells was performed. Fifteen days after inoculation only stage I granulomas were seen, while between 28 and 60 days, there was a steady progression through granuloma stages such that by day 60, granulomas of all 4 stages were seen. Acid-fast bacilli were present in moderate-to-large numbers in stage I granulomas 15–60 days after inoculation. Stage IV granulomas contained large numbers of acid-fast bacteria. Abundant iNOS immunoreactivity was associated with granulomas from day 15 through day 60 but was minimal from day 90 to the termination of the experiment. The relative number of CD4+ and CD68+ cells remained constant throughout the study. In contrast, at time points >60 days, numbers of CD8+ and γ/δ T cells diminished. Tuberculous granulomas are dynamic lesions that follow an orderly progression through disease stages. Diminished expression of iNOS and reduced numbers of CD8+ and γ/δ T cells late in the progression of tuberculous granulomas may represent a failure of the host response to control infection.
Keywords
Mycobacterium bovis, the causative agent of bovine tuberculosis, infects over 50 million cattle world-wide and results in economic losses of over $3 billion annually. 42 Within the host, M. bovis is known to persist within granulomas, distinct lesions represented by a caseonecrotic core surrounded by epithelioid macrophages, T cells, B cells, Langhans-type multinucleated giant cells, and fibroblasts. 43 Survival of the host depends on the ability to limit mycobacterial proliferation through effective granuloma formation. Such formation involves a complex array of migration and activation of immune cells, which is initiated by cytokine expression triggered by mycobacterial antigens. 43
It is widely accepted that protective immunity to mycobacterial agents depends on interactions between macrophages and T cells. 21 The dynamic response of circulating T-cell subsets has been investigated in experimentally infected cattle. 35 During the first 16 days after inoculation, there is a decrease in γ/δ T cells, followed by an increase in γ/δ T cells. These changes are followed by an increase in the CD4 : CD8 T-cell ratio, which peaks 44 days after inoculation. 35 By 95 days after inoculation, the ratio decreases to below preinoculation levels. Changes in the CD4 : CD8 ratio are due to an initial increase in CD4 T cells, followed by an increase in CD8 T cells. 35
Other cattle studies examining changes at the site of lesion development have shown an overall decrease in total T cells, with an increase in CD68+ cells and γ/δ T cells in morphologically advanced granulomas, compared with morphologically immature granulomas from cattle examined 29 weeks after inoculation. 45 A separate study showed that γ/δ T cells were most numerous in developing lesions up to 21 days after inoculation. 4 Increases in transforming growth factor-beta and type I procollagen were also seen in more advanced granulomas. 45
Nitric oxide (NO), the product of the enzyme nitric oxide synthase (NOS), is a highly reactive nonpolar gas that leads to a number of biochemical reactions that ultimately result in inactivation of various pathogens. 6 NO has been found to be involved in granuloma formation of both infectious and noninfectious granulomas of humans and animals. 12 However, NO has not been implicated in all types of granulomas; for example, evidence of NO is not detectable in foreign body granulomas and nonspecific granulomatous lymphadenitis. 12 In mammalian cells, NO production is catalyzed by 3 distinct isoenzymes: inducible NOS (iNOS), endothelial NOS, and neuronal NOS. 6 In macrophages and other cells, iNOS is stimulated by endotoxin, cytokines, and other activators. 17 Once produced, NO reacts with superoxide to generate peroxynitrite and other reactive nitrogen intermediates. Peroxynitrite is a powerful oxidant, which damages proteins, lipids, membranes, and deoxyribonucleic acid. 6
Murine macrophages have been shown to inhibit growth and kill M. bovis bacillus Calmette Guerin (BCG) and Mycobacterium tuberculosis in an interferon-gamma (IFN-γ)–dependent mechanism through the production of NO. 5, 13 Inhibition was abrogated through the use of arginine inhibitors that prevent NO synthesis. 5, 13 Although experimental evidence exists that NO plays a role in controlling infection with members of the M. tuberculosis complex 5, 13, 28 and bovine macrophages have been shown to express iNOS in vitro 2, 17, 47 and in vivo, 14, 16, 19 the expression of iNOS in bovine tuberculosis appears to be distinct from that seen in the murine tuberculosis model. In contrast to those in mice, bovine macrophages require additional signals from bacterial products (e.g., lipopolysaccharide, lipoarabinomannan) in addition to IFN-γ for maximal NO production. 18, 20 Moreover, in vitro, it appears that levels of NO produced by bovine macrophages are insufficient to control intracellular replication of virulent M. bovis. 3, 9
Previous studies in cattle have examined in vitro responses of M. bovis-infected macrophages, temporal changes in circulating leukocytes, or in situ cellular composition of granulomas in cattle at a single fixed time point after inoculation; however, few studies have examined temporal changes in tuberculous lesions from cattle experimentally inoculated with the same dose of M. bovis and examined at various time points after inoculation. The objective of the current study was to examine the morphologic and cellular changes in granulomas of the medial retropharyngeal lymph nodes of cattle intratonsilarly inoculated with M. bovis at various times after inoculation. The intratonsilar method of inoculation provides an ideal model for such an evaluation as inoculum is placed in a small defined area (palatine tonsillar crypt), resulting in reproducible lesions in an organ of suitable size to allow for thorough examination (medial retropharyngeal lymph node). 30, 33
Materials and Methods
Inoculum preparation
M. bovis strain 1315 was used in the present study. Strain 1315 was originally isolated from a white-tailed deer in northeast Michigan. Virulence, lesion distribution, lesion severity, and disease progression have been shown to be similar in cattle inoculated with strain 1315 of deer origin compared with cattle inoculated with isolates of bovine origin. 31 Inoculum consisted of mid-log-phase M. bovis grown in Middlebrook's 7H9 liquid media supplemented with 10% oleic acid-albumin-dextrose complex (Difco, Detroit, MI) plus 0.05% Tween 80 (Sigma Chemical Co., St. Louis, MO) grown for 10 days at 37°C. To harvest bacilli from the culture media, we pelleted cells by centrifugation at 750 × g and washed them twice with 1 ml of phosphate-buffered saline solution (0.01 M, pH 7.2) and diluted them to the appropriate density. Enumeration of bacilli was by serial dilution plate counting on Middlebrook's 7H11 selective media (Becton Dickinson, Cockeysville, MD). Inoculum was stored in 1-ml aliquots at −80°C until used. Twenty-four hours after freezing, a single 1-ml aliquot was removed, and bacilli were enumerated by plate counting of serial dilutions on Middlebrook's 7H11 medium. At the time of inoculation, aliquots of inoculum were thawed and diluted to the appropriate concentration on the basis of plate counts as described above. Plate counts were repeated the day of inoculation to retrospectively confirm inoculum dosage.
Animals and experimental inoculation
Thirty-two, 6-month-old, cross-bred cattle were selected from a tuberculosis-free source on the basis of negative reactivity to M. bovis purified protein derivative using the Bovigam (Prionics AG, Schlieren, Switzerland) assay for IFN-γ production as per manufacturer's instructions. Cattle were inoculated intratonsilarly with 1 × 105 colony forming units of M. bovis. Four calves each were euthanized and examined 15, 28, 42, 60, 90, 180, 270, and 370 days after inoculation. At necropsy, the following tissues were collected and processed for isolation of M. bovis and microscopic analysis as described 32 : palatine tonsil, lung, liver, mandibular, parotid, medial retropharyngeal, tracheobronchial, mediastinal, hepatic, mesenteric and prefemoral lymph nodes, nasal turbinate, spleen, kidney, and brain. Lymph nodes were cross-sectioned at 0.5-cm intervals and examined. Each lung lobe was examined separately and cross-sectioned at 0.5- to 1.0-cm intervals. Lungs and lymph nodes were subjected to semiquantitative scoring of gross lesions adapted from previous studies. 44 Lung lobes (left cranial, left caudal, right cranial, right caudal/middle, and accessory) were subjected to the following scoring system: 0, no visible lesions; 1, no external gross lesions, but lesions seen upon slicing; 2, <5 gross lesions of <10 mm in diameter; 3, >5 gross lesions of <10 mm in diameter; 4, >1 distinct gross lesion of >10 mm in diameter; and 5, coalescing gross lesions. Scoring of lymph node gross lesions was based on the following scoring system: 0, no visible lesions; 1, small focal lesion (1–2 mm in diameter); 2, several small foci; and 3, extensive lesions. Tissues collected for microscopic analysis were fixed by immersion in 10% neutral buffered formalin and included all tissues collected for bacteriologic examination. For microscopic examination, formalin-fixed tissues were processed by routine paraffin-embedment techniques, cut in 5-μm sections, and stained with HE. Adjacent sections were cut from samples containing lesions suggestive of tuberculosis (caseonecrotic granulomata) and stained by the Ziehl-Neelsen technique for identification of acid-fast bacteria, the Masson trichrome method for collagen, and the Von Kossa method for calcium salts. When present, microscopic granulomas were staged (I–IV) on the basis of adaptations of criteria previously described. 37, 45 Stage I (initial) granulomas were characterized by accumulations of epithelioid macrophages with low numbers of lymphocytes and neutrophils. Multinucleated giant cells may have been present, but necrosis was absent. Acid-fast bacilli, when present, were seen within macrophages or multinucleated giant cells. Stage II (solid) granulomas were characterized by accumulations of epithelioid macrophages surrounded by a thin connective tissue capsule. Infiltrates of neutrophils and lymphocytes may have been present as well as multinucleated giant cells. Necrosis when present was minimal. Acid-fast bacilli, when present, were seen within macrophages or multinucleated giant cells, as well as within necrotic material. Stage III (necrotic) granulomas were characterized by complete fibrous encapsulation. Necrotic cores were surrounded by a zone of epithelioid macrophages admixed with multinucleated giant cells and lymphocytes. Stage IV (necrotic and mineralized) granulomas were characterized by a thick fibrous capsule surrounding irregular multicentric granulomas with multiple necrotic cores. Necrotic cores contained foci of dystrophic mineralization. Epithelioid macrophages and multinucleated giant cells surrounded necrotic areas, and there were often moderate-to-marked infiltrates of lymphocytes. Acid-fast bacilli were often present in moderate numbers and primarily located within the caseum of the necrotic core.
For immunohistochemistry, samples of medial retropharyngeal lymph nodes and lung were collected both by snap-freezing in liquid nitrogen-cooled isopentane as well as fixed by immersion in 10% neutral buffered formalin as described above.
Immunohistochemistry
Formalin-fixed sections of medial retropharyngeal lymph nodes from each animal were processed for immunohistochemistry for iNOS and CD68 (Table 1) as described previously. 1, 16 Frozen sections of medial retropharyngeal lymph nodes were processed for immunohistochemistry as described previously, 15, 22 using antibodies to the T-cell markers CD4, CD8, and γ/δ (Table 1). The percentage of immunolabeled T cells for each of the antibodies was estimated according to the following scale: no labeling (−), 5% or less (±), 6–20% (+), 21–50% (++), and more than 50% labeled cells (+++).
Primary/secondary antibodies, suppliers, and conditions.
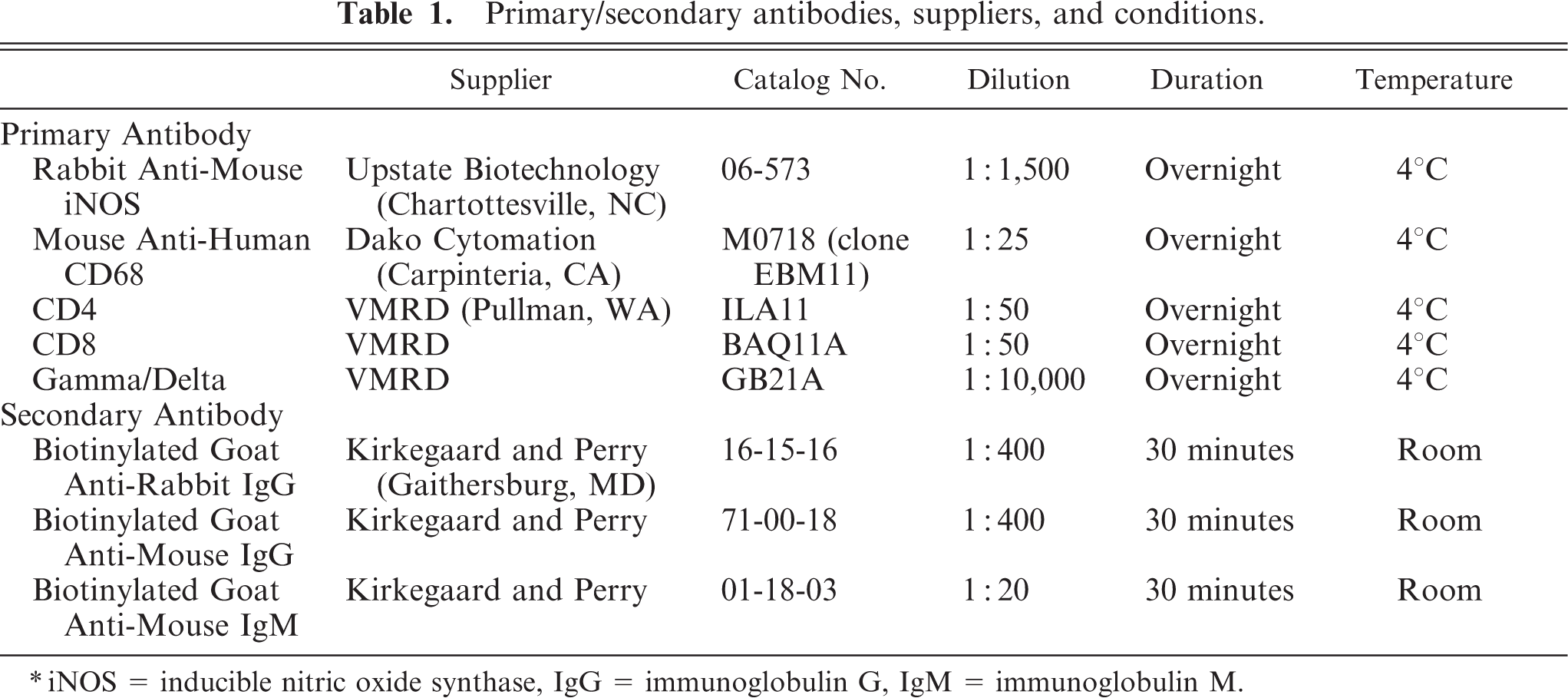
∗iNOS = inducible nitric oxide synthase, IgG = immunoglobulin G, IgM = immunoglobulin M.
Morphometry
Digital images of sections from all medial retropharyngeal lymph nodes containing microscopic lesions consistent with M. bovis infection were obtained with a light microscope (Nikon Eclipse E800, Nikon Co., Tokyo, Japan) and digital camera (Spot RT, Diagnostic Instruments Inc., Sterling Heights, MI). Images were transferred to a computer with software for image analysis (Image Pro-Plus, Media Cybernetics, Silver Spring, MD) to calculate the total granuloma area per slide and area of granuloma demonstrating iNOS immunoreactivity. Distances were calibrated for each microscope objective with the image analysis software and a standard micrometer bar. iNOS immunolabeling was calculated by instructing the software to identify positive iNOS labeling (black DAB-Ni reaction product, Vector, Burlingame, CA). Similarly labeled cells were then electronically identified, and the total area of iNOS immunolabeling was computed. Inclusion of background labeling was avoided by careful identification of iNOS immunolabeling in the software instruction phase.
Statistical analysis
Mean group values for lesion scores or iNOS immunoreactivity were compared using one-way repeated measures analysis of variance (GraphPad Prism, GraphPad Software, San Diego, CA). Differences between means were then compared using the Bonferonni method. A P value < .05 was considered significant.
Results
Lesion development
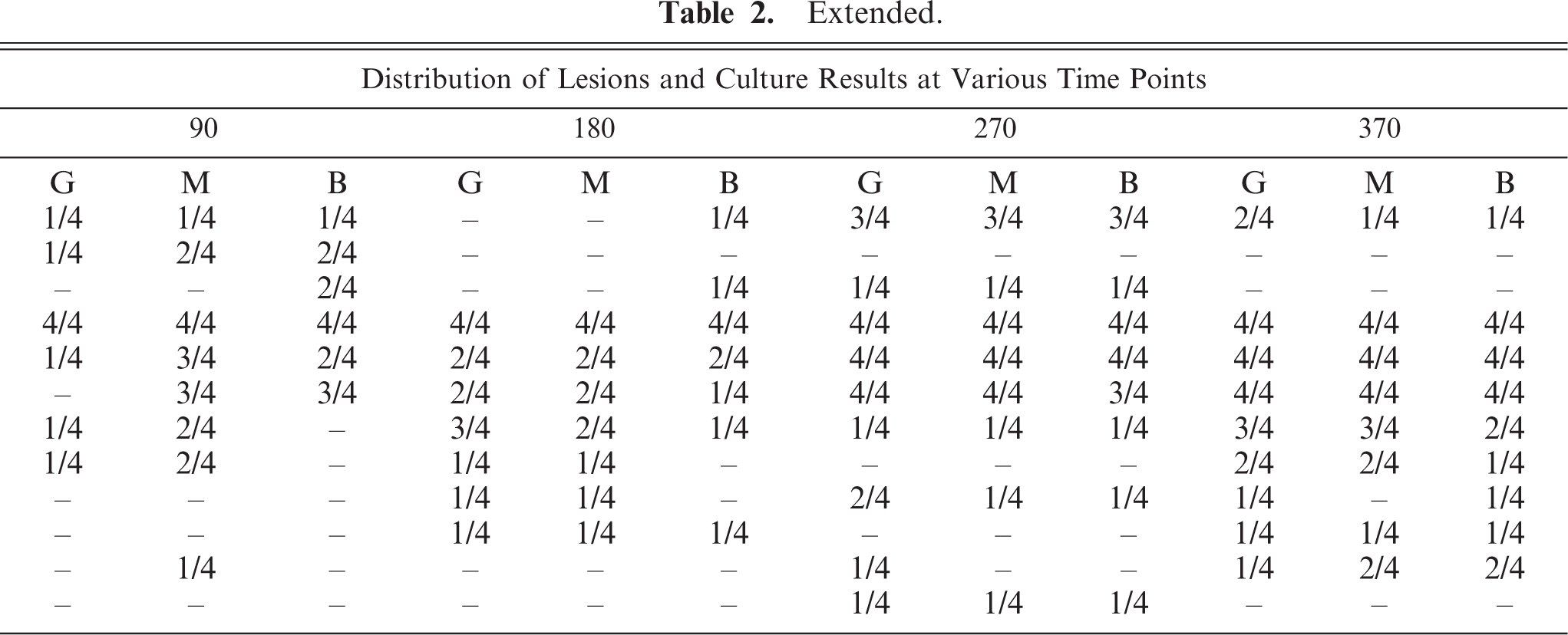
M. bovis was isolated from medial retropharyngeal lymph nodes as early as 15 days after inoculation (Fig. 1). Microscopic lesions consistent with tuberculosis were seen in medial retropharyngeal lymph nodes of 2/4 calves 15 days after inoculation and in 4/4 calves 28 days after inoculation and at every time point thereafter ( Table 2). Gross lesions were first seen in the medial retropharyngeal lymph node 28 days after inoculation and in the mediastinal and tracheobronchial lymph nodes 42 days after inoculation in 2/4 and 1/4 calves, respectively. Gross lesion scores increased rapidly between 15 and 42 days after inoculation in the medial retropharyngeal lymph node (Fig. 2) and lung (Fig. 3), remaining relatively constant thereafter. Gross lesions of lymph nodes were generally caseonecrotic in nature, with liquefactive lesions first seen 42 days after inoculation (Fig. 1). Liquefactive lesions in the medial retropharyngeal lymph nodes were seen in 1–2 calves at each time point between 42 and 270 days and in all 4 calves 370 days after inoculation. Microscopically, central necrosis was seen in lymph node granulomas as early as 28 days after inoculation. Peripheral fibrosis, demonstrated by Masson trichrome staining, and central mineralization, demonstrated by Von Kossa staining, were first seen 60 days after inoculation (Fig. 1).
∗Four calves were examined at each time point.
†G = gross, M = microscopic, B = bacteriologic, – = no isolation of M. bovis and no gross or microscopic lesions present; LN = lymph node.
Extended.

Schematic representation of the chronology of morphologic and immunohistochemical changes in the medial retropharyngeal lymph nodes of calves inoculated intratonsilarly with 1 × 105 colony forming units of Mycobacterium bovis and examined at various times after inoculation.
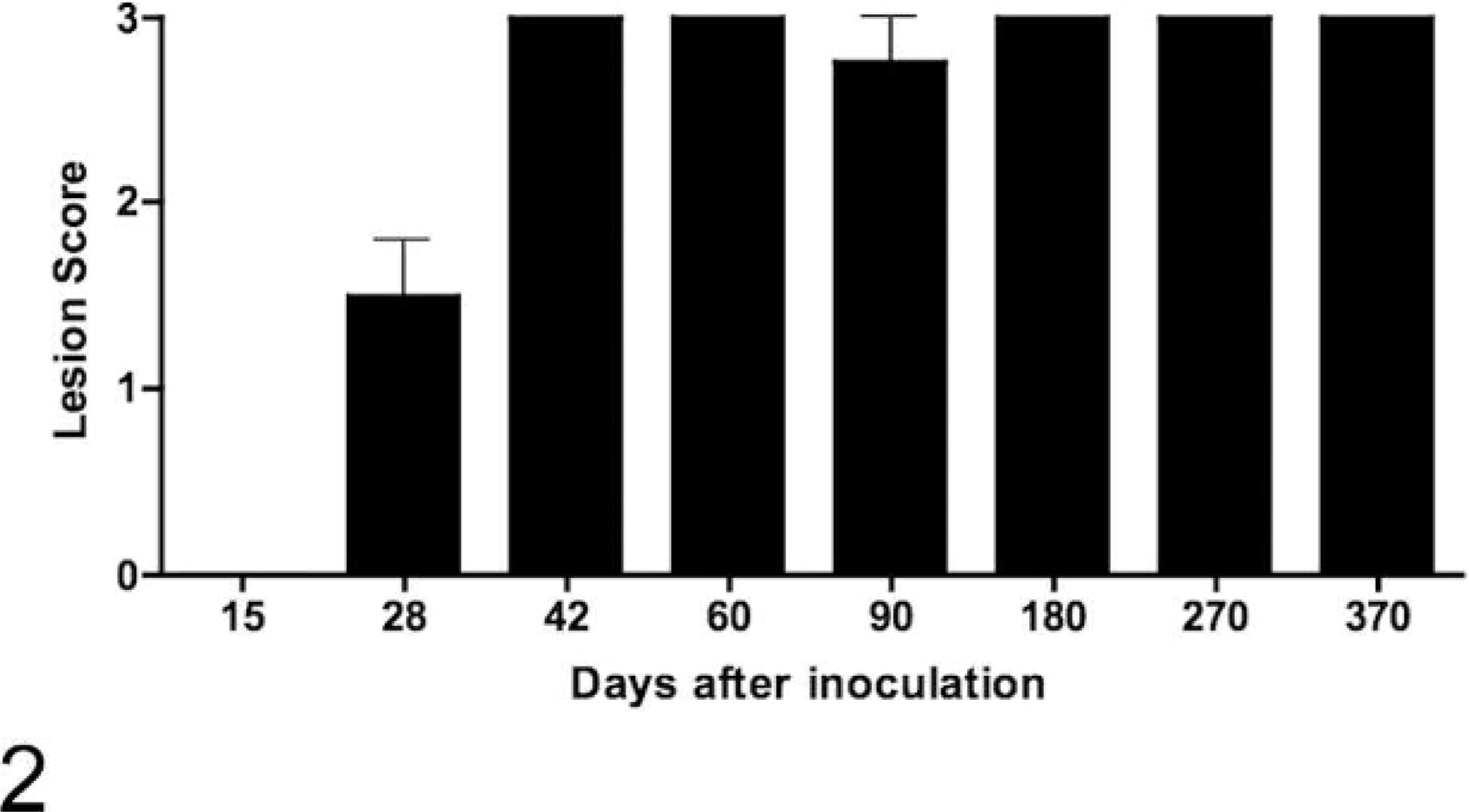
Gross lesion scores of medial retropharyngeal lymph nodes of calves inoculated intratonsilarly with 1 × 105 colony forming units of Mycobacterium bovis and examined at various times after inoculation. Data represent the mean ± SE of 4 calves at each time point.
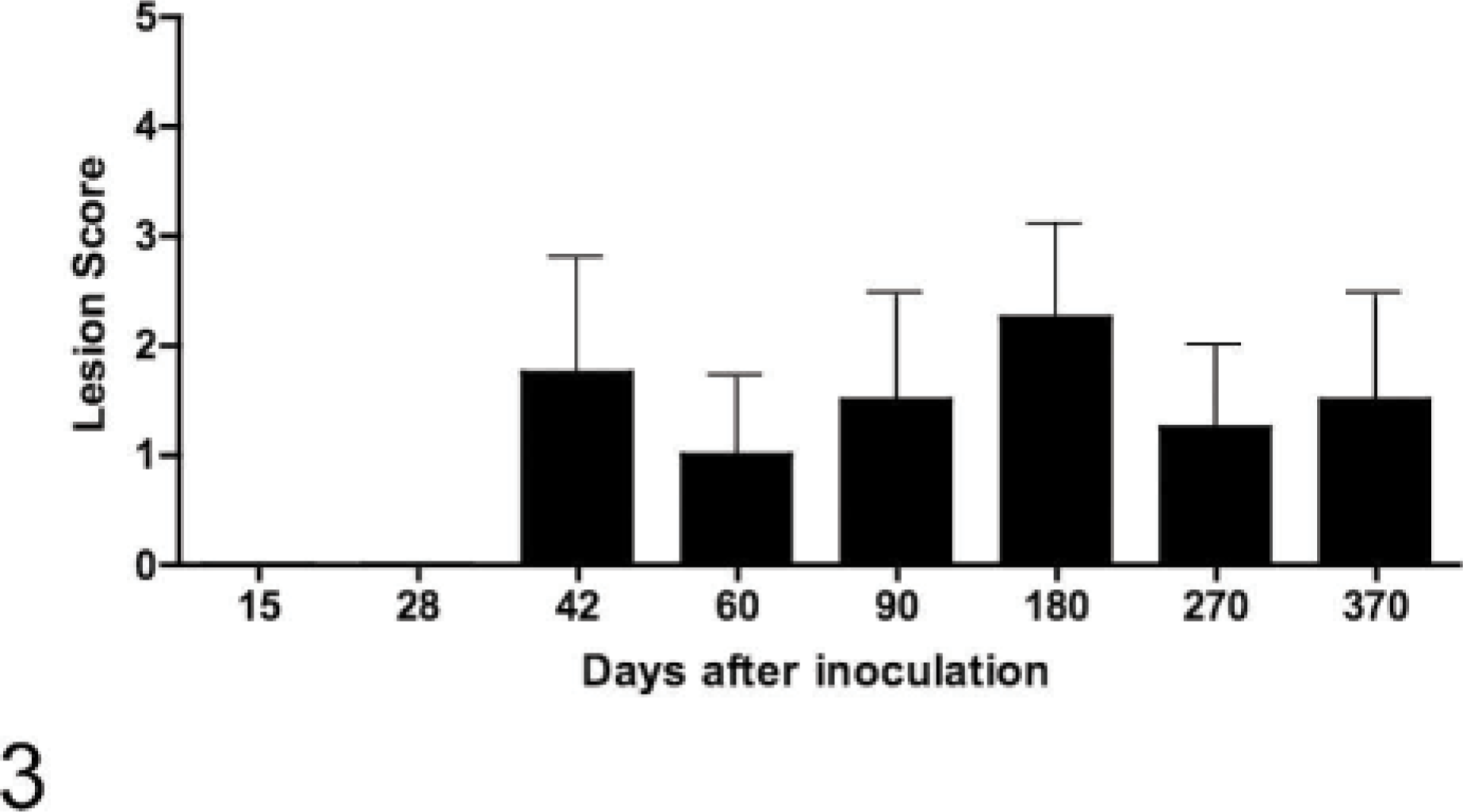
Gross lesion scores of lungs of calves inoculated intratonsilarly with 1 × 105 colony forming units of Mycobacterium bovis and examined at various times after inoculation. Data represent the mean ± SE of 4 calves at each time point.
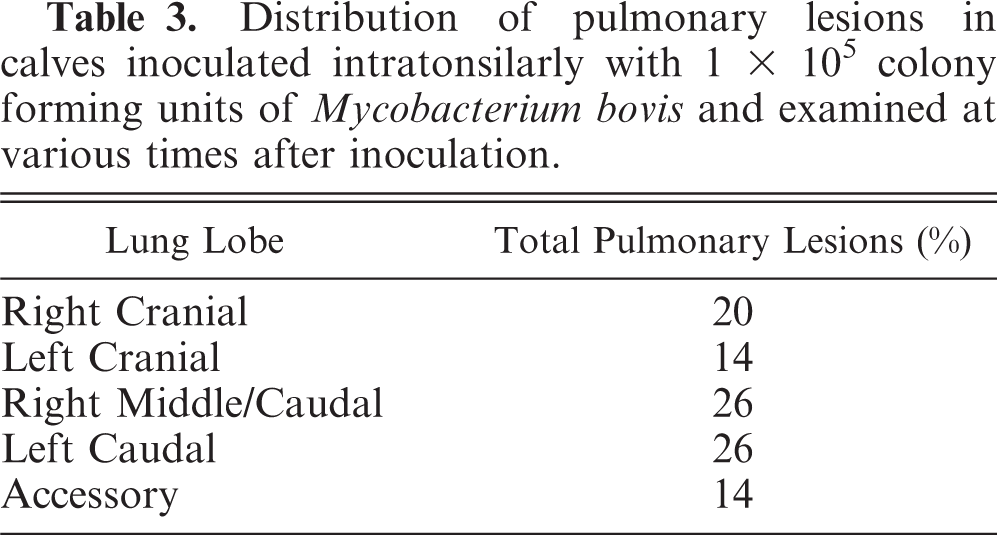
Gross and microscopic lesions were first seen in the lungs 42 days after inoculation. Fifty-two percent of pulmonary lesions were present in the left caudal and right caudal/middle lung lobes combined (Table 3), with the right cranial lobe being the third most common lung lobe with 20% involvement. M. bovis was first isolated from extracranial (not tonsils or lymph nodes of the head) and extrapulmonary sites 42 days after inoculation when M. bovis was isolated from 3/4 hepatic lymph nodes, 1/4 mesenteric lymph nodes, and 1/4 liver samples. Gross or microscopic lesions were first seen in extracranial and extrapulmonary sites 60 days after inoculation (Table 2).
Distribution of pulmonary lesions in calves inoculated intratonsilarly with 1× 105 colony forming units of Mycobacterium bovis and examined at various times after inoculation.
Granuloma formation
Fifteen days after inoculation, only stage I granulomas were seen within medial retropharyngeal lymph nodes (Table 4). Between 28 days after inoculation and 60 days after inoculation, there was a steady advancement through granuloma stages such that by 60 days after inoculation granulomas of all 4 stages were seen. Through the termination of the study, variable numbers of all 4 stages of granulomas were seen. In later time points (>60 days), stage I granulomas were often seen in proximity to granulomas of more advanced stages (III–IV), characteristic of “satellite granulomas” (Fig. 4). Acid-fast bacilli were present in moderate-to-large numbers in stage I granulomas 15 to 60 days after inoculation (Table 5). Bacilli were seen within macrophages and multinucleated giant cells. In contrast, stage I granulomas present ≥90 days after inoculation (satellite granulomas) contained few-to-no acid-fast bacilli. With the exception of day 180, stage IV granulomas contained large numbers of acid-fast bacteria (Table 5). Invariably, acid-fast bacilli were present extracellularly within the necrotic caseum of stage IV granulomas and rarely within macrophages or multinucleated giant cells.
Summary of histologic staging of granulomas (stages are defined in the text) within the medial retropharyngeal lymph node of cattle experimentally inoculated with Mycobacterium bovis and examined at various time points after inoculation.

Summary of numbers of acid-fast bacilli within granulomas of various stages of development (stages are defined in the text) in medial retropharyngeal lymph nodes of cattle experimentally inoculated with Mycobacterium bovis and examined at various time points after inoculation.

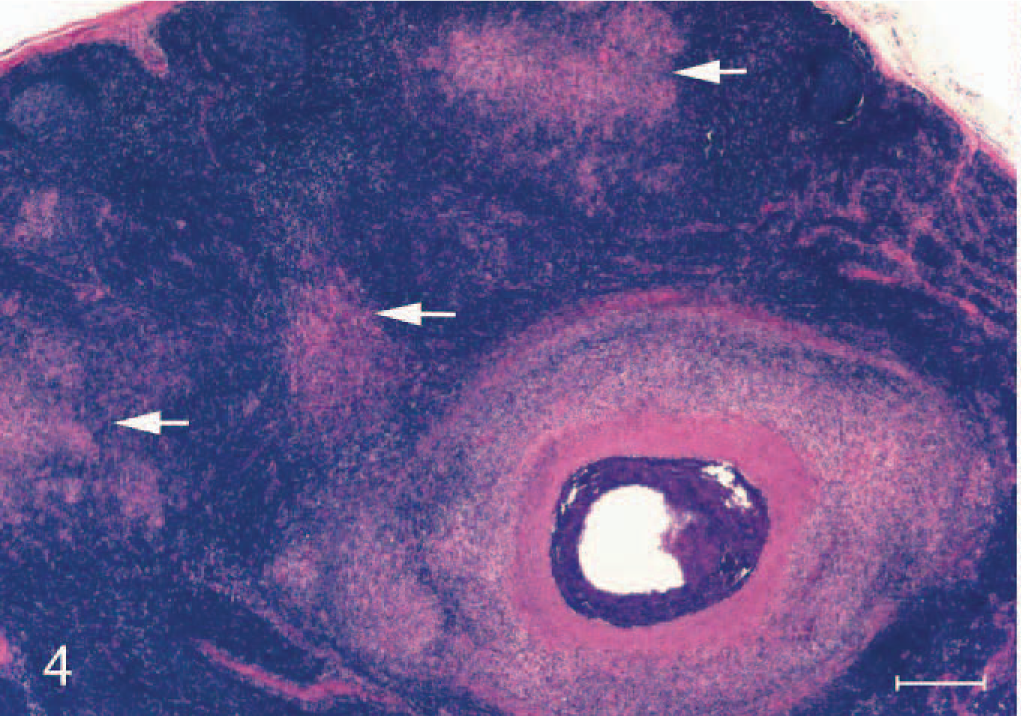
Section of medial retropharyngeal lymph node from a calf inoculated intratonsilarly with 1 × 105 colony forming units of Mycobacterium bovis and examined 180 days after inoculation. Note stage III caseonecrotic granuloma with central mineralization and peripheral fibrosis with nearby stage I satellite granulomas (arrows) composed of macrophages and multinucleated giant cells. HE. Bar = 250 μm.
iNOS and cell subsets
Immunoreactivity for iNOS was present with the appearance of the first microscopic lesions 15 days after inoculation. iNOS-positive cells consisted of macrophages and multinucleated giant cells (Fig. 5). Abundant iNOS immunoreactivity was associated with granulomas through day 60 but was minimal from day 90 after inoculation to the termination of the experiment (Fig. 6). In granulomas containing necrotic centers, iNOS immunoreactivity was noticeably limited to a narrow cellular zone surrounding the necrotic caseum. Immunoreactive cells with morphologic characteristics of activated macrophages were seen as well as other cells with distinct spindloid morphology (Fig. 7). In all cases with positive iNOS immunoreactivity, staining was invariably limited to cells associated with the granuloma and absent in the surrounding tissue. CD68+ cells (activated macrophages) were a prominent feature of granulomas at all time points examined (Fig. 8).
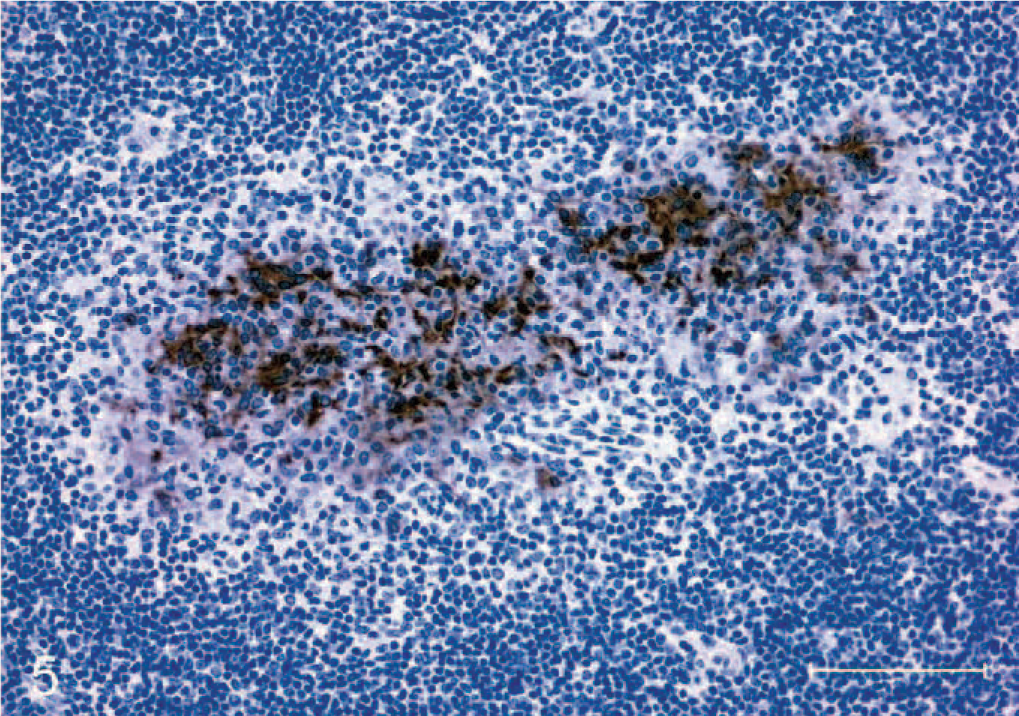
Section of medial retropharyngeal lymph node from a calf inoculated intratonsilarly with 1 × 105 colony forming units of Mycobacterium bovis and examined 15 days after inoculation. Note granuloma composed of macrophages and multinucleated giant cells demonstrating strong inducible nitric oxide synthase immunoreactivity. Immunoperoxidase method, Harris hematoxylin counterstain. Bar = 100 μm.
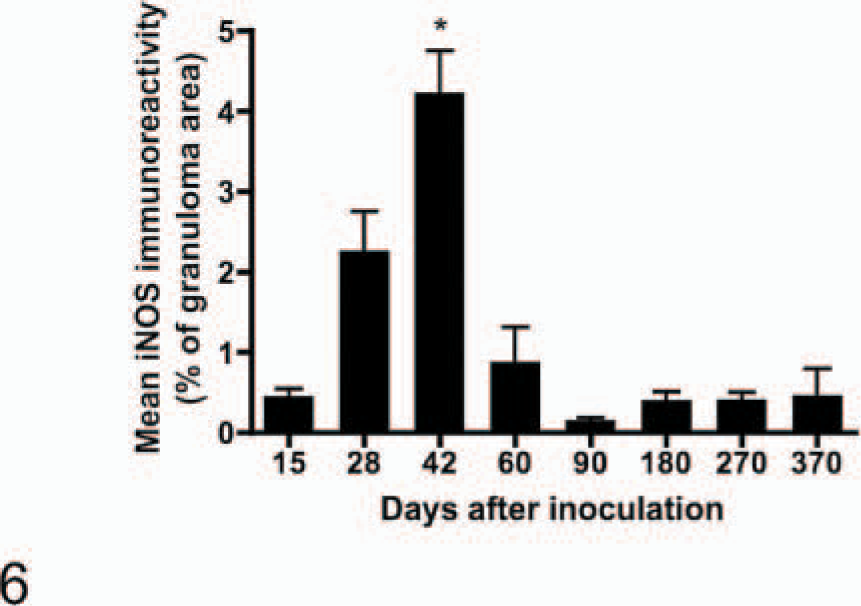
Area of granulomas within medial retropharyngeal lymph nodes demonstrating inducible nitric oxide synthase immunoreactivity. Calves were inoculated intratonsilarly with 1 × 105 colony forming units of Mycobacterium bovis and examined at various times after inoculation. Data points represent the mean ± SE of lymph nodes from 4 calves. ∗ = significantly (P < .001) greater than the mean at all other days examined.
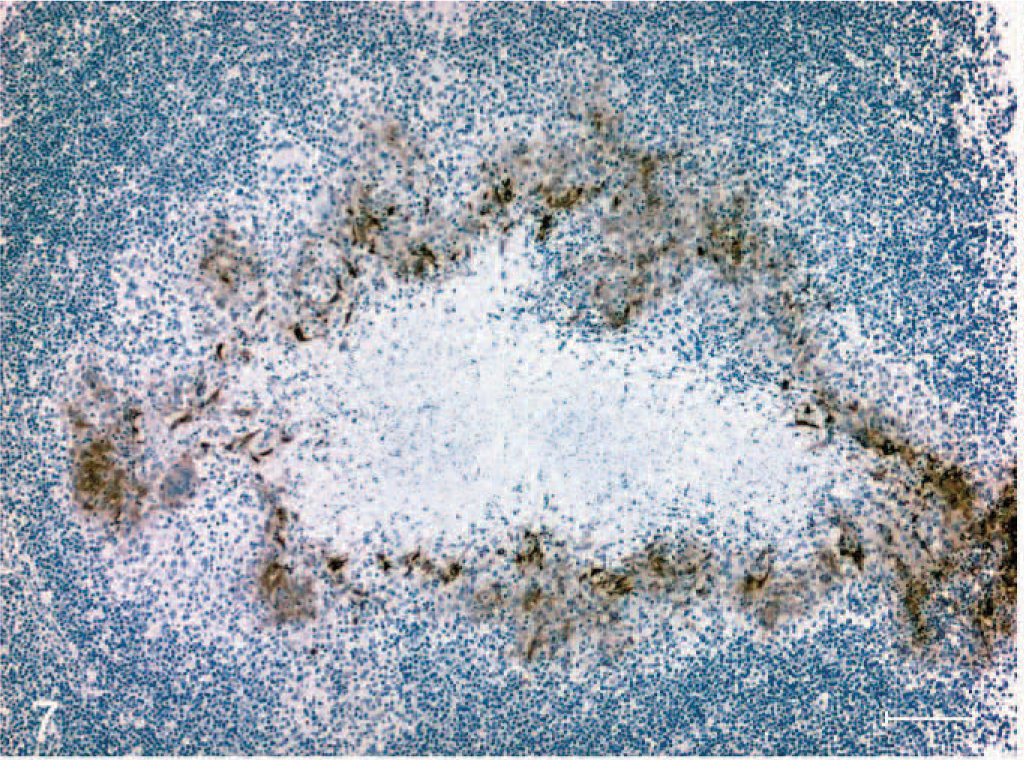
Section of medial retropharyngeal lymph node from a calf inoculated intratonsilarly with 1 × 105 colony forming units of Mycobacterium bovis and examined 15 days after inoculation. Note stage II granuloma composed of a necrotic core surrounded by epithelioid macrophages and multinucleated giant cells. Inducible nitric oxide synthase immunoreactivity is strongest in a narrow zone of inflammatory cells surrounding the necrotic core. Immunoperoxidase method, Harris hematoxylin counterstain. Bar = 100 μm.
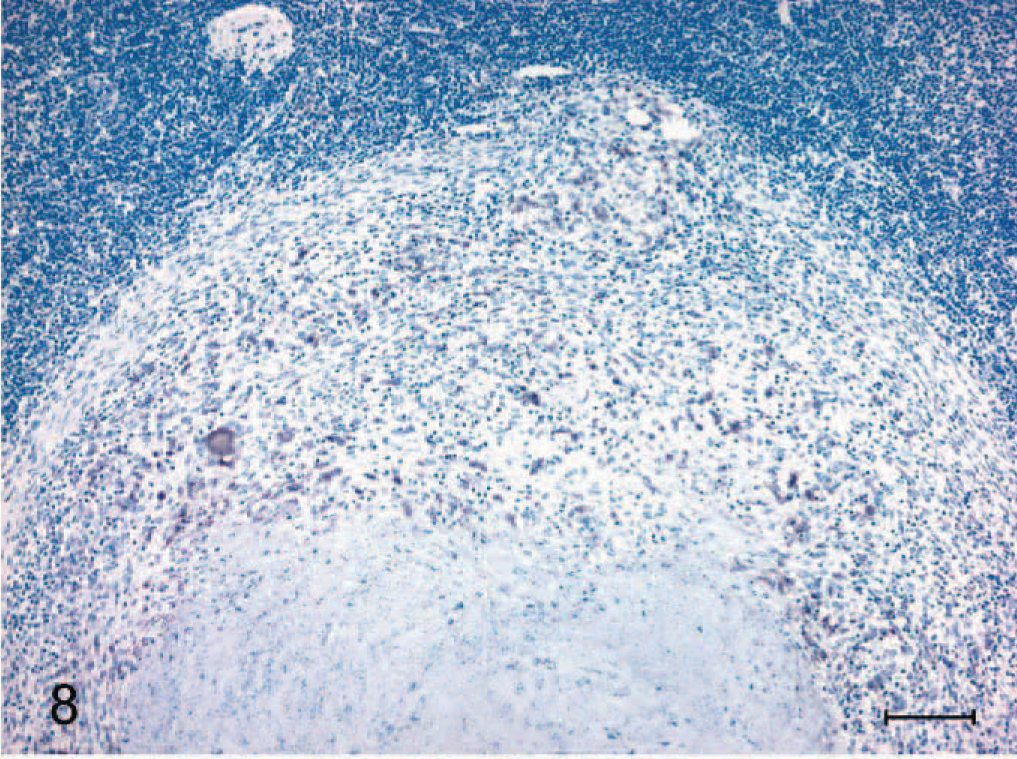
Section of medial retropharyngeal lymph node from a calf inoculated intratonsilarly with 1 × 105 colony forming units of Mycobacterium bovis and examined 180 days after inoculation. Note CD68+ immunoreactivity within numerous cells surrounding a necrotic core of a stage III granuloma. Immunoperoxidase method, Harris hematoxylin counterstain. Bar = 100 μm.
Lesions at all time points contained larger numbers of CD4+ T cells with lesser numbers of CD8+ T cells and few γ/δ T cells (Fig. 9). Although the relative number of CD4+ cells remained constant throughout the study, at time points >60 days after inoculation, scores of +, ++, or +++ decreased from 50 to 6% for γ/δ T cells and from 100 to 56% for CD8+ T cells (Table 6).
Semiquantitative scoring of immunolabeling for CD4+, CD8+ or γ/δ T cells in granulomas in medial retropharyngeal lymph nodes of cattle experimentally inoculated with Mycobacterium bovis and examined at various time points after inoculation.
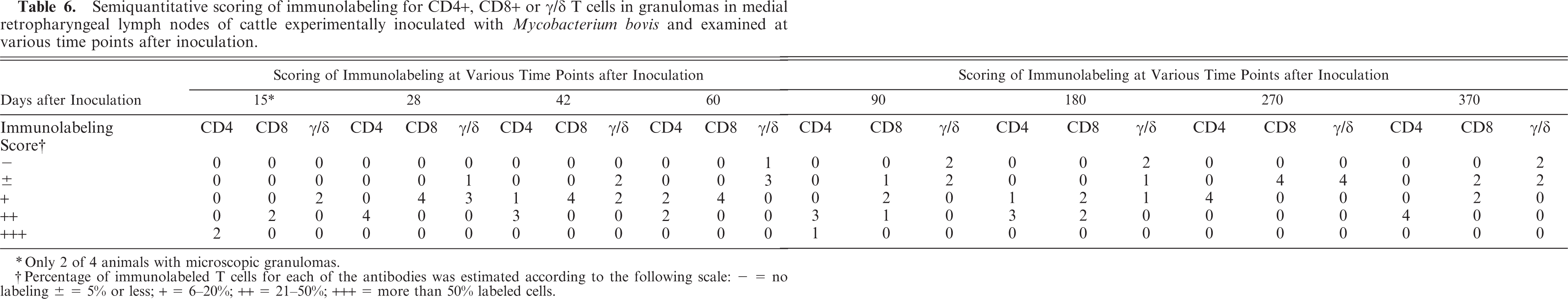
∗Only 2 of 4 animals with microscopic granulomas.
†Percentage of immunolabeled T cells for each of the antibodies was estimated according to the following scale: ± = 5% or less; + = 6–20%; ++ = 21–50%; +++ = more than 50% labeled cells.

Section of medial retropharyngeal lymph node from a calf inoculated intratonsilarly with 1 × 105 colony forming units of Mycobacterium bovis and examined 180 days after inoculation. Note immunoreactivity for CD4+ (A), CD8+ (B), and γ/δ (C) T cells within tuberculous granulomas. Immunoperoxidase method, Harris hematoxylin counterstain. Bar = 100 μm.
Discussion
The present study demonstrates that iNOS is expressed in granulomas from cattle experimentally inoculated with M. bovis; however, expression decreases in granulomas of longer duration. Few studies have examined the presence of iNOS in situ over the course of a disease process. In caprine arthritis encephalitis virus infection of goats, iNOS expression was a feature of late-stage chronic arthritis and not involved in the early development of disease. 23 Moreover, in affected joints of goats with arthritis, iNOS-expressing cells were CD68- and major histocompatibility complex II (MHCII)-negative and thought to be fibroblast-like synoviocytes. 23 It is possible that the spindloid cells exhibiting iNOS immunoreactivity in the present study were fibroblasts. Cells with similar morphology have been described in a recent report of iNOS immunoreactivity in bovine granulomas. 10
Recently iNOS expression has been documented in granulomas from cattle naturally infected with M. bovis. 34 Expression varied between cattle, likely due to the unknown duration of infection and stage of granuloma development. In the present study, iNOS immunoreactivity decreased in later stages of disease when stage III and stage IV granulomas were most numerous. It is within these same granuloma stages that numbers of acid-fast bacilli are highest. In other mycobacterial diseases of cattle such as paratuberculosis, iNOS expression has been shown to be minimal or absent in granulomas with high numbers of acid-fast bacilli. 16 Similar to the present study, iNOS immunoreactivity in human granulomas, caused by mycobacterial and nonmycobacterial infectious agents, is greatest in smaller granulomas and minimal in large granulomas with extensive necrosis. 12 Interestingly, in the present study, iNOS expression was limited to cells within the granuloma and not present in surrounding tissue, suggesting that iNOS expression is dependent on conditions within the granuloma microenvironment.
In human granulomas, iNOS expression has been positively associated with the expression of Th1-type cytokines such as IFN-γ and negatively correlated with expression of Th2-type cytokines such as IL-4. 11 Th1-type cytokines induce iNOS, which oxidizes L-arginine to form NO. 27 In contrast, Th2-type cytokines induce an alternative pathway where L-arginine is metabolized by arginase to L-ornithine and urea, inhibiting NO production through iNOS. 27 Thus, the increased expression of iNOS early in disease with decreased expression in advanced disease is in agreement with the generally held belief that Th1-type responses prevail early in mycobacterial infections, while Th2-type responses prevail in later disease stages.
Morphologically, stage I granulomas, 15–42 days after inoculation, were similar to stage I granulomas 60–370 days after inoculation (satellite granulomas), with the exception of the close proximity of the latter to stage IV granulomas. However, the lack of acid-fast bacilli in satellite granulomas suggests that although similar in cell type, the microenvironment of satellite granulomas differs substantially from that seen in early stage I granulomas. It is likely that due to the more advanced stage of disease, macrophages recruited to satellite granulomas have experienced different signaling and activation events compared with macrophages that constitute early stage I granulomas.
T-cell-mediated immunity is considered essential for clearance of mycobacterial infections. 29 The predominant T-cell subtype in granulomas of all stages was CD4+, with fewer CD8+ and γ/δ T cells. CD4+ T cells play a central role as IFN-γ produced by CD4+ T cells activates macrophages to facilitate killing of intracellular mycobacteria. However, in response to stimulation with M. bovis purified protein derivative, CD4+, CD8+, and γ/δ T cells from cattle have all been shown to proliferate, and CD4+ and CD8+ T cells from M. bovis-infected cattle have been shown to produce IFN-γ. 24, 46
The results of the present study are in agreement with previous findings in both humans and cattle. In tuberculous granulomas of unknown duration from human lymph nodes, CD4+ and CD8+ T cells represent approximately 79% of the total cell population within lesions. 36 Similar to findings in the present study, others have demonstrated in cattle that γ/δ T cells were most numerous in developing lesions compared with more advanced lesions. 4 Unlike the present study, Wangoo et al. 45 reported an overall decrease in T cells and an increase in γ/δ T cells in morphologically advanced granulomas compared with morphologically immature granulomas from experimentally infected cattle examined 29 weeks after inoculation. Differences between the 2 studies may be the result of difference in inoculum strain, inoculum dosage, route of inoculation, or duration of infection (203 days versus 370 days).
Grossly liquefactive lesions in the medial retropharyngeal lymph nodes were seen as early as 42 days in some calves and in all calves 370 days after inoculation. The conversion of a solid or semisolid caseous lesion to one of a more liquid nature is similar to the liquefaction of tuberculous pulmonary lesions described in humans and experimentally infected rabbits and due to the action of various proteases and nucleases. 7 The presence of liquefied lesions is thought to be important in the dissemination of disease within the host as well as to other susceptible hosts.
Microscopic or gross pulmonary lesions were first seen 42 days after inoculation, with most lesions present in the caudal lung lobes. The predilection for lesion development in caudal lung lobes has been previously reported in cattle, where 90% of pulmonary lesions are found in the diaphragmatic lobes, approximately 50% of which are located in the distal one third. 25, 26, 41 Such a lesion distribution may be due to the fact that the majority of lung parenchyma is located within the right and left caudal lobes. Alternatively, regional lesion distribution within the lung could also be due to factors such as a difference in oxygen tension. A higher ventilation/perfusion ratio exists in cranioventral regions of the bovine lung compared with the caudodorsal region, creating a relative hypoxia in the caudodorsal region. 38, 39 Such hypoxia may result in suboptimal macrophage function. 8 Interestingly, the distribution of pulmonary lesions in tuberculous cattle is dissimilar to the typical cranioventral distribution of many bacterial pneumonias of cattle where, like tuberculosis, aerosol exposure is presumed. Regional lesion distribution has also been noted in human tuberculosis, where there is a predilection for granuloma development in the apical lung lobes. 40
Granulomas in lymph nodes of experimentally inoculated cattle represent dynamic lesions that, early in development, are characterized by iNOS-expressing macrophages and multinucleated giant cells, moderate numbers of acid-fast bacilli, minimal necrosis, mineralization, peripheral fibrosis, and a heterogeneous lymphocyte population of CD4+, CD8+, and γ/δ T cells. With disease progression, there is an increase in the numbers of acid-fast bacilli, necrosis, mineralization, and peripheral fibrosis with a decrease in iNOS-expressing cells, CD8+, and γ/δ T cells. Although there is an orderly progression through lesion stages as disease advances, within a given lymph node at any time, there may be granulomas of various stages of development that likely represent lesions with differing microenvironments.
Footnotes
Acknowledgements
We thank L. Wright, D. Weuve, D. Robinson, D. Ewing, and W. Varland for animal care and B. Lyon, S. Zimmerman, B. Olthoff, and J. Pollock for technical assistance. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture.