Abstract
A 2–year-old, captive-born, clinically healthy male, rhesus macaque, was euthanatized as part of an experimental study. At necropsy, diffuse pale streaking of the trunk, lumbar, and limb muscles were noted macroscopically. On histology, numerous elongated cysts that contained crescent-shaped basophilic spores were found in the fibers of skeletal muscles. Scattered affected myofibers were degenerate and accompanied by eosinophilic-to-granulomatous inflammation. Sarcocysts had prominent villus-like projections with the morphology of a type 11 sarcocyst wall similar to Sarcocystis neurona but possessing many more villus microtubules than is reported for S. neurona. In addition, bradyzoites were very long, up to approximately 12 um in length. The protozoa were consistent with a Sarcocystis sp., based on histology and ultrastructure, however, a definitive identification of the species was not possible. Nonspecific immunohistochemical crossreaction with Sarcocystis cruzi antisera was observed. The 18S ribosomal deoxyribonucleic acid sequence showed 91% similarity to Sarcocystis hominis, 90% similarity to Sarcocystis buffalonis, and 89% similarity to Sarcocystis hirsuta. Interestingly, the ITSI sequence showed very little homology to any sequence in GenBank, suggesting that this is possibly a unique Sarcocystis sp. Sarcocystosis is often considered an incidental finding, particularly in wild-caught animals, with little clinical significance. However, as demonstrated in this report and others, disseminated sarcocystosis can occur in captive-born rhesus macaques with or without clinical signs. In some cases interference with research results can occur; including death in fulminant cases.
Keywords
Sarcocystosis has been reported in primates, other animal species, and humans. 4 Although common, this parasite is usually considered nonpathogenic and an incidental finding during necropsy, particularly in wild-caught animals. In this report, we describe a case of chronic eosinophilic polymyositis associated with disseminated sarcocystosis in a captive-born, young rhesus macaque.
A 3.1-kg, 2-year-old, captive-born, clinically healthy male rhesus macaque (Macaca mulatta) was received after an uneventful quarantine from an outdoor breeding facility in southeast USA. The monkey was negative, by serology, to the following agents: tuberculosis, Cercopithecine herpesvirus 1 (B-virus), measles, simian retrovirus/D, simian T-cell leukemia virus-1, and simian immune-deficient virus. In addition, the animal was negative to gastrointestinal parasites, Salmonella spp., Shigella spp., and Campylobacter spp . Blood samples were collected for complete blood count and serum chemistry. Phosphorus and aspartate aminotransferase were found slightly elevated (9.2 mg/dl, normal 5.1 ± 0.7 mg/dl; and 66 U/l, normal 47 ± 13 U/l, respectively); and the following were found below normal levels: glucose (48 mg/dl, normal 74 ± 12 mg/dl), creatinine (0.5 mg/dl, normal 0.7 ± 0.1 mg/dl), and calcium (8.5 mg/dl, normal 10.3 ± 0.5 mg/dl). The hypoglycemia was most likely because of fasting of the animal before anesthesia, and the other changes were not considered clinically significant. At physical examination, the monkey was found to be clinically healthy and subsequently was enrolled in an institutional animal care and use committee–approved viral pathogenesis study. The animal was inoculated with a viral construct and was euthanatized 3 days later as part of the experimental study. At necropsy, diffuse pale streaking of the trunk, lumbar, and limb muscles was noted. No other gross lesions were observed. Tissue samples were collected and fixed in 10% neutral buffered formalin, embedded in paraffin, sections cut at 3–5 μ and processed routinely for histologic examination. For ultrastructural examination, formalin-fixed skeletal muscle was transferred to half-strength modified Karnovsky's fixative before 2 washes in 0.2 M sodium cacodylate and after fixation in 2% osmium tetroxide reduced with 2.5% potassium ferrocyanide. After fixation, the tissue was washed in 0.2 M sodium cacodylate, dehydrated through a graded ethanol series before infiltration and embedment in Eponate 12 epoxy resin. Thick sections were cut, mounted on glass slides and stained with toluidine blue O, and examined by light microscopy. Thin sections were cut, mounted on 150-mesh copper grids, stained with 4% uranyl acetate in 75% ethanol and Reynold's lead citrate, and examined in a Zeiss 906E transmission electron microscope at 60 kv accelerating voltage. Immunohistochemical staining was performed on 4-μm sections of formalin-fixed paraffin embedded tissue by using rabbit polyclonal antisera against Sarcocystis neurona, Sarcocystis falcatula, Neospora caninum, and Toxoplasma gondii, as previously described, 11 with the exception that the S. falcatula optimal dilution was 1 : 1,200. Sarcocystis cruzi polyclonal antisera 1 was graciously supplied by Dr. J. P. Dubey and was used at a 1 : 200 dilution by using the same procedures. For antigen retrieval, the slides were pretreated by incubation in prewarmed 0.4% pepsin (w/v in 0.1 N Cl) for 15 minutes at 37°C. The positive control was a bovine fetal lung infected with presumptive S. cruzi merozoites. Staining was also run with a S. neurona specific monoclonal antibody in tissue-culture supernatant at a 1 : 700 dilution as previously described. 9 Equine brainstem that contained S. neurona served as the positive control. To further characterize the organism, deoxyribonucleic acid (DNA) was extracted and polymerase chain reaction amplified from formalin-fixed and paraffin-embedded macaque tissues. Briefly, paraffin tissue sections were placed in 2.0-ml Microfuge tubes and was heated at 70°C for 15 minutes followed by the addition of 180 μl ATL buffer (Qiagen). The tubes were heated at 70°C for several hours, with frequent vortexing, then were centrifuged at 14,000 rpm (20,000 g) for 3 minutes. Proteinase K, 40 μl, was added, and the Qiagen tissue extraction protocol was followed, except, in the final step, the DNA was eluted with 60 μl of 95°C Millipore H2O. Of this DNA solution, 1 μl was used in a 50 μl polymerase chain reaction (PCR). The PCRs were carried out in a GeneAMP 2400 thermocycler under the following reaction conditions: 5.0 μl 10×PCR Buffer (Applied Biosystems, Foster City, CA) w/MgCl2 (15 mM), 200 μM dNTPs, 1 μM of each primer (S5 forward, S2 reverse), and 1.5U Taq polymerase. 5 After initial denaturation of templates and primers (94°C, 3 minutes), 30 cycles of the following conditions were used: 94°C for 1 minute, 55°C for 45 seconds, 72°C for 1 minute, followed by a 5-minute extension at 72°C; 10 μl of PCR product was electrophoretically separated on a 2% agarose gel stained with ethidium bromide and visualized under ultraviolet light. PCR products from positive bands were cleaned by using the ExoSAP-IT PCR clean-up system and sequenced at the DBS sequencing facility at University of California at Davis.
On histology, numerous elongated tissue cysts that contained crescent-shaped protozoal zoites were found in the fibers of skeletal muscles. The numerous cysts were much more noticeable on cross-section (Fig. 1). Scattered affected myofibers were degenerate and accompanied by eosinophilic-to-granulomatous inflammation (Fig. 2). Peripheral lymph nodes were hyperplastic with plasmacytosis and focal eosinophil infiltrates. By transmission electron microscopy, sarcocysts had prominent villus-like projections with the morphology of a type 11 sarcocyst wall similar to S. neurona but possessing many more villus microtubules than is reported for S. neurona 3 (Fig. 3). The tissue cyst wall was measured from a total of 8 cysts by transmission electron microscopy. Measurements were made on villous projections cut lengthwise in profile. In these fields, the length of the longest profile and the width of the broadest profile were measured, as well as the broadest width in the adjacent granular layer. Villous projections ranged from 2.74 to 6.32 μm in length and 0.54 to 1.17 μm in width (average of 4.47 × 0.88 μm). The granular layer ranged from 0.68 to 1.49 μm in width (average of 0.92 μm). The size of both the villous projections and the width of the granular layer varied markedly and correlated with the overall corresponding tissue-cyst diameter. For example, in the smallest cyst, the projections and the granular layer width were approximately a half of that of the larger tissue cysts. Bradyzoites were very long and narrow. A total of 11 longitudinal profiles were measured through their longest axis and broadest point. Attempts were made to measure longitudinal profiles that included an anterior apicomplex, but, in some instances, the profiles included anterior ends that did not include a visible apicomplex. These profiles appeared to be very near the apicomplex. The individual bradyzoite ranged from 10.03 to 13.63 μm in length by 1.90 to 3.78 μm in width, with an average of 11.8 × 2.67 μm, respectively. Bradyzoites had nuclei located in the posterior of the zoites, with numerous micronemes oriented parallel to the pellicle and anterior to the nuclei (Fig. 4). There were several large electron-dense granules located primarily anterior to the nuclei and several additional membrane-bound granules that contained peripheral electron-dense material with variable-sized central clear regions. Amylopectin granules, Golgi, and multiple profiles of mitochondria were also present. Twenty-two longitudinal subpellicular microtubules were present beneath the inner cell membrane (Fig. 5). Bradyzoites in larger cysts were often packeted and separated by fine granular septae. Metrocytes were present within septae. With the exception of the S. cruzi antisera, neither the bradyzoites or the cyst wall reacted by immunohistochemistry with any of the antibodies. Approximately half of the bradyzoites reacted positively to the S. cruzi antisera, creating a mosaic pattern of positive- and negative-reacting bradyzoites. This reaction was interpreted as a nonspecific cross-reaction to common shared epitopes in a select subset of the bradyzoites. The 18S ribosomal DNA (∼350 bp) sequence showed 91% similarity to Sarcocystis hominis, 90% similarity to Sarcocystis buffalonis, and 89% similarity to Sarcocystis hirsuta. Interestingly, the ITS1 (first internal transcribed sequence) showed very little homology to any sequence in GenBank. Only about 44 bp of 320 bp matched up with a number of other apicomplexan ITS1 sequences.
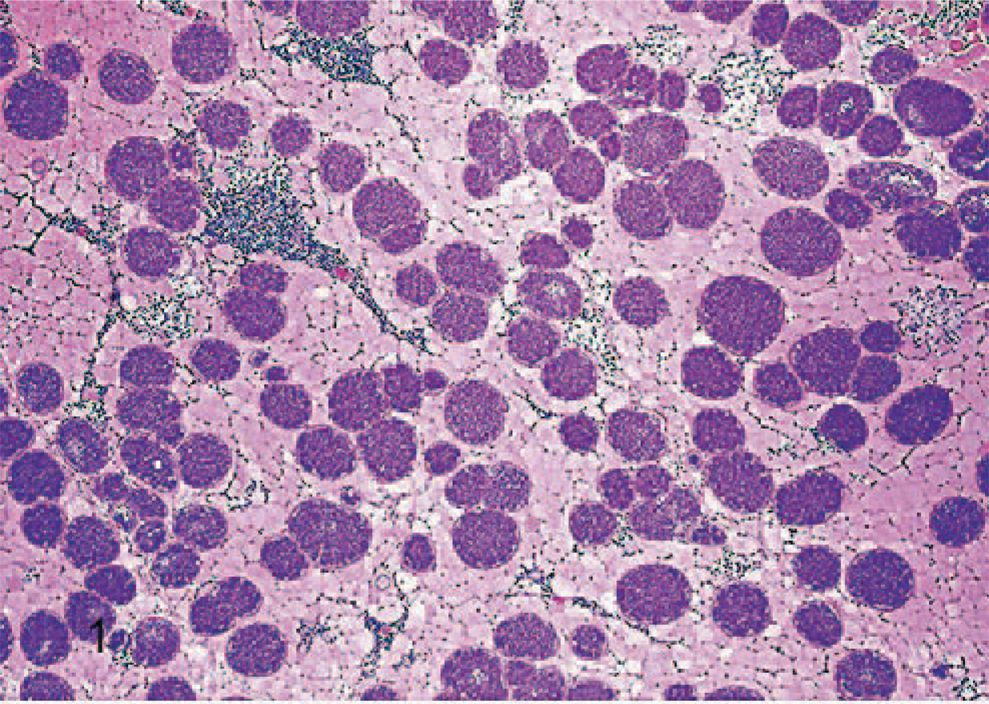
Skeletal muscle; rhesus macaque. Skeletal muscle showing numerous cross sections of sarcocysts. HE.
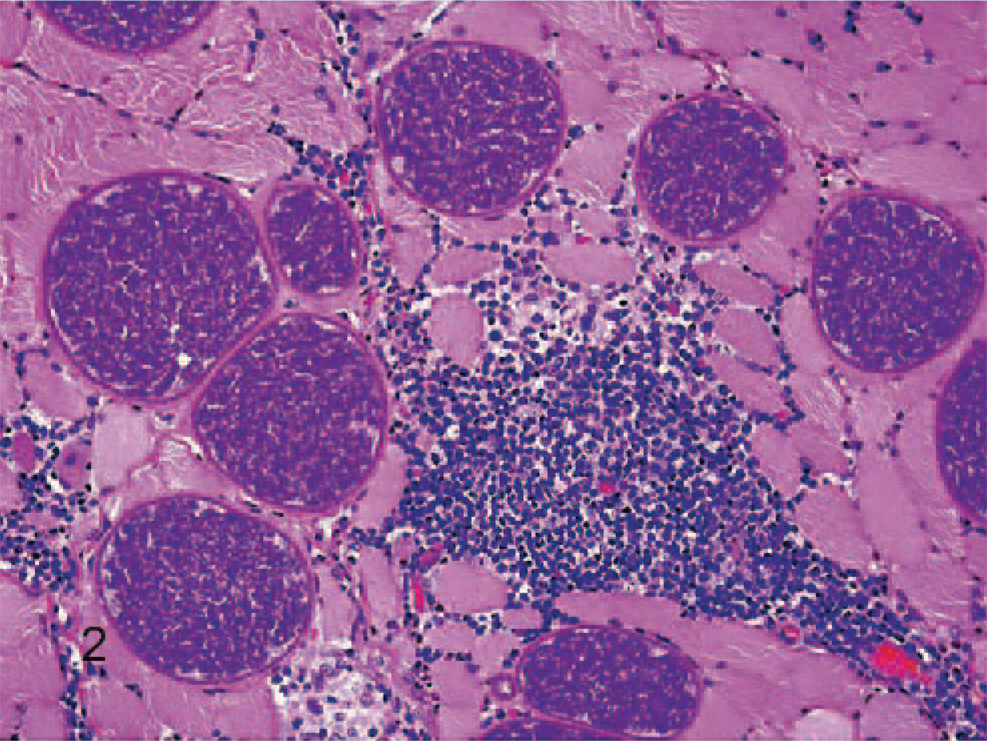
Skeletal muscle; rhesus macaque. Higher magnification, showing sarcocysts and necrotic myofibers accompanied by inflammatory infiltrate composed of eosinophils, plasma cells, lymphocytes, and macrophages. HE.
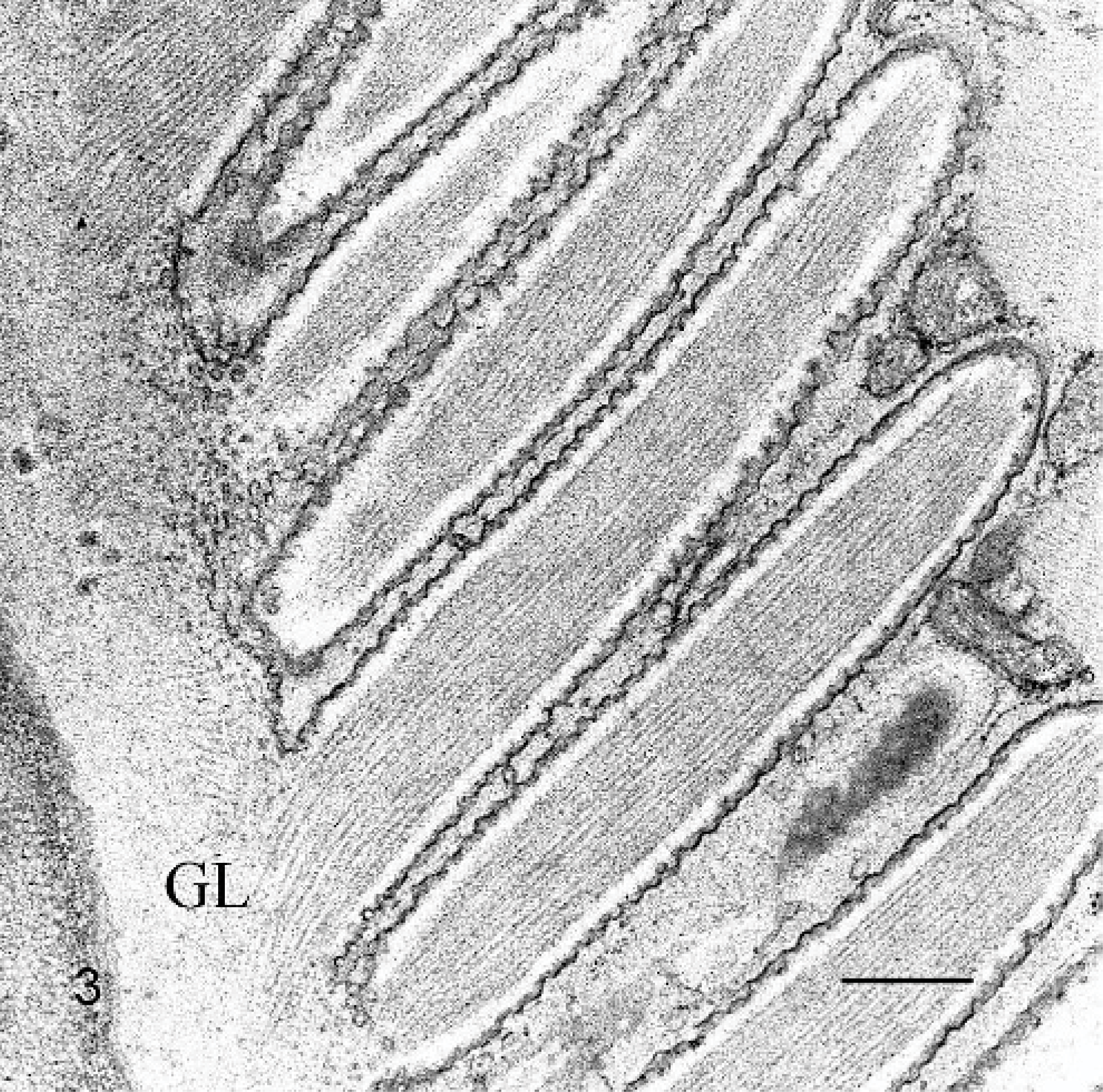
Skeletal muscle; rhesus macaque. Transmission electron micrograph showing cyst wall with long villous projections, a fine wavy or cobbled membrane, and a thin dense surface layer. Numerous fine microtubules in the villous projections extend a short distance into the underlying granular layer (GL). Transmission electron microscopy. Bar = 500 nm.
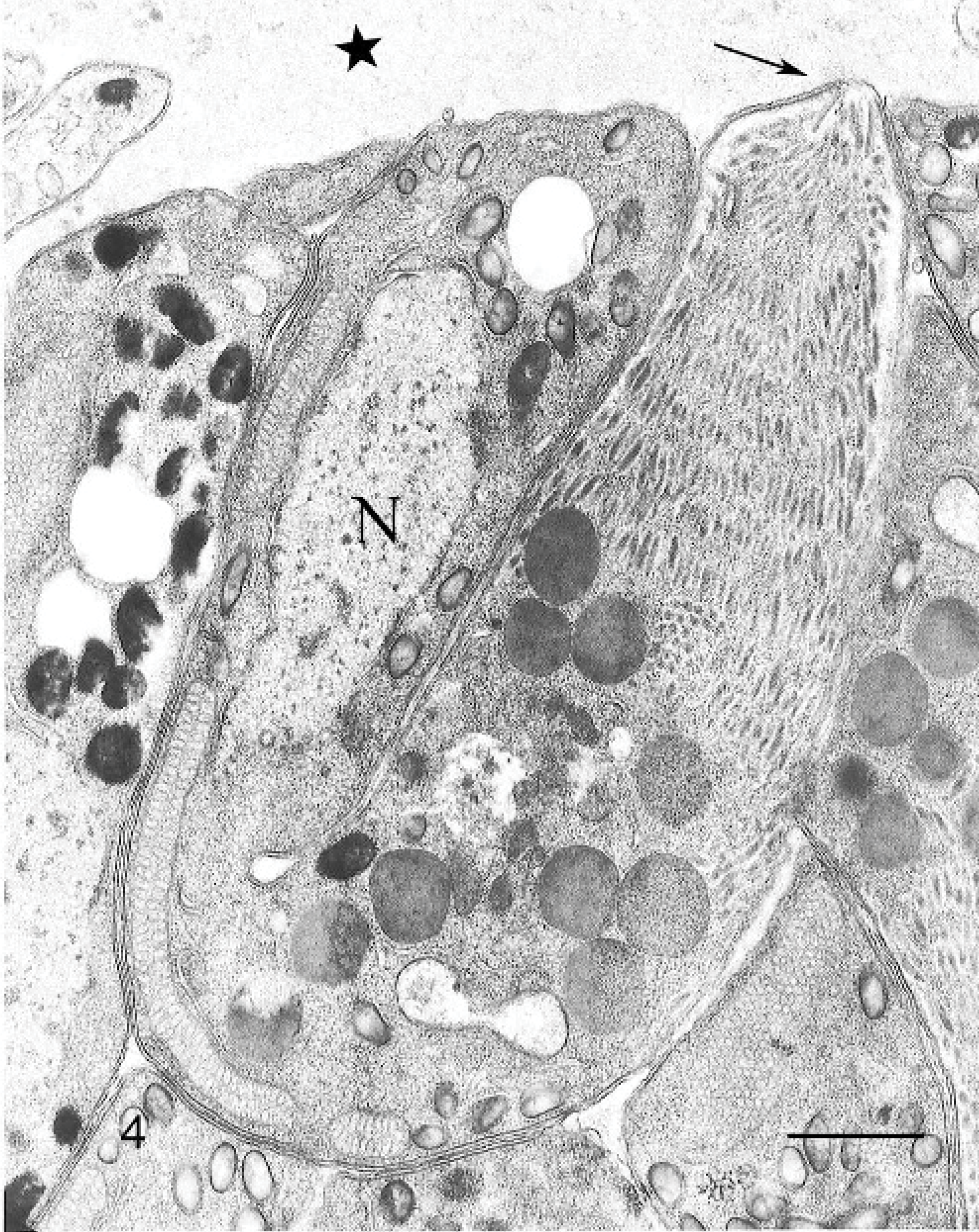
Skeletal muscle; rhesus macaque. Transmission electron micrograph of a bradyzoite. A conoid and an adjacent polar ring are visible at the anterior end (arrow). There are large numbers of micronemes, filling the anterior end, that are arranged in an array parallel to the outer membrane (no rhoptries were seen). In addition, there are 2 types of dense membrane-bound granules that consist of large round granules, with a uniform dense staining pattern, and additional smaller, slightly elongated granules that have very dense peripheral staining, with slightly less dense or hollow-appearing central regions. These latter granules are present both anterior and posterior to the nucleus (N), which is located in the posterior half of the bradyzoite. In addition, 3 profiles of long mitochondria, a portion of the Golgi apparatus anterior to the nucleus, and a single round empty space, at the posterior end, that is thought to be an amylopectin granule are also visible. The granular layer of the cyst wall (star) with a portion of a metrocyte lies next to the bradyzoite. Transmission electron microscopy. Bar = 1 μm.
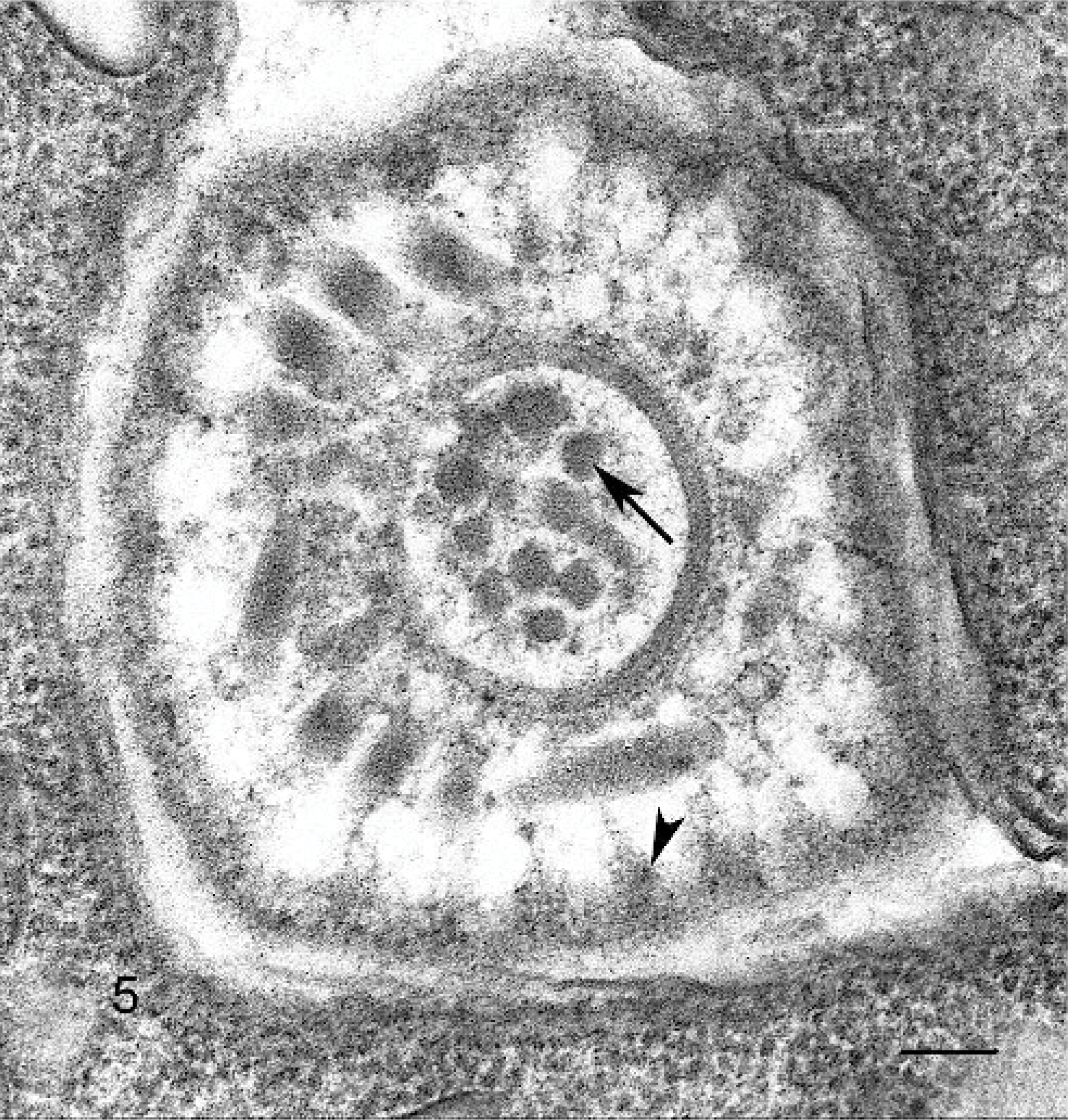
Skeletal muscle; rhesus macaque. Cross-cut through anterior end of a bradyzoite, showing the conoid, with central cross-sections of presumptive micronemes (arrow), additional micronemes around the conoid, and tangential sections of 22 electron dense projections extending from the outer membrane that represent tangential profiles of subpellicular microtubules (arrow head). Transmission electron microscopy. Bar = 100 nm.
Sarcocystosis has been reported in rhesus macaques and is often considered an incidental finding, particularly in wild-caught animals, with little clinical significance. However, Terrell and Stookey 13 reported a severe case of chronic eosinophilic myositis in a rhesus macaque infected with Sarcocystis kortei. More recently, Lane et al. 7 reported a case of fulminant sarcocystosis in a captive-born rhesus macaque. In this last case, the lesions involved generalized endothelial infection with an unidentified Sarcocystis sp. Immature and mature schizonts found within endothelial cells were most prevalent in the cardiac muscle, mature tissue cysts were not found in myocardium but were seen in skeletal muscle. 7 Mandour 8 described Sarcocystis nesbitti in rhesus macaques. In addition, an unidentified Sarcocystis sp. has been reported in rhesus macaques. 10 However, these sarcocysts had a cyst-wall morphology type 1, different from the cyst-wall morphology observed in the sarcocysts from this report. A captive-born rhesus macaque experimentally infected with simian immunodeficiency virus was reported to have chronic necrotizing encephalomyelitis with intralesional protozoal schizonts similar to S. neurona, however, no cysts were observed. 6 We were unable to determine if the sarcocyst observed in our case was similar to S. kortei, because the ultrastructural description was not performed in the case reported by Terrel and Stookey. 13
By histology and ultrastructure, the cyst-wall morphology in the present report was consistent with a type 11 sarcocyst. 4 However, a definitive identification of the species was not possible. In addition, the 18S rDNA and the ITS1 sequence analysis showed very little homology to any known Sarcocystis sequence in GenBank. The ITS1 sequence separates the 18S and 5.8S rDNA genes, and, as the name implies, it is transcribed but then cut out during the ribosomal processing. Because it is not part of the ribosome, it tends to be a much less conserved gene sequence, which makes it useful for differentiating closely related species. Given these results, we believe that this is possibly a unique Sarcocystis sp. or at least one that has not been cataloged before in GenBank.
Identification of the tissue-cyst stage of Sarcocystis spp. is very difficult, because standard immunohistochemical reagents that are used to identify the merozoite stage often are not specific for the tissue-cyst biological stage. 2, 12 Although the tissue cyst-wall ultrastructural characteristics, as described by Dubey et al. 4 are useful as part of the general characterization and identification of a Sarcocystis sp., the ultrastructural features by themselves should not be considered sufficient to speciate a Sarcocystis that has not been previously well characterized in a given host species. Species identification should require some second confirmatory test methodology, such as PCR, or immunohistochemistry.
The source of the infection in this case was most likely feces from a feral carnivore. However, a definitive host was not identified. As demonstrated in this report and by others, disseminated sarcocystosis can occur in outdoor-housed, captive-born, rhesus macaques with or without clinical signs. In some cases, interference with research results can occur, and death has occurred in fulminant cases.
Footnotes
Acknowledgements
This research was supported by the Intramural Research Program of the National Institutes of Health, National Institute of Allergy and Infectious Diseases (NIAID), Comparative Medicine Branch, the Office of Research Support, and a NIAID contract to SoBran Inc. We thank Dr. Randy Elkins, John DeLeonardis, and Brad Fisher for support, and Larry Faucette for histotechnology, Bob Nordhausen for electron microscopic technical support, and the Davis CAHFS histology laboratory for immunohistochemical technical support.
