Abstract
Melanomas are the most common malignant oral neoplasm in dogs. Osteocartilaginous differentiation in oral melanomas is a rare feature described both in veterinary and human medicine. Here, 10 cases of this type of neoplasm were used to study their immunohistochemical, biological, and clinical characteristics. Reactivity for S100 and melan A antigen was evaluated, and 4 prognosis factors (mitotic index, invasiveness of epithelium, nuclear atypia, and proliferation index) were analyzed and correlated with the clinical course of the neoplasms after diagnosis. Immunohistochemical analysis of the studied neoplasms, including the osteocartilaginous areas, showed positive immunoreaction for S100 and melan A, except in one dog, which was negative for melan A. Analysis of the results showed that oral melamonas with osteocartilaginous differentiation have a clinical course similar to that of other melanomas in the oral cavity. Analysis of the mitotic index and the expression of proliferation marker Ki-67 could be useful tools for predicting the biological behavior of these neoplasms.
Keywords
Oral melanomas are relatively common neoplasms that represent approximately 1% of canine pathology accessions received in veterinary diagnostic laboratories. 16 Melanomas are located preferentially in the gingival mucosa, although they can also occur in the labial mucosa, tongue, palate, or tonsils. In contrast with dermal melanomas, which are usually benign, nearly all melanomas of the oral cavity are considered malignant because of their infiltrative growth, which frequently recurs after surgery, and because of their capacity to induce both local and distant metastasis. 1, 5, 19
Microscopically, the morphology of these neoplasms is highly variable, and several subtypes appear: epithelioid, fusiform, mixed, or clear cells. 6 This fact, together with the existence of amelanotic melanomas, represents a challenge for the accurate diagnosis of these tumors. Immunohistochemistry has been used as a supplementary tool for diagnosing malignant melanomas 18, 21 ; most melanomas are positive for vimentin and S100 antibodies, although recently a more specific marker, melan A, has been introduced in veterinary diagnosis. 17
Osteocartilaginous differentiation within malignant melanoma is a very rare occurence, although cases have been reported in both dermal 14 and oral 2, 16 melanomas. Because there are very few reported cases of osteocartilogenic oral melanomas, their prognosis and the pathogenesis of the osteocartilaginous elements remain unclear. The aim of the present study was to improve our knowledge of the expression of prognostic factors and the clinical course of this tumor type and to compare it with the classical form of oral melanoma.
Ten canine neoplasms from the oral cavity were used in this study (Table 1). The cases were taken from the files of the Anatomic Pathology Service of the Veterinary Teaching Hospital of the University of Murcia and from a private laboratory (Histovet, Barcelona, Spain). The samples used were submitted to the laboratories during the period 2002 through 2006. The breed, age, and sex of the dogs, together with the location of the tumors, are summarized inTable 1. The neoplasms were diagnosed from their microscopic characteristics as malignant melanomas with areas of osteocartilaginous differentiation. These areas were predominantly cartilaginous, except in dog No. 8, where only osteogenic differentiation could be observed. All the tumors were pigmented to a variable degree, except in dog Nos. 2, 4, and 8, which were considered amelanocytic melanomas.
Characteristics of the dogs and location of tumors.

To assess the initial diagnosis, an immunohistochemical analysis was carried out as follows: Selected 4-μm histologic sections from formalin-fixed and paraffin-embedded samples were stained by hematoxylin and eosin (HE) and by the avidin-biotin-peroxidase complex (ABC) method with 7 primary antibodies, whose clone, dilution, and pretreatment for antigen retrieval are summarized inTable 2. All the antibodies were purchased from Dako Corporation (Carpinteria, CA). Counterstaining involved Mayer's hematoxylin while both positive and negative control sections were included for each antibody.
Characteristics of the primary antibodies used in the immunohistochemical diagnosis carried out in the present study.
Once the diagnosis had been confirmed, several prognostic factors were analyzed: 1) Nuclear atypia, following the method reported by Spangler and Kass, 20 using an incremental scale from 1 to 10, signifying the subjectively estimated percentage of nuclei involved; 2) mitotic index (MI), calculated as the average number of mitoses per field after observation of 10 high-power fields (hpfs; 400×) per tumor; 3) invasiveness of the epithelium or presence of ulcerative lesions; and 4) cell proliferation index, which was evaluated by measuring the expression of the proliferation marker Ki-67 in the nuclei of neoplastic cells, because increased expression of the marker has been associated with malignancy in melanomas. 11, 15 The ABC technique was carried out using a monoclonal antibody (clone MIB-1, Dako Corporation), observing 600 nuclei in each tumor and calculating the percentage of positive nuclei. As additional information, we included the cell type of the tumor and the degree of pigmentation, which was evaluated using a subjective scale 16 running from nonexistent, scant (pigment in fewer than 30% cells), moderate (pigmentation in 31% to 80% of cells), to abundant (pigment in more than 80% of cells). Finally, the clinical course of the disease after collection of surgical biopsies was obtained by a phone survey to the referring veterinarians.
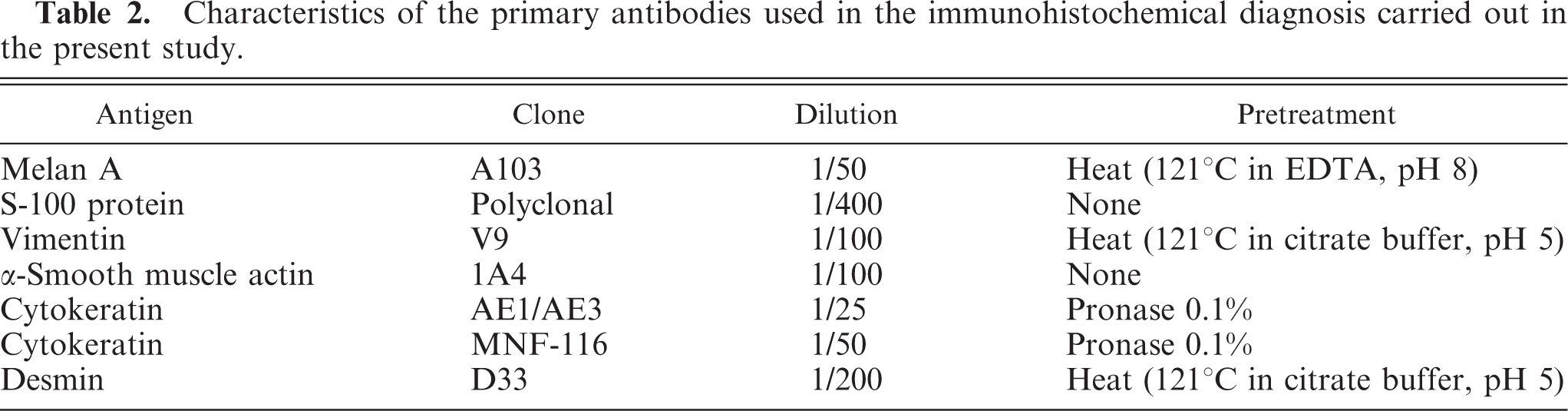
Microscopic observation of the tumors showed 3 cell-type variants (epithelioid, fusiform, and mixed; detailed inTable 3). Areas of ostecartilaginous differentiation were observed in all the dogs except dog No. 8 (only osteoid). The extent of the osteocartilaginous areas was varied; for example in dog No. 3, a cartilaginous area with small osteoid areas appeared in 50% of the observed section of the tumor, while in other tumors (dog Nos. 1, 4, 5, and 6) the cartilaginous area was less than 10% of the observed section. Three degrees of differentiation could be observed in the osteocartilaginous areas, appearing sometimes in the same case: 1) well-differentiated, with or without osteoid matrix, and small areas with chondrocytes located within lacunae and abundant basophilic extracellular matrix; in some areas, the matrix became acidophilic and fibrillar, resembling osteoid matrix; 2) moderately differentiated, peripherally to the well-differentiated areas or in independent areas, polyhedric chondroid cells located between slightly basophilic matrix showed moderate anaplasia and slight anisokaryosis, with large vesicular nuclei with 1 to 2 or even 3 evident nucleoli; nonchondroid anaplastic tumoral cells were seen interspersed within this areas (Fig. 1); 3) poorly differentiated: substantial anaplasia was observed in the chondroid areas, with pleomorphic cells with basophilic cytoplasm, large nuclei with evident nucleoli, marked anisokaryosis, presence of multinucleated giant cells and frequent mitosis. These anaplastic areas were observed only in dog Nos. 7, 9, and 10.
Characterization of the cell type, pigmentation, prognostic factors, and clinical course of the neoplasms used in the present study.
∗ Subjectively estimated percentage of nuclei with atypia, i.e., 7 means that 70 % of nuclei showed atypia; NA = not applicable.
† Percentage of nuclei with positive staining for the proliferation marker Ki-67.
Dog No. 9. Fig. 1a. Cartilaginous areas (asterisks) in an oral melanoma. The cartilaginous areas are surrounded by neoplastic melanogenic cells. HE. Bar = 200 μm. Fig. 1b. Detail of Fig. 1a where anaplastic chondroid cells are observed among a slightly basophilic matrix. Cells are polyhedric with large nuclei, and some of them are showing evident nucleoli. Mitoses can be observed (arrows). Nonchondroid neoplastic cells containing melanin are also observed. HE. Bar = 50 μm.
Dog No. 8 had no chondroid cells or basophilic matrix, but abundant osteoid formations were distributed in large and anastomosing trabeculae. Tumoral cells were closely lining these trabeculae, and some of them appeared to be entrapped in the osteoid matrix.
In relation with the nonosteocartilaginous areas of the studied neoplasms, immunohistochemical analysis demonstrated an intense positive immunoreaction for vimentin in all the tumors. Immunolabeling with S100 and melan A also provided positive results for all the tumors, except in dog No. 10, which was negative for melan A. Labeling using S100 antibodies was stronger in all the dogs than labeling with melan A antibodies, as observed from the number of positive cells and from the intensity of the immunoreaction. No conclusions can be obtained from the differences in the intensity of the labeling, because different types of primary antibodies are used in the immunohistochemical techniques (polyclonal vs monoclonal). However, the use of melan A markers can be justified in our study because other S100-positive tumors, such as malignant peripheral nerve sheath tumors, show divergent osteocartilaginous differentiation. 3 Dog No. 10 was diagnosed as melanoma on the basis of the S100 positive reaction and the presence of moderate pigmentation. Pigmented melanomas positive for S100 but negative for melan A have been described. 10
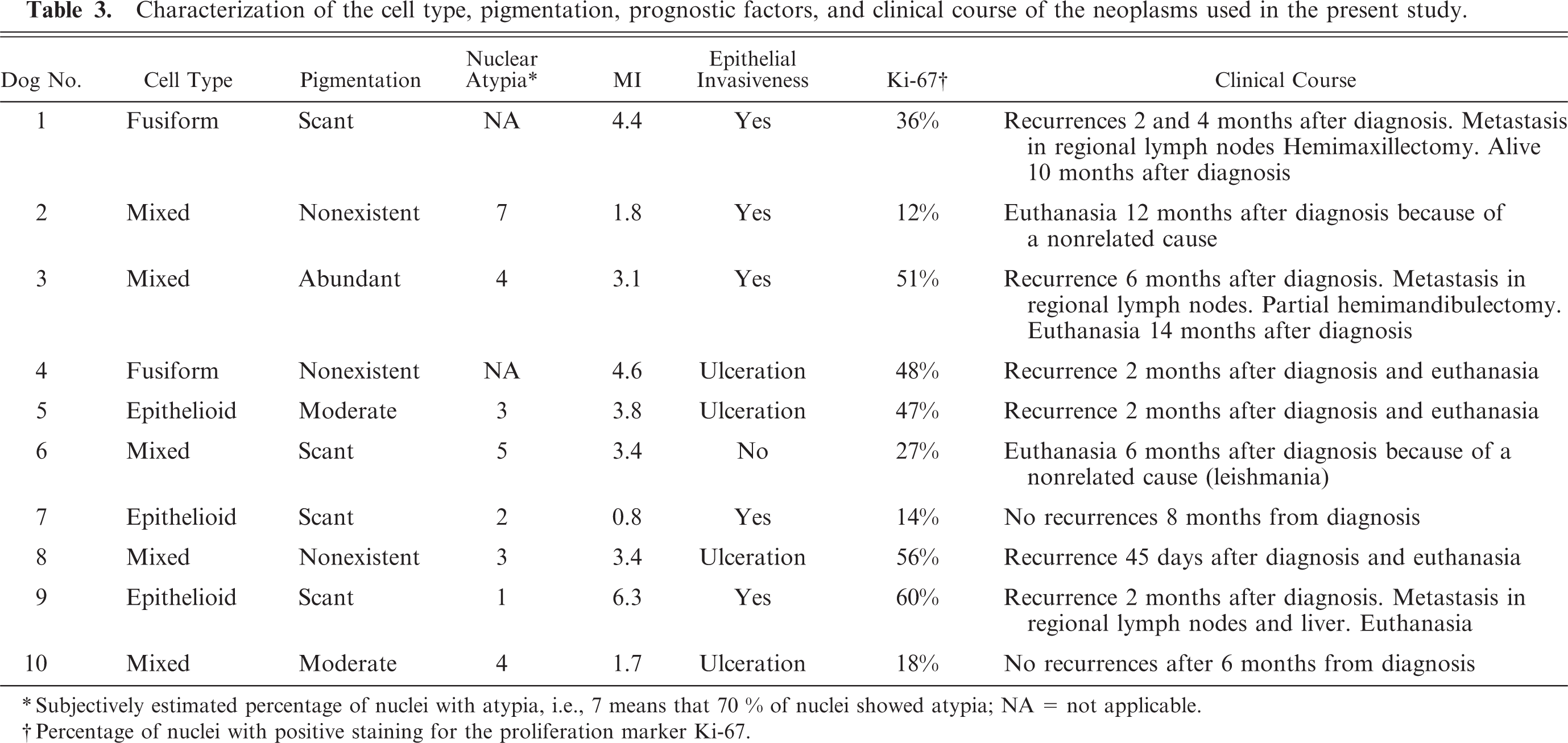
The immunohistochemical results of the osteocartilaginous areas was varied; in some cases (dog Nos. 2, 3, and 9) numerous chondroid anaplastic cells showed positive immunoreaction for melan A (Fig. 2), to the same extent as the typical melanocytic areas of the tumor; in other dogs (dog Nos. 1, 5 and 7) only small nests of positive-melan A cells were observed in the cartilaginous areas; no immunoreaction for melan A in cartilaginous areas was observed in any of the other dogs. The level of immunolabeling when S100 antibody was used was always strong (Fig. 3) in all the osteocartilaginous areas, especially in highly differentiated cells. No immunoreaction for cytokeratin, desmin, or actin was observed in any of the studied cases.
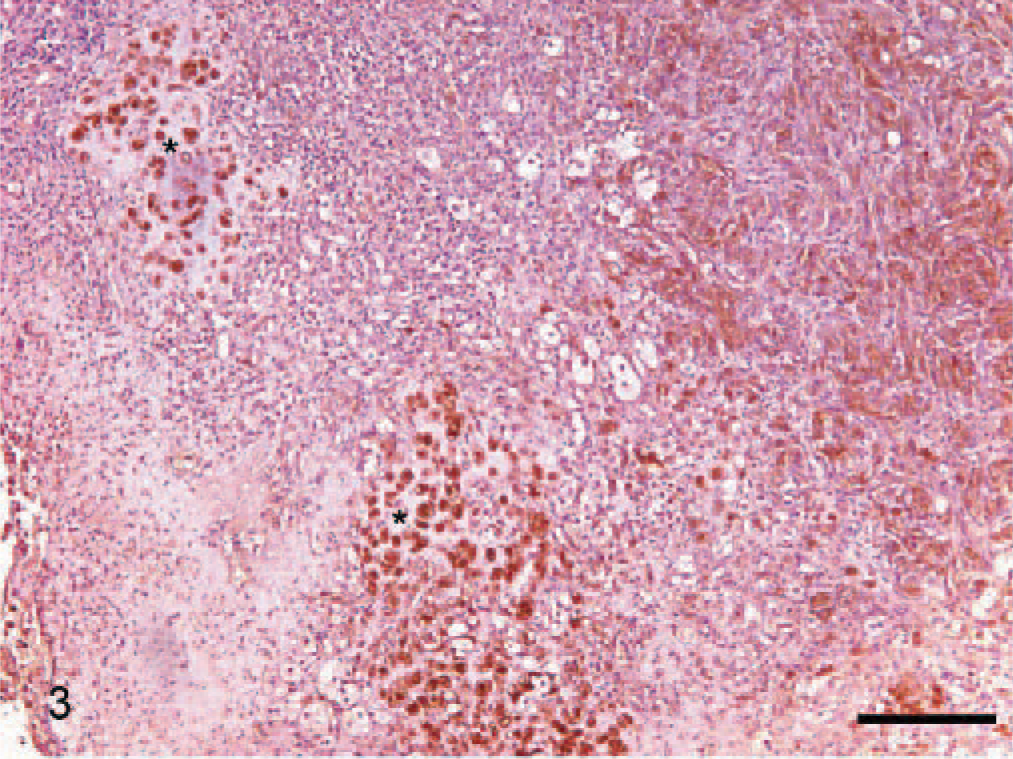
Dog No. 2. Immunolabeling for S100 protein in an oral melanoma. Cartilaginous areas (asterisks) were positive for S100 immunolabeling. ABC, Mayer's hematoxylin counterstain. Bar = 320 μm.
The analysis of four selected prognosis factors and the clinical course of the neoplasms after diagnosis provided a basis for the diagnosis and prognosis of future cases of this rare variant of oral melanoma (seeTable 3). The average mitotic index was 3.33 mitoses per hpf, very similar to the index reported by Ramos-Vara et al 16 as the mean of 129 oral melanomas. MI has been found useful for predicting malignant behavior in some reports 2, 20 but lacked significance in others. 8, 16 In our study, although the number of cases was low, MI seemed to be useful for predicting the biological behavior of the neoplasms, as the 4 highest MI showed recurrence 60 days after diagnosis and the 3 lowest MI showed no recurrence during the followup period (measured in days). All the tumors, except those in dog No. 6, showed invasion of the epithelium or ulceration. No relation was observed in our study between nuclear atypia and recurrence or metastasis. However, Spangler and Kass 20 demonstrated the usefulness of nuclear atypia in canine oral melanomas after an analysis of 104 cases; probably a larger number of cases is necessary for evaluating this prognostic factor. A close relation between prognostic factor and clinical course was found in the present study in the case of the proliferation index, measured as expression of the proliferation marker Ki-67 in the nuclei of neoplastic cells. Use of the proliferation index as a prognostic tool has been well established in recent years. 11, 15 The 6 neoplasms that showed a very high expression (>30% positive cells) in their nuclei (Fig. 4) showed recurrences (and sometimes metastasis) after 6 months. In short, our observations suggest that a MI >3 per hpf and a proliferation index >30% predict the early recurrence and possible metastasis of an oral melanoma with osteocartilaginous differentiation. Excluding the 10 cases of oral melanomas with osteocartilaginous differentiation presented in this study, to our knowledge only 4 other cases have been reported in veterinary medicine, 2, 9, 16 although in only 2 of them, detailed descriptions were available. 2, 9 Altogether, the epidemiologic data of the osteocartilaginous melanomas were similar to the data about classical oral melanomas reviewed by Smith et al. 19 As in the classical oral melanomas, osteocartilaginous melanomas affected old dogs (mean age of 11.8 years in our study, 12 and 11 years in the other reports) and had a certain predilection for small breeds; the cocker spaniel was the most represented (3 of the 12 total cases). A slight predilection for one sex could be observed in our cases (6 males versus 4 females), although this fact is probably meaningless because of the small number of cases. Finally, the most widely represented location was the gingival mucosa (10 of 14), and the labial mucosa was the target in the other four cases. No data about the clinical course, as a definitive measure of the biological behavior of this kind of tumor, has been reported previously; in our study, malignant behavior (recurrence and/or metastasis) was confirmed in 60% of the dogs, which is similar or even higher than the percentages reported recently in classical oral melanoma studies by Spangler and Kass 20 (59%) and Ramos-Vara et al 16 (43%). Only 3 cases of metastasis were observed in our study, although 3 dogs were euthanized without necropsy after early recurrence. Seven animals were euthanatized during the followup because of causes related (n = 5) or not related (n = 2) with the tumors. The average survival time after surgery for the 7 animals was 168 days, again similar to the data reported by Spangler and Kass 20 (118 days) and Ramos-Vara et al 16 (173 days). In conclusion, the clinical features and biological behavior of oral melanomas with osteocartilaginous differentiation seems to be similar to the behavior described for oral melanomas in 2 recent extensive retrospective studies.
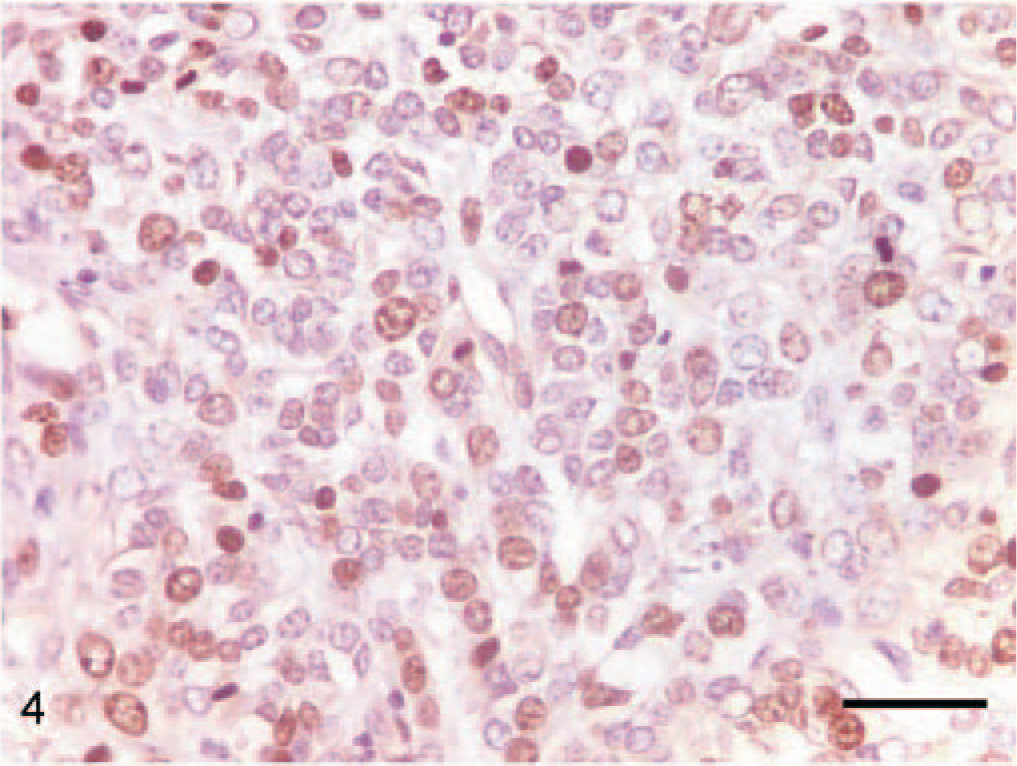
Dog No. 8. Expression of the proliferation marker Ki-67 in the nuclei of the neoplastic cells in an oral melanoma. Note the high number of positive nuclei. ABC, Mayer's hematoxylin counterstain. Bar = 40 μm.
Malignant melanoma with osteocartilaginous differentiation is a very rare variant of malignant melanoma, not only in veterinary medicine (1.5% of the canine oral melanomas described in a retrospective study 16 ) but also in human medicine, where only 20 cases have been described from 1985 to 2005. 4, 22 The origin of the osteocartilaginous components are not well determined but may result from pseudosarcomatous differentiation of neoplastic melanocytes or from metaplasia of the surrounding stroma in response to osteogenic factors produced during invasion by the melanoma cells. Although no samples from metastasis were analyzed in our study, reports in human cases have described the persistence of osteochondroid formation in lymph node metastasis of this kind of melanoma. 13 In our study, the close apposition of neoplastic melanocytic cells to the chondroid or osteoid areas, the existence of clearly anaplastic chondroid cells, and the positive cellular immunolabeling in the osteocartilaginous areas for S100 in all the tumors and for melan A in 6 of the cases, all indicated that the neoplastic melanocytic cells themselves were involved in the development of the osteocartilaginous areas. The occurrence of multyphenotypic cells, including mesenchyme and neuroectoderm, in a tumor in the head is no surprise because the mesenchyme in this region is derived from the neural crest. 12 Thus, an interesting hypothesis is that both typically melanocytic neoplastic cells and the cells of the osteochondroid component could be derived from common multipotent precursors. A similar pathogeny, with 2 populations and a common origin, has been postulated for the malignant ectomesenchymoma, 7 a rare soft tissue tumor described in human medicine composed of both mesenchymal (including cartilage) and neuroectodermal elements (including melanocytes); however, the clinical features and behavior of these tumors are very different from the behaviour of oral melanomas.
In summary, oral melanonas with osteocartilaginous differentiation show a clinical course similar to other melanomas in the oral cavity; analysis of the mitotic index and the expression of proliferation marker Ki-67 could be a useful tool for predicting the biological behaviour of these neoplasms. Also, our findings suggest that the osteochondroid formation observed could be a result of melanocytotic histogenesis.
Footnotes
Acknowledgements
We thank Juan Sanchez and the technical staff of Histovet (St Quirze del Valles, Barcelona) for their excellent assistance.
