Abstract
Intramedullary cavernous malformations (CVMs) of the spinal cord were diagnosed in 2 adult dogs that presented for paraparesis. An intramedullary spinal cord lesion was identified on a myelogram in the first dog, and expansion of the vertebral canal was evident on radiographs in the second. Extensive intraparenchymal hemorrhage was found on gross postmortem examination in both dogs, and a distinct lobulated intramedullary mass was evident in the second dog. Microscopically, both lesions were composed of dilated, thin-walled vascular channels with little-to-no intervening neural parenchyma. Both dogs had evidence of channel thrombosis along with perilesional hemorrhage and hemosiderin accumulation. The second dog had additional degenerative changes, including thickened fibrous channel walls with hyalinization, foci of mineralization, and occasional tongues of entrapped gliotic neuropil. CVMs appear to be an uncommon cause of both acute and chronic spinal cord disease in the dog.
Vascular malformations of the spinal cord are an infrequent cause of neurologic disease in both humans and animals. 1, 3, 9, 10, 12 These lesions are considered non-neoplastic hamartomas of blood vessels and can be classified on the basis of histopathology, anatomic location, and angioarchitecture. The most widely accepted classification system of central nervous system vascular malformations in people separates lesions into arteriovenous malformations (AVMs), cavernous malformations (CVMs), venous malformations, and capillary telangiectasia. 9, 12 Venous malformations and capillary telangiectasia do not carry a significant risk for acute hemorrhage and have been rarely described in the spinal cord. 12 Spinal cord AVMs in people can be further divided into dural arteriovenous fistulae and intradural AVMs, which include glomus, juvenile, and fistulous subtypes. 6 Human spinal AVMs and CVMs can be distinguished on the basis of distinct imaging and histopathologic findings. 4, 5, 12 Antemortem classification of spinal vascular hamartomas is clinically important in people since the treatment of AVMs and CVMs is usually different. To date, there have been only a few reports of CVMs in the brain and spinal cord of dogs, and consequently very little is known about these lesions in this species. 8, 11, 13 In this article, we describe the clinical and histopathologic features of intramedullary CVMs of the spinal cord in 2 adult dogs that both presented for paraparesis.
Dog No. 1, a 10-year-old intact male Catahoula Leopard dog, presented to the referring veterinarian with a 1-day history of pelvic limb discomfort and lameness. Over the next 3 days, the dog developed pelvic limb ataxia and progressive paraparesis and was referred to the North Carolina State University–Veterinary Teaching Hospital (NCSU-VTH). On initial neurologic examination at the NCSU-VTH, the dog was nonambulatory paraparetic. Abnormal findings included pelvic limb postural reaction deficits and absence of the cutaneous trunci reflex caudal to the eighth thoracic vertebra (T8). The lesion was localized between the third thoracic and third lumbar (T3 to L3) spinal cord segments, with a specific localization to the T6-T7 spinal cord segments based on the cutaneous trunci reflex cut-off. There were no specific findings on routine blood work; urinalysis revealed marked hematuria and proteinuria. The dog was started on amoxicillin-clavulanic acid pending an aerobic urine culture, and a transdermal fentanyl patch (75 μg/hr) was placed.
Lateral spinal radiographs revealed caudal lumbar spondylosis deformans. Cisternal cerebrospinal fluid analysis was unremarkable other than a mild elevation in protein (0 nucleated cells/μL and 27.6-mg/dL protein). A myelogram disclosed an intramedullary lesion from approximately T5 to T8 (Fig. 1A, B). CT was performed immediately after the myelogram but did not provide any additional information. Intramedullary neoplasia, hematomyelia, or an ischemic myelopathy secondary to fibrocartilaginous embolism was considered the most likely differential diagnosis. One day following the myelogram, the dog became painful along the mid-thoracic vertebrae, and neurologic status deteriorated to paraplegia. The dog was treated supportively in the hospital, but clinical signs progressed to pelvic limb analgesia 2 days later. The dog was euthanized because of a poor prognosis for recovery.
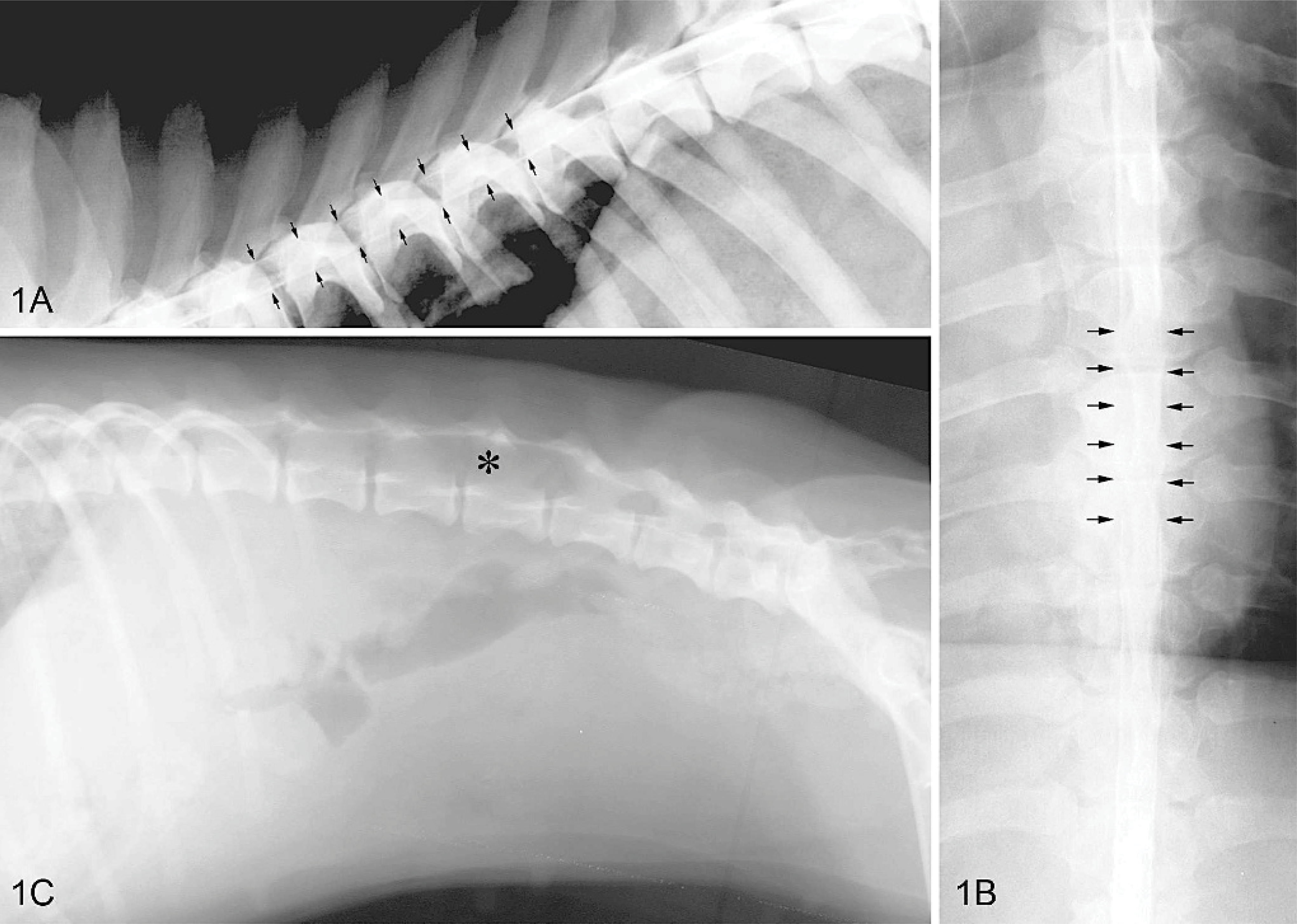
Lateral myelogram; dog No. 1. Subtle thinning of the dorsal and ventral contrast columns is seen over the sixth, seventh, and eighth thoracic vertebrae (arrows). Fig. 1B. There is complete attenuation of contrast medium on ventrodorsal views of the myelogram (arrows) at the same level. A lack of spinal cord compression on orthogonal radiographs indicates an intramedullary lesion. Fig. 1C. Lateral abdominal radiograph; dog No. 2. Note the marked expansion of the vertebral canal between L3 and L5 (asterisk) due to vertebral remodeling from a chronic intramedullary space-occupying mass.
Gross postmortem examination revealed locally extensive spinal cord hemorrhage and mild enlargement of the seventh and eighth thoracic spinal cord segments. A cross-section of the enlarged spinal cord appeared dark red and friable with a thin rim of normal parenchyma compressed against the dura mater. On microscopic examination of paraffin-embedded sections stained with HE, nearly 60% of the spinal cord was replaced by a nonencapsulated, spheric mass of thin-walled, blood-filled, vascular channels (0.1–50 mm in diameter) that were closely apposed with scant intervening spinal cord parenchyma (Fig. 2A). Vascular channels were lined by a single endothelial layer and supported by a thin fibrous wall (Fig. 3A). The largest channel was nearly occluded by a laminated, mixed blood and fibrin thrombus with very early cellular reorganization at its attachment sites to the wall. Mild patchy hemorrhage and accumulation of a few hemosiderin-laden macrophages were present in the adjacent neuropil. Factor VIIIRa immunohistochemical staining confirmed the endothelial lining of vascular channels, and Masson trichrome staining highlighted fine collagenous support stroma. Verhoeff-Van Gieson staining did not reveal an internal elastic lamina as seen in normal dural arteries, and elastin fibers were not present. Smooth muscle actin (SMA) immunohistochemical staining revealed scattered individual and small clusters of moderately positive cells in the walls and at the attachment sites of the thrombus. A discrete layer of SMA-positive smooth muscle cells, as seen in the tunica media of normal arteries and veins, was not seen. Throughout the adjacent, compressed spinal cord, there was axonal degeneration and demyelination associated with gliosis and mild-to-moderate vascular hypertrophy and hyperplasia. Central areas of white and gray matter hemorrhage and necrosis extended into adjacent spinal cord segments. The morphologic diagnosis was intramedullary spinal cord CVM.
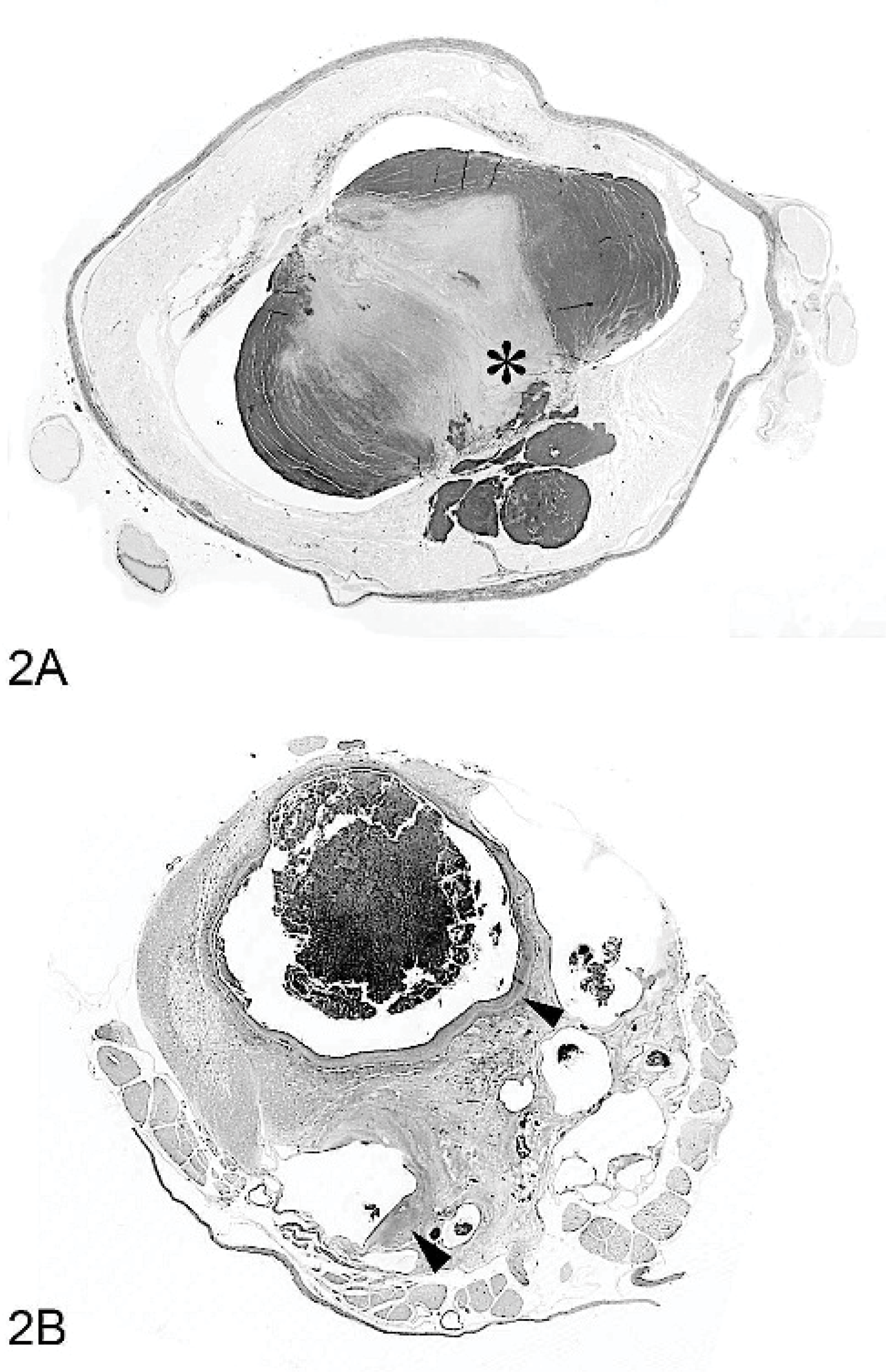
Spinal cord, T7-8; dog No. 1. A transverse section of the spinal cord contains an intramedullary expansile mass of small and large, thin-walled vascular channels. The largest channel contains a central attached thrombus (asterisk).
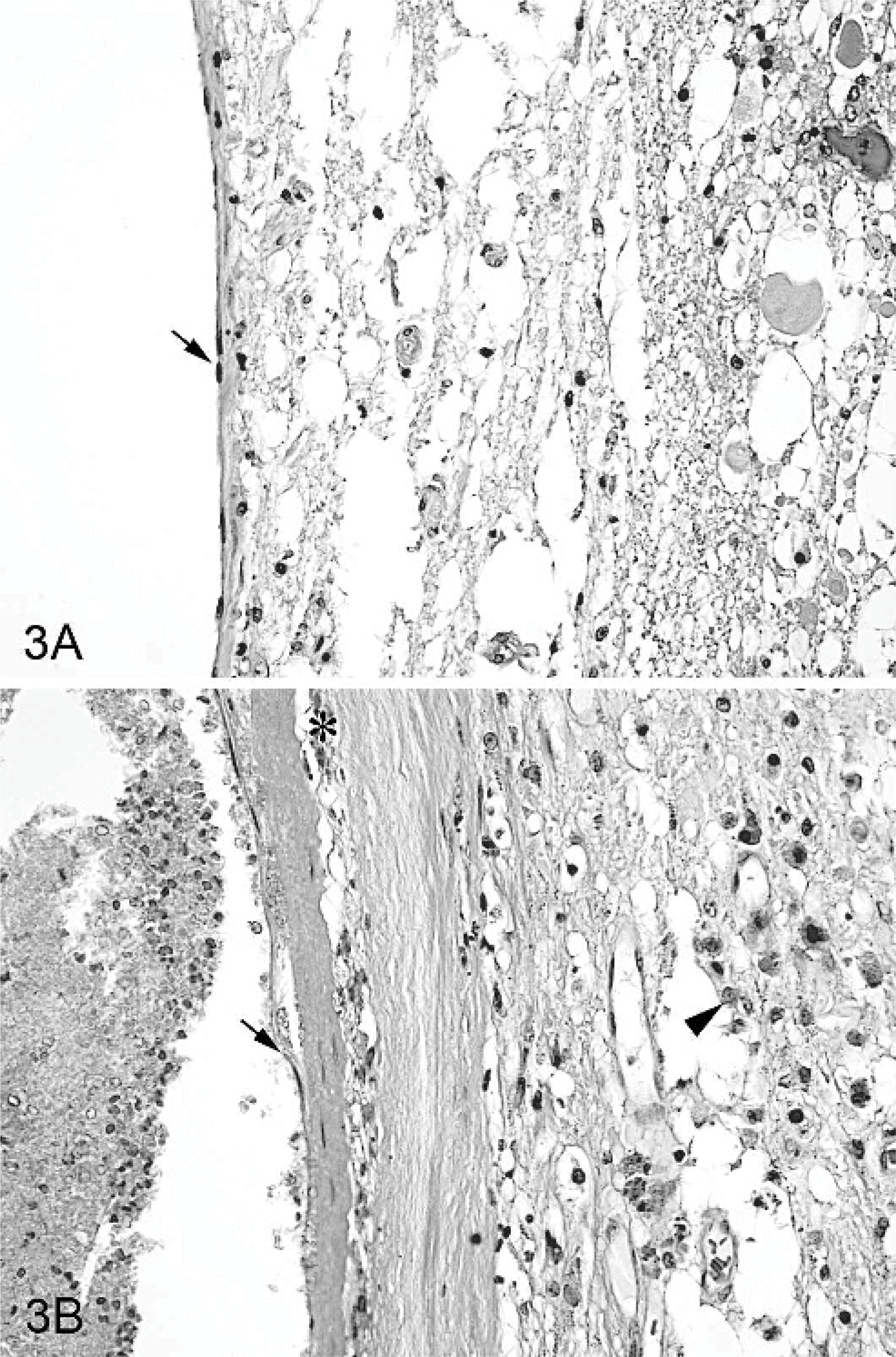
Spinal cord, T7-8; dog No. 1. Endothelial cells (arrow), supported by a thin layer of fibrous connective tissue, line the vascular channel walls. Adjacent degenerating neural parenchyma is vacuolated and contains pale eosinophilic axonal spheroids.
Dog No. 2, an adult female Beagle, was found stray and brought to a local emergency clinic because of numerous abrasions over the dorsal lumbar spine. Physical examination found the animal to be dehydrated, weak, and in poor body condition. Multiple infected wounds were found on the dorsum in the mid lumbar region, and palpation of the area elicited pain. Decreased proprioceptive positioning of both pelvic limbs, and fecal and urinary incontinence were noted. Notable laboratory findings included a marked neutrophilic leukocytosis, pyuria, and bacteruria. Pyometra was suspected, but abdominal radiographs were inconclusive. Expansion of the vertebral canal and thinning of the dorsal lamina from L3 to the cranial aspect of L5 were visible on the radiographs (Fig. 1C). The dog was treated empirically with antibiotics and intravenous fluids. Complete blood count was repeated 3 days later, and the leukocytosis had resolved. Intravenous fluids were discontinued, and prednisone was added to the treatment regimen. The dog was referred to NCSU-VTH after approximately 2 weeks of hospitalization because of persistent pelvic limb deficits and fecal and urinary incontinence.
On neurologic examination, the dog was paraparetic, with a wide-based pelvic limb posture. Proprioceptive positioning and other postural reactions were decreased in both pelvic limbs. Pelvic limb withdrawal and perineal reflexes were both decreased, but patellar reflexes and tail tone were normal. The lesion was localized to the L6-S3 spinal cord segments, and on the basis of radiographs, the most likely differential diagnosis was spinal cord neoplasia. The dog was euthanized and submitted for necropsy.
At necropsy, there was a large, firm, lobulated mass within the spinal cord at the level of the L4 vertebra with associated hemorrhage. The vertebral canal was expanded by the mass, and the vertebral pedicles bowed around the enlarged spinal cord. On microscopic examination, the spinal cord was replaced by a centrally located mass with similar morphology to the mass described in the first dog (Fig. 2B). There was slightly more intervening neuropil between some but not all of the vascular channels. The largest channel was 8 mm in diameter and contained an attached thrombus composed primarily of fibrin and erythrocytes, in which there was surface endothelialization. There was extensive, asymmetric, patchy-to-segmental, fibrous thickening and hyalinization of the vascular channel walls. There was occasional connective tissue mineralization of channel walls and a few small vessels in the scant neuropil incorporated into the mass but not in the adjacent dura. Hematoidin and macrophages filled with hemosiderin were prominent in thickened channel walls and the adjacent neuropil (Fig. 3B). In larger channels, occasional small tonguelike projections of gliotic neuropil extended from the walls into the vascular lumens (“gliotic tongues”). Prominent Masson trichrome staining of the thin walls was similar to that of the first dog, but there was less staining of thickened and degenerative areas. An internal elastic lamina was absent, and thin-walled channels lacked elastin fibers; however, Verhoeff-Van Gieson staining did reveal mild elastosis with scattered irregular areas of fine elastin accumulation in thickened and degenerative walls of some channels. SMA staining of individual cells was rare in the thin walls of channels, but there was prominent, patchy, and irregular SMA staining of cells in the thickened walls. Factor VIIIRa staining confirmed a simple endothelial lining of vascular channels. Axonal degeneration, parenchymal compression, and atrophy were also observed in the adjacent spinal cord along with gliosis. The final diagnosis was intramedullary CVM of the spinal cord.
CVMs of the central nervous system, also known as cavernomas, cavernous angiomas, or cavernous hemangiomas, are well described in people, but there is little published information on these tumors in animals. 12 Grossly, CVMs in people have a “mulberry-like” appearance and may be sharply demarcated from the adjacent parenchyma by a yellow rim representing hemosiderin staining and gliosis. Microscopically, channels lack clear features of arteries or veins and are lined by a single layer of endothelium supported by a thin connective-tissue matrix. 5, 12 The 2 canine spinal cord masses reported here are morphologically consistent with CVMs reported in people. In both cases, the focal, intramedullary, expansile masses were composed of dilated and thin-walled vascular channels with little-to-no intervening neural parenchyma. 2, 5 As is common in human lesions, thrombosis, hemorrhage, and hemosiderin accumulation were observed in both cases reported here. In dog No. 2, there were additional degenerative changes that have been reported in people, including thickened fibrous channel walls with hyalinization, foci of mineralization, mild elastosis, and occasional tongues of entrapped gliotic neuropil. 5 Human CVMs and our canine cases lack clear features of either arteries or veins seen in spinal cord AVMs. Morphologic features of capillary telangiectasia or venous malformations reported in the brain were not seen our dogs. 12
To these authors' knowledge, there is only 1 other description of a spinal cord CVM in a dog. 13 As a result, very little is known about the natural progression, imaging findings, treatment, and histopathologic features in this species, although some information may be inferred from human medicine. The etiology of these masses is unknown, although in humans, there is a familial form that is inherited in an autosomal dominant pattern with incomplete penetrance. 12 Most human spinal CVMs are believed to be congenital in origin, although symptoms usually develop in the third or fourth decade of life. 2 Both dogs described in this report were mature; however, the other report of a canine CVM of the spinal cord was in a 4-month-old animal. 13
CVMs are thought to grow through recurrent hemorrhage and thrombosis of vascular sinuses causing acute expansion. 5 Symptoms may follow an acute, progressive, or episodic course. 2, 5 Acute signs are usually caused by hemorrhage into the vascular spaces or the adjacent spinal cord (hematomyelia). The initial onset of symptoms may be sudden, and neurologic deterioration usually progresses over a few hours to days, as seen in the first dog reported here. 6 However, symptoms of CVMs in many people progress over months to years, with either a slow or episodic deterioration of neurologic function. 2 The most common clinical course appears to be episodic neurologic dysfunction, with variable recovery following deterioration. 4 Dog No. 1 had an apparently acute and progressive clinical course. A chronic course, either progressive or punctuated, was likely for dog No. 2 based on the vertebral remodeling and histopathologic evidence of chronic hemorrhage. However, because the dog was found as a stray, the true history is not known.
Myelography and postmyelographic CT findings may be normal or nonspecific in human CVMs, as seen in the first dog reported here. 5 Magnetic resonance imaging (MRI) and selective spinal angiography may, however, permit definitive diagnosis and therapeutic planning for endovascular occlusion (embolization), surgical ligation/resection, or both. 4, 6 CVMs have a characteristic MRI appearance of a central core of mixed signal intensity from hemoglobin at varying stages of evolution. A hypointense peripheral rim representing hemosiderin deposition is frequently seen on T2-weighted images. 4 Unlike most AVMs, CVMs are angiographically occult because of their low blood flow. While endovascular occlusion plays a significant role in the treatment of AVMs in people, surgical excision is the treatment of choice for CVMs. Microsurgical techniques and ultrasonic aspirators have helped to provide a generally good postoperative outcome following resection of spinal CVMs in people. 2, 5 No attempts have been made to remove either intracranial or spinal CVMs in dogs, and so the outcome with surgery is unknown. There is a report of successful surgical treatment of a spinal vascular hamartoma in a dog, suggesting that surgery can be effective for some spinal vascular malformations. 7 The increased use of MRI in veterinary medicine may lead to an increase in the number of CVMs that are identified and subsequently treated. 8 Prompt surgical intervention may have prevented the devastating clinical deterioration that occurred in the first dog.
