Abstract
Although it has been shown that auricular chondritis in rats is caused by the use of metal identification ear tags, the pathogenesis remains unclear. Based on the hypothesis that the auricular chondritis is caused by metal ions released from metal identification ear tags, we investigated the pathogenesis in male C57BL/6 mice tagged with metal identification ear tags. Twenty-six weeks after the attachment of the ear tags, visible increases in the thickness of the auricle were observed, and the concentrations of copper and iron in the tagged ears were significantly increased (P <.05) in the tagged ears compared with the untagged ears. There was up-regulation of metallothionein (MT)-I and MT-II mRNA in the tagged ears, and this was confirmed by immunohistologic staining of the destroyed cartilage. Histopathologically, there were observed severe chondritis with extensive granulomatous inflammation, newly formed cartilage nodules, and osseous metaplasia accompanied by cellular infiltrates, such as CD4 T lymphocyte, macrophages, neutrophils, and mast cells, and expression of Th1 cytokines, such as interferon-gamma, tumor necrosis factor-alpha, and interleukin-2 in the tagged ear. Based on these results, we concluded that the release of copper and iron ions from the metal ear tags played a major role in the onset of auricular chondritis. Subsequent cellular interactions, such as CD4 T cells, macrophages, fibroblasts, and mast cells, mediated by cytokines, such as tumor necrosis factor-alpha and interferon-gamma, caused an autoimmune response that may have led to the progression of auricular chondritis as an autoimmune disease.
Introduction
Spontaneous auricular chondritis, also known as auricular chondropathy, is characterized grossly by bilateral nodular to diffuse thickening of auricles in aged rats 5 and bears some resemblance to relapsing polychondritis (RP) in humans. 15 Histopathologically, the lesions have been described as consisting of focal or multifocal granulomatous inflammation with destruction of the cartilage plate, formation of new cartilaginous nodules, and osseous metaplasia. Similar lesions have been experimentally induced in the animal model for human RP, type-II collagen immunized rats 6, 20 and mice, 10 and HLA-DQ6/8 double and NOD.DQ8.Aβo (Ag7-/-) transgenic mice, 3, 19, 30 suggesting that an immunologic process is related to the pathogenesis.
In some reports, it has been suggested that the individual identification method using metal ear tags plays a role in the development of spontaneous lesions. 18, 21 Similarities in the auricular lesions include 1) lesions developed bilaterally in the later stages but developed initially in the tagged ear; 2) traumatic insult from the ear tagging procedure healed within a few weeks, and only after a prolonged time interval did the tagged auricle begin to increase in thickness with accompanying erythema indicative of inflammation; and 3) lacerations in most animals that lost their metal ear tags healed without any ear deformity. Moreover, other methods of identification, such as nylon ear tags, ear punching/notching, and color markings on the dorsum, and traumatic insults to the pinnal cartilage do not cause similar lesions. 17, 21 Based on these similarities, it is reasonable to suppose that the lesions are triggered by a factor other than traumatic insult due to ear tagging.
In this study, we hypothesized that metal ions released by metal ear tags may play a key role in the onset of auricular chondritis associated with ear tagging. Based on this hypothesis, a comprehensive study was performed using mice to better understand the initial pathologic events resulting from the use of metal ear tags.
Materials and Methods
Animals
Forty 5-week-old male SPF C57BL/6 mice obtained from Hoshino Laboratory Animals (Saitama, Japan) were examined. The animals were maintained in a specific pathogen free-facility and were housed individually in polycarbonate cages (136 × 208 × 115mm; Clea Japan Inc., Tokyo, Japan). Sterilized drinking water and commercial diet (Oriental Yeast, CRF-1, Tokyo, Japan) were given ad libitum. Ambient lighting was automatically regulated on a 12-hour light/day cycle. All animal studies were approved by the Shiseido Research Center Subcommittee on Research Animal Care. Upon arrival in our facilities at 5 weeks of age, metal ear tags, for identification purposes, were attached to the base of the right ear, penetrating through skin and cartilage, using a specifically designed applicator. The metallic ear tags are composed of a nickel-copper alloy called “monel metal.” Using an X-ray analytical microscope (XGT5000; Horiba, Kyoto, Japan) operating at 50 kV and 0.1 mA, we analyzed the metal components of the ear tags and showed that they consisted of 63.9% ± 0.3 nickel, 33.7% ± 0.3 copper, 1.5% ± 1 iron, and 0.87% ± 0.1 manganese.
Measurement of the ear thickness
Auricular lesion was monitored grossly from the day the ears showed redness and swelling, indicative of inflammation. All mice were regularly checked for any other clinical manifestations. To quantify the gross changes observed in the ears, changes in ear thickness was measured using a calibrated gauge (Digimatic caliper; Mitsutoyo Co., Ltd., Kanagawa, Japan) once every 2 weeks. Each ear was measured at 3 different points/areas (medial, center, and lateral areas) of the auricle in millimeters and are reported as mean ± SE. In addition, microcomputed tomography of tagged and untagged (contralateral) ears 26 weeks after attachment of metal ear tags was done using the eXplore Locus MicroCT System (GE Healthcare, Waukesha, WI, USA).
Ear tissue sample preparation
Affected mice were euthanized with an overdose of isoflurane, and the tagged and untagged (contralateral) ears were surgically removed 26 weeks after the tagging procedure. Each ear sample was longitudinally halved from the center. For histopathology, all ear sample halves were fixed in 10% buffered formalin for 24 hours at 4°C. The other halves (case Nos. 1–13) were randomly selected for analysis, i.e., 3 for RNA isolation (case Nos. 1–3), 5 for metal content and cytokine measurement (case Nos. 4–8), and 5 for immunohistochemistry (case Nos. 9–13). For analysis of RNA isolation, metal content, and cytokine measurement, each pinna (approximately 50 mg) was thoroughly washed in PBS, wiped of excess buffer solution, and frozen immediately using liquid nitrogen. The frozen ear samples were pulverized using a Cryo-Press device (Microtec, Chiba, Japan). For immunohistochemistry, ear tissues were embedded in optimal cutting temperature compound (Sakura Finetek USA Inc., Torrance, CA, USA) and snap frozen in liquid nitrogen.
Histolopathology and immunohistochemistry
Formalin-fixed halves of ear samples were embedded in paraffin; sectioned at 5 μm; and stained with HE, toluidine blue, periodid acid-Schiff, and Giemsa's method for light microscopic observation. All major organs/tissues, including trachea, larynx, nose, and bronchus, from each mouse were routinely processed for histopathology. Ear tissue sections were also prepared for immunohistochemical staining of anti-MT and anti-PCNA (PC10) staining. The sections (case Nos. 9–13) were deparaffinized in xylene and rehydrated with serial dilutions of graded alcohol (100%, 90%, and 70% ethanol/distilled water), and endogenous peroxidase was blocked with 3% hydrogen peroxide in methanol for 10 minutes at room temperature.
Frozen sections were prepared for anti-CD4, anti-CD8, anti-Mac-1, and anti-B220 staining. The sections were cut at 5 to 7 μm, air dried, and fixed in acetone for 2 minutes at −20°C. Endogenous peroxidase activity was blocked with 0.3% hydrogen peroxide in PBS (frozen sections) for 10 minutes at room temperature. Serial sections were incubated with corresponding primary antibodies overnight at 4°C. After incubation with the primary antibody, tissues were applied with a biotinylated goat anti-mouse IgG as secondary antibody, followed by streptavidin-peroxidase conjugate as described by the manufacturer (BD Biosciences Pharmingen, San Diego, CA, USA). After 3 washings with PBS, the antibody binding sites (positive reactions) were stained with 3,3′-diaminobenzidine (Dako Cytomation Inc., Carpinteria, CA, USA). Color development was monitored by visual inspection and stopped by rinsing in distilled water. Sections were counterstained with hematoxylin, dehydrated, cleared, and mounted with Mount-Quick (Daido Sangyo, Saitama, Japan). The antibodies used in this study were anti-MT-I/II (E9; DAKO, 1:100), anti-PCNA (PC-10 clone; Santa Cruz Biotechnology, Santa Cruz, CA, USA, 1:600), rat anti-mouse CD4 (RM4-5; BD Biosciences Pharmingen, San Diego, CA, USA, 1:100), rat anti-mouse CD8a (Ly-2; Pharmingen, 1:200), rat anti-mouse Mac-1 (M1/70; Pharmingen, 1:200), and rat anti-mouse CD45R/B220 (RA3-6B2; Pharmingen, 1:200).
Analysis of metal content in ear tissue
Frozen pulverized ear samples were dissolved in 50 mg/200 μL (PBS with 0.5% BSA). After letting them stand on ice for 20 minutes, the cell extracts were centrifuged at 12,000 rpm (× 11,000 g) for 20 minutes at 4°C, and the resulting supernatant was used for copper, iron, nickel, and manganese estimation by X-ray fluorescence spectrometer (RIX2000; Rigaku Corp., Tokyo, Japan). Metal levels were expressed as μg/g tissue, and data were displayed as mean ± SE.
Cytokine measurements
Frozen pulverized ear tissue samples were prepared as described for the analysis of metal content. Interleukin (IL)-2, IL-4, IL-5, IL-10, IL-12 (p70), granulocyte-macrophage colony-stimulating factor, interferon (IFN)-γ, and tumor necrosis factor (TNF)-α content in cell extracts were measured by multiplex cytokine array analysis using the Bio-Plex protein multiarray system (Bio-Rad, Tokyo, Japan) according to the recommendations of the manufacturer as described previously. 34 Cytokine levels were expressed as pg/g tissue, and data were reported as mean ± SE.
Reverse transcriptase polymerase chain reaction
Frozen pulverized ear samples were dissolved in 1 mL of Isogen reagent (Nippon Gene, Toyama, Japan), and total RNA was isolated as recommended by the manufacturer. Total RNA concentration and purity were determined by measuring the OD260 and OD260/280, respectively, on a NanoDrop spectrophotometer (NanoDrop Technologies, Rockland, DE, USA). Total RNA samples were reverse transcribed with a GeneAmp RNA polymerase chain reaction (PCR) kit (Applied Biosystems, Foster City, CA, USA). Briefly, 1 μg of RNA was added to a tube containing 5 mM MgCl2, 1 mM each of dNTP (A,T,G,C), 50 mM KCl, 10 mM Tris-HCl (pH 8.3), 2.5 μM random hexamer, 1 U of RNase inhibitor, and 2.5 U of murine leukemia virus reverse transcriptase. The mixture was incubated at 42°C for 60 minutes, heated to 95°C for 5 minutes, and placed on ice until used for PCR. The single-stranded cDNA was then amplified by PCR in a total volume of 20 mL containing 7 pmol each of specific sense and antisense primers, 1.5 mM MgCl2, 2.5 mM each of dNTP (A, T, G, C), 50 mM KCl, 10 mM Tris-HCl (pH 8.3), 0.5 units of AmpliTaq DNA polymerase (Applied Biosystems). The PCR condition was 94°C for 5 minutes, 92°C for 30 seconds, 55°C for 30 seconds, and 72°C for 30 seconds for 25 to 30 cycles and a final extension time of 10 minutes at 72°C. Polymerase chain reaction products were analyzed by electrophoresis through 2.0% agarose gel. The primers used in this study were murine MT-I (sense, 5′-ATGGACCCCAACTGCTCCTGCTCCACC-3′; antisense, 5′-GGGTGGAACTGTATAGGAAGACGCTGG-3′) and MT-II (sense, 5′-GCTCCTGTGCCTCCGATGGA-3′; antisense, 5′-GAGAACGGGTCAGGGTTG-3′) and β-actin (sense, 5′-CCGTGAAAAGATGACCCAGATCATG-3′; antisense,5′-GATGCCACAGGATTCCATACCCAAG-3′). The mRNA expression of β-actin was used as an internal standard for comparisons between samples.
Statistical analysis
Data were analyzed with Student's t-test using SAS statistical program (SAS software ver. 8.2, SAS Institute Inc., Tokyo, Japan) and were considered statistically significant at P < .05.
Results
Gross findings
Clinically, the ear lesions were initially observed only on tagged ears within 1 week after the attachment of the metal ear tag. The area surrounding the tag showed varying degrees of erythema and swelling. Although the lesions healed within 2 weeks in most mice, about 32.5% (13 of 40) of the tagged mice started to develop hyperemia and palpable thick firm nodules. In the remaining 27 mice, no gross pathologic changes were observed at the end of study. Compared with the untagged ears, obvious increases in the thickness of tagged ears were observed, and the differences were statistically significant from approximately 18 weeks after tagging (Fig. 1). Affected ears were thickened twice or more with erythema in comparison with the untagged ear auricles at the end of study (Fig. 2). In addition, no gross lesions were observed in the other cartilage organs at the necropsy (data not shown).
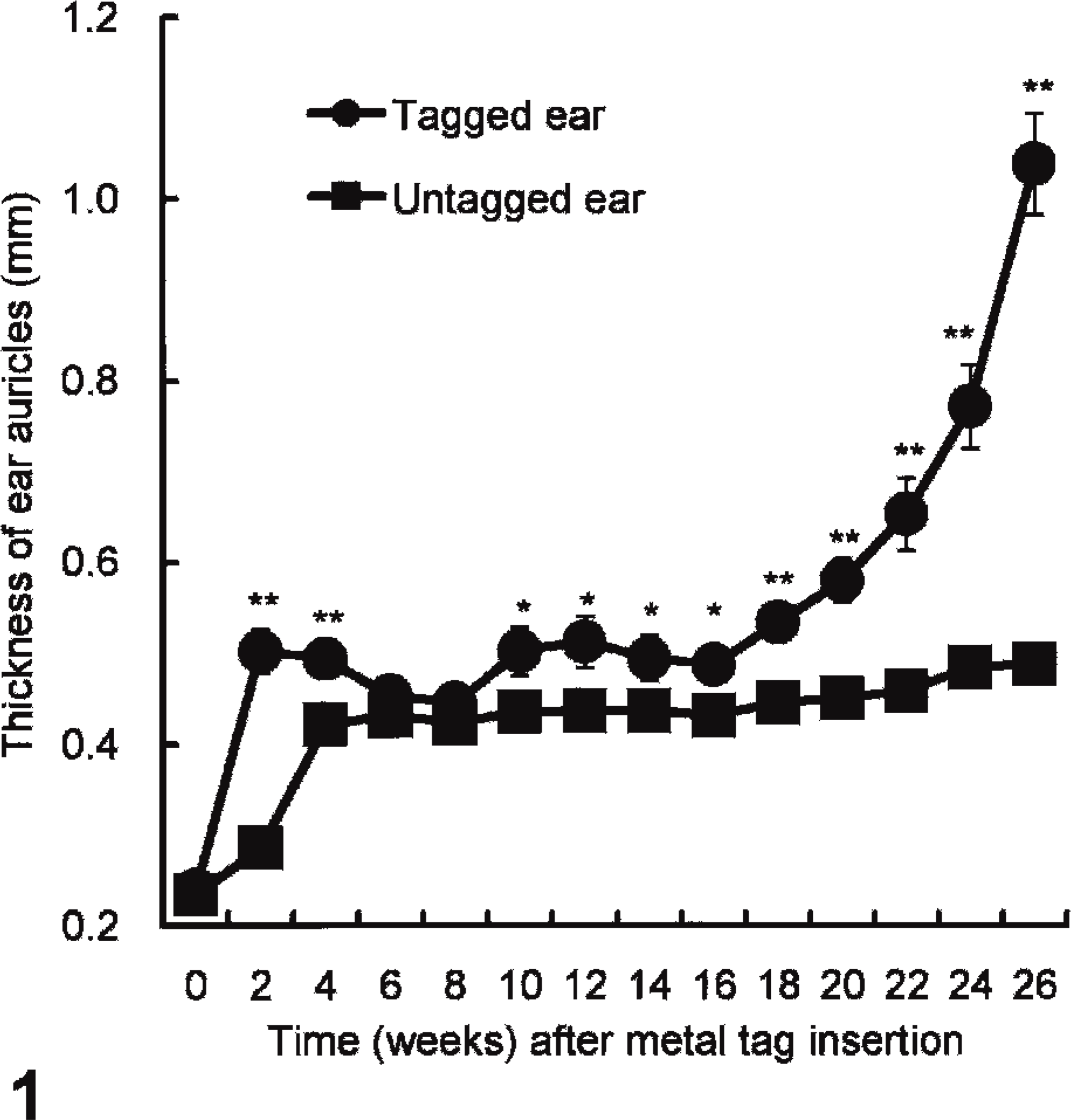
Average change in the thickness (mm ± SE) of the tagged and untagged ear auricles over time (weeks) after insertion of the ear tag. (∗P < .05, ∗∗P < .01; Student's t-test).
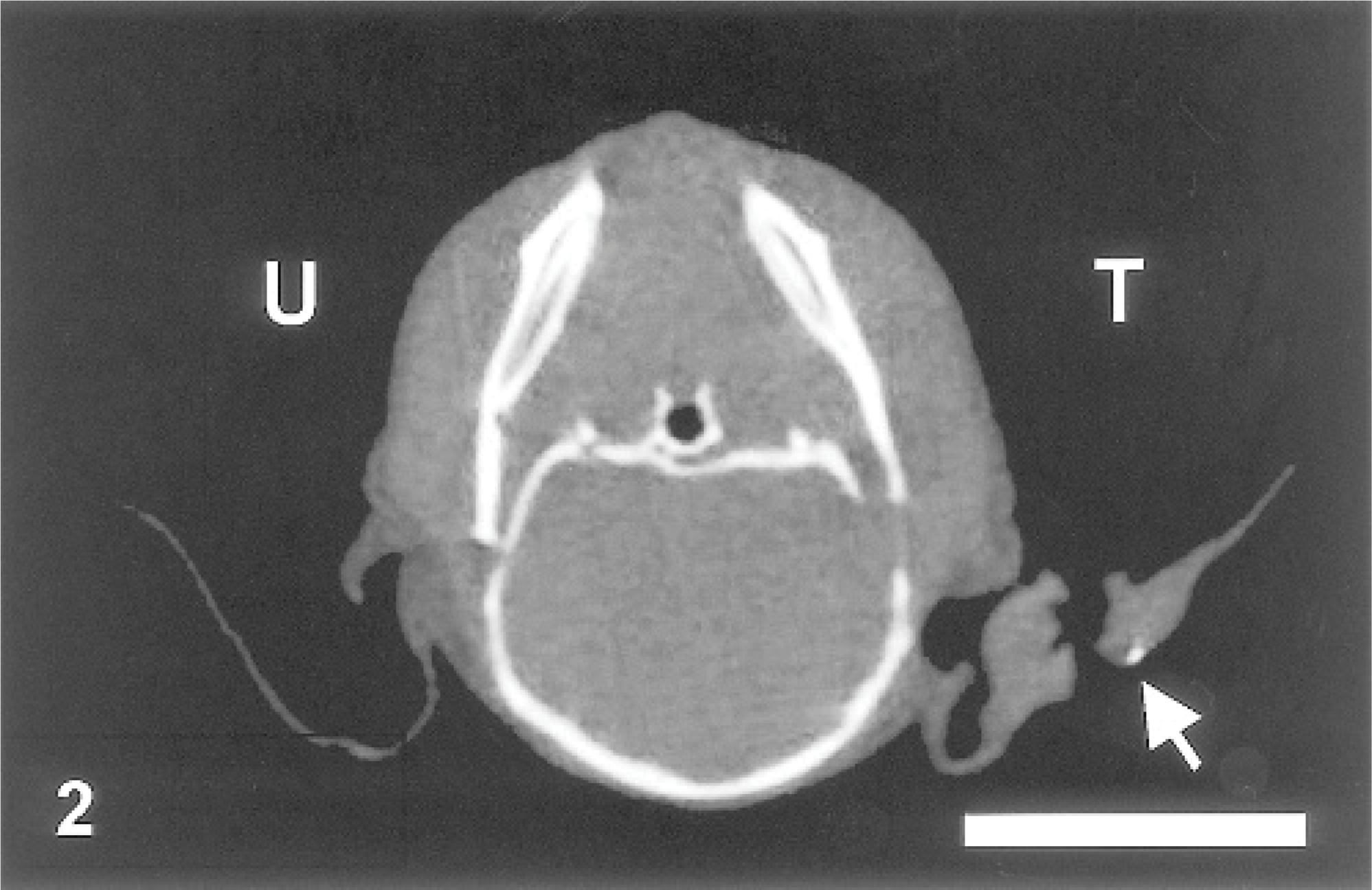
Computed tomography (CT) scan. Ear; mouse No. 1. Micro-CT image of the tagged (T) and untagged (U) ear auricles 26 weeks after metal tag insertion. Arrow indicates bone metaplasia. Bar = 1 cm.
Metal concentration in ear auricles
The concentrations of copper and iron of tagged ear tissues samples were significantly higher than those of untagged ears (P < .05) (Table. 1). Nickel concentrations were not significantly different, and no manganese was detected.
Metal concentrations(μg/g tissue) in tagged and untagged ear tissue samples (n = 5).
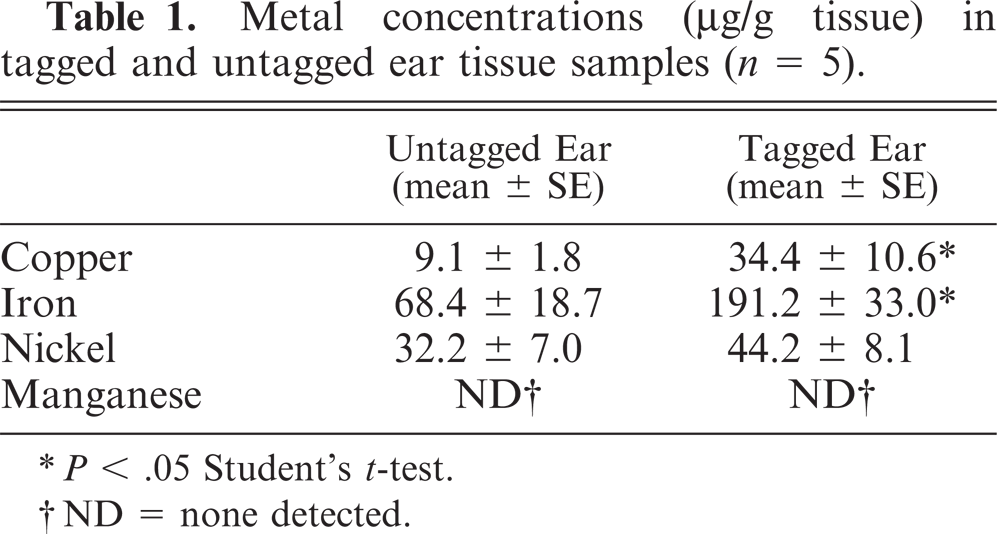
∗P < .05 Student's t-test.
†ND = none detected.
Analysis of MT-I and MT-II isoform specific expression using reverse transcriptase polymerase chain reaction
To confirm whether auricular chondritis is associated with mRNA expression of MT-I and MT-II, we performed reverse transcriptase polymerase chain reaction, with β-actin as an internal control. Expression of MT-I and MT-II mRNA in tagged ears was clearly enhanced compared with untagged ears (Fig. 3).
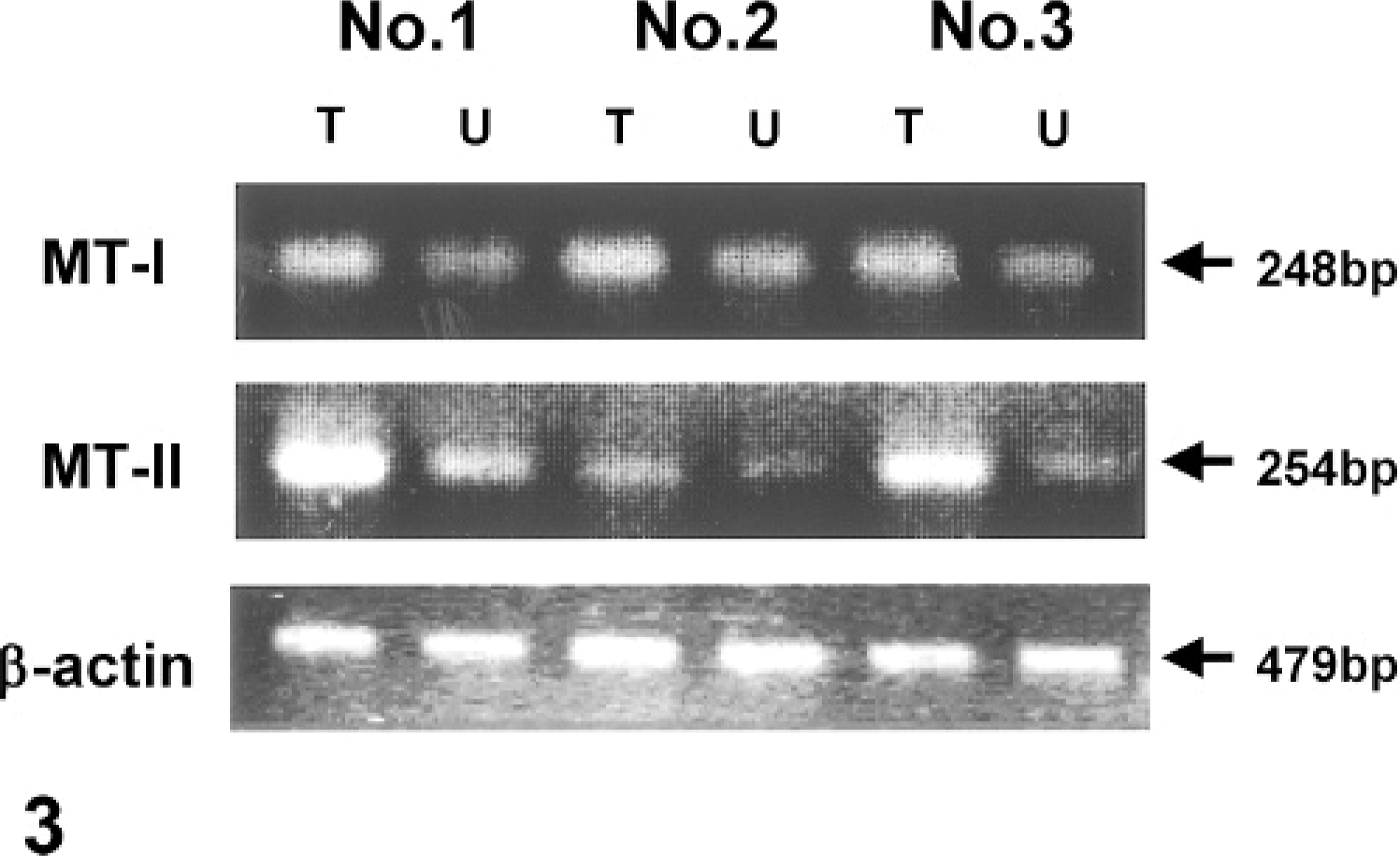
MT-I and MT-II expression in ear tissue samples from tagged (T) and untagged (U) ears using reverse transcriptase polymerase chain reaction (Nos. 1–3).
Cytokine production
Cytokine production in ear tissue samples were evaluated using a multiplex cytokine array analysis (Table 2). Two to 4-fold increases in the concentrations of IL-2, IFN-γ, and TNF-α were significantly higher in tagged ears (P < .05). Concentrations of IL-4, IL-5, IL-10, IL-12, and granulocyte-macrophage colony-stimulating factor were not significantly different between the tagged and untagged ears.
Cytokine concentrations (pg/g tissue) in tagged and untagged ear tissue samples (n = 5).
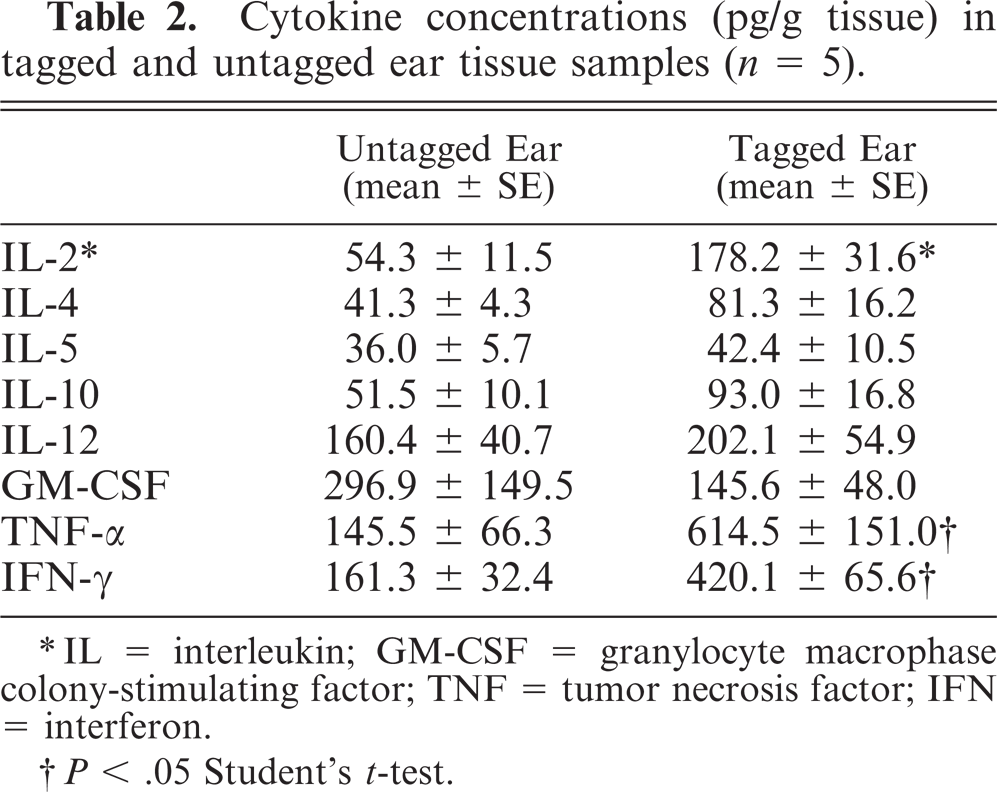
∗IL = interleukin; GM-CSF = granylocyte macrophase colony-stimulating factor; TNF = tumor necrosis factor; IFN = interferon.
†P < .05 Student's t-test.
Histopathological findings
Although auricular chondritis were seen in tagged mice with ear thickness, no typical features of the lesions were found in those without ear deformity. Histopathologic changes consisted of various stages and were observed only in the tagged ears. The lesions were characterized mainly by destruction of the normal cartilage plate with granulomatous inflammation consisting of fibrous histiocytic reaction and infiltration of mononuclear cells (Fig. 4). Newly formed proliferating cartilage nodules, occasional multinuclear giant cells, and osseous metaplasia developed around the destroyed cartilage (Fig. 5). Increased numbers of infiltrating mast cells at the area of destructed and newly formed cartilage and in the upper dermis were detected (Fig. 6). Gram and periodic acid-Schiff stains were negative for microorganisms, and no abnormality related to auricular chondritis was observed in other cartilaginous organs (data not shown).
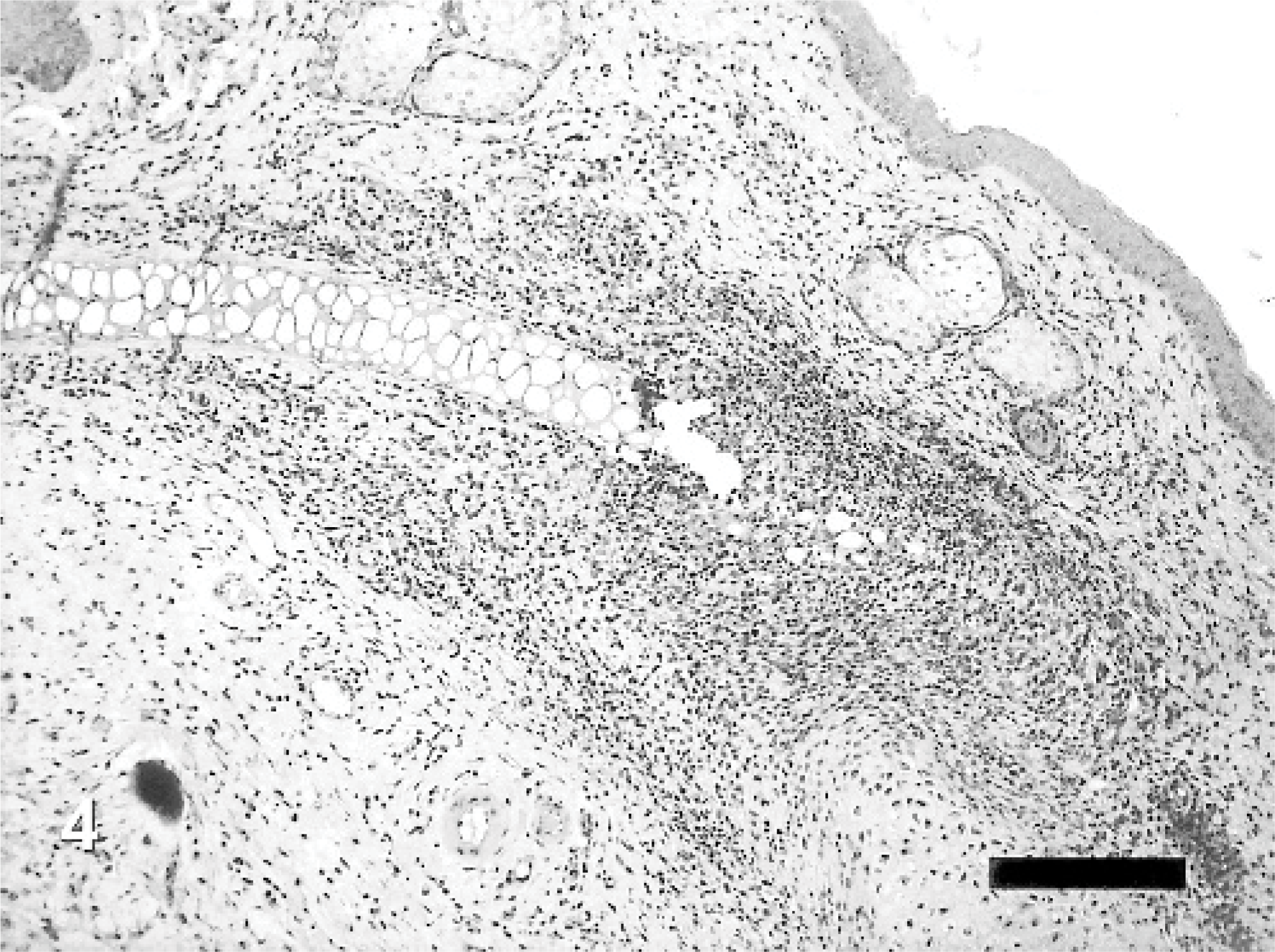
Auricular chondritis in a tagged ear; mouse No. 1. The lesion consists of focal granulomatous inflammation with mononuclear cell infiltrate and destruction of the normal cartilage plate. HE. Bar = 200 μm.
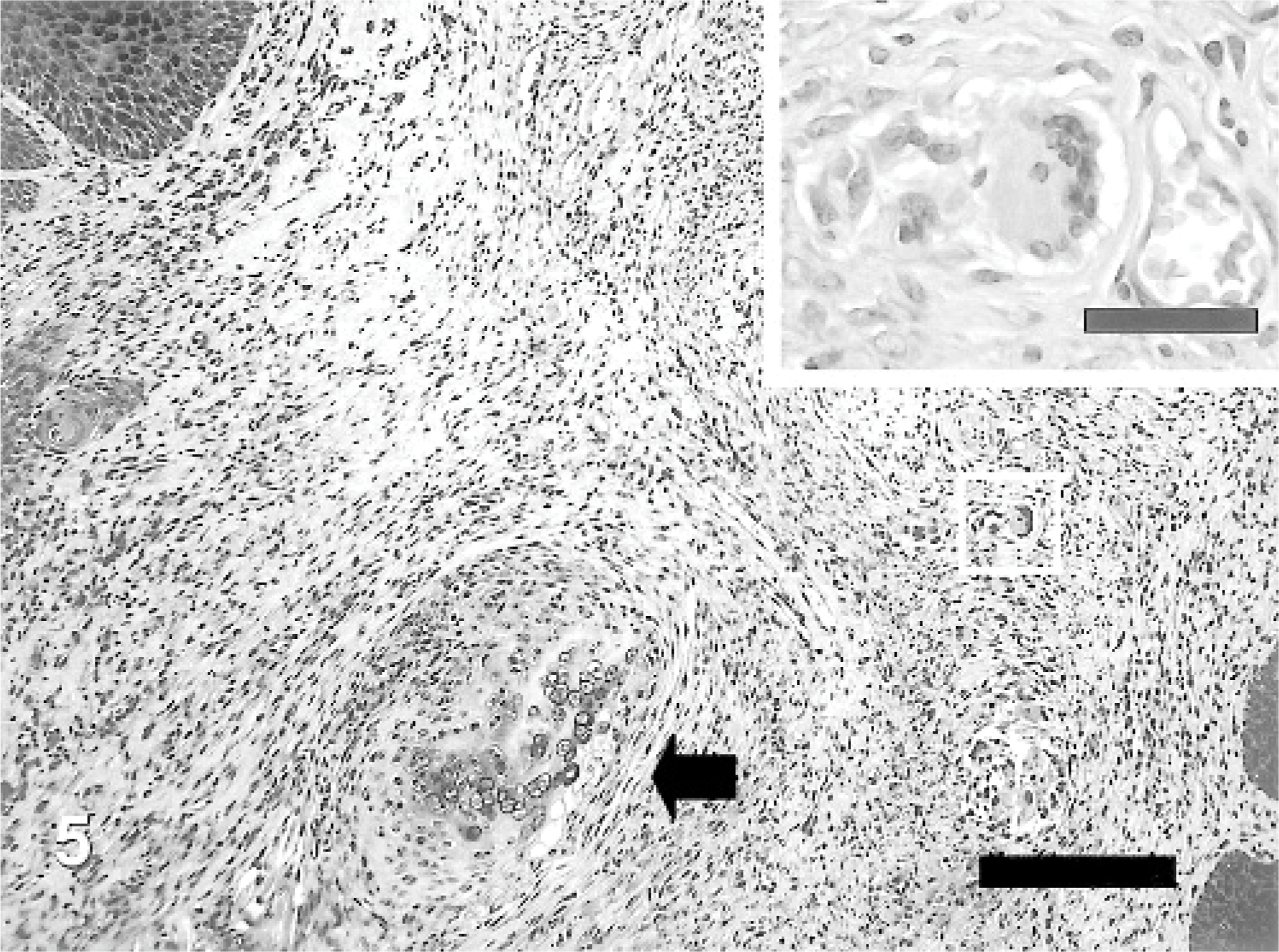
Auricular chondritis in a tagged ear; mouse No. 2. Newly formed cartilaginous nodules (arrow) around the destroyed cartilage plates with multinucleated giant cells. HE. Bar = 100 μm. Inset shows a higher magnification of a portion of a multinucleated giant cell. HE. Bar = 30 μm.
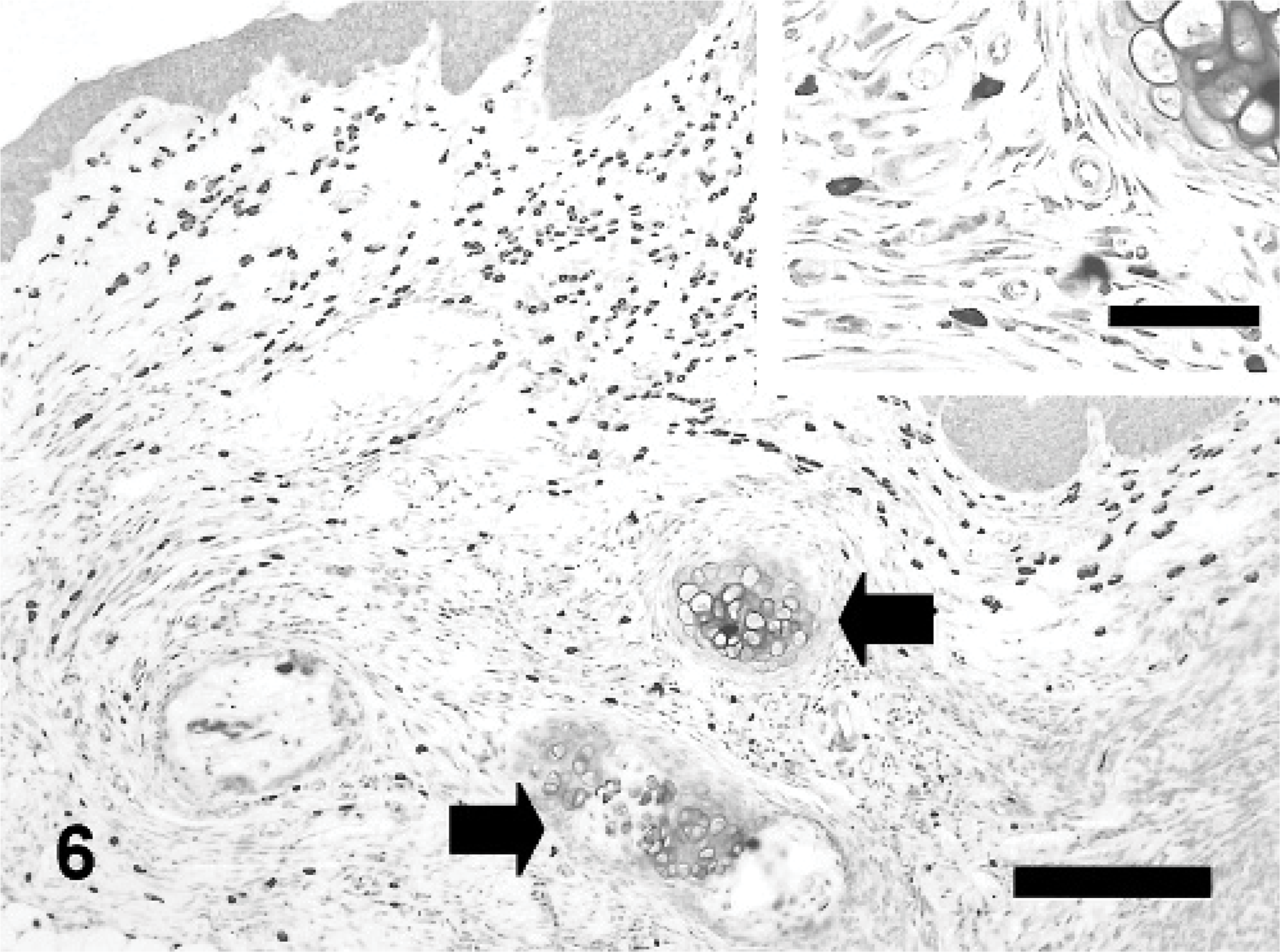
Auricular chondritis in a tagged ear; mouse No. 3. Aggregates of mast cells in the area of destroyed and newly formed cartilage (arrows). Toluidine blue. Bar = 200 μm.
Immunohistochemical examination
Immunohistochemical localization of MT in the tagged and untagged ears was performed. In tagged ears, MT-positive stained cells were found mainly in inflammatory cells, such as fibroblasts, neutrophils, and macrophages around granulomatous areas (focal or multifocal) in the dermis (Fig. 7A). In addition, chondrocytes around the destroyed cartilage plates and the basal cells of epidermis also showed immunopositivity for MT staining. MT-positive reactions in these cells were detected not only in the cytoplasm but also in the nucleus. On the other hand, no activity of MT was found in the areas surrounding the newly formed cartilage nodules. In the untagged ears, expression of MT-I and MT-II mRNA was very low, and immunostaining for MT was barely visible (data not shown). PCNA-positive cells were frequently observed in the nuclei of the fibrocytes found in the areas with destroyed cartilage and nodules (Fig. 7B). Extensive infiltration of predominantly CD4-positive and Mac-1−positive mononuclear cells were found with weakly staining CD8-positive and B220-positive cells in areas with granulomatous inflammation (Fig. 7C–F).
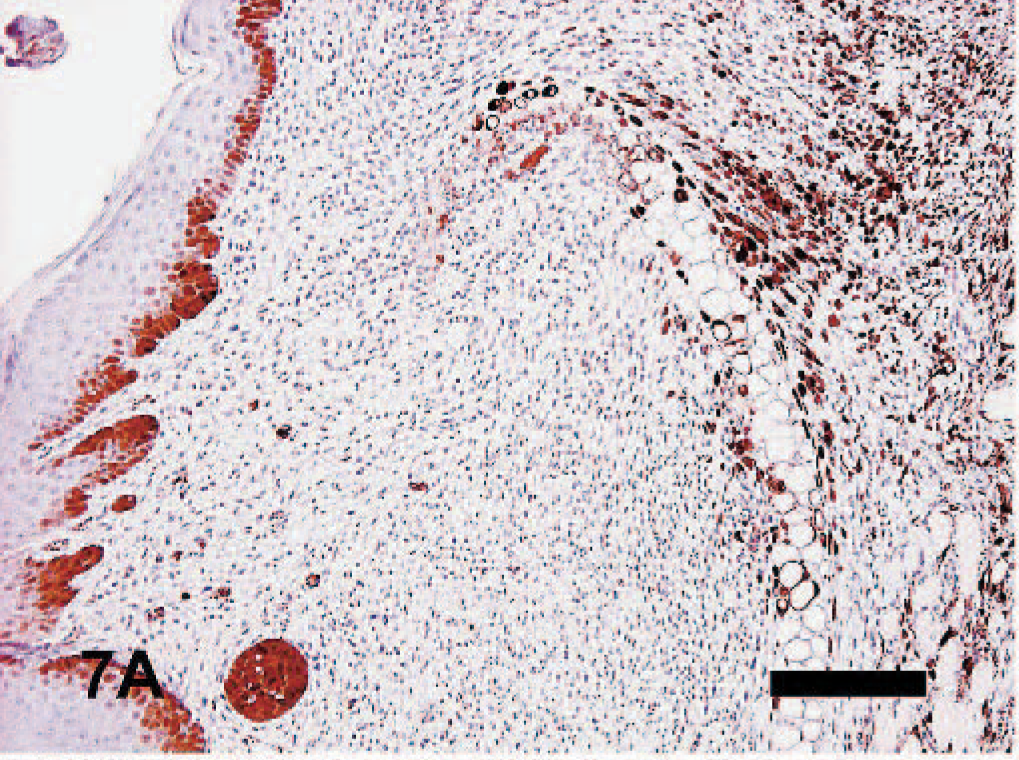
Auricular chondritis in a tagged ear; mouse No. 9. MT-I− and MT-II− positive cells in areas with granulomatous inflammation and around the destroyed cartilage plate (arrow). Immunohistochemistry for MT-I and MT-II with hematoxylin counterstaining. Bar = 200 μm.
Discussion
Spontaneous and experimentally induced auricular chondritis have been reported in rats. 4 The auricular lesions with accompanying arthritis induced in rats and mice by immunization with type-II collagen is a well-known animal model for RP in humans. 10, 20 Although the pathogenesis remains unclear, the auricular lesions were limited to the pinnae in spontaneous cases. 4 Ear lesions associated with metallic ear tags have been described in Wistar [Crl:(WI)BR] rats and FVB/N mice used in long-term studies. 2, 36 These findings differ clearly from our observations because the chronic, proliferative inflammatory reactions and tumor development were seen only in the tagged ears, leaving the untagged ear lesion free. In this study, we observed inflammatory nodular thickening in ears tagged with metal in aged C57BL/6 male mice. The clinical and histopathologic features of the chronic inflammation observed in mice with metal ear tags resembled those reported for induced or spontaneous auricular changes in rats. 4, 20 This spontaneous auricular disease has not been described in other laboratory animal species, although inflammation as an extension of dermatitis may involve the auricular cartilage. 4 Although we observed bilateral (tagged and untagged) inflammatory nodular thickening of the ear pinnae in aging male C57BL/6 mice (approximately 60 weeks after tagging) in preliminary study, lesions were more marked in ears with metal ear tags (unpublished data), suggesting auricular chondritis may be induced not only in the rat but also in mice. Ear thickening induced by traumatic insult at the time of tag insertion to the auricular cartilage was transitory and eventually healed without complications. 18 Because other methods of identification, such as the nylon ear tag, notch, and punch, and traumatic insults to the pinnal cartilage do not cause similar lesions, 17, 21 the auricular changes may be a specific reaction to metallic ear tags.
In this study, we hypothesized that the spontaneous auricular chondritis was caused by metal ions released from metallic ear tags. The analysis of metal ion content showed significantly higher concentrations of copper and iron, components of the metal ear tag, in tagged ears compared with untagged ears. The most remarkable finding from the reverse transcriptase polymerase chain reaction analysis was the up-regulation of MT-I and MT-II mRNA in affected ear tissues, in accordance with the observed increase in the concentration metal ions. Moreover, immunohistochemical staining also revealed MT-positive cells in the early stages of auricular chondritis. Generally, MT expression is induced by many heavy metal ions, and the protective role of MT against copper and iron toxicity is well established. 1, 9, 13, 26 Iron and copper ions are known as suppliers of reactive oxygen species that induce inflammation and fibrosis in the skin. 28, 35 Oxidation of cartilage collagen can cause destruction by altering the properties of collagen fibrils, thereby making them more brittle and prone to mechanical fatigue failure. 33 Thus, reactive oxygen species derived from heavy metal ions released from metal ear tags may play a key role in onset of auricular chondritis associated with the use of metal ear tags for identification purposes.
Many investigators reported about the skin sensitization potential of metal ions in laboratory animals. 11 Some metals are well-known contact allergens capable of inducing a delayed-type hypersensitivity response in susceptible individuals upon prolonged direct exposure. 7, 12, 32 It has been reported that a positive reaction for copper (copper chloride), not iron, can be detected in local lymph node assay in skin sensitization tests. 16, 24 In this study, there were significant increases of copper concentration in tagged ear tissue compared with untagged ears. In addition, immunohistochemical studies revealed that CD4-positive T lymphocytes were more prominent rather than CD8-positive T lymphocytes in inflammatory areas. Furthermore, expression of Th1-type cytokines, such as IFN-γ, IL-2, and TNF-α, were significantly increased in tagged ears. From these observations, delayed-type allergic contact dermatitis against metal ions, such as copper, was thought to lead to auricular chondritis in tagged mice. However, it is difficult to substantiate that the auricular chondritis was only caused by allergic contact dermatitis, because the lesions were observed frequently on both the tagged and untagged ears as the animal aged. Moreover, the lesion was ear specific, with no other cartilaginous organs affected. Thus, it is probable that auricular chondritis caused by ear tagging is induced by autoimmune disease triggered by the presence of metal ions.
Histopathologically, granulomatous inflammation with destruction of the normal cartilage plate with massive infiltration of inflammatory cells, particularly mast cells, was observed. Macrophages, fibroblastic synoviocytes, lymphocytes, and neutrophils are usually recognized as important contributors to rheumatoid arthritis (RA) joint pathology. However, many recent studies indicate that mechanisms wherein mast cells play a part in the pathogenesis of RA involve secretion of potent proinflammatory cytokines, such as TNF-α, which have been implicated in the pathogenesis of arthritis, as well as secretion of other mediators of inflammation and tissue destruction, such as matrix metalloproteinases. 31, 37 Mast cells may also contribute to the activation of macrophages through the production of IFN-γ. 23 Moreover, it was reported that CD4-positive memory T cells were biased toward Th1 cells in RA synovium and peripheral blood. 22
Although it remains unclear how CD4-positive T cells are generated and activated, CD4-positive T cells seem to be the prime mediators of RA. 8, 25 In our data, activation and recruitment of infiltrated cells, such as CD4-positive T lymphocytes, Mac-1-positive macrophages, PCNA-positive fibroblasts, neutrophils, and mast cells, and increased cytokine production, such as TNF-α and IFN-γ, were observed in the tagged ears. Although there are no previous reports on spontaneous auricular chondritis, our immunohistochemical and cytokine data resemble those of other reports on experimentally induced auricular changes in animal models of RP and RA as autoimmune diseases. 3, 14, 27, 38 These findings suggest that auricular chondritis in mice with metallic ear tags is provoked by an autoimmune response, such as RP and RA. The degeneration of epidermal or dermal proteins by reactive oxygen species derived from copper and iron ions released from the metal ear tags may be sources of autoantigen. Reactive oxygen species have also been suggested to play a key role in activation of mast cells by heavy metals. 29
In conclusion, although the results of the present study did not completely elucidate the etiology of auricular chondritis in mice with metal ear tags, it is reasonable to speculate that its onset is caused by metal ions released from the metal tag and that subsequently autoimmune processes led to cellular interactions of CD4 T cells, macrophages, fibroblasts, and mast cells, mediated by cytokines, such as TNF-α and INF-γ. This phenomenon observed in mice with no immunologic induction may be a useful animal model for studying the autoimmunity of RP and other inflammatory autoimmune diseases in humans.
Footnotes
Acknowledgements
The authors thank Masataka Morita and the animal care technicians for the breeding and care of the mice, Naoko Takahashi for skillful technical assistance, and Daisuke Yoshizawa and Kenji Kanda for giving us the opportunity to use an X-ray fluorescence spectrometer. Finally, we are also indebted to Dr. Ignacia S. Braga-Tanaka III for her review and helpful comments.
