Abstract
An 8-month-old PML/RARα knock-in female mouse developed a promyelocytic-like myeloid leukemia with an expected latency. At necropsy, besides the typical findings associated with myeloid leukemia, a severe unilateral hydronephrosis was observed. By histopathologic examination, 2 polypoid adenomas arising from the transitional epithelium of the renal pelvis and ureter were detected. The epithelial cells of the polypoid adenomas showed accumulation of hyaline eosinophilic material within the cytoplasm. Large amounts of extracellular eosinophilic crystals were also associated with the transitional cell adenomas. Immunohistochemical analysis revealed that the eosinophilic intracytoplasmic material and the extracellular eosinophilic crystals were composed of Ym proteins. A unilateral hyaline droplet tubular nephropathy was associated with the myeloid leukemia. Expression of Ym proteins characterized both the neoplastic myeloid infiltrates and the tubular hyaline droplets. In the present PML/RARα knock-in female mouse, the accumulation of Ym proteins associated with the myeloid leukemia and with the polypoid adenomas of the transitional epithelium underlies 2 distinct pathogenetic mechanisms.
The proteins of the Ym family are encoded by a group of closely clustered genes identified on mouse chromosome 3. At the mRNA level, 4 isoforms of the Ym family have been identified: Ym1, Ym2, Ym3, and Ym4. 7 The Ym1, Ym2, and Ym3 isoforms share a 95% nucleotide sequence homology among them, whereas Ym4 shares 85% identity and has been shown to represent a pseudogene. 7 These proteins are novel mammalian lectins 1 sharing similarities with chitinases from lower organisms. The tissue-specific constitutive expression of Ym1 and Ym2 isoforms in the murine tissues has been investigated at the mRNA and protein level. The Ym1 isoform is expressed by myeloid cells and macrophages in the bone marrow and in the spleen and by pulmonary macrophages, 6, 9, 10 while the Ym2 isoform is expressed in the squamous epithelium of the forestomach. 9, 10 Overproduction and accumulation of Ym chitinase-like lectins have been identified in different pathologic conditions in the mouse. Ym chitinase-like lectins are implicated in the genesis of eosinophilic crystals described in the lungs in association with the so-called Acidophilic Macrophage Pneumonia or with other types of inflammation in both wild-type and genetically engineered mice. 3, 5, 10 Expression and extracellular crystallization of Ym chitinase-like lectins have also been identified in hyperplasia associated with epithelial hyaline change in the glandular stomach, 10 in hyperplastic conditions associated with inflammation of the bile ducts, 5, 10 and in subcutaneous abscesses in the p47 phox -deficient mouse model of chronic granulomatous disease. 5 Furthermore, the overproduction of Ym chitinase-like lectins by neoplastic myeloid cells and the role of these proteins in the genesis of a paraneoplastic hyaline droplet and crystalline nephropathy have been described in murine models of acute myeloid leukemia. 8
An 8-month-old C57BL/6 × 129/SvJ PML/RARα knock-in female mouse 11 developed a disseminated acute myeloid leukemia with an expected latency. At necropsy, a typical splenomegaly was detected and an unexpected severe unilateral hydronephrosis was also evident. The tissues of the mouse were collected and fixed in 10% neutral-buffered formalin and processed for histologic evaluation with hematoxylin and eosin. Additional serial sections of the spleen and of both kidneys were submitted for immunohistochemical analysis using a rabbit polyclonal antibody recognizing both Ym1 and Ym2 proteins 10 and a commercially available rabbit polyclonal antibody anti-human lysozyme (EC 3.2.1.17 DAKO, Glostrup, Denmark). Histopathologic examination confirmed the presence of a disseminated myeloid leukemia diffusely involving the spleen and the bone marrow and infiltrating the liver, the lungs, the kidneys, and the lymph nodes. In all tissues examined, the infiltrates of neoplastic myeloid cells were characterized by the accumulation of abundant amounts of brightly eosinophilic crystalline material both in extracellular location and within the cytoplasm of large macrophage-like cells (Fig. 1). Histopathologic examination of the hydronephrotic kidney showed an extensive infiltration of neoplastic myeloid cells in close proximity to the renal pelvis encroaching upon the uretero-pelvic junction. Moreover, both the pelvis and the ureter showed the presence of locally extensive polypoid adenomas arising from the transitional epithelium (Fig. 2). The polypoid adenomas were organized in exophytic masses composed of nests and tubules of moderately differentiated transitional epithelial cells within a dense fibrous stroma. Scattered groups and nests of more undifferentiated transitional epithelial cells were detectable in some areas of the proliferative lesions. Moderate numbers of lymphocytes and neutrophils as well as some immature myeloid cells were detectable within the stroma. The transitional cells of the polypoid adenomas showed the accumulation of coarsely granular or homogeneous hyaline eosinophilic material within the cytoplasm. Needle-shaped and splinter-shaped eosinophilic crystals were also accumulated in extracellular location associated with the transitional epithelial cells of the polypoid adenomas (Fig. 3). The marked hydronephrosis observed macroscopically was secondary to the obstruction of the lumen of the pelvis and ureter caused by the proliferative lesions. In the contralateral kidney, mild interstitial infiltration of neoplastic myeloid cells and a moderate unilateral hyaline droplet tubular nephropathy were detectable. Immunohistochemistry for Ym1/Ym2 proteins showed variable moderate to intense specific reactivity of the eosinophilic crystals associated with the neoplastic myeloid cells and the transitional epithelium (Fig. 4). Most neoplastic myeloid cells in the leukemic infiltrates displayed a granular cytoplasmic immunoreactivity for Ym1/Ym2 proteins (Fig. 5) but were negative for lysozyme. Moreover, hyaline droplets within the cytoplasm of cortical tubular epithelium of the kidney with hyaline droplet nephropathy were also immunoreactive for Ym1/Ym2 proteins (Fig. 5). Of interest, tubular hyaline droplets were also immunoreactive for lysozyme. Neither epithelial hyaline change nor accumulation of extracellular eosinophilic crystals was detectable in the lungs, glandular stomach, or bile ducts of this mouse.
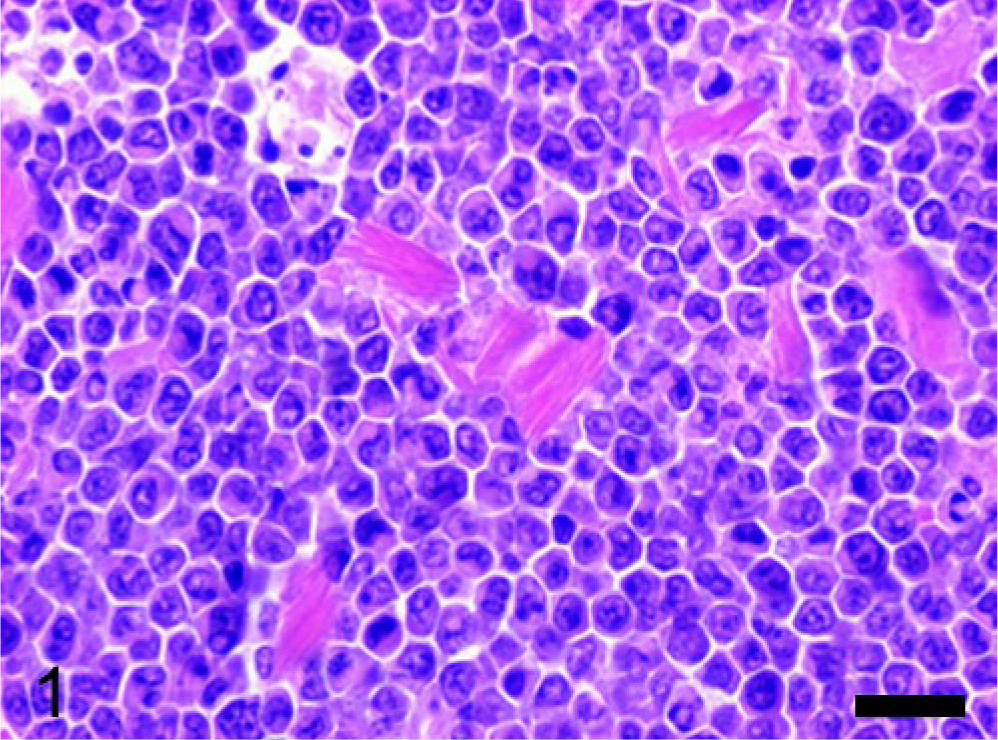
Spleen; mouse. Diffuse infiltration of neoplastic myeloid cells associated with accumulations of eosinophilic crystals. HE stain. Bar = 25 μm.
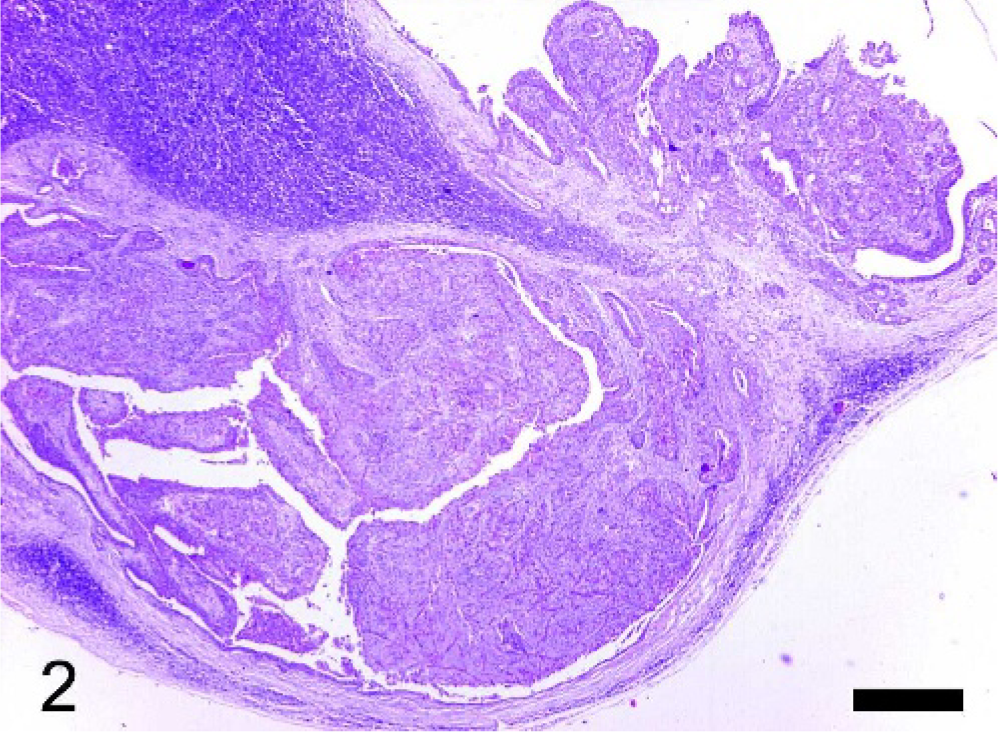
Kidney; mouse. Exophytic polypoid adenomas arising from the transitional epithelium of the renal pelvis (upper right) and ureter (lower left). HE stain. Bar = 570 μm.
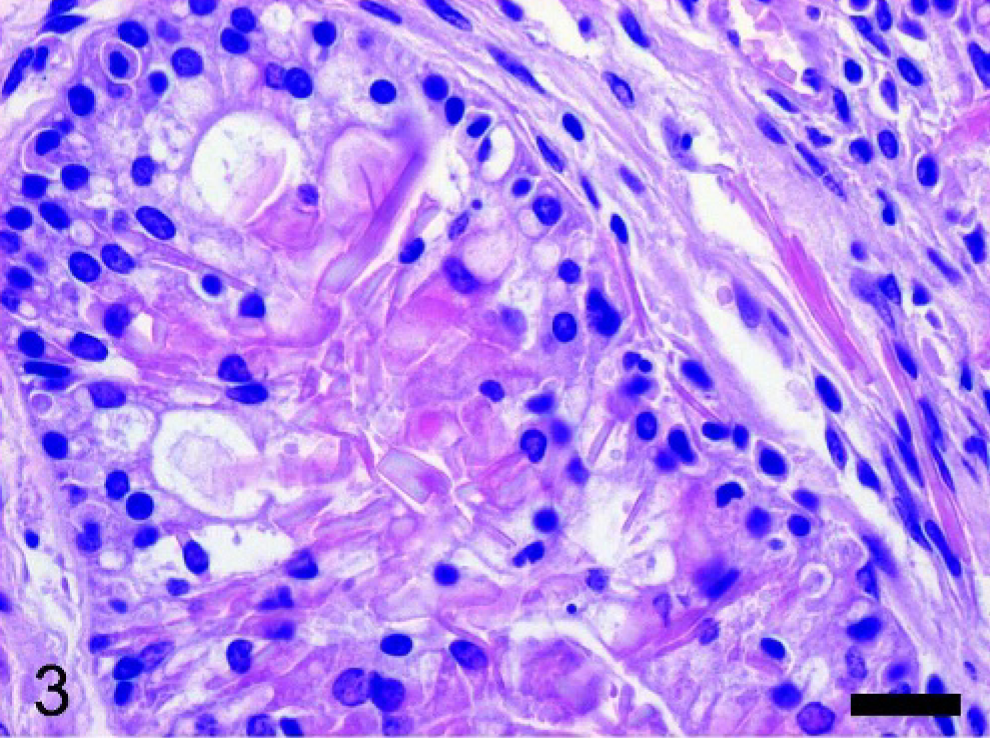
Kidney; mouse. Prominent accumulation of hyaline eosinophilic crystalline material associated with the transitional epithelial cells of the polypoid adenomas. HE stain. Bar = 25 μm.
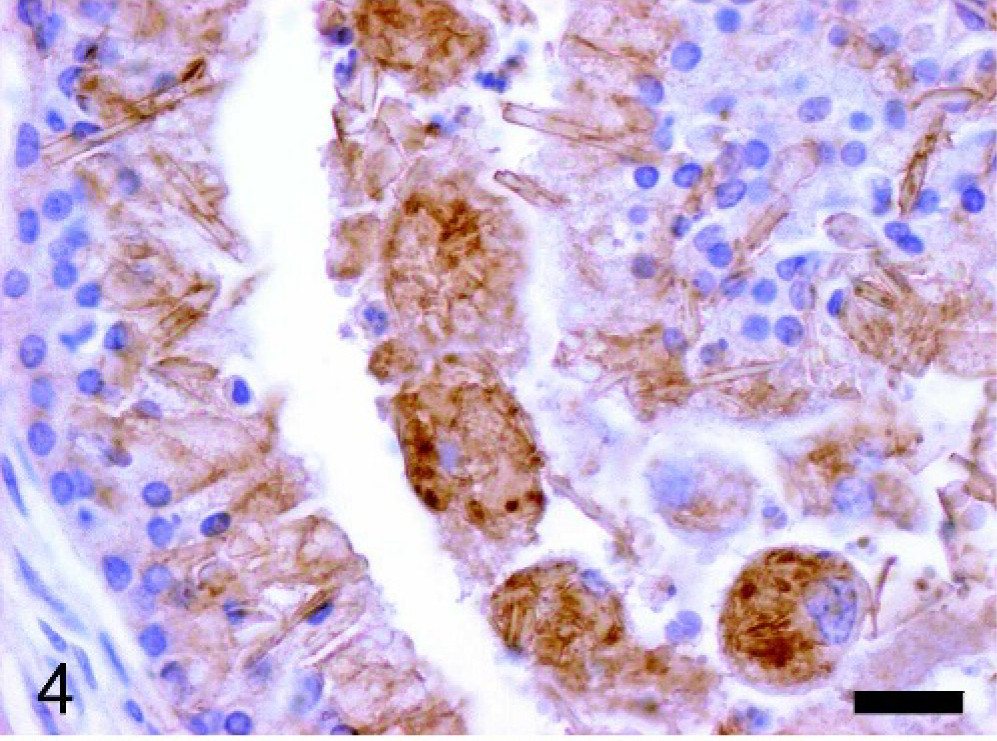
Renal pelvis; mouse. The extracellular crystals associated with the transitional epithelial cells of the polypoid adenomas display consistent immunoreactivity for Ym1/Ym2 proteins. Ym1/Ym2 immunohistochemistry, Mayer's hematoxylin counterstain. Bar = 25 μm.
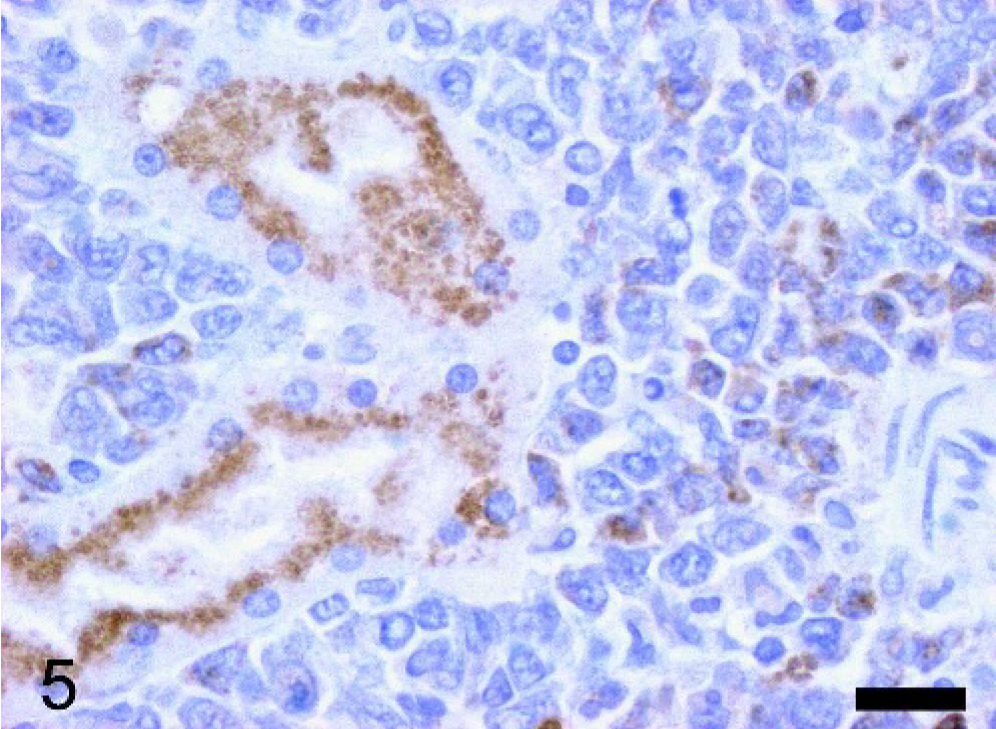
Kidney; mouse. In the contralateral kidney, ← droplets within the cytoplasm of tubular epithelial cells are immunoreactive for Ym1/Ym2 proteins. Moderate granular cytoplasmic immunoreactivity for Ym1/Ym2 proteins is evident in neoplastic myeloid cells infiltrating the interstitium. Ym1/Ym2 immunohistochemistry, Mayer's hematoxylin counterstain. Bar = 25 μm.
This is the first report describing the presence of eosinophilic crystals composed of Ym proteins associated with proliferative lesions arising from the transitional epithelium of the renal pelvis and ureter. Our findings parallel the morphologic evidences described in other organs such as the glandular stomach, the bile ducts, 4 and the lungs.
The constitutive expression of the Ym proteins in pulmonary alveolar macrophages and in the squamous epithelium of the forestomach suggest a role for these lectins as surveillance/defence mechanisms in tissues that are naturally exposed to possibly harmful exogenous environmental factors. Moreover, heparin/heparan sulfate proteoglycans have been identified as the possible endogenous substrates of the Ym1 isoform, suggesting a role of these proteins in tissue remodelling associated with inflammation. 1, 6 Indeed the Ym protein-related epithelial hyaline change and accumulation of eosinophilic crystals observed in hyperplastic lesions of the glandular stomach and bile ducts are constantly associated with variable degrees of inflammation, 5, 10 although the precise relationship between these 2 findings has not been thoroughly assessed. In the PML/RARα knock-in female mouse we describe there was a mild inflammation associated with the transformed transitional epithelium, and a much more prominent infiltration of neoplastic myeloid cells was present in close proximity to the transitional cell polypoid adenomas.
The primary overexpression and accumulation of Ym proteins in the urinary tract has not been previously described. A weak expression of the Ym2 mRNA has been identified in the normal kidney, 7 but this finding was not confirmed by other investigators. 9 It seems still unclear whether epithelial cells of the kidney and lower urinary tract harbor a constitutive expression of any Ym chitinase-like lectins isoforms; nevertheless, our findings clearly demonstrated an overproduction and extracellular accumulation of these proteins associated with the transformed transitional epithelial cells. It is interesting to remember that granular eosinophilic cytoplasmic inclusions have been described in the superficial cells of the transitional epithelium of the urinary bladder of mice exposed to anthraquinone. 2 Although these eosinophilic cytoplasmic inclusions in transitional epithelial cells are thought to represent degradative products in response to injury, their biochemical characterization is still lacking. It would be interesting to investigate whether these eosinophilic cytoplasmic inclusions in transitional epithelial cells are composed of Ym chitinase-like lectins.
The Ym1 isoform has shown to be expressed by myeloid cells in hematopoietic tissues such as the bone marrow and the spleen, 6, 9 thus the overexpression and accumulation of these proteins associated with the progression of a myeloid leukemia as in our mouse is not surprising. In 1 of the 2 kidneys, a hyaline droplet tubular nephropathy was evident, and the hyaline droplets within the renal tubules were immunoreactive for Ym proteins. This finding is in agreement with our previous data that have demonstrated the role of Ym proteins in the development of a distinctive hyaline droplet and crystalline nephropathy observed in other murine models of PML/RARα-induced myeloid leukemia. 8 Tubular hyaline droplets were also immunoreactive for lysozyme. The coexpression of Ym proteins and lysozyme in renal tubular hyaline droplets has not been previously reported. The mechanism responsible for this finding is not completely clear. Neoplastic myeloid cells were negative for lysozyme, and this would exclude that the lysozyme colocalizing with Ym proteins in tubular hyaline droplets might be tumor derived. Furthermore, this result would support the diagnosis of a myeloid leukemia and exclude a differential diagnosis with histiocytic sarcoma. Since lysozyme may be found in epithelial cells of renal tubules, an up-regulated expression of lysozyme in tubular epithelium as a consequence of tubular dysfunction caused by an accumulation of proteins might be hypothesized. Indeed lysozyme-positive hyaline droplets have been described in the renal tubular epithelium of rats bearing tumors (fibrosarcoma and meningioma) that did not show primary expression of lysozyme. 12, 13
The case we describe is unique and characterized by an unusual combination of findings whose pathogenetic mechanisms cannot be easily dissected. We identified an overproduction and crystallization of Ym proteins associated both with the polypoid adenomas of the transitional epithelium and with the neoplastic myeloid cells, although the accumulation of Ym proteins underlies distinct pathogenetic meanings in the 2 different lesions.
Footnotes
Acknowledgements
We wish to express our gratitude to Dr. Shioko Kimura, Dr. Jerrold Ward, and Dr. Miriam Anver for providing the Ym1/Ym2 antibody and for performing immunohistochemical staining. We gratefully acknowledge the precious help of Dr. Enrico Radaelli for the immunohistochemical staining for lysozyme, and the technical assistance of Marco Colombo for photographic editing.
