Abstract
Discoid lupus erythematosus (DLE) is a well-known autoimmune disorder described in dogs and humans. In dogs, DLE is considered the second most common immune-mediated dermatitis and is usually localized to the nasal planum. DLE does not evolve to generalized disease, however lesions may spread to the bridge of the nose and less commonly may extend to periocular region, pinnae, distal limbs, and mucocutaneous junctions (lips, oral cavity, and genital region). A 4-year-old male Bavarian Mountain Scenthound developed a chronic, erosive, cutaneous lesion located exclusively in the perianal region without facial skin involvement. Clinical signs included erythema, depigmentation, severe alopecia, crusting, and ulceration. Histologically, the hallmarks of the changes were an interface dermatitis consisting of plasma cells, lymphocytes, neutrophils, and macrophages, hydropic degeneration of basal cells, few apoptotic cells in the basal layer, pigmentary incontinence, and a focal thickening of the basement membrane, which was characterized by linear deposition of IgG. Despite the unusual localization the lesion was diagnosed as DLE based on the characteristic histologic and immunohistologic features. Following diagnosis, corticosteroid therapy resulted in a complete resolution of perianal lesions.
Interface skin diseases are characterized by a diffuse, bandlike infiltrate of mononuclear cells in the superficial dermis that often obscures the dermoepidermal junction. Individual cells within the basal cell layer and also stratum spinosum undergo hydropic degeneration and also apoptosis. Pigmentary incontinence and thickening of the basement membrane represent other significant features of this disease complex. 11 The differential list for interface dermatitis is extensive and includes systemic and discoid lupus erythematosus (DLE), vesicular cutaneous lupus erythematosus of the Shetland Sheepdog and Collie, pemphigus erythematosus (PE), fixed drug eruption, and the interface impersonator mucocutaneous pyoderma (MP). 11
Canine DLE is a relatively benign cutaneous disease without systemic involvement. DLE lesions initially develop at the dorsal junction of nasal planum and haired skin or along the ventral or medial aspects of alar folds. Lesions may develop also periocularly and in the ear pinnae with a bilateral symmetrical pattern. 3, 10 Occasionally, lesions may be present on the oral mucosa, tongue, pinnae, trunk, distal extremities, genitals, and the perianal region. 2, 4 A discrete erosive anal disease and histologic lesions suggestive of mucocutaneous lupus erythematosus was already described in eight dogs suffering from severe dyschezia. 6 Several breed predilections for DLE (“Collie nose”) have been mentioned and sun exposure aggravates the disease in about 50% of cases. 8 In contrast to helper T-cell predominated human DLE, many plasma cells are found in the dog indicating an important role for antibodies in the pathogenesis of the canine counterpart. 8 Nevertheless, the pathogenesis of this autoimmune skin disorder including the role of B cells and plasma cells is still not fully understood. However, there are obvious differences in the underlying immune mechanisms of DLE compared to systemic lupus erythematosus (SLE). In contrast to SLE, antinuclear antibodies are normally not found in DLE. 4
A 4-year-old male Bavarian Mountain Scenthound was presented with a 1½–year history of a painful superficial erosive dermatitis of the perianal region without anal sac involvement (Fig. 1A). The dog was pretreated with Depot-Medrate (Methylprednisolon-21-acetate, Pfizer), Atopica® (Cyclosporine, Novartis, 5 mg/kg body weight once daily), Trental® (Pentoxifylline, Hoechst Marion Roussel, 10 mg/kg body weight three times daily), and several antibiotics with no clinical improvement. There were no other skin problems or oral lesions observed and reported. Furthermore, no lesions were found in the rectal mucosa by coloscopy. Though treatment was suspended, the lesions persisted. Therefore, after 10 weeks 2 biopsies were submitted and routinely processed for histopathologic examination. Sections of 4 μm were stained with hematoxylin and eosin or periodic acid–Schiff (PAS).
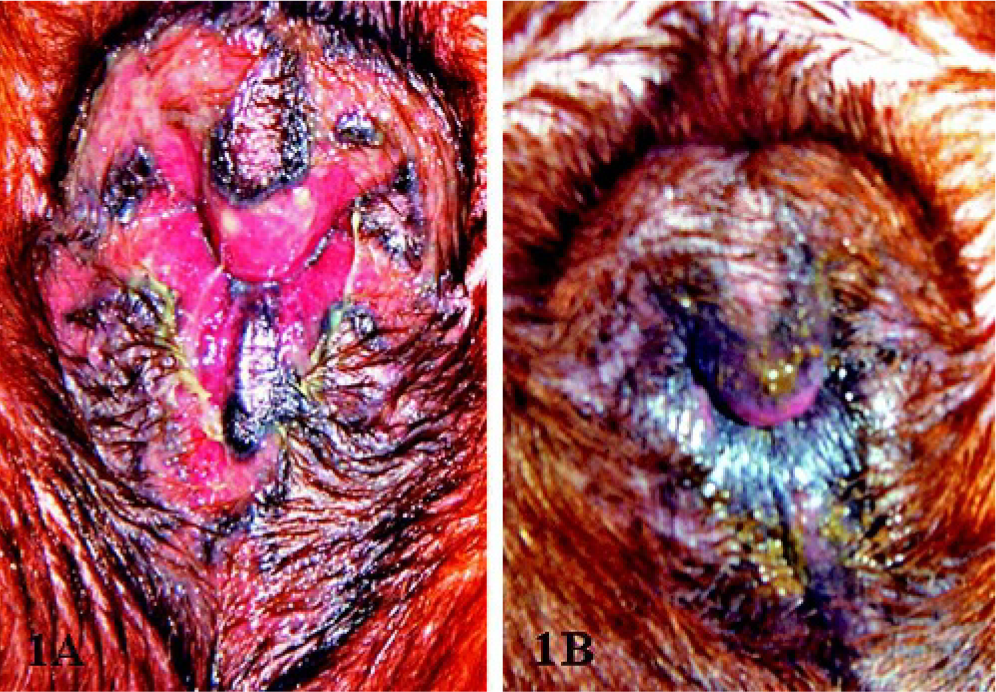
Perianal region. Bavarian Mountain Scenthound. Characteristic DLE lesions with alopecia, erythema, erosions, and crusting prior to corticosteroid therapy.
Immunohistochemical examination was carried out by a peroxidase method with 3,3-diaminobenzidine as chromogen, using antibodies directed against IgA (1 ° 8,000), IgG (1 ° 2,000), IgM (1 ° 2,000; Bethyl Lab., Montgomery, TX, USA), and C3 (1 ° 80; Nordic Immunology Lab., Tilburg, NL). 12
Histopathologic examination of the skin revealed a moderate hydropic degeneration of basal keratinocytes, individual apoptotic keratinocytes (Civatte bodies) in the basal cell layer, and a moderate pigmentary incontinence in the dermis (Fig. 2). A prominent, diffuse, partly perivascular linear band of inflammatory cells consisting of elevated numbers of plasma cells, lesser numbers of small mature lymphocytes and macrophages, and multifocal aggregates of neutrophils was noted obscuring the dermoepidermal junction (Fig. 3). There were also multifocal erosions and a substantial exocytosis of neutrophils leading to intracorneal pustules partly mixed with eosinophilic granulocytes. Deposits of PAS-positive material were detected around keratinocytes in the stratum spinosum and at the dermoepidermal junction resulting in a focal thickening of the basement membrane (Fig. 4).
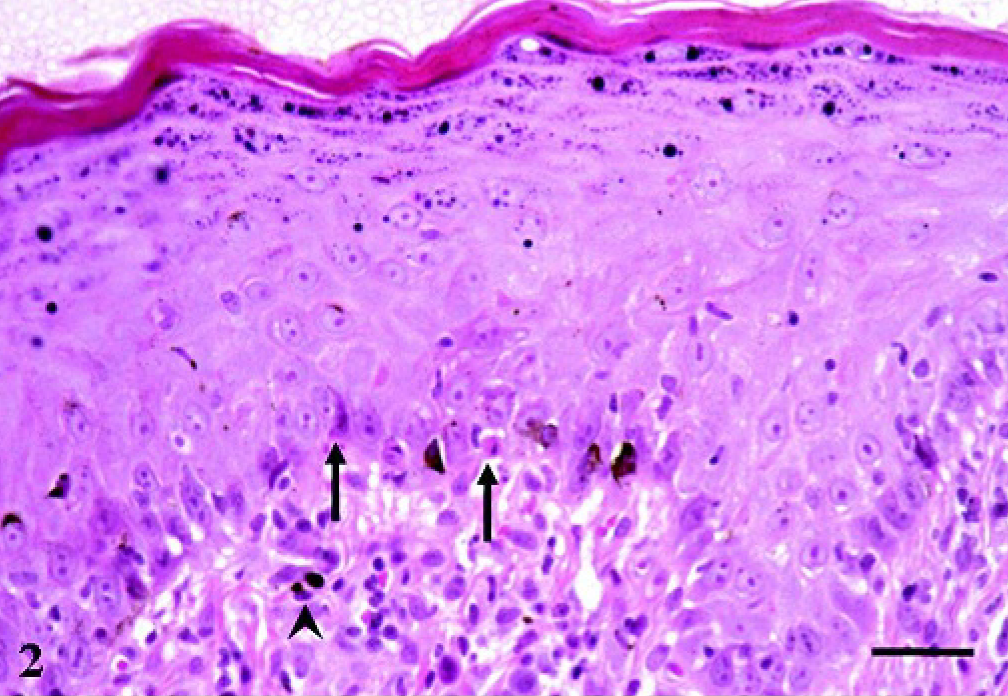
Skin biopsy, perianal region. Bavarian Mountain Scenthound. Apoptotic keratinocytes (arrow) and pigmentary incontinence in the corium (arrowhead). HE staining. Bar = 30 μm.
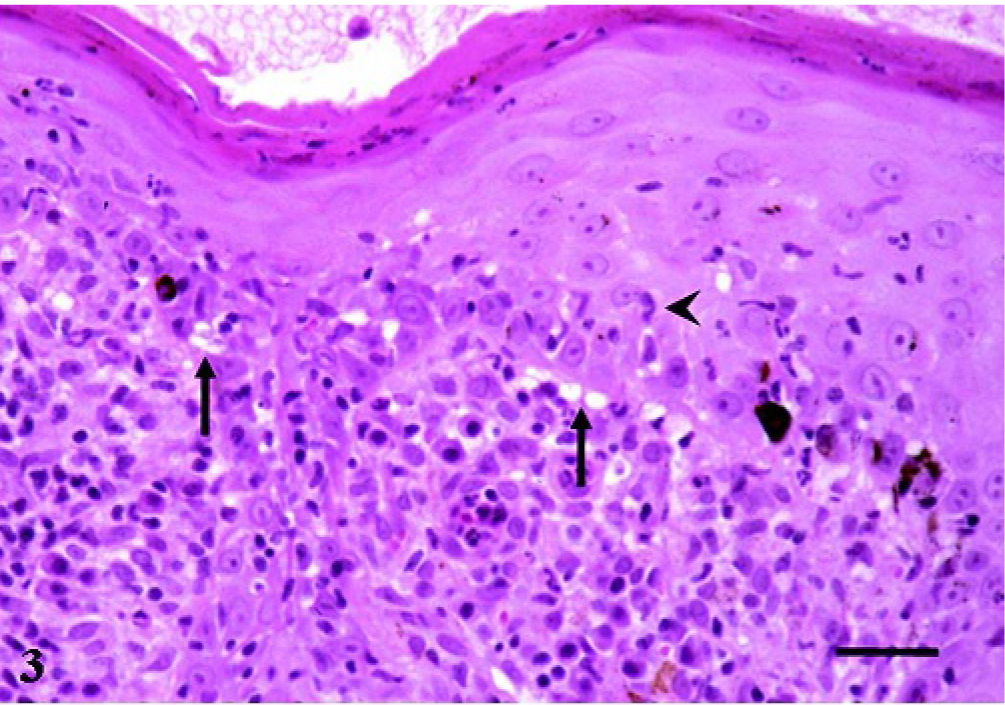
Skin biopsy, perianal region. Bavarian Mountain Scenthound. Moderate bandlike infiltration consisting of plasma cells, lymphocytes, neutrophils, and macrophages. Vacuolar degeneration of basal cells (arrows), and exocytosis of neutrophils (arrowhead). HE staining. Bar = 29 μm.
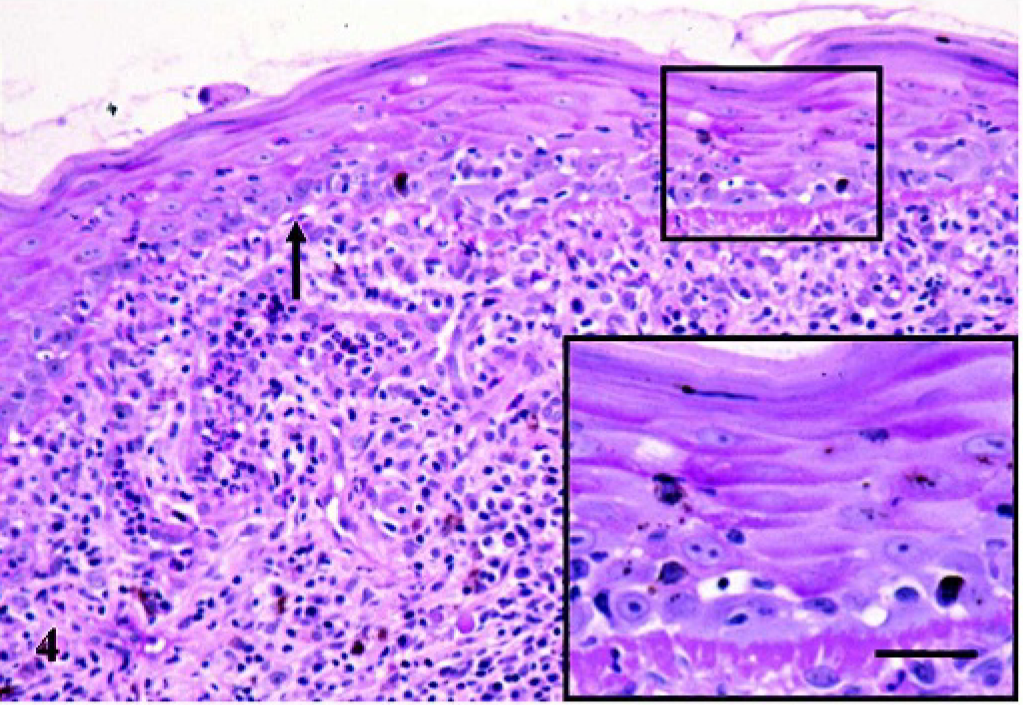
Skin biopsy, perianal region. Bavarian Mountain Scenthound. Focal, moderate thickening of the basement membrane compared to an unaltered segment (arrow). PAS staining. Bar = 48 μm. Inset: Higher magnification demonstrating thickened PAS-positive basement membrane. Bar = 23 μm.
Immunohistology revealed a focal, beehive, intercellular reaction in the epidermis and a continuous band of IgG at the dermoepidermal junction about 2–15 μm in width (Fig. 5). In addition, some segments of the basement membrane were also slightly to moderately positive for IgM and IgA immunoglobulins. Incubation with the anti-C3 antibody resulted in a slight and diffuse, presumably nonspecific immunoreactivity of the epidermis.
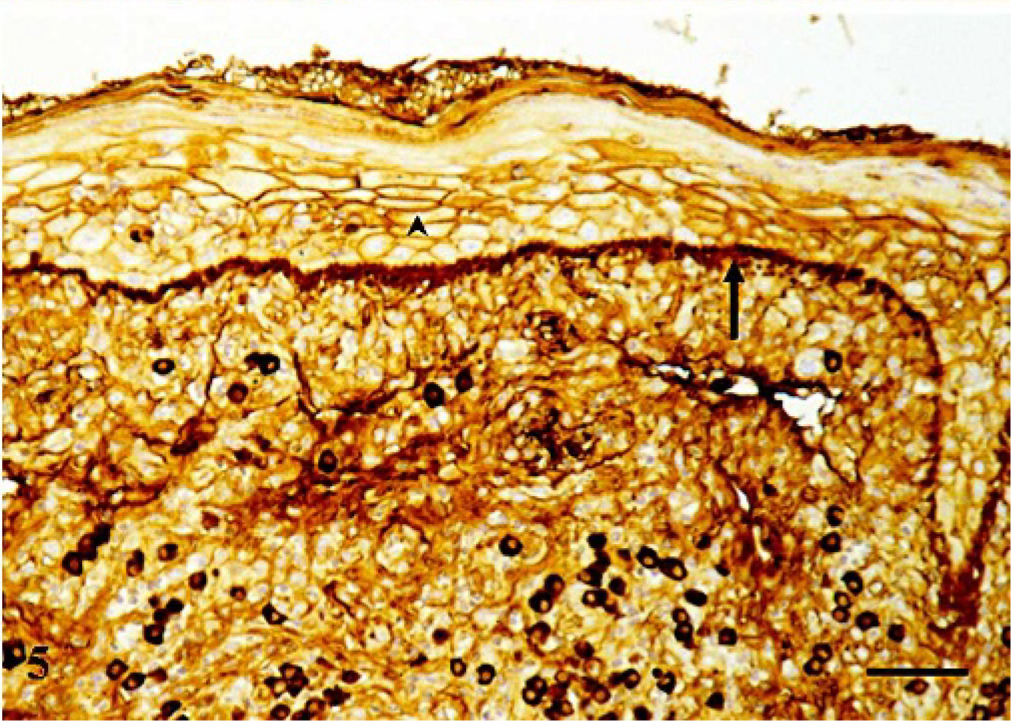
Skin biopsy, perianal region. Bavarian Mountain Scenthound. Strong immunoreaction at the dermoepidermal junction (arrow) and artifactual, beehive intercellular IgG deposits in the epidermis (arrowhead). Immunohistochemistry for IgG immunoglobulin, counterstained with hematoxylin. Bar = 48 μm.
Following the diagnosis of DLE a corticosteroid therapy consisting of Prednisolon-5 (Prednisolone-acetate, CP-Pharma, 1 mg/kg body weight once daily as an initial dose for 8 days, than tapered down slowly to 0.25 mg/kg body weight once daily as a permanent medication) was initiated. In addition, during the first 3 weeks of therapy Cefaseptin forte® (Cefalexin, Chassot, 25 mg/kg body weight twice a day) was given. After several weeks of treatment a complete recovery of the perianal skin lesions was noted (Fig. 1B).
The dog of this report showed typical features of DLE using a combination of histochemical as well as immunohistochemical methods. In particular, the typical interface dermatitis with pigmentary incontinence, vacuolated and apoptotic basal cells indicated DLE. 11 The chief differential diagnoses were fixed drug eruption, PE, and MP. 11 However, anamnesis excludes a fixed drug eruption, because no clinical improvement was seen after suspension of treatment. Besides, apoptotic cells in the stratum spinosum or satellitosis around apoptotic keratinocytes characteristic of a fixed drug eruption were absent. 11 A cleft formation inside the stratum granulosum and acantholytic cells in intraepidermal pustules characteristic for PE were lacking. 5 Furthermore, uncomplicated MP is unlikely based on the presence of significant interface damage.
Vesicular cutaneous lupus erythematosus of the Shetland Sheepdog and Collie represents another interface dermatitis, which was described so far only in the adult Shetland Sheepdog, Rough Collie, and their crossbreeds. Lesions may involve the mucocutaneous junctions including the anus. Nevertheless, typical lesions in the intertriginous areas of the groin and axillae were absent in the present case. Besides, compared to DLE the bandlike dermal infiltrate is normally less severe and the basal cell degeneration more intense in this disease. 4
In addition, lichen planus (LP) another interface dermatitis in humans, which may cause erosive or ulcerative lesions at the anogenital region, has to be considered as differential diagnosis. 9 However, the high amount of plasma cells in the present case represents a very unusual finding for erosive LP (H. H. Kreipe, personal communication) and the basement membrane should be negative for immunoglobulin deposition as well. Nonetheless, the similarity in the clinical presentation makes it tempting to speculate that this could be a case of erosive LP in a dog.
Immunohistology showed the characteristic thickening of the basement membrane owing to a deposition of IgG, IgM, and IgA immunoglobulin subclasses. 7 IgG immunoglobulin was also detected in the extracellular space between keratinocytes of the stratum spinosum, which might point towards a deposition of autoantibodies against components of intercellular adherence molecules characteristic of an autoimmune disorder of the pemphigus complex. 1, 5 However, an intraepidermal deposition of IgG immunoglobulin subclasses can also be detected in nonautoimmune lesions including pyoderma owing to nonspecific diffusion of serum antibodies into the epidermis through vascular leakage and damaged basement membrane (W. B., personal observation). As already mentioned by others, owing to possible false positive and negative results, immunohistology can never establish a final diagnosis, but only helps to strengthen a histologic diagnosis of autoimmune skin diseases. 2, 12
The facial skin is the site most commonly involved in DLE. In contrast, only the perianal region was involved in the present case, which was reported only in 1 other study. 6 The authors described 8 dogs with a histopathologic diagnosis of DLE. Clinical inspection revealed 1 or more painful discrete anal erosions, which bled easily on palpation. Histopathology was characterized by an interface dermatitis, focal hydropic degeneration of basal epidermal cells leading to a cleft formation and ulceration, apoptotic keratinocytes, focal thickening of the basement membrane, and a bandlike infiltration of the dermis with lymphocytes and plasma cells. Pigmentary incontinence was not reported. These results resembled the histopathologic features seen in the present case. Nevertheless, PAS-staining and immunohistology was not applied in the aforementioned study. In addition to oral corticosteroid therapy, other drugs were used if problems persisted, which included oxytetracycline, niacinamide, and chlorambucil. Two dogs could be taken off therapy after 6 months, 1 dog had to be euthanatized owing to aggressive behavior, and the other 5 dogs needed continuous low-dose corticosteroid therapy to be maintained in remission. In the present case low-dose corticosteroid therapy initially combined with antibiotics was sufficient to achieve remarkable results.
In conclusion, a case of a chronic, erosive, cutaneous lesion in a dog with histologic and immunohistologic features of DLE was presented. Interestingly, the facial skin was not involved, and lesions were restricted to the perianal region. Nevertheless, the cause of this particular case of autoimmune disorder remains unclear. A more thorough immunologic workup, including testing for circulating autoantibodies, and long-term clinical follow-up on additional cases of this unique dermatosis may help to determine how best to classify it.
Footnotes
Acknowledgements
We thank Prof. Dr. Hans-Heinrich Kreipe, Department of Pathology, Medizinische Hochschule Hannover, for helpful discussions and to Klaus-Peter Kuhlmann for excellent technical assistance.
