Abstract
In 2001, the first case of bovine chlamydial abortion was reported in canton Graubunden, Switzerland. In this region, Chlamydophila (Cp.) abortus is endemic in small ruminants. Hence, we aimed to investigate the incidence of chlamydia-related abortions in cattle from Graubunden. During breeding seasons of 2003-2004, formalin-fixed and paraffin-embedded placenta specimens (n = 235) from late-term abortions in cattle were analyzed by histopathology, immunohistochemistry with a Chlamydiaceae-specific monoclonal antibody against chlamydial lipopolysaccharide (LPS), and 2 different polymerase chain reaction (PCR) methods (16 S ribosomal ribonucleic acid [rRNA] PCR, intergenic spacer [IGS-S] PCR), followed by PCR product sequencing. In 149 of 235 cases (63.4%), histopathologic lesions such as purulent and/or necrotizing placentitis were observed. Chlamydial antigen was clearly demonstrated in immunohistochemistry in only 1 of 235 cases (0.4%). Cp. abortus or Cp. psittaci was found in 12 of 235 (5.1%) and 10 of 235 cases (4.2%) by 16 S rRNA PCR and IGS-S PCR, respectively. However, we detected, by 16 S rRNA PCR, 43 of 235 cases (18.3%) to be positive for chlamydia-like organisms. In contrast to the situation in small ruminants in the canton Graubunden, bovine abortion from Cp. abortus seems not to play an important role. Nevertheless, zoonotic potential should be taken into account when handling abortion material from cattle. The significance of chlamydia-like isolates other than Waddlia chondrophila remains an open question in abortion and needs further investigation.
Chlamydiae are implicated in a wide variety of clinically and economically important diseases in livestock and companion animals. 6
In cattle, several disease syndromes caused by chlamydiae infection, including abortion and other urogenital tract infections, pneumonia, conjunctivitis, enteritis, polyarthritis, encephalomyelitis, and mastitis, as well as subclinical infections, have been reported around the world. 10, 11, 17, 21, 23 Three chlamydial species that cause these conditions are known to infect cattle: Chlamydophila (Cp.) abortus, Cp. pecorum and the “chlamydia-like” Waddlia chondrophila. 13
Chlamydial abortion in cattle because of Cp. abortus occurs during the sixth to eighth months of gestation, particularly among heifers in their first pregnancy. Some cows give birth to chlamydiae-infected, weak, premature calves. Among experimentally induced abortions, placentitis is the most consistent and striking pathologic feature. 11, 17 Cp. abortus is also known to cause zoonotic infection in humans, where the greatest threat is to pregnant women and results in spontaneous abortion. 14
Cp. pecorum has been associated with conjunctivitis, encephalomyelitis, enteritis, pneumonia, and polyarthritis in cattle. 6, 13, 14 In contrast to Cp. abortus, the zoonotic potential of Cp. pecorum is unknown. 13
W. chondrophila type strain WSU 86–1044 was implicated as an abortigenic agent in 1986 when it was detected in lung, liver, and other tissues of an aborted bovine fetus in the USA. 5, 6, 12 The role of Waddlia in abortion is not clear, nor is its range of hosts; the diseases that it causes are not known either. 4
In Switzerland, 14,000 to 28,000 bovine abortion cases are reported every year, resulting in a loss of 22 to 45 million Swiss Francs per year. 9
The first case of bovine abortion in Switzerland from Cp. psittaci was reported in the canton Graubunden in 2001. 19 The first human zoonotic infection and abortion caused by Cp. abortus from caprine abortion material occurred in the same region concurrently. 18
An earlier study that examined the causative situation of sheep and goat abortions in Switzerland indicated that Cp. abortus is the leading cause of infectious abortion, inducing 39% of sheep abortions and 23% of goat abortions. 3 Seroprevalence in the Swiss sheep population was highest in Graubunden (43%) compared with 10 other Swiss cantons (2–29%). 1 In this study, we addressed whether the occurrence of chlamydia-related abortions in cattle was comparable with small ruminants aborting in the canton Graubunden.
Materials and Methods
Abortion cases
Placental tissue (1–4 cotyledons per case) of 235 randomly selected late-term cattle abortions, as well as 235 serum samples from dams were collected during the breeding seasons 2003–2004 and were submitted to the Cantonal Laboratory of Veterinary Bacteriology, Chur, Switzerland. Placental smears were prepared and stained by Koester and Stamp to determine the presence of Coxiella burnetii. Serum samples from dams were serologically investigated for the presence of antibodies against Brucella abortus and bovine herpesvirus infection (IBR-IPV). Subsequently, placenta samples were fixed in 4% formalin and embedded in paraffin.
Histopathology
Hematoxylin and eosin (HE) stained histologic sections of all placenta specimens (235) were examined for the type and the degree of placentitis and/or vasculitis. In cases, where fungal etiology was suspected, additional adjacent sections were stained by periodic acid–Schiff (PAS) and Grocott's methenamine silver.
Immunohistochemistry for chlamydiae
Paraffin sections were investigated for the presence of chlamydial antigen by using a Chlamydiaceae family–specific mouse monoclonal antibody (Ab) directed against the chlamydial lipopolysaccharide (LPS; Clone ACI-P, Progen, Heidelberg, Germany). Detection was performed with a detection kit (Dako ChemMate; Dako, Glostrup, Denmark) according to the manufacturer's instructions. Briefly, paraffin sections were deparaffinated in xylene and rehydrated through graded ethanol to water. Antigen retrieval was performed by 10-minute enzyme digestion (Pronase; Dako). To inhibit the endogenous peroxidase activity, the slides were immersed in peroxidase-blocking solution for 5 minutes at room temperature (RT) and incubated with the primary antibody diluted in Ab diluent with background-reducing components for 60 minutes at RT. The sections were incubated for 30 minutes at RT with the link-Ab, developed in 3-amino, 9 ethyl-carbazole (AEC) substrate solution for 10 minutes at RT, and counterstained with hematoxylin.
A negative control of each section was performed by using the Ab diluent instead of the primary Ab. Intestinal tissue from gnotobiotic piglets experimentally infected with porcine Chlamydia suis strain S45 was used as a positive control. 8
Deoxyribonucleic acid extraction for polymerase chain reaction screening
Sections (30–60 μm) were cut from each paraffin block and were placed in a sterile microcentrifuge tube. Paraffin was removed by extraction with 1.2 ml of xylene. After centrifugation at 13,000 ×g for 5 minutes, residual xylene was removed by twice extraction with 1.2 ml ethanol. Samples were centrifuged (13,000 ×g, 5 minutes), and ethanol was carefully removed. Deoxyribonucleic acid (DNA) for polymerase chain reaction (PCR) analysis was extracted from the tissue pellet by using a commercial DNA extraction kit (DNeasy Tissue kit; Qiagen, Hilden, Germany).
PCR detection of chlamydial DNA
Each sample was investigated for the presence of chlamydial DNA by using 2 different primer sets:
Primer set 16SIGF/16SIGR, targeting the 16 S ribosomal ribonucleic acid (rRNA) gene described by Everett and others, modified.
7
Primer set cIGS1f/cIGS2r, targeting the intergenic spacer region (IGS-S) between the 16 S and the 23 S rRNA gene.
(i) A 278-base pair (bp) fragment was amplified with the primer pair (16SIGF: 5′-GATGAGGCATGCAAGTCGAACG-3′; 16SIGR: 5′-CCAGTGTTGGCGGTCAATCTCTC-3′) complementary to a conserved region within the 16 S gene (16 S rRNA PCR).
Briefly, 1 μl of sample DNA was tested in 3.5 mM MgCl2, 0.4 mM of deoxyribonucleotides (dNTP) (Roche Diagnostics, Basel, Switzerland), 1 μM of each forward (16SIGF) and reverse (16SIGR) primer (Microsynth, Balgach, Switzerland), and 2 U DNA polymerase (AmpliTaq Gold; Applied Biosystems, Rotkreuz, Switzerland) and, with distilled water reaction, was brought to a final volume of 50 μl.
PCR cycling conditions consisted of initial denaturation (95°C, 15 minutes), followed by 45 cycles of denaturation at 94°C for 30 seconds, primer annealing at 70°C for 30 seconds, and extension at 72°C for 45 seconds, with a final extension step for 5 minutes at 72°C. The reactions were carried out on a TGRADIENT thermocycler by Biometra, Göttingen, Germany.
Identities of all positively tested 16 S PCR products were determined by direct sequencing of the 16 S rRNA gene PCR product from both strands with primer 16SIGF and 16SIGR, respectively.
(ii) A 352-bp fragment of the IGS-S rRNA gene region was amplified by using forward primer cIGS1f (5′-CAAGGTGAGGCTGATGAC-3′) and reverse primer cIGS2r (5′- TCGCCTKTCAATGCCAAG-3′).
One μl of extracted DNA was added to a final concentration of 3.5 mM MgCl2, 0.4 mM of dNTP (Roche Diagnostics), 0.25 μM of forward (cIGS1f) and 0.5 μM reverse (cIGS2r) primer (Microsynth), 2.5 U DNA polymerase (AmpliTaq Gold; Applied Biosystems), and distilled water, to a final volume of 50 μl, was added.
PCR product amplification was achieved in 40 cycles (TGRADIENT thermocycler; Biometra) with initial denaturation (94°C, 10 minutes), denaturation at 94°C for 30 seconds, annealing at 48°C for 30 seconds, and extension at 72°C for 45 seconds.
Sensitivity of the IGS-S PCR compared with the 16 S rRNA PCR was determined by spiking water with 10-fold dilutions of genomic Cp. abortus–DNA (10,000 to 0,1 template/μl). Genomic DNA of Cp. abortus strain S26/3 was prepared according to the following protocol: confluent HEp-2 cell layers were infected with Cp. abortus S26/3 multiplicity of infection (MOI) = 0.5 for 3 consecutive days. Thereafter, every third day supernatant was removed and was exchanged with fresh growth medium. Supernatant was collected 216 hours after infection and centrifuged at 250 ×g to remove remaining cell debris. Dimethyl sulfoxide (DMSO; 10%) was added to cleared supernatant, and 1-ml aliquots were stored at -80°C. For preparation of genomic chlamydial DNA, 2 ml of frozen aliquots were used. For protein digestion, supernatant was adjusted to 200 mM Tris/Cl (pH 8.0), 25 mM ethylenediamine-tetraacetic acid (EDTA; pH 8.0), 300 mM NaCl, 2% sodium dodecyl sulfate (SDS), and a final concentration of 50 mg/ml proteinase K was added, and proteins were digested overnight at 55°C. The remaining protein parts were removed by 3 repetitive phenol/chloroform extractions followed by an ethanol precipitation. We obtained, from these 2 aliquots, a total of 200 ng chlamydial genomic DNA measured by ultraviolet (UV)-spectrometry (in a Spectrophotometer DU 800, Beckmann Coulter, Fullerton, CA).
Accordingly, genomic DNA templates per volume were calculated, and DNA was used for PCR titration experiments.
Negative controls were performed by using a reaction mixture with water (H2O) instead of the template DNA.
Ten μl of each PCR product were electrophoresed in a 1.5% agarose gel, stained with ethidium bromide and observed under UV illumination. The desired fragments were excised and further purified for sequencing with the MinElute Gel Extraction Kit or MinElute PCR Purification Kit (Qiagen).
Sequencing
Sequencing was performed in collaboration with the sequencing service of the University of Zurich, with an ABI Prism 377 DNA sequencer (Applied Biosystems) or Applied Biosystems 3100 (Synergene Biotech). The obtained sequences were compared with the sequences available in GenBank by using the BLAST server from the National Center for Biotechnology Information (http://www.ncbi.nlm.nih.gov/BLAST).
Results
Abortion cases
Examinations of placental smears for Coxiella burnetii and of sera from the dams for Brucella abortus and IBR-IPV were negative in all 235 cases.
Histopathologic findings
Necrosis of trophoblastic epithelium of various degrees and/or abundant infiltration by neutrophils was present in 149 of 235 HE-stained placental specimens (63.4%). Multifocal to coalescing or diffuse, purulent to necrotizing, or necrotizing placentitis was diagnosed in these 149 cases. Most of the cases (104/149) showed both purulent and necrotizing changes, whereas 37 of 149 cases had only necrotizing placentitis, and 8 of 149 cases displayed purulent inflammation of epithelial parts without necrosis of trophoblastic epithelium. Vasculitis was diagnosed in 26 of 235 cases (11.1%). Mainly, arterioles within the intercotyledonary area displayed cellular infiltrates within their walls and consisted of neutrophilic granulocytes, as well as mild to moderate fibrinoid vessel-wall necrosis. Severe, diffuse purulent to necrotizing placentitis and vasculitis with myriad, intralesional fungal hyphae was present in 3 of 26 cases. The examination of the placental tissue was limited in 44 of 235 cases (18.7%) because of severe autolysis. In the remaining 42/235 cases (17.9%), no pathologic changes of the placenta were observed.
Immunohistochemistry
Positive antigen labeling was present in 9 of 235 cases (3.8%); among these 9 samples, only 1 placenta showed obvious multiple positive labeling (0.4%; Fig. 1). The remaining 8 cases (3.4%) displayed only single positive cells and, therefore, were judged as questionable.
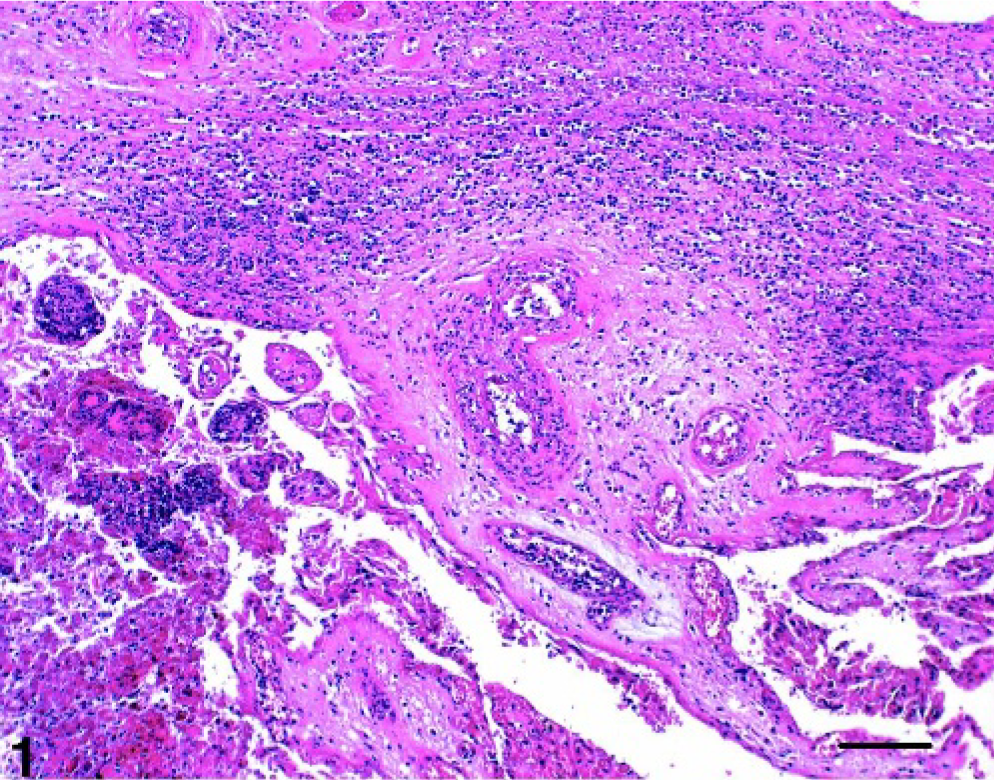
Placenta; cow No. 1. Purulent to necrotizing placentitis and vasculitis. Positive IHC and PCR for Chlamydiae. HE. Bar = 100 μm.
PCR detection
Comparison of 16 S rRNA and IGS-S PCR sensitivity
Amplification of the Cp. abortus S26/3 DNA template by using 16SIGF/16SIGR and cIGS1f/cIGS2r primer pairs produced roughly 278-bp and 352-bp fragments, respectively. The PCR sensitivity was compared in serial dilution steps (10,000 to 0.1 templates) of Cp. abortus DNA in distilled water. By using the 16 S primer set, we were able to detect as few as 1 to 10 DNA templates, whereas the detection limit of the IGS-S primer set was 10 DNA templates. The detection limit of both assays (16 S rRNA and IGS-S PCR) tested on the serial dilutions after the DNA extraction process was 10 DNA templates.
PCR detection of chlamydial DNA in placental samples
In the 16 S rRNA PCR assay, 55 of 235 cases (22.1%) were positive, 6 samples (2.55%) of these were 98 to 100% identical to Cp. abortus, and another 6 cases (2.55%) were 96 to 100% identical to Cp. psittaci (case Nos. 1–12). Forty-three cases showed 82 to 100% sequence similarity to the newly described chlamydia-like organisms. From these 43 cases, 9 cases revealed high sequence similarity to Parachlamydia acanthamoebae (91–99%), whereas, in the other 34 cases, a definitive identification was not possible (case Nos. 18–61). Thus, these isolates were referred to as chlamydia-like organisms.
By IGS-S PCR, 10 of 235 cases (4.2%) were positive for Cp. abortus or Cp. psittaci. Nine of these cases showed high similarity values (98–99%) to Cp. abortus, whereas 1 case was 99% identical to Cp. psittaci (case Nos. 1–5, 13–17).
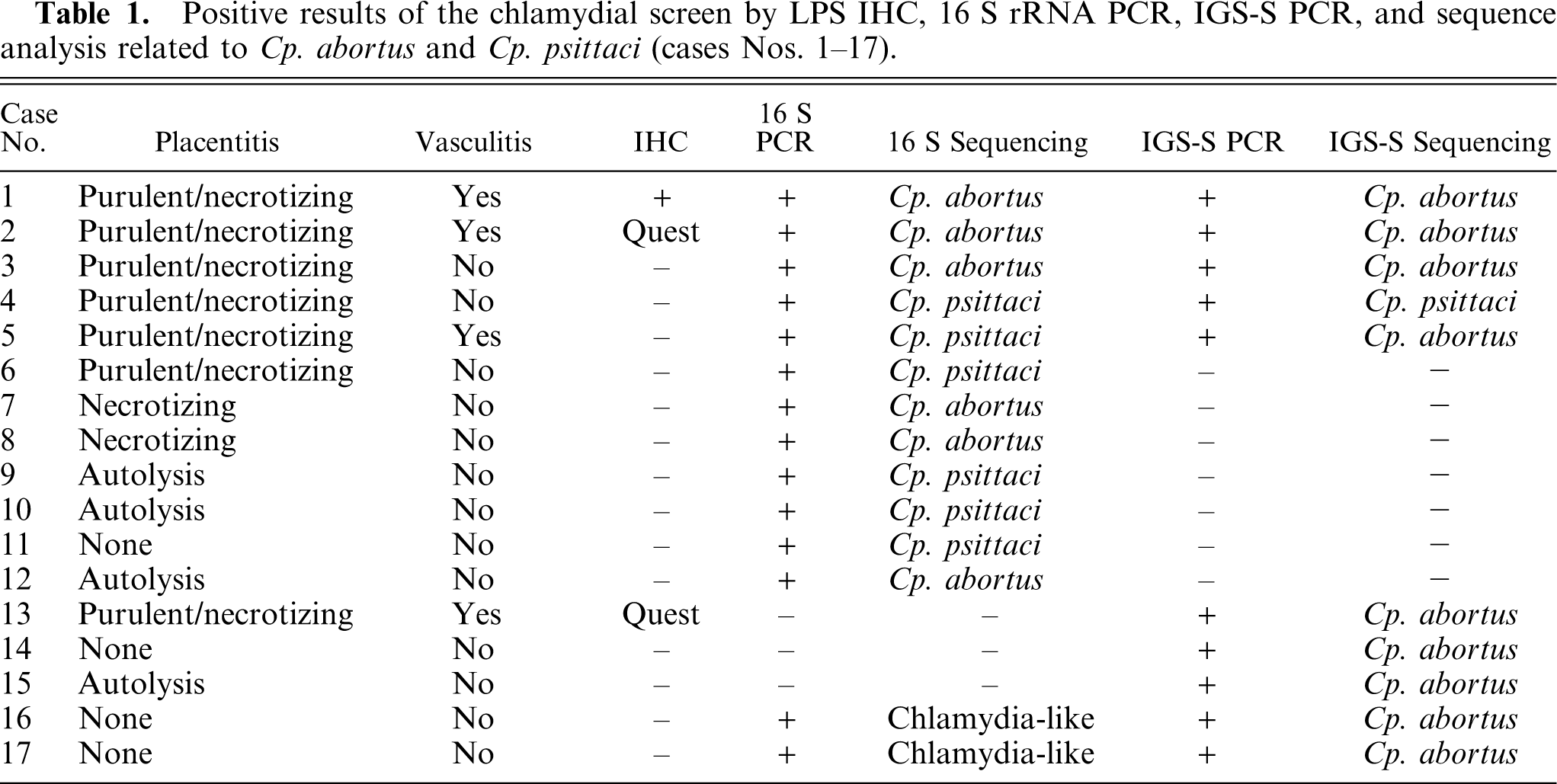
Histologic diagnosis, immunohistochemical, PCR (16 S rRNA PCR and IGS-S), and sequencing results of the cases positive for Cp. abortus and/or Cp. psittaci (cases Nos. 1–17) are displayed in Table 1.
Positive results of the chlamydial screen by LPS IHC, 16 S rRNA PCR, IGS-S PCR, and sequence analysis related to Cp. abortus and Cp. psittaci (cases Nos. 1–17).
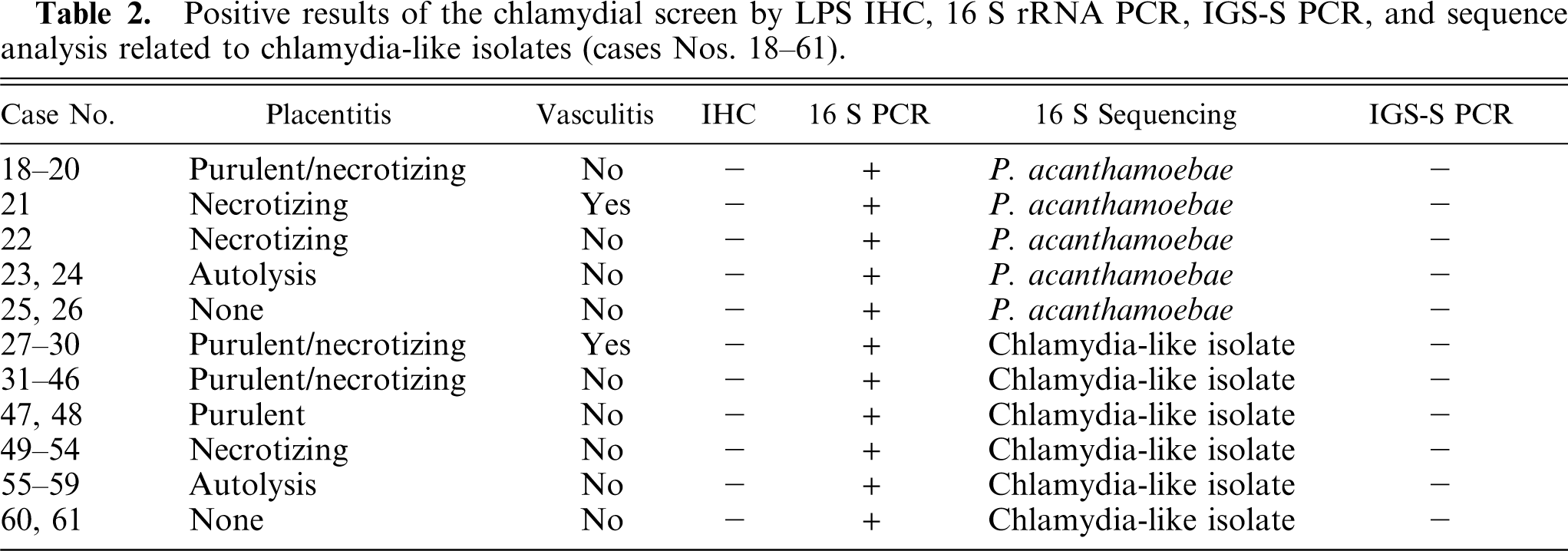
Cases Nos. 18–61 positive for chlamydia-like organisms are described in Table 2.
Positive results of the chlamydial screen by LPS IHC, 16 S rRNA PCR, IGS-S PCR, and sequence analysis related to chlamydia-like isolates (cases Nos. 18–61).
Nine of 235 cases (3.8%) displayed placental lesions and were positive by PCR for Cp. abortus or Cp. psittaci (16 S rRNA and/or IGS-S) and hence were definitively diagnosed as chlamydial abortion (cases Nos. 1–8, 13; Figs. 1 and 2).
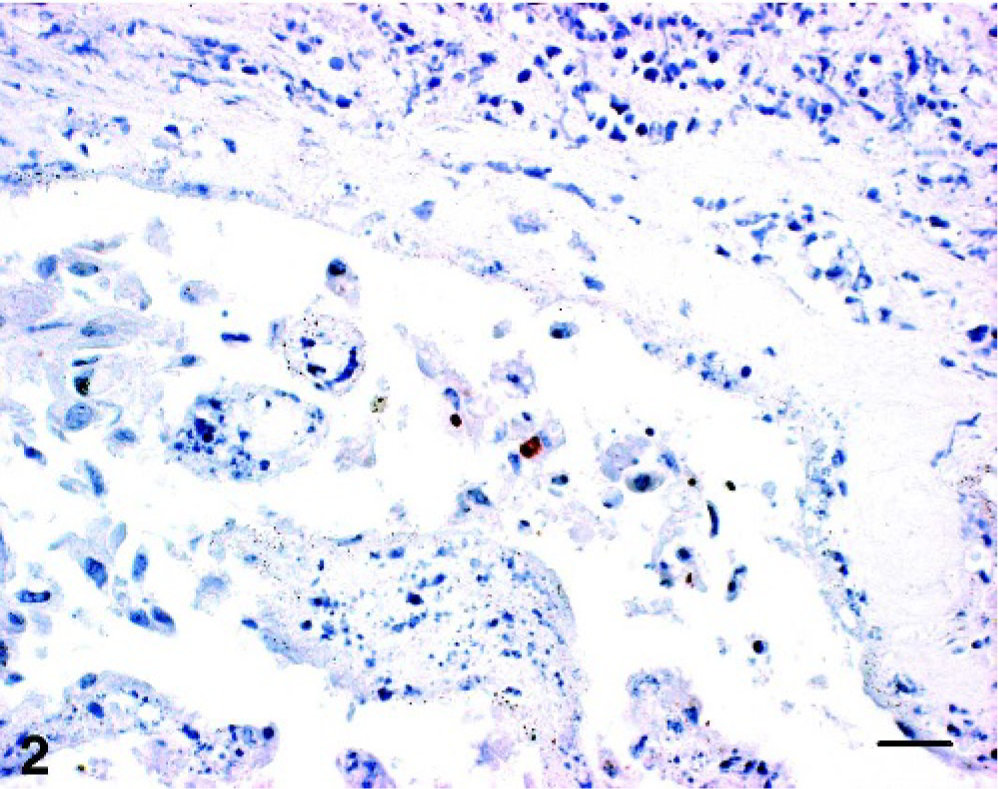
Placenta; cow No. 1. Positive granular reaction within trophoblastic epithelium. Chlamydial LPS IHC; AEC/peroxidase method, hematoxylin counterstain. Bar = 20 μm.
Discussion
Cattle abortion of unknown infectious cause still remains a major economic problem. Hence, in this study, we focussed on new or not well-defined abortigenic agents, such as on the incidence of chlamydial abortion in cattle. We concentrated our examination on placenta specimens from late-term abortions, because it was shown experimentally that chlamydiae multiply primary in the cotyledons, where they cause severe inflammation and necrosis that leads to abortion. 11, 17
The Swiss cattle population is composed of 1.58 million cattle in 65,000 registered cattle herds, from which 78,000 cattle (2,350 herds) are located in the canton Graubunden, which belongs to a mountain region of Switzerland. About 1,500 late-term abortions (more than 6 months of gestation) are reported annually in Graubunden. We selected this mountainous region of Switzerland because of its high seroprevalence in sheep (43%), suggesting this canton is endemic for chlamydial abortion. 1
In 9 of 235 cases (3.8%), placentitis was present and chlamydial antigen (IHC) and/or Cp. abortus/Cp. psittaci DNA could be found within the lesions (cases Nos. 1–8, 13). Therefore, we assumed that bovine abortion from Cp. abortus/Cp. psittaci was not of great importance in the canton Graubunden. Previous studies reported high seroprevalence of chlamydiae in cattle by complement-fixation test (CFT) or enzyme-linked immunosorbent assay (ELISA). 2, 15 These observations were partly seen because many cattle harbor chlamydiae in the intestinal tract, which stimulates a low CFT titer, leading to false-positive results. Thus, detection of seropositivity in cattle is considered an indication of chlamydial infection but not necessarily of chlamydial disease unless a rise in titer can be demonstrated.
Nevertheless, among the chlamydial-positive cases, the detected histopathologic placental lesions (purulent to necrotizing placentits, and vasculitis) in cattle were similar to those found in sheep. However, placentitis (purulent, purulent to necrotizing or necrotizing) was very often observed (149/235 cases), the combination of placentitis and vasculitis was only prevalent in 26 of 235 cases (11.1%). From these 26 cases, 3 had a fungal etiology. Regarding the etiologic agent, based on our sequence result, we found similarities to Cp. abortus but also to Cp. psittaci, which is closely related to Cp. abortus. In case No. 5, Cp. abortus was detected by IGS-S PCR, whereas 16 S rRNA PCR detected Cp. psittaci. PCR-induced mutations during the amplification process leading to sequence variation might be unlikely and did not skew the results. There were not many options for mutations because (i) we sequenced directly PCR products and hence averaged our PCR sequences and (ii) short PCR products do not leave many options for mutations. Hence, it is most likely that case No. 5 is a mixed infection of Cp. abortus and Cp. psittaci.
The natural method of spread of the infection in cattle is not known, but the disease can be reproduced by parenteral inoculation of the agent. 11, 17 In general, infectious elementary bodies can be shed in feces, nasal, ocular or vaginal discharges, uterine fluids, urine, or semen, depending on the particular syndrome. 17 In sheep flocks, the aborting ewe is shedding masses of infectious elementary bodies in uterine fluid, the aborted fetus, and the placenta, resulting in infection of other ewes. 21 It is assumed that transmission in cattle occurs similarly; however, this has not been verified. In contrast to the endemic situation of newly infected sheep and goat flocks, of which up to 60% of the animals suffer from abortion, the 7 cases found in cattle occurred in different herds. Close contact between ruminant species on farms suggests that chlamydiae might have been transmitted from infected sheep to cattle, but this could not be confirmed.
The amount of chlamydial antigen within the bovine placenta estimated in the IHC seemed to be much lower than in sheep placenta affected by ovine enzootic abortion (OEA). Similar observations were already published by McKercher 16 in 1969, who reported that, in contrast to OEA, which can be diagnosed by the demonstration of elementary bodies in smear preparations of the placentas from aborting ewes, elementary bodies are found only rarely in aborted bovine placentas and then in very low numbers. To date, the reason for this finding remains unclear, but it could be hypothesized that cattle might be less susceptible to chlamydial abortion than sheep and goats.
In previous studies, often only PCR was used to diagnose chlamydial abortion in cattle. Because it is known that many cattle harbor chlamydiae in their intestinal tract and almost all placentas collected after fetal delivery are contaminated with fecal material, a high rate of false-positive PCR results can occur. The combined methods of IHC, 16 S rRNA PCR, and IGS-S PCR used in our study enabled us to confirm that the pathogen was actually present within the typical lesions. Placental specimens with PCR assays (16 S rRNA, IGS-S) positive for chlamydial DNA but without evidence of placentitis were not definitively considered as bovine chlamydial abortion.
Overall, we concluded that a diagnosis of bovine chlamydial abortion should include the presence of placentitis and the demonstration of the etiologic agent within the lesion by PCR and/or immunohistochemistry.
Chlamydia-like organisms were detected in 43 of 235 cases (18.3%) by 16 S rRNA PCR. Failure to detect chlamydia-like organisms by IGS-S PCR is most likely because of the lower sensitivity of IGS-S PCR method compared with 16 S rRNA PCR. We tended to assume that only low genome numbers of these organisms were prevalent in our placental specimens. Sequencing of PCR products surprisingly revealed no similarities to W. chondrophila but to P. acanthamoebae. In 5 of 9 cases positive for P. acanthomoebae, placentitis could be found, in 1/9 case vasculitis was detectable. In 28 of 34 cases (82.3%) positive for chlamydia-like purulent and/or necrotizing placentitis was obvious; 4 of 28 cases displayed additional vasculitis.
Although WSU 86-1044, Simkania negevensis, and P. acanthomoebae formed a cluster, the 3 species are as far apart from one another as they are from the chlamydiae. 20 Chlamydia-like organisms could just be detected by 16 S rRNA PCR assay and not in the IHC. To date, it is known that some new species do not have a LPS at all or possess the LPS in a truncated form. Therefore, we are not able to detect them in IHC when using an antibody directed against the LPS. Production of antibodies directed against the new species and their evaluation for IHC is in process.
Footnotes
Acknowledgements
This study was kindly supported by the Federal Veterinary Office, Bern, Switzerland (project number 1. 03. 07). We are grateful to the laboratory technical staff of the Institute of Veterinary Pathology, University of Zurich and of the Cantonal Laboratory of Veterinary Bacteriology, Chur, Switzerland.
