Abstract
Intoxication by pods of Prosopis juliflora (mesquite beans) causes an impairment of cranial nerve function in cattle and goats. In goats, vacuolation of neurons in the trigeminal motor nuclei has been reported. To study the lesions in cattle caused by consumption of P. juliflora pods and dry ground pods, eight 6- to 12-month-old male cattle were divided into 4 groups: group 1 was fed a ration containing 50% of pods; groups 2 and 3 received a ration containing 50 and 75% of dry ground pods, respectively; group 4 was the control. After 200 days, all cattle were killed and sampled for histologic evaluation. Samples of the trigeminal motor nucleus were examined by electron microscopy. All cattle from groups 1, 2, and 3 showed clinical signs resulting from impaired function of cranial nerves V, IX, X, and XII, starting 45-75 days after consumption of the plant. The main histologic lesions were vacuolation and loss of neurons in trigeminal motor nuclei and other motor cranial nerve nuclei with Wallerian-like degeneration in the cranial nerves. Mild denervation atrophy was observed in the masseter and other masticatory muscles. On electron microscopy, neurons of the trigeminal nuclei had markedly swollen mitochondria, with the mitochondrial cristae displaced peripherally, disoriented and disintegrating. Intoxication by P. juliflora seems to have a novel pathogenesis, characterized by a selective, primary, chronic, and progressive injury to mitochondria of neurons of the trigeminal and other cranial nerve nuclei. Cranial nerve degeneration and denervation atrophy of the muscles occurs as a consequence of the neuronal lesion.
Keywords
Introduction
Prosopis juliflora (mesquite beans), a xerophilous tree introduced to northeastern Brazil in the 1940s, grows rapidly to a height of 8 to 12 m. Currently, it is estimated that there are approximately 150,000 hectares cultivated with P. juliflora in northeastern Brazil, mainly in the semi-arid region. Because of the combination of low costs, high palatability and nutritional value, the fruit pods (“mesquite beans”) from this plant or the dry ground pods are used to feed dairy and beef cattle, sheep, goats, swine, chickens, and rabbits. It has also been used for human consumption as bread, biscuits, jellies, sweets, and spirits. 11
Intoxication of cattle after consumption of P. juliflora pods has been reported in the USA, 6, 7 Peru, 2 and Brazil. 8 Clinical signs, which are more prominent during eating and rumination are characterized by masseter muscle atrophy, involuntary movements and protrusion of the tongue, a dropped (slack) mandible, tilting of the head during chewing, profuse salivation, yawning, and dysphagia. Continuous chewing, nervousness, ruminal atony, anemia, submandibular edema, and gradual emaciation are also observed. 11 Affected cattle waste away and die. If the plant is removed from the diet at the first stages of the disease, clinical recovery can be observed, but, in most cattle, recovery does not occur, and the clinical signs persist.
In Brazil, the disease was reported in the semi-arid regions of the states of Paraíba in 1981 and Rio Grande do Norte in 1986. 11 Since 1992, the number of outbreaks increased, and 50% of cattle were affected on some farms. 4 The disease was produced experimentally in cattle fed a ration containing 50 or 100% of the pods. All animals had clinical signs 3 months after the onset of the feeding trial. 10
The disease also occurs spontaneously in goats in Peru and was produced experimentally in this species by feeding pods of P. juliflora as the main source of food. 2 Clinical signs were characterized by twitching of the lips, head tremors, salivation, and emaciation. 2 In Brazil, intoxication was produced in goats after ingestion of food containing either 60 or 90% of P. juliflora pods for 270 days. The first clinical signs were observed after 210 days of ingestion. Histologic lesions were characterized by diffuse vacuolation of neurons in the trigeminal motor nuclei, Wallerian-like degeneration of the trigeminal and mandibular nerves, and denervation atrophy of the masseter, temporal, hyoglossal, genioglossal, styloglossal, medial pterygoid, lateral pterygoid, and mylohyoid muscles. 12 Trigeminal motor neuron vacuolation was also observed in a goat ingesting leaves, pods, or beans of the related plant Prosopis glandulosa (honey mesquite). 13 Denervation atrophy of the masseter is also observed in intoxicated cattle, but vacuolation of neurons had not been observed in this species, probably because surviving neurons had recovered from the vacuolation after withdrawal of the plant. 11
The objectives of this paper are to report the toxicity of dry ground P. juliflora pods, the histologic lesions in cattle intoxicated experimentally by P. juliflora pods and the dry ground pods, and the ultrastructural lesions in the trigeminal motor nuclei.
Material and Methods
Eight 6- to 12-months-old male crossbred steers were numbered 1 to 8 and randomly divided into 4 groups of 2 animals each. Group 1 (steer Nos. 3 and 6) was fed a ration containing 50% of the dry matter weight of P. juliflora pods. Groups 2 (Nos. 1 and 2) and 3 (Nos. 4 and 8) received a ration containing 50 and 75% of the dry matter weight of dry ground P. juliflora pods, respectively. The dry ground pods are produced by grinding the pods after dehydration in a hot furnace. In groups 1–3, the ration was complemented with Cynodon (Tifton) hay. Group 4 (Nos. 5 and 7) was the control and received only Tifton hay. The daily amount of food for each experimental animal corresponded to 3% of the body weight. All cattle were observed daily for signs of clinical disease. Neurologic examinations performed weekly focused particularly on motor and sensory cranial nerve functions.
After 200 days of consumption, all cattle were killed by electrocution, examined postmortem and widely sampled for complete histologic evaluation. Samples of lung, liver, kidney, pancreas, thyroid, adrenal, spleen, lymph nodes, muscles (masseter, medial pterygoid, lateral pterygoid, temporal, digastric, styloglossal, hyoglossal, buccinator, genioglossal, and geniohyoid), heart, forestomachs, abomasum, large and small intestine, peripheral nerves (trigeminal, maxillary, mandibular, hypoglossal, facial, and lingual), trigeminal ganglia, and paravertebral ganglia were fixed in 10% neutral formalin, embedded in paraffin, and sectioned at 6 μm. After fixation in 20% neutral formalin, transverse sections of the brain were made. The brain stem was sectioned in 3- to 4-mm-thick transverse sections, and all sections were processed for histology. Samples of cerebral cortex (frontal, occipital, temporal, and parietal), internal capsule and basal nuclei, and transverse and longitudinal sections from the cervical, thoracic, and lumbar spinal cord were also studied. All sections were stained by HE, and sections of the nervous system were also stained with Luxol fast blue for myelin.
For electron microscopy, samples of the left trigeminal motor nucleus were fixed in 2% glutaraldehyde with 2% paraformaldehyde in 0.4 M cacodylate buffer (pH 7.4), postfixed in 1% osmium tetroxide buffered in 0.4 M sodium cacodylate (pH 7.4), and embedded in Epon 812. Semithin sections were stained with methylene blue. Ultrathin sections were stained with lead citrate and uranyl acetate and examined with an EM 109 Zeiss transmission electron microscope at 80 Kv.
Results
All 6 cattle from groups 1, 2, and 3 showed motor and sensory cranial nerve dysfunction. The first clinical signs were a decreased response of the facial skin to pinprick, awkward mastication of food, and drooling of saliva. These signs were noted in all animals from the 3 groups, starting 45 to 75 days after the start of consumption. When the tongue was pulled out, a decreased tone was detected in all cattle from groups 2 and 3 and in one from group 1, 45 to 75 days after P. juliflora consumption. An inability to close the mouth because of a dropped jaw and reduced tone on manipulation of the mandible, difficulties in prehending food, and yawning were observed after 80 days consumption in 2 animals (one from group 1 and one from group 2). Delay or inability to swallow prehended food was observed in all cattle from groups 1 and 3 and in one from group 2, starting at day 160. One animal from group 1 also had tongue protrusion, tilting the head with dropping of food from the mouth during chewing, swallowing impairment, mandibular tremors during chewing or rumination, bradycardia, ruminal atony, and, after 110 days, became dehydrated. Excessive tongue movements were observed in 1 animal from group 1. All clinical signs observed persisted until the end of the experiment at 200 days. In the control group 4, both animals ate normally, gained weight, and had no neurologic abnormalities. At necropsy, no macroscopic lesions were observed in all cattle ingesting P. juliflora pods or dry ground pods. The main histologic lesions were observed in the trigeminal motor nuclei (Table 1). In these nuclei, a few neurons had fine vacuolation of the perikaryon that presented a granular or spongy appearance (Figs. 1, 2). Dark distended nuclei, sometimes displaced to the margin of the perikaryon were observed in some neurons (Fig. 1). Occasionally, ghost neurons, characterized by a pale perikaryon with dissolution of the Nissl substance, and undefined borders were observed. On other occasions, a round cavity bordered by eosinophilic material suggested neuronal loss. Axonal spheroids were rarely observed (Fig. 2). Reactive astrocytes with vesicular dilated nuclei and scant eosinophilic cytoplasm were observed in low number within the trigeminal motor nuclei. Mild neuronal loss was detected in all treated animals when compared with control animals.
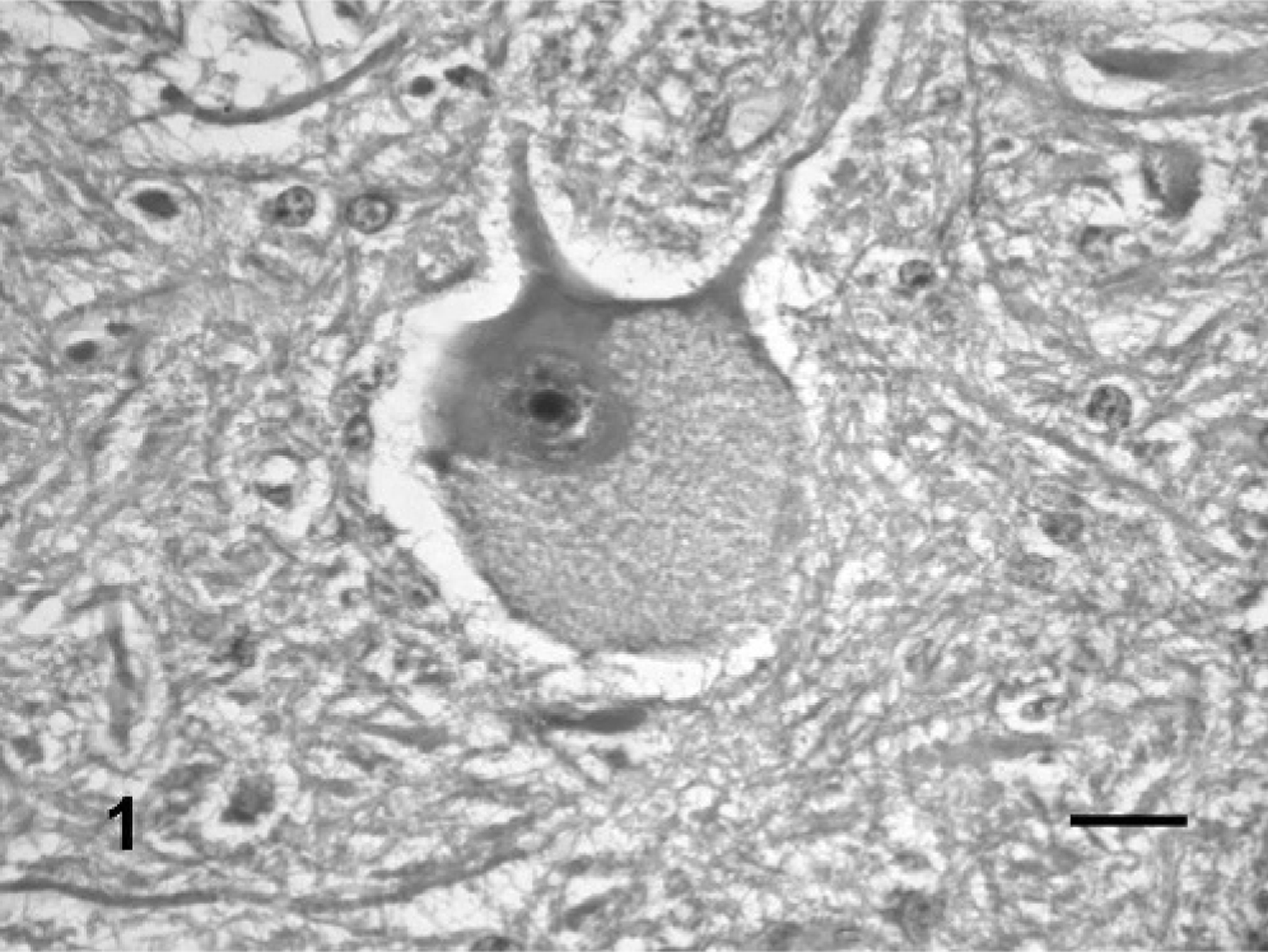
Neuron of the trigeminal motor nuclei; steer No. 3. The neuron has a diffusely vacuolated perikaryon and a dark emarginated nucleus. HE. Bar = 25 μm.
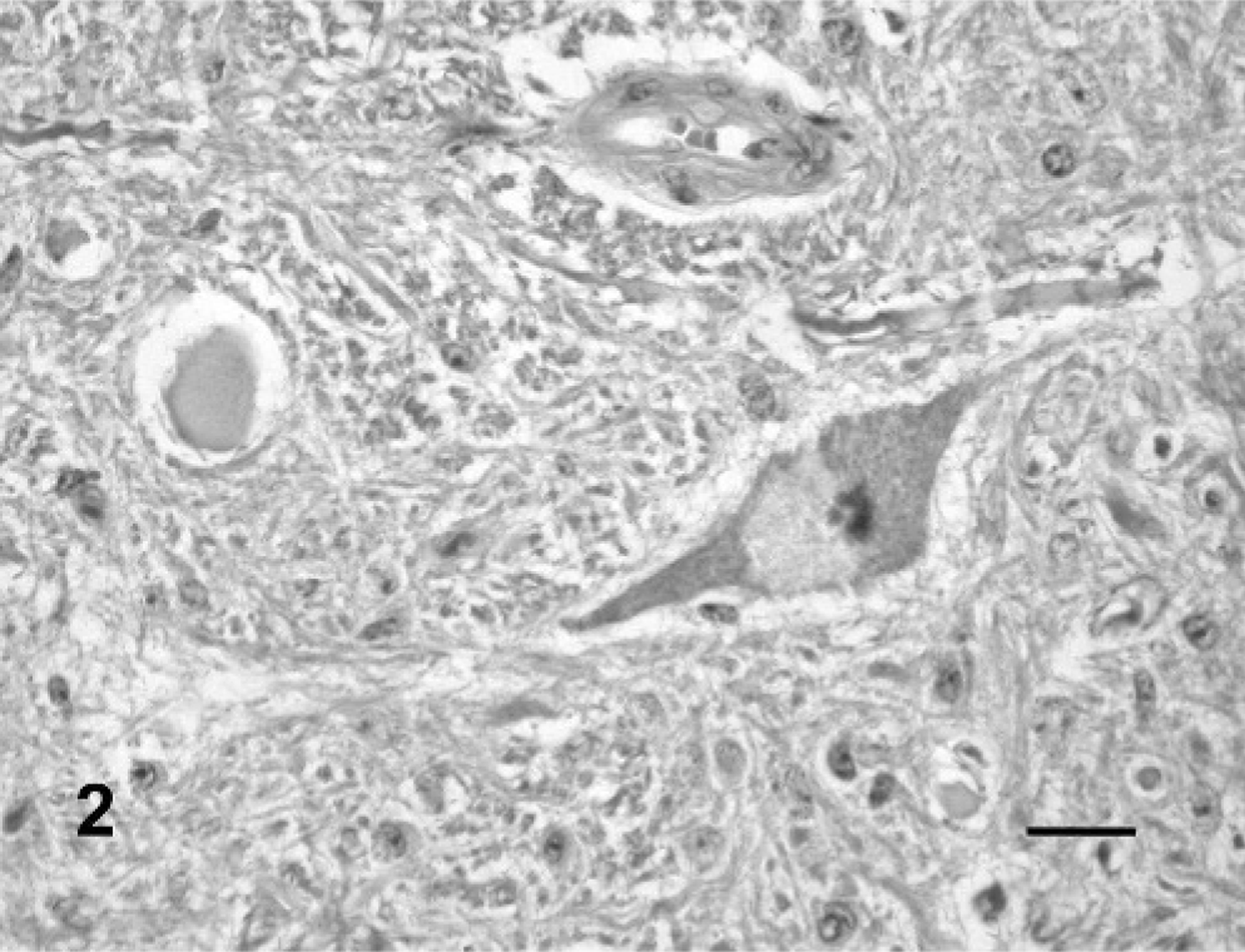
Neuron of the trigeminal motor nuclei; steer No. 4. The neuron has partially vacuolated perikaryon and a dark nucleus. An axonal spheroid is also observed. HE. Bar = 25 μm.
Histologic lesions in cranial nuclei and nerves in steers from groups 1(50% of pods in the food), 2 (50% dry ground pods), 3 (75% dry ground pods), and 4 (control).∗
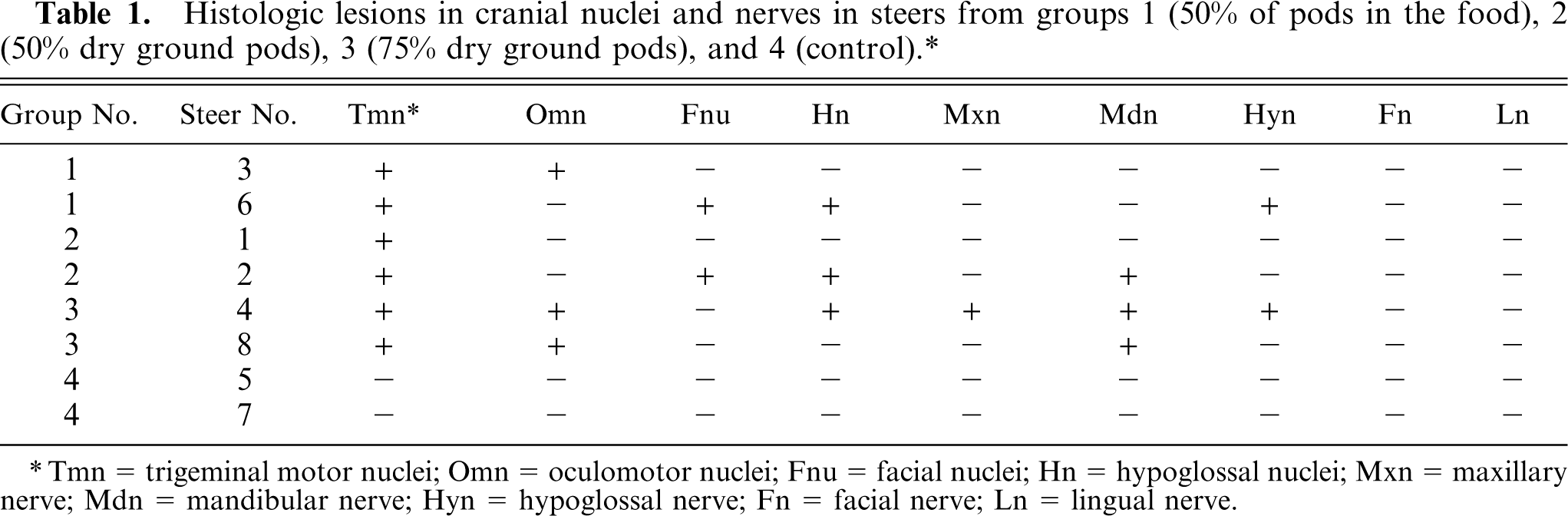
Tmn = trigeminal motor nuclei; Omn = oculomotor nuclei; Fnu = facial nuclei; Hn = hypoglossal nuclei; Mxn = maxillary nerve; Mdn = mandibular nerve; Hyn = hypoglossal nerve; Fn = facial nerve; Ln = lingual nerve.
In some sections, mild perivascular cuffing of mononuclear cells was observed. Similar but milder lesions were observed occasionally in neurons of the facial, hypoglossal, and oculomotor nuclei. Wallerian-type degeneration suggested by short chains of 2 to 5 vacuoles side by side, occasionally containing eosinophilic myelin residues or some macrophages were observed in the intracranial roots of facial, hypoglossal, and oculomotor nerves (Fig. 3). Wallerian-like degeneration of variable severity was observed also in the maxillary, mandibular, hypoglossal, facial, and lingual nerves. The presence of neurohistologic lesions in each animal is shown in Table 1.
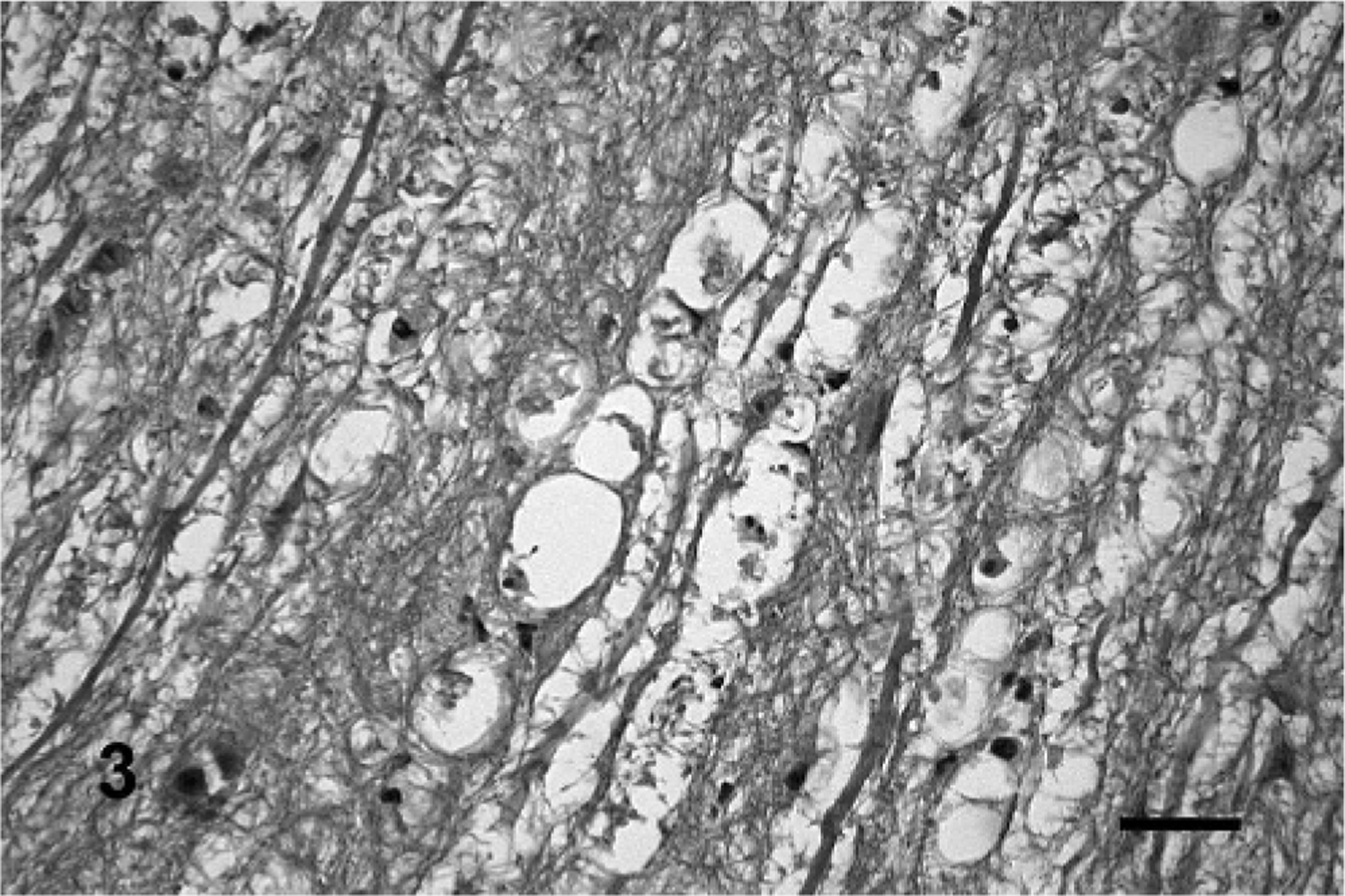
Intracranial trigeminal roots; steer No. 4. The intracranial root has Wallerian-type degeneration indicated by fragmentation or loss of axons and short chains of 2 to 5 vacuoles (myelin ellipsoids) side by side. Eosinophilic myelin residues and a few macrophages are also observed. HE. Bar = 40 μm.
Mild denervation atrophy characterized by fiber-size variation with fibers of decreased size and some angular fibers, abundant internal nuclei, and occasional vacuolated fibers were observed in the different muscles studied (Fig. 4), especially the masticatory muscles. The distribution of lesions in the different muscles is presented in Table 2.
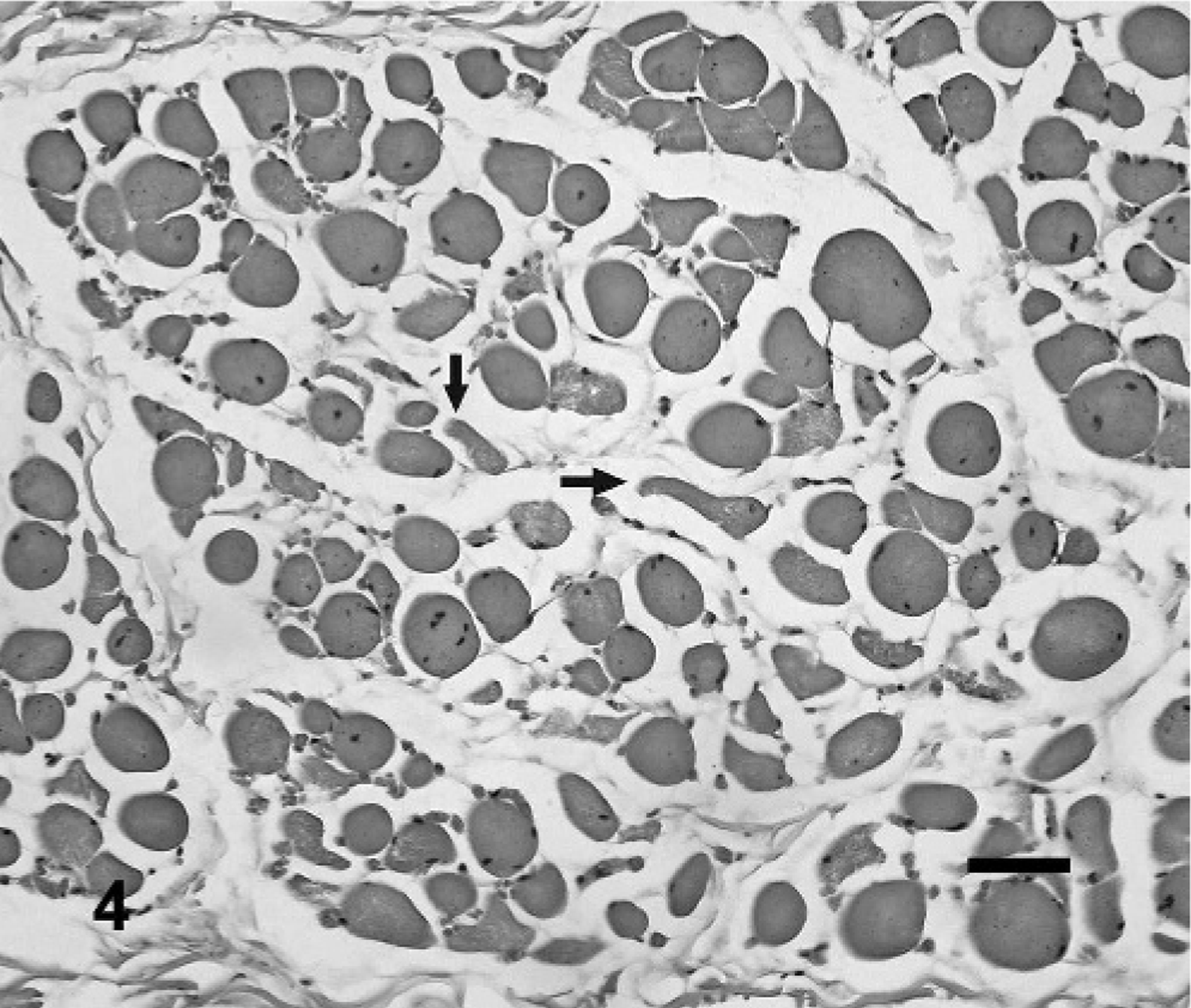
Buccinator muscle; steer No. 1. Myopathic changes are indicated by the fiber-size variation, abundant internal nuclei, and occasional vacuolated fibers. Changes suggestive of denervation atrophy are fibers of decreased size and small angular fibers (arrows). HE. Bar = 40 μm.
Histologic lesions in the muscles in steers from groups 1(50% of pods in the food), 2 (50% dry ground pods), 3 (75% dry ground pods), and 4 (control).∗
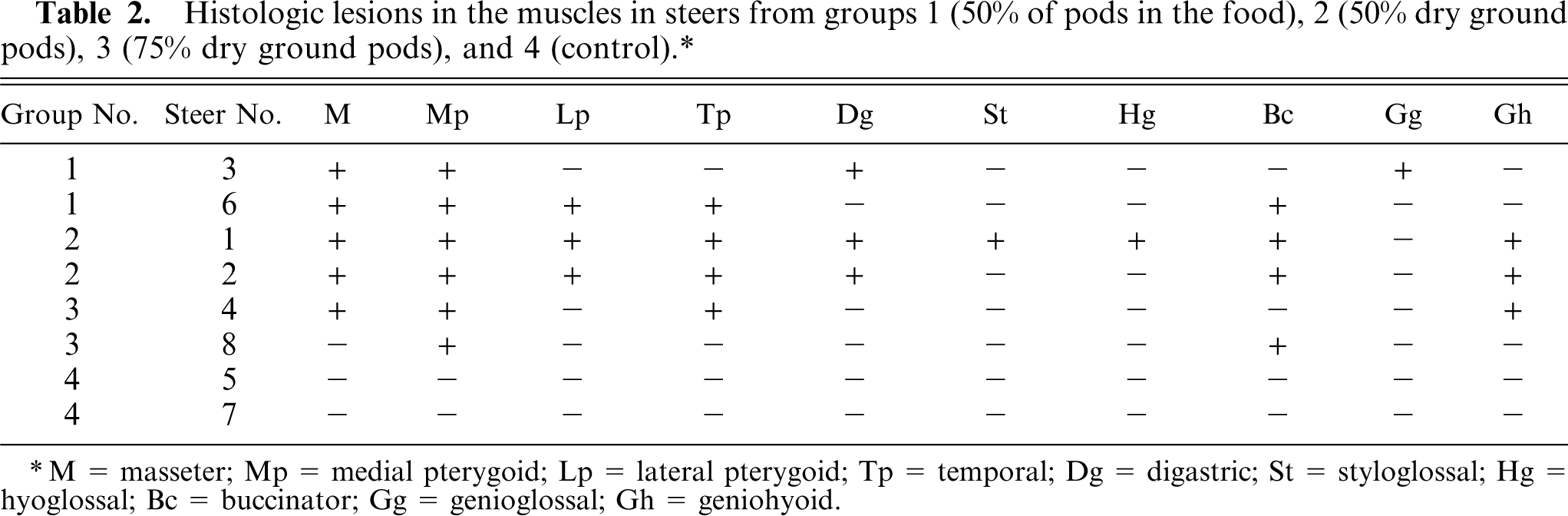
M = masseter; Mp = medial pterygoid; Lp = lateral pterygoid; Tp = temporal; Dg = digastric; St = styloglossal; Hg = hyoglossal; Bc = buccinator; Gg = genioglossal; Gh = geniohyoid.
No significant lesions were observed in the CNS of the control cattle and in other tissues of experimental and control cattle.
In semithin sections of the trigeminal nuclei, many neurons had diffuse vacuolation of the perikaryon, sometimes with a dark nucleus. Neurons from control animals appeared normal. In some neurons, small blue dark round bodies were dispersed in the perikaryon between the vacuoles. The major ultrastructural finding in neurons of the trigeminal nuclei was markedly swollen mitochondria, corresponding to the vacuoles observed at light microscopy. The mitochondrial cristae were displaced peripherally, disoriented, and disintegrating (Fig. 5). In severely affected mitochondria, the cristae were extremely shortened or absent (Fig. 6). In the inner chamber of some mitochondria membrane rests representing a result of damage of the organelle were seen (Fig. 6). Intramitochondrial dense granules were absent. In the control animals, the mitochondria had normal appearance (Fig. 7). Dilated mitochondria similar to those of the perikaryon were found in neuronal processes. The Golgi complex was dilated, but the endoplasmic reticulum was not altered. There was an increased number of lysosomes consistent with secondary lysosomes with membranous to granular/amorphous electron-dense residual bodies (Fig. 5), corresponding to the small blue bodies dispersed in the perikaryon seen in the semithin sections. In one degenerated neuron, paracrystalline filaments were observed; these were 10–12 nm wide, highly organized, and separated by an electron-lucent 10-nm-wide band either in parallel or in lattice-like arrays (Fig. 8).
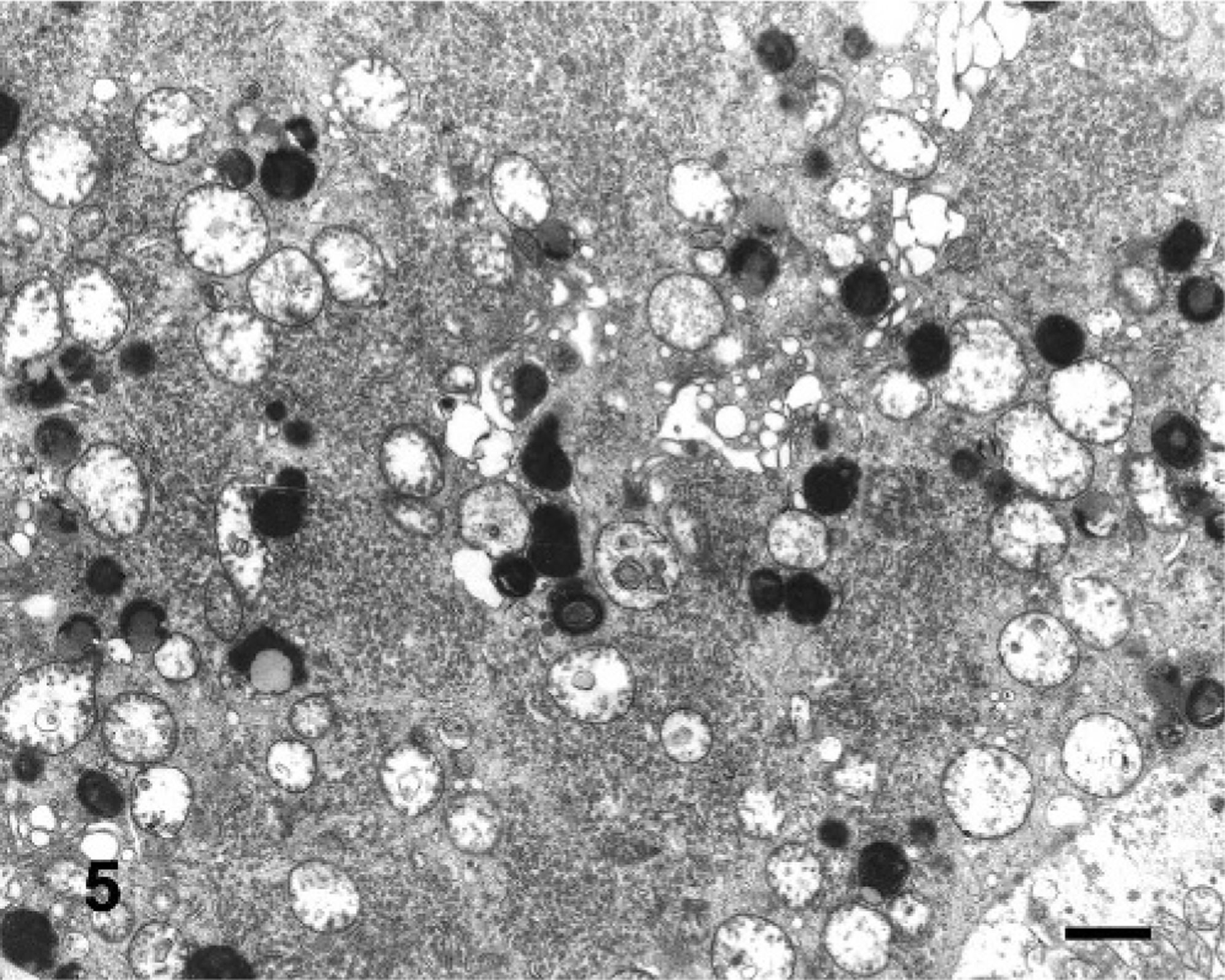
Electron microscopy micrograph. Neuron of the trigeminal motor nuclei; steer No. 6. Perikaryon contains distended mitochondria with shortened and disintegrating cristae. The rough endoplasmic reticulum was well preserved. There were numerous lysosomes with residual bodies. Bar = 1 μm.
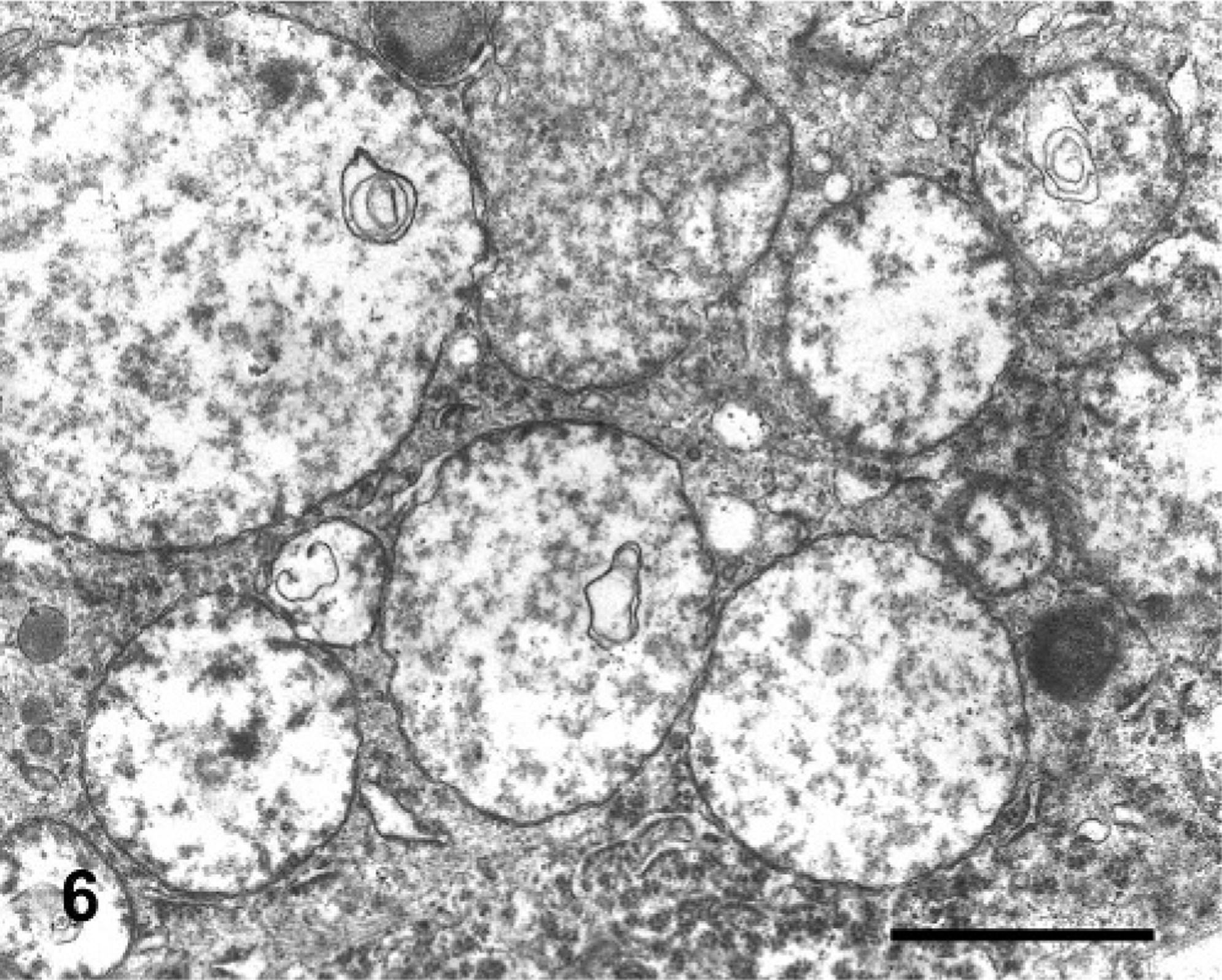
Electron microscopy micrograph. Neuron of the trigeminal motor nuclei; steer No. 6. Perikaryon with extremely distended mitochondria, showing disruption of the matrix. The cristae were shortened, disintegrated, or largely absent. The outer mitochondrial membrane was not altered. Concentric membranous bodies were seen in some mitochondria. Lysosomes showed residual bodies. Bar = 1 μm.
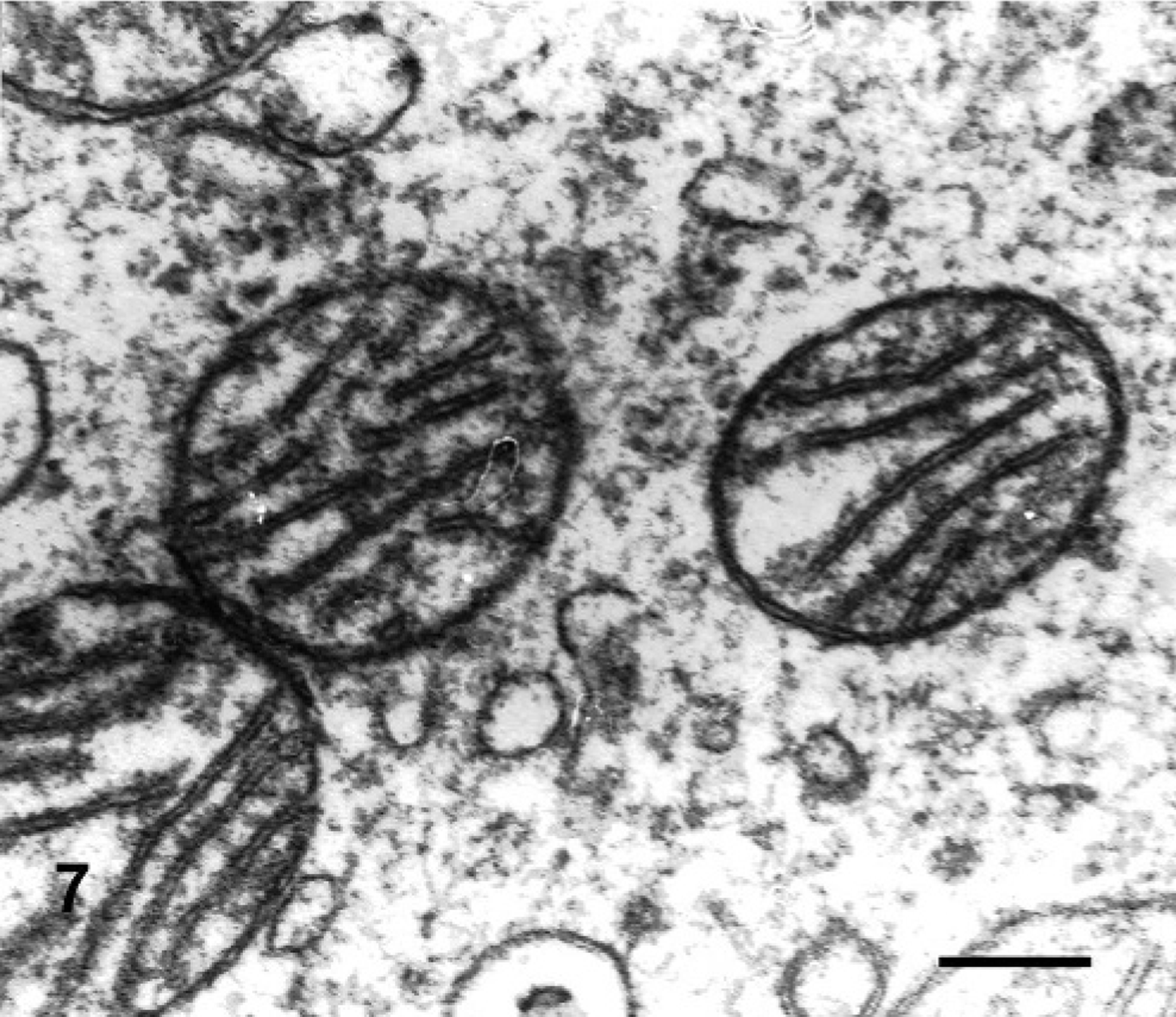
Electron microscopy micrograph. Neuron of the trigeminal motor nuclei; steer No. 7 (control). Perikaryon of a neuron of the trigeminal nucleus. Mitochondria were well preserved. Bar = 0.5 μm.
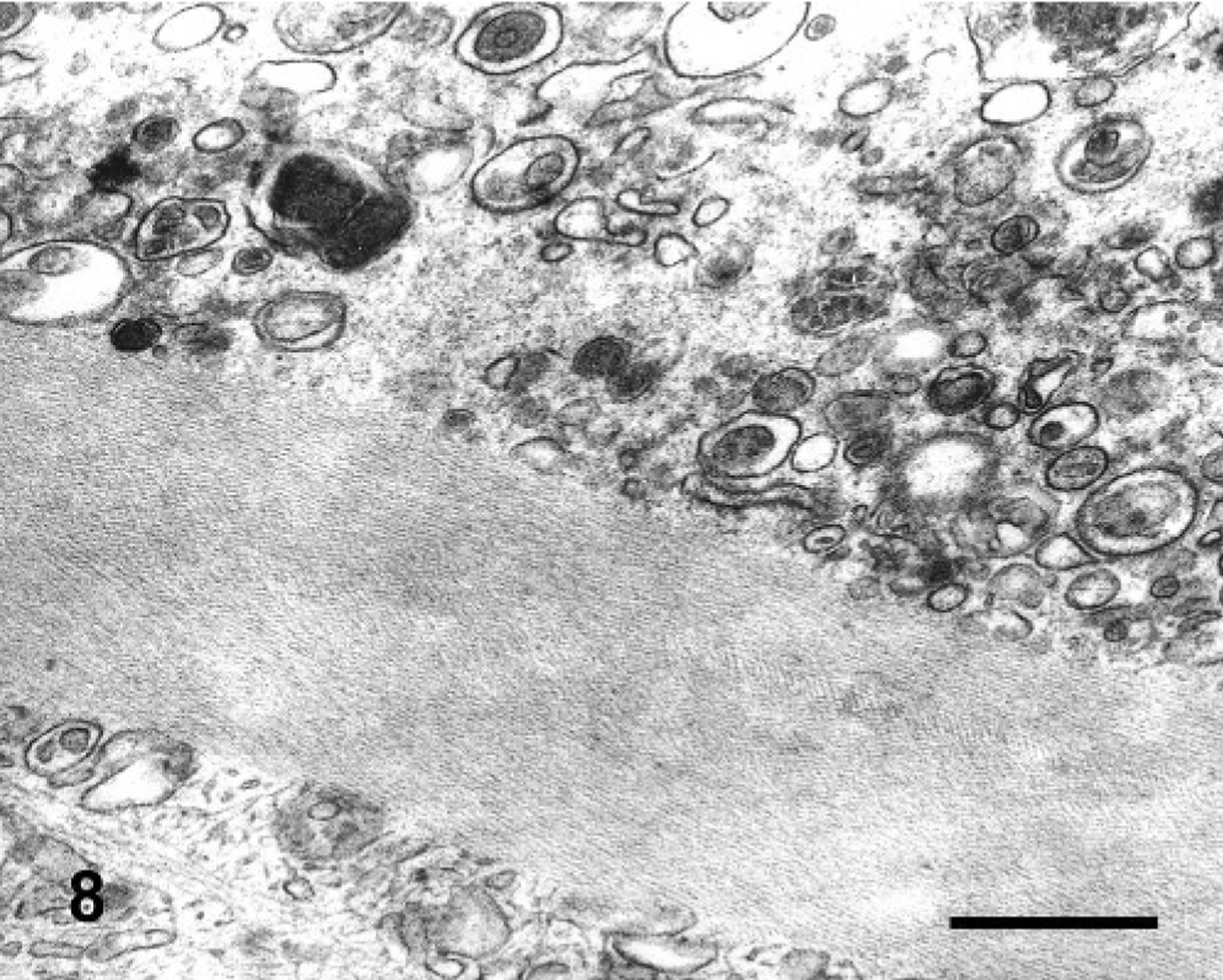
Electron microscopy micrograph. Neuron of the trigeminal motor nuclei; steer No. 4. Perikaryon containing a Hirano body with the typical fibrils in parallel or latticelike pattern. Bar = 0.5 μm.
Discussion
The present experiment confirmed the toxicity of P. juliflora pods and the dry ground pods for cattle, with the first clinical signs appearing after 45 to 75 days of ingestion. Regardless of whether cattle were fed a diet containing 50% pods (group 1) or either 50 or 75% dry ground pods (groups 2 and 3, respectively), the onset and the nature of the clinical deficits were essentially the same. Clinical signs were similar to those observed previously, after the experimental administration of P. juliflora pods to cattle. 7, 10 In our experiments, intoxication was produced by the ingestion of dry ground P. juliflora pods. The toxicity of the dry ground pods not previously reported, is an important point, because this is the best form to store the pods and is largely used to produce concentrate food for different domestic animals in the northeastern region of Brazil.
The neurologic deficits observed suggested degeneration of neurons in the nuclei of several cranial nerves (CN): the dropped jaw and lack of mandibular tone indicated the motor division of CN V, whereas the facial hypalgesia pointed to sensory V (spinal nucleus of V or possibly the trigeminal ganglion); the decreased tongue tone indicated CN XII; the dysphagia suggests nucleus ambiguus (CN IX and X); the drooling of saliva also indicated dysphagia. On histologic study, the most important lesions were vacuolation and loss of neurons in trigeminal motor nuclei and Wallerian-like degeneration of the trigeminal nerve and Wallerian-type degeneration of intracranial nerve roots. Changes in neurons of other nuclei, including the oculomotor, facial, and hypoglossal, were more subtle than those observed in the trigeminal motor nuclei, but the Wallerian-like degeneration observed in the nerves and nerve roots demonstrated that these other cranial nerve nuclei were also affected. While involvement of other nuclei had not been reported previously in cattle, vacuolation of neurons of the trigeminal motor and oculomotor nuclei and Wallerian-like degeneration of cranial nerves were previously reported in the experimental intoxication by P. juliflora pods in goats. 12 In this species, the lesions were more marked, probably because goats ingested the pods for a longer period (270 days). In field and experimental conditions, goats are more resistant to the intoxication than cattle. 11 Vacuolization of the trigeminal motor nuclei is also reported in the spontaneous intoxication by Prosopis glandulosa in goats. 13
As suggested by the clinical signs, the main effect of the intoxication is an impairment of CN function, mainly affecting the trigeminal motor nerve but also other cranial nerves. The histologic study suggests that the primary lesion was neuronal degeneration followed by neuronal loss with Wallerian-like degeneration of the cranial nerves, leading to denervation atrophy of the masticatory and lingual muscles. In spontaneous cases studied previously, the histology of the CNS revealed a moderate loss of neurons in the trigeminal motor nuclei. The remaining neurons were not vacuolated. The GFAP stain in these natural cases revealed a widespread, mild astrocytosis in affected nuclei. Because those cattle probably did not ingest P. juliflora pods for some months before necropsy, the absence of neuronal vacuolation in the remaining neurons in the trigeminal nuclei suggests that these surviving neurons recovered from the vacuolation after withdrawal of the plant. 11 In this experiment, denervation atrophy of the masseter and other masticatory muscles was mild, probably because of the relatively short period of ingestion. In spontaneous cases in which animals ingested the pods for longer periods, masseter atrophy was severe and most muscle fibers were replaced by fat or connective tissue. 11
In the semithin sections of the brain stem containing the trigeminal motor nuclei, vacuolization was much more evident than the lesion suggested by the routine hematoxylin and eosin histologic sections, and affected many neurons in the nuclei. On the ultrastructural study, it was found that the vacuolation observed at light microscopy was caused by markedly swollen mitochondria. The swelling of mitochondria after injury results from the entry of solutes and water into the mitochondrial matrix. 9 In severe swelling, the mitochondria are transformed into structureless vacuoles that contribute to the vacuolar appearance of swollen cells. When primary injury to mitochondria is the cause of swelling, these organelles may be massively enlarged. 3 Mitochondria are susceptible to a variety of toxins that interfere with their biochemical function, 1, 3 but the molecular pathogenesis of this intoxication and the basis for the selective effect on cranial motor neurons remain to be investigated. Despite the severe mitochondrial injury, no calcium-sequestering granules, a very common finding in cell injury with mitochondrial swelling, were found in this study. The paracrystalline filaments observed in neurons are very similar to the Hirano bodies described in some chronic degenerative disease of humans, such as Pick's disease, Alzheimer's disease, in scrapie-affected animals, and Kuru-affected chimpanzees. 5 F-actin, along with other proteins, was reported as the main component of the filaments. In our case, Hirano bodies were observed only in degenerating neurons, and we cannot speculate about the significance of this incidental finding.
No references could be found in the veterinary or human literature describing selective mitochondrial injury to neurons of the cranial nerve nuclei or other locations in the CNS. P. juliflora toxicity seems to have a novel pathogenesis for a plant intoxication, characterized by a selective, primary, chronic, and progressive injury to mitochondria of neurons of the trigeminal and other motor cranial nerve nuclei, with extreme and diffuse enlargement of mitochondria and cristolysis followed by neuronal degeneration and loss. Cranial nerve degeneration and denervation atrophy of the muscles occurs as a consequence of the neuronal lesion. Difficulty in prehending, chewing, and swallowing food slowly leads to wasting and death.
Footnotes
Acknowledgements
We thank Dr. A. de Lahunta for assistance in correlating the neurologic and neuropathologic changes described in this paper. This work was financially supported by the Programa de Apoio a Núcleos de Excelência (PRONEX), Grant N° 661069/1997 and by the National Council of Scientific and Technologic Development (CNPq), Grant N° 465681/00-0.
