Abstract
Eosinophilic crystalline pneumonia is an idiopathic disease that occurs in many strains and stocks of mice, more commonly in strains on a C57BL/6 background. The disease occurs sporadically in most strains of mice and varies from mild and subclinical to severe and fulminating, sometimes resulting in respiratory distress and death. In this study, 94 aged male and female 129S4/SvJae mice were evaluated for eosinophilic crystalline pneumonia lesions. There was an 87% incidence, with females overrepresented. Histologically, there were multifocal to coalescing inflammatory infiltrates composed of numerous large eosinophilic macrophages and multinucleate cells admixed with eosinophils, neutrophils, lymphocytes, and plasma cells within alveolar and bronchiolar spaces, associated with refractile, brightly eosinophilic, angular crystals. Alveolar macrophages and multinucleate cells contained fine needlelike to rectangular intracytoplasmic crystalline material. Similar crystals were often free within alveoli and conducting airways, often associated with mucous metaplasia of bronchiolar epithelium. This disease may occur spontaneously or in concert with other pulmonary lesions, such as pulmonary adenomas, lymphoproliferative disease, allergic pulmonary disease, and parasitic or fungal infections. The characteristic crystals morphologically resemble Charcot-Leyden crystals, which represent eosinophil breakdown products in humans with eosinophil-related disease. However, crystals in eosinophilic crystalline pneumonia are composed predominantly of Ym1 protein, a chitinase-like protein associated with neutrophil granule products and secreted by activated macrophages. The function of Ym1 protein is not fully understood but is believed to be involved in host immune defense, eosinophil recruitment, and cell-cell and cell-matrix interactions consistent with tissue repair. The mechanism of induction of eosinophilic crystalline pneumonia with Ym1 crystal formation is unknown.
Keywords
Eosinophilic crystalline pneumonia, also known as acidophilic macrophage pneumonia or crystalline pneumonitis, is a sporadic, idiopathic pulmonary disease of laboratory mice, more commonly affecting certain naturally occurring strains of laboratory mice (C57BL/6, 129Sv), 9, 16, 17 specific strains developed from naturally occurring recessive mutations such as Ptpn6^me (motheaten) mice, 2, 9, 12, 14, 17 severe combined immunodeficiency (SCID) mice, 4 as well as some knockout mice (such as CYP1A2 null) on a C57BL/6 or 129 background. 16 Embryonic stem (ES) cells from the 129 mouse strain have been used extensively for targeted mutagenesis in the development of genetically engineered mice. 13 However, because of accidental and deliberate outcrossing over the years, the 129 strain has been subdivided into several substrains, resulting in extensive genetic variability across substrains, as well as in the ES cells derived from them. 13 The genetic variability among the 129 substrains results in different substrain background phenotypes. Although ES cells from the 129 substrains are easy to manipulate in culture by using these ES cells to derive transgenic and knockout models will result in accumulation of background “nonlesions” specific to the substrain but unrelated to the transgene or knockout construct. 13 This is of prime importance to consider when developing a genetically engineered mouse model, particularly when background lesions that are not caused by the genetic construct are present in the organ system or systems of interest to the investigator.
Eosinophilic crystalline pneumonia can be subclinical to fatal, may occur alone or in conjunction with other pulmonary diseases, and tends to increase in incidence with age. 4, 6, 8, 9, 11– 17 In the 129Sv strain, females are reportedly more often affected than males. 8, 16 The pneumonia is characterized by patchy to lobar lung consolidation, caused by alveolar filling, with large numbers of foamy to acidophilic macrophages and giant cells, admixed with variable numbers of other inflammatory cell types, eosinophilic variably sized acicular crystals within macrophages and multinucleate giant cells, as well as extracellularly within alveolar spaces and bronchiolar lumina. 4, 6, 9, 14, 15, 16
Eosinophilic crystals in various organs of laboratory mice have been described as early as 1905; 4, 5 however, the disease condition of eosinophilic crystalline pneumonia was not described until 1942, when it was characterized by Green. 3, 9 Eosinophilic crystals have been documented in the lower pulmonary tract, hepatobiliary tract, as well as hematopoietic tissues, such as lymph nodes, spleen, and bone marrow. 4, 9, 11, 16 The most severe manifestation and the principal site of accumulation of crystals occurs in the lungs as eosinophilic crystalline pneumonia. 4 The following report describes the gross and histopathologic features of eosinophilic crystalline pneumonia in a series of 94 aged 129S4/SvJae laboratory mice and discusses the biochemical nature of the crystals that are a pathognomonic feature of the disease.
Materials and Methods
Signalment, necropsy, and histopathologic findings from a set of 94 aged 129S4/SvJae mice have been reported previously. 15, 16 In the present study, we provide a more thorough description of the histopathology, electron microscopy, and Ym1 immunohistochemistry from the same set of mice. Mice were bred in our facilities and were maintained at 5 animals per cage for up to 27 months of age. Mice were allowed to live out their life spans in compliance with the recommendations of the Guide for the Care and Use of Laboratory Animals of the National Research Council. 15, 16 Few mice died spontaneously, and mice were euthanized when signs of progressive deterioration because of aging changes were observed. Common causes of death or illness included macrophage pneumonia and tumors of the lung and Harderian gland. More than a half of 129S4/SvJae mice survived to 24 months of age; the mean survival of all mice studied was approximately 102 to 104 weeks for male and female mice, respectively.
Tissues were collected in 10% neutral buffered formalin and embedded in paraffin. Tissue sections were deparaffinized with xylene and ethanol by standard methods, and stained with hematoxylin and eosin (HE). For Luna histochemical staining, tissue sections were deparaffinized and hydrated in distilled water, stained in hematoxylin for five minutes, then differentiated in 1% acid alcohol and rinsed in tap water. Slides were then dipped in lithium carbonate solution until sections turned blue, then rinsed with tap water for 2 minutes. Sections were then routinely dehydrated and mounted. Immunohistochemical staining was performed on formalin-fixed tissues embedded in paraffin and sectioned at 4 to 6 μm. Tissues were incubated with rabbit polyclonal anti-Ym1 antibody at dilutions of 1°1,000. The antibody was prepared by Shioko Kimura (National Cancer Institute, Bethesda, MD) by immunizing rabbits with 2 peptide sequences (Macromolecular Resources, Fort Collins, CO), QYNFDGLNLDWQYPGSRGSPPK and DLHDPKDGYTGENSPLYKSPYD, derived from peptide sequencing of the band from the sodium dodecyl sulphate gel from eosinophilic crystalline lesions in the gastric epithelium of CYP1A2 mice. 16 These peptide sequences matched those of secretory Ym1 when subjected to a GenBank search (GenBank U56900). Based on the published sequence, which exhibited good antigenicity by computer analyses, an antibody was made by using part of these peptide sequences with extension of amino acid residues 130 to 151 and 203 to 234, respectively. 16
All lung sections were incubated with the primary antibody for 60 minutes at 4°C by using the Vectastain ABC Rabbit Elite Kit (Vector Laboratories, Inc., Burlingame, CA). Diaminobenzidine (Dako, Carpinteria, CA) was the chromagen with hematoxylin as the counterstain. For transmission electron microscopy (TEM), the lung tissue was fixed in 2% glutaraldehyde in 0.1 M cacodylate buffer (pH 7.4) overnight. The tissue was washed with cacodylate buffer and postfixed with 1% OsO4 tetroxide for 2 hours. The tissue was washed again with 0.1 M cacodylate buffer, serially dehydrated in ethanol and propylene oxide, and embedded in Eponate 12 resin (Ted Pella, Redding, CA). Thin sections (∼80 nm) were obtained by using the Leica ultracut-UCT ultramicrotome (Leica, Deerfield, IL), placed onto 400-mesh copper grids and stained with saturated uranyl acetate in 50% methanol and then with lead citrate. The grids were viewed in the Philips 410 electron microscope (FEI, Hillsboro, OR) at 80 kV, and images were recorded on Kodak (Rochester, NY) SO-163 film.
Results
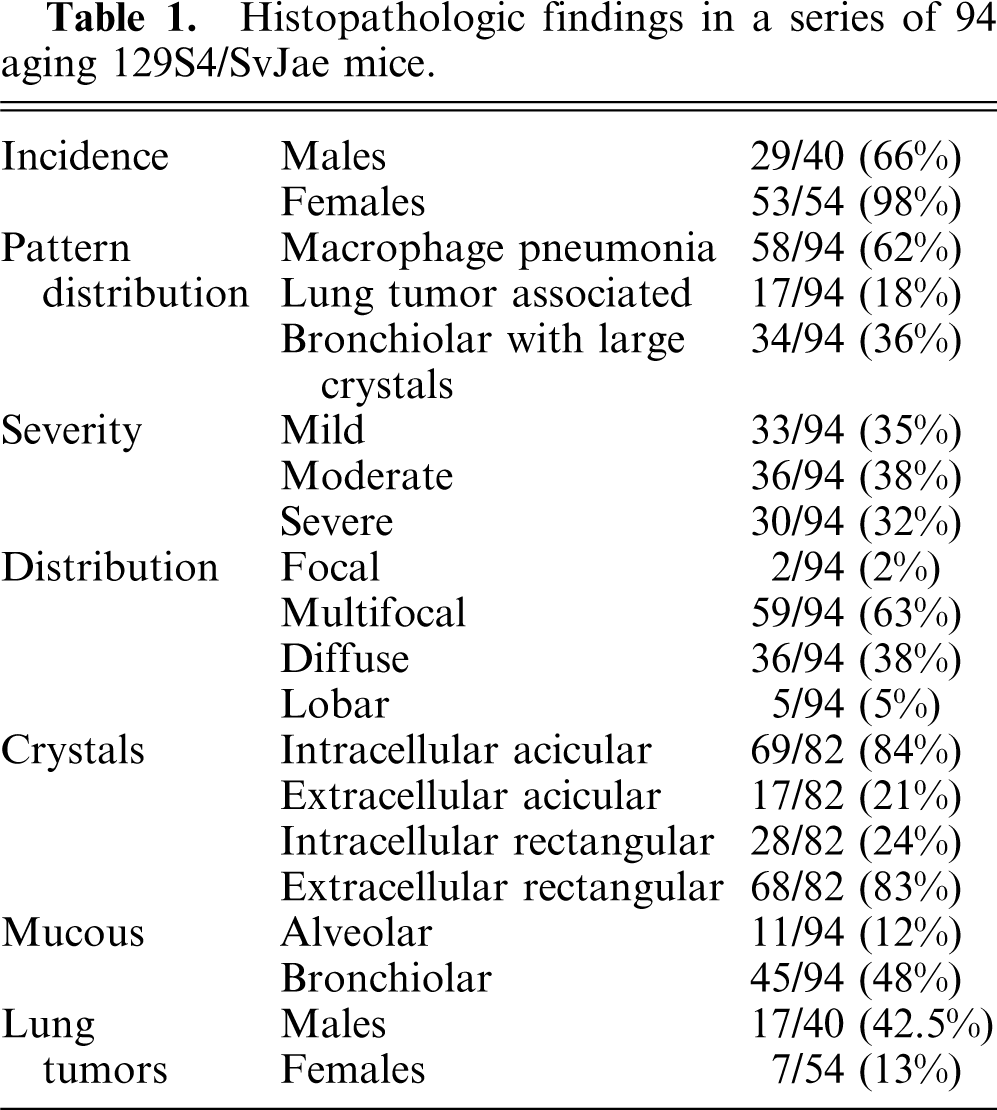
The incidence of eosinophilic crystalline pneumonia in this study of mice was approximately 87%, with females greatly overrepresented (29/40 males [66%]; 53/54 females [98%]) (Table 1). As reported in the previous study of this group of mice, 15 gross lesions ranged from multifocal parenchymal infiltrates to lobar to diffuse lung involvement, characterized by whitish-tan to red discoloration, consolidation, and failure of lungs to collapse upon opening the thoracic cavity (Fig. 1). Histologically, further assessment of this group of pulmonary lesions in the current study indicated 3 main patterns of lung lesions (Table 1), with variable intensity and distribution, often with a multifocal to coalescing pattern (Fig. 2). The first pattern consisted of lobar to diffuse interstitial inflammatory infiltrates of variable severity, composed predominantly of large numbers of activated macrophages and numerous multinucleate cells with abundant eosinophilic and homogenous cytoplasm admixed with fewer eosinophils, lymphocytes, and occasional neutrophils, associated with moderate to severe lymphoplasmacytic perivascular and peribronchiolar cuffing (Fig. 3). Within macrophages and giant cells, as well as free within alveoli, were variably sized, fine acicular (needle shaped) and rectangular, partially refractile eosinophilic crystals (Fig. 4). The second lesion pattern was consistent with macrophage reaction to pulmonary neoplasia, such as type II cell adenomas and adenocarcinomas (Fig. 5). In this lesion, there were few to no crystals, and the macrophage infiltrate was usually localized only to the region of the lung tumor. The third pattern was usually focal to multifocal and focused on bronchioles, with large rectangular crystals in airways, and minimal macrophage infiltrates (Fig. 6). The first pattern predominated in a majority of aged 129 mice, whereas lesions with the latter 2 patterns were sporadic, involving only a few animals. These findings suggest that there are 3 types of lung lesions, with macrophage predominance in aged 129 mice.
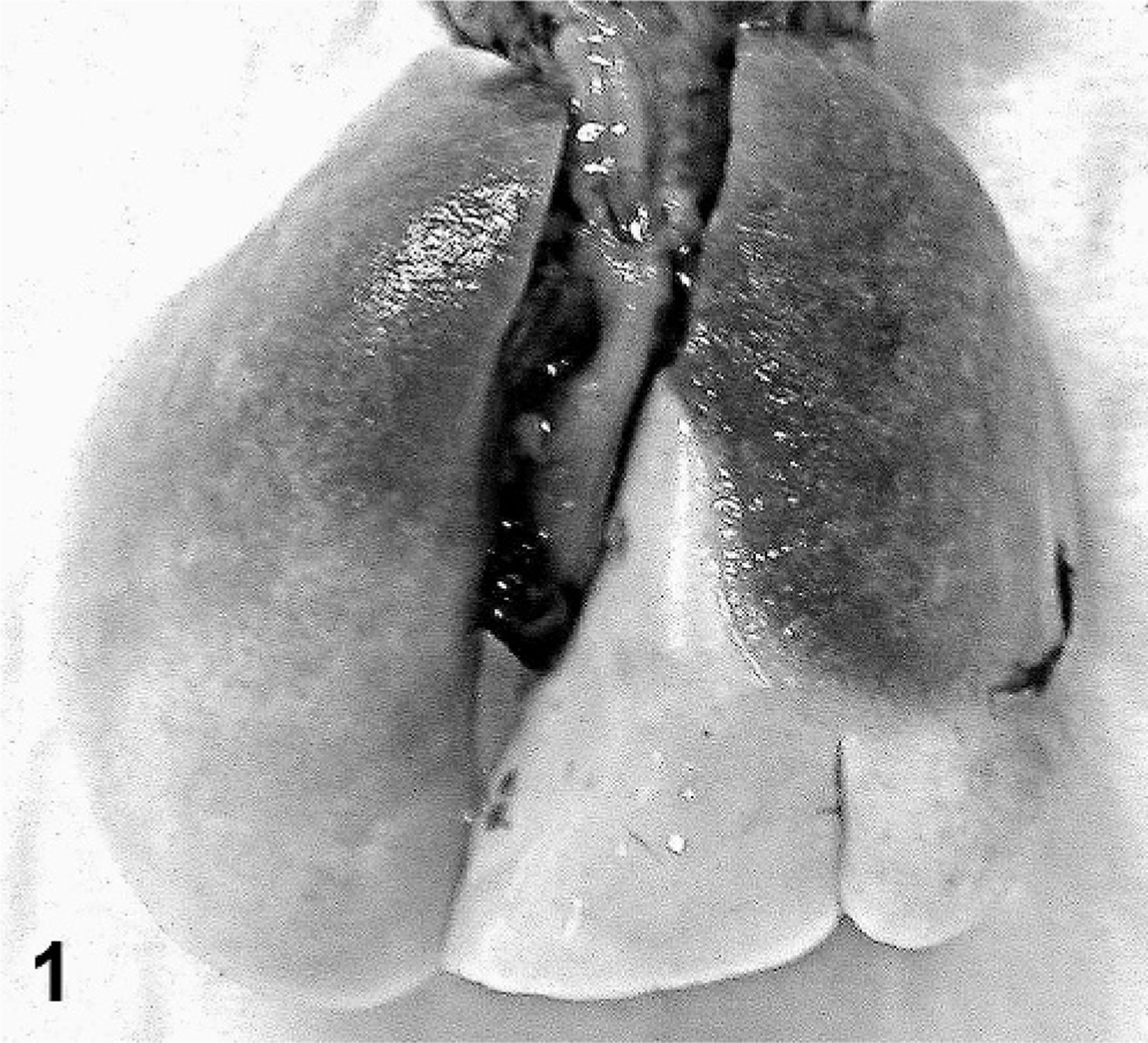
Lungs; 129S4/SvJae mouse. There is lobar consolidation with whitish tan to dark red mottling, and affected areas are firm and meaty on palpation and fail to collapse.
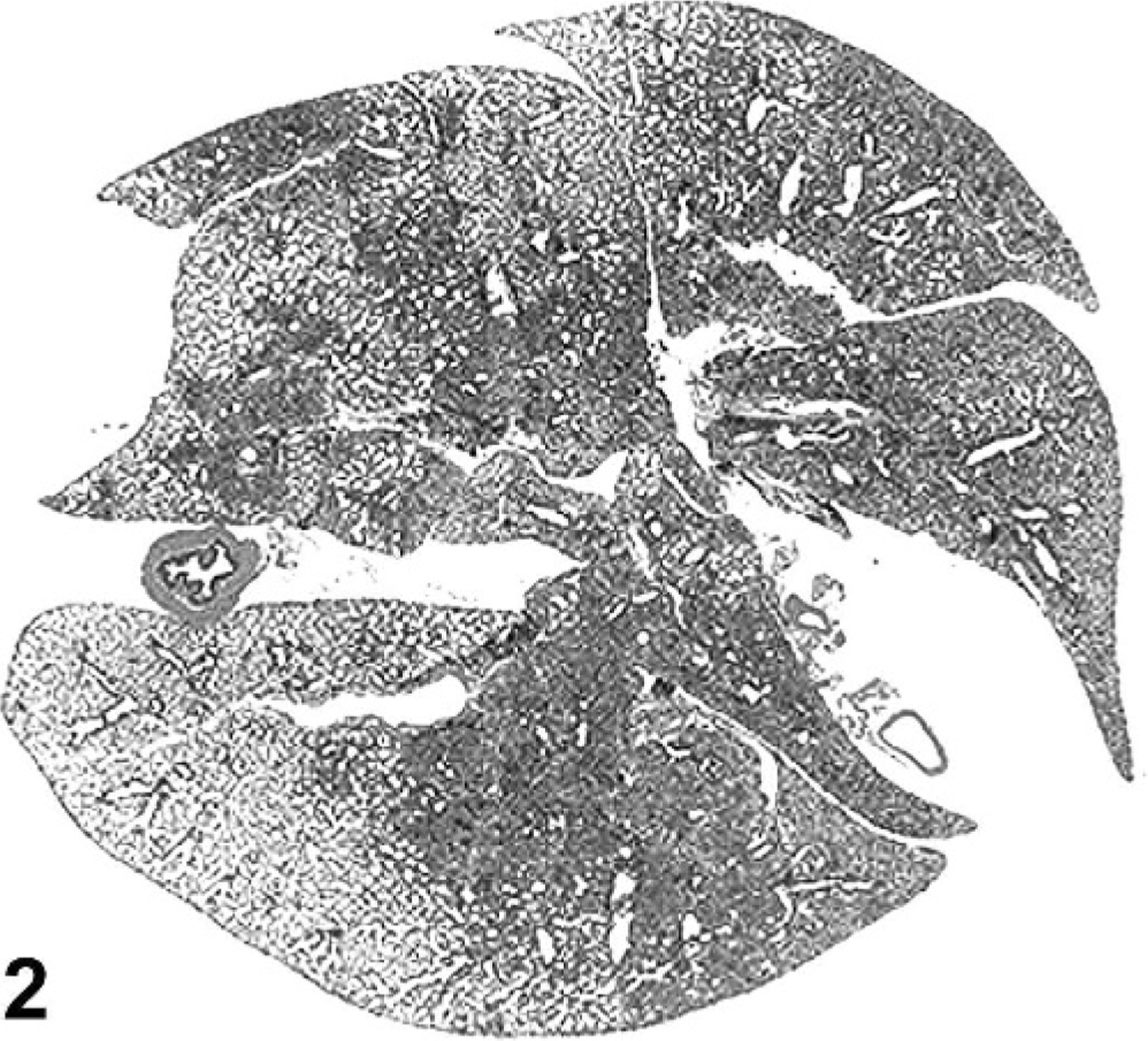
Lung; 129S4/SvJae mouse. There is a severe multifocal to coalescing inflammatory infiltrate involving all lung lobes. HE.
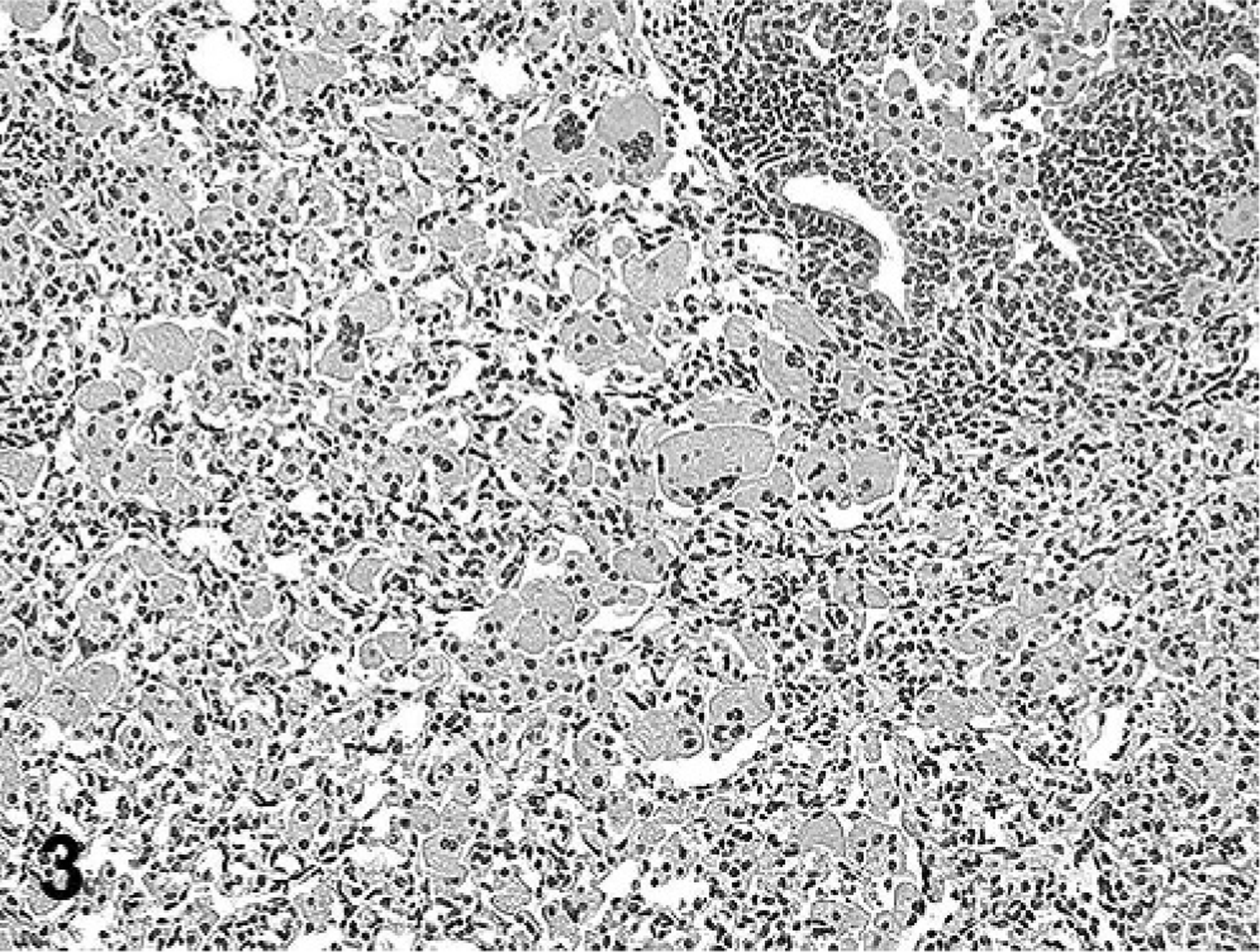
Lung; 129S4/SvJae mouse. There is a marked inflammatory infiltrate filling alveoli, composed predominantly of large eosinophilic macrophages distended with large amounts of intracytoplasmic eosinophilic crystalline material. HE.
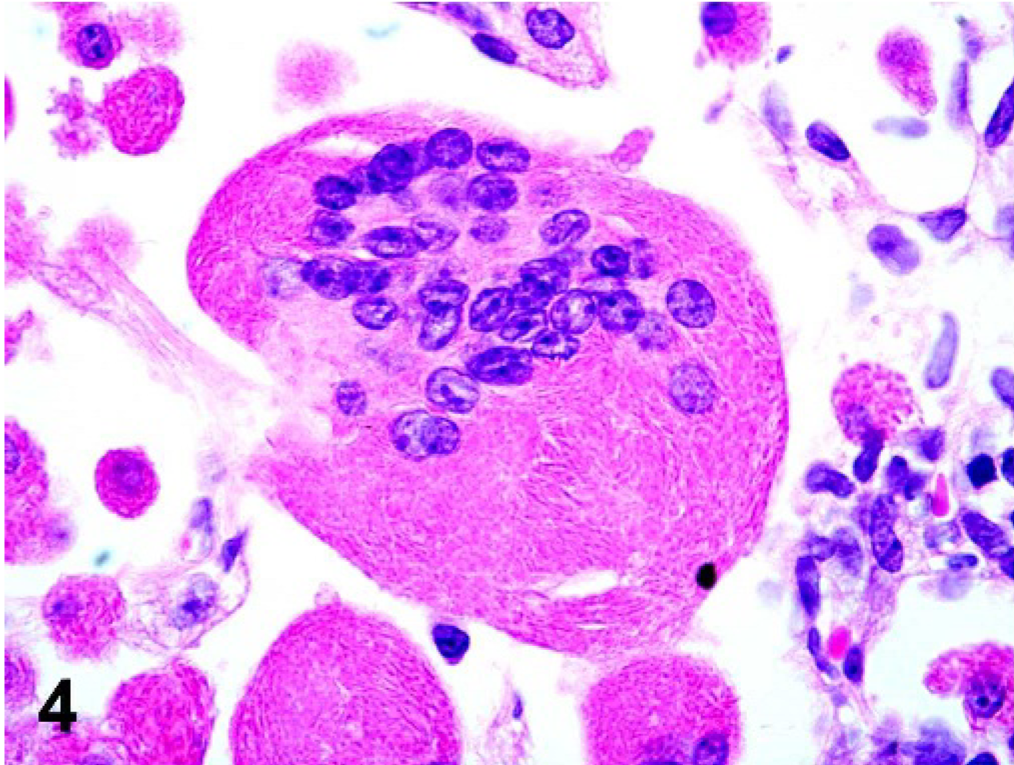
Lung; 129S4/SvJae mouse. The cytoplasm of a multinucleate giant cell is distended with fine acicular eosinophilic crystalline material. HE.
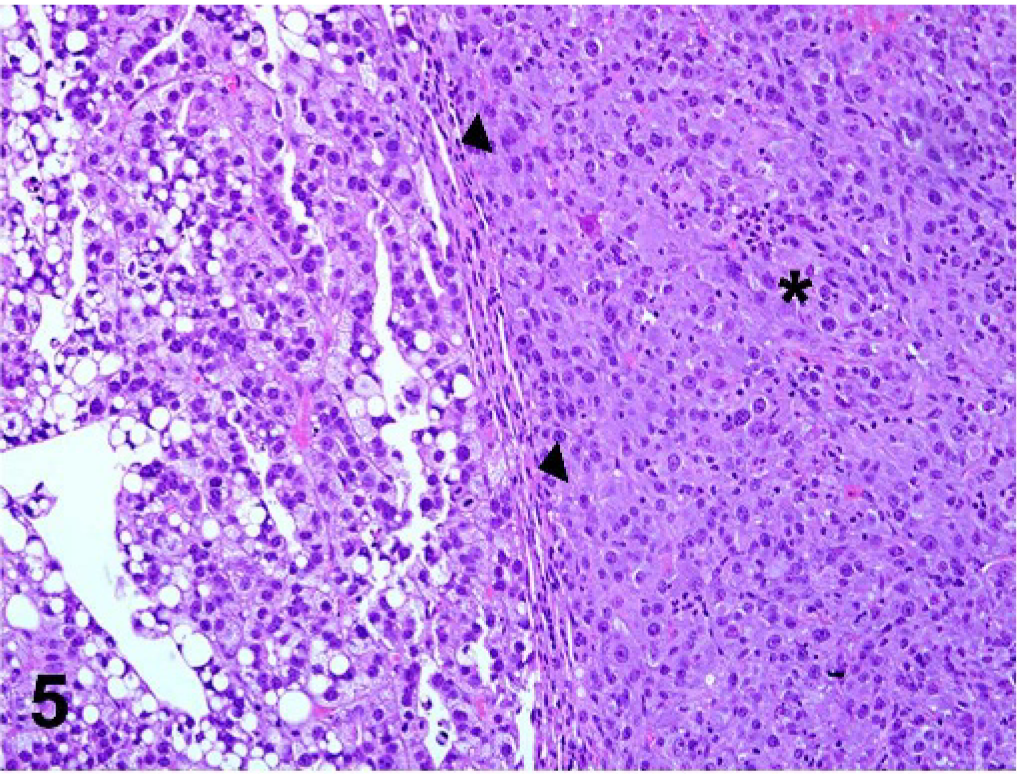
Lung; 129S4/SvJae mouse. Pulmonary adenoma (arrowheads); there is a moderate to severe inflammatory infiltrate (asterisk) composed predominantly of large eosinophilic macrophages with few to no crystals, surrounding a pulmonary adenoma. HE.
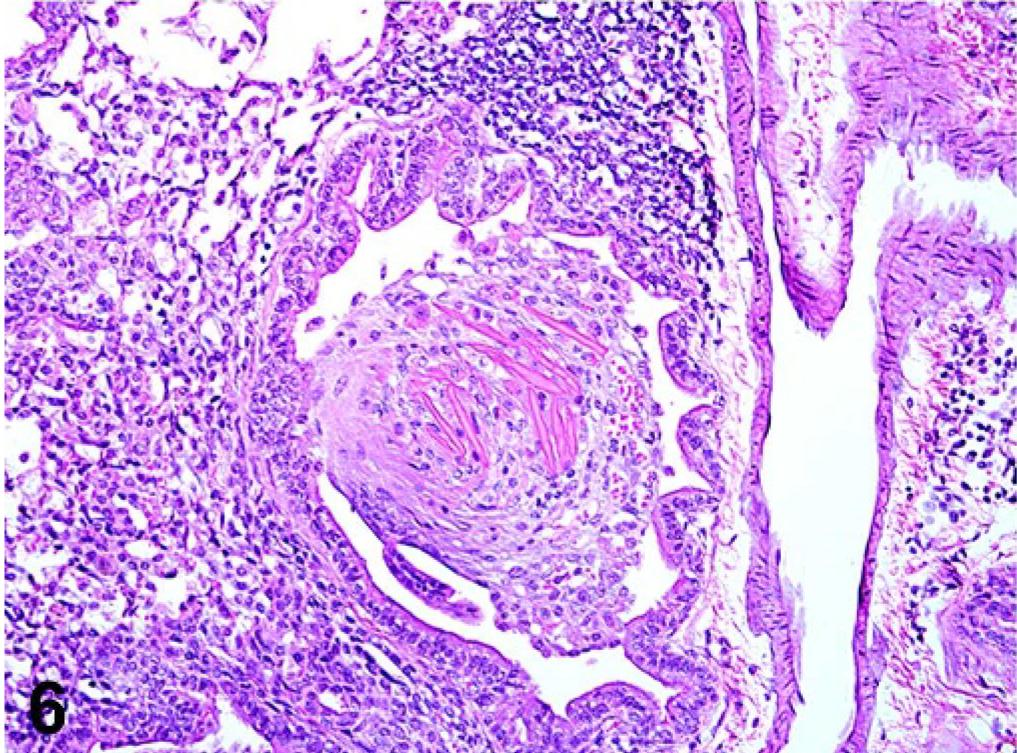
Lung; 129S4/SvJae mouse. Large rectangular intrabronchiolar eosinophilic crystals distend airways causing bronchiolitis obliterans, associated with variable intra-alveolar and intrabronchiolar inflammatory macrophage and neutrophil infiltrates. HE.
Histopathologic findings in a series of 94 aging 129S4/SvJae mice.
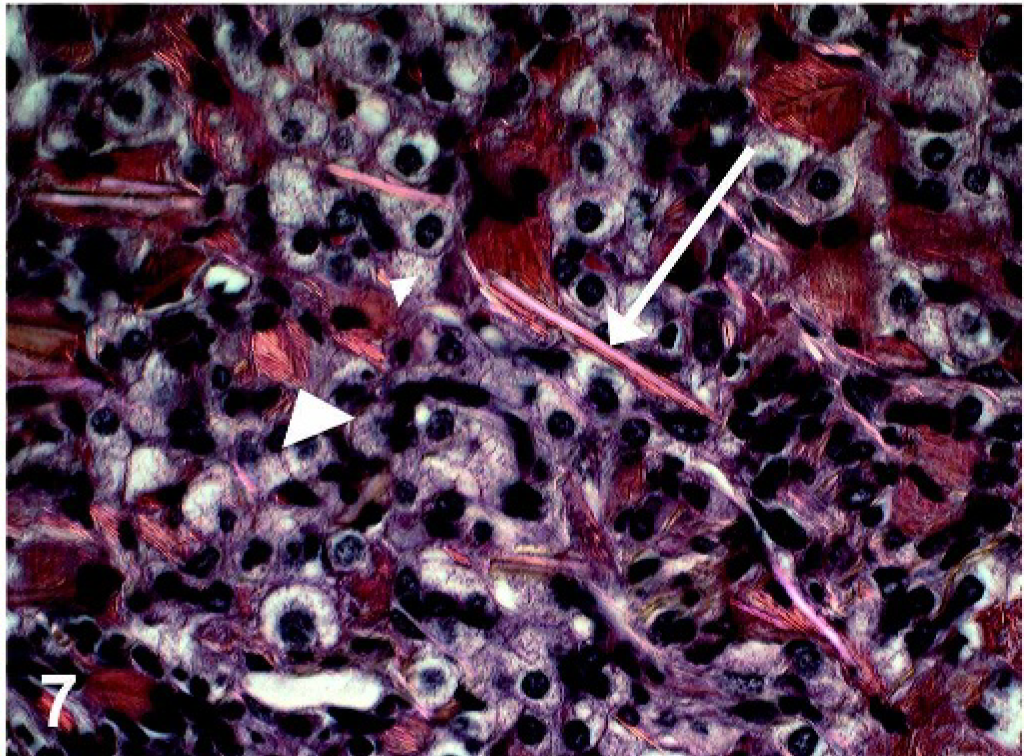
Under polarized light, crystals were variably birefringent, with the largest extracellular crystals exhibiting the most birefringence and less intense birefringence noted from intrahistiocytic crystals (Fig. 7). Luna histochemical stain for eosinophils demonstrated bright red staining of large extracellular crystals, as well as acicular intrahistiocytic crystalline material (Fig. 8). Immunohistochemically, antibodies to Ym1 protein showed positive immunoreactivity restricted to intrahistiocytic crystalline material and extracellular large rectangular and fine acicular crystals (Fig. 9). When using TEM, intracellular crystals were characterized by electron dense, rectangular, curved, or rhomboid structures with parallel walls and often with pointed ends (Fig. 10).
Lung; 129S4/SvJae mouse. Under polarized light, eosinophilic intracellular (arrowhead) and extracellular (arrow) crystals are partially birefringent. HE.
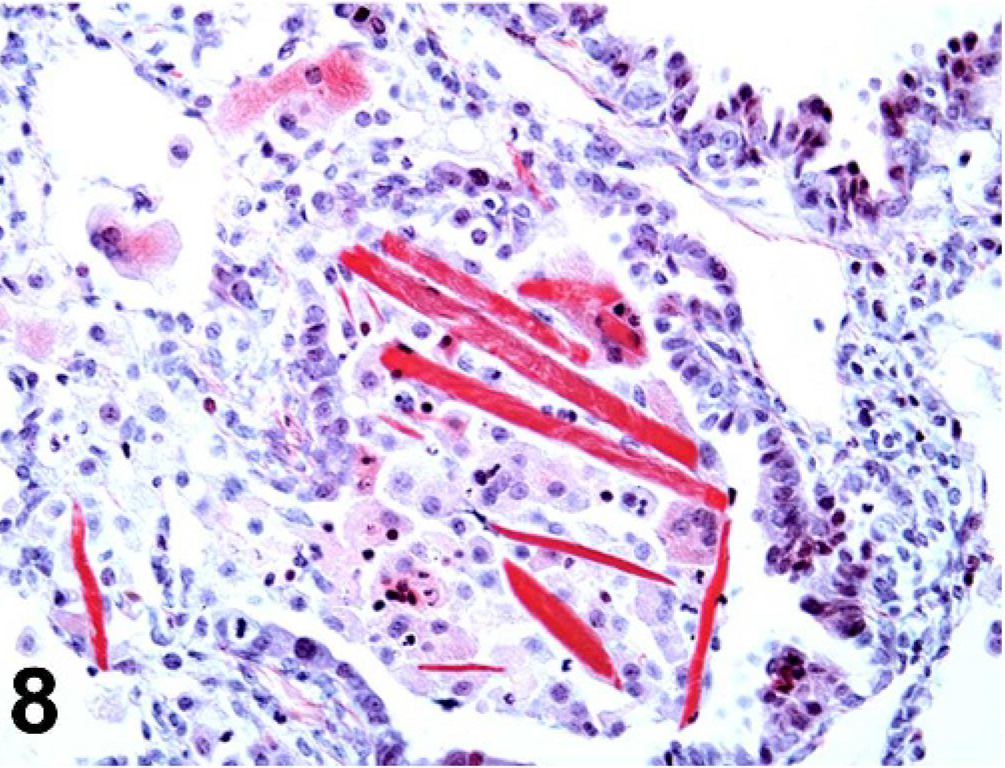
Lung; 129S4/SvJae mouse. Large extracellular eosinophilic crystals stain bright red with Luna stain, whereas fine intrahistiocytic crystalline material stains faint red. Luna histochemical stain.
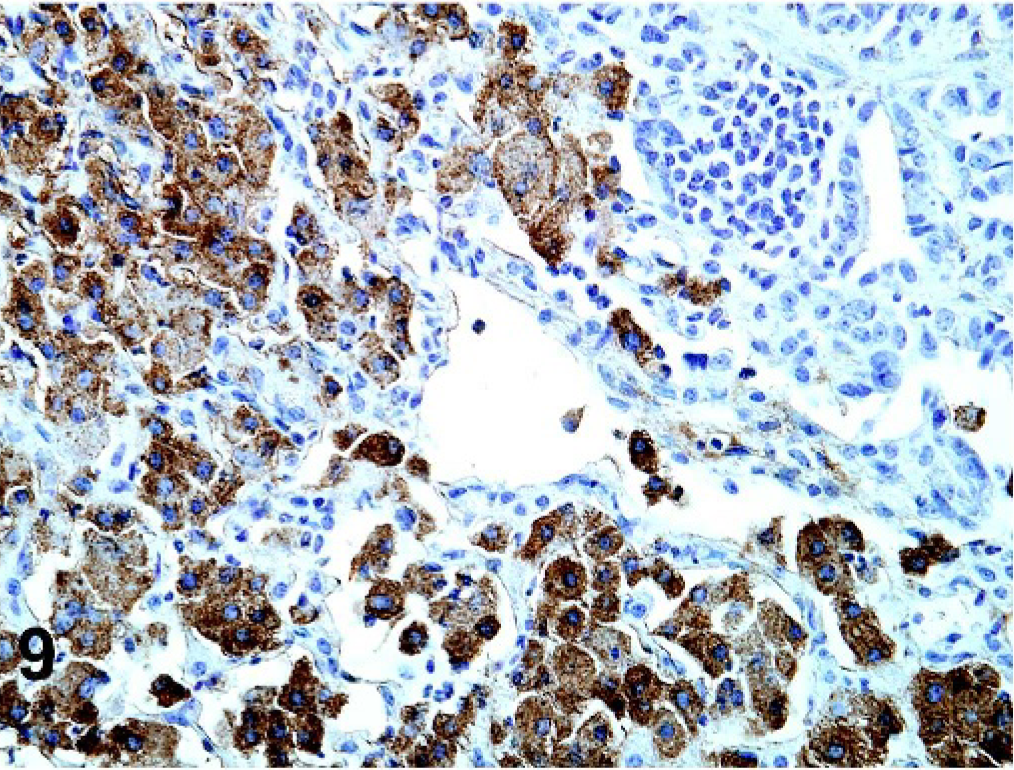
Lung; 129S4/SvJae mouse. There is positive immunoreactivity to antibodies against Ym1 protein in both large extracellular and fine intracellular crystalline material. Anti-Ym1 antibody. Counterstain = Meyer's hematoxylin.
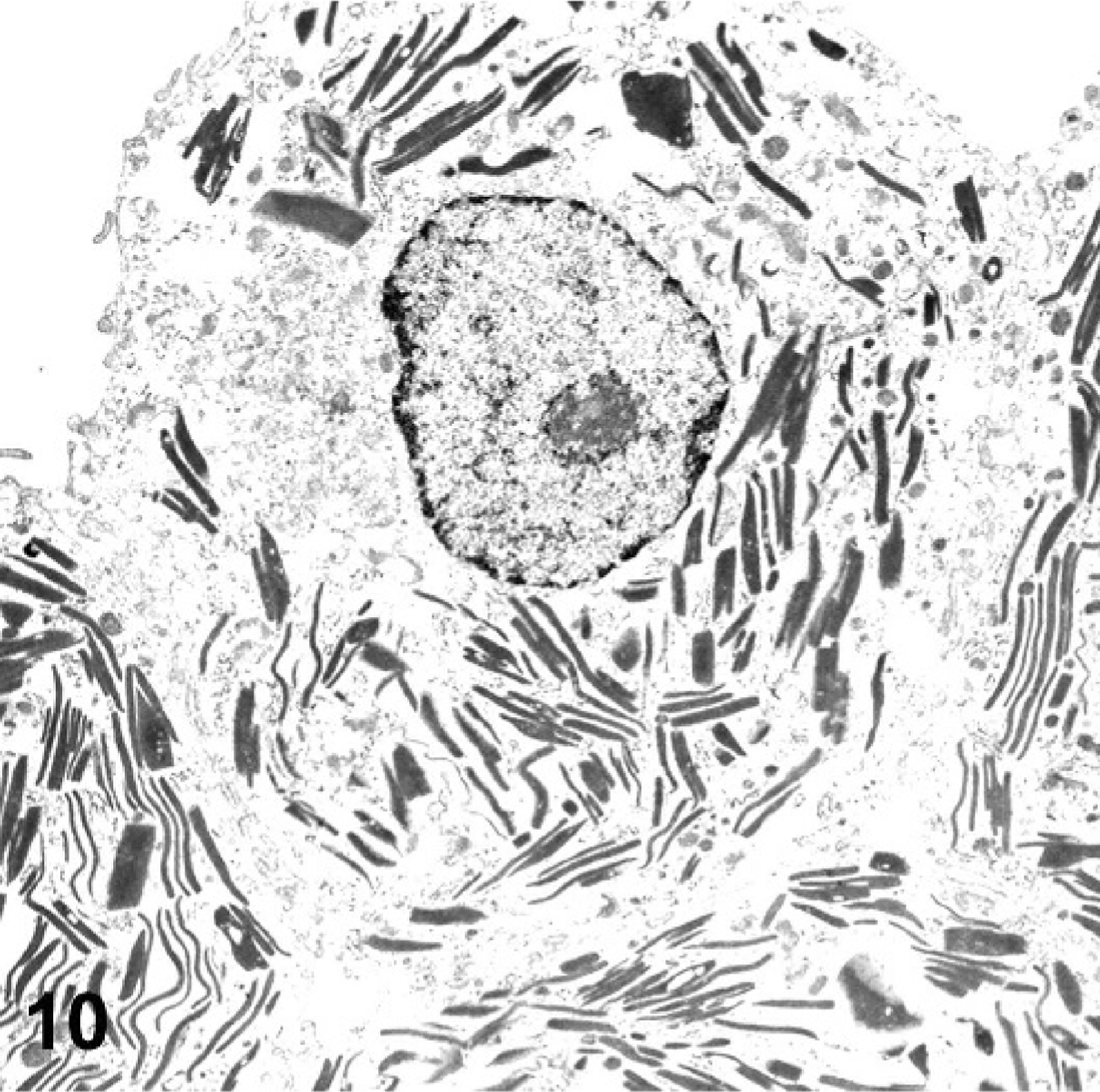
TEM; Lung; 129S4/SvJae mouse. Intrahistiocytic crystalline material is characterized by markedly electron dense, amorphous, rectangular to curved material, ranging from 100 to 200 nm in diameter, with parallel walls and blunted to pointed ends. Uranyl acetate.
Discussion
Laboratory mice used in transgenic mouse model development express phenotypic background lesions, depending on their strain of origin. Certain strains, such as the C57BL/6 strain 9, 16, 17 and its derivatives (p47phox knockout mouse, 6 motheaten [Ptpn6^me] mouse 2, 9, 13, 14 ) have a higher incidence of this lesion than in other strains. Also, commonly affected are mice with recessive mutations affecting the immune system, such as severe combined immunodeficiency (SCID) mice 4 and knockout models targeting components of the immune system, such as the CD40L knockout model and alpha/beta T cell receptor knockout model. 4, 6
In addition, eosinophilic crystalline pneumonia may occur in mice with concurrent pulmonary disease. 2, 4, 14 Eosinophilic crystalline pneumonia has been associated with infectious, neoplastic, hypersensitivity, and lymphoproliferative diseases. Eosinophilic crystalline pneumonia has been reported to occur concurrently with pulmonary adenomas; 4, 8, 16 parasitic and fungal diseases, such as pulmonary pneumocystosis, nematodiasis, and cryptococcosis; 2, 4, 8, 16 and mouse models of hypersensitivity or allergic diseases, particularly asthma. 4, 6, 19 Several cases of eosinophilic crystalline pneumonia have been reported in mouse models of allergic airway disease used by exposure to egg albumen (J. M. Ward, personal communication), as well as BALB/c mice sensitized and challenged with ovalbumin. 19 Upregulation of Ym1 protein in association with eosinophilic crystalline pneumonia has also been reported with a number of allergic, infectious, and neoplastic diseases. 4, 6, 11, 17, 19
The characteristic crystals associated with eosinophilic crystalline pneumonia were previously believed to be Charcot-Leyden crystals, unique to humans and non-human primates, which occur in conjunction with eosinophilic-rich diseases, such as helminth infestations and asthma. 2, 4, 8, 9 However, the primary component of Charcot-Leyden crystals is lysophospholipase, an autocrystallizing protein of eosinophils and basophils related to carbohydrate-binding galectins. 4, 6, 17, 18 In contrast, the primary component of eosinophilic crystals in eosinophilic crystalline pneumonia is Ym1 protein, also called T-lymphocyte dependent eosinophilic chemotactic factor, 4, 11, 17 a member of the chitinase family of proteins of which the function is not currently known. 8, 10, 11, 17 This protein is implicated in processes such as host defense, 2, 4, 7 cell-to-cell and cell-to-matrix interactions, 10, 11 and hematopoiesis. 10, 11 There are currently 4 known Ym proteins, Ym1, Ym2, Ym3, and Ym4. 4 These proteins are chitinaselike proteins 6, 8 but do not possess any chitinase enzymatic activity. 10, 17 Because Ym1 has been shown to have 95% sequence identity with Ym2, there may be some antibody cross-reactivity; however, when using mass spectrometry, pulmonary crystals have been determined to be predominantly composed of Ym1 protein, 4 and differential tissue expression between Ym1 and Ym2 has been shown; Ym1 is expressed in high levels in lung and spleen but not stomach, and Ym2 is expressed highly in stomach but not lung or spleen. 16
The function of Ym1 is unknown; however, several characteristics of the protein suggests its important roles in hematopoiesis, tissue remodeling, or immune responses. The protein is somewhat chemotactic for eosinophils, 2, 6, 8, 16 stored primarily in neutrophil granules 6, 8, 11 and to a lesser extent in macrophages, in which it is elicited in Th2-mediated immune responses. 1, 6, 10 Because of its chitinaselike structure, Ym1 protein has been implicated in immune responses to organisms that contain chitin, such as fungal and parasitic organisms. 4, 7, 10, 16 Macrophages activated by type 2 cytokines, particularly interleukin (IL) 4 and IL-13, produce large amounts of Ym1, 8, 10, 17 resulting in extravasation of eosinophils and recruitment of T cells in response to parasitic infections. 16 It has been suggested that the homology of Ym1 with other chitinase family members implicates the ability of Ym1 to bind with glycosaminoglycans such as heparin and heparan sulfate on the surface of cells or in the extracellular matrix, implying cell-to-cell or cell-to-matrix regulation of cell migration, adhesion, and differentiation. 7, 11, 17 Binding specificity toward these extracellular matrix constituents strongly implies a role in tissue repair. 10 Lastly, Ym1 has a possible functional role in hematopoiesis. As in the lung, Ym1 protein is expressed constitutively in adult hematopoietic tissues, such as bone marrow and spleen. 7, 11 Immature neutrophils and macrophage progenitors in the bone marrow and spleen reportedly express Ym1 protein. 11 Needle-shaped crystals with Ym1 immunoreactivity have been reported in the cytoplasm of macrophages forming erythroblastic islands in the bone marrow, and immature neutrophils show immunoreactivity to Ym1 protein, which they lose over time with maturation. 11 In addition, murine fetal and neonatal liver, a prominent site of extramedullary hematopoiesis, contains Ym1 immunoreactive cells, while adult murine liver does not. 11 Interestingly, in a mouse model of myelogenous leukemia with hyaline droplet nephropathy, the intracellular and intratubular protein was determined to be Ym1 protein, implicating the circulating and tissue infiltrating tumoral myeloid cells in the production of the protein. 8
In conclusion, eosinophilic crystalline pneumonia is an idiopathic pulmonary lesion that occurs sporadically in some laboratory mouse strains and with high incidence in the 129S4/SvJae strain. In contrast to previous studies on eosinophilic crystalline pneumonia, the histopathologic results of this retrospective study suggest that eosinophilic crystalline pneumonia may not represent a single entity but rather a histopathologic feature or range of features common to at least 3 separate etiologies. In addition, this study characterizes the immunohistochemical and electron microscopic features of the pulmonary and intrahistiocytic crystals that form an important component of this lesion in 129/SvJae mice. The characteristic crystals accompanying the lesion are composed of Ym1 protein, a chitinaselike protein that has been implicated in a number of biological processes such as host immune defense, cell-to-cell and cell-to-matrix interactions, and hematopoiesis. Eosinophilic crystalline pneumonia is a background lesion commonly in aged mice of the 129SvJae strain, which may confound studies of aged transgenic or knockout mice that are engineered from this strain. Investigators designing a genetic construct on this background must be aware of this strain-related lesion, particularly when designing studies either based on the effects of aging, or those based on pulmonary pathophysiology. In addition, the embryonic stem-cell line J1 originates from the 129/SvJae strain, 11 so eosinophilic crystalline pneumonia may be expected in genetically engineered mice originating from these embryonic stem cells. The laboratory animal veterinarian and the veterinary pathologist must be aware of this background strain related lesion and be able to identify it as “nonlesion” not associated with the effects of the knockout or transgene when characterizing these models. Such knowledge pertaining to background lesions in laboratory mice will help prevent mischaracterization of transgenic mouse models as they relate to human disease. Knowledge and familiarity with laboratory strains and their association with eosinophilic crystalline pneumonia is critical, and caution must be adopted before a pulmonary phenotype is assigned to a genetically engineered mouse.
Footnotes
Acknowledgements
Supported, in part, by a National Institute of Allergy and Infectious Diseases contract to SoBran, Inc, a National Cancer Institute (NCI) contract to SAIC Frederick, and the Intramural Research Program of the National Institutes of Health (NIH), Office of Research Services, NCI, Center for Cancer Research, where MJH is a Molecular Pathology Graduate Fellow in the NCI and Michigan State University Graduate Partnership Program. We are thankful for the excellent histotechnology assistance of Cindy Erexson and Larry Faucette, electron microscopic expertise by Patricia Zerfas (Division of Veterinary Resources, Office of Research Services, NIH), and aid of Dr. Shioko Kimura and Frank Gonzalez. We would like to thank Dr. Mark Simpson for assistance with publication funding.
