Abstract
A 17-year-old Quarter horse mare was referred to Cornell University for postmortem examination after 72 hours of encephalopathy that consisted of depression, mania, and blindness. A plasma sample and cerebral spinal fluid demonstrated hyperammonemia. Gross necropsy examination findings included the following: mild icterus, a transmural mass in the glandular portion of the gastric fundus, multiple masses throughout the liver, and a large tumor thrombus in the portal vein. Microscopically, the gastric mass, hepatic masses, and portal vein thrombus were composed of similar neoplastic epithelial cells that formed variably sized acini and branching cords separated by a dense desmoplastic stroma. Throughout the cerebral frontal cortex were numerous Alzheimer type II astrocytes. Hepatic encephalopathy was caused by gastric adenocarcinoma, with metastasis to the liver and the portal vein. The clinical and pathologic lesions from this unique case, as well as hyperammonemia and portal vein thrombosis in the pathogenesis of hepatic encephalopathy, are discussed.
Gastrointestinal adenocarcinoma is rare in the horse, with the majority of reported cases involving the colon. 1 In a review of equine neoplasia, Cotchin 4 cites a Dutch report published in 1941 by Thoonen and Ide, which, to our knowledge, is the only primary equine gastric adenocarcinoma reported in the literature. The majority of the reported cases of intestinal adenocarcinoma in horses resulted in chronic colic. Metastasis was common, primarily involving the mesenteric lymph nodes, lungs, or liver. 15 Recently, an intestinal adenocarcinoma in a horse was associated with neurologic signs, including hind-limb paresis and hyporeflexia secondary to extension of sublumbar lymph-node metastasis into the vertebral canal. 15
Signs consistent with hepatic encephalopathy have classically been associated with Theiler's disease (serum sickness) in adult horses or with hepatic failure from toxins (e.g., urea poisoning, pyrrolizidine alkaloids, and alsike clover). 12 Other, less frequent, causes of encephalopathy include uremic encephalopathy secondary to renal failure, a recently recognized idiopathic hyperammonemia syndrome similar to transient hyperammonemia of adult humans, and an idiopathic hyperammonemia syndrome associated with Clostridium sordelli infection. 3, 6, 8, 13 An acquired portosystemic shunt because of portal vein thrombosis, to our knowledge, has been described only once in the horse. 2 In a previous study that reported tumor metastasis to the liver in 5 horses, 1 horse developed clinical signs of hepatic encephalopathy. 16 In the case presented, hepatic metastasis, with portal vein tumor thrombosis, resulted in clinical signs of hepatic encephalopathy.
The horse had been in good health until 72 hours before death when the mare had acute encephalopathy. The horse was vaccinated annually against rabies and Eastern and Western equine encephalomyelitis, and had never received equine antiserum.
On initial examination, the horse was afebrile but severely depressed, head pressing, and blind. Symptomatic treatment with polyionic intravenous fluids, broad spectrum antibiotics, and anti-endotoxic doses of flunixin meglumine did not result in improvement. The mare became progressively more encephalopathic over the following 72 hours, displaying intermittent periods of severe depression and extreme maniacal behavior that could not be adequately controlled by sedation with either opioids or α2 agonists. The mare was euthanized on humane grounds 72 hours after encephalopathic signs were first noted.
Routine hematology and serum biochemistry profiles performed approximately 6 hours before euthanasia demonstrated a stress leukogram, and slight elevations in the hepatocellular enzyme sorbitol dehydrogenase (19.4 IU/l; normal range, 2–11 IU/l), the hepatobiliary enzymes gamma-glutamyl transpeptidase (55 IU/l; normal range, 8–33 IU/l), and alkaline phosphatase (308 IU/l; normal range, 75–220 IU/l). The total serum bilirubin was elevated, at 3.7 mg/dl (normal range, 0.7–2.6 mg/dl), with the unconjugated fraction comprising 95% of the total value. Other findings included hyperglobulinemia (4.7 g/dl; normal range, 2.7–4.0 g/dl), hyperglycemia (153 mg/dl; normal range, 75–117 mg/dl), and a mild increase in serum activity of creatine kinase (1016 IU/l; normal range, 93–348 IU/l). The plasma ammonia level was 148 µmol/l. The plasma ammonia level of a healthy control horse measured simultaneously was 12 µmol/l. The published normal range of blood ammonia in horses is 8–63 µmol/l. 11 In a review of 50 equine cases of primary hepatic disease, 18 horses presented with hepatic encephalopathy. 14 All of these horses had plasma ammonia levels higher than 90 µmol/l with one half of these encephalopathic cases also having hyperglycemia. The ammonia level in a sample of cerebrospinal fluid from the current case, obtained at postmortem was 86 µmol/l. Normal cerebral spinal fluid ammonia levels are presumed to be similar to plasma levels, however, data in horses are not available.
At necropsy, the mare was in good nutritional condition, with minimal autolysis and moderate body-fat stores. The oral and conjunctival mucous membranes, sclera, and subcutaneous fat were pale yellow. Numerous contusions were present on the head, predominately near the eyes, and there was bilateral hyphema. The peritoneal cavity contained 1–2 liters of yellow-orange, turbid serosanguineous fluid with numerous fibrous and fibrinous tags adherent to the serosal surfaces of the viscera.
Within the mucosa of the greater curvature of the stomach, there was a locally extensive 2 × 3 cm firm, round, pink, pitted area rimmed by red, elevated nodular tissue, forming an overall stellate-shaped mass measuring 6 × 7 × 2 cm. The mass extended transmurally to a locally extensive 1.2 × 2.7 cm, firm, white, collagenous serosal plaque (Fig. 1).
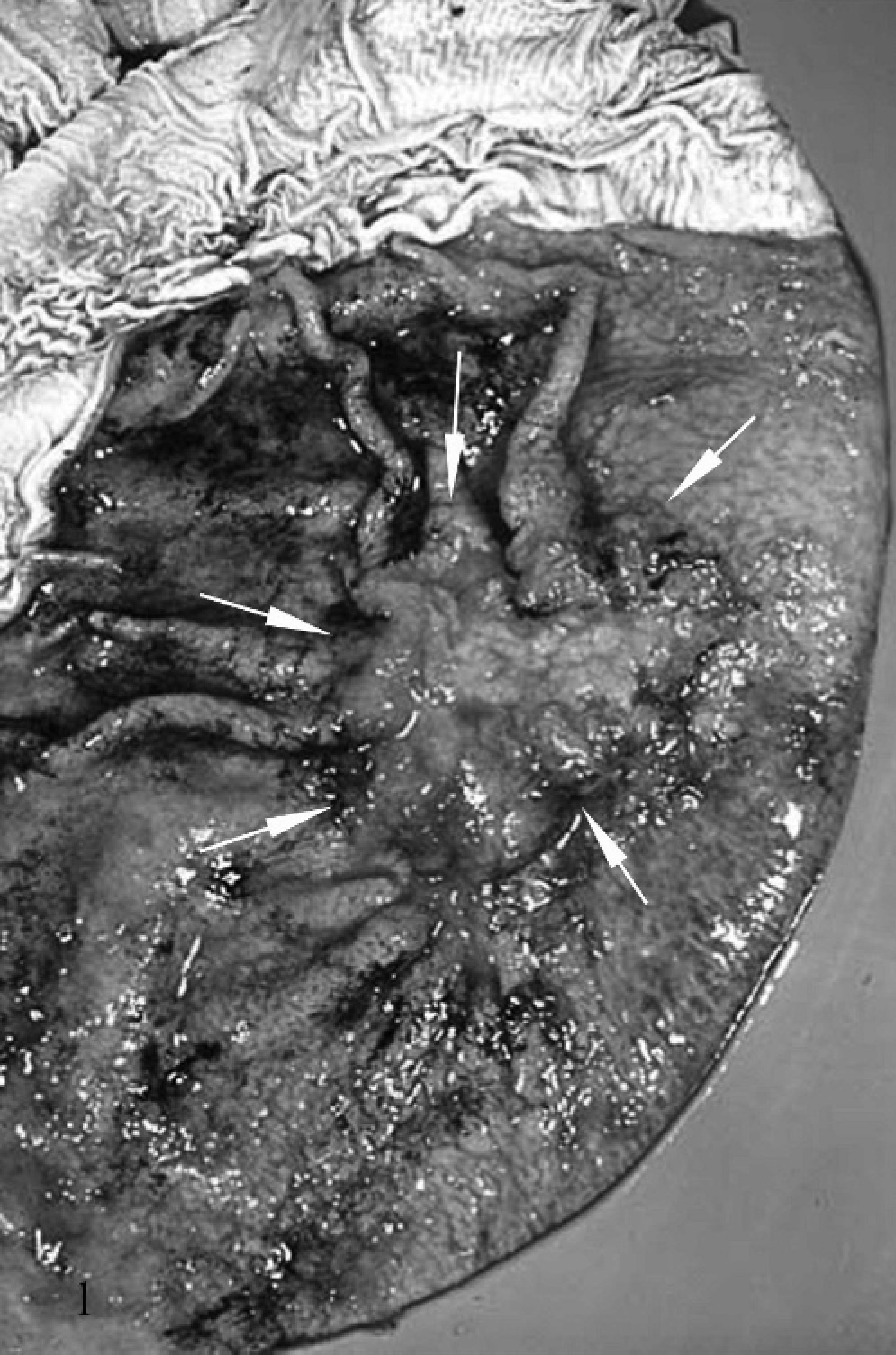
Stomach; 17-year-old Quarter horse mare No. N97-454. The mucosal surface of the opened stomach with a locally extensive, firm, stellate, transmural mass within the glandular portion (arrows).
Along the free edge of the attachment of the triangular ligament to the right liver lobe, there was a botryoid cluster of multiple 1- to 3-cm-diameter coalescing, firm, white round to oval nodules, which extended from the capsular surface of the liver into the parenchyma. On section, the masses were round, with multiple small nodules throughout each mass. Scattered similar nodules were present within the right medial and left lateral liver lobes (Fig. 2).
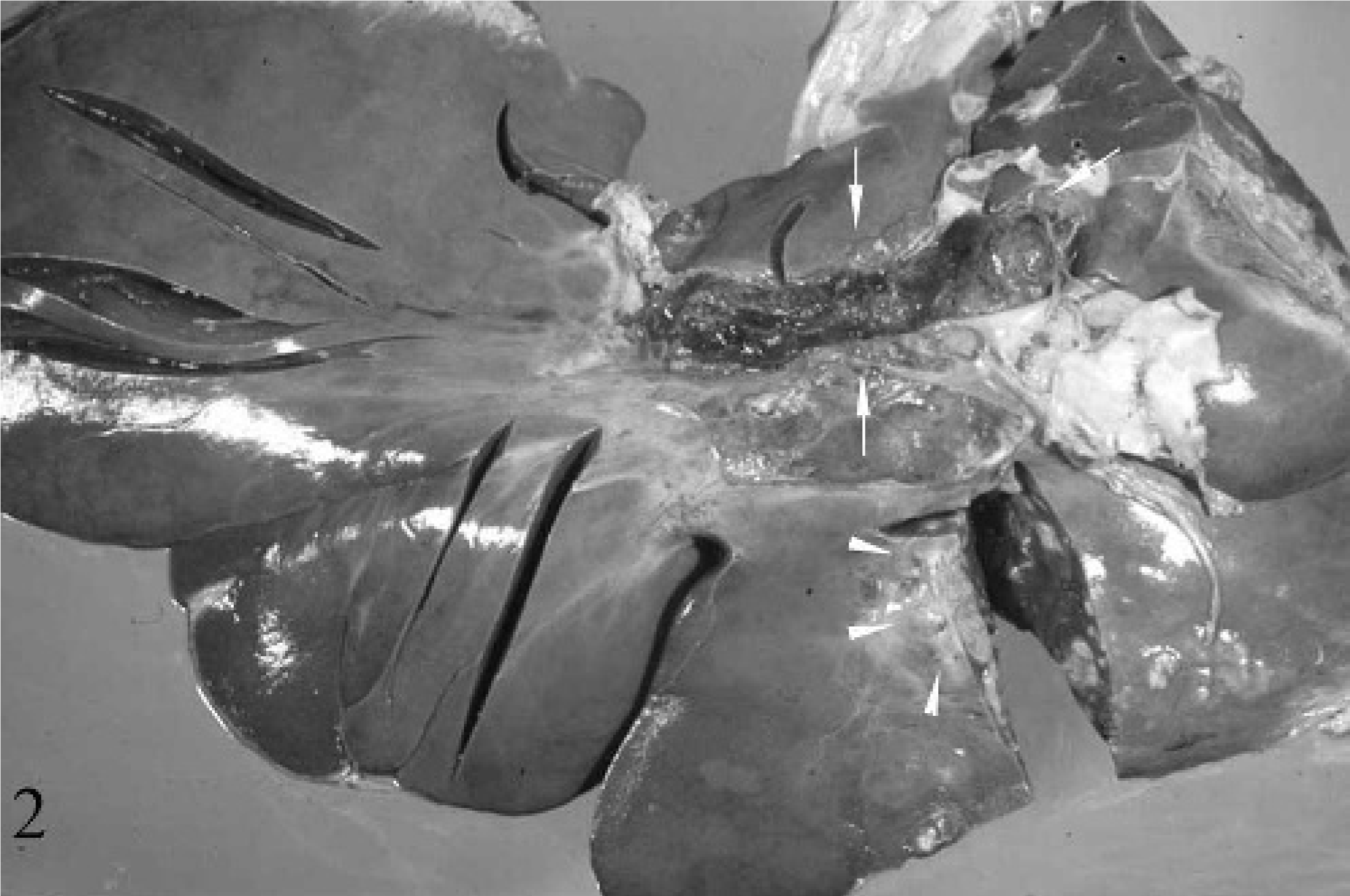
Liver; 17-year-old Quarter horse mare No. N97-454. The portal vein (arrows) was occluded by a large, dark red, firmly adhered tumor thrombus that extended from several, multifocal, stellate, pale tan to yellow, firm masses within the right lateral liver lobe (arrowhead).
At the liver hilus, the portal vein was filled with an approximately 4-cm-diameter, 23-cm-long, organized, variegated, and firmly adhered thrombus (Fig. 2). The mass in the left lateral liver lobe extended into the portal vein, obliterating the vessel wall at the distal aspect of the thrombus. The brain was grossly normal.
Sections of liver, portal vein, kidney, stomach, heart, lung, anterior chamber of the eye, and several sections of brain were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned routinely at 4–6 µm, and stained with hematoxylin and eosin. The hepatic mass, portal thrombus, and gastric mass were also stained with alcian blue, periodic acid–Schiff reagent (PAS), and routine immunohistochemical procedures by using antibodies to cytokeratin (Boehring-Mannhein, monoclonal, trypsin pretreatment, 1 ° 100) and vimentin (Dako, monoclonal, trypsin pretreatment, 1 ° 40). Immunohistochemistry with antibodies to glial fibrillary acidic protein (GFAP) (Dako, polyclonal, no pretreatment, 1 ° 3,000), vimentin (Dako, monoclonal, trypsin pretreatment, 1 ° 40), and S-100 (Dako, polyclonal, no pretreatment, 1 ° 200) were performed on a single section of frontal cortex.
On histopathologic examination, the gastric wall was distorted by a transmural, desmoplastic mass with central expansion by disorganized, often radiating columns of acini and small clusters of epithelial cells. The cells were pseudostratified to simple columnar, with finely stippled euchromatic oval nuclei and a single prominent central nucleolus (Fig. 3).
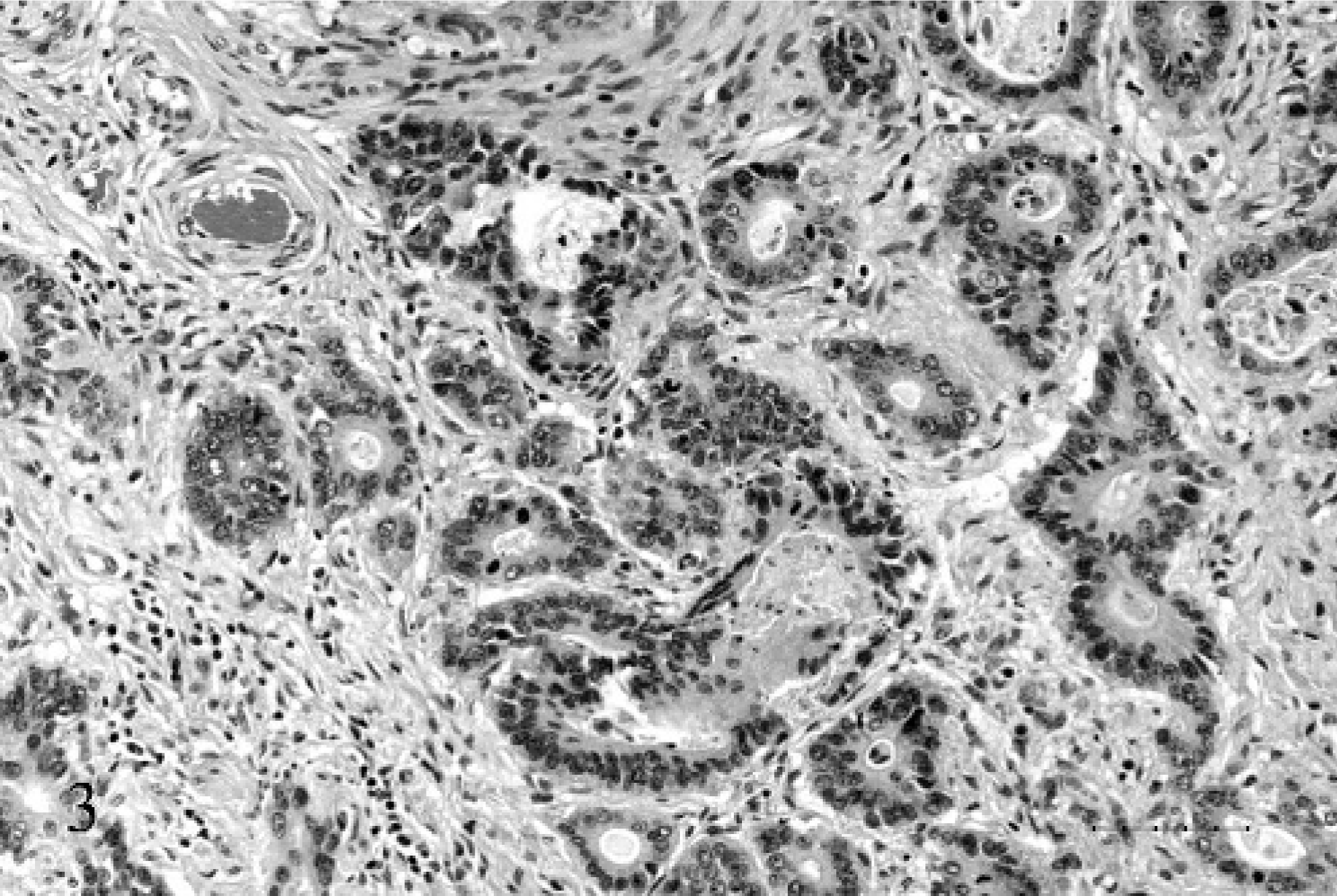
Stomach; 17-year-old Quarter horse mare No. N97–454. The mass is composed of variably sized acini and elongate columns of epithelial cells separated by a desmoplastic and multifocally necrotic stroma. HE. Bar = 50 µ.
They had scant to abundant eosinophilic cytoplasm, with a prominent brush border, and occasional cells contained vacuoles filled with mucin that stained positively with PAS and alcian blue. There were 0–4 mitoses/400× high power field. The acini were filled with necrotic debris and mucus. Similar material was adherent to the luminal surface. The acini were supported by a desmoplastic stroma that formed a central plaque on the serosa and that was multifocally infiltrated with lymphocytes and plasma cells.
Expanding the portal vein and obliterating the lumen, was a mass composed of similar fronds and acini of epithelial cells, admixed with hemorrhage, cellular debris, and fibrin (Fig. 4). The mass was anchored to the vessel wall at 1 edge and was obliterating the intima and the endothelium. The epithelial cells were supported by an interlacing network of fibrovascular stroma, which was largely obliterated by neutrophils and cellular debris (Figs. 5, 6).
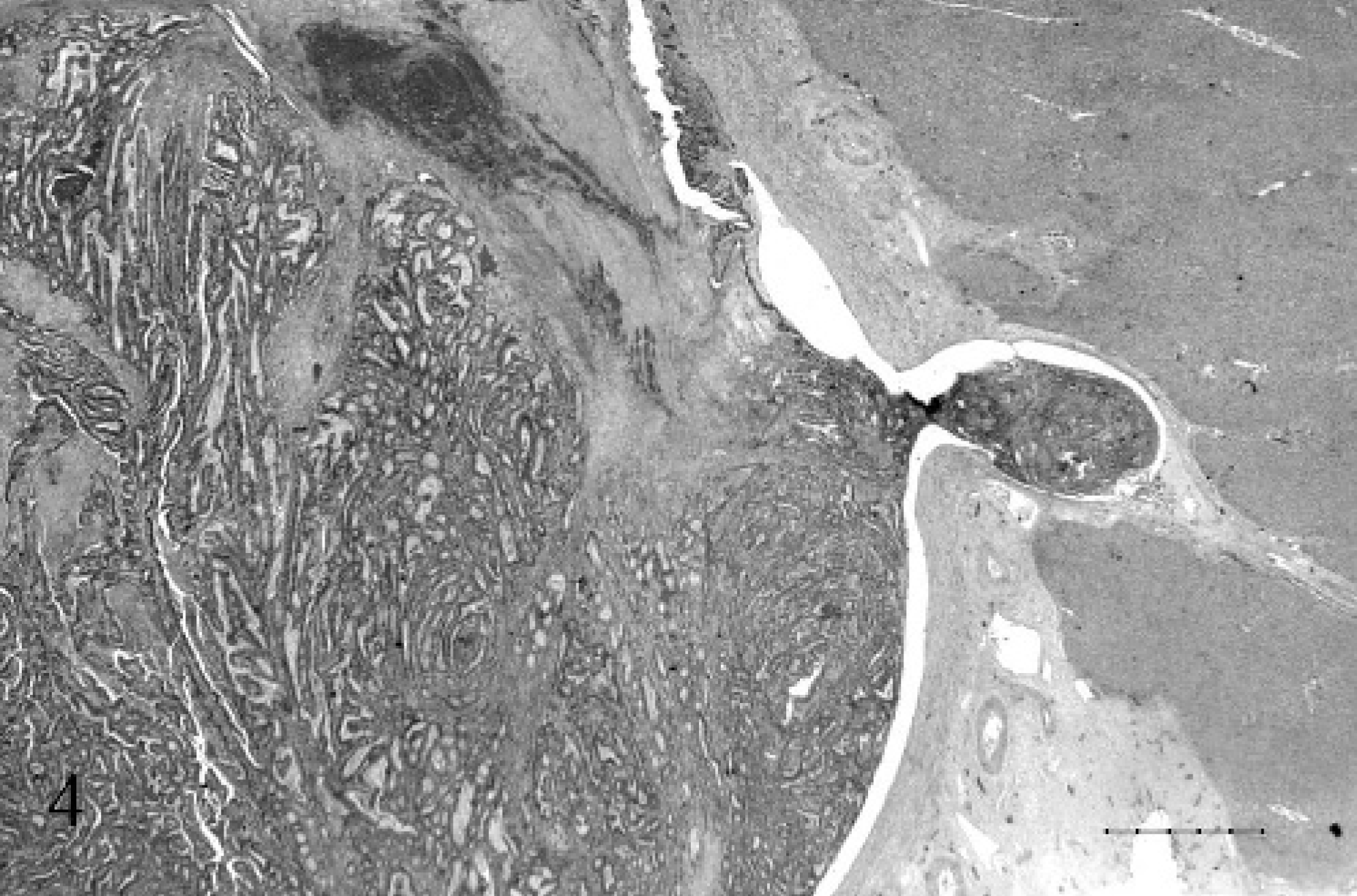
Liver and portal vein; 17-year-old Quarter horse mare No. N97–454. The portal vein was completely occluded by a firmly adhered tumor thrombus. HE. Bar = 1000 µ.
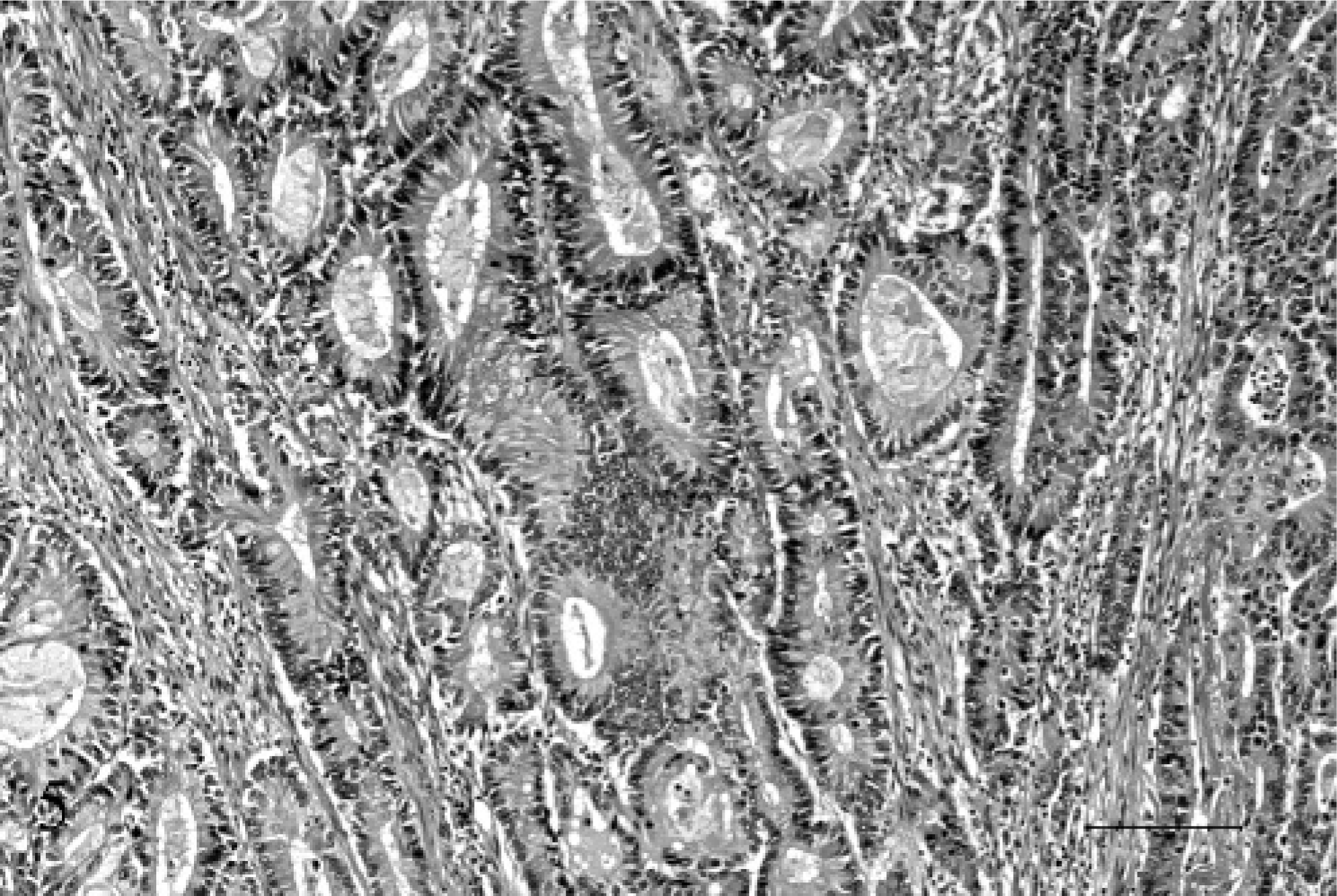
Portal vein; 17-year-old Quarter horse mare No. N97–454. The thrombus was composed of columnar epithelial cells forming acini, separated by a fine interlacing network of fibrovascular stroma, which was multifocally necrotic and infiltrated by neutrophils admixed with cellular debris. At one edge, the mass was anchored to the vessel wall and was obliterating the intima and endothelium. HE. Bar = 100 µ.
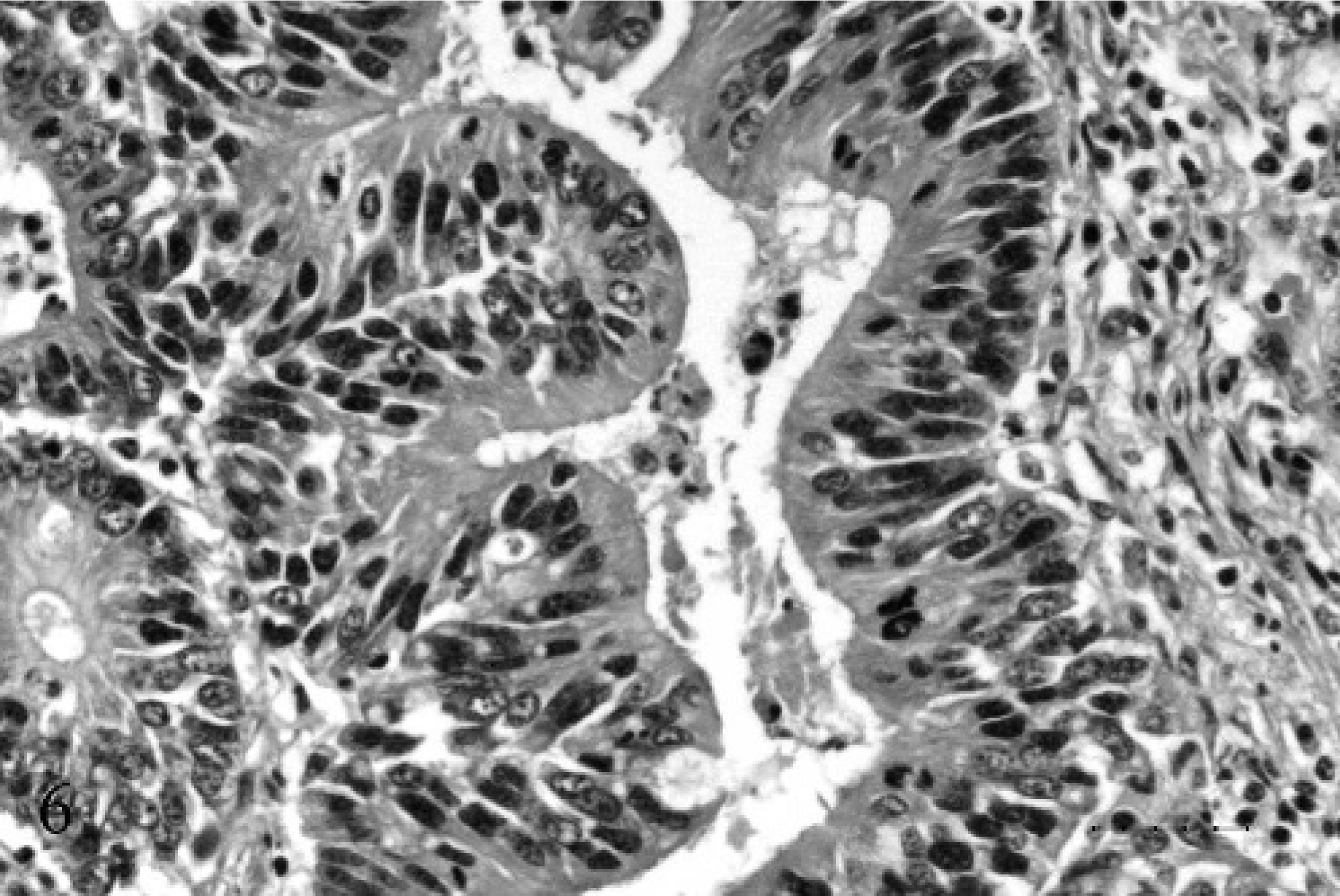
Portal vein; 17-year-old Quarter horse mare No. N97–454. The cells were pseudostratified to simple columnar, with finely stippled euchromatic to hyperchromatic oval nuclei and a single prominent central nucleolus. They had abundant eosinophilic cytoplasm and occasional cells contained vacuoles filled with PAS and alcian blue positive mucin. HE. Bar = 25 µ.
Within the stomach, approximately 40–50% of the neoplastic epithelial cells were strongly positive for cytokeratin, approximately 30–40% were moderately positive, and the remaining 10–20% were weakly positive or negative. In the portal vein, <10% were weakly positive or negative for cytokeratin and >90% were moderately to strongly positive. The supporting fibroblastic stromal cells were uniformly strongly positive for vimentin; however, the neoplastic epithelial cells were negative for vimentin.
Within the frontal-lobe gray matter, there were Alzheimer type II astrocytes characterized by clusters of 2–4 swollen, 20- to 30-µm-diameter open-faced irregular oval to indented astrocytes (Fig. 7). These cells were negative to weakly positive for GFAP, negative for vimentin, and negative for S-100.
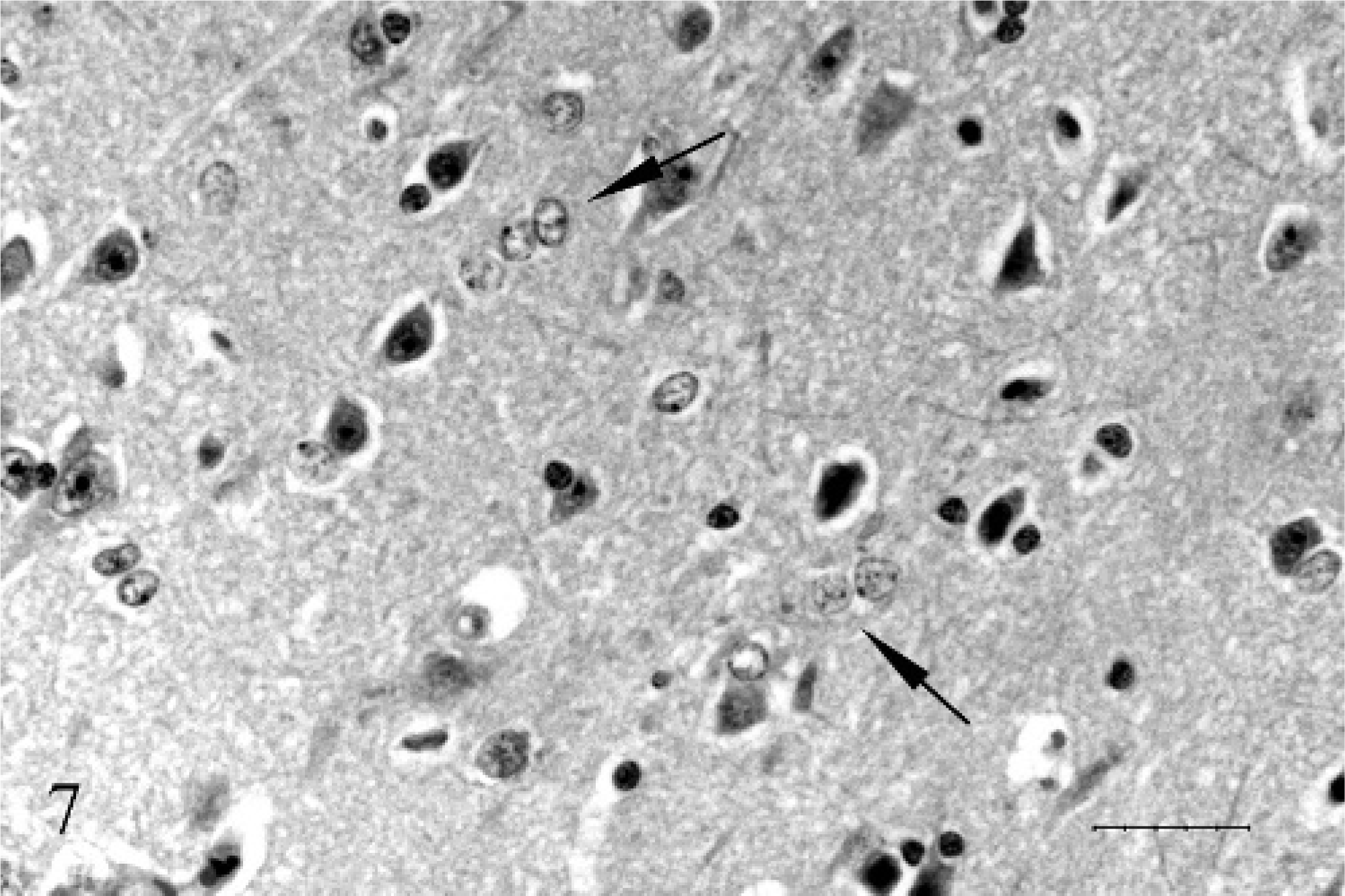
Frontal lobe gray matter; 17-year-old Quarter horse mare No. N97–454. The astrocytes are swollen 20- to 30-µm-diameter open-faced irregular oval to indented and form clusters of 2 to 4 (Alzheimer type II astrocytes). HE. Bar = 25 µ.
Squamous-cell carcinoma is the most frequently reported neoplasm originating in the equine stomach. Rarely, other masses have been reported, including a leiomyosarcoma and a hyperplastic polyp. 1 With all tumor types, these horses generally present with signs of anorexia and weight loss, with early metastasis of the squamous-cell carcinomas to involve the peritoneum, liver, and lung.
Portal vein thrombosis has rarely been reported in the horse. A yearling Thoroughbred gelding was previously reported to have presented with hepatic encephalopathy associated with an arteriovenous anomaly and portal vein thrombosis. 2 Epiploic foramen entrapment has also been reported to induce hepatic ischemic necrosis and portal vein thrombosis. 5 Hepatic encephalopathy secondary to liver metastasis of an unspecified neoplasm was previously reported; however, portal vein involvement in that case was not cited. 16 The pathogenesis of portal vein thrombosis in other animals may be from tumor invasion, states of hypercoagulability, retrograde intravascular growth of an intrahepatic tumor, or an inflammatory process. 12 Thrombophlebitis, omphalophlebitis, and sepsis are the most common causes in cattle and sheep. In dogs, tumor invasion, pancreatitis, and nephrotic syndrome, with subsequent loss of antithrombin III, may cause thrombosis of the portal vein, caudal vena cava, or renal arteries. 14
Portal tumor thrombosis is a rare complication of gastric carcinomas in humans, with a reported incidence of 0.05–0.95%. 9 Portal thrombosis in adult humans is more commonly associated with cirrhosis, hepatobiliary malignancies, and pancreatitis, occasionally associated with pancreatic carcinomas and rarely with prostatic carcinomas. 10 The majority of these cases presented with clinical signs of nausea, loss of appetite, or abdominal pain. Rarely, portal tumor thrombosis in humans has been associated with obstructive jaundice or hyperammonemia with clinical hepatic encephalopathy. 10
The astrocytic changes and laboratory results in this equine case were consistent with hyperammonemia-associated encephalopathy. Hepatic encephalopathy, uremic encephalopathy, and a recently recognized idiopathic encephalopathy of horses are associated with hyperammonemia. Ammonia has deleterious affects on the central nervous system and may have direct effects on neuronal membranes. Classically, hepatic encephalopathy and hyperammonemia in horses have been associated with the development of characteristic Alzheimer type II astrocytes. Ammonia causes downregulation of GFAP messenger ribonucleic acid, which is evident in these cells as a decrease in GFAP staining via immunohistochemistry. 8
Ammonia is produced by deamination of amino acids by the action of the bacterial urease on ingested proteins and ammonium salts in the gastrointestinal tract. Approximately 80% of this ammonia is then absorbed into the portal circulation. The ammonia is subsequently detoxified by conversion to urea in the liver via the urea cycle. 7 Hyperammonemia results when there is a failure to convert ammonia to urea. Hyperammonemia in this case was most likely because of shunting of blood from the liver because of portal vein thrombosis. Extrahepatic shunts were not noted at the time of necropsy, however, given the large size of the mesenteric root of the horse, numerous small shunts could have been present and not detected.
The case presented represents a unique manifestation of a rare tumor in the horse. Gastric adenocarcinoma with metastasis to the liver is a common presentation in the dog, however, even in this species, portal vein thrombosis is an unusual occurrence. 1
Footnotes
Acknowledgements
We would like to thank Alexis Wenski-Roberts for photomicroscopy and imaging.
