Abstract
Development of the primary and secondary lymphoid organs is a tightly controlled process. These tissues are highly organized to maximize efficiency of the immune response. Spontaneous and targeted mutations in laboratory mice have led to better understanding of the molecular interactions and signaling pathways essential to the development and organization of lymphoid tissues, and the functional consequences of loss or disruption of the normal structures. on the basis of studies of mutations in mice and other species, it has been determined that a wild-type allele of the Foxnl gene is required for normal thymic development and function. The Tlx1, Bapx1, Tcf21, Wt1 and Dh genes are essential for development of the spleen, while mutations of Nkx2-3, Lta, Ltb, Ltbr, Map3k14, Relb, Tnf, Tnfrsf1a, Cxcl13, Blr1 (Cxcr5), or cpdm genes result in disruption of normal splenic microarchitecture. The requirements for organized lymph nodes vary according to anatomic location, but most rely on Id2 (Idb2) and Rorc, in addition to lymphotoxins and Tnfrsf1 1a, Tnfsf11, Relb, Map3k14, Cxcl13, and Blr1 genes. Development of Peyer's patches is dependent on Id2 and Rorc genes, lymphotoxins, and Relb, Map3k14, Il7r, and cpdm genes. Less is known about the requirements for nasal-associated lymphoid tissues (NALT), but Id2 is a requirement. Here we review abnormalities of lymphoid organ development in immunodeficient mutant mice, including spontaneous and targeted mutations of Id2, Rorc, Tnf, Tnfrsf1a, Lta, Ltb, Ltbr, Tnfrsf11a, Tnfsf11, Relb, Map3k14, IL7r, Blr1, and Cxcl13 genes.
Introduction
Studies of mutant mice with dysfunctional immune systems have provided useful tools to enhance our understanding of the immune system and the immune response. Often the genes disrupted in such mice have human homologues, and mutations arising in these genes result in immunologic disease. Examples include the genetically athymic nude mouse (Foxn1nu ), which was first described in 1966, and was followed by a report of the human homologue in 1999. 33, 35 More recently, the IPEX (immune dysfunction, polyendocrinopathy, enteropathy, X-linked) syndrome has been identified as the human homologue of the mouse scurfy (Foxp3) mutation, which was first described more than 40 years ago. 12, 115, 140 As in these cases, most mice with spontaneous mutations resulting in immunodeficiency are first discovered because of grossly identifiable abnormalities in other organs, such as the skin.
Lymphoid tissues are usually classified as primary and secondary lymphoid organs, but this distinction is not absolute. Secondary lymphoid tissues such as the gut-associated lymphoid tissue (GALT) play an important role in B-cell lymphopoiesis in several species, including ruminants and rabbits, 52, 93, 106, 109 although primary lymphoid tissues can play a role in the initiation of antigen-specific immune responses. In this review, we have retained the classic division of “primary” and “secondary” lymphoid organs as a means of organizing this text. The primary lymphoid organs in humans and mice are the thymus and bone marrow. This review focuses on structural aspects of lymphoid organ development and will not include a discussion of the bone marrow as it does not contain discretely organized tissue that can be identified by light microscopy as the site of B-cell development.
The adaptive immune response is the result of highly regulated interactions among antigen-presenting cells, T lymphocytes, and B lymphocytes. These interactions take place in organized lymphoid tissues where the potential for interactions between antigens and rare antigen-specific lymphocytes is maximized. In mice, the secondary lymphoid organs that develop in antigen-independent manner include the spleen, lymph nodes, mucosal-associated lymphoid tissues, such as nasal-associated lymphoid tissue (NALT) and Peyer's patches, as well as other GALT. In other species, this list is more extensive and includes tonsils, diffuse conjunctiva-associated lymphoid tissue, ductal-associated lymphoid tissue, and bronchus-associated lymphoid tissue (BALT). Additionally, organized lymphoid tissues can be induced in other tissues by antigenic stimulus and cytokines.
Recent studies of spontaneously occurring and targeted mutations in mice have shed light on the molecular interactions and signaling pathways that orchestrate the development of secondary lymphoid organs and the microenvironments necessary for optimal adaptive immune responses (Figs. 1, 2). 36, 81 In the following text, we discuss the lymphoid organs in mouse models that have gross or microscopic abnormalities in these tissues (summarized in Table 1), discuss homologous human abnormalities, and review known developmental requirements and markers for each. Table 2 includes a list of allelic variations of mutant mice discussed in the text.
Development of secondary lymphoid organs in mutant mice.∗
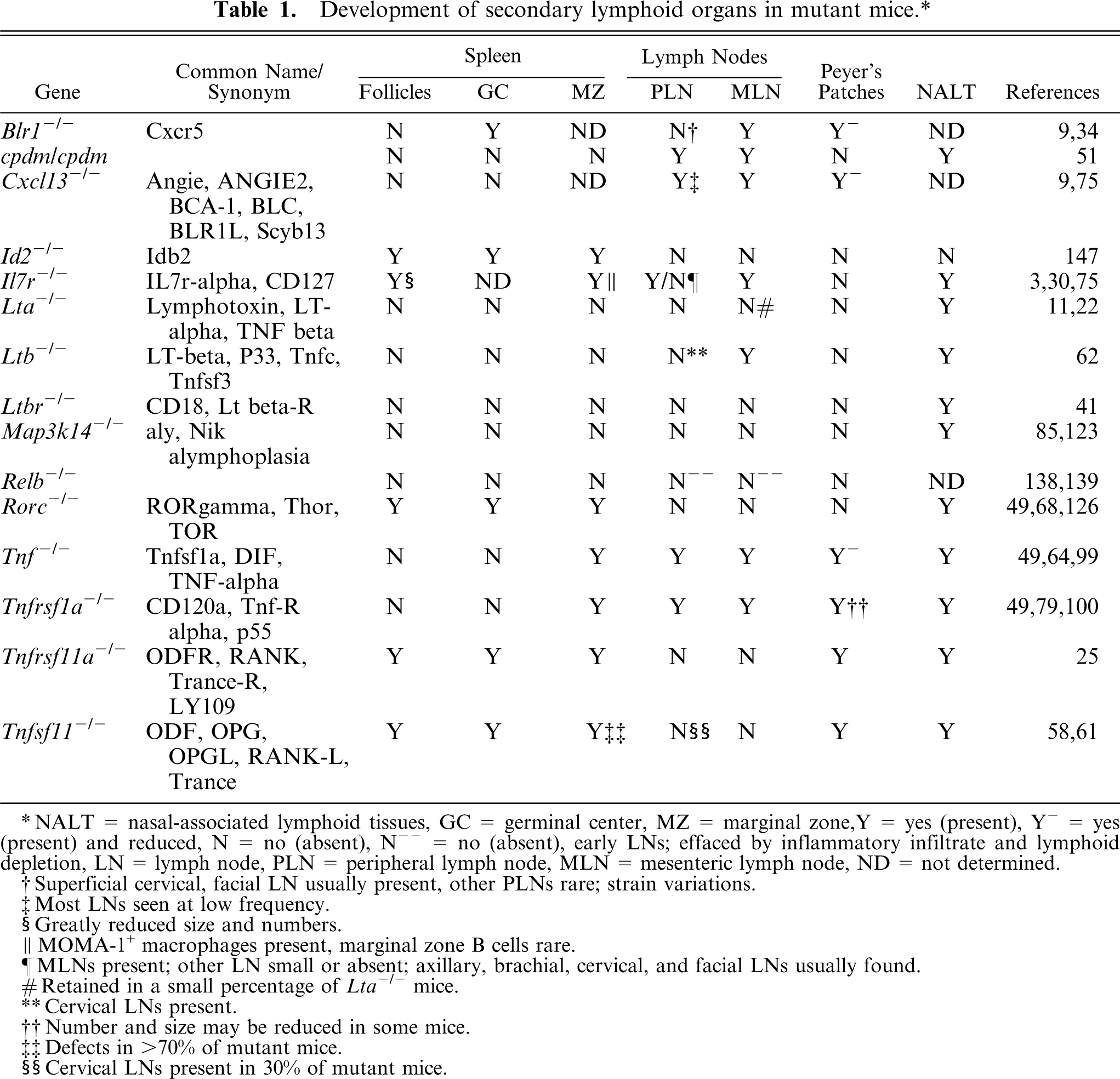
NALT = nasal-associated lymphoid tissues, GC = germinal center, MZ = marginal zone, Y = yes (present), Y− = yes (present) and reduced, N = no (absent), N = no (absent), (absent), N− = no (absent), early LNs; effaced by inflammatory infiltrate and lymphoid depletion, LN = lymph node, PLN = peripheral lymph node, MLN = mesenteric lymph node, ND = not determined.
Superficial cervical, facial LN usually present, other PLNs rare; strain variations.
Most LNs seen at low frequency.
Greatly reduced size and numbers.
MOMA-1+ macrophages present, marginal zone B cells rare.
MLNs present; other LN small or absent; axillary, brachial, cervical, and facial LNs usually found.
Retained in a small percentage of Lta−/− mice.
Cervical LNs present.
Number and size may be reduced in some mice.
Defects in >70% of mutant mice.
Cervical LNs present in 30% of mutant mice.
Tm, targeted mutation; S, spontaneous; Tg, transgene; PP, Peyer's patches; NK, natural killer (cells); AU,; TNF, tumor necrosis factor; LPS, lipopolysaccharide; FDCs, follicular dendritic cells.
Random insertion of a class-I major histocompatibility complex transgene resulting in functional disruption of the Relb gene.
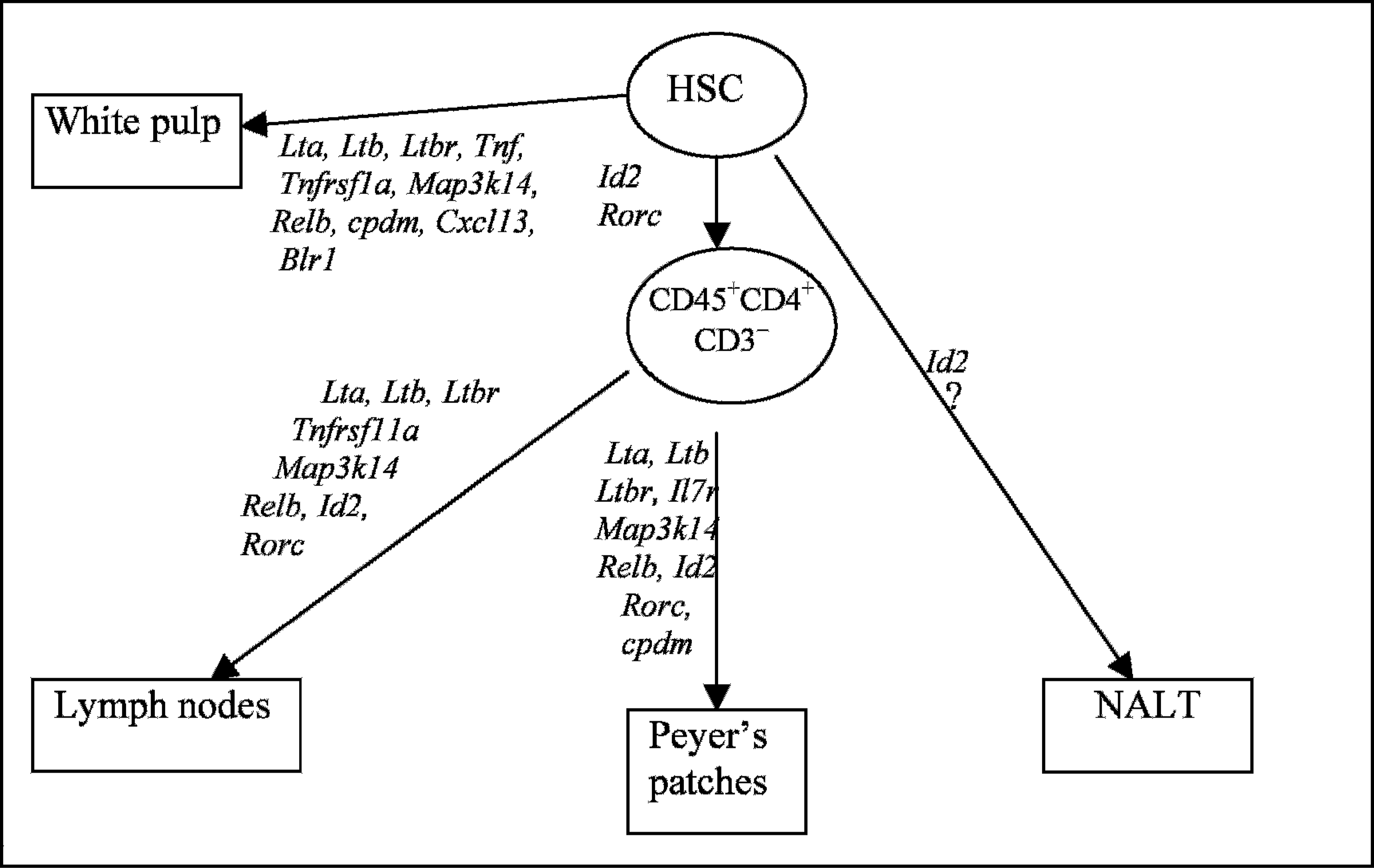
Genes involved in development of secondary lymphoid tissues. HSC = hemopoietic stem cell; NALT = nasal-associated lymphoid tissue.
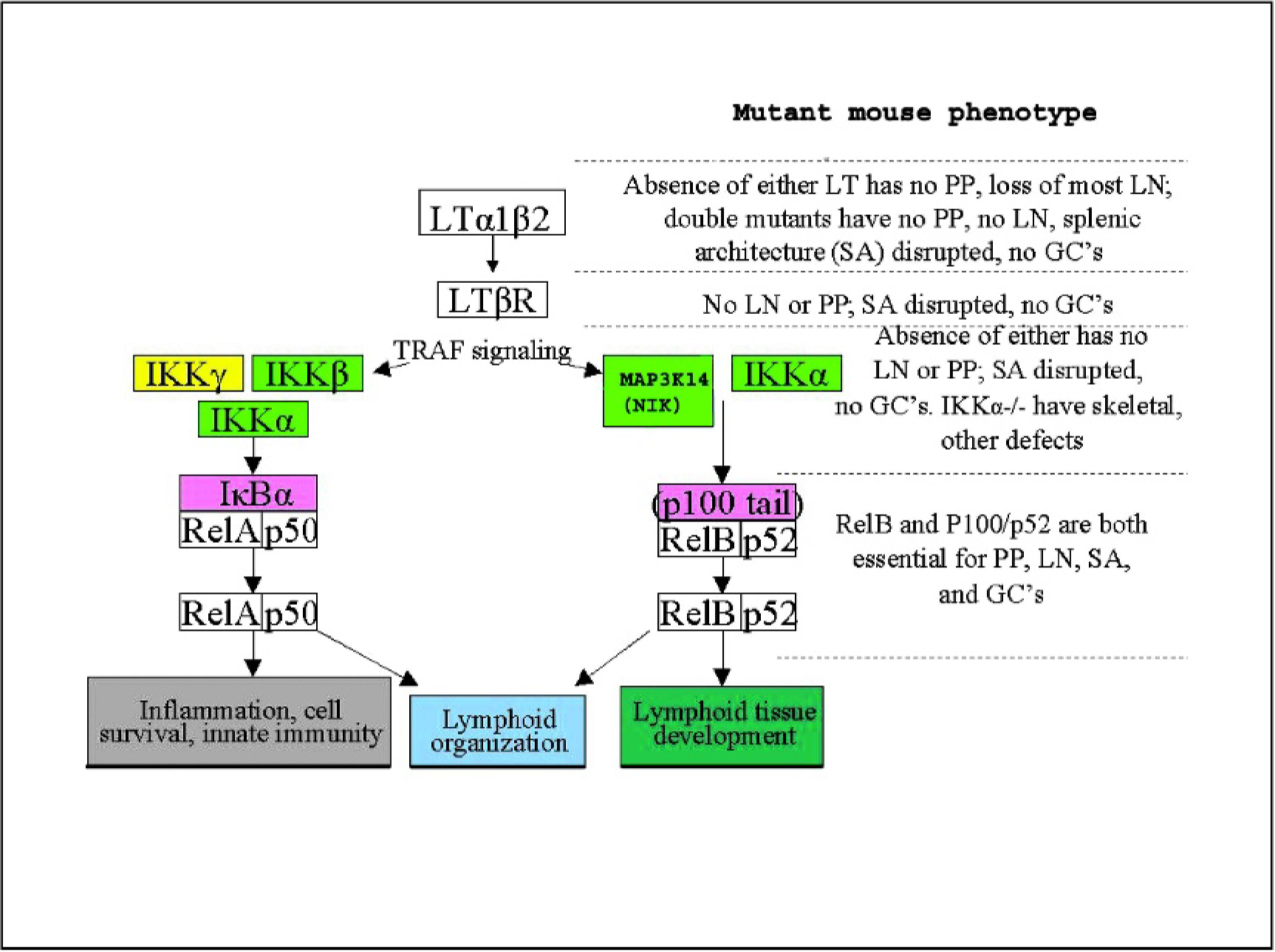
Signaling through the lymphotoxin β-receptor (LTβR). Adapted from references 81,105, and 145. GC = germinal center, IKK = inhibitor of kappaβ kinase, LN = lymph node, LT = lymphotoxin, MAP3K14 = mitogen activated protein kinase kinase kinase 14, NIK = nuclear factor kappaβ-inducing kinase, PP = Peyer's patches, TRAF = tumor necrosis factor receptor-associated factor.
Among the many informative and useful immunodeficient mouse models are those that have severe immune deficiency, not because of abnormal lymphoid organ development per se, but because of intrinsic defects in immune cells that affect lymphoid populations and, therefore, the size and cellularity of these organs. Two widely used models, mice with the severe combined immunodeficiency (SCID) mutation and mice with targeted deletion of Rag1 and Rag2, are briefly addressed here, but they are otherwise outside the scope of this review.
The SCID mouse mutation was reported in 1983. 14 Mice homozygous for what is now known as the Prkdcscid mutation, disrupting the protein kinase, DNA-activated, catalytic polypeptide gene, lack functional T and B cells owing to a defect in the joining of coding ends during V(D)J (variable, diversity, and joining segment) recombination. Most homozygous mice have little or no detectable IgM, and no IgG or IgA. The T cells that are produced are predisposed to lymphoma, and the mice also have DNA repair defects resulting in increased susceptibility to irradiation. 13, 14, 40, 73 The thymus and secondary lymphoid organs are small and effectively lack lymphocytes. The Prkdcscid mice also lack follicular dendritic cells (FDCs) in lymphoid organs, because mature B and T cells are required for the development of FDCs in these tissues. 53 Some Prkdcscid mice can develop partial immunoreactivity (evidenced by detectable IgM concentration), thought to be the result of occasional joining of the coding ends, such that some functional T cells and IgM-producing B cells are found, particularly in older mice or those not housed in a specific pathogen-free environment. 2, 14, 19, 32
Severe combined immunodeficiency syndromes in humans have many causes including mutations of the interleukin (IL-7) receptor or common cytokine (IL-2) receptor gamma chain, adenosine deaminase deficiency, Janus kinase 3 deficiency, and RAG1/RAG2 proteins. 16, 107, 120 Although mutations affecting the DNA–protein kinase subunit product of PRKDC homologs result in SCID phenotypes of variable severity in mice, horses, and dogs, 69, 83, 141 mutations in the human PRKDC gene are not known to be associated with human SCID.
Mice with targeted mutations of recombination activation genes Rag1 or Rag 2 have a phenotype similar to that of the Prkdcscid mouse; Rag1 and Rag2 null mice have B- and T-cell development arrested at an early stage. The absence of mature B and T cells is attributable to failure to initiate V(D)J recombination; they do not tend toward leakiness and, therefore, can have a more severe combined immunodeficiency than do most scid mice. Immunoglobulin M is undetectable. The thymus is small to undetectable. Peyer's patches and lymph nodes are small and lymphopenic, and the spleen has low cellularity. 1, 86, 122
Various forms of human SCID have been attributed to impairment of RAG1 or RAG2 proteins and diminished efficiency of V(D)J recombination. Omenn syndrome in humans is an autosomal recessive disease that manifests as severe immunodeficiency with skin disease, hypereosinophilia, and hypogammaglobulinemia, in which mutations of RAG1/RAG2 genes are often found. 117, 120, 128, 135, 136
Thymus
The thymus is located in the cranial mediastinum of the thorax. Ectopic thymic tissue is sometimes found associated with the thyroid. The stromal network of the thymus consists of epithelial cells and scattered mesenchymal cells. Although earlier studies indicated a mixed ecto-endodermal origin of the epithelial cells, recent work suggests that the thymus epithelium is entirely derived from endoderm. 45 Around postconception day (PCD) 11, epithelial buds separate from the third pharyngeal pouch to give rise to the thymus and parathyroid glands. 21, 124 Around PCD 12, the thymic epithelium becomes populated with lymphocyte precursors. The development and maturation of T cells is determined by sequential interactions with distinct populations of these epithelial cells and with bone marrow-derived dendritic cells. In turn, differentiation and organization of the epithelium depend on signals provided by developing T cells. The fully developed thymus is a lobulated organ, with each lobule divided into cortical and medullary regions (Fig. 3). The cortex is densely populated with immature T cells, whereas the medulla is less densely populated and contains more mature T cells. At least 95% of the developing T cells undergo apoptosis as a result of failure to express T-cell receptors or by the process of negative selection. Apoptotic remnants are common in the numerous macrophages throughout the cortex and medulla. Monoclonal antibodies are useful for characterizing the organization of the thymic epithelium. The monoclonal antibodies ER-TR4 and ER-TR5 bind to cortical and medullary epithelial cells, respectively. 133 Thymic corpuscles are clusters of keratinizing epithelial cells in the thymic medulla that are positive for KRT1-19 (keratin complex 1, acidic, 19; keratin 19 in humans) and other keratin markers on the basis of results of immunohistochemical analysis. Recently, a role has been discovered for thymic medullary epithelium in the induction of central immune tolerance through expression of peripheral antigens in a small number of these cells. 60 This process is controlled at least in part by the Aire (autoimmune regulator) gene. 7
Thymus; wild-type control mouse, normal architecture. HE.
DiGeorge/velocardiofacial syndrome is a common primary immunodeficiency disorder in humans (estimated incidence 1 ° 4,000 live births) characterized by facial and vascular malformations and hypoplastic or absent thymus and parathyroid glands. Most cases are associated with deletion of chromosome 22q11.2, 44, 116 and recently, the T-box 1 (TBX1) gene has been implicated as a candidate in some cases. 15, 44, 144 Features that overlap with DiGeorge syndrome are found in mutant mice with targeted mutations of genes encoding the transcription factors homeobox A3 (HOXA3), or paired box 1 and 9 (PAX1 and PAX9), and with defective retinoic acid signaling or synthesis. 26, 77, 103, 134 Those studies indicate a role for these factors in the development of pharyngeal organs. Thymic hypoplasia has also been observed in mice with mutations of the IL-7 receptor (IL7r) gene. 101
The nude mouse mutation was first described in 1966. 33 In 1968, lack of a thymus was discovered in nude mice; this abnormality results in leukopenia, immunodeficiency, and susceptibility to infectious organisms. 97 The nude gene Foxn1 encodes the transcription factor FOXN1, a member of the winged-helix/forkhead family. 91 Foxn1 is exclusively expressed in epithelial cells, mainly in the thymus and skin. In nude mice and mice with a targeted disruption of the Foxn1 gene, development of the thymus is halted at an early stage, prior to the differentiation of cortical and medullary epithelial cells (Fig. 4). 90 Mutations in the Foxn1 gene have also been discovered in rats and in humans, and result in the same phenotype of athymia and macroscopic hairlessness. 35, 91
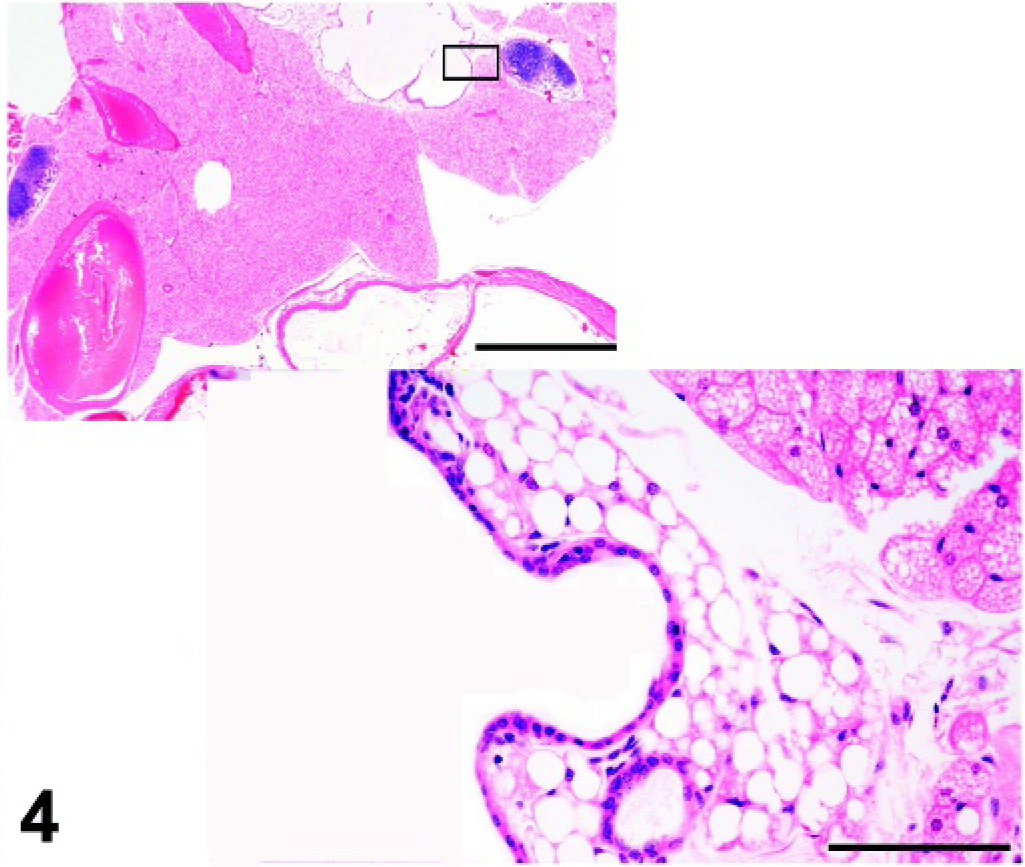
Thymic remnant with cyst; homozygous Foxn1nu (athymic nude) mutant mouse. HE.
Secondary Lymphoid Organs
Lymph nodes and mucosal lymphoid tissues
Lymph nodes are strategically distributed throughout the body. The first protrusions of venous endothelial cells that will form the early lymph sacs develop around PCD 11.5. Lymphatic anlagen are formed when stromal tissues begin to invade this primitive structure. 81 Visualization of lymph nodes in mice can be aided by subcutaneous injection of carbon particle suspensions CH40, CH1500AA, 47 India ink, or dyes such as trypan blue or Evans blue, 42 which are transported to the draining lymph nodes via afferent lymphatics. Accumulation of dye in the lymph nodes stains the tissues within 30 minutes to 2 hours. Alternatively, 300 µl of 1% pontamine/Chicago sky blue in saline can be administered intraperitoneally in mice. 9, 130 This stains most tissues blue, but after 2–3 weeks, the blue dye is retained principally by the lymph nodes.
Organized mucosal lymphoid tissues include Peyer's patches in the small intestine and NALT (considered the equivalent of human tonsils 10, 20 ) in the proximal respiratory tract. Unlike these structures, the BALT does not develop in mice without antigenic stimulation, 24, 87, 96 and is not discussed here.
Peyer's patches are found in the antimesenteric wall of the small intestine. The average number of Peyer's patches varies from about 6 to 10 among mouse strains. Peyer's patches can be identified macroscopically, but historically, microscopic examination of serial sections of Swiss rolls of the small intestine were necessary to determine whether Peyer's patches were absent or underdeveloped. An elegant method of whole-mount immunostaining with monoclonal antibodies has been developed to identify early stages of Peyer's patch development in fetal or neonatal intestines. 4 Early anlage is documented by positive staining for VCAM1 and ICAM1 at PCD 15.5, followed by CD3−CD4+IL−7Rα+Ia+ cell colonization at PCD 16.5, and finally, mature B and T cells (CD3+, CD45+) beginning at PCD18.5.
The NALT is composed of lymphoid aggregates that have a symmetric bilateral distribution in the ventrolateral wall of the nasopharyngeal duct, dorsal to the soft palate in mice. 39 It is present in cross-sections of the nose anterior to the first molars. Peyer's patches and NALT do not have afferent lymphatics, and antigens enter these lymphoid tissues via specialized cells, known as (microfold) M-cells, in the epithelium overlying the lymphoid nodules.
Disrupted development of lymph nodes and Peyer's patches
Early development of lymph nodes (Figs. 5–9) and Peyer's patches (Figs. 10–15) appears to depend on the colonization of anlagen by lymphoid tissue inducer (LTi) cells that interact with stromal organizer cells. The LTi cells are CD45+, CD3−, and CD4+. 82 These cells also express IL7Rα, BLR1, CCR7, and α4β7. 75, 82 Mice with deletion of the helix-loop-helix inhibitor of DNA binding Id2 or the RAR-related orphan receptor γ (RORC) have undetectable LTi cells and lack lymph nodes and Peyer's patches. 27, 68, 126, 147
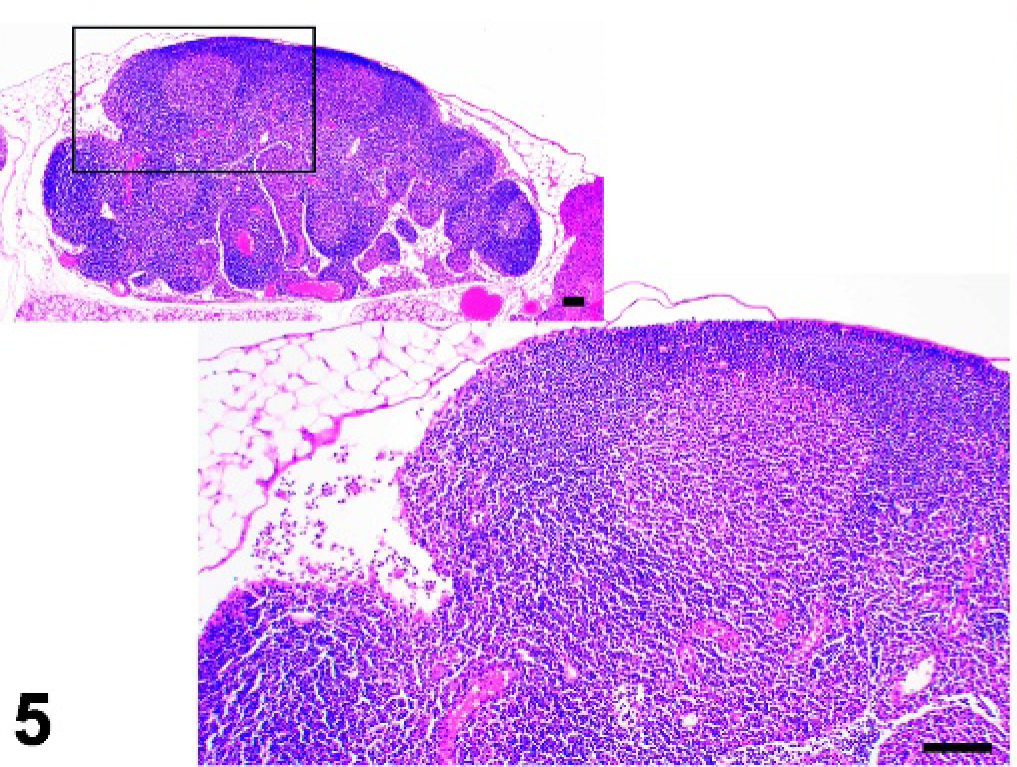
Mandibular lymph node; wild-type control mouse, normal architecture. HE.
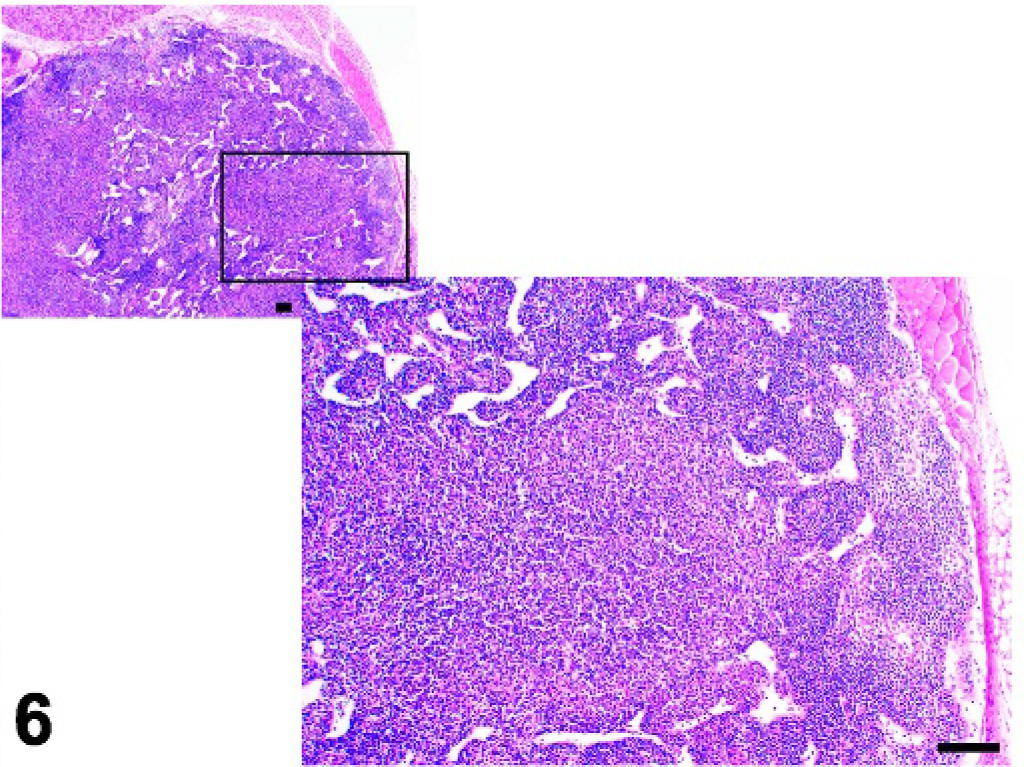
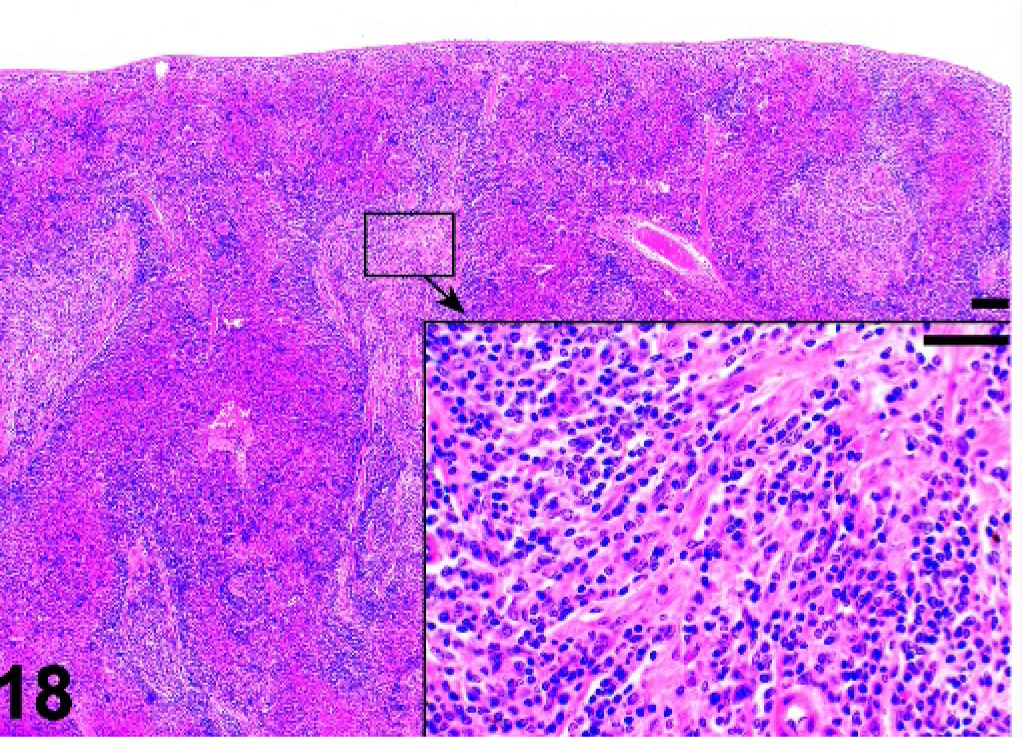
Mandibular lymph node; cpdm/cpdm mutant mouse, lack of B-cell follicles and germinal centers. HE.
Cytokines of the tumor necrosis factor (TNF) superfamily play an important role in lymphoid organogenesis by controlling the proliferation of critical precursor cells, and by regulating cell-to-cell interactions. The cytokine lymphotoxin (LT) is expressed as a secreted homotrimer, LTα3, and a membrane-bound heterotrimer, LTα1β2. The soluble form of LT, LTα3, binds to TNFR1A and a receptor known as TNFRSF14 or herpesvirus entry mediator (HVEM). The membrane-bound form is expressed on the LTi cells and binds to the lymphotoxin B receptor (LTBR) on stromal organizer cells. 36, 80 Ltbr −/− mice and Lta−/− mice have no peripheral (PLN) or mesenteric lymph nodes (MLN), and no Peyer's patches, indicating a pivotal role of LT signaling in the interaction between LTi and organizer cells, and in the development of these lymphoid tissues. 22, 41 In contrast to Ltbr−/− and Lta−/− mice, Ltb−/− mice lack Peyer's patches and PLN, but have MLN and, sometimes, cervical lymph nodes. 11, 22, 62 Signaling through TNFR1A or TNFRSF14 may rescue the development of MLN in Ltb−/− mice. 119 This suggests that organogenesis of MLN and PLN is predominantly controlled by signaling via LTBR, with an auxiliary role for related receptors in the development of MLN. The role of TNF signaling through the TNF receptor in the development of lymph nodes and Peyer's patches is not entirely clear; however, the lack of B-cell follicles and germinal centers, and reduced size or number, or both, of Peyer's patches (which are undetectable in some Tnfrsf1a−/− mice) indicates involvement in the normal developmental and organizational processes. 64, 92, 100
The expression of LTα1β2 on LTi cells is increased by IL7Rα signaling and by TNFSF11 (RANKL or TRANCE). 148 This is consistent with defective development of Peyer's patches and many peripheral lymph nodes in IL7r−/− mice and absence of lymph nodes and reduced numbers and size of Peyer's patches in Tnfsf11−/− and Tnfrsf11a−/− mice. 25, 58, 61, 75 The IL7 receptor is a heterodimer of the IL7Rα and the IL2Rγ common chain and its signaling involves the JAK3 pathway. Mice that do not express the IL2Rγ common chain or JAK3 lack most peripheral lymph nodes, 18, 98 further supporting a role for IL7 in lymph node development. Similarly, signaling through TNFRSF11A involves the adaptor molecule TRAF6, and absence of TRAF6 results in defective lymph node development. 88 These studies suggest nonoverlapping functions of IL7 and TNFSF11 in the development of Peyer's patches and lymph nodes. Homozygosity for the spontaneous mutation, chronic proliferative dermatitis (cpdm), has a phenotype that is complementary to that of the Tnfsf11−/− and Tnfrsf11a−/− mice. Whereas the Tnfsf11−/− and Tnfrsf11a−/− mice have Peyer's patches, but lack lymph nodes, adult cpdm/cpdm mice have peripheral and mesenteric lymph nodes, although the normal architecture is disrupted, and they lack Peyer's patches. 51 This suggests that the cpdm mutation affects a gene that is specifically needed for the development of Peyer's patches. The cpdm mutation maps to mouse chromosome 15, but the affected gene has not yet been identified. 127
Ligand-binding at the TNF superfamily receptors, including the LTBR, activates certain NF-κB signaling pathways. NF-κB is a collection of heterodimers composed of various combinations of the proteins RELA, RELB, c-REL, NFKB1 (p50), and NFKB2 (p52). The heterodimers are kept in inactive form in the cytoplasm by binding of an inhibitor (NFKB1B, formerly IκB) or by similar inhibitory domains. Activation results in phosphorylation of the inhibitors by inhibitor of kappa B kinases (IKBK) and release of the active form of NF-κB, which translocates to the nucleus and is capable of DNA binding. 105 These NF-κB transcription factors induce transcription of various genes associated with cellular proliferation and apoptosis, inflammation, lymphoid development, and the immune response, including cytokines, chemokines, adhesion molecules, and major histocompatibility (MHC) molecules.
The organogenesis of lymph nodes and Peyer's patches is completely disrupted in avian reticuloendothelial viral oncogene-related B (Relb) mutant mice 138, 139 and in mutations disrupting mitogen-activated kinase kinase kinase 14 (MAP3K14, formerly known as NF-κB-inducing kinase or NIK) activity. 85, 123 Mice lacking p52 activity have a similar phenotype. The phenotype of these mice resembles that of Ltbr −/− mice, and it has been documented that LTBR-induced binding of the RELB-p52 heterodimer requires MAP3K14 activity. It is, therefore, likely that RELB and MAP3K14 are essential components of this LTBR signaling pathway (Fig. 2). 145
Signaling through the LTBR can also activate the RelA-p50 heterodimer; however, this form of NF-κB is produced by most cell types and mostly relies on IL1 and/or TNF receptor stimulation. 105, 145 Targeted mutation of the Rela gene in mice results in embryo lethality attributable to massive apoptosis of the liver. Through the creation of Rela−/− , Tnfrsf1a−/− (Tnfr1−/− ) mice, it has been determined that RELA has a protective effect against TNF receptor-induced apoptosis of hepatocytes, and that RELA and TNF contribute to the normal lymphoid architecture of the spleen. 113
The interaction between LTα1β2 on LTi cells and LTBR on stromal organizer cells results in the secretion of chemokines including chemokine (C-X-C motif) ligand 13 (CXCL13, formerly known as B-cell chemoattractant or BLC, a ligand of BLR1) as well as CCL19 and CCL21 (ligands of CCR7). These chemokines amplify the recruitment of LTi cells into the anlagen of lymph nodes and Peyer's patches. This concept is supported by the finding that CXCL13 and its receptor BLR1 (CXCR5) are required for the development of most peripheral lymph nodes. Cxcl13 −/− and Blr1 −/−mice lack most PLN, but have MLN and superficial cervical lymph nodes. 9, 75 Peyer's patches are present, but their numbers and size are reduced in comparison with those in wild-type mice. Mice deficient in CCR7 or the CCR7 ligands, CCL19 and CCL21 (absent in paucity of lymph node T cell or plt/plt mice), occasionally lack inguinal lymph nodes. Crosses of Cxcl13−/− with plt/plt mice resulted in more general deficiency of PLN, although the MLN were still present, suggesting that the orchestrated expression of multiple chemokines is required for proper development of lymph nodes and Peyer's patches. 75
Disrupted development of NALT
Although the NALT and Peyer's patches are mucosal lymphoid tissues and intimately associated with mucosal epithelium, the signaling pathways that induce these structures are different. This was first evident in cpdm/cpdm mice, which lack Peyer's patches but have bilateral NALT, although the structures are underdeveloped. 51 The formation of NALT is dependent on the expression of the Id2 gene, but does not require IL7R, LTBR, or MAP3K14 signaling. 39 Similar to Peyer's patches and lymph nodes, it has been reported that NALT development is initiated by colonization of anlagen by CD45+CD4+CD3− LTi cells. 39, 82 Surprisingly, however, NALT is present and well developed in Rorc-deficient mice, which lack the LTi cells that are critical to lymph node and Peyer's patch development suggesting that a different type of LTi is necessary for development of the NALT. 49 Similar to that in the cpdm/cpdm mice, the NALT is present, but sparse and disorganized in IL7r −/−, Lta −/−, Ltb−/− , and Map3k14−/− mice. Other requirements for NALT organogenesis are unclear, and NALT development has not been thoroughly investigated in many different mutant mice strains.
Spleen
The splenic anlage can be identified in the dorsal mesogastrium of the mouse embryo at PCD 12.5; 57 however, it can be visualized in whole embryos or sectioned tissues as early as PCD 10.5 by T-cell leukemia homeobox 1 gene expression (Tlx1 or Hox11). 23, 111 Hemopoietic cells populate the spleen after PCD 15.5. 118
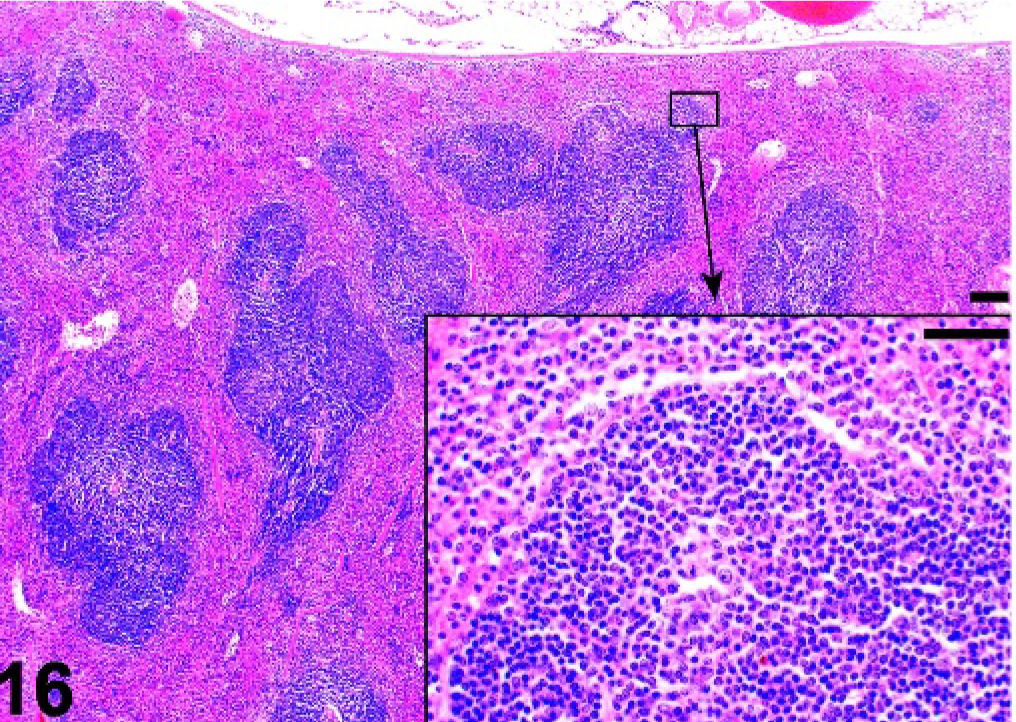
The spleen (Fig. 16) can be separated into the red pulp, which is involved in red blood cell (RBC) and megakaryocyte production and the removal of senescent RBCs, and the white pulp, which is the lymphoid component involved in immunologic defense against blood-borne pathogens. The white pulp consists of a T cell-rich compartment, the periarteriolar lymphocyte sheath (PALS), surrounded by B cell-rich follicles. The follicles contain FDCs, which are mesenchymal cells that do not originate in the bone marrow. 54 These cells retain antigens in the form of immune complexes on their cell surface and make them available to follicular B cells. The PALS and follicles are enveloped by the marginal zone, which separates the lymphoid tissue from the red pulp. The marginal zone consists of a marginal sinus, which is lined by endothelial cells that express the adhesion molecule mucosal vascular addressin cell adhesion molecule 1 (MADCAM1) 66 surrounded by a network of reticular cells that is populated with B cells and macrophages. At least 2 populations of macrophages are recognized, the metallophilic macrophages (MOMA-1) that are localized between the B- and T-cell areas and the marginal sinus, and marginal zone macrophages localized between the marginal sinus and the red pulp. 65 A population of CD21hi, CD23low “marginal zone B cells” active in the T cell-independent humoral immune response is intermixed with the marginal zone macrophages. 31, 55 Monoclonal antibodies against MADCAM1, MOMA-1, and marginal zone macrophages (MARCO and ER-TR9) are useful to identify the marginal zone. 55, 65, 66
Spleen; wild-type control mouse. Spleen with normal organization of the white pulp. B-Cell follicles are evident. HE.
In the following sections, mouse mutations that result in absence of the entire spleen (asplenia) are discussed, followed by a discussion of mutations that cause disruption of the architecture of the white pulp.
Asplenia
Congenital asplenia in humans usually occurs in the context of situs abnormalities such as those seen in situs ambiguous. 43, 112, 125 However, rare cases of isolated congenital asplenia have been described. The familial forms are often dominant conditions and clinically are characterized by increased susceptibility to pneumococcal infections. 43
Absence of the spleen was described more than 35 years ago in mice with the dominant hemimelia (Dh) mutation. 46 Homozygous Dh/Dh mice die in utero, but heterozygous Dh/+ mice have skeletal and visceral defects including asplenia and enlarged lymph nodes. The dominant hemimelia gene is located on mouse chromosome 1, and the mutation is thought to disrupt epithelial-mesenchymal interactions in the developing embryo. 70
Asplenia has recently been reported in several mutant mice, often in combination with other visceral and skeletal abnormalities such as those seen in Dh/+ mice. An exception is mice with a targeted mutation of the T-cell leukemia, homeobox 1 (Tlx1) gene, also called homeobox 11 (Hox11) on mouse chromosome 19. 23, 110 Tlx1−/− mice are viable, and their internal organs develop normally apart from the absence of a spleen. 110 They have been used to study the role of the spleen in immune responses. 56 A splenic anlage can be identified in Tlx1−/− embryos, but development of the spleen is halted at PCD 13.5, and the rudimentary tissue is not colonized by hemopoietic cells. 23
A similar interruption of spleen development occurs in mice that are deficient in the transcription factors TCF21 (capsulin) and bagpipe homeobox gene homolog 1 (BAPX1), and the Wilms tumor homolog (WT1), 50, 71, 74, 131 but the mutations cause embryo or neonatal death owing to hypoplasia of the lungs and other visceral organs. Absence of the spleen occurs in some mice with a targeted mutation of the homeobox-containing NK2 transcription factor-related locus 3 (Nkx2-3 or Nkx2.3), whereas other Nkx2-3 −/− mice have small spleen with abnormal microarchitecture of the white pulp. 94, 95
Disrupted development of the splenic white pulp
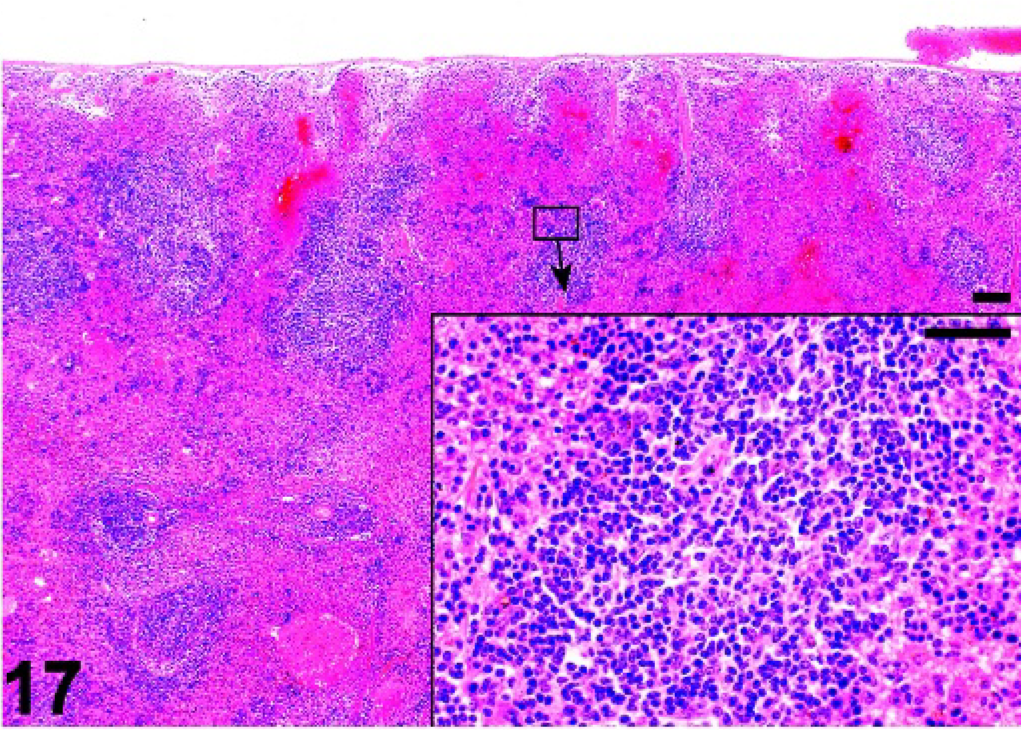
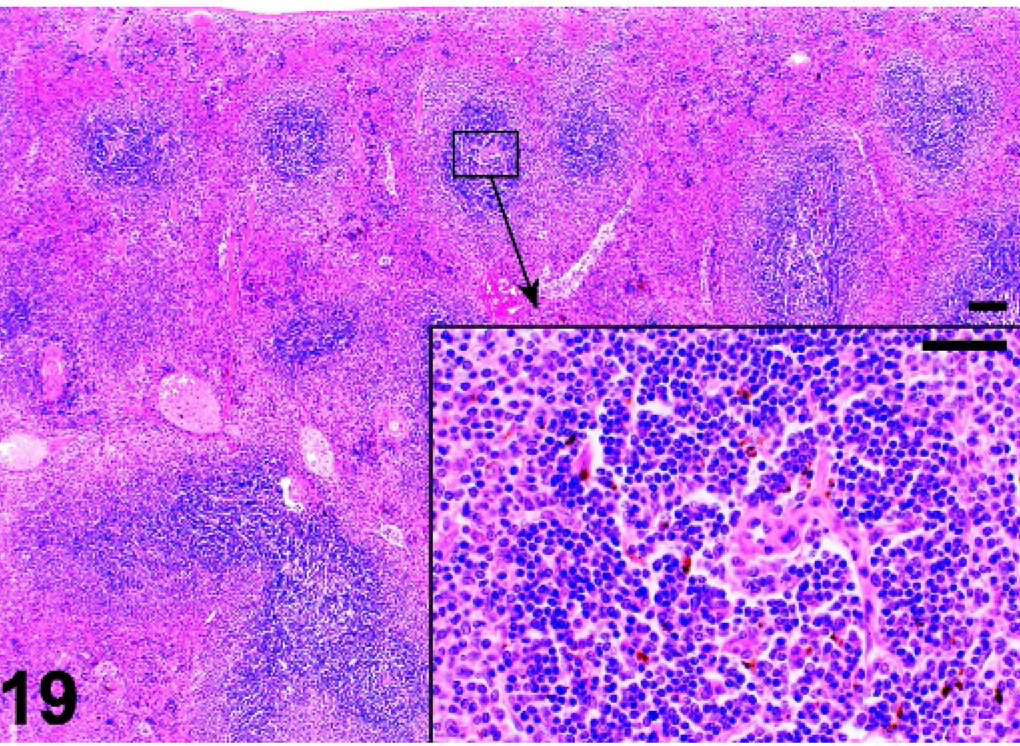
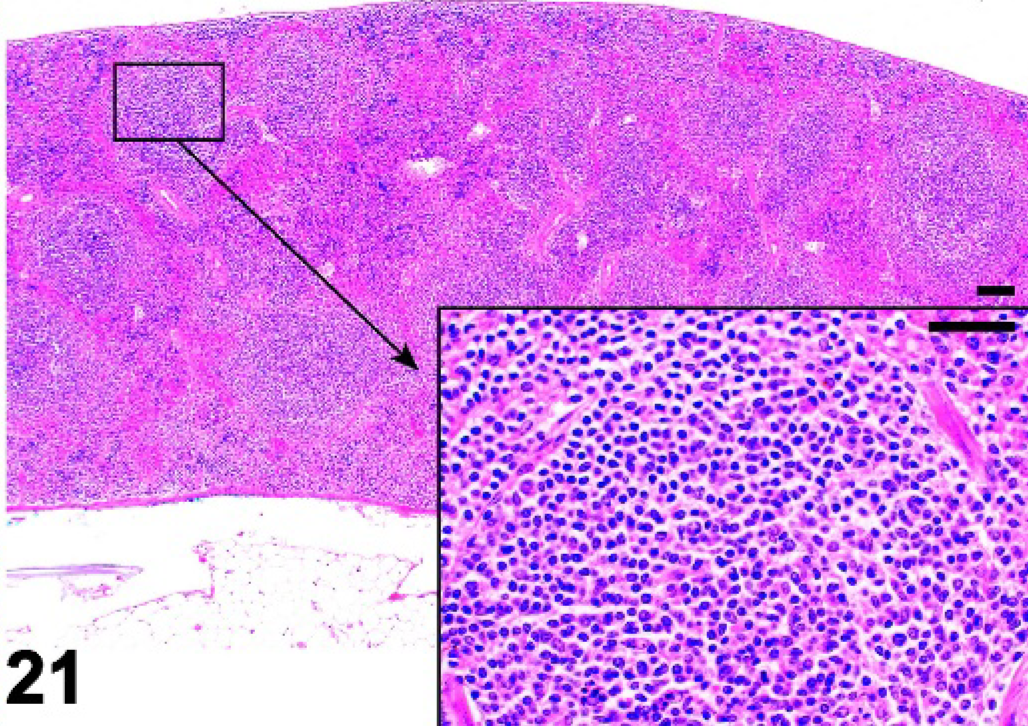
Mice lacking certain NF-κB transcription factors such as Relb (Fig. 17), those with disruptions of genes for lymphotoxins alpha (α) or beta (β) (Lta, Ltb), the receptor Ltbr, Map3k14 (Nik, or alymphoplasia, aly), and chronic proliferative dermatitis (cpdm) (Fig. 18) mice have severely disturbed microarchitecture of the white pulp as well as defects in lymph node and Peyer's patch organogenesis. These mice do not have separate B- and T-cell areas in the spleen, and they lack follicles, FDCs, and the marginal zone. The development of follicles and FDCs also depends on TNF, as Tnf −/− and the TNF receptor Tnfrsf1a −/− mice lack these components (Figs. 19, 20). 64, 79, 99, 100 However, these mice retain distinct B- and T-cell areas and the marginal zone. Mutant mice lacking the IL7 receptor retain marginal zone-specific macrophages and normal separation of red and white pulp (Fig. 21); however, the spleen is small with greatly reduced T-cell areas, reduced size and numbers of B-cell follicles, and few marginal zone B cells. 30 Figures 22–24 demonstrate the wild-type marginal zone, its disruption in the IL7 mutant mouse, and its absence in the Relb mutant.
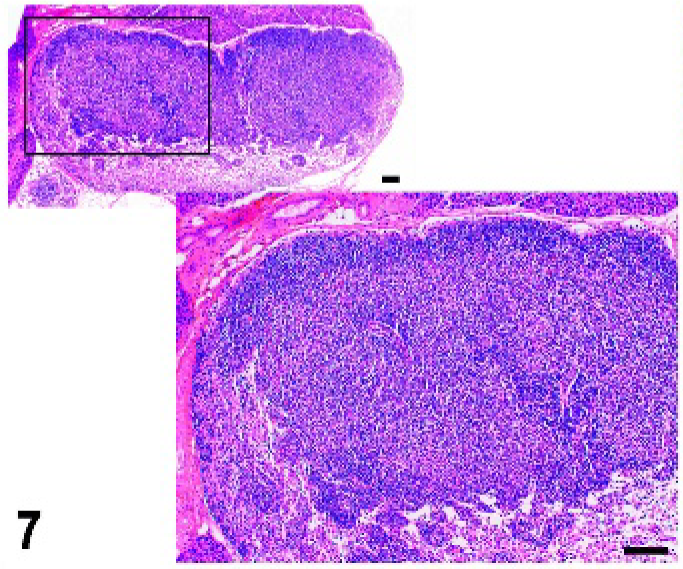
Mandibular lymph node; Tnfrsf1a−/− mutant mouse, lack of B-cell follicles and germinal centers. HE.
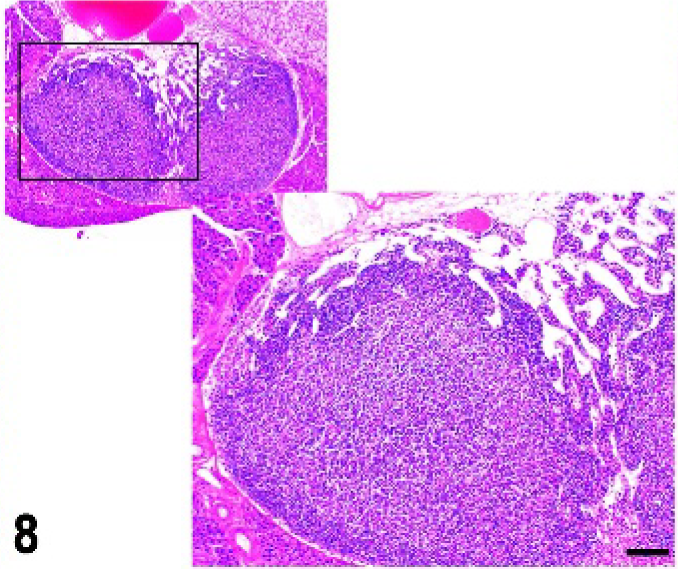
Mandibular lymph node; Tnf−/− mutant mouse, lack of B-cell follicles and germinal centers. HE.
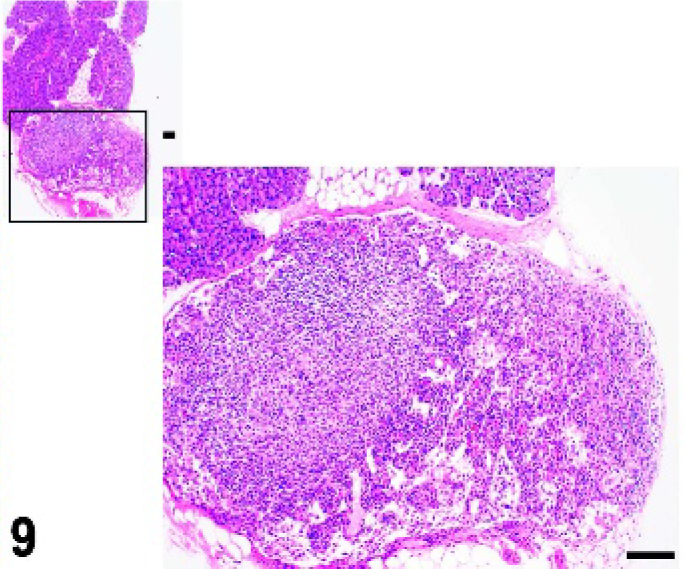
Mandibular lymph node; IL7r−/− mutant mouse. The lymph node is small, sparsely populated and lacks follicles. HE.
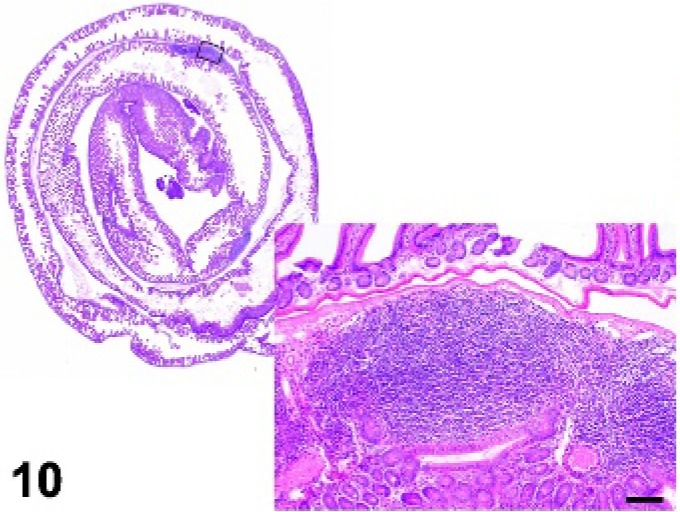
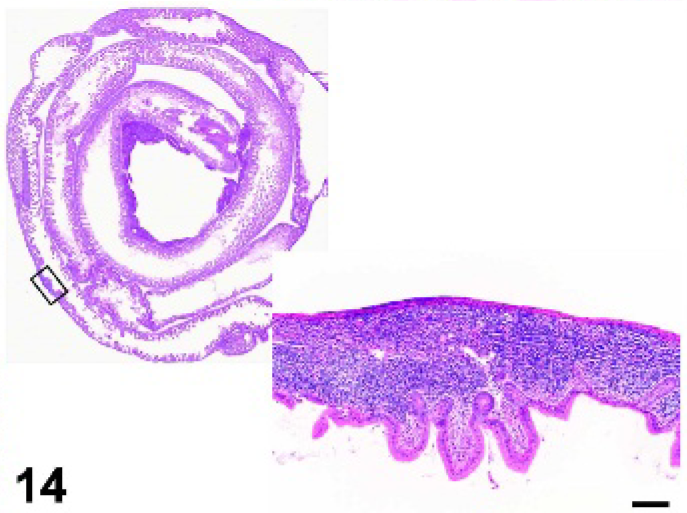
Small intestine; wild-type control mouse, small intestine with several Peyer's patches. HE.
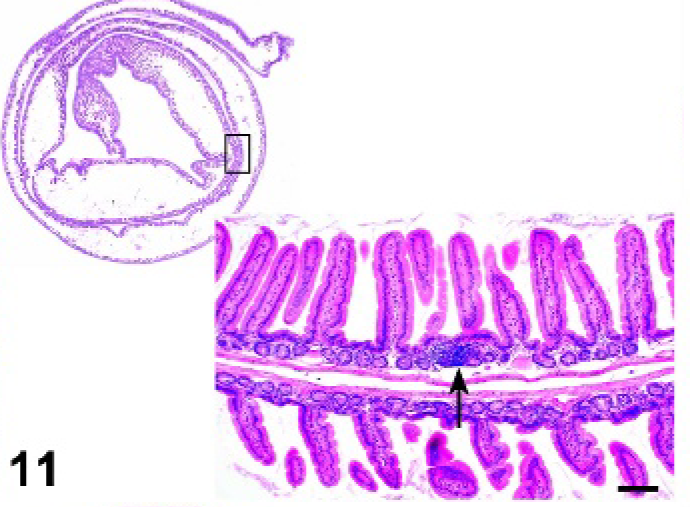
Small intestine; Relb −/− mutant mouse. Small intestine lacks Peyer's patches. Inset. Small lymphoid aggregate (arrow). HE.
Small intestine; cpdm/cpdm mutant mouse. Small intestine lacks Peyer's patches. HE.
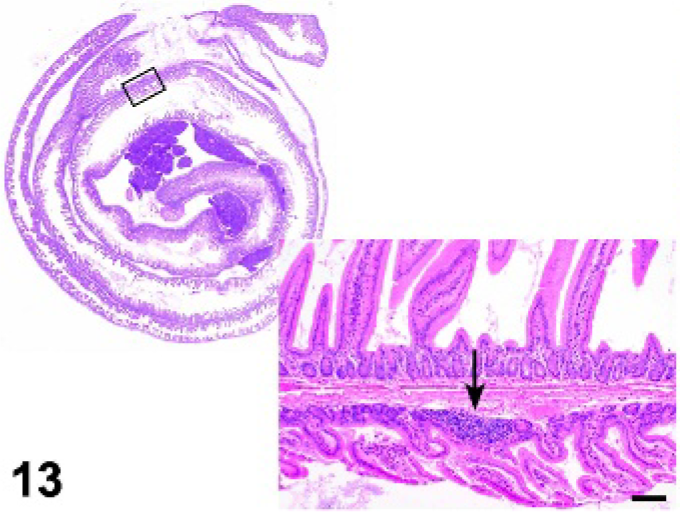
Small intestine; Tnfrsf1a−/− mutant mouse. Reduced size and numbers of Peyer's patches (arrow). HE.
Small intestine; Tnf−/− mutant mouse. Reduced size and numbers of Peyer's patches. HE.
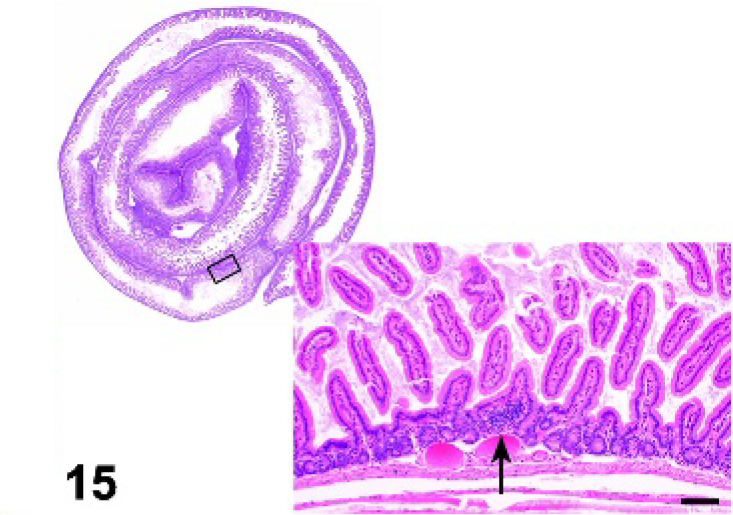
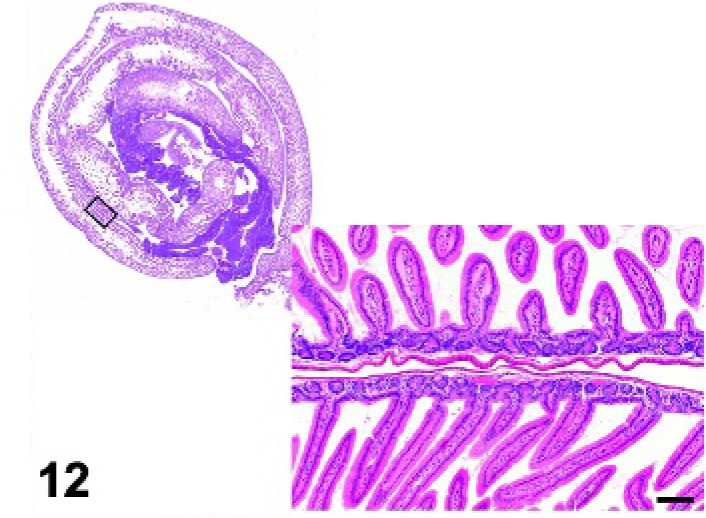
Small intestine; IL7r−/− mutant mouse. Small intestine lacks Peyer's patches. Inset. Small lymphoid aggregate (arrow). HE.
Spleen; Relb− /− mutant mouse. White pulp is small and lacks well defined follicles. HE.
Spleen; cpdm/cpdm mutant mouse. White pulp is small with a few lymphocytes and fibrosis. Notice lack of lymphoid follicles. HE.
Spleen; Tnfrsf1a−/− mutant mouse. White pulp is well defined, but lacks lymphoid follicles. HE.
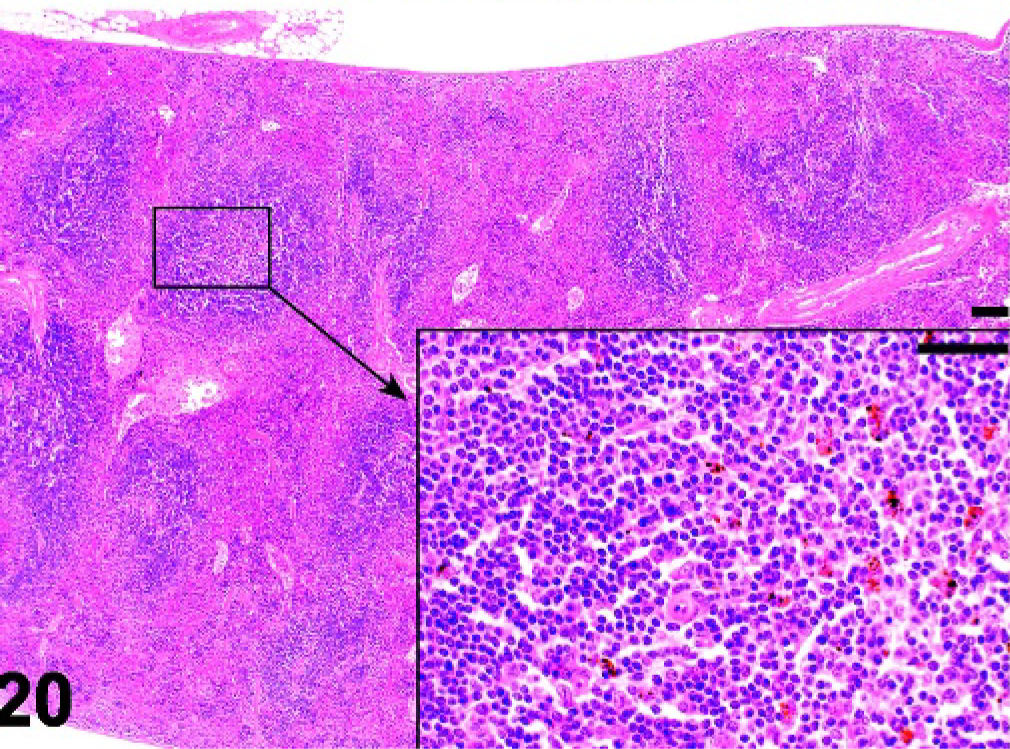
Spleen; Tnf−/− mutant mouse. White pulp is well defined, but lacks lymphoid follicles. HE.
Spleen; IL7r−/− mutant mouse. Spleen is small (notice thickness of cross section). White pulp is well defined but has reduced size and number of lymphoid follicles. HE.
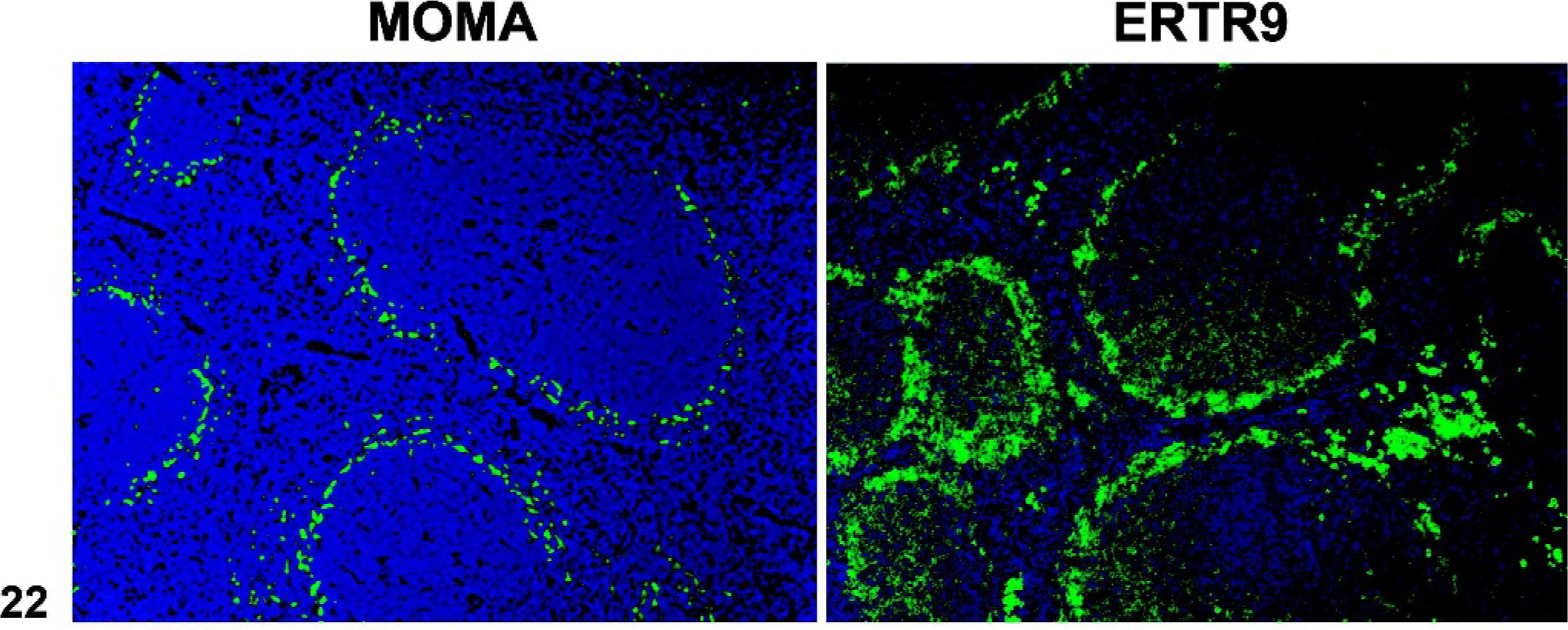
Spleen; wild-type control mouse. The monoclonal antibodies MOMA-1 (left) and ER-TR9 (right) label metallophilic macrophages of the inner marginal zone, and marginal zone macrophages of the outer zone, respectively. Immunofluorescence on frozen sections.
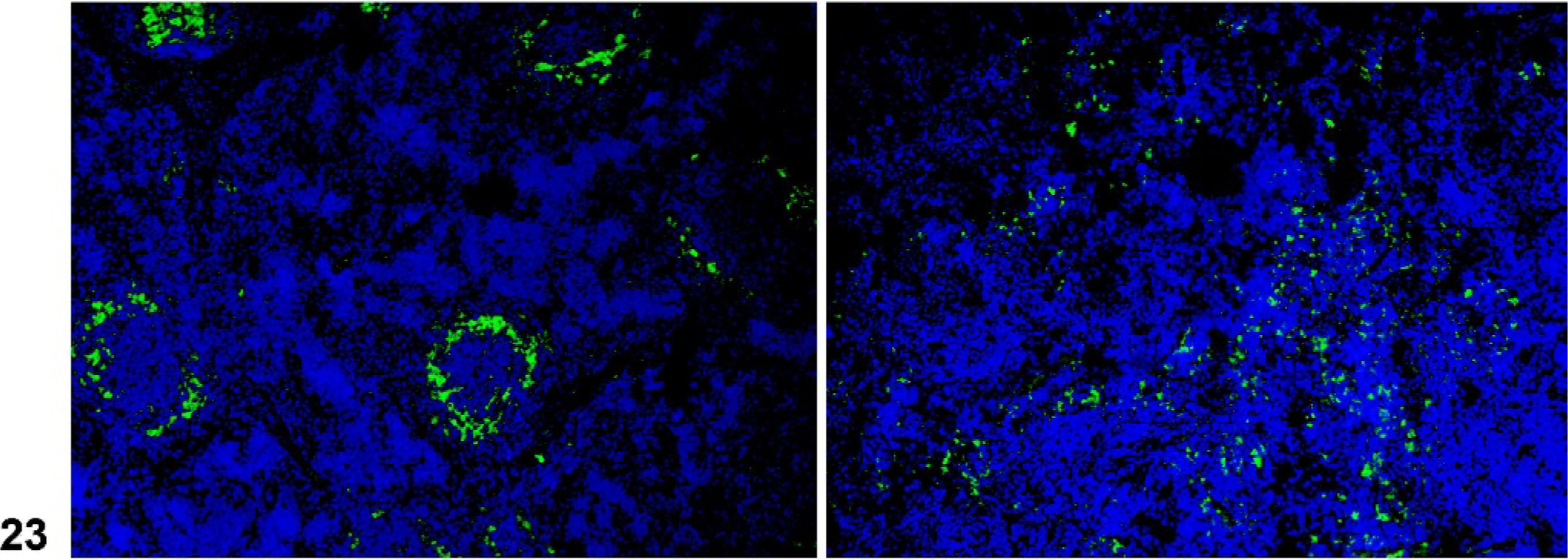
Spleen; IL7r−/− mutant mouse. Abnormal marginal zone as indicated by reduced inner marginal zone, (identified by MOMA-1, left), and lack of an outer marginal zone (right). Immunofluorescence on frozen sections.
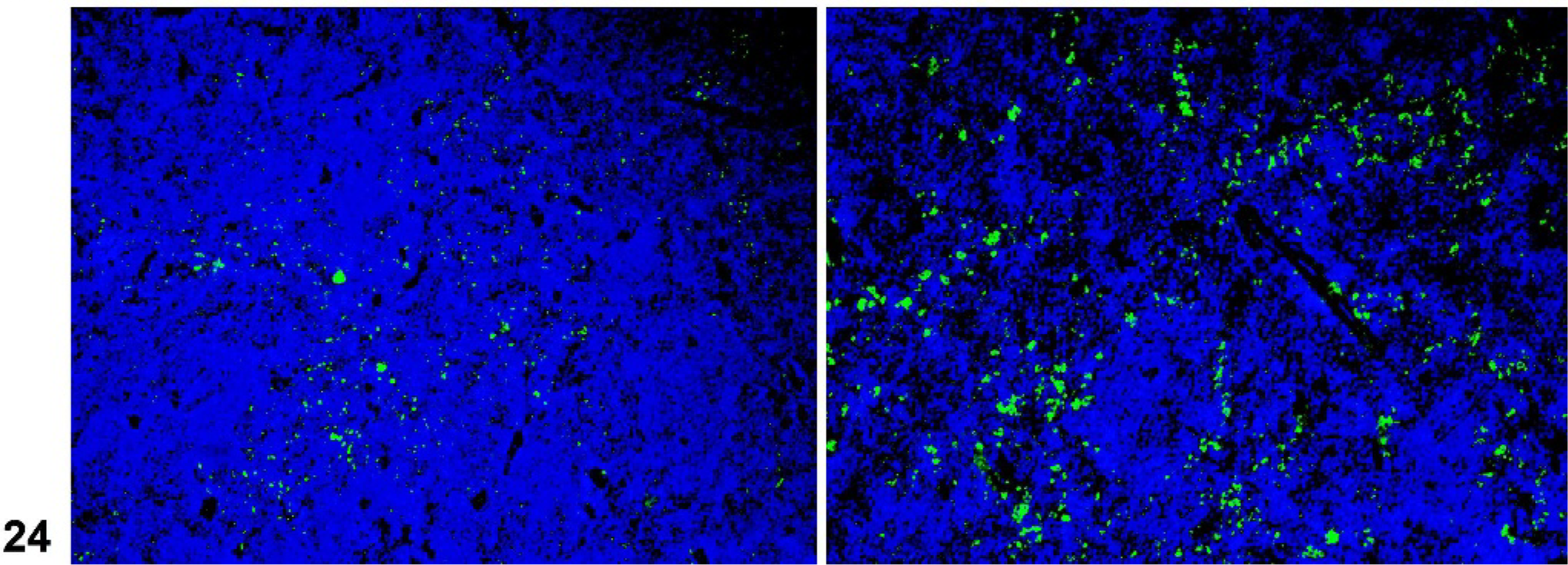
Spleen; Relb− /− mutant mouse. Lack of a marginal zone. Immunofluorescence on frozen sections.
Reconstitution experiments with purified lymphocytes from mutant mice have indicated that the expression of the membrane-bound trimeric form of lymphotoxin (LTα1β2) by B cells delivers the signal for FDC differentiation and formation of organized lymphoid follicles. 37 The expression of LTα1β2 by B cells is up regulated by CXCL13, and indeed, Cxcl13 −/− mice lack follicles and FDCs. 8, 9 The architecture of the white pulp of the spleen in Id2- and Rorc-deficient mice is intact, suggesting that the CD45+CD4+CD3− cells are not essential for the development of the white pulp of the spleen. Reconstitution of normal splenic architecture in IL7r−/− mice following adoptive transfer of normal spleen cells also supports this finding. 30
Conclusion
Detailed examination of lymphoid organs in mutant mice has led to significant advances in our understanding of the molecular interactions that are involved in lymphoid organogenesis. The development of these highly organized tissues depends on carefully orchestrated expression of multiple genes with various sets of genes contributing to the development of PLN, MLN, Peyer's patches, and NALT (Table 1). The LT pathway has been identified as pivotal for the development of lymph nodes, Peyer's patches, and the white pulp of the spleen, whereas the signals for development of the NALT are not yet fully known.
Footnotes
Acknowledgements
This work was supported in part by grants from the National Institutes of Health (T32RR07068; CA34196; RR00173; and AR49288). We thank Drs. Derry Roopenian and Leonard Shultz for their time and efforts in review of this manuscript, and B. Shirley and J. Denegre for their assistance with fluorescent imaging.

