Abstract
An atypical case of severe soft-tissue mineralization in a 3-week-old foal from a herd of Andalusian horses is described. The herd clinical history and the laboratory findings were compatible with a diagnosis of secondary hyperparathyroidism due to a mineral imbalance in the diet (low calcium and high phosphorus intake). Mares showed a marked increase in serum parathyroid hormone (PTH) approximately 10 times normal levels. Serum PTH was marginally elevated in foals. Clinical signs (unthriftiness, painful joints, lameness in one or more limbs, and stiff gait) were more pronounced in foals than in mares. Two foals died and necropsy of one of them revealed extensive soft-tissue mineralization of arterial walls and pulmonary parenchyma. Clinical signs in mares and foals resolved by 4 weeks after diet adjustment.
Soft-tissue mineralization occurs as a result of hypercalcemia and/or hyperphosphatemia, leading calcium and phosphorus crystals to be deposited extracellularly adjacent to elastic fibers. This condition usually affects the wall of arteries and smaller vessels. In adult horses, soft-tissue mineralization has been reported in association with vitamin D toxicosis due to excessive vitamin D supplementation or caused by accidental ingestion of toxic plants (Cestrum and Solanum spp.). 8, 13 Affected horses may exhibit hypercalcemia and hyperphosphatemia, although normal calcium and phosphorus plasma levels can also be found. 7, 8
In boars 6 and marmosets, 9 soft-tissue mineralization has been reported in cases of nutritional secondary hyperparathyroidism (NSHP).
Horses fed a diet containing an excess of phosphorus relative to the amount of calcium increase parathyroid hormone (PTH) secretion and develop NSHP. Parathyroid hormone, acting on the kidney and bone, has an hypercalcemic and hypophosphatemic action. Thus, increased PTH levels can maintain extracellular calcium and phosphorus within normal limits in the face of a low calcium/high phosphorus intake. However, this new equilibrium is achieved at the expense of increasing bone resorption, which can lead to osteodystrophy. 11, 12 This article describes an unusual case of severe soft-tissue mineralization in a suckling foal from a herd of mares receiving a diet containing excess phosphorus and low calcium.
A farm with a small breeding stock of Andalusian horses (11 brood mares) had suffered two perinatal casualties during the same reproductive season. The second foal (3 weeks old) that died was necropsied by the attending veterinarian. Gross changes included severe pulmonary congestion and petechiation in the lungs, endocardium, pericardium, and kidneys (cortex and medulla). Specimens of the heart, lungs, liver, spleen, and kidneys were fixed in 10% saline formalin. Tissue samples were routinely processed and paraffin embedded. Four-micrometer-thick tissue sections were stained with the hematoxylin and eosin and Von Kossa techniques.
The most relevant histopathological finding was extensive and severe mineralization of capillaries, arterioles, and arteries in the lungs, heart, kidneys, and spleen. Mineralization was particularly severe in the lungs, where numerous different sized mineralized foci were observed in interalveolar septa and in the walls of numerous arterioles and arteries (Figs. 1, 2) in both cranial and caudal lobes. These foci stained positive with the Von Kossa method. Inflammatory reaction or tissue damage, such as necrosis, was not observed at the periphery of mineralization foci indicating a metabolic origin. Congestion was severe in the lungs, liver, and kidneys. Small hemorrhages were observed in the endocardium, pericardium, cortex, and medulla of the kidneys.
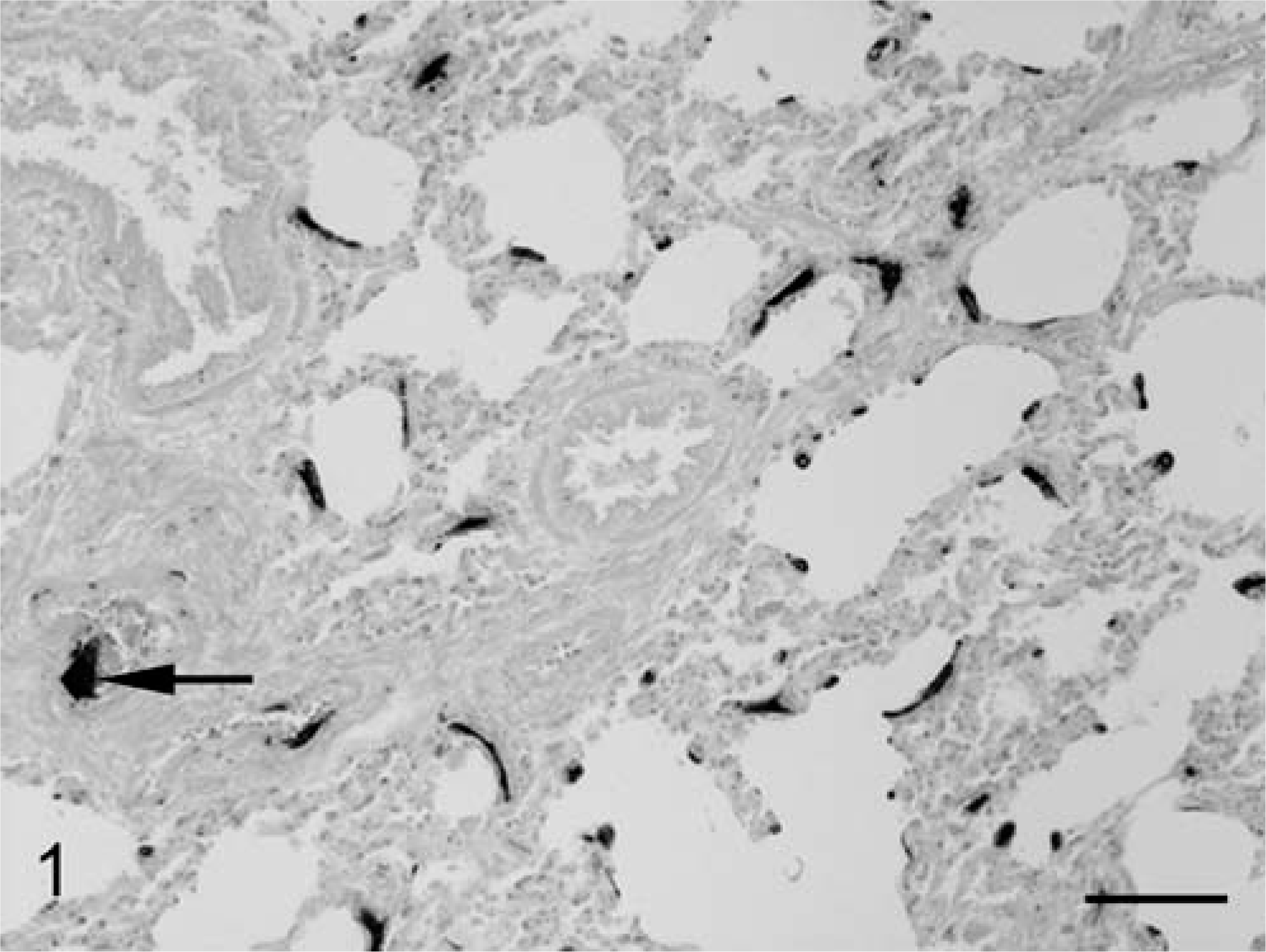
Horse, lung; section showing severe mineralization of the wall of several arteries (arrow) and numerous mineralization foci in the interalveolar septa. Von Kossa. Bar = 100 µm.
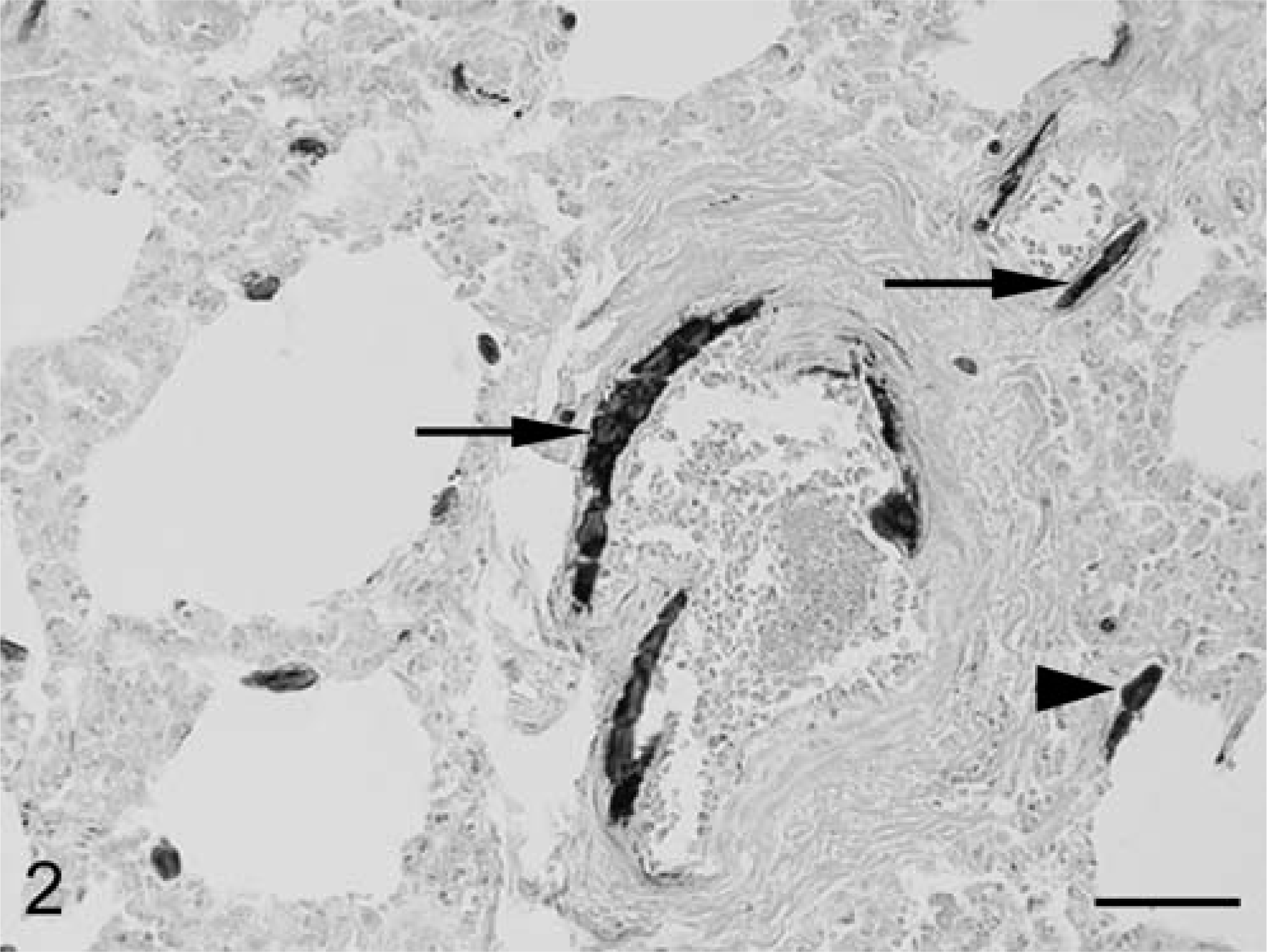
Horse, lung; detail of two arteries showing extensive mineralization in the tunica media (arrows) and mineralization foci in the interalveolar septa (arrowhead). Von Kossa. Bar = 50 µm.
The Field Service of the School of Veterinary Medicine of the University of Cordoba was asked to carry out a survey of the herd. The mares had been vaccinated against rhinopneumonitis, influenza, and tetanus and had been dewormed. No history of perinatal deaths had been reported in previous reproductive seasons. Mares (n = 11) and foals (n = 9, age 2–6 weeks old) were housed in stalls at nights and in a wide pen during the day. Mares were fed two times a day a home-made diet containing oats, beans, sunflower seeds, wheat bran, and wheat straw. No mineral supplementation was provided. All foals were suckling and they also had free access to the mares' food. Animals had unrestricted access to water.
Upon presentation, the horses did not show ostensive clinical signs, although the herd had an unthrifty look and some animals suffered subtle lameness, stiff gait, and painful joints. In general, these signs were more prominent in foals than in mares. Rectal temperature was normal in mares and foals. A complete blood count and a routine biochemistry panel were run in all animals. No relevant hematological or biochemical abnormalities were observed in mares or foals.
Mineral metabolism was evaluated by measuring the plasma concentrations of total calcium (Cat), ionized calcium (Ca2+), phosphorus (P), PTH, and 1,25-dihydroxycholecalciferol (1,25-D). Cat and P were quantified by routine spectrophotometric techniques (Sigma Diagnostics, St. Louis, MO, USA). Ca2+ was measured with a selective electrode (Bayer Diagnostics, Barcelona, Spain). PTH was quantitated by the use of an immunoradiometric technique (Intact-PTH, Nichols Diagnostics, San Juan Capistrano, CA, USA) that has been previously validated in horses. 4 1,25-D concentration was measured with a radioimmunoassay (Immunodiagnostic Systems, Boldon, UK).
Table 1 shows the blood parameters of mineral metabolism in mares and foals. Both Cat and Ca2+ were within the normal range in mares and in foals. When compared with the foals, the mares had slightly lower calcium levels. Plasma P was normal in the mares. Phosphorus concentration was much higher in foals than in mares. PTH values in mares (mean ± SE) were very high (423 ± 192 pg/ml), whereas foals had marginally elevated PTH levels (105 ± 16 pg/ml). Plasma 1,25-D concentration was 27.6 ± 4 pg/ml (mares) and 23.2 ± 3.1 pg/ml (foals). Because there is controversy in the literature about the normal levels of 1,25-D in horses, 1,25-D levels were also measured in six plasma samples from healthy adult horses, obtaining a value of 27.7 ± 2.8 pg/ml (range 18–36 pg/ml). Analysis of the diet revealed an unbalanced mineral content: Ca = 0.18% and P = 0.58% (dry matter).
Plasma values (mean ± standard error) of parameters of mineral metabolism in the mares and foals. ∗
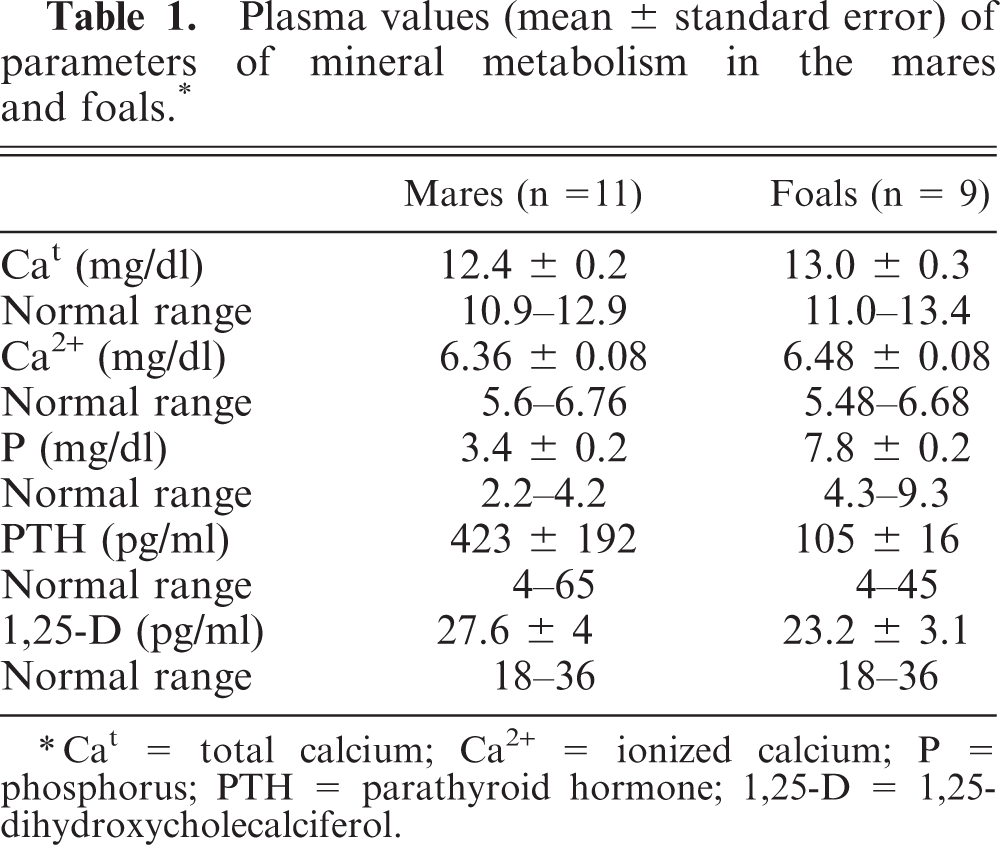
∗Cat = total calcium; Ca2+ = ionized calcium; P = phosphorus; PTH = parathyroid hormone; 1,25-D = 1,25-dihydroxycholecalciferol.
Based on these data, a diagnosis of NSHP was established. NSHP is the most common disorder of mineral metabolism in horses. 11, 12 Advanced cases of NSHP show bone deformities of the calvaria as a result of fibrous osteodystrophy (big head). Less affected animals usually present lameness, as a consequence of bone microfractures, and skeletal deformities affecting long bones. 11, 12 In the herd of the present study, bone deformities were not noticed but unthriftiness and lameness were observed. When the clinical signs are subtle, diagnosis of NSHP is difficult. In the past, urinary fractional excretion (FE) of calcium and phosphorus have been used for diagnostic purposes. 12 By definition, a true diagnosis of hyperparathyroidism can only be made when high PTH levels are demonstrated. In addition, the degree of increase in PTH can be used as an estimation of the severity of bone resorption. The mares under study had PTH concentrations that were four times higher than the upper limit of the normal range and approximately 10 times the mean normal PTH concentration. 1, 2, 4 The serum PTH in foals was lower than in mares. Healthy foals have been reported to have lower serum PTH levels than adult horses. 5 In addition, because, in the present case, most of the calcium and phosphorus intake of the foals was through the milk, they would be less affected by the mineral imbalance of the diet. It is also interesting to note that serum P in foals, although within the normal range, 3 was much higher than in mares.
As previously stated, demonstration of elevated PTH levels in the presence of normal or decreased plasma calcium is diagnostic of secondary hyperparathyroidism; however, the diagnosis of NSHP must be confirmed by diet analysis. In the present case, analysis of the diet demonstrated a low intake of calcium and an excessive intake of phosphorus.
The owner was advised to change the diet and a new diet with normal amounts of Ca (0.5%) and P (0.3%) was formulated. A progressive decrease in clinical signs was noted over a 4-week course. No subsequent casualties were reported and, on reexamination after 1 month on the new diet, the herd was considered normal. Although PTH levels were not as elevated in foals as in mares, the fact that their clinical signs subsided after changing the diet would indicate that they were also affected by the mineral imbalance of the diet.
The pathology findings in the necropsied foal were indicative of extensive soft-tissue mineralization. This condition may be associated to inflammation or to metabolic disorders. In the foal reported here, the absence of inflammatory reaction and tissue damage suggested a metabolic origin. In horses, generalized soft-tissue mineralization has been associated with vitamin D toxicosis due to excessive vitamin D supplementation or by the accidental ingestion of toxic plants. 13 Vitamin D intoxication was considered an unlikely diagnosis in this foal because the animals did not receive vitamin D supplementation and no increase in plasma 1,25-D was found. Although 1,25-D levels were not measured in the necropsied foal, it is likely that they were similar to these of the surviving foals because all of them received the same diet and were of similar age.
Chronic renal failure is another condition that may result in soft-tissue mineralization. 13 This disease was ruled out because no histopathologic changes were found in the kidneys.
In horses, soft-tissue mineralization associated to NSHP has not been reported, probably because adult horses are able to increase PTH levels to maintain phosphorus and calcium concentrations within normal ranges, as occurred in the mares of the present study. However, in other species, boars 6 and marmosets, 9 soft-tissue mineralization has been described in association with NSHP. The soft-tissue mineralization of the foal in this report may have been related to the imbalance of the diet. Factors that may contribute to development of calcifications in the foals of this herd are the relatively low PTH levels (when compared with adults on the same diet) and the increased plasma P concentration. Parathyroid hormone-related peptide (PTHrP) has been reported to prevent vascular calcifications. 10 Because PTHrP and PTH act on the same receptor and have similar biological actions, the lower PTH concentration may have predisposed the foals of this herd to suffer vascular calcifications. In addition, the elevated plasma P of the foals can be particularly relevant because it will contribute to increase the CaxP product (101 ± 3.4 mg/dl in the foals of this farm), thus promoting soft-tissue mineralization.
In conclusion, the severe soft-tissue mineralization described in this study is atypical because of the age of the affected foal (3 weeks old). Because the problem appeared in a herd affected by NSHP and NSHP has been reported to cause calcifications in other species, it is likely that soft-tissue mineralization in this foal may be related to NSHP. The relatively low PTH and high P levels in the foals, when compared with the adult horses of this herd, may have predisposed them to soft-tissue mineralization.
Footnotes
Acknowledgements
The authors are grateful to Manuel Ruiz for help collecting clinical data.
